Abstract
Background:
There is increasing interest in better understanding the immune cell composition of human milk and how these cells interact with neonatal immune development. However, consistent methods for immune cell isolation from human milk are lacking.
Research Aim:
Our aim was to systematically compare available cell isolation techniques to isolate T lymphocytes from human milk samples.
Methods:
This repeated measures study design compared three cell isolation methods using human milk samples: (1) centrifugation, (2) immunomagnetic bead isolation, and (3) a combination of both methods. We assessed the proportion and viability of CD3+, CD4+, CD25+ and regulatory T cells using flow cytometry in isolated cells to compare the performance of these isolation methods.
Results:
Immunomagnetic separation is a feasible method to isolate T lymphocytes in human milk, similar to blood. It improves target cell enrichment and cell viability compared to centrifugation, which may be an advantage when the goal is to characterize rare cell types or when cells are further used in functional assays. No excess cell activation (CD25 positivity) was observed with the use of magnetic beads.
Conclusion:
Immunomagnetic separation of human milk T lymphocytes may have advantages over centrifugation depending on the intended downstream use of cells.
Background
Human milk is not only a source of nutrition for infants, but it also contains a large variety of maternal cells, including immune cells. Increasing attention is turning to how these cells interact with the infant’s immune system (Camacho-Morales et al., 2021), and whether they are retained by the infant, leading to a state of maternal microchimerism (Molès et al., 2017). This maternal microchimerism of immune cells through lactation likely supports neonatal and infant immune system maturation, tissue repair, and immune tolerance (Dutta & Burlingham, 2009; Srivatsa et al., 2003). Immune cells include myeloid precursor cells, dendritic cells, macrophages (Ichikawa et al., 2003), innate lymphoid cells (ILCs); (Baban et al., 2018), natural killer (NK) cells (Li et al., 2017), as well as different T lymphocytes subsets, such as cytotoxic T cells (Cabinian et al., 2016), and regulatory T cells (Tregs; Cérbulo-Vázquez et al., 8). Non-immune cells in human milk include stem cells, myoepithelial cells, lactocytes, and progenitor cells (Witkowska-Zimny & Kamińska-El-Hassan, 2017).
Tregs are essential in regulating the immune response to pathogens and environmental antigens, in maintaining immunological homeostasis and tolerance towards autoantigens in order to avoid autoimmunity (Vignali et al., 2008), and in controlling allergic responses in the infant (Černý et al., 2018). We have recently demonstrated that by 3 weeks of age, the proportion of peripheral Tregs is nearly twice as high in exclusively breastfed healthy full-term neonates as in exclusively formula-fed healthy full-term infants (Wood et al., 2021).
In order to better understand the role of immune cells in human milk in shaping the neonatal immune system, reliable, standardized cell isolation methods are necessary to minimize the variation of results arising from the use of the inconsistent methodology in a biological sample that is already intrinsically subject to wide heterogeneity. For instance, human milk varies extensively in nutritional and immunological composition during the different stages of lactation (Bardanzellu et al., 2017). Colostrum, the first stage of lactation after delivery, has high immunological components in comparison to transitional and mature human milk (Pang & Hartmann, 2007). Maternal diseases and infections, such as mastitis or preterm delivery, are also well-known factors affecting the cellular composition of human milk (Ballard & Morrow, 2013).
Previous work on immune cells in human milk has used various isolation techniques and protocols. Researchers have employed different centrifugation settings (i.e., velocity, time, temperature) and dilution rates to isolate lymphocyte populations from human milk; however, there is not a standardized protocol resulting in variable cell retrieval rates across studies (Armistead et al., 2023; Baban et al., 2018; Bertotto et al., 1990; Cabinian et al., 2016; Cérbulo-Vázquez et al., 2018; Hassiotou et al., 2012; Jain et al., 1991; Järvinen & Suomalainen, 2002; Jin et al., 2011; Lazar et al., 2021; C. Li et al., 2017; S. Li et al., 2019; Ten-Doménech et al., 2023; Trend et al., 2015; Wirt et al., 1992).
