Abstract
Background
When mothers are confronted with milk supply problems, taking domperidone is regularly suggested. However, domperidone has been associated with sudden cardiac death and caution in prescribing is advised. In 2016, a multidisciplinary group of authors from a tertiary academic hospital (Erasmus MC) published a clinical protocol in a leading Dutch physicians’ journal to support Dutch family physicians in prescribing domperidone to stimulate lactation.
Research aim
To explore consumer and health care provider perspectives and experiences regarding the prescribing of domperidone for lactation insufficiency following publication of a national clinical protocol.
Methods
A cross-sectional qualitative study was performed using semi-structured interviews (N = 40) based on a topic list covering the prescribing process. Participants were mothers (n = 18) who had been advised to try domperidone to boost their milk supply between November 2016 and May 2018, their International Board Certified Lactation Consultants (n = 9), and their family physicians (n = 15). Another group of participants (mothers; n = 6) answered short questionnaires. All interviews were recorded, transcribed and analyzed using ATLAS.ti software. The resulting list of codes was organized according to the topics.
Results
In the process leading to domperidone use to stimulate lactation, participant family physicians relied on the IBCLC, pharmacist, or mother to guide the prescription of domperidone, often citing the published national clinical protocol as back up. The medical safeguards incorporated in the protocol (e.g., taking medical history, physical exam, performing electrocardiograms, limiting dosage) were usually not implemented.
Conclusions
Though the availability of a national clinical protocol in which the prescribing of domperidone for lactation is supported appeared to increase the willingness of participant family physicians to prescribe, gaps were identified between clinical practice and this clinical protocol for prescribing domperidone.
Keywords
Background
It is important to support breastfeeding so that mothers can meet their breastfeeding goals (Chantry et al., 2015), because of the beneficial influences on mothers’ psychological state (Krol et al., 2014; Stuebe et al., 2012) and physiological health (e.g., each year of breastfeeding is associated with a 4.3% decrease in relative risk of breast cancer; Rollins et al., 2016; Victora et al., 2016). Furthermore, feeding human milk substitutes has been associated with negative consequences for infants’ health (e.g., an increased risk for the infant of developing infections, Rollins et al., 2016; Victora et al., 2016). The most frequently cited reason for cessation of breastfeeding in developed countries is “not enough milk,” whether real or perceived (Odom et al., 2013). Treatment of medical breastfeeding problems requires exploration of efforts, risks, and benefits in a shared decision process with due attention to the mother’s preferences for specific outcomes (Truglio-Londrigan & Slyer, 2018).
Milk production consists of three phases; Lactogenesis I, II, and III. Lactogenesis I, in which glandular breast tissue proliferates and lactocytes are activated, occurs from the second half of pregnancy to the first 2 days postpartum. This is followed by Lactogenesis II, the onset of copious milk production, which occurs between Day 3 and Day 8 postpartum and is under endocrinal control, requiring high levels of serum prolactin and low levels of progesterone for milk production, and oxytocin for milk release. Failure of Lactogenesis I and II are together termed primary lactation insufficiency. Causes include pituitary dysfunction, medication, nicotine, obesity, and anatomical and neurological abnormalities (Fu et al., 2015; Ramji et al., 2016).
Next, the production of milk comes under autocrine control, Lactogenesis III. When breasts are not drained sufficiently, for instance due to insufficient frequency of sucking stimulation, feedback inhibitor of lactation (FIL) builds up and blocks protein synthesis and eventually reduces the number of secretory cell surface receptors for prolactin, leading to secondary lactation insufficiency (Neifert, 2001). The prevalence of these problems—primary and secondary together—has been estimated at approximately 15% for primiparous mothers of term infants (Neifert, 2001; Stuebe et al., 2014).
Presumably, many cases of secondary lactation insufficiency can be prevented or solved by non-pharmacological interventions. Treatment of lactation insufficiency should therefore start with optimization of breastfeeding management before turning to medications that augment lactation, known as galactagogues (Grzeskowiak & Amir, 2014; Grzeskowiak et al., 2018). The most widely studied galactagogue is domperidone, favored because of its relatively satisfactory safety profile for both mother and child (Cuppers-Maarschalkerweerd et al., 2017; Grzeskowiak & Amir, 2014). Domperidone is assumed to work by enhancing serum prolactin levels (Drugs and Lactation Database, 2018; Hale & Rowe, 2018).
It is of note that both the efficacy and safety of domperidone to stimulate lactation have been questioned. For instance, an often quoted review concluding that domperidone results in an increase in daily milk production of 74.72% (95% CI [54.57, 94.86]) depends largely on a trial criticized for its lack of randomization and blinding, as well as for skewing effects due to the initial very low milk output of three out of the seven participants receiving a placebo (Drugs and Lactation Database, 2018; Grzeskowiak et al., 2018; Hale & Rowe, 2018; Paul et al., 2015). In studies in which volume is employed as an indicator, the possibly limited efficacy for term infants becomes clear. For example, Grzeskowiak et al. (2018) conclude that domperidone use resulted in a moderate short-term increase in expressed milk volume, among participants who birthed preterm infants, of 88.3 ml/day (95% CI [56.8, 119.8]), which represents almost 40% of total daily milk intake for a preterm infant weighing 1.5 kg receiving enteral feeds of 150 ml/kg/day. By contrast, milk adequacy for a term infant has been defined as ≥ 500 ml/day by Week 6 (Hill et al., 2005).
