Abstract
Despite increased prevalence of domestic violence and abuse (DVA), victimization through DVA often remains undetected in mental health care. To estimate the effectiveness of a system provider level training intervention by comparing the detection and referral rates of DVA of intervention community mental health (CMH) teams with rates in control CMH teams. We also aimed to determine whether improvements in knowledge, skills and attitudes to DVA were greater in clinicians working in intervention CMH teams than those working in control teams. We conducted a cluster randomized controlled trial in two urban areas of the Netherlands. Detection and referral rates were assessed at baseline and at 6 and 12 months after the start of the intervention. DVA knowledge, skills and attitudes were assessed using a survey at baseline and at 6 and 12 months after start of the intervention. Electronic patient files were used to identify detected and referred cases of DVA. Outcomes were compared between the intervention and control teams using a generalized linear mixed model. During the 12-month follow-up, detection and referral rates did not differ between the intervention and control teams. However, improvements in knowledge, skills and attitude during that follow-up period were greater in intervention teams than in control teams: β 3.21 (95% CI 1.18-4.60). Our trial showed that a training program on DVA knowledge and skills in CMH teams can increase knowledge and attitude towards DVA. However, our intervention does not appear to increase the detection or referral rates of DVA in patients with a severe mental illness. A low detection rate of DVA remains a major problem. Interventions with more obligatory elements and a focus on improving communication between CMH teams and DVA services are recommended.
Keywords
Background
Domestic violence and abuse (DVA) here defined as “any incident of threatening behavior, violence or abuse (physical, sexual, emotional and/or material) between adults who are or have been intimate partners, family members, friends or otherwise closely related (Stuurgroep multidisciplinaire Richtlijnontwikkeling in de GGZ, 2009)”, is a major problem worldwide and it affects people in all social classes (Devries, Mak, Bacchus et al., 2013; Devries, Mak, Garcia-Moreno et al., 2013; Ellsberg et al., 2008; Gracia et al., 2019; Henning & Feder, 2004; Hines & Douglas, 2012; Hines & Douglas, 2015; Sanz-Barbero et al., 2018).It can have serious consequences, which can be both psychological (Chandra et al., 2009; Devries, Mak, Bacchus et al., 2013; Satyanarayana et al., 2015) and physical (Campbell, 2002), as well as short term or chronic (Campbell, 2002; Ferrari et al., 2016; Heyman et al., 2009; Loxton et al., 2017; Oram et al., 2013; Reid et al., 2008; Trevillion et al., 2012).
Contrary to popular belief, preexisting mental illness is associated with a higher risk of DVA victimization. This association is much higher than the association with mental illness and DVA perpetration (Khalifeh & Dean, 2010; Swartz & Bhattacharya, 2017). A Dutch cross-sectional study showed that serious mentally ill (SMI) patients are three times more at risk to be a victim of personal violence compared to the general population (Kamperman et al., 2014). Furthermore, in the general population, men are more often perpetrators of DVA and women are more often victims. However, in SMI patients this difference is greatly reduced as both genders are at a high risk of victimization of DVA (Carmo et al., 2014; Howard et al., 2010; Jonas et al., 2014). Several studies have shown that male SMI patients are more likely to be a victim of DVA compared to the general population (Khalifeh et al., 2015), while they are also less likely to seek help (Huntley et al., 2019). Most interventions are focused on female victims of DVA, and very little interventions are geared toward both genders (Rivas et al., 2019), consequently creating a gap in literature and interventions for DVA victims.
Despite the high risk of victimization, only 10% to 30% of all victims of DVA are detected in mental health care (Howard et al., 2010). This indicates that a large group of SMI patients will not receive the care they need. In response, universal DVA screening has been proposed and studied as a means to address the issue (O’Doherty et al., 2014). In their meta-analysis, O’Doherty et al., (2014) conclude that universal screening increases detection of DVA at antenatal clinics. However, the findings did not hold in the context of hospital outpatient screening. They conclude that studies on universal screening so far have been successful in finding only a modest amount of absolute cases, especially considering the high prevalence of DVA victimization (World Health Organization, 2013).
Reasons for the low DVA detection rates in mental health care vary ranging from a lack of knowledge about DVA to not finding DVA a priority in the treatment of mental illness (Rose et al., 2011). As previously mentioned, males are likely to be considered perpetrators of DVA, even though their SMI status makes them more vulnerable to becoming victims. This knowledge gap is problematic as current detection tools are not sensitive to differences arising from gender (Arkins et al., 2016). It is important, therefore, to target both male and female patients when screening for DVA, especially in the context of mental health care. Another source of concern is that interventions often target narrowly defined elements of the health care system instead of operating at a wider systems level. This has been identified as effective ground for improvement, in response of which interventions have started incorporating training for all staff in a health care system (Ghandour et al., 2015; Hamberger et al., 2015; Garcia-Moreno et al., 2015). Furthermore, previous system-level programs were shown to be both effective and sustainable by implementing a training course for clinicians about detection of patients who are victim of DVA, along with the provision of a referral and care pathway and also include training for DVA professionals on mental health further along the referral line (Feder et al., 2011; Trevillion et al., 2014). The training on mental health for DVA professionals is key to increase a mutual understanding between mental health care professionals and DVA professionals which would improve collaboration and ultimately could greatly increase health care for SMI patients and/or victims of DVA (Feder et al., 2011; Trevillion et al., 2014). Considering the possible life-long consequences of victimization of DVA for people with SMI, the detection and treatment of DVA needs to be improved by both being sensitive to gender differences and introducing interventions at a system level. In mental health care so far, no studies have launched a system-level intervention.