Magnetic-activated cell sorting (MACS) using antibody-coated magnetic beads can isolate T lymphocytes, including the Treg subset, from whole blood. Commercial kits employ positive or negative selection for immunomagnetic separation of the target cells. Positive selection uses magnetic antibodies that directly bind to target cells, such as anti-CD3 antibodies in the case of T cells, retaining these cells on a magnetic column during isolation. On the contrary, during negative selection, a magnetic antibody cocktail is used to retain all other anticipated cell types from the sample on the column except for the target cells. Immunomagnetic isolation is easy to use, enriches the target cells, and is a well-established method in blood samples (Chiu et al., 2019; Clarke & Davies, 2001; Schuler et al., 2011). Positive immunomagnetic bead selection of lymphocytes is considered to be a feasible method in human milk samples. Indeed, one of the above human milk studies employed immunomagnetic cell isolation (Bertotto et al., 1990); however, the performance of this method in human milk compared to centrifugation alone has not yet been evaluated for the selection of T lymphocytes. Therefore, the aim of our study was to systematically compare available cell isolation techniques, including centrifugation and immunomagnetic separation, to isolate T lymphocytes from human milk samples.
Methods
Research Design
This study includes a set of method optimization experiments, and a single group of human milk donors was included. The study design was a repeated measures design. This design controls for individual differences since each participant serves as their own control. It also requires fewer participants than other designs to detect differences between different cell isolation techniques. The study was conducted in accordance with the Declaration of Helsinki, and approved by the New Zealand Health and Disability Ethics Committee (HDEC 2022 AM 11657).
Key Messages
Human milk is intrinsically heterogeneous. Therefore, reliable and standardized cell isolation methods are necessary to minimize methodologic variation in results.
The performance of immunomagnetic cell isolation is one method used for cell isolation in human milk, but it has not been compared to centrifugation alone and has not yet been evaluated for the selection of T lymphocytes.
Immunomagnetic separation is a feasible method to isolate T lymphocytes in human milk, which is similar to the process that is used for isolation from blood samples.
Immunomagnetic separation improves target cell enrichment and cell viability from human milk compared with the process of centrifugation.
Setting and Relevant Context
This study was conducted in a developed country with a healthcare system that promotes breastfeeding. The study area was culturally and socioeconomically diverse, with ethnicities including European, Māori, and Pacific people. The infants of all study participants were born in a hospital setting. Although lactation advice is broadly available to new mothers both in the community and hospital settings, only one in five infants are exclusively breastfed to 6 months of age, with rates consistently lower for Māori and Pacific people. Local major barriers to breastfeeding include perinatal distress, socioeconomic pressure, and insufficient parental leave (Castro et al., 2017).
Sample
The target population for this study was defined as healthy lactating mothers. Samples of freshly expressed mature human milk (10–30 ml collected at the start of the feed) were obtained from eight lactating mothers who were exclusively breastfeeding and not regularly using breast pumps between February and May 2023. Participants did not receive any compensation for participating in this study. Using electric breast pumps, milk was expressed into sterile plastic containers. The time between human milk sample collection and complete processing was consistently less than 3 hours, and samples were kept at room temperature during this time. Mothers were donors with no reported acute or chronic illness at the time of sample collection. Infants of mothers were not ill or unwell at the time of sample collection. The only inclusion criterion was giving birth at Auckland City Hospital, Auckland, New Zealand. Exclusion criteria were any acute or chronic illness during sample collection, including acute or chronic infections. While this was an exploratory study, our sample size was guided by assuming a detectable difference of 20% in the measured flow cytometry parameters, based on the available literature.
Measurement
Milk samples from all participants were subjected to three different cell isolation methods, as detailed below: centrifugation, immunomagnetic separation, and the combination of centrifugation and immunomagnetic separation. Isolated cells from all three methods were processed using the same flow cytometry protocol as detailed below.