Key Messages
It is unclear how consumer and healthcare provider perspectives and experiences regarding the prescribing of domperidone for lactation insufficiency were affected following publication of a national clinical protocol in the Netherlands.
The availability of a national clinical protocol supporting the prescribing of domperidone for lactation appeared to increase the willingness of doctors to prescribe.
Gaps were identified between clinical practice and this clinical protocol for prescribing domperidone.
Publishing an evidence-based protocol for the off-label prescription of domperidone in a medical journal is no guarantee for implementation of safety measures in the doctor’s office.
With respect to safety considerations, a retrospective, population-based cohort study was performed in which at least one in five participants filled a minimum of one prescription for domperidone in the 6 months postpartum. In total, 21 cases of ventricular arrhythmia were identified, indicating that these events are very rare. Of these 21, 16 participants had a prior history of ventricular arrhythmia, including the six participants who were taking domperidone. Due to the small number of events, the hazard ratio for those exposed to domperidone was reported as not statistically significant (HR = 2.25, 95% CI [0.84, 6.01]). For participants with BMI > 25, the reported hazard ratio was 3.22 (95% CI [1.00, 10.4]) (Smolina et al., 2016).
The other literature pertaining to the safety of domperidone for lactation is limited to case studies. One concerns a mother who was hospitalized after taking domperidone for lactation and was discovered to have congenital long QT syndrome. (Blusztein et al., 2017). Researchers of four case studies have reported withdrawal symptoms after extended use for lactation, including insomnia, anxiety, and tachycardia (90–160 mg daily for 14 weeks, 80 mg daily for 8 months, 80 mg daily for 9 months, 30 mg daily for 10 months). These cases required very slow tapering to resolve symptoms (Doyle & Grossman, 2018; Manzouri & Mink, 2017; Papastergiou et al., 2013; Seeman, 2015).
Domperidone was developed and is registered in the Netherlands and in many other countries as an anti-emetic; its use as lactation stimulant is off-label (Passier, 2014). By contrast, domperidone is not approved for any indication in the United States, due to safety issues (Osadchy et al., 2012). Sale of domperidone was available over the counter at Dutch drugstores before 2009. In that year sale was limited to pharmacies due to the indications of an association between domperidone, QT-prolongation, and cardiac adverse events. However, a prescription was not yet compulsory at this time (Michaud & Turgeon, 2013). Since July 2014, based on further evidence, dispensing domperidone in the Netherlands has required a prescription by a physician (College ter Beoordeling van Geneesmiddelen [Medicines Evaluation Board], 2014; Pharmacovigilance Risk Assessment Committee, 2014). As a result of both changing regulations and news coverage concerning domperidone, Dutch physicians, since, have been prescribing domperidone less often (Berger et al., 2017).
In response to the denied requests, particularly of mothers of preterm infants experiencing dwindling milk production while pumping to meet their hospitalized child’s demands, a multidisciplinary group of authors (Van Paassen et al., 2016) working at a Dutch university medical center published an article to support Dutch family physicians in prescribing domperidone to stimulate lactation in November 2016 in the Nederlands Tijdschrift voor Geneeskunde (NTvG [Dutch Journal for Medicine]). The authors of the NTvG article argued in favor of using domperidone to stimulate lactation for an extended duration, advising dosages starting at 10 mg 3 times a day, going up to 20 mg 3 times a day if use of the lower dosage proved insufficient (Van Paassen et al., 2016). The authors advised performing an electrocardiogram (ECG) after 2 weeks of use to rule out untoward consequences of employing domperidone on heart rhythm and provided a step-by-step plan (Figure 1). In this study, the Dutch NTvG article was considered to be a national clinical protocol in order to expedite exploration of perspectives and experiences in relation to this protocol since official national guidelines are lacking in the Netherlands.
