Abstract
Background:
Mother’s breastmilk is the gold standard for feeding preterm infants. Preterm delivery may be precipitated by inflammatory maternal states, but little is known about milk cytokine profiles and how they correlate with markers of infant gut inflammation (i.e., stool calprotectin) in this vulnerable population.
Research Aim:
To assess cytokines and inflammatory markers in milk from parents of very preterm infants over time as well as correlations between milk and infant’s stool calprotectin.
Method:
This is a secondary analysis of milk samples collected during OptiMoM, a triple-blind randomized clinical trial of infants born < 1250 g (NCT02137473). Longitudinally collected samples were analyzed for cytokines, choline, and inflammatory markers (C-reactive protein [CRP], IFN-γ, IL-10, IL-1β, IL-1ra, IL-6, IL-8, TNF-α). Infant stools were collected for longitudinal calprotectin analysis. Generalized estimating equations quantified longitudinal profiles of milk markers and stool calprotectin, their associations, and the correlation between free choline and C-reactive protein over follow-up.
Result:
Participants included 92 parents and infants (median weeks of gestation 27.3, median birth weight 845 g, and prevalence of male infants 45%). In all, 212 milk samples and 94 corresponding stool calprotectin levels were collected 1–11 weeks postpartum. C-reactive protein was present in much higher concentrations than other markers, and was highest in Week 1 postpartum. It decreased over time. IL-8 and free choline also changed over time while other markers did not. There was no correlation between any milk markers and stool calprotectin.
Conclusion:
Milk from mothers of very preterm infants has detectable inflammatory markers, some of which change over time. Research is needed to determine if infant outcomes are associated with these markers.
Keywords
Key Messages
Preterm birth is associated with maternal inflammation; little is known about cytokines and inflammatory markers in very preterm milk or how they change over time.
C-reactive protein was present in high concentrations and decreased over time. IL-8 and free choline also changed over time; other inflammatory markers did not.
More research is needed to determine potential associations between milk inflammatory markers and infant outcomes in this vulnerable population.
Background
Mother’s own milk (MOM) is the gold standard for feeding very preterm infants, and reducing short-term and life-long morbidities (Miller et al., 2018; Zambruni et al., 2017). MOM is not just food, but a complex milieu of growth factors, stem cells, cytokines, and other bioactives that likely contribute to its ability to improve neonatal outcomes (Perrella et al., 2021; Ruiz et al., 2017). MOM cytokines and their potential effect on the infant are a relatively new area of research. The presence of both pro- and anti-inflammatory cytokines in MOM suggests specific biologic roles that remain to be elucidated, and limited studies suggest a potential association with infant outcomes (Baumgartel et al., 2016). Although limited research has focused on term infants, MOM cytokines appear to change over time and vary by term versus preterm delivery (Baumgartel et al., 2016; Chollet-Hinton et al., 2014; Groer et al., 2014; Ustundag et al., 2005) and by geographic or socioeconomic setting (Ruiz et al., 2017).
There is little published research specific to the MOM cytokine profile of parents who deliver very low birth weight (VLBW, < 1500 g) infants admitted to neonatal intensive care (NICU) settings, for whom maternal risk factors may vary significantly from term infants. Early delivery is common with maternal diagnoses associated with inflammation, such as obesity and pre-eclampsia (Jayaram et al., 2020; Liu et al., 2019), which affect maternal serum cytokine profiles and inflammatory markers such as C-reactive protein (CRP) and tumor necrosis factor alpha (TNF-α) (Catalano & Shankar, 2017; Chen et al., 2017; Gaillard et al., 2016; Kwiatkowski et al., 2016; Liu et al., 2019; Shin et al., 2017), and plausibly could affect the MOM fed to these infants (Baumgartel et al., 2016; Erbağcı et al., 2005; Fujimori et al., 2015).
Studies of MOM CRP are limited to a few studies of term mothers (Fujimori et al., 2015, 2017; Whitaker et al., 2018). In term mothers, elevated maternal serum CRP has been negatively correlated to MOM free choline, a nutrient essential for fetal brain development, which is of concern as MOM from mothers who deliver preterm infants may be already lower in choline (Maas et al., 2016; Ozarda et al., 2014).
The role of MOM cytokines and inflammatory markers is unknown. The very preterm population is at much higher risk of both exposure to prenatal and post-natal inflammation and a resultant risk of neurodevelopmental challenges (Yanni et al., 2017). It is reasonable to infer that MOM cytokines may influence the neonate’s vulnerable gut, although it is unclear if pro-inflammatory MOM cytokines may be harmful, protective, or have no impact (Garofalo, 2010). Stool calprotectin is a marker of gut inflammation, and high levels can correlate with gastrointestinal distress and necrotizing enterocolitis (NEC; MacQueen et al., 2018). Baumgartel et al. (2016) reported stool calprotectin levels in VLBW infants at 3 weeks of life, a peak time for NEC, showing a trend with pro-inflammatory interleukin (IL)-6 in MOM, but studies of associations between MOM inflammatory markers and stool calprotectin are lacking.
