Abstract
Background:
Toxic trace elements could enter human milk through mothers’ food consumption, drinking water, air, or incidental soil ingestion, and are of concern to the nursing infant.
Research Aim:
To determine the concentration of toxic trace elements (lead and arsenic) in Peruvian mothers’ milk and their association with blood concentrations in their own infants 3–20 months old.
Method:
This exploratory, cross-sectional study, carried out in Peru, included breastfeeding mother/child dyads (N = 40). Following standardized protocols, biospecimens of human milk and child’s blood were collected.
Results:
Lead and arsenic concentrations in milk were above the method detection limits in 73% and 100% of samples with median concentrations of 0.26 µg/L (IQR = 0.10, 0.33 µg/L) and 0.73 µg/L (IQR = 0.63, 0.91 µg/L), respectively. Concentrations of lead and arsenic in blood were 2.05 µg/dL (SD = 1.35), and 1.43 µg/dl (geometric mean: SD = 1.39), respectively. Blood lead concentrations in 12.5% (n = 5) of the samples were above the U.S. Center for Disease Control and Prevention reference value (< 3.5 µg/dl), and over half of arsenic concentrations were above the acceptable levels of < 1.3 µg/dl (Mayo Clinic Interpretative Handbook). Our results showed that for every one-month increase in age, lead blood concentrations increased by 0.1 µg/dl (p = 0.023). Additionally, every 1 µg/L increase in the mother’s milk arsenic was associated with a 1.40 µg/dl increase in the child’s blood arsenic concentration.
Conclusions:
Implementing effective interventions to decrease the toxic exposure of reproductive-aged women is needed in Peru and worldwide.
Key Messages
Toxic elements are high in Peru’s environment, and the association between the level of toxic elements in human milk and the breastfeeding child’s blood is unknown.
Most of the milk samples collected from Peruvian mothers exceeded the method detection limit for lead and arsenic.
Every 1 µg/L increase in the mother’s milk arsenic was associated with a 1.40 µg/dl increase in her children’s blood arsenic concentrations.
Exclusively breastfed infants 6 months old or younger had the lowest arsenic and lead blood concentrations.
Background
Human milk is crucial for the proper development of the new infant and is recognized as the gold standard for infant nutrition. Some toxic trace elements can enter human milk through food consumption, drinking water, air, or incidental soil ingestion, and are of concern to the nursing infant (Çebi & Şengül, 2022; Olowoyo et al., 2021). Toxic elements can be transferred to milk through maternal blood, even at relatively low concentrations. Lead and arsenic may represent a risk because they are associated with delayed neurodevelopment (Bauer et al., 2020; Pajewska-Szmyt et al., 2019).
In 1989 the World Health Organization and the International Atomic Energy Agency (WHO & IAEA, 1989) defined a baseline for human milk trace element concentrations still largely in use by researchers. Toxic elements in human milk may vary in concentrations depending on the level of contamination in the maternal environment, and they have been studied in developing countries associated with industrial, mining, and agricultural zones (Bansa et al., 2017; Çebi & Şengül, 2022; Mandiá et al., 2021; Motas et al., 2021; Olowoyo et al., 2021; Vahidinia et al., 2019).
Peru, a country in South America, is known for its mines and agricultural areas. In several districts of Peru, drinking water has widespread arsenic concentrations greater than 10 µg/L, which exceeds the WHO provisional guideline (Fano et al., 2020; Fano-Sizgorich, 2021; George et al., 2014; Tapia et al., 2019). Furthermore, soil contamination with lead is extensive in Peruvian mining/agricultural towns (van Geen et al., 2012). To our knowledge, there is no published research on the concentration of lead and arsenic in the human milk of Peruvian mothers. Hence, this study, carried out in Peru, aimed to determine concentrations of lead and arsenic in milk and their association with blood concentrations of lead and arsenic in their breastfeeding infants, aged 3 to 20 months.
Method
Research Design
This exploratory cross-sectional study aimed to determine the concentration of toxic elements (lead and arsenic) in Peruvian mothers’ milk and their association with blood concentrations in their own infants 3 to 20 months old. The Ethical Medical Committee of the University of San Luis Gonzaga-Ica, Peru, granted IRB approval on October 2018 (IRB# 001-2018); additionally, the University of Kentucky approval protocol was IRB#61106.
Setting and Relevant Context
This study was carried out in Peru as part of a Fulbright Scholar program of the Principal Investigator (PI) in the city of Ica, Peru. Peru is known for its high rate of exclusive breastfeeding (68%) compared with other countries in South America (Santos et al., 2022). In Peru, the Ministry of Health certifies Maternity Units in hospitals as Baby-Friendly Hospitals (BFH). It is an initiative to increase breastfeeding (Ministry of Health Peru, 2019). It is also known that 76% of Peruvian women of reproductive age do not have formal work (Instituto Nacional de Estadisca, Peru, 2023), give birth in birthing clinics which are labor/delivery rooms in outpatient clinics, and 65.5% have vaginal births (MINSAL Peru, 2019). The Growth and Development Control Program run by the Peruvian Ministry of Health (CRED is the Spanish acronym), is carried out in all outpatient clinics by nurses or nurse technicians. Care includes an assessment of child growth and development, as well as providing vaccinations (Ministry of Health Peru, 1999). Although Peru is generally known for having drinking water with widespread arsenic level concentrations and agricultural areas with lead contamination (George et al., 2014; van Geen et al., 2012), we were unable to find a report on the specific city where participants were recruited.