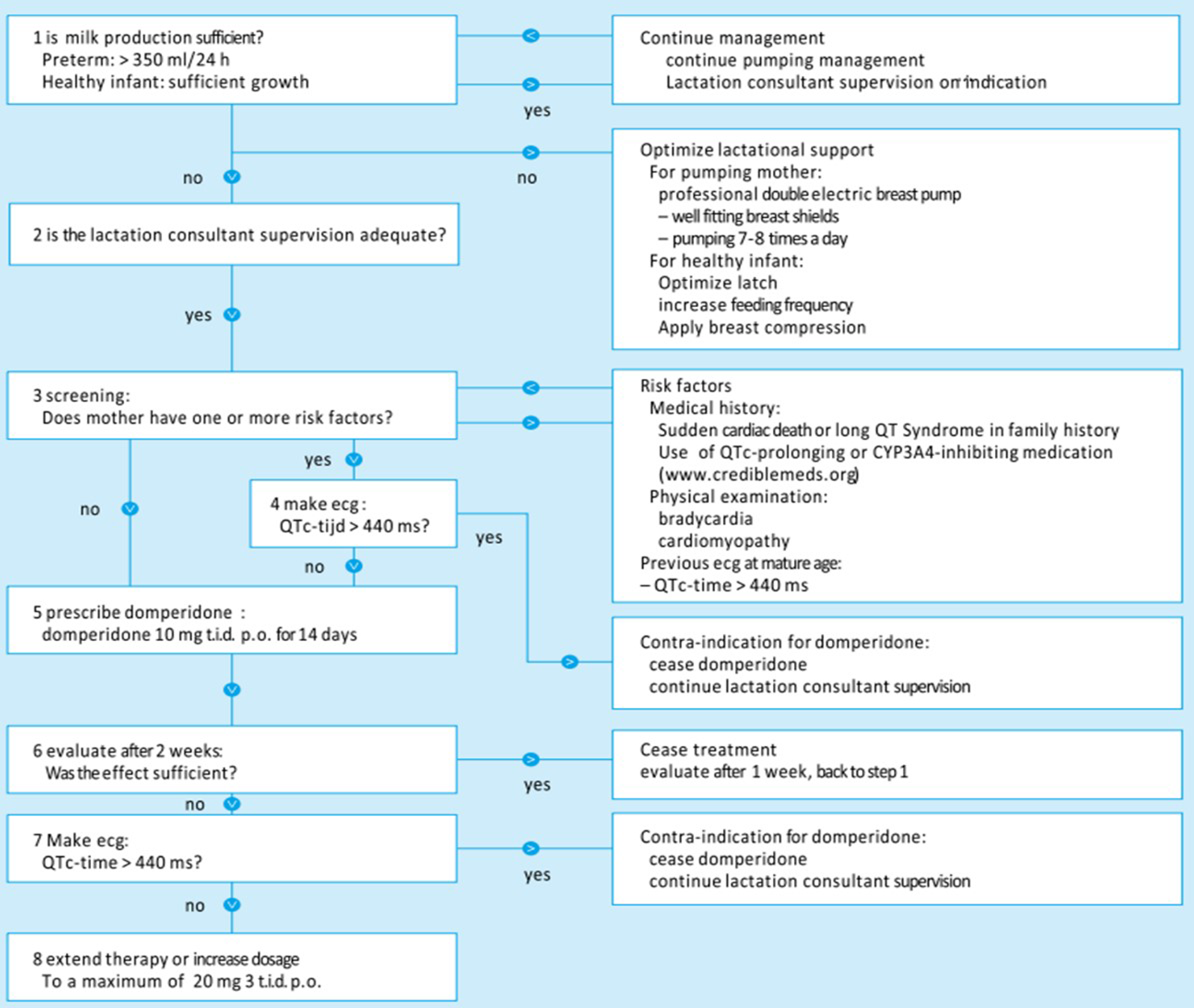
Steps in Case of Insufficient Milk Production. Note. Original in Dutch (Van Paassen et al., 2016).
It is unclear how consumer and health care provider perspectives and experiences regarding the prescribing of domperidone for lactation insufficiency were affected following publication of this national clinical protocol. Our study aim was to explore the consumer and health care provider perspectives and experiences with respect to domperidone prescription for lactation insufficiency.
Methods
Design
This was a retrospective, cross-sectional qualitative study. The design enabled us to explore and understand the perceptions of consumer and health care providers regarding the prescribing process of domperidone for lactation insufficiency (Colorafi & Evans, 2016). The study was given a waiver by the Medical-Ethical Evaluation Committee of Erasmus Medical Center in Rotterdam as, in accordance with the Dutch Medical Trial Act, it does not compromise patient integrity.
Setting
Participants were recruited through a Dutch website designed for this purpose (www.meermelkonderzoek.nl) in January 2018 and advertised through Dutch social media. The Netherlands is a small country with a well-developed health infrastructure. Dutch government is responsible for the accessibility and quality of the health care system. People with low incomes receive compensation to help them pay their private insurance fees. For costs accrued in hospitals and for the use of medication, there is a mandatory annual total deductible of €385 (US$401). Insurance companies provide a package with a government-defined set of insured treatments, to date not including specialized lactation support. Some lactation support is provided by maternity nurses in the first 8 days after birth and by midwives and gynecologists during pregnancy and the first 6 weeks postpartum, although their training in this area is limited. Many mothers opt for additional health insurance packages, which may cover some lactation support by IBCLCs. Services provided by the family physician are not included in the mandatory deductible of €385, so there is no financial barrier to visiting first line physician health care.
There are about 400 IBCLCs in the Netherlands. Of the 120 hospitals, most offer support by an IBCLC for in-hospital mothers and infants on certain days of the week. IBCLCs working at Dutch hospitals may combine their institutional work with private lactation practices. In addition, all Dutch municipalities offer support to parents through Centers for Youth and Family, some of which employ IBCLCs who offer limited services for free.
