Abstract
Background:
Raw, never stored or pasteurized mother’s own milk (MOM) is not always available to feed preterm infants; however, storage and pasteurization of MOM diminishes some bioactive components. It can be difficult to feed raw MOM to preterm infants due to transportation and storage of small volumes that might be pumped away from the infant, and a concern that they might harbor bacteria. However, the higher availability of bioactive components in raw MOM may provide benefits to preterm infants compared to frozen or pasteurized MOM.
Research Aim:
To systematically review and summarize the results of studies on feeding raw MOM versus frozen or pasteurized MOM to preterm infants born at less than 37 weeks of gestation.
Methods:
Four databases were searched (Cochrane, Embase, Ovid MEDLINE, and Web of Science) for this systematic review. Of 542 studies identified, nine met inclusion criteria and were critically evaluated using the quality assessment tool for quantitative studies by the Effective Public Health Practice Project. Studies were organized using the Breastfeeding Challenges Facing Preterm Mother–Infant Dyads theoretical framework.
Results:
Included studies evaluated the outcomes of preterm infants fed raw versus pasteurized MOM (n = 7, 77.8%) or raw versus frozen MOM (n = 2, 22.2%). Researchers found that raw MOM did not increase infant infections and may have improved health and growth outcomes for study participants.
Conclusion:
There is laboratory evidence supporting the safety and efficacy of the use of raw MOM for preterm infants. A raw MOM diet is recommended for preterm infants by professional organizations. Despite this, it may not be universally prioritized and could require purposeful implementation by each institution. Further research is needed to pursue the potential benefits of a raw MOM diet for preterm infants.
Keywords
Key Messages
Raw, never stored or pasteurized, mother’s own milk is not frequently used to feed preterm infants; however, storage and pasteurization of mother’s own milk diminish some bioactive components.
This review found positive growth and health benefits and no negative outcomes with a raw mother’s own milk diet versus frozen or pasteurized mother’s own milk.
Raw mother’s own milk has the potential to improve preterm infant health outcomes but has not been fully explored in the preterm infant population and requires further study.
Background
High-dose mothers’ own milk (MOM) from birth to neonatal intensive care unit (NICU) discharge may help reduce healthcare costs (Meier et al., 2017). Researchers have found that MOM may reduce preterm infant morbidity, including improved neurological outcomes to 7 years of age (Belfort et al., 2022), decrease the risk of bronchopulmonary dysplasia (BPD; Xu et al., 2020), and reduce the risk of necrotizing enterocolitis (NEC) and sepsis (Cortez et al., 2018; Gopalakrishna et al., 2019). The health benefits of MOM are due primarily to nutritional and bioactive components that have synergistic mechanisms of action and change with the stage of lactation (Gila-Diaz et al., 2019). Bioactive components including secretory immunoglobulin A (sIgA; Palmeira & Carneiro-Sampaio, 2016), hormones (Mazzocchi et al., 2019), stem cells (Kakulas, 2015), oligosaccharides, probiotic microorganisms (Pannaraj et al., 2017), and antioxidants (Paduraru et al., 2018), aid infant growth and development (Meier et al., 2018) and improve neonatal immunological health (Munblit et al., 2017).
However, the storage of MOM and the passage of time diminish some of its bioactive components (Lawrence, 1999; Paulaviciene et al., 2020; Vass et al., 2020). For example, stem cells—believed to influence immune development and assist in intestinal tissue repair (Molès et al., 2018)—begin to decrease several hours after milk extraction (Kakulas, 2015). Some countries routinely pasteurize MOM, which further diminishes its bioactive properties (Aceti et al., 2020; Agence Francaise de Securite Sanitaire des Aliments, 2005; Vass et al., 2020). However, healthcare organizations, for example, the American Academy of Pediatrics (AAP) and the European Society of Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) recommend raw mother’s own milk (rMOM) for preterm infants (American Academy of Pediatrics [AAP], 2012).
In the neonatal intensive care unit (NICU), rMOM is often not prioritized, possibly owing to the tiny volumes consumed in the first weeks to months of life, pumping that occurs at locations away from the infants, centralization of milk management, and perceived risk for cytomegalovirus (CMV) transmission. For these reasons, much of the pumped MOM is frozen or pasteurized until needed. In contrast, the healthy term infant can receive fresh MOM during direct breastfeeding.
Conceptual Framework
This review was framed by the Breastfeeding Challenges Facing Preterm Mother–Infant Dyads framework (Figure 1; Lau, 2018). This framework describes concepts that influence human milk production for mothers of preterm infants in the NICU and demonstrates how improving the mother's ability to pump can positively affect her ability to provide sufficient human milk. While a human milk diet improves the infant's overall growth and health, raw human milk may further augment those benefits. The aim of this review was to summarize the results of feeding raw MOM versus frozen or pasteurized MOM to preterm infants born at less than 37 weeks gestation.