We sought to describe longitudinal cytokine and CRP MOM profiles, choosing MOM cytokines previously reported in the limited literature to be associated with maternal inflammation. These included IL-1β, IL-1ra, and IL-8 which have been associated with pre-eclampsia (Erbağcı et al., 2005); IL-6 and TNF-α, which may be associated with maternal body mass index (BMI), pre-eclampsia, and infant adiposity (Baumgartel et al., 2016; Erbağcı et al., 2005; Fujimori et al., 2015; Young et al., 2017; Zambruni et al., 2017); anti-inflammatory IL-10, which may correlate with NEC risk (Baumgartel et al., 2016); interferon (INF)-γ, which may upregulate pro-inflammatory cytokines (Chatterton et al., 2013) and is found in high levels in colostrum (Moles et al., 2015); and CRP, which has been studied in normal and overweight maternal populations (Fujimori et al., 2015, 2017). Finally, we studied free choline, which may negatively correlate with MOM CRP (Ozarda et al., 2014). Adult dietary choline intake is inversely related to serum inflammatory markers, including CRP and TNFα likely through a variety of signaling pathways (Mazidi et al., 2019). Overall, our aim was to describe longitudinal cytokine and CRP MOM profiles in a diverse population of mothers of very preterm infants, as well as to determine if infant stool calprotectin correlated with MOM cytokine and inflammatory marker profiles.
Methods
Research Design
This study is a secondary analysis of a prospective cohort (OptiMoM), a triple-blind randomized clinical trial (NCT02137473; O’Connor et al., 2018). This study was a convenience sample—the secondary use of previously meticulously collected longitudinal MOM samples in a high-risk population has facilitated additional studies with minimal resource utilization. Both the original trial and this secondary analysis were approved by Research Ethics Boards at both hospitals where enrollment occurred.
Setting and Relevant Context
Enrollment in the original study took place in two NICUs in Toronto, Canada, that serve as the primary referral quaternary NICU and one of two level three perinatal centers for a catchment of 6 million people. All hospitals are publicly funded with free access to all legal residents. Toronto is one of the most culturally diverse cities in North America, with a majority of residents born outside of Canada. Some 10% of residents live in poverty. Universal healthcare provides parents with access to lactation support clinics and hotlines, in addition to the lactation support provided in-hospital by full-time International Board Certified Lactation Consultants (IBCLCs) in each NICU, who see all parents of hospitalized infants. Nearly all mothers initiate lactation, and over 80% are lactating at NICU transfer or discharge. Hospital-grade breast pumps are available in the NICU and at low rental cost for home use.
Sample
The OptiMoM study was a randomized controlled trial of human milk-based versus bovine-based fortifier that included 127 infants and their mothers (O’Connor et al., 2018). Infants were eligible if their birth weight was < 1250 g and they were consented for supplemental donor milk. Exclusion criteria included receipt of formula or fortifier prior to enrollment, no feeds within 14 days of birth, or congenital anomalies affecting growth. Measurement of inflammatory markers in MOM to answer the study questions of the present manuscript are reported for the first time. Mothers of multiples were included, with the first infant enrolled in the original study utilized for infant-level data. Of the original OptiMom 127 mother/infant dyads, 92 had frozen MOM samples that met inclusion criteria (Figure 1). Participants whose infants died or who were lost to follow-up during the original study were not re-approached. In these few cases, only data available from the original study were utilized and MOM samples only retested for components that fell within the nutritional realm (free choline).
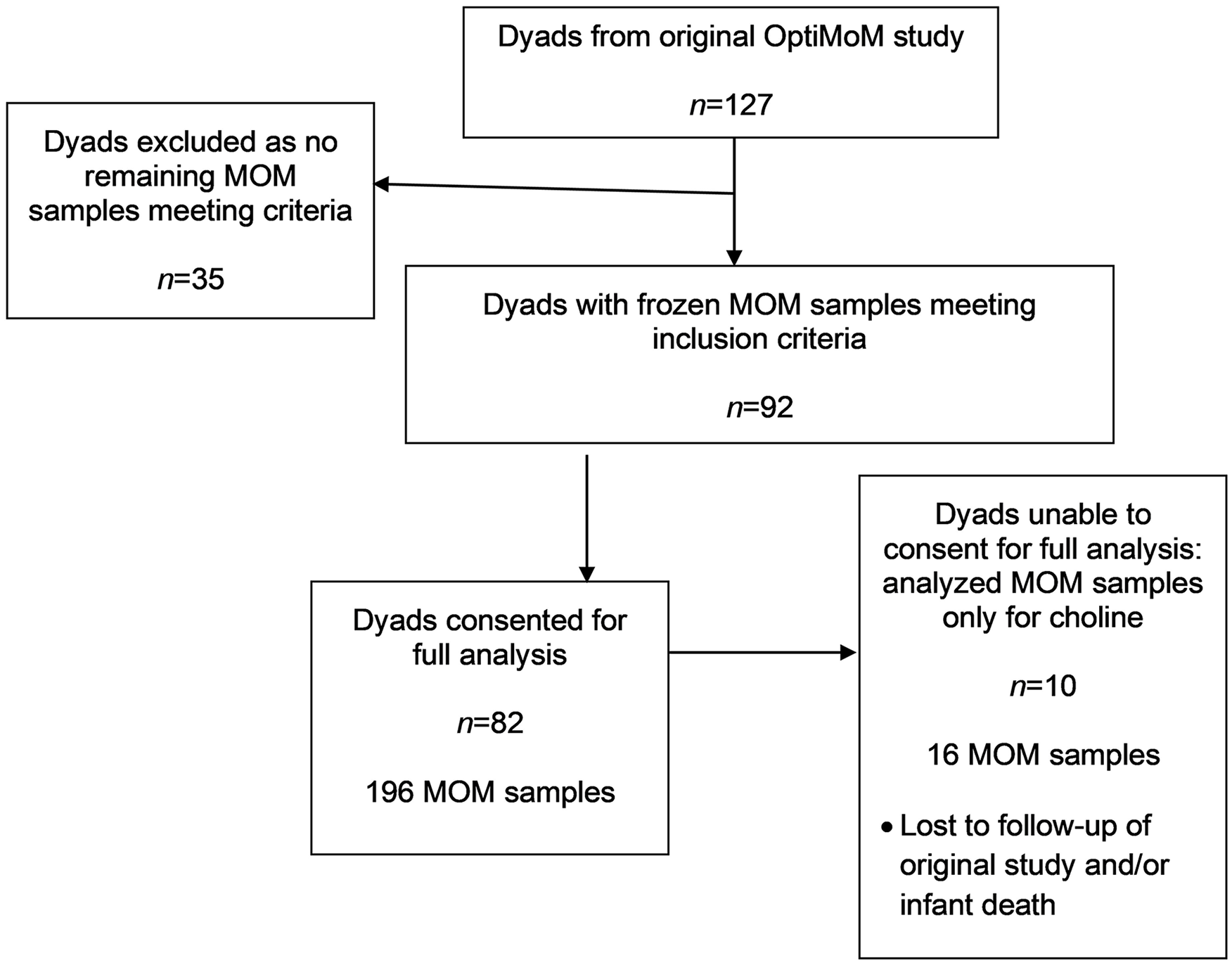
Study Flow Diagram.