The majority of Dutch mothers work part-time, on average 26.6 hr a week. They are entitled to 16 weeks of maternity leave, which should start at the latest 4 weeks before the term date. According to the latest report, 80% of Dutch mothers initiate breastfeeding after birth. Among infants, 57% still were exclusively breastfed after 1 month and 47% after 3 months (Peeters et al., 2015).
Sample
The target population consisted of breastfeeding mothers who had asked for, or been advised to use domperidone for lactation insufficiency, and their IBCLCs and family physicians. Mothers were eligible to participate if they had been advised to use domperidone for lactation insufficiency in the period after the publication of the national clinical protocol in the NTvG article (November 9, 2016). If a mother had not been advised to use domperidone, or had asked for domperidone before the national clinical protocol was published, she was excluded from the study.
Via the website, 32 mothers applied for an interview. Two other mothers were approached after an interview with their IBCLC. Selection of participants (mothers) was based on convenience, starting at the top of the list and proceeding to the next name if the phone call was not answered. Two mothers who had not given birth themselves, did meet the inclusion criteria since they had been advised to use domperidone to develop lactation after a surrogacy birth. After the first 16 interviews with participants (mothers) had been conducted and analyzed, a short email questionnaire was sent to all the mothers who had signed up through the website and who had not yet been interviewed to check data saturation. This questionnaire was returned by 10 participants (mothers). Based on their answers, two more participants (mothers) were interviewed, making a total of n = 18, achieving data saturation.
Through the website, 11 IBCLCs applied directly, four of them together with one of their clients. Following the first interviews, it was decided to select participants (IBCLCs) based on client cases; the participants (mothers) were given case numbers (M1, M2, etc.), with corresponding labels for their IBCLCs (L1, L2, etc.). Interviews were conducted with eight of these participants (IBCLCs), after which data-saturation was achieved.
After the interviews with the participants (mothers), interviews were conducted with 13 of their family physicians (D1, D2, etc.). Two family physicians, although declining to be interviewed, filled out questionnaires containing the same questions as used for the interviews; these were included in the data analysis. Two family physicians refused to participate. One participant (mother) did not ask her physician for domperidone. Analysis of the interviews and questionnaire responses combined with the data obtained from the participants (mothers) and their IBCLCs suggested that data saturation was also reached with respect to the family physicians (Figure 2).
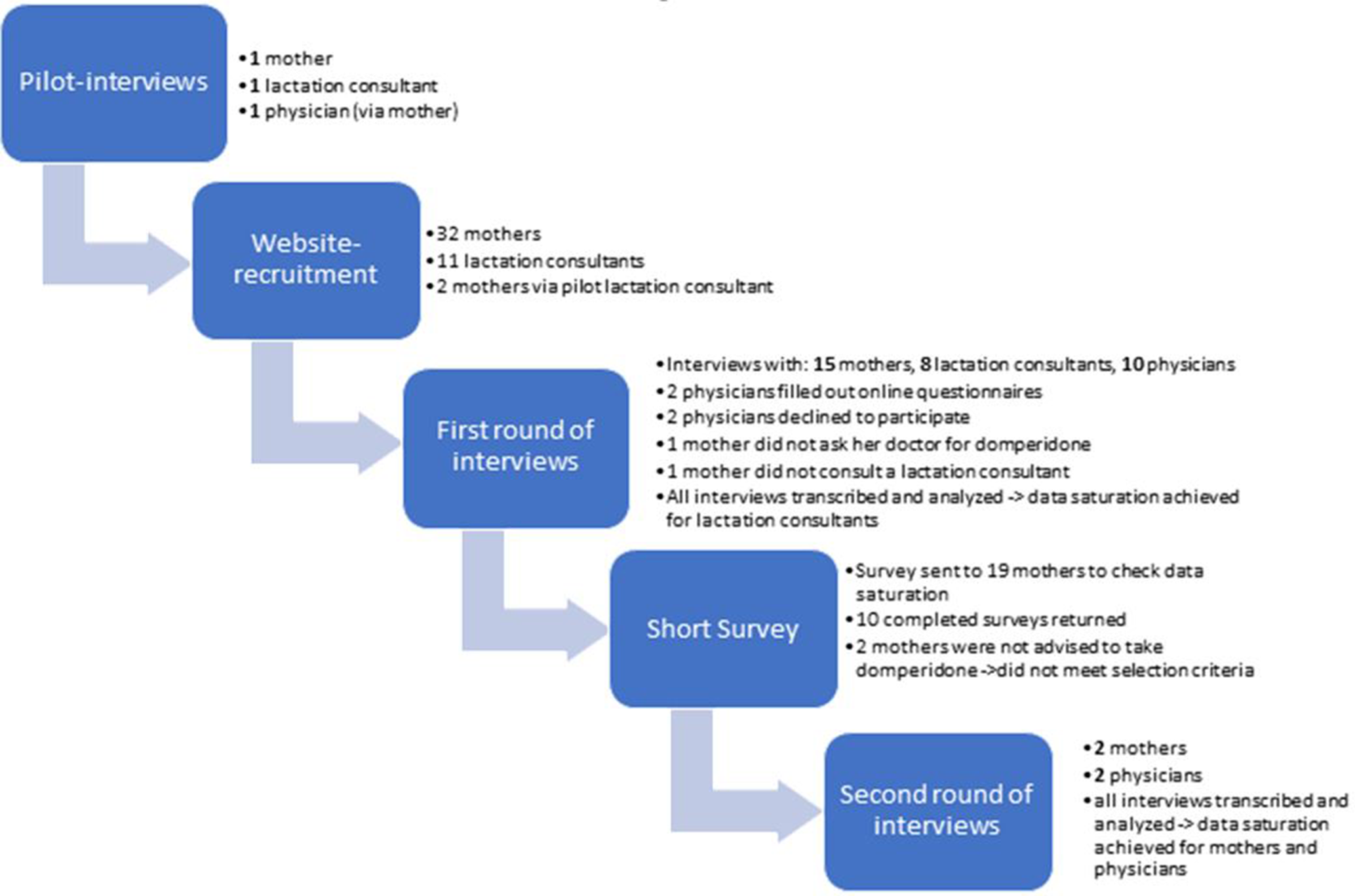
Flow Chart of Participant Selection.
