Abstract
Background
Failures in communicating risks associated with consumer products, including pharmaceuticals, foods, and medical devices, have repeatedly contributed to preventable morbidity and mortality and have undermined public trust in product safety.
Purpose
To argue that the prevailing consumer product risk communication paradigm is structurally inadequate and to propose an integrated framework that shifts risk communication from a reactive to a proactive public health function.
Research Design
Conceptual and policy analysis informed by lessons from major safety crises and established principles of risk communication and risk perception science.
Study Sample
Not applicable (no human participants or clinical dataset).
Data Collection and/or Analysis
We synthesize recurring system-level vulnerabilities in consumer product oversight (eg, fragmented regulation, reliance on industry self-reporting, misaligned corporate incentives, and limited incorporation of risk perception science) and integrate these insights with evidence-based communication principles to derive a proposed framework emphasizing transparency, clarity, independent validation, and accountability.
Results
We identify persistent, predictable failure points that enable delayed recognition and disclosure of risk, inconsistent public messaging, and inadequate accountability, collectively fostering a reactive posture that permits avoidable harm. We propose an integrated framework with four pillars: (1) proactive transparency across the product lifecycle, (2) evidence-based communication clarity tailored to how individuals perceive and act on risk information, (3) independent validation to reduce conflicts of interest and improve credibility, and (4) systemic accountability mechanisms that align corporate behavior with public health goals.
Conclusions
Incremental refinements to current regulations are unlikely to resolve the recurring, structural drivers of risk communication failure. An integrated, proactive framework is an urgent public health imperative to prevent harm, better equip clinicians and consumers to make informed decisions, and restore confidence in the safety of consumer goods.
Keywords
Introduction
Each year, preventable morbidity and mortality arise directly from failures communicating risks associated with widely used consumer products, including pharmaceuticals, medical devices, and foods. Delayed warnings about drug side effects contribute to adverse events. Inadequate alerts about contaminated food lead to infectious disease outbreaks. Obscured risks in medical devices result in patient injury and revision surgeries. These are not isolated incidents but symptoms of systemic weaknesses in how risks are identified, assessed, and communicated. Although regulatory frameworks and legal doctrines exist, they have proven insufficient to consistently compel the timely, clear, and actionable communication necessary to prevent harm. Clinicians stand at a critical juncture, interpreting product information for patients and observing potential adverse events. Yet, they, too, are often hampered by delayed or incomplete safety disclosures from manufacturers and regulators.
We argue that the prevailing reactive, compliance-focused model of risk communication is fundamentally inadequate and poses an ongoing threat to public health. The current system remains overly reliant on measures triggered by a crisis, vulnerable to corporate pressures that can prioritize profit over timely disclosure. It frequently fails to incorporate established principles of risk perception. We believe that only a paradigm shift towards a proactive, transparent, and ethically grounded framework can effectively mitigate these dangers and restore essential public trust among consumers, clinicians, and the institutions designed to protect them.
Systemic Failures Undermining Timely Disclosure
The landscape of consumer product oversight suffers from critical structural flaws. Fragmentation across agencies (eg, FDA, CPSC in the United States) coupled with limited resources for independent postmarket surveillance creates significant vulnerabilities. This requires heavy reliance on industry self-reporting, establishing an untenable conflict of interest where fiduciary duty to shareholders can directly oppose the public health imperative for rapidly disclosing emerging hazards. The FDA’s GRAS (Generally Recognized As Safe) process for food additives, allowing manufacturers self-affirmation of safety, exemplifies a broader regulatory philosophy that can systematically underestimate the pressures against transparency. 1 This dependence allows crucial safety information to be delayed or minimized when companies fear financial or reputational repercussions.
The catastrophic 2008 Chinese milk scandal, causing widespread renal injury in infants due to melamine adulteration, starkly illustrates the lethal potential of these combined failures. 2 Initial warnings were suppressed, prioritizing economic concerns over immediate public health action. By the time the truth emerged, the damage was extensive, and global trust was shattered. While an extreme example, the underlying dynamics, including fragmented authority, incentives favoring concealment, and delayed action, are recurrent themes in less dramatic but harmful product safety failures across jurisdictions.
Furthermore, legal doctrines intended to ensure safety can paradoxically impede effective communication. While the “failure to warn” principle creates manufacturer liability, 3 as affirmed in cases like Wyeth v. Levine, the threat of litigation often incentivizes defensive communication strategies. This can manifest as “overwarning”: burying critical safety information within the dense, legalistic text that few consumers or clinicians can parse effectively, or strategic delays to manage liability exposure rather than prevent patient harm. This environment risks transforming risk communication from a vital public health function into a calculated legal tactic, fundamentally misaligning its purpose. Corporate structures emphasizing short-term profits can exacerbate these tendencies, pressuring managers to downplay or defer disclosures that might impact quarterly earnings.