There were 268 longitudinal infant stool calprotectin samples available. To study associations between MOM markers and stool calprotectin, MOM samples were matched to a stool sample within a 10-day window, ensuring that MOM samples were collected before the stool. Once matched with MOM samples, we utilized 94 samples between Days 8 and 38 post birth, with 26 samples from Week 1, 32 at week 2, 20 at week 3, and 16 at Week 4. In total, 29 participants contributed two samples and 36 participants contributed one. As this was an exploratory secondary analysis of previously collected data, no power calculations were performed.
Measurement
Participant demographics, clinical maternal characteristics, longitudinal MOM markers and stool calprotectin were summarized. Continuous variables were summarized as medians and interquartile ranges (IQRs). Dichotomous and polychotomous variables were summarized as numbers and proportions of patients. Median and IQRs of MOM markers were grouped together by week postpartum. Inflammatory markers in MOM had varying detection limits and incomplete observations, given that some samples were below detection limits. To reduce variability in MOM collection timing, we chose samples that were ± 2 days from the end of each postpartum week (e.g., postpartum Day 5–9, or postpartum Day 12–16). All milk analyses were performed by the Analytical Facility for Bioactive Molecules, Toronto, Canada in 2019 to 2020. Milk samples were thawed and centrifuged. The skimmed supernatant was separated into aliquots and used in duplicate for all analyses. Magnetic bead assays have been validated and utilized in previous human milk studies (Groer et al., 2014). Therefore, a custom cytokine multiplex bead panel assay (HCYTOMAG-60K-07 human cytokine magnetic kit; Millepore Sigma) was performed for the following markers with noted detection limits in parentheses: IL-1β (1.09 pg/ml), IL-1ra (0.95 pg/ml), IL-6 (1.90 pg/ml), IL-8 (1.69 pg/ml), IL-10 (0.81 pg/ml), IFN-γ (0.66 pg/ml), and TNF-α (1.00 pg/ml). CRP was quantified with a magnetic bead multiplex assay (HNDG2MAG-36K-01 Neurodegenerative MAG Panel 2; Millipore Sigma) using assay buffer. Samples were run at a 1:100 dilution with a detection limit of 0.003 pg/ml. Sample data were processed with Milliplex Analyte (Version 5.1.0.0). Free choline was quantified using a choline colorimetric enzyme assay kit (MAK056; Sigma-Aldrich) with a detection limit of 0.01 nmol/µl. Choline results were multiplied by the molecular weight of choline and converted to pg/ml.
Infant stools were collected weekly during the original study. Calprotectin was measured within 7 days prior to commencing fortification (100 ml/kg enteral intake) and at 7 and 14 days thereafter via ELISA (Eurospital, n.d.; O’Connor et al., 2018); these data were used again in this secondary analysis. Calprest has a published detection range of 15.6 to 500 µg calprotectin/g stool.
Data Collection
For the original OptiMoM study, subjects were recruited between August 2014 and November 2015. For this study, participants previously enrolled in the OptiMoM trial were eligible if they still had frozen longitudinally collected MOM sample(s) available during postpartum Weeks 1, 2, 3, or 4 (immediate postpartum era), and/or Weeks 7–8 or 10–11 (late postpartum era). Samples were taken by study coordinators from MOM that had been stored at −20 °C after pumping and thawed to prepare NICU infant feedings. Once a week, leftover unfortified milk was refrozen at −80 °C and stored for future analyses. Stools had been collected weekly from diapers and stored at −80 °C. In the original study, stools were collected both before and after fortification had started; no differences had previously been seen in stool calprotectin nor in feeding interruptions based on fortifier type (O’Connor et al., 2018).
Parents signed informed consent for the original study for themselves and their infants. As the present study required data from the electronic medical record (EMR) that had not been collected (pre-eclampsia, preterm labor, premature rupture of membranes, diabetes, infant diet at NICU discharge), ethics approval was obtained to re-approach previous participants (ethics approvals #100061228 and #18-0153-E). Eligible participants were re-consented via telephone by a study coordinator from the original study to access their EMR and use frozen MOM samples for testing outside of the original consent of “nutritional components.” No compensation was given. Newly obtained data were added to the original password protected data set and stored in a password protected drive on hospital password protected computers. No identifying data were obtained or stored in the new data set and, to prevent bias, previous blinding was not broken.