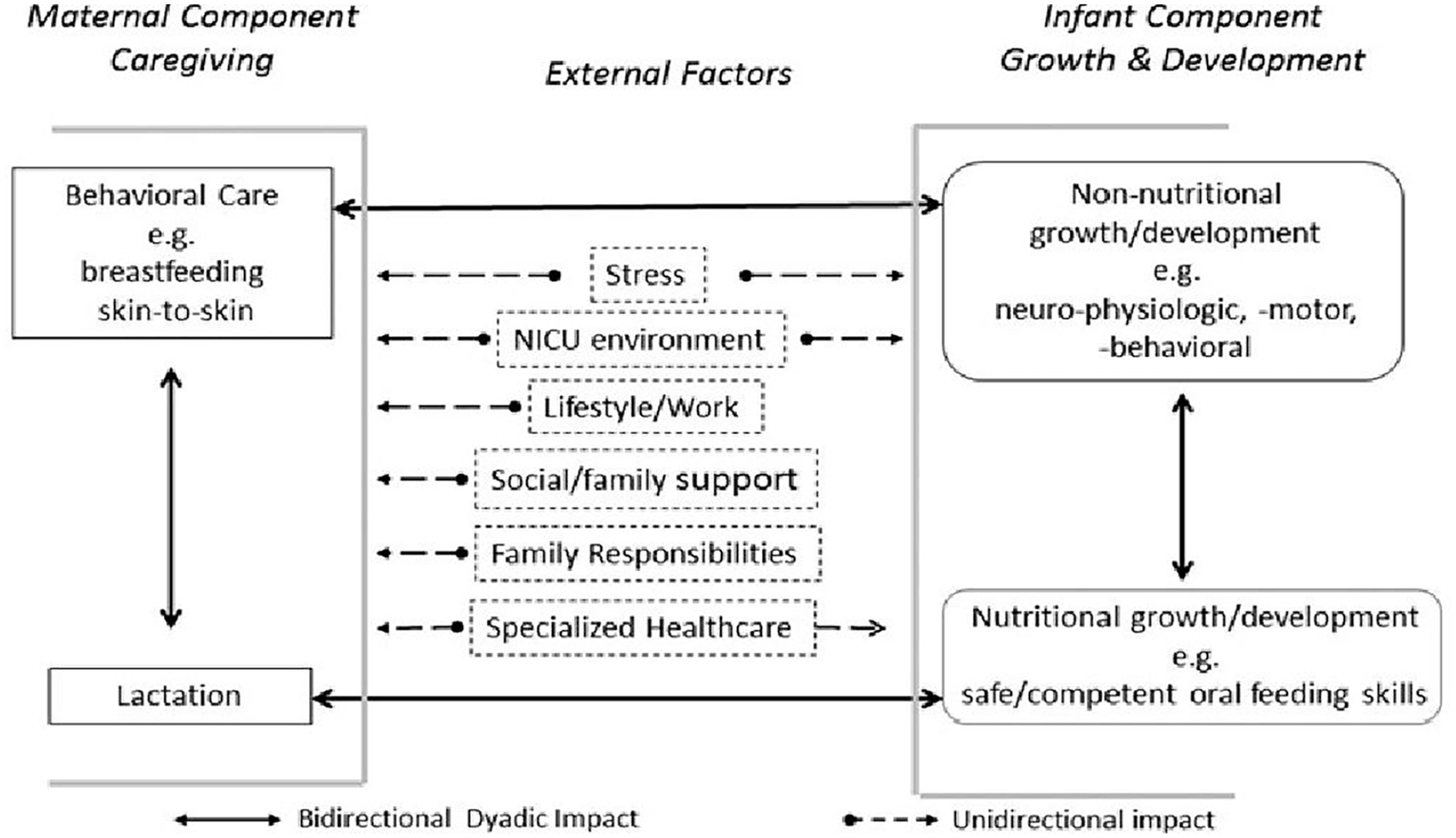
Conceptual Model of Breastfeeding Challenges Facing Preterm Mother–Infant Dyads (Lau, 2018).
Methods
Research Design
A systematic review was used to identify studies that compared the effects of feeding raw MOM versus altered MOM (pasteurized or frozen) for preterm infants’ health. The approach allowed us to identify relevant research and critically appraise the findings. The protocol and search criteria were registered on PROSPERO (CRD42022367550).