The Perception Gap: Ignoring the Science of Communication
Effective risk communication demands more than factual accuracy; it requires a sophisticated understanding of human psychology. Decades of research in cognitive psychology illuminate how risk perception is shaped by heuristics (eg, the availability heuristic making dramatic events seem more likely), framing effects (eg, “95% safe” vs “5% risk”), and critical, trust in the information source. 4 Yet, these foundational principles are routinely ignored in corporate and even regulatory communication strategies, leading to predictable failures in conveying urgency, fostering understanding, and enabling appropriate protective action by the public and healthcare providers.
The failure is not a lack of knowledge but a lack of application. The stark contrast between Johnson & Johnson’s handling of the 1982 Tylenol tampering crisis, characterized by rapid, transparent action and empathetic communication that ultimately rebuilt trust, and the distrust sown by initial denials in the melamine crisis 2 underscores this point. Effective communication that accounts for perception is achievable but requires deliberate integration of psychological insights. Ignoring the recipient’s cognitive and emotional landscape ensures that even well-intentioned warnings may fall short, failing to change behavior or breeding cynicism and mistrust.
A Framework for Proactive and Responsible Risk Communication
Comprehensive Framework for Proactive Risk Communication in Consumer Product Safety, Outlining Seven Core Components With Implementation Strategies and Common Pitfalls to Avoid
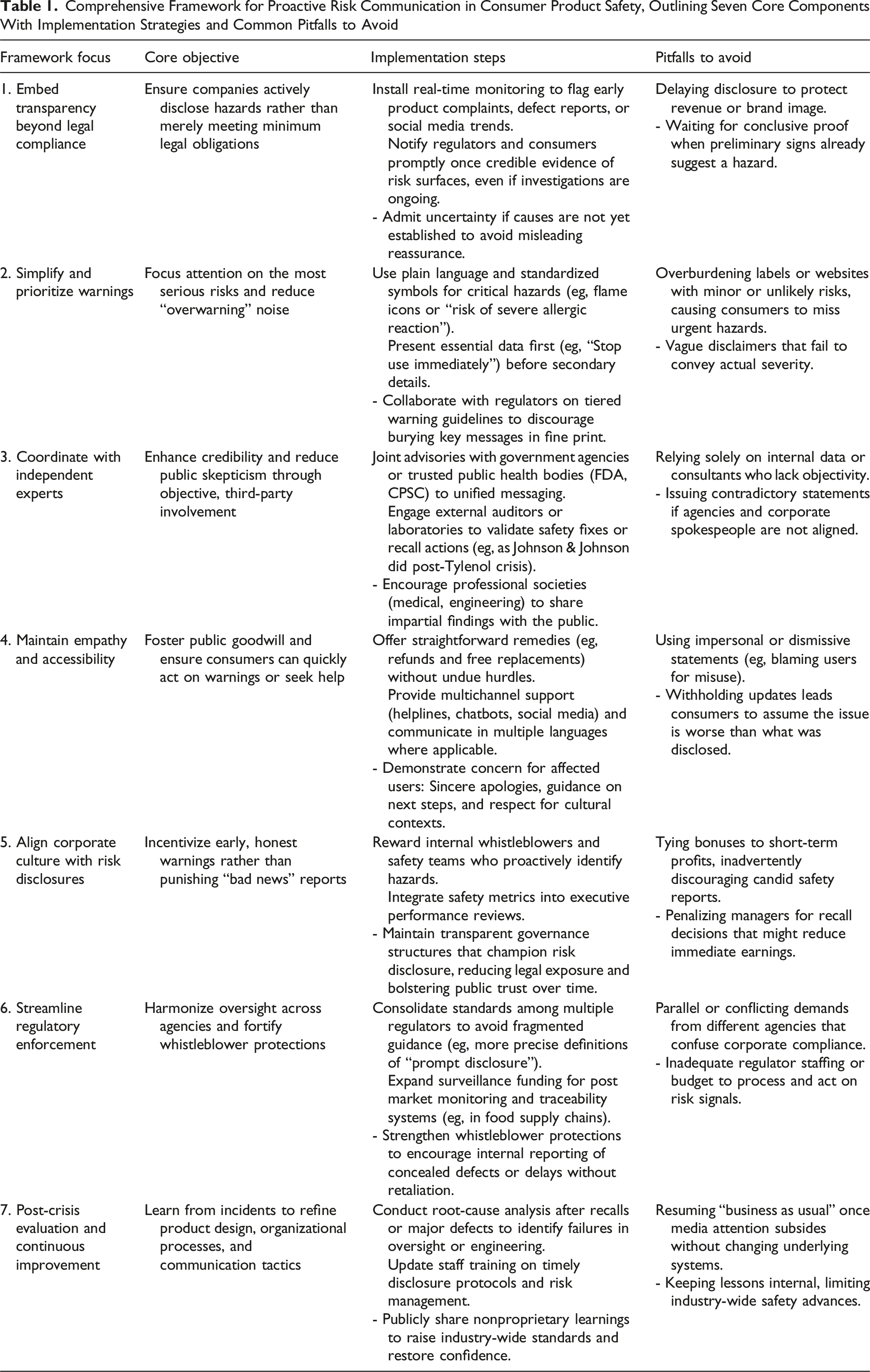
Embed Proactive Transparency Beyond Compliance
To counter the systemic bias towards delayed disclosure, organizations must implement robust early warning systems insulated from commercial pressures, with pre-defined thresholds for mandatory public notification. A dedicated, empowered Consumer Safety Officer is crucial. This shifts the default from reactive silence to proactive transparency.
Prioritize Clarity and Actionability in Warnings
Tackling ineffective ‘overwarning’ driven by legal defensiveness, this component mandates tiered warnings using plain language, standardized visuals, and rigorous comprehension testing. The goal is to ensure critical information is immediately identifiable and actionable for consumers and clinicians.
Enhance Credibility through Independent Collaboration
Addressing the inherent credibility deficit of purely corporate messaging, this principle requires structured engagement with independent experts, regulators, and public health bodies for message co-development, data validation, and joint communication during crises leveraging trusted voices.
Center Empathy and Accessibility
Recognizing that communication is human interaction, this involves multi-channel, culturally sensitive outreach using accessible language and formats, particularly for vulnerable populations. It also includes streamlined remedy processes, demonstrating genuine concern for affected individuals.
Align Organizational Culture and Incentives with Safety