Data Analysis
For measurements subject to a detectable range, we reported summary statistics (e.g., weekly medians) by setting incompletely observed measurements that were below lower detection limits to a value half of the lower detection limit. For all subsequent data analysis, we log transformed MOM markers and stool calprotectin to address skewness in distributions. In all analyses except descriptive statistics, left-censored incomplete observations (i.e., measurements below detectable ranges) were imputed 15 times each using censored likelihood multiple imputation (Boss et al., 2019). Parameter estimates and corresponding 95% confidence intervals (CIs) were obtained using Rubin’s rule to combine imputation results (Li et al., 1991; Little & Rubin, 2019). All analyses assumed a significance level of 5% and were performed using R (Version 3.5.3) with the packages of geepack, splines and lodi (R Foundation for Statistical Computing, 2020).
Longitudinal MOM inflammatory markers
To visualize time profiles of MOM markers over time, we used observations before 10 weeks (ranging from 5–54 days of life). For stool calprotectin, we applied the same approach on samples obtained before 7 weeks post-birth (Days 8–38). In this exercise, we modeled the time effect using generalized estimating equations (GEEs) with an identity link function and independent working covariance structure (Prentice & Zhao, 1991). The corresponding 95% CIs were calculated using robust sandwich estimators (Liang & Zeger, 1986).
Associations with stool calprotectin
Next, we assessed associations between MOM markers and stool. Specifically, we modeled the time effect and milk effect on log calprotectin using GEEs with independent working correlation structure. Both time and milk markers were modeled using natural cubic splines. Finally, we quantified associations between free choline and CRP over time. Specifically, we considered time, free choline and their interaction on CRP in GEE models with an identity link function and independent working covariance structure. The time effect was modeled non-linearly with natural cubic splines with 4 degrees of freedom.
Results
Characteristics of the Sample
Participant characteristics are shown in Table 1. Infants were born at a median gestational age of 27.3 (IQR 25.4, 29.2) weeks and with a median birth weight of 845 g (IQR 729, 1030) to participants who were 33 years old (IQR 31, 37). In all, 212 MOM samples were available, with 196 eligible for all analyses with re-consent; the remaining samples were tested only for choline. Figure 2 shows percentages of samples with levels below detection limits for each inflammatory marker. In addition, despite multiple dilutions, two samples (1% of the total) had CRP levels above detection limits, as did one sample (0.5%) for free choline.
Participant Characteristics (N = 92).
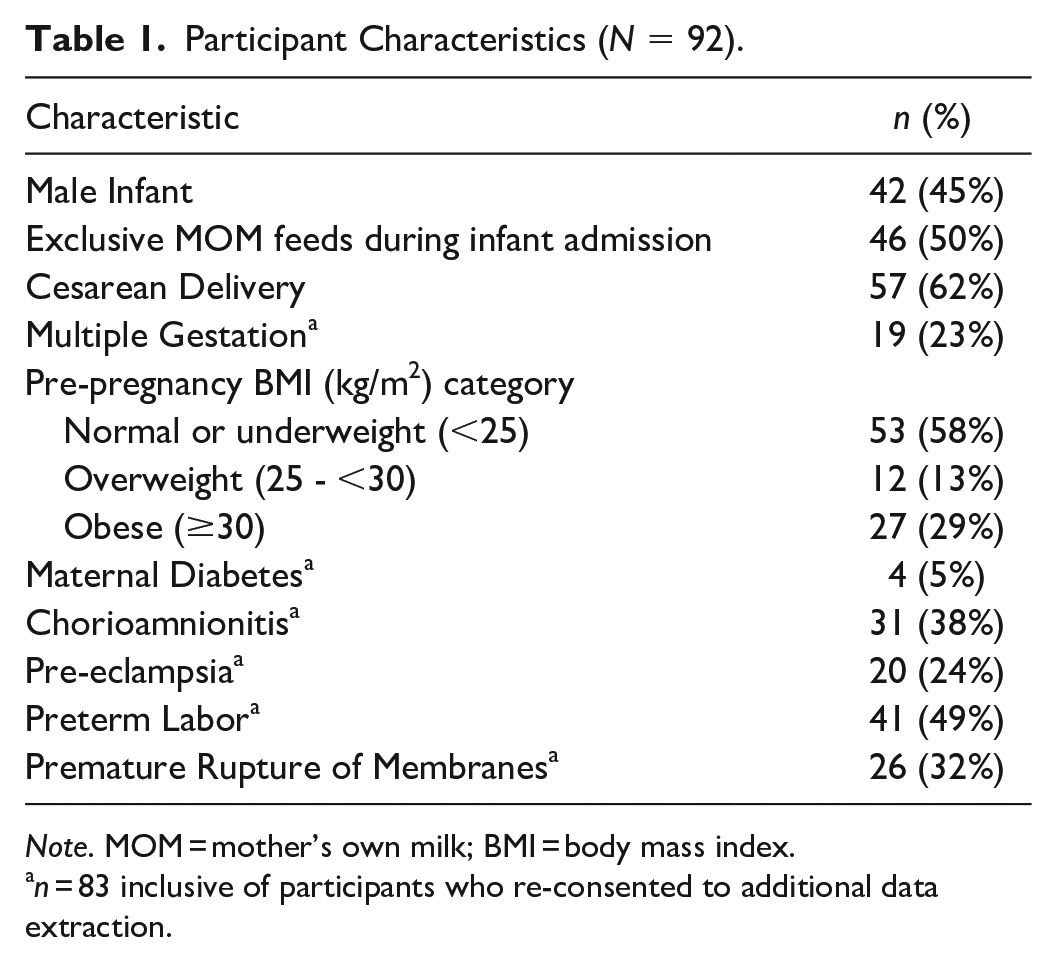
Note. MOM = mother’s own milk; BMI = body mass index.
n = 83 inclusive of participants who re-consented to additional data extraction.