We interviewed a total of 40 participants: mothers who had been advised to try domperidone to boost their milk supply between November 2016 and May 2018 (n = 18), their IBCLCs (n = 9), and their family physicians (n = 13). Two additional participants (family physicians) answered a questionnaire based on the original topic list (n = 2). Another group of participants (mothers; n = 6) answered short questionnaires.
Data Collection
Data were collected between November 2016 and May 2018. Participants (mothers and IBCLCs) were recruited through a website containing information about the project (see Figure 2). Family physicians and IBCLCs were approached after the interviews with the mothers. Informed consent of all participants was requested at the start of each interview and mothers were asked to sign forms giving consent for interviews with their IBCLC and family physician, which were shared with the involved health care providers.
The interviews, performed by the primary researcher, were semi-structured, using topic lists developed to explore the experiences of the participants (see Table 1). The topics were chosen to cover the full prescribing process, from a mothers’ request for help concerning milk supply problems, to non-pharmacological interventions to resolve these problems, to galactagogues in general and domperidone in particular, including dosing, therapy duration, and the NTvG article on domperidone to stimulate lactation. The topics were identical for all three groups but worded to fit the participant’s role in the prescribing process. All interviews were recorded and transcribed verbatim. Personal information about the participants was left out of the transcripts and stored in a separate, secured location.
Topic List Used for Participant Interviews.

Note. NTvG = Nederlands Tijdschrift voor Geneeskunde [Dutch Journal for Medicine] (Van Paassen et al., 2016). ECG = electrocardiogram.
Data Analysis
We performed a qualitative thematic analysis to explore consumer and health care provider perspectives and experiences regarding the prescribing of domperidone for lactation insufficiency. At the start of each interview with a mother participant, demographic data were collected including age and profession. Descriptive statistics were used to analyze these data. Transcripts were analyzed with ATLAS.ti Version 8 (ATLAS.ti Scientific Software Development GmbH, Berlin, Germany), using an iterative coding process. An initial coding scheme was developed by the first author (LTB). The resulting list of codes was organized according to the topics, leading to agreement on existing themes (see Table 2). Based on recurring themes, and supported by additional questionnaires filled out by six participants (mothers) who had signed up but had not yet been interviewed, we determined that data saturation had been attained for the three participant groups (mothers, IBCLCs, and doctors) and data collection was ceased.
Data Analysis Structure Table.

Results
Characteristics of the Sample
The mothers ranged in age from 27–44, with an average age of 33; other characteristics can be found in Table 3.
Characteristics of Participating Mothers (N = 18).
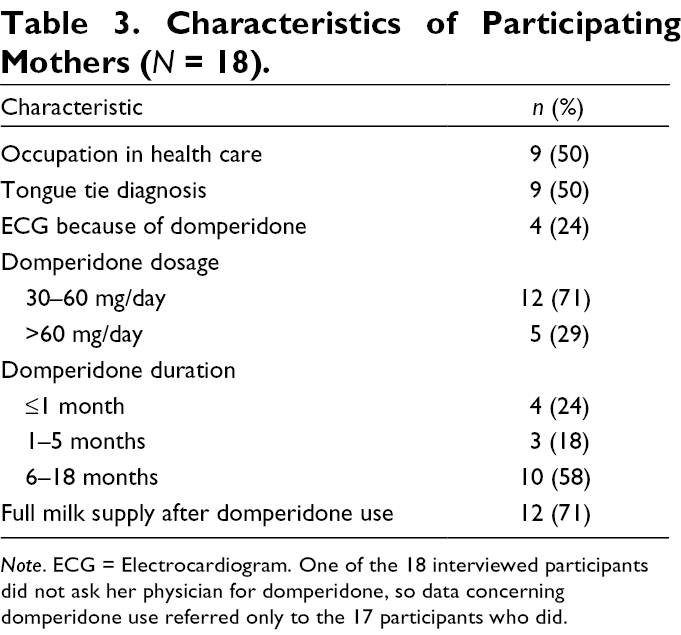
Note. ECG = Electrocardiogram. One of the 18 interviewed participants did not ask her physician for domperidone, so data concerning domperidone use referred only to the 17 participants who did.