Sample
Given the exploratory nature of this study, no a priori power analysis was conducted. The target population was mother–child dyads, and our target sample size was 40 participant dyads. Inclusion criteria were that the mother had a currently breastfeeding child aged 3 to 20 months, who was healthy by the mother’s report at recruitment, and did not have a known congenital disease. Exclusion criteria were an inability to communicate in Spanish or a child with disabilities. This study defined breastfeeding using WHO recommendations for infant feeding. Exclusive breastfeeding included no other foods or liquids other than human milk for the first 6 months, followed by the introduction of complementary food while continuing breastfeeding for up to 2 years of age (WHO, 2021). A sample of 40 biosamples was considered appropriate for testing the levels of trace elements.
Data Collection
Recruitment and data collection were conducted between May and June 2019 from four well-childcare primary healthcare outpatient clinics in Peru. The clinics were at the city’s periphery and served a marginalized and low-income population. All mothers granted informed consent for themselves and their child. Trained Peruvian research assistants interviewed participants in a private room in the clinics that were assigned for this purpose. Samples were collected following standardized protocols previously validated (Linares et al., 2020). Each participant was assigned a numerical code to protect their personal and health information, the study data collected with the survey, and the identification of biosamples. Only research personnel had access to data that was kept in an office in the host institution in Peru. Participants were compensated with $50 Peruvian soles (around $15 US dollars) for their participation.
Measurement
Demographic and Personal Characteristics
The research assistants collected demographic information (one-time point), including questions about family sociodemographic characteristics, environmental and housing conditions, and complementary food in the child’s diet. The survey was developed by the research team by adapting a previously used demographic questionnaire (Linares et al., 2019), and included questions assessing the mother’s age, time living in the area, education completed, work status, income, and health insurance. Reproductive questions included parity, type of delivery for the breastfeeding child, characteristics of that pregnancy, and anthropometric characteristics of the child as a newborn. A set of questions was incorporated from the Peruvian National Household Survey (Instituto Nacional de Estadistica e Informatica [INEI], 2019), which evaluates home conditions, including the number of rooms, construction type, and water and electricity access. Finally, a set of questions was adapted from the Infant Feeding Scale (IFS) to identify food and beverages consumed by the child (Horodynski et al., 2011). No further reliability analyses were completed for demographic questions collected during this study. After completing the survey, each participant’s milk was collected, then researchers performed a venipuncture for blood collection and collected anthropometric measurements on the child.
Trace Elements in Milk
Lead and arsenic were measured in the maternal participant’s milk to compare with reference values reported by WHO and IAEA (1998): lead 2–5 µg/L, and arsenic 0.2–0.6 µg/L. For the milk collection, participants were in a private place with research personnel who explained the procedure. A study pump (Medela® Pump in Style Advanced, Electric Breast Pump) was used to collect the samples. Strict protocols were followed to avoid contamination in collecting milk samples, including the use of gloves when handling samples, new tubes and flanges in each collection, and cleaning the nipple area with sterile gauze and water. These procedures have been validated in a previous study (Linares et al., 2020). No specific time from the last nursing was required to collect 5 ml of milk. The samples were collected between 10 am and 12 pm from one breast/chest in a Bisphenol-A (BPA) and phthalates-free container. Milk samples were transported on ice (0 °C) to a local lab where they were stored at −20 °C until shipment to the United States.
Infant’s Anthropometric Measures
Weight (gm), height (cm), and head circumference (cm) were measured using a calibrated scale, a calibrated stadiometer, and a disposable paper tape measure. Measurements were validated by completing them in duplicate; a process completed by two trained Peruvian research assistants. We used the WHO (2006) child growth standards for infants between 0 to 24 months to calculate age- and sex-specific percentile (U.S. Centers for Disease Control and Prevention [CDC], 2021). Weight/length (W/L) percentile allows for evaluating healthy growth comparison at different ages during recruitment.
Trace Elements in Blood
Lead and arsenic were measured in the participant infant’s blood and compared with reference values: lead < 3.5 µg/dl (U.S. CDC, 2020), and arsenic < 1.3 µg/dl (Mayo Clinic Laboratories, 2020). A certified pediatric nurse, also Peruvian, collected 1 ml of blood following a protocol that has been previously validated (Linares et al., 2021). Blood was collected in a metal-free Vacutainer EDTA tube, Hemogard® (Becton Dickinson, Franklin, NJ, USA). Blood samples were transported on ice to a local lab and were stored at −20 °C until they could be shipped to the United States for analysis.
Transport and Analysis of Biosamples
Biosamples were shipped by a courier company that ensured the appropriate temperature (at least −20 °C) of blood and human milk samples at all stages of the shipping process. Arsenic and lead are chemical elements, so they cannot be degraded, and no volatile metabolites are expected in blood and human milk (Berlin et al., 2005; Unrine et al., 2019). Both blood and human milk can be stored frozen (−20 °C) to avoid degradation of the matrix without affecting total elemental concentrations (Berlin et al., 2005). All samples were shipped to the University of Kentucky and stored in the Clinical Center for Translational Science Laboratory at −80 °C. Frozen samples of blood were transferred to the Environmental Chemistry and Toxicology Laboratory, University of Kentucky, for elemental analysis.