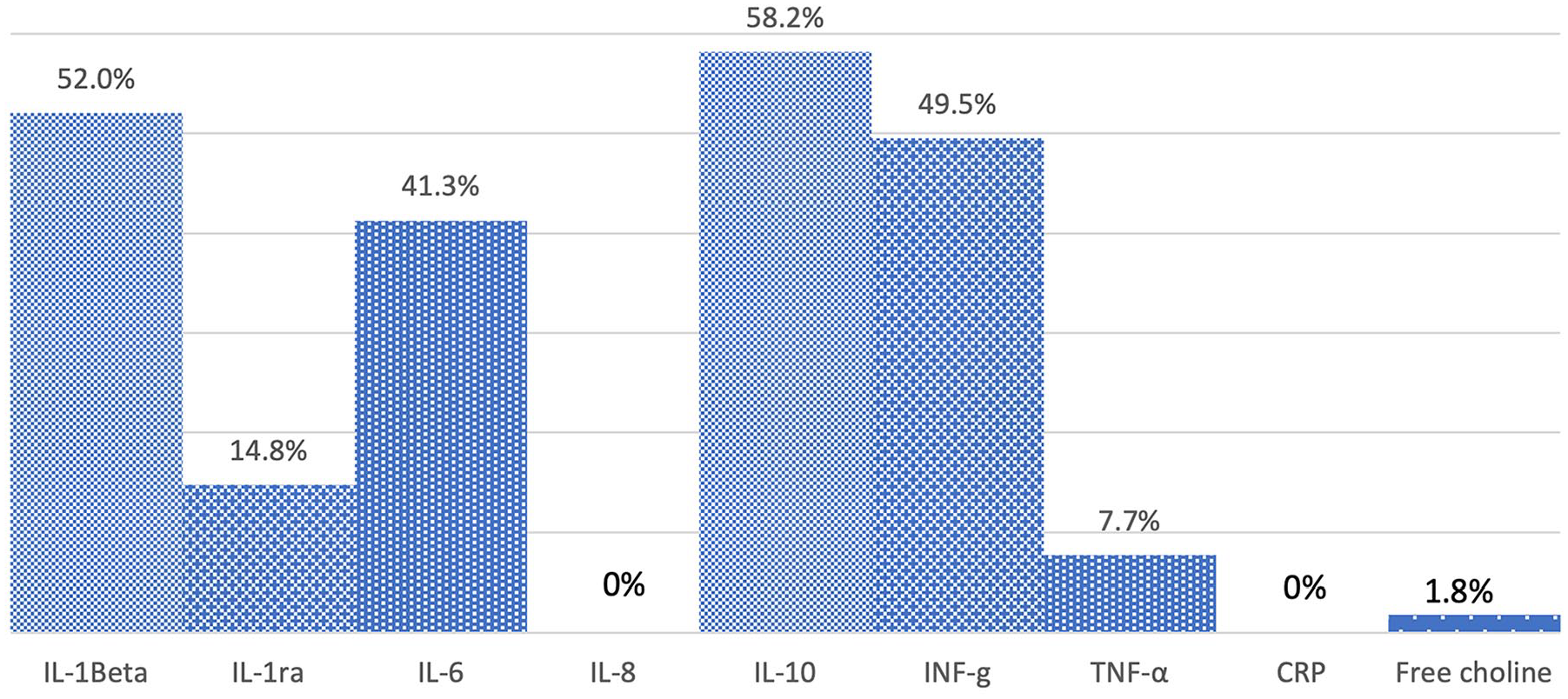
Percentage of Mother’s Own Milk Samples With Values Below Detection Limit.
MOM inflammatory markers
Median values for inflammatory markers which include imputed values for levels below detection limits were summarized longitudinally (Table 2). Estimated time profiles of concentrations are presented in Figure 3 with 95% CIs. At a 5% significance level, our regression results suggested that IL-8, free choline, and CRP concentrations changed over time. Indeed, in Figure 3 we observed decreases in IL-8 concentrations and free choline 1–2 weeks postpartum, then little to no subsequent change. CRP concentrations decreased until 4 weeks postpartum, with similar 4 week and 7–8 week postpartum levels. There were no significant differences in the change of other analytes over time. The regression suggested that the association between free choline and CRP changes over time (p < .001). In a heat map (Figure 4), we observed a stronger inverse association at later milk collection days.
Median Levels of MOM Inflammatory Markers (pg/ml) and Stool Calprotectin (µg/g) by Collection Week (N = 212).