Consumer and Health Care Provider Perceptions
We analyzed consumer and health care provider perceptions using the seven topics which formed the basis of the interviews (Table 2).
Mothers’ Requests for Help
The national clinical protocol suggested two parameters to define lactation insufficiency: A pumped volume equal to or less than 350 ml/24 hr in the case of a preterm infant, or insufficient growth in healthy infants. In practice, the participant’s (mother’s) own definition of her problem as lactation insufficiency was accepted by their respective family physicians. Neither participants (mothers) nor their IBCLCs tended to describe their milk supply problems in terms of objective parameters.
When a participant (mother) reported a sick or preterm or very allergic child, both participants (mothers and their IBCLCs) said mothers were more motivated to continue their attempts to increase their milk production when confronted with lactation insufficiency. The participating mother of a preterm infant expressed shock when family physicians suggested opting for formula. On the other hand, participating IBCLCs stated that the chance of producing enough milk is actually greater in the hospital environment, due to closer supervision by professional nurses and IBCLCs.
The intrinsic motivation of the participant mothers influenced their wish to try domperidone. Participant mothers sometimes stated that they could not accept that their body failed to produce enough milk. All participating IBCLCs observed that some participant mothers were very motivated to breastfeed and then chose to use domperidone, whereas others just gave up. Participating family physicians noted that they might prescribe domperidone for the simple reason that mothers tell them that they just really want to breastfeed. Participating mothers commented that their motivation was pre-structured by external guidelines, for example by the World Health Organization’s (WHO; Kramer & Kakuma, 2012) advice to breastfeed exclusively for 6 months, or for 2 years alongside other foods.
Interventions for Milk Supply Problems
The national clinical protocol incorporated the suggestion that increasing milk supply by frequent feeding and pumping should be attempted before turning to domperidone. In practice, of the interviewed mothers, 15 (83%) did put a lot of effort into optimizing breastfeeding management (often including taking herbal galactagogues, among them fenugreek and milk thistle, which they did not consider to be medicines), because taking a medicine to improve production was a radical decision for them. Participants (IBCLCs) also stated that optimization of non-pharmacological interventions should precede advising domperidone use. Other participants (family physicians) were less clear about this, although four of them (D3, D4, D6, and D11) did say that they first inquired about breastfeeding management in order to limit pharmaceutical intervention. The main advice given by participating family physicians was bedrest, preferably together with the infant, sometimes to the frustration of participant mothers who felt they could not follow this advice because of work or other children.
A specific cause of secondary lactation insufficiency that was mentioned in the interviews with participant mothers and their IBCLCs was latch difficulty due to tongue-tie. Of the participant mothers (n = 18) interviewed for this study, nine (50%) stated that their children were diagnosed with and treated for tongue-tie. Participating family physicians reported their experience of being asked about tongue-tie, with one remarking that “You usually can’t get away without a referral.”
Domperidone
When non-pharmacological treatment options had been exhausted or deemed inappropriate, the advice provided in the national clinical protocol was to prescribe domperidone. All but one of the 13 participating family physicians (92%) in this study stated that they did not know about this indication prior to the request of the participating mother, with one commenting that she had had a similar request from another mother in the months following the first one. Participating family physicians said that they were surprised when they were told that you could use domperidone for lactation. Participants (IBCLCs) noted that mothers tend to give up if family physicians are reluctant to prescribe, which is often caused by lack of familiarity with the indication. This lack of knowledge is not limited to domperidone’s mechanism of action; both participating mothers and IBCLCs noted that family physicians often do not know about or confuse the physiological action of prolactin and oxytocin, a hormone which is used in its synthetic form, syntocinon, as a nasal spray to aid in milk ejection.
Of the 17 participant mothers who presented to family physicians, 16 (94%) did so after an IBCLC had already assessed the (presumed) etiology of the lactation insufficiency. In cases of suspected primary insufficiency, suggestions of possible causes by participants (IBCLCs) remained conditional, even depending on the benefit of domperidone use. Participants (IBCLCs) noted that when domperidone had no or insufficient benefit, they suspected primary insufficiency. If participants (IBCLCs) suspected primary insufficiency based on lack of breast growth during pregnancy or directly after delivery, they were sometimes reluctant to advise domperidone because they expected the mother in question would have to use it for an extended period, often with limited success. This was in contrast to mothers who experienced secondary lactation insufficiency after initial sufficient production who, according to the participants (IBCLCs), usually establish a full milk supply after a short period of domperidone use (< 1 month).
The NTvG Article
The availability of the NTvG article describing the national clinical protocol affected whether domperidone was prescribed or not. Participants (mothers and IBCLCs) said they felt that providing family physicians with the article increased their readiness to prescribe. Participants (family physicians) who read the article said this led to less reluctance in prescribing domperidone for lactation. Not all participating family physicians had read the article, and two noted that they may have read it in the journal but did not explicitly recall doing so. Two physicians were not willing to read the article when it was suggested by the participant mother. This type of refusal may be associated with time constraints; one family physician who refused to read it also declined to participate in the present study, stating that she did not have enough time.