Sample: Defining the Articles Reviewed
Table 1 summarizes the elements of the nine studies included in this review. Sample sizes ranged from 12 to 926 preterm infants. Publication dates ranged from 1984 to 2023 with seven of the nine studies (77.8%) published in the last 10 years. Studies were from Sweden (n = 1), Belgium (n = 3), France (n = 2), India (n = 1), and China (n = 2). Minimal race and ethnicity information was provided in the included studies. All studies had a quantitative design; three were randomized controlled trials (33.3%), one was a randomized crossover (11.1%), four were prospective observational studies (44.4%), and one was a retrospective observational study (11.1%). All had at least one aim that compared the results of raw MOM (rMOM) and altered MOM (aMOM) on preterm infant outcomes.
Study Characteristics for Raw Versus Frozen or Pasteurized Mother’s Own Milk Review (N = 9).
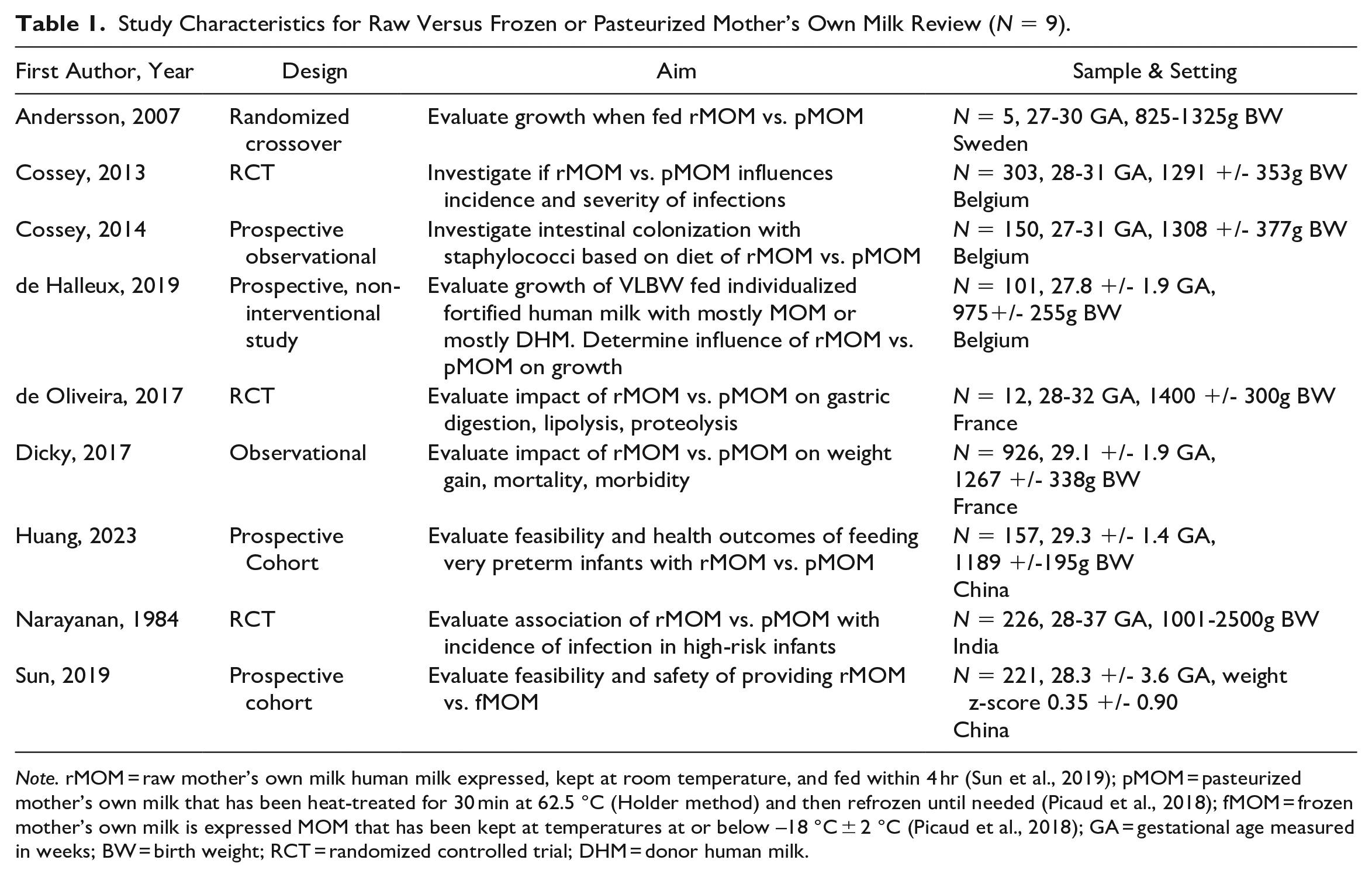
Note. rMOM = raw mother’s own milk human milk expressed, kept at room temperature, and fed within 4 hr (Sun et al., 2019); pMOM = pasteurized mother’s own milk that has been heat-treated for 30 min at 62.5 °C (Holder method) and then refrozen until needed (Picaud et al., 2018); fMOM = frozen mother’s own milk is expressed MOM that has been kept at temperatures at or below –18 °C ± 2 °C (Picaud et al., 2018); GA = gestational age measured in weeks; BW = birth weight; RCT = randomized controlled trial; DHM = donor human milk.