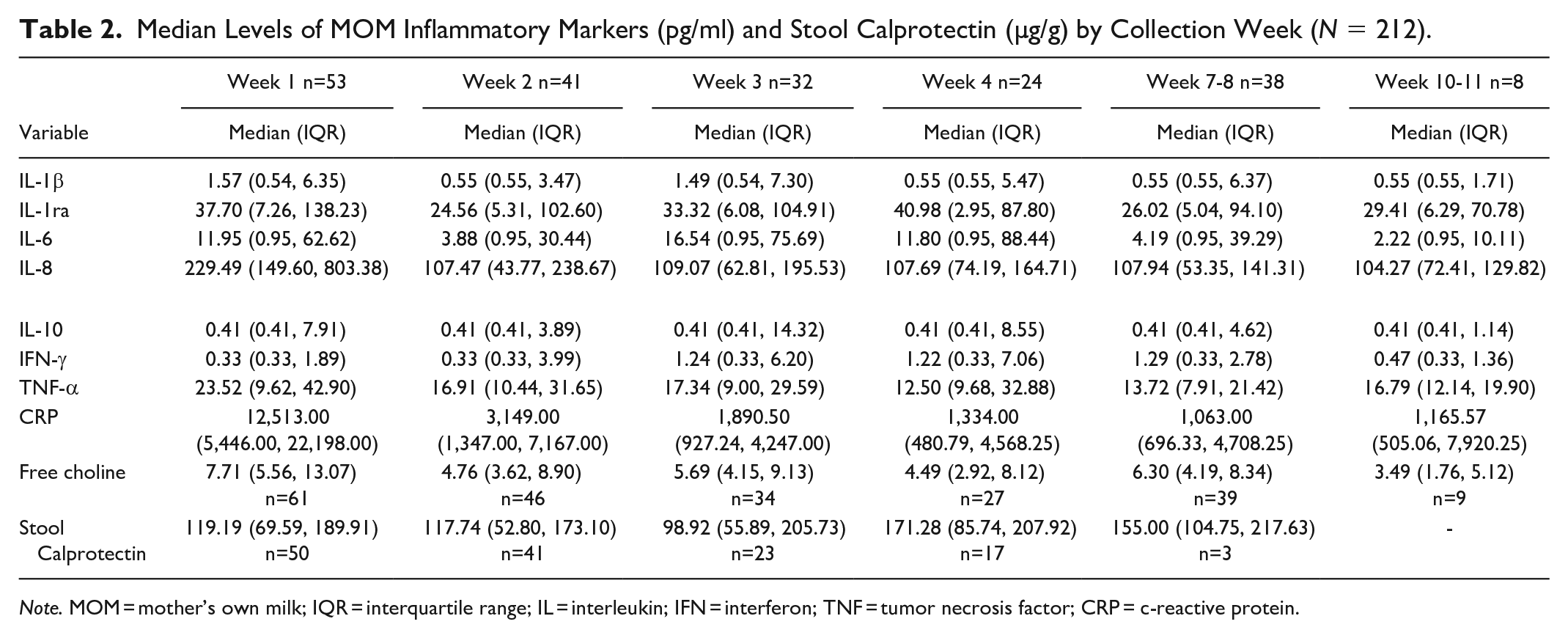
Note. MOM = mother’s own milk; IQR = interquartile range; IL = interleukin; IFN = interferon; TNF = tumor necrosis factor; CRP = c-reactive protein.
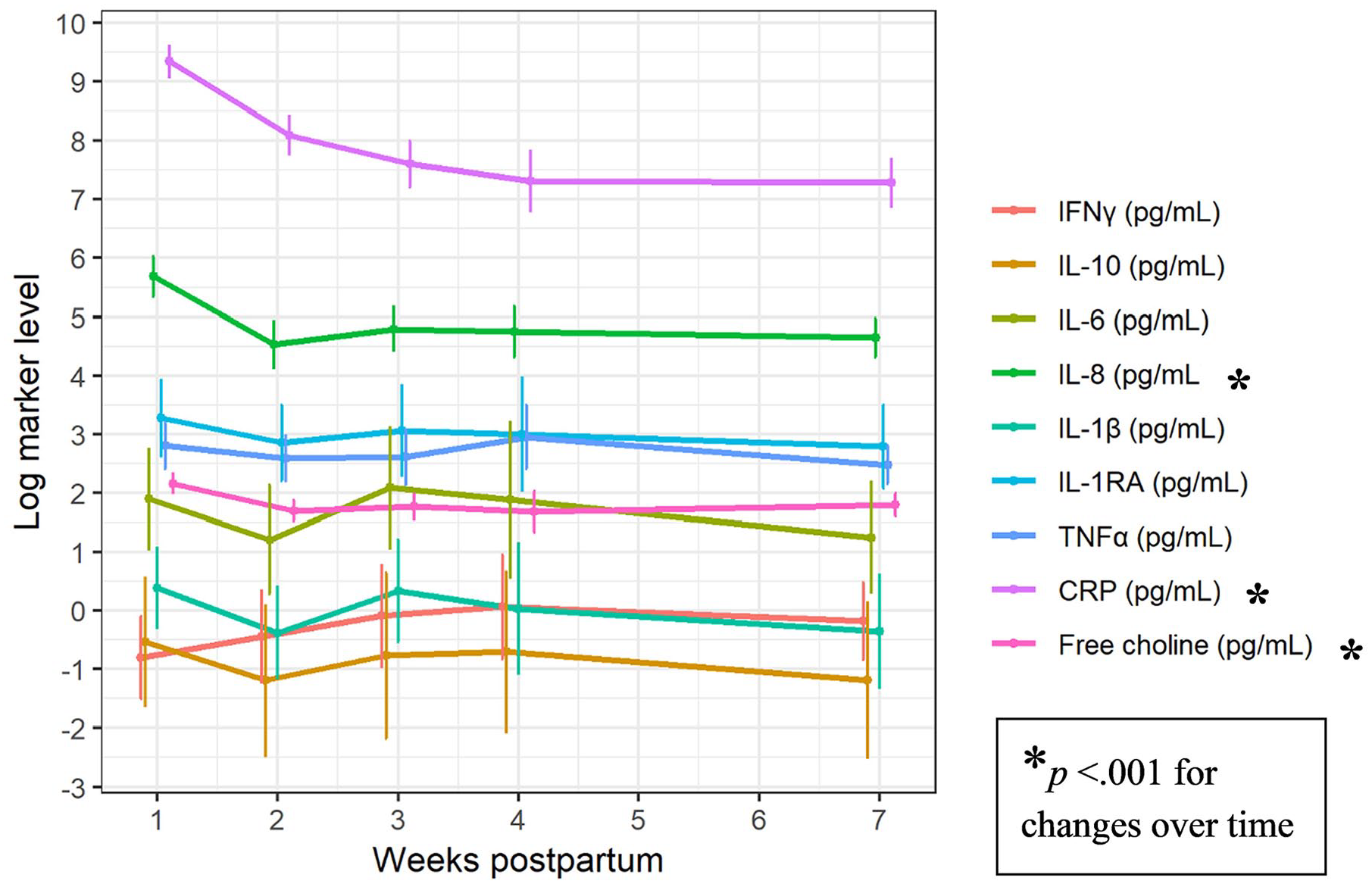
Changes in Mother’s Own Milk Inflammatory Markers Over Time.