This component targets the underlying incentive structures because policies alone are insufficient. It involves integrating safety metrics into performance, protecting whistleblowers, and fostering leadership that unequivocally prioritizes ethical disclosure over short-term financial gain.
Advocate for Streamlined Regulation and Robust Enforcement
To create an environment conducive to transparency, this principle calls for harmonizing regulatory standards, enhancing independent surveillance capabilities, strengthening whistleblower protections, and implementing enforcement that meaningfully penalizes concealment while potentially recognizing proactive disclosure.
Implement Post-Crisis Evaluation and Continuous Improvement
Ensuring organizational learning involves systematic root-cause analyses after incidents, updating training based on findings, and sharing nonproprietary lessons to elevate industry-wide practices, creating a feedback loop for sustained improvement.
Conclusion
The recurring cycle of consumer product safety failures, marked by delayed warnings and eroded trust, demonstrates the profound inadequacy of our current risk communication paradigm. The consequences are measured in economic losses and preventable suffering, injury, and death. Continuing with the status quo, characterized by reactive measures, conflicting incentives, and insufficient regulatory reach, guarantees further preventable harm and more profound public cynicism.
As outlined here, the transition to a proactive, transparent, and ethically grounded framework is therefore not merely advisable but constitutes an urgent public health imperative. This requires concerted action: Industry leaders must champion cultural transformation and invest in robust safety and communication systems prioritizing public well-being. Regulators must evolve towards harmonized standards, enhanced independent surveillance, meaningful enforcement against non-disclosure, and stronger whistleblower safeguards. The medical and public health communities, including clinicians on the front lines, are vital in advocating for these reforms, contributing to surveillance through diligent reporting, and helping patients navigate complex product safety information. Adopting a genuinely responsible approach to risk communication is an ethical necessity and a fundamental requirement for protecting public health in an increasingly complex global marketplace.
Footnotes
Author’s Note
AI was used to proofread and to change syntax on several sentences for phrasing purposes to get under word count, however, was later edited over. AI was not involved in anything relating to idea generation, formatting, or any other reason.
Ethical Considerations
This manuscript presents a theoretical framework based on literature review and analysis of existing cases. No primary data collection involving human subjects was conducted, therefore IRB approval was not required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health NIDDK U24 DK132733, P30 DK040561, and UE5 DK137285 (FCS).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