Centrifugation optimization
Our centrifugation protocol was tested using frozen mature human milk samples in preliminary experiments obtained from separate healthy donors and thawed in a 37°C water bath before processing. The protocol was based on previously available data in the literature (Armistead et al., 2023; Baban et al., 2018; Bertotto et al., 1990; Cabinian et al., 2016; Cérbulo-Vázquez et al., 2018; Hassiotou et al., 2012; Jain et al., 1991; Järvinen & Suomalainen, 2002; Jin et al., 2011; Lazar et al., 2021; C. Li et al., 2017; S. Li et al., 2019; Ten-Doménech et al., 2023; Trend et al., 2015; Wirt et al., 1992) and our preliminary results were based on viable cell counts following centrifugation, determined through microscopy following Trypan Blue staining by a single operator using a Neubauer Improved Haemocytometer Counting Chamber. Running the samples at 4°C was found to aid the natural separation and manual removal of the fat layer without compromising the cell retrieval, so this temperature was selected for the final protocol. At this temperature, the lipid layer becomes solid on cooling, allowing more efficient removal as it does not promptly dissolve back into the skim milk supernatant. A 1:1 dilution was found to yield similar cell counts to both higher and lower dilution factors, while improving the separation of the fat and skim milk layer with the cell pellet at the bottom. Dilution likely minimized contamination of the pellet with the milk during manual removal, allowing the supernatant to wash out excess fat when poured off, thus minimizing the number of cells trapped in the lipid layer.
The final optimized centrifugation protocol is described herein: 1 ml aliquots of fresh mature milk samples were diluted 1:1 in PBS and centrifuged at 800 g (1850 RPM) for 10 minutes at 4°C. The dense lipid layer was removed with a Pasteur pipette, and the skim milk supernatant was poured out. The cell pellet was then resuspended in 1 ml PBS, transferred to a new tube, and pelleted again by centrifugation at 800 g (1850 RPM) for 10 minutes at 22°C. The supernatant was discarded, and the pellet was retained for downstream application.
Immunomagnetic separation
One ml aliquots of fresh milk samples from each of the eight donors were analyzed using flow cytometry following cell isolation by three different methods: centrifuged cells using the optimized centrifugation protocol (described in the Results section); centrifuged cells that also underwent immunomagnetic separation; and a 1:1 PBS diluted sample that underwent immunomagnetic separation without centrifugation. A peripheral blood mononuclear cell sample, isolated using standard gradient centrifugation, was also included as a control for immunomagnetic isolation. CD3 cells from samples subjected to immunomagnetic isolation were positively selected by incubation with anti-human CD3 microbeads (Miltenyi Biotech, #130-050-101) according to the manufacturer's guidelines for blood samples. The volume of microbead antibody was set at 20 μl for 1 ml milk aliquots following titration of antibody concentrations of 10 μl, 20 μl, and 40 μl. Following magnetic isolation, samples were centrifuged at 800 g (1850 RPM) for 10 minutes at 22°C and cell pellets were frozen at -80°C in fetal bovine serum (FBS) + 10% dimethyl sulfoxide (DMSO) until further processing.
Flow cytometry analysis
Fluorescent antibody staining
Frozen cells were thawed together for flow cytometry analysis in a 37°C water bath. Thawed samples were washed in PBS and centrifuged at 800 g (1850 RPM) for 7 minutes at 22°C. The supernatant was discarded, and the cell pellet was stained with Live/Dead Aqua viability dye (Invitrogen, #L34966) and incubated in the dark at room temperature for 30 minutes. Following incubation, samples were washed in MACS buffer (PBS, 2% FBS, 4 mM EDTA), and centrifuged at 800 g (1850 RPM) for 7 minutes at 22°C. The supernatant was discarded, and the cell pellet was stained with anti-CD3 (Biolegend, #344824), anti-CD4 APC-Cy7 (Biolegend, #344616), and anti-CD25 PE/Fire700 (Biolegend, #356146) monoclonal anti-human antibodies as per manufacturer guidelines. The samples were then incubated in the dark for 30 minutes on ice, washed with MACS buffer, and centrifuged at 800 g (1850 RPM) for 7 minutes at 22°C. The cell pellets were run on a BD FACS Aria II flow cytometer. Data were analyzed using the FlowJo software (Becton Dickinson & Company, v10.9).
Gating strategy
First, lymphocytes were identified based on forward and side scatter characteristics. Next, doublets were excluded based on forward scatter area and height characteristics. Live cells were identified based on Live/Dead Aqua negativity, T cells were identified based on CD3 positivity, and CD4 positive cells were identified within the CD3 positive cell population. CD25 positive cells were identified within the CD4 positive cell population. Tregs were identified as CD3+ CD4+ CD25 high cells (Figure 1). Data were analyzed with FlowJo software (Version 10.9).