The samples (both whole blood and milk) were microwave digested (CEM MARS 6, Matthews, NC, USA) in ultrapure nitric acid (Aristar Ultra, VWR, Randor, PA, USA), diluted with ultra-pure water, and analyzed to determine trace-element concentrations using inductively coupled plasma mass spectrometry (ICP-MS; Agilent 7900, Santa Clarita, CA, US) based on previously validated methods (Palmer et al., 2006). Quality control procedures included analysis of reagent blanks, initial calibration verification, and analysis of standard reference materials (NIST SRM 955c, Toxic Metals in Caprine Blood, National Institute of Standards and Technology, Gaithersburg, MD, US).
Data Analysis
Descriptive statistics, including means and standard deviations, frequency distributions or medians, and interquartile ranges (IQR), as appropriate, were used to summarize study variables. There were no missing data. Prior to analysis, milk lead levels below the method detection limit (BDL) were substituted for half the method detection limit (MDL for lead = 0.19 µg/L; ½ MDL = 0.095 µg/l). This process was required for approximately 27% (n = 11) of milk lead observations. The distributions of both arsenic and lead were non-normally distributed, so medians and IQR ranges were used to summarize levels descriptively. The distribution of arsenic concentrations in the blood was also not normally distributed; therefore, values were log-transformed prior to regression modeling for this outcome, and geometric means and standard deviations were used to summarize blood arsenic concentrations descriptively. Bivariate associations among study variables and lead blood concentrations or log-transformed arsenic blood concentrations were evaluated using Pearson’s correlation coefficients, one-way analysis of variance (ANOVA), two-sample t-tests or Spearman’s correlation coefficients, as appropriate. Linear regression modeling examined predictors of infant blood concentration for each contaminant (or log-transformed contaminant, as appropriate) separately. Each linear regression model included the respective participant’s milk concentration and any infant, maternal, or environmental characteristics significant in the bivariate analysis. All data analysis was conducted using SAS (Version 9.4), with a significance alpha level of 0.05.
Results
Characteristics of the Sample
The mean age of maternal participants was 28.3 years (SD = 7.5), and slightly less than half were first-time mothers. The mean infant’s age was 11.2 months (SD = 4.4), with a range of 3 to 20 months of age. More than half of the infant participants were female. Participant dyads from four clinics were included in the study. Most infants had a healthy body weight (90%, n = 36) based on height-for-weight percentile (WHO, 2006). Most participants reported having water access at home, and slightly less than half lived in a house cement construction (Table 1).
Characteristics of Participants (N = 40).
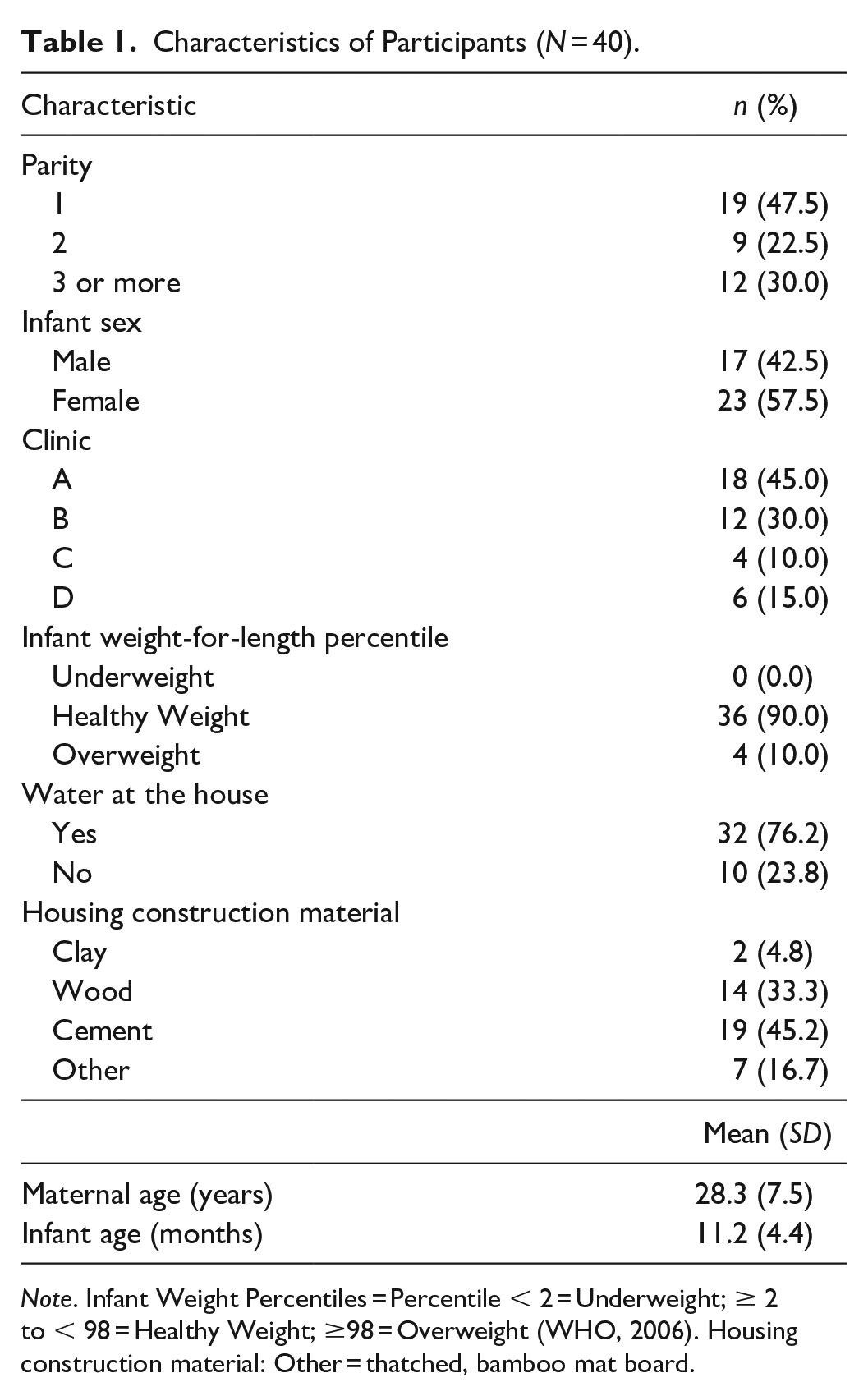
Note. Infant Weight Percentiles = Percentile < 2 = Underweight; ≥ 2 to < 98 = Healthy Weight; ≥98 = Overweight (WHO, 2006). Housing construction material: Other = thatched, bamboo mat board.