We included peer reviewed studies published in English. All research methods that addressed the aim were considered (Figure 2). In addition, the bibliographies of the included studies were reviewed. The PRISMA checklist was used to ensure transparency and thoroughness (Page et al., 2021).
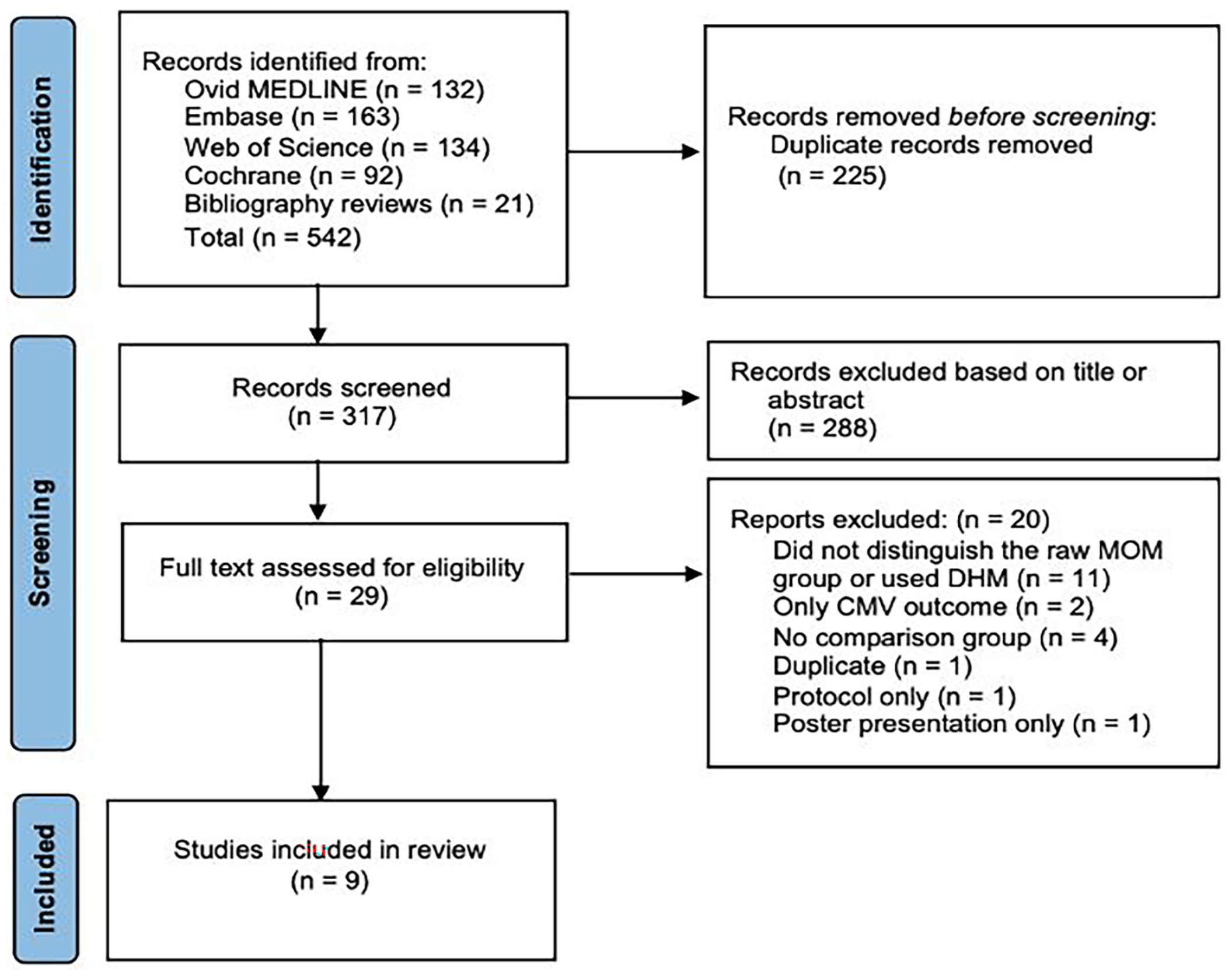
PRISMA Flow Diagram for Identification of Studies Included in Review.
Articles were excluded if the rMOM group was not defined, if they used donor human milk (DHM), did not include results (protocol only), or did not include a comparison group. Studies that only included CMV infection as an outcome were excluded as postnatally-acquired CMV is controversial, making the results difficult to interpret (Gunkel et al., 2018; Park et al., 2021).