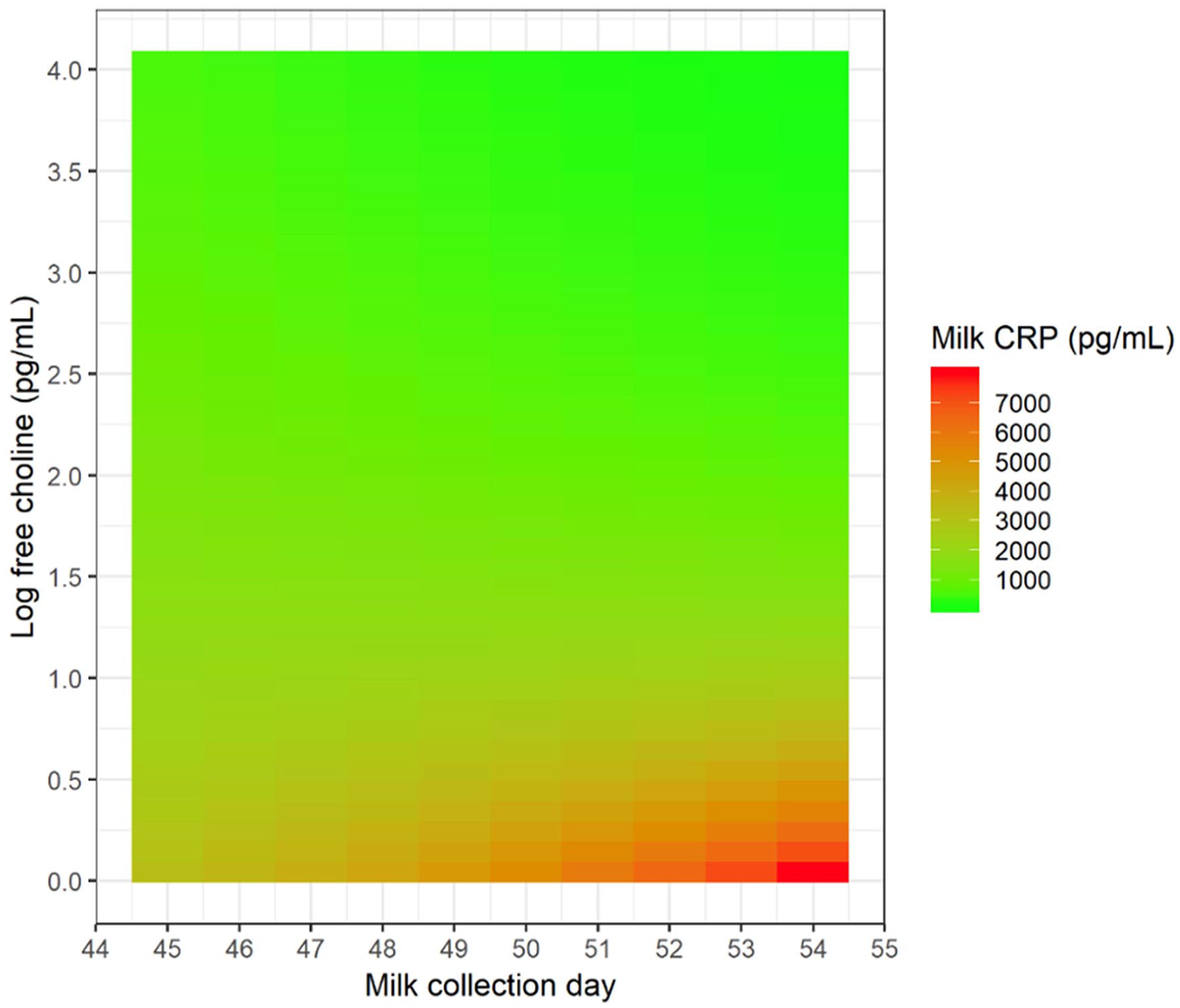
Association Between Free Choline and CRP Changes Over Time.
Stool calprotectin
Median values for stool calprotectin were summarized longitudinally (Table 2). The median lag time between milk and stool samples was 4 (IQR 2, 7) days. These matched stool calprotectin levels over time and are detailed in Table 2. Regression results revealed that calprotectin levels did not significantly change over time (p = 1.0). At a 5% significance level, our regression results suggested no association between stool calprotectin and any MOM inflammatory markers.
Discussion
To our knowledge, this is the largest most comprehensive report of longitudinal MOM inflammatory markers in the VLBW population, and the first to include longitudinal CRP. CRP showed the largest changes over time. Very high levels of CRP in Week 1 plummeted by Week 2 and continued to trend down, but remained about 100 times higher than most other MOM markers. Free choline also showed statistically significant changes over time, with the highest levels in Week 1. Adult dietary choline intake is inversely related to serum CRP (Detopoulou et al., 2008). Previous publications have shown a negative correlation between maternal serum CRP (Ozarda et al., 2014) and mature milk free choline. We found that the association between MOM free choline and CRP changed over time, with the strongest inverse relationship at later collection days. It was somewhat reassuring that the high CRPs in early postpartum days were not as strongly associated with low free choline, a nutrient important for brain development.