Complementary Food to Child’s Diet by Age
All children participants were breastfed in this sample (N = 40), but we divided the infant participants into two groups based on age, with a cut-off point of 6 months to analyze the diet (Table 2). Of the sample, 43% of infants 6 months old or younger were supplemented with formula, and one infant was receiving complementary food; 52% of infants older than 6 months (n = 17) were supplemented with milk besides their mother’s own milk, and most consumed evaporated milk instead of infant formula. There was no difference in juice ingested by child’s age, but a significantly higher proportion of children older than 6 months were eating vegetables (p < 0.001) and beef (p < 0.001) compared with infants 6 months old or younger. Although not meeting the threshold for statistical significance, herbal tea was consumed more commonly in infants older than 6 months (82%, n = 27) compared with their counterparts 6 months old or younger (43%, n = 3, p = 0.052).
Status of the Receipt of Complementary Food by Infant’s Age (N = 40).
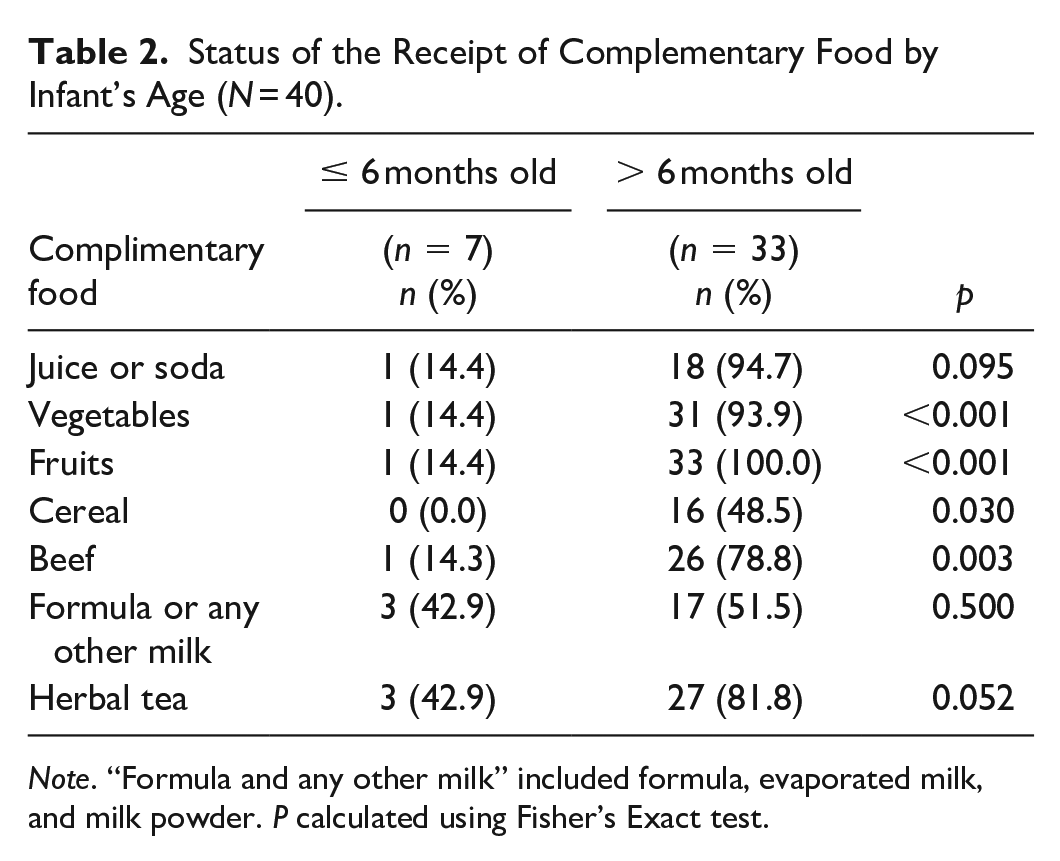
Note. “Formula and any other milk” included formula, evaporated milk, and milk powder.
P calculated using Fisher’s Exact test.