Data Collection
Given the sparsity of evidence and no previous review published using this search criteria, no date limits were placed on the search. The search was completed with the assistance of an academic research librarian on January 17, 2023, using the Cochrane Database of Systematic Reviews, Embase, Ovid MEDLINE, and Web of Science. The search was completed using the following keywords: human milk, frozen OR pasteurized, fresh OR raw, prematurity, and low birthweight. The full search criteria are reported with registered protocol on PROSPERO CRD42022367550.
Measurement
In the literature, “fresh MOM” and “raw MOM” are used interchangeably to mean human milk expressed, kept at room temperature, and fed within 4 hr (Sun et al., 2019). Frozen MOM (fMOM) is defined as expressed MOM that has been kept at temperatures at or below −18 °C ± 2 °C (Picaud et al., 2018), and pasteurized MOM (pMOM) is defined as MOM that has been heat-treated for 30 min at 62.5 °C (Holder method) and then refrozen until needed (Picaud et al., 2018). There is evidence that frozen and pasteurized MOM undergo similar changes to bioactive protein compositions (Kaya & Çınar, 2023; Paulaviciene et al., 2020). In this review, fMOM or pMOM is referred to as altered MOM (aMOM) and was compared with rMOM.
Studies were initially screened by the primary author (JG) based on titles and abstracts. The review software PICO Portal removed all duplicates (PICO Portal, 2022). The remaining articles underwent an independent, full-text review by the primary author (JG) and another master’s prepared nurse, to minimize bias. A data coding sheet with selected study information was compiled independently by each reviewer for each included study with the following headings: citation, country, setting, definition of rMOM or aMOM, study design, study aim, sample characteristics, inclusion criteria, exclusion criteria, analysis, findings, and recommendations. Any disagreements between the two reviewers were discussed and resolved. The percent agreement between the independent coders was calculated using Cohen’s kappa (κ = 1.0).
The findings were organized by the guiding framework concepts of non-nutritional growth/development and nutritional growth/development. Non-nutritional growth/development is defined as the infant's overall health and medical status, and nutritional growth/development is defined as the infant's anthropometric growth and development of oral feeding skills (Lau, 2018).
Data Analysis
Study strengths and limitations were extracted and evaluated by all authors. The Effective Public Health Practice Project (EPHPP) quality assessment tool checklist for quantitative studies was used to evaluate internal methodological congruence and risk of bias as strong, moderate, or weak (Thomas et al., 2004). Quality and bias were evaluated based on the selection of participants, study design, confounders, blinding, data collection, withdrawals and dropouts, intervention integrity, and analysis (Thomas et al., 2004). The reviewers discussed and agreed upon the final quality rating of the studies. Following quality appraisal, the study results were discussed based on concepts from the guiding conceptual model of Breastfeeding Challenges Facing Preterm Mother–Infant Dyads (Lau, 2018).
Results
Quality Analysis
Table 1 summarizes the elements of the nine studies included in this EPHPP (Thomas et al., 2004). Two of the nine studies were appraised as weak owing to the possibility of sampling bias related to small sample size and potential for crossover effects (Andersson et al., 2007) or risk of bias from participants having a short hospital stay (< 4 days; Narayanan et al., 1984). One study was appraised as moderate owing to the risk of sampling bias (de Oliveira et al., 2017). The remaining six studies were appraised as strong (Cossey et al., 2013, 2014; de Halleux et al., 2019; Dicky et al., 2017; Huang et al., 2023; Sun et al., 2019). A summary of included study variables, measurement processes, key findings, and quality is provided in Table 2.
Summary of Variables, Measurement, Analysis Processes, and Quality for Included Studies Evaluating any Effects of Fresh MOM Versus Frozen or Pasteurized MOM (N = 9).