IL-8, a pro-inflammatory chemotactic cytokine, was also present in high levels, about 10 times higher than other markers, and also was highest in Week one, a somewhat similar finding to Ustundag et al. (2005), who assessed this in mothers of preterm (but not VLBW) infants, in which IL-8 levels in the 1st week were higher than Week 8 postpartum. We postulate that the peri- and immediate postpartum period is a peak time of maternal inflammation, with high rates of time-limited diagnoses such as surgical delivery (62% of mothers), pre-eclampsia (24%), and chorioamnionitis (38%), whose effects would be expected to quickly lessen. Chollet et al. (2014) reported that MOM cytokines decreased over time in a healthy term population, but CRP was not measured. The potential effect of elevated MOM CRP in the 1st week of life is unclear. This is when VLBW infants are typically started on small volume MOM feeds but are also at their most vulnerable. CRP has potentially antimicrobial properties, but arguably “inflamed milk” could contribute to intergenerational transference of disease risk (Whitaker et al., 2018). The next steps for our research team are to further study potential associations between various maternal “inflammatory diagnoses” such as obesity/overweight and pre-eclampsia and these MOM markers.
Most MOM markers in this longitudinal survey did not show statistically significant changes in the 11 weeks of milk collection, unlike the aforementioned Chollet study (Chollet-Hinton et al., 2014). IL-6 is a pro-inflammatory cytokine that has been associated with NEC but also potentially protective for intraventricular hemorrhage (Baumgartel et al., 2016). Interestingly, although levels did not change in a statistically significant way in the 11 weeks of our study as a whole, they did increase between postpartum Weeks 1–4 (Figure 3), a peak time for NEC (Baumgartel et al., 2016). These results are similar to an increase during the 2nd week postpartum, reported in another preterm cohort (Ustundag et al., 2005). NEC was very rare in our cohort (n = 2). In both of these cases, they had normal stool calprotectin and their mothers had MOM IL-6 levels below the median in the 1st week postpartum, which were the only samples they contributed.
Unlike Baumgartel et al. (2016), we did not find a trend between IL-6 and stool calprotectin (see the Supplemental Figure in the online supplemental material), a marker that is elevated in NEC, nor between any MOM marker and calprotectin, although our sample size was relatively small. It is unclear to what extent inflammatory markers are digested, or how or if they could interact with or affect the infant gut, but it is somewhat reassuring that we do not have direct evidence of a relationship with infant gut inflammation. However, our study, similar to the Baumgartel et al. (2016) cohort, is limited in our ability to study this relationship between MOM components and stool calprotectin, given that only 50% of infants received exclusive MOM feeds, so 50% would have received feeds with pasteurized DM, which likely had a different inflammatory marker profile. Of note, formula was not used when stool was being collected in the original study. Groer (Groer et al., 2014) reported that DM had significantly lower levels of multiple cytokines, including IL-6, compared to MOM from mothers of VLBW infants in the first 5 weeks postpartum. However, as few VLBW infants receive exclusive MOM during their NICU stay, our data is likely representative. A strength of our study was the inclusion of a large number of VLBW infant–mother dyads from a racially and sociodemographically diverse urban NICU with repeated MOM sampling with stool samples from the same time frame.
Limitations
This is a single centre study. Although it was a diverse urban cohort, it may not be representative of all populations. We had decreasing MOM and stool samples over time, so not all mothers were represented at all time points. Given the low number of early milk samples due to infant feeding requirements, we also started our analysis at Day 5, so these data likely represent only transitional and mature milk, not colostrum. Some inflammatory markers, such as TNF-α and IFN-γ, have been reported to have much higher levels in colostrum than mature milk in the extremely low birth weight population (Moles et al., 2015), so our data likely do not represent the full longitudinal experience of MOM fed to these vulnerable infants. In addition, as seen in Figure 1, four of our markers were present in low concentrations in many samples, with more than 40% of the samples having undetectable IL-1beta, IL-6, IL-10, and IFN-γ, using our commercially available cytokine multiplex bead panel assay; similar rates below detection limits were reported for IL-10 and IFN-γ, as well as for IL-1ra, but not for IL-1beta or IL-6, in mature milk from mothers of LBW infants in a low income setting (Zambruni et al., 2017). These findings could be either a limitation of available testing capabilities or an accurate reflection of very low levels in samples, particularly of mature milk (Moles et al., 2015). Further optimization of cytokine analysis for the complex biologic fluid that is MOM would be ideal for future studies.
Conclusion
We have described longitudinal MOM inflammatory markers in a VLBW population, data that are currently lacking in the literature. Further studies are needed to determine associations with specific maternal inflammatory diagnoses, such as obesity, pre-eclampsia, and preterm labor, as well as the potential short- and long-term clinical ramifications of such markers in MOM.
Supplemental Material
sj-tif-1-jhl-10.1177_08903344231192441 – Supplemental material for Inflammatory Markers in Mother’s Own Milk and Infant Stool of Very Low Birthweight Infants
Supplemental material, sj-tif-1-jhl-10.1177_08903344231192441 for Inflammatory Markers in Mother’s Own Milk and Infant Stool of Very Low Birthweight Infants by Rebecca Hoban, Hadar Nir, Emily Somerset, Jordan Lewis, Sharon Unger and Deborah L. O’Connor in Journal of Human Lactation
Footnotes
Authors’ Note
Author Contributions
Disclosures and Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The original study (PIs SU and DOC) was funded by Canadian Institute for Health Research (CIHR NCT02137473). This secondary analysis was funded from research start-up funds for Rebecca Hoban from the Department of Paediatrics of the Hospital for Sick Children.
Ethics/Consent Statement
Consent was obtained for the original study as well as secondary analyses and approved by the REB/IRBs of the Hospital for Sick Children and Mount Sinai Hospital (Toronto, Canada).
Supplemental Material
Supplementary Material may be found in the “Supplemental material” tab in the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