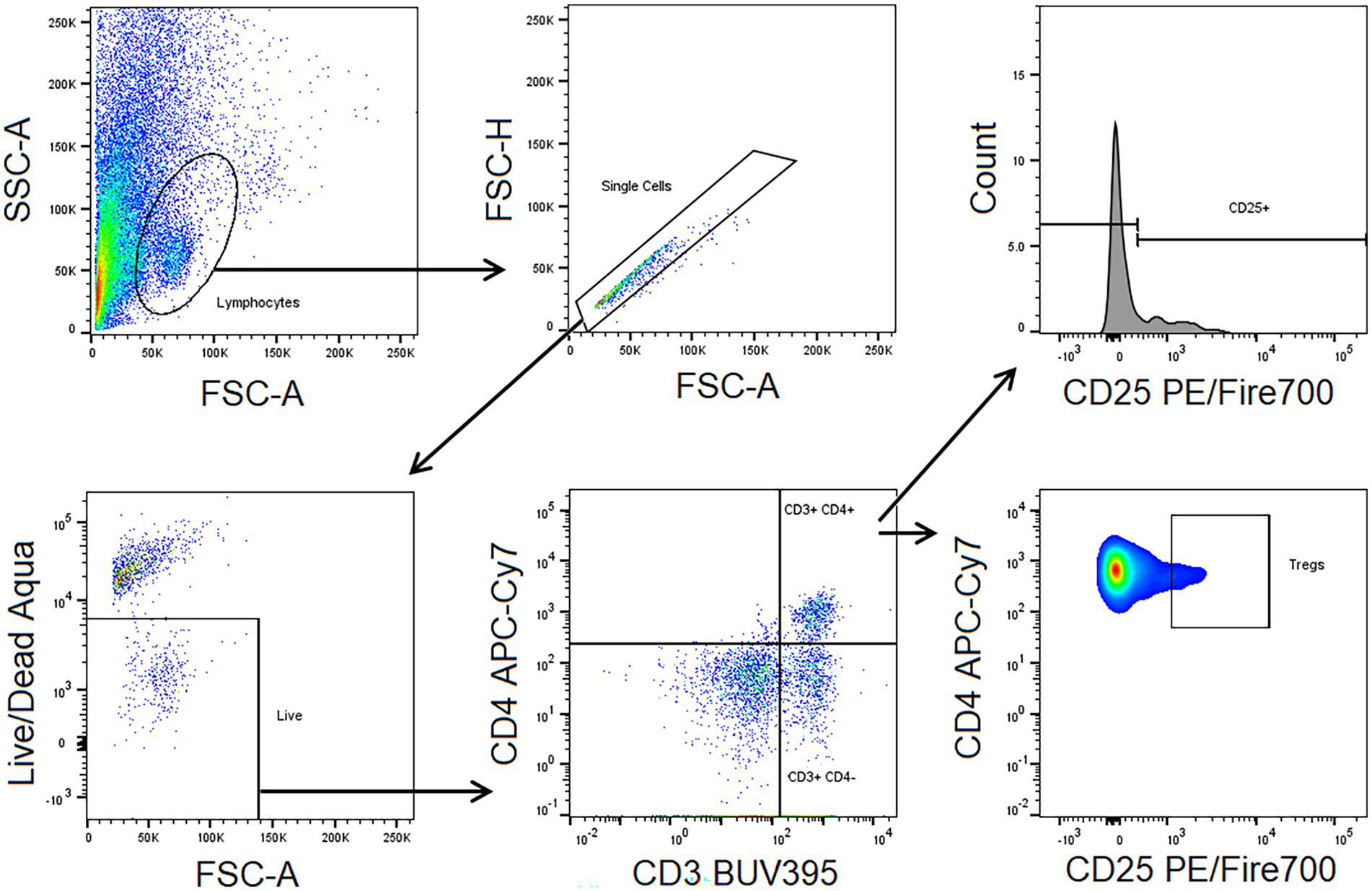
Gating Strategy of Flow Cytometry Measurements Depicting the Sequence of Gates.