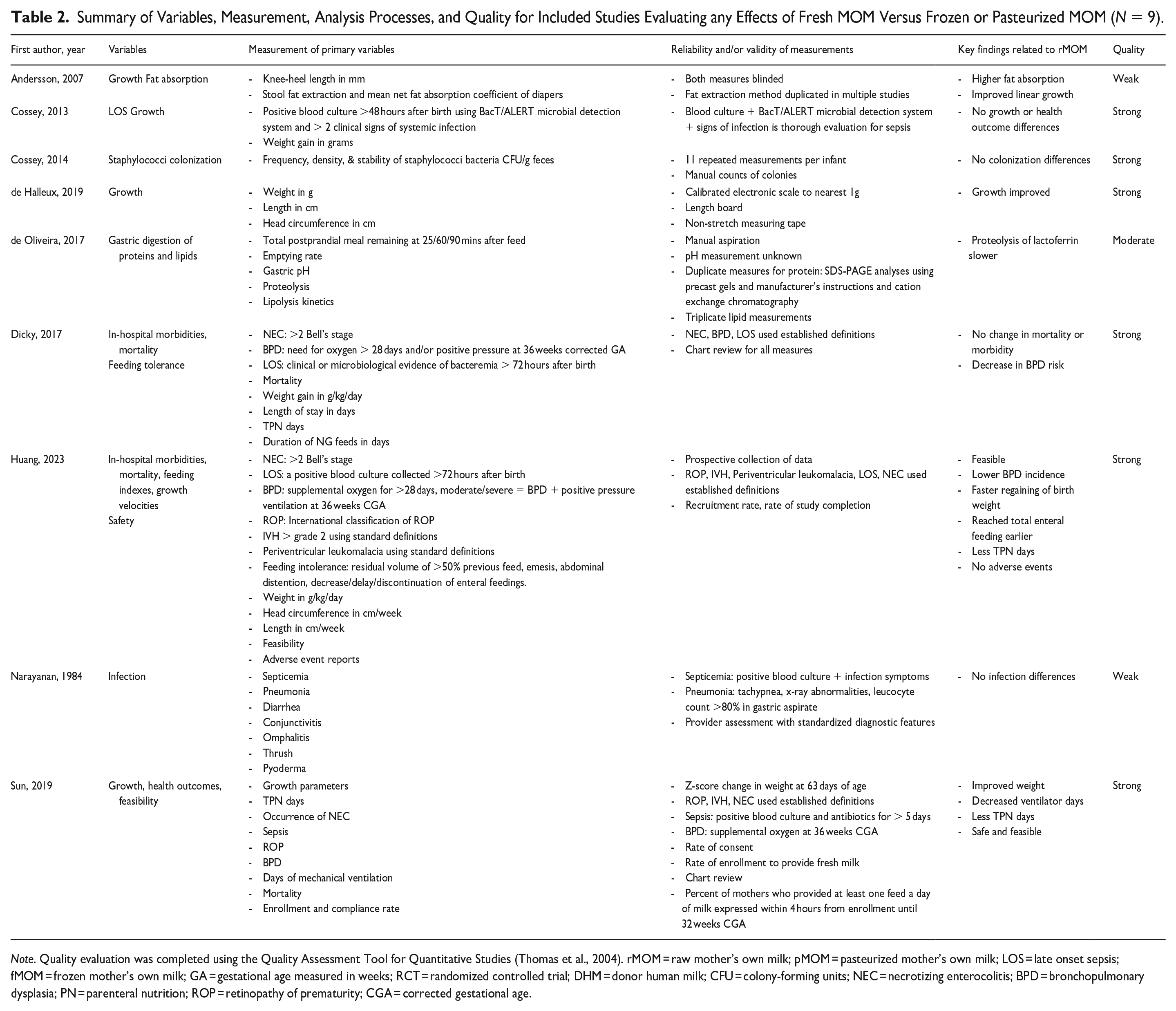
Note. Quality evaluation was completed using the Quality Assessment Tool for Quantitative Studies (Thomas et al., 2004). rMOM = raw mother’s own milk; pMOM = pasteurized mother’s own milk; LOS = late onset sepsis; fMOM = frozen mother’s own milk; GA = gestational age measured in weeks; RCT = randomized controlled trial; DHM = donor human milk; CFU = colony-forming units; NEC = necrotizing enterocolitis; BPD = bronchopulmonary dysplasia; PN = parenteral nutrition; ROP = retinopathy of prematurity; CGA = corrected gestational age.
Nutritional Growth and Development
Of the nine studies, five examined growth-related outcomes comparing rMOM to aMOM (Andersson et al., 2007; Cossey et al., 2013; de Halleux et al., 2019; Huang et al., 2023; Sun et al., 2019). Cow’s milk based human milk fortifier was added equally across the study groups in four of the studies (Cossey et al., 2013; de Halleux et al., 2019; Huang et al., 2023; Sun et al., 2019). No fortifiers were used in one of the studies that reported growth-related outcomes (Andersson et al., 2007). Significantly improved weight gain with rMOM was noted in three studies, demonstrated by a faster regaining of birth weight (p = .021; Huang et al., 2023), an increased weight gain of 1.96 g/kg/day (p < .001; de Halleux et al., 2019), and improved z-score weight gain (p = .01, CI [0.12, 3.67]; Sun et al., 2019). None of the included studies reported negative growth findings.