Trace Elements Concentrations in Milk
Lead and arsenic were above the MDL in 70% (n = 28) and 100% (N = 40) of the milk samples, respectively. The median concentration of lead and arsenic among participant’s milk samples were 0.26 µg/L (IQR = 0.10, 0.33 µg/L) and 0.73 µg/L (IQR = 0.63, 0.91 µg/L (Table 3), respectively. We found that only one (2.5%) participant’s milk sample had a lead concentration above the acceptable threshold range provided by the WHO and IAEA (2–5 µg/L); while nearly two-thirds (65%, n = 26) of the arsenic concentrations in the milk were higher than the WHO and IAEA (1989) range recommended. Maternal age was the only characteristic significantly associated with higher concentrations of lead and arsenic in participants’ milk (lead r = 0.40, p = 0.01; arsenic r = 0.31, p = 0.04, results not shown in table).
Participant Lead and Arsenic Concentrations: Infant Blood and Maternal Milk Compared With Reference Values (N = 40).
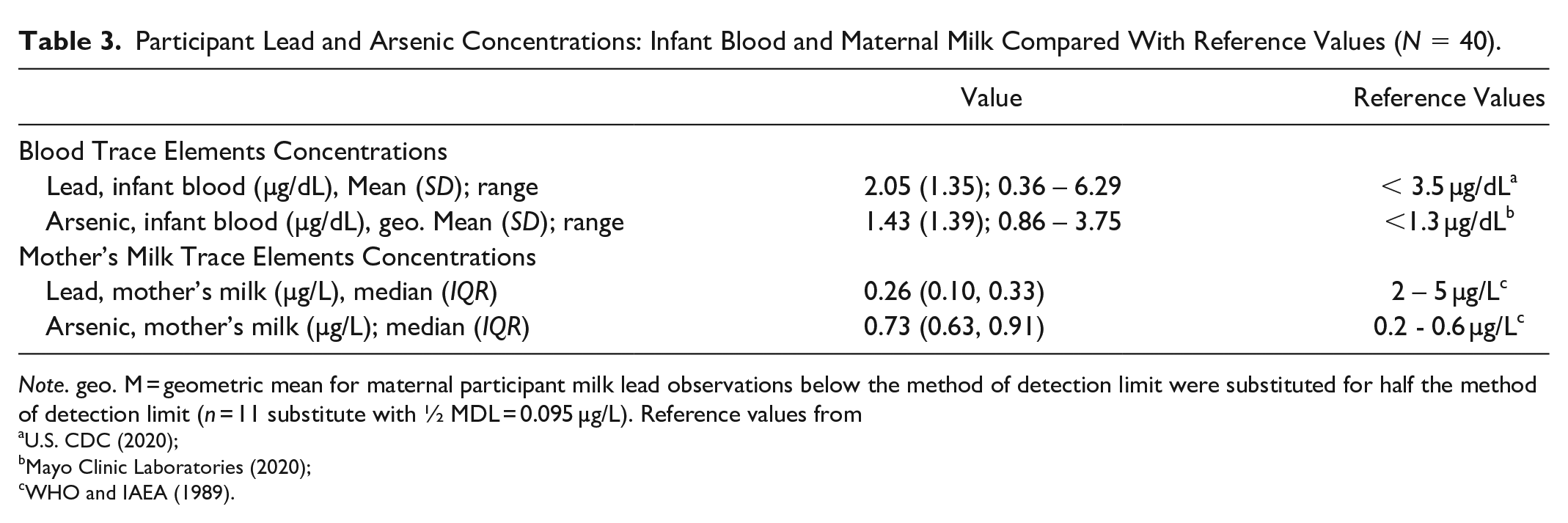
Note. geo. M = geometric mean for maternal participant milk lead observations below the method of detection limit were substituted for half the method of detection limit (n = 11 substitute with ½ MDL = 0.095 µg/L). Reference values from aU.S. CDC (2020);bMayo Clinic Laboratories (2020);cWHO and IAEA (1989).
Trace Elements Concentrations in Blood
Lead and arsenic were above the method detection limit in all infant participants. The average concentration of lead and arsenic in blood were 2.05 µg/dl (SD = 1.35), and 1.43 µg/dl (geometric mean; SD = 1.39), respectively (Table 3); 12.5% (n = 5) of lead concentration in the blood samples were above the U. S. CDC (2020) blood lead reference value (3.5 µg/dl), and over half (55%, n = 22) of arsenic concentration on infant participant’s blood samples were above the acceptable levels of < 1.3 µg/dl (Mayo Clinic Laboratories, 2020). There was a significant, positive correlation between infant participant age and blood concentration of lead (r = 0.37, p = 0.018 (Table 4). Blood lead concentrations were not significantly associated with maternal age, parity, infant weight-for-length percentile, infant sex, clinic location, housing materials, access to water, and lead or arsenic levels in their mother’s own milk. Arsenic blood concentration strongly correlated with arsenic level in the infant participant’s own mother’s milk (rho = 0.53, p < 0.001). Blood arsenic concentration was not associated with maternal age, parity, infant age, infant weight-for-length percentile, infant sex, clinic location, housing materials, or access to water. Maternal participants’ arsenic and lead milk levels were not significantly correlated (rho = 0.15, p = 0.36)
Peruvian Children’s Blood Levels of Lead and Arsenic Associated With Demographic and Environmental Characteristics, and Mother’s Milk Trace Element Concentrations a (N = 40).