Data collection
Milk samples were collected from participants between February and May 2023. Informed consent was obtained by the principal investigator from all subjects involved in the study. Participants self-reported being generally healthy and having no acute or chronic illness, including infections, at the time of recruitment. Hospital notes from participants were not accessed as part of this study, and data from medical records were not collected. Samples were de-identified at the time of sampling by a unique study code. De-identification was performed by the principal investigator, and flow cytometry data were stored on secure, password-protected computers.
Data analysis
Repeated measures ANOVA tests with Tukey’s multiple comparisons tests were used to compare the tested centrifugation and immunomagnetic isolation methods; p values less than 0.05 were regarded as significant, and less than 0.1 showed a trend. Sphericity was not assumed, and the Geisser-Greenhouse correction was applied. Gaussian data distribution was assumed based on the Shapiro-Wilk and Kolmogorov-Smirnov tests for normality. Dependent variables measured included the lymphocyte count, the proportion of live cells within lymphocytes, the proportion of CD3+ cells and CD4+ cells within live lymphocytes, the proportion of CD25+ cells and Tregs within CD4+ cells, the mean fluorescence intensity of CD25, as well as CD3+, CD4+, CD25+ and Treg cell counts. Statistical analysis was carried out with GraphPad Prism 9 software.
Results
Characteristics of the Sample
Samples were provided at a median postpartum age of 16 weeks (interquartile range [IQR] 10–34 weeks). The median gestational age at delivery was 35 weeks (IQR 27–39 weeks), and four babies were born preterm (< 37 weeks of gestation).
A repeated measures ANOVA was conducted to compare the effects of three isolation methods (immunomagnetic separation applied to diluted milk samples, immunomagnetic separation applied to cell pellets from centrifuged milk samples, and centrifugation alone) on cell counts and proportions. Analysis of flow cytometry data for CD3+, CD4+, and CD25+ cells allowed comparison of isolation techniques with regard to cell proportions and cell counts (Figure 2).
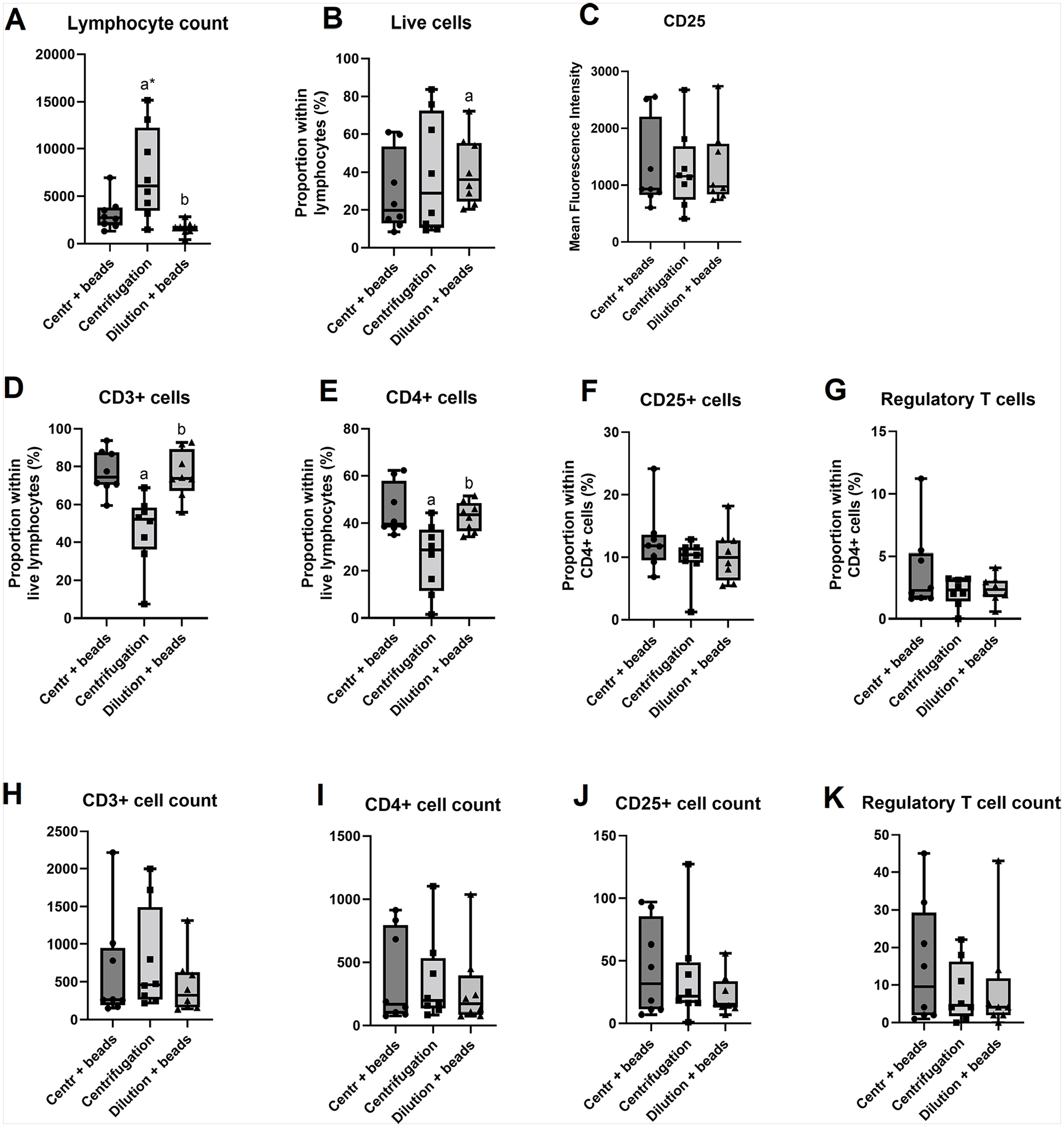
Cell Count and Proportion Comparisons Between the Three Isolation Techniques on Eight Human Milk Donor Samples. A – Lymphocyte count, B – Live cells, C – CD25, D – CD3+ cells, E – CD4+ cells, F – CD25+ cells, G – Regulatory T cells, H – CD3+ cell count, I – CD4+ cell count, J – CD25+ cell count, K – Regulatory T cell count
Lymphocyte Count