Non-Nutritional Growth and Development
Seven of the included studies reported a decreased or unchanged risk of deleterious health outcomes for preterm infants fed rMOM (Cossey et al., 2013, 2014; de Oliveira et al., 2017; Dicky et al., 2017; Huang et al., 2023; Narayanan et al., 1984; Sun et al., 2019). Three studies specifically evaluated the risk for preterm infant infections (Cossey et al., 2013; Narayanan et al., 1984; Sun et al., 2019). One found a significant decrease in NEC or mortality (relative risk [RR] 0.45, CI [0.21, 0.96], p = .04; Sun et al., 2019). Two other studies found no significant differences in the incidence of infection (Cossey et al., 2013; Narayanan et al., 1984). In one study, a protective effect from infection with rMOM was seen when both rMOM and pMOM were alternated with human milk substitute (Narayanan et al., 1984). However, this practice is no longer considered ethical, given the poor outcomes associated with human milk substitute use (Brown et al., 2019).
A decrease in common reasons for preterm infant morbidity was found in three studies (Dicky et al., 2017; Huang et al., 2023; Sun et al., 2019). A significant decrease in BPD (OR = 0.40, 95% CI [0.27, 0.67], p < 0.001) was found by Dickey et al. (2017), who compared NICUs with a feeding policy that supported the use of rMOM versus those with policies requiring pasteurization of MOM for infants < 1500 g. In a prospective cohort study (N = 157), Huang et al. (2023) also found an improvement in BPD for preterm infants fed rMOM (OR = 0.345, 95% CI [0.15, 0.77], p = 0.010). Finally, one study demonstrated an improvement in survival without severe complications in infants fed rMOM (OR = 2.590, 95% CI [1.22, 5.49], p = 0.013; Huang et al., 2023).
Other potential effects related to an rMOM diet were found in the included studies. Cossey et al. (2014) found that preterm infant diet did not affect the frequency, density, or stability of Staphylococci colonization, a common cause of preterm bloodstream infections. Furthermore, de Oliveira et al. (2017) found that the enteral feeding type also did not affect gastric digestion, but pasteurization of MOM increased the proteolysis of lactoferrin (p < 0.01). Finally, Sun et al. (2019) and Huang et al. (2023) found that a diet of rMOM decreased the days of total parenteral nutrition (p = 0.02 and p = 0.045, respectively).
Discussion
This review found some positive growth and health benefits with rMOM versus aMOM including a significant decrease in BPD (Dicky et al., 2017; Huang et al., 2023) and improved anthropometric growth (Andersson et al., 2007; de Halleux et al., 2019; Sun et al., 2019). Additionally, two studies directly demonstrated the feasibility of feeding rMOM in multi-cite and single-site studies, respectively (Huang et al., 2023; Sun et al., 2019). Importantly, there were no negative growth or health effects seen with feeding rMOM versus aMOM.
Nutritional Growth and Development
Benefits of rMOM identified in this review may be attributed to the digestibility of rMOM for preterm infants (Miller et al., 2018) or related to the evidence of 17% higher fat absorption demonstrated with rMOM versus pMOM (Andersson et al., 2007). Promoting preterm infant growth can be challenging, given the myriad obstacles related to immaturity. Despite this, efforts to support preterm infant growth are important as infant growth has been associated with improved long-term cognitive outcomes (Barreault et al., 2019; Ottolini et al., 2020).
Bioactive factors, like lactoferrin, interact with intestinal bacteria to establish a healthy intestinal microbiome, decrease infection and decrease gut inflammation in preterm infants (Thai & Gregory, 2020). Researchers have demonstrated that a diet of rMOM conveys more lactoferrin than aMOM (Paulaviciene et al., 2020). This has the potential to improve preterm infant health by decreasing inflammation and providing immune support (Maffei & Schanler, 2017; Spiegler et al., 2016). Conversely, pasteurization of MOM increases the proteolysis of lactoferrin, which may decrease its absorption and, therefore, its health benefits (de Oliveira et al., 2017).
Several of the statistically significant outcomes attributed to rMOM may not be clinically significant. For example, the birth weight is expected to be regained by 14 days of life. Regaining birth weight at day 6.9 versus day 7.5, a difference of 14.4 hr, may not be clinically significant (Huang et al., 2023).
Non-Nutritional Growth and Development
Dicky et al. (2017) separated rMOM and pMOM groups based on NICU policies and did not provide individual infant diet information, thereby precluding specific conclusions related to the effect of preterm infant diet. The improvements in BPD rates found by Dicky et al. (2017) may have been due to other unmeasured variables in the units supporting the use of rMOM. However, Huang et al. (2023) also found improvements in BPD rates, thus strengthening the possible association between an rMOM diet and decreased BPD rates.
Understanding the effect of rMOM on the most vulnerable preterm infants is limited by the difficulty in capturing the extremely preterm population, as demonstrated by only three studies including extremely preterm infants who were < 27 weeks gestational age at birth (de Halleux et al., 2019; Huang et al., 2023; Sun et al., 2019). Further still, the study by Narayanan et al. included infants up to 37 weeks gestational age.