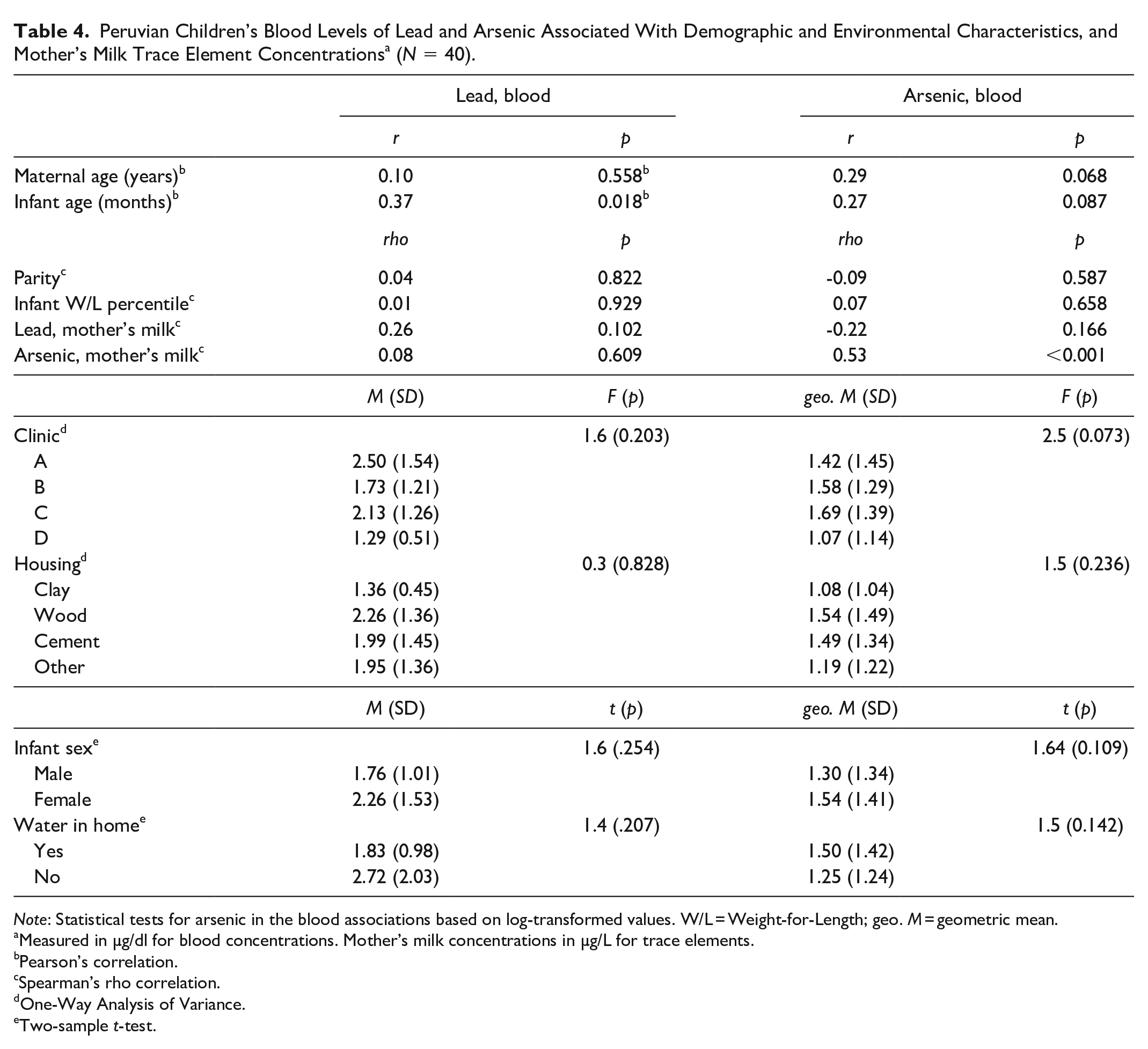
Note: Statistical tests for arsenic in the blood associations based on log-transformed values. W/L = Weight-for-Length; geo. M = geometric mean.
Measured in µg/dl for blood concentrations. Mother’s milk concentrations in µg/L for trace elements.bPearson’s correlation.cSpearman’s rho correlation.dOne-Way Analysis of Variance. eTwo-sample t-test.
We evaluated if infant groups divided by age showed significant differences in lead and arsenic blood levels. Infant participants 6 months old or younger had significantly lower levels of toxic elements than their counterparts older than 6 months (lead = 1.15 vs. 2.24 µg/dl, p = 0.008; and arsenic = 0.11 vs. 0.16 µg/dl, p < 0.001, respectively).
Predictors of Trace Elements in Infant Participant’s Blood
A linear regression model (Table 5) was run with previously significant variables (e.g., infant age and concentration of toxic elements in milk) to evaluate predictors of infant participants’ blood concentration of lead and arsenic, with separate models for each contaminant. The multiple linear regression modeling of lead concentration in infants’ blood was significant overall (F = 3.3, p = 0.047; adjusted R-squared = 0.11). Infant age was significant in this model, with an average expected increase in lead blood concentration of 0.1 µg/dl for every 1-month increase in age (beta = 0.11, SE = 0.05, p = 0.023). Lead concentration in the maternal participants’ milk was not associated with infant blood level in the adjusted model. The linear regression modeling of arsenic in infant participants’ blood was significant overall (F = 20.3, p < 0.001; adjusted R-squared = 0.33), and arsenic concentration in mothers’ milk was a significant predictor (p < 0.001). After transforming the beta coefficient reflecting estimated change related to log-transformed arsenic level within individual breastfeeding dyads, every 1 µg/L increase in arsenic in maternal milk concentration was associated with a 1.40 µg/dl increase in the infant blood arsenic concentration (geometric beta = 1.40, SE = 1.08), on average.