There was a significant effect of the isolation method on lymphocyte count (F = 8.613, p = 0.017, R2 = 0.552, with post-hoc Tukey’s multiple comparisons tests revealing that centrifugation alone resulted in significantly higher and more variable lymphocyte counts compared to both magnetic isolation methods (Figure 2A). This reduction in cell count with the magnetic beads is attributed to the loss of non-T cells from the total lymphocyte population as the beads targeted and retained CD3+ T cells only as opposed to centrifugation alone.
Viability
The proportion of live cells within the lymphocyte population appeared higher in diluted magnetically isolated samples than in centrifuged samples with or without magnetic isolation (F = 8.608, p = 0.021, R2 = 0.564, Figure 2B). This indicates better viability of cells without centrifugation.
Lymphocyte Subsets
While the lymphocyte cell count is higher in the centrifuged sample (Figure 2A), this method returned the lowest CD3+ proportion (F = 9.319, p = 0.016, R2 = 0.571, Figure 2D), again an attribute of the larger total lymphocyte population in the centrifuged sample which also contains cell types other than T cells. While the absolute CD3+ cell counts were comparable for all three methods (Figure 2H), the proportion of these cells within the total lymphocytes was much higher in both magnetically isolated samples (Figure 2D), indicating better selection of the target cells with the magnetic beads.
The same pattern was noted for CD4+ cells where absolute cell counts were comparable (Figure 2I), but both magnetically isolated samples returned a higher CD4+ proportion within lymphocytes (F = 8.03, p = 0.01, R2 = 0.534, Figure 2E).
CD25+ cell proportions (Figure 2F) and absolute cell counts (Figure 2J), as well as CD25 mean fluorescence intensities (MFIs; Figure 2C) were comparable across all isolation methods. The comparable expression of the CD25 activation marker across all isolation techniques indicates that the beads did not inadvertently activate cells. The proportion and absolute cell count of a rare cell T lymphocyte subset—Tregs—was also similar across the three methods (Figures 2G and K), although a higher trend of cell counts with the combination of centrifugation and magnetic beads may be present over the other two methods.
Discussion
Using immunomagnetic cell separation, we aimed to create an optimized protocol to isolate T lymphocytes, specifically Tregs, from human milk. This work was the first attempt to compare T cell isolation methods in human milk systematically. Antibody-coated magnetic beads are used to isolate T lymphocytes and other immune cell subsets of interest from blood, based on established literature (Chiu et al., 2019; Clarke & Davies, 2001; Schuler et al., 2011) in the field of immunology. This study demonstrates that immunomagnetic separation of T lymphocytes is also feasible from human milk in combination with an optimized centrifugation protocol.