However, not all participants (family physicians) based their judgement on the article or the evidence it is based on. In practice, participating family physicians relied on others, frequently the mother. The background of the patient seemed to matter; eight (47%) of the 17 mothers participating in this study who used domperidone were health care professionals (four professional nurses, one midwife, one physical therapist, one psychologist, one physician). Family physician participants took into account the occupation of the participating mothers if they were nurses or midwives or fellow physicians, with one stating: “I didn’t know anything about domperidone, but I did it because she is a midwife” (D3). Participating family physicians also relied on fellow family physicians, specialists (usually pediatricians) and IBCLCs. One participant (IBCLC) pointed out that she was lucky, because in her region family physicians call her regularly, in contrast to the experience of other IBCLCs (L14).
A few of the participating family physicians mentioned consulting the Pharmacotherapeutic Compass (the Dutch reference guide for all medications registered, and some non-registered medications), which does not include a description of the use of domperidone for lactation insufficiency and in fact contains the advice to use restraint in prescribing domperidone as an antiemetic to lactating mothers (Zorginstituut Nederland, 2018). As a second step, three participants (family physicians) discussed the request with the local pharmacist, ultimately leading to approval of the participating mother’s request. Reliance on the pharmacist’s judgement in one case resulted in dosage instructions differing from the regime recommended in the clinical protocol:
Then I consulted the pharmacist and he said “yes, that is possible—it’s an off-label use, but it’s possible.” And he advised: “Begin with [two 10 mg tablets] 3 times per day… and then, as soon as the milk production gets going, you can lower it to [one 10 mg tablet] 3 times per day…. Honestly, I can’t give you more information over the reasoning behind that—then I’d have to refer you to the pharmacist” (D1).
Participants (IBCLCs) noted that pharmacists often are more reluctant than family physicians to recommend dosages higher than 20 mg of domperidone 3 times a day.
Participants (IBCLCs) commented that often parents reacted to hearing that the family physician might be reluctant to prescribe by deciding not to ask at all. A longstanding positive relationship between patient and family physician was given by participants (family physicians as well as mothers) as a reason to feel more positive about prescribing domperidone. One participating mother (M4) said she fabricated a reason for asking for domperidone, saying that her production had diminished after the return of her menses, because she thought this reason would be more acceptable to her family physician than “just having trouble pumping at work.” When participants (family physicians) did critically discuss with their patients whether or not they should prescribe domperidone, two participants reported that they had the feeling they were being put under pressure to prescribe anyway. When participants (family physicians) were reluctant to prescribe, but did not explain why, it sometimes led to friction with their patient. Two participant mothers (M5 and M18) said their family physicians, who at first had refused their request for domperidone, eventually relented under protest, stating they would prescribe “at the own risk of the patient.” According to one participating mother, her family physician even put this in writing (D5, who refused to participate in this study). The other participating family physician (D18), explained that she had not believed domperidone would have any impact on lactation, but had felt uncomfortable saying this, because the mother, IBCLC, and maternity nurse were all present and were adamant that domperidone would provide the solution.
This statement by a family physician participant, who at first had refused to prescribe domperidone due to concerns regarding medication safety, illustrates all of the above:
I realized it had to do with a pediatric nurse and particularly someone with whom a positive connection existed, and I didn’t feel comfortable. So, I did a search, as a result of which I found the NTvG article. Then her colleague called, the same day, the pediatrician who had advised her to try domperidone, to clarify and to direct me to the article I had already found (D4).
Verification of Suitability/Screening
After determining that domperidone was a suitable treatment for lactation insufficiency in general, it was necessary to verify whether it was suitable for the particular patient. Screening for risk factors (Figure 1) is described in the national clinical protocol. In this section, we discuss the extent to which these safeguards were implemented in the prescribing process of domperidone for lactation.
Importantly, one of the three participants (mothers) whose family physicians had requested ECGs was told by her family physician to discontinue use of domperidone after the ECG, because of bradycardia, although the QT-time was within normal limits. The ECG was repeated 2 weeks after having quit domperidone; the bradycardia was gone. The participant (family physician; D6) involved in this case said she had read about ECGs and domperidone in the clinical protocol.
A special group of mothers with primary lactation insufficiency are mothers who want to feed an infant that has not grown in their own uterus. Two participants (mothers) in this study wanted to relactate due to hysterectomies for children born after gestational surrogacy. Protocols for induced lactation using domperidone have been published (Goldfarb & Newman, 2015; Newman, 2014) and were quoted by participants (IBCLCs and mothers). Within these protocols, higher dosages, up to 90 mg 3 times a day, were suggested. Although the two participants (family physicians) prescribed this amount, the pharmacies refused to dispense it. After discussion with the participating mother’s IBCLC, one of these participants received the higher dosage after all.
Of the 17 participants (mothers) interviewed in this study who took domperidone for lactation insufficiency, 12 (71%) reported achieving a full milk supply without need for supplementation. Of these 12 participants, nine used a dosage of 30 mg/day, of which six tapered to 20 mg/day and later three of these tapered to 10 mg/day. Both participants (mothers) who achieved full milk supply using higher dosages (90 mg/day) were relactating after previous normal lactation periods (see Table 3).