Multiple Linear Regressions Modeling the Association between Concentrations of (1) Lead and (2) Arsenic in Peruvian Children’s Blood and Their Mother’s Milk (N = 40).
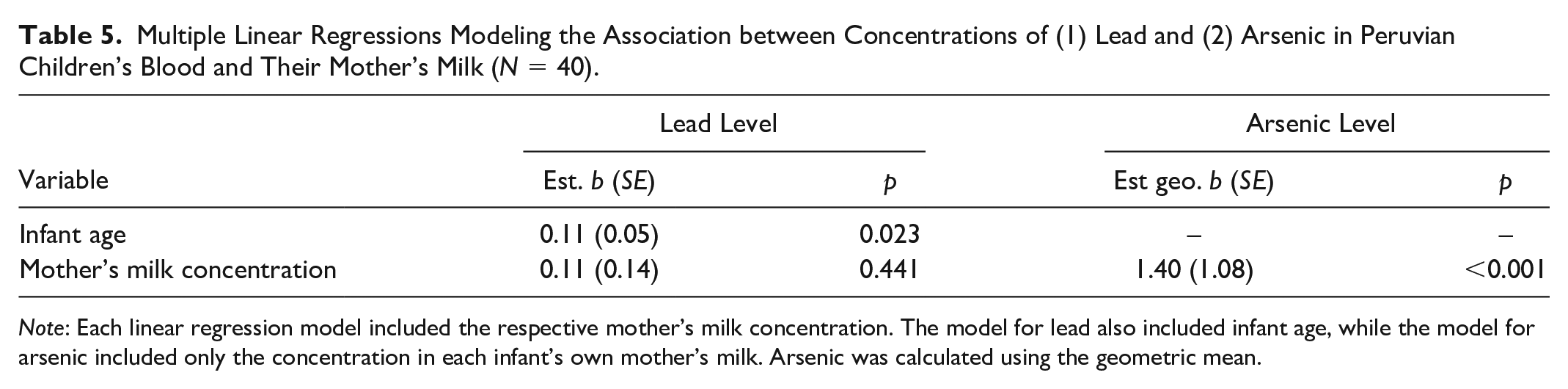
Note: Each linear regression model included the respective mother’s milk concentration. The model for lead also included infant age, while the model for arsenic included only the concentration in each infant’s own mother’s milk. Arsenic was calculated using the geometric mean.
Discussion
Most of the samples of maternal participant’s milk had concentrations above the method detection limits for lead, and all mothers participating had arsenic detected in their milk. However, only one sample of milk was over the threshold range of lead concentrations given by the WHO and IAEA (1989; 2–5 µg/L). The research team was unable to find previous studies in this population to compare the results, since this is the first study in Peru. A few studies in Latin America, especially in Brazil, have reported a variety of lead concentrations in human milk differing by living in an agricultural area (0.26 µg/L; Cardoso et al., 2014), a rural area surrounded by a mine (4.3 µg/L); or an urban area near a mine (12.6 µg/L; Marques et al., 2013). Other researchers have reported lead concentrations in human milk in Argentina of 0.59 µg/L (Klein et al., 2017). It is important to note that even when these levels seem to be relatively low, the mother’s milk is the only source of food recommended for the first 6 months of life of the infant (Meek et al., 2022; WHO, 2021); hence, a small level of contamination is a matter of more significant concern when a variety of foods is consumed in the diet.
Even though Peru is one of the countries in Latin America already identified with elevated water arsenic concentrations (George et al., 2014; Tapia et al., 2019), to our knowledge, this is the first study that evaluated arsenic concentrations in Peruvian mothers’ milk. Our results showed that the median arsenic concentration in this sample was 0.73 µg/L, with 65% (n = 26) of the milk samples over the WHO and IAEA (1989) threshold concentrations (0.2–0.6 µg/L). We only found one study reporting concentrations of arsenic in the human milk of native Andean women exposed to high levels of environmental arsenic. The reported average concentration of arsenic in their milk was 2.3 µg/kg (Concha et al., 1998). Concha et al. (1998) were among the first teams to report concentrations of arsenic in milk, even when maternal arsenic concentrations in blood and urine were high. It was suggested that the mammary gland is capable of avoiding the secretion of arsenic into human milk, providing a protective low exposure to the child (Concha et al., 1998). Moreover, the protective characteristic of mother’s milk was shown in a study in Sweden, where inorganic arsenic—the more toxic form of arsenic compounds—was not detected in a sample of human milk with high concentrations of total arsenic (Björklund et al., 2012). Human milk has been described for its unique health benefits and limited ecological footprint, which are associated with decreased production of waste and the emission of pollutants (Martínez et al., 2019; Zielinska & Hamulka, 2019).
We found that our lactating participant’s age was positively associated with lead and arsenic concentrations in their milk. Several researchers have reported that maternal age is linked to increasing concentrations of some toxic elements in milk, including lead and arsenic. It has been suggested that age influences the deposit of toxic trace elements in the body (Olowoyo et al., 2021; Pajewska-Szmyt et al., 2019).