The representative flow cytometry graphs below (Figure 3), comparing human milk samples (Figures 3B–D) with a peripheral blood mononuclear cell sample control (Figure 3A) following CD3+ magnetic bead isolation, revealed less defined lymphocyte populations with a higher degree of cell contamination in human milk than blood. Human milk samples had a distinct band of events outside the lymphocyte gate, potentially indicating lipid globules, cellular debris, or other non-immune cell types, such as stem or epithelial cells, that could have been trapped on the magnetic columns during isolation, thus contaminating the samples eluted from the columns.
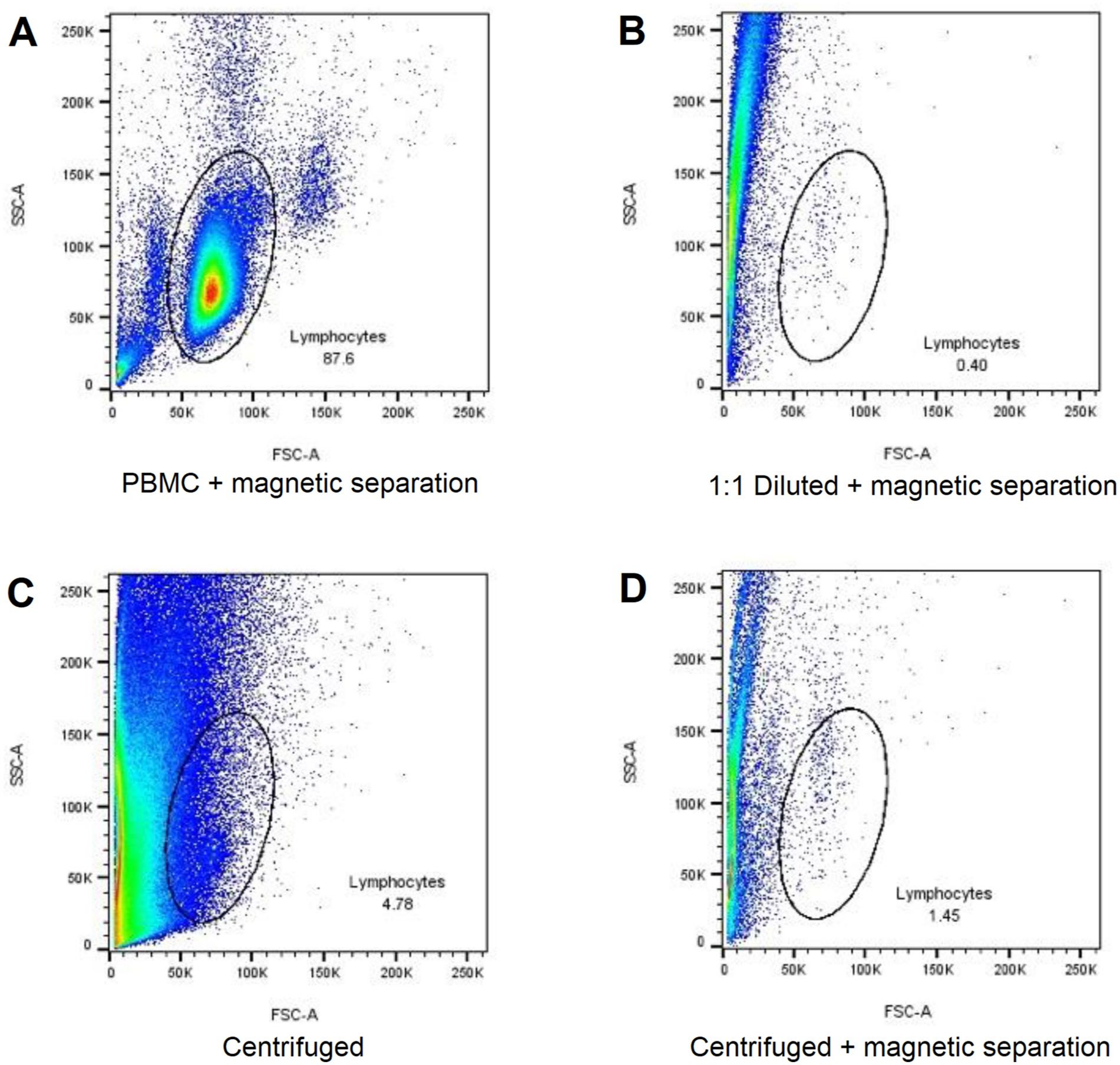
Representative Flow Cytometry Dot Plot Graphs of Blood and Human Milk Samples Highlighting the Percentage Lymphocytes of Total Events.