In two studies, the baseline characteristics differed (de Oliveira et al., 2017; Sun et al., 2019). In the study by de Oliveira et al. (2017) the macronutrients of the rMOM and pMOM differed significantly; however, growth-related conclusions were not drawn in this study. Baseline infant characteristics differed significantly for antenatal betamethasone provided to the rMOM group in the study by Sun et al. (2019) which could have confounded the positive outcomes in the rMOM group.
Implications for Practice
Human milk, specifically MOM, in any form, is the best diet for preterm infants (AAP, 2012). Nursing interventions encouraging mothers to pump and provide human milk, regardless of hospital policy regarding storage or pasteurization, are vital to improving preterm infant outcomes (Hallowell et al., 2016). Nurse support for practices that improve MOM production, like skin-to-skin holding or breastfeeding education, can significantly increase breastfeeding outcomes for preterm infants (Conde-Agudelo & Díaz-Rossello, 2016; Yang et al., 2019). Additionally, policy and best-practices guidance on how to safely prioritize a rMOM diet for preterm infants in the NICU is needed.
Neonatal providers should know that national guidelines support the use of rMOM (AAP, 2012), and rMOM may be beneficial, as demonstrated by the studies in this review. However, all outcomes evaluated in this review were only supported by one or two studies, making the magnitude of any effects difficult to fully understand.
Implications for Research
Further evaluation of the potentially positive effects of feeding rMOM versus aMOM is needed. Given that most NICUs do not routinely pasteurize MOM, studies that compare the effects of rMOM versus fMOM would be of the most importance. Sun et al. (2019) and Huang et al. (2023) demonstrated potential benefits of rMOM for decreasing common preterm morbidities, but studies with larger sample sizes are needed. Sun et al. (2020) has since published a protocol to evaluate the effects of rMOM versus fMOM for preterm infants in a large, multicenter study in China.
In future studies, ensuring that an adequate number of infants less than 25 weeks gestation are included would be helpful to ensure safety at the limits of viability. Furthermore, even though bench science suggests that freezing or pasteurizing MOM may damage its beneficial bioactive components (Aceti et al., 2020; Lawrence, 1999; Paulaviciene et al., 2020; Sheen et al., 2021; Vass et al., 2020), more studies are required to determine whether rMOM is superior to aMOM for preterm infant health outcomes. Moreover, future studies should quantify infant diets by the amount of aMOM, rMOM, or DHM received versus a binary outcome to understand any related outcomes.
Studies evaluating the effects of rMOM on preterm infant outcomes are complex, as mothers often pump in locations outside of the NICU; therefore, practices like pumping at the infant's bedside would be a convenient way to access rMOM. Understanding the feasibility, barriers, and effect on the mother’s breastfeeding experience of bedside pumping would support future studies requiring access to rMOM. Lastly, given that rMOM promotes a healthy preterm microbiome compared with DHM (Hård et al., 2019), evaluating the effect of rMOM versus aMOM on the preterm infant microbiome would further elucidate effects of an rMOM diet.
Limitations
This review was limited by the number and quality of studies that directly compared rMOM versus aMOM and the heterogeneity of outcomes which precluded firm conclusions. All of the studies were completed in Europe (n = 6) or Asia (n = 3), and given the disparateness of the unit policies regarding MOM usage, small sample sizes, and the variety of populations, the results are difficult to generalize. Also, the included study outcomes may have unknown confounding biases that would distort the interpretation of the results. Selection bias may have occurred despite the use of a research librarian search support and two reviewers for the study selection. Finally, excluding studies not available in English may have biased the findings.
Conclusions
Despite national guidelines supporting the use of rMOM for preterm infants (AAP, 2012) and multiple laboratory studies finding benefits of rMOM that could improve the health of preterm infants (Cheng et al., 2021;; Kakulas, 2015; Mazzocchi et al., 2019; Paduraru et al., 2018; Palmeira & Carneiro-Sampaio, 2016; Pannaraj et al., 2017), an rMOM diet is not universally prioritized and requires purposeful implementation by each institution. More studies are needed to directly evaluate the effect of an rMOM versus aMOM diet for preterm infant health and growth outcomes. Overall, the nine studies in this review did not demonstrate harm when feeding rMOM versus aMOM. There is some evidence that a diet of rMOM may support positive health and growth outcomes compared to aMOM, but more research is needed. Finally, caution in generalizing these results is warranted given the sparsity of evidence.
Footnotes
Acknowledgements
We thank Natalie Jackson, PhD, APRN, for contributing time as the second reviewer.
Author Contributions
Disclosures and Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary Material may be found in the “Supplemental material” tab in the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