It is a concern that we found that all infant participants’ blood samples had detectable concentrations of lead and arsenic. In general, the levels of trace elements in this Peruvian sample of infants were typical blood levels. However, we need to be cautious, because high levels of lead in infant blood have been associated with neurodevelopmental effects (Bauer et al., 2020; U.S. CDC, 2020). Arsenic is also associated with neurotoxicity, especially in early-age exposure (Bauer et al., 2020; Olowoyo et al., 2021). Infant age was significantly associated with concentrations of trace elements in the blood. Older infants had higher concentrations of trace elements in their blood, which could be related to the fact that they have already initiated complementary food that could be sources of lead and arsenic. In our sample, all infants older than 6 months were eating complementary food, while infants 6 months old or younger only got their mother’s milk or their mother’s milk supplemented with infant formula. Even though only 12.5% (n = 5) of infant blood samples had concentrations of lead above the limit suggested by the U. S. CDC (2020) (< 3.5 µg/dl), it is essential to note that there is no safe level of lead in the blood, especially in early childhood (Vorvolakos et al., 2016). The final model showed that lead blood concentration increased 0.1 µg/dl for every 1-month increase in age of the participant infants. This finding could support the hypothesis that human milk decreases waste production and the emission of pollutants and protects against pollution in infants younger than 6 months old (Martínez et al., 2019; Zielinska & Hamulka, 2019).
Surprisingly, lead blood concentration in our infant participants was not associated with the levels of lead in their own mother’s milk in this Peruvian sample. This contrasts with other researchers reporting that concentrations of lead in infant blood did significantly correlate with the levels in their own mother’s milk (Ettinger et al., 2004). In the study from Ettinger et al. (2004); however, infants were 1 month old, which varied from our study in which the infant participants were between 3 to 20 months of age at the time the samples were drawn.
In contrast, arsenic concentrations in infants’ blood were over the acceptable levels of < 1.3 µg/dl (Mayo Clinic Laboratories, 2020); in the final model, we showed that these were significantly associated with concentrations of arsenic in each infant’s own mother’s milk. Our results showed that every 1 µg/L increase in arsenic in mother’s milk was associated with 1.40 µg/dl increase her child’s blood concentration.
Contamination of the environment with trace elements and the potential entry into human milk could be unsafe for breastfeeding dyads. In general, researchers have concluded that the benefits of breastfeeding outweigh the potential risks of contamination (Motas et al., 2021; Pajewska-Szmyt et al., 2019; Rebelo & Caldas, 2016). Moreover, researchers have reported that infant formula milk is not exempt from contamination. Contamination can occur in human milk substitutes during the process of fabrication, handling, or manufacturing, with harmful elements including lead, arsenic, lithium, indium, pharmaceutical residues, mycotoxins, or packing product (Martín-Carrasco et al., 2023; Maruszewska et al., 2021).
Limitations
Our study had several limitations that need to be considered. Levels of lead and arsenic in the participant mothers’ blood or other body fluid were not measured, which would have been helpful to compare with levels that we did measure in the milk. The team planned to measure arsenic in nail samples; however, because most mothers worked in agricultural areas, they kept their nails short, making it impossible to get the samples. Additionally, measurements of other potential sources of contamination, including water, soil, cooking ware, or cooking fuel that participants were using, were not collected. Finally, the infants’ ages were diverse, which increased the variability in the relevance of their own mother’s milk as the principal source of food. We recommend caution in the generalizations of these findings.
Conclusion
Arsenic concentrations in our participants’ milk were significantly associated with arsenic concentrations in their infants’ blood while lead was not. Maternal age was the only factor associated with levels of arsenic in milk. Infants 6 months old or younger and only getting their own mother’s milk exclusively had lower levels of lead and arsenic in their blood than infants older than 6 months who already incorporated complementary food. This finding suggests that exclusive breastfeeding was protective for increased levels of trace elements in Peruvian infants younger than 6 months. We suggest a call to health authorities and researchers to continue monitoring levels of contamination in human milk with the goal of implementing effective interventions to decrease exposure to harmful elements in women of reproductive ages. This would maintain the high quality of human milk and support exclusive breastfeeding as the gold standard for infant food globally.
Supplemental Material
sj-docx-1-jhl-10.1177_08903344231212430 – Supplemental material for Concentrations of Lead and Arsenic in Mother’s Milk and Children’s Blood in Peruvian Breastfeeding Dyads
Supplemental material, sj-docx-1-jhl-10.1177_08903344231212430 for Concentrations of Lead and Arsenic in Mother’s Milk and Children’s Blood in Peruvian Breastfeeding Dyads by Ana Maria Linares, Amanda Thaxton-Wiggins and Jason M. Unrine in Journal of Human Lactation
Footnotes
Acknowledgements
The authors acknowledge the assistance of N. Pastor, J. C. Tantalean, and Dr. Castaneda from the University of San Luis Gonzaga-Ica, Peru. The authors also acknowledge the assistance of S. Shrestha from the University of Kentucky Department of Plant and Soil Sciences.
Author Contributions
Disclosures and Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project or publication has different sources of funding:
1. Fulbright Foundation through a Research Award to the first author of this publication.
2. Research Pilot Fund, College of Nursing, University of Kentucky
3. NIH National Center for Advancing Translational Sciences through grant number UL1TR001998. The content is solely the responsibility of the authors and does not necessarily represent the official views of NIH.
4. UK-CARES through Grant P30 ES026529. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIEHS.
Supplemental Material
Supplementary Material may be found in the “Supplemental material” tab in the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