Nevertheless, immunomagnetic isolation enriched the target cells within the obtained samples (Figure 2D) compared to centrifugation alone, meaning that subsequent use of the sample will have less interference or contamination by other cells and milk components. This may be a benefit in experiments where the aim is to phenotype rare cell types, such as Tregs. However, the disadvantages of this technique include the high cost of antibody-coated magnetic beads and the need to process each sample individually, using the magnetic column, thus prolonging overall sample processing times.
A further concern regarding the use of magnetic antibodies may be their effect on cell activation. According to our results, the similar CD25 protein expression of cells across all methods supports the use of magnetic bead antibodies to isolate cells where downstream applications are either phenotyping or cellular assays, as the cells do not seem to be excessively activated in comparison to centrifugation alone.
In the centrifuged sample, the absolute lymphocyte count is higher as the population contains a variety of immune cells such as B lymphocytes, NK cells, and others. This method is cost effective and can be applied in experiments where a large event size is a requirement. However, lower event numbers of less frequent cell populations may be a disadvantage if the aim of the experiment is to characterize rare cell types. Additionally, the decreased viability with centrifugation is a necessary consideration if the intended use of cells is in functional assays or cell cultures over phenotyping.
Using magnetic isolation following centrifugation while removing a large amount of debris and fat (Figure 3D) returned similar CD3+ proportions and cell counts to magnetic isolation alone (Figure 2H). This indicates that although debris removal was expected to be easier with magnetic isolation following centrifugation, the advantages of removing the fat and skim milk manually with centrifugation are not necessarily reflected in the cell retrieval. A potential advantage of the combination of centrifugation and magnetic isolation over either of the two methods used alone may be a higher trend of counts of rare cell types, such as Tregs (Figure 2K); however, this increase was not present in other investigated cell types in the current study. This work was necessary in light of the growing interest in investigating the immune cell composition of human milk, and the lack of methodological consensus in earlier literature.
Limitations
A limitation of our current study was the small sample size of donors, particularly when considering the heterogeneity and dynamic composition of human milk across different stages of lactation. The impact of expression technique, milk maturity and maternal illness on the cellular composition of human milk was also not assessed in the current study but merits further investigation in future studies. While we acknowledge that factors such as time of day or time since the last feed contribute to the variability of lymphocyte counts in human milk, the individual effects of these factors were not assessed. Similarly, in the current study, we only applied a limited number of fluorescent cell surface antibodies, as our aim was method optimization rather than in-depth phenotyping of the isolated cells. A further limitation is that factors such as time of day and time since the last feed—known to influence lymphocyte counts—were not standardized for sample collection.
Conclusion
Overall, immunomagnetic separation is a feasible method to isolate T lymphocytes in human milk, similar to blood. Immunomagnetic isolation improves target cell enrichment and cell viability compared to centrifugation alone, which may be an advantage when the goal is to characterize rare cell types, or when cells are further used in functional assays. However, a drawback of this technique may be the cost and prolonged sample processing times. The choice of immune cell isolation technique in human milk (magnetic separation versus centrifugation) depends on the intended downstream use of cells and assay requirements for each experiment.
Footnotes
Acknowledgements
The authors wish to thank Professor Frank Bloomfield (Liggins Institute, University of Auckland, Auckland, New Zealand) for his contribution to the study design and critical reading of the manuscript.
Authors’ Note
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Contributions
Disclosures and Conflicts of Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mariana Muelbert and Gergely Toldi are advisors of students Noor Radhi and Ayamita Paul. The authors declare no other disclosures or conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Auckland Medical Research Foundation (Grant Number: 1122015).