Discussion
Although we found that availability of a national clinical protocol supporting the prescription of domperidone for lactation appeared to increase the willingness of physicians to prescribe, it is unclear whether or not the NTvG article was accepted as a national clinical protocol in practice. In addition, there is no international consensus on best practice for prescribing domperidone. Other protocols have been published which, for instance, do not recommend an ECG after 2 weeks of treatment. Nevertheless, regarding the NTvG article as a national clinical protocol enabled us to explore perspectives and experiences in the absence of official national guidelines in the Netherlands.
Gaps were identified between clinical practice and the recommendations in this clinical protocol. For instance, to ascertain the need for medical intervention, participating family physicians relied on the patient’s and the IBCLC’s definition of milk supply problems, rather than assessing the situation themselves. In order to inform appropriate prescription of domperidone, it might be beneficial if family physicians inquired more systematically about milk production and infant growth.
All but one of the 13 (92%) participants (family physicians) stated that they did not know about this indication prior to the request of the participating mother. This is in accordance with quantitative data from 2016, provided by the Netherlands Institute for Health Services (Nivel) research. Over approximately 500 family practices, domperidone was prescribed to approximately 0.01% of all pregnant women (Karin Hek, Nivel, personal communication, January 22, 2018).
In contrast to prior quantitative studies in which the pharmacological treatment of lactation insufficiency by physicians was analyzed (Axelsson et al., 2014; Grzeskowiak et al., 2013), our aim was to provide insight into the motivations of mothers, IBCLCs, and family physicians. One of the strengths of the study was our attention to the diversity of these viewpoints.
The topics structure we have used resembles the 6-step method of the WHO’s (Keijsers et al., 2015) guide to state-of-the-art prescribing. To improve the prescribing process of domperidone, a review and possibly an update of the national clinical protocol incorporating the WHO model would be desirable. This should also include comparison with other published protocols for the prescription of domperidone, for instance one published by Haase et al., 2016 that contains more elaborate handouts and checklists for history taking and physical exams, as well as concrete advice on tapering domperidone use to the minimally necessary dosage to maintain milk supply.
Aside from exploring the prescribing process of domperidone for lactation insufficiency, we have raised questions about the way family physicians handle requests for off-label pharmacological treatment initiated by their patients. We have indicated that publishing an evidence-based protocol is no guarantee for implementation in the family physician’s office. It may be better to opt for regulation and a more official status for comparable protocols. Strategies aimed at improving the translation of protocols into clinical practice should be considered in follow up research.
Other ways of recruiting participants, for instance through a prospective study or through experimental research, are recommended for future studies. A prospective study would also enable the formulation of a diagnostic model that could provide a firmer basis for categorizing mothers as suffering from primary or secondary lactation insufficiency. This, in turn, would have the potential to inform the design of quantitative researchers who aim to elucidate the effectivity of domperidone for primary and secondary lactation insufficiency, including dosage and duration.
Limitations
As the study progressed, it became clear that pharmacists also play an important role in the prescribing process of domperidone for lactation. Due to time constraints it was not feasible to include pharmacists in this study. This is a limitation which should be addressed in further research.
By using semi-structured interviews, this study focused solely on the impression that interviewees had of their own actions. It would be valuable to contrast this with direct observation of interactions between patients and health care providers.
Selection effects may well have had an influence on our results. It is, for example, remarkable that over half of the mothers had a health care profession. However, the observation that physicians referred to the profession of the mother if she was working in health care remains valid.
Conclusion
The availability of a national clinical protocol supporting the prescribing of domperidone for lactation appeared to increase the willingness of physicians to prescribe. However, gaps were identified between clinical practice and this clinical protocol. Screening safeguards that should identify contra-indications for domperidone were frequently not implemented in practice. Participating mothers regularly used much higher dosages. Success of the intervention was not evaluated by participating physicians. Understanding of consumer and health care provider perspectives should inform future clinical protocols concerning medication for lactation insufficiency.
Supplemental Material
Supplementary Material 1 - Supplemental material for Exploring the Prescribing Process of Domperidone for Low Milk Supply: A Qualitative Study Among Mothers, IBCLCs, and Family Doctors
Supplemental material, Supplementary Material 1, for Exploring the Prescribing Process of Domperidone for Low Milk Supply: A Qualitative Study Among Mothers, IBCLCs, and Family Doctors by Lara A. Tauritz Bakker, Liset van Dijk, and Patricia M. L. A. van den Bemt in Journal of Human Lactation
Footnotes
Authors’ Note
The abstract of this article has been published before in Breastfeeding Medicine (2019): Abstracts From The Academy of Breastfeeding Medicine 24th Annual International Meeting, Blackburn, United Kingdom October 16–19, 2019.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LT was a final year medical student at Erasmus University, Faculty of Medicine, Rotterdam, the Netherlands; this research represented her compulsory Master’s research project to obtain the MD title. Her projected graduation date is October 2020. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