The current Better Reduction and Assessment of Violence (BRAVE) study is the first full-scale cluster randomized controlled trial aiming at improving detection and referrals on DVA in mental health care, using a gender sensitive, system-level program that targeted community mental health (CMH) teams, and also provided mental health training for DVA professionals.
Aims
We conducted a cluster randomized controlled trial using a DVA intervention developed for this study called the BRAVE intervention. On a system level, our aim was to compare the detection and referral rates of DVA of CMH teams who received the BRAVE intervention and those in the control condition who did not. We expected the detection and referral rates of DVA to increase more in the intervention condition than in the control condition. On an individual level, we also aimed to determine whether clinicians in the intervention group showed comparatively higher improvements in (a) knowledge; (b) perceived knowledge; (c) skills in handling DVA; and finally (d) attitudes toward DVA.
Methods
Participants and Study Setting
The trial period was from February 2016 to February 2017 for all Rotterdam teams and February 2017 to February 2018 for all The Hague teams. Participation in the BRAVE study was voluntary, and consent was assumed from participation. The delayed start of The Hague teams was the consequence of organizational changes among participating CMH institutions in an early phase of the project before randomization. A typical CMH team consists of 9-14 of diverse mental health clinicians. The composition of each team varies slightly, but each team employs a psychiatrist, psychologists, general nurses, social psychiatric nurses, and social workers. Teams that provided outpatient care to SMI patients 18 years or older, with ≤ 20% of employees working in more than one team and who had a functioning electronic patient file (EPF) with at least 12 months of historic data, were eligible for the study. The included CMH teams had a mean caseload of 195 patients.
The Medical Ethical Committee (METC) of the Erasmus Medical Center in Rotterdam, the Netherlands granted ethical approval for this study (approval granted on 10th of June 2015, MEC-2015-409). The study protocol has been published previously (Ruijne et al., 2017). Team managers and the board directors of the participating institutions provided consent for participation of the CMH teams. Since the intervention was aimed at the clinician working in CMH teams, the METC did not find informed consent from patients necessary. The trial has been registered on the ISRCTN register (number: ISRCTN14115257). This article is reported according to the CONSORT extended guidelines (Campbell et al., 2012).
Design
In this cluster randomized controlled trial, we studied the impact of the BRAVE intervention in CMH teams on the detection and referral rates of DVA in SMI patients. CMH teams from the Rotterdam and The Hague area functioned as clusters. Rotterdam and The Hague are two major and ethnically diverse cities in the Netherlands. We randomly assigned 24 CMH teams to the BRAVE intervention or to the control condition with an allocation ratio of 1:1 (Figure A.1). Randomization was performed with block sizes of 2, using a web-based computer-generated scheme in ten-ALEA (randomization software version 2.2) by an independent researcher. The socioeconomic status of the service region of each included CMH team (dichotomized into high vs. low) was used as stratification factor. Due to the nature of our intervention, it was not possible to blind the CMH teams for allocation status.
Intervention
The BRAVE intervention consists of three parts: (a) a training course for CMH teams to increase knowledge, attitudes, and skills in managing DVA, (b) a knowledge, attitudes, and skills training course on mental illness for DVA professionals, and (c) the provision and implementation of a direct care referral pathway between CMH services and DVA services for victims of DVA. The BRAVE intervention is based on the successful LARA intervention (Trevillion et al., 2014) and adapted to the Dutch context and circumstances and has described in the published protocol (Ruijne et al., 2017).
A knowledge, attitudes and skills training about DVA for CMH teams. All CMH clinicians from the intervention teams received an eight-hour training course, divided into two four-hour sessions. The training course is based on two clinical handbooks for health care for victims of domestic violence and abuse or sexual violence and amended to fit our patient population (Tobler et al., 2008; World Health Organization, 2013) and was adjusted in order to be able to focus on both male as female victims of DVA and mental health patients. The training met the standards for courses on DVA of the World Psychiatry Association (Richards et al., 2003). The training covered four themes: (a) facts and theories about DVA; (b) identification and documentation techniques for the assessment of DVA; (c) safety for both patient as CMH clinician; and (d) treatment and/or follow-up which consisted of a referral pathway and proposes strategies to empower and support patients who are victims of DVA. The training consisted of lectures, documentary media, and interactive workshops among other similar teaching methods. The information in the training focused on both male and female victims and also addressed possible cultural differences participants could notice in their diverse patient population. Participants were informed about the mandatory Dutch decision-making process for referral after DVA is detected or suspected. During the training and follow-up period we recommended participants to add questions about DVA to their routine enquiry and report detected cases of DVA in a special DVA form embedded in the EPF. Additionally, participants received a referral pathway with the details and contact information of shelters and DVA services for victims or perpetrators of DVA. Mean attendance rate per team was 86% (range 67%-100%). After the training all CMH clinicians in the intervention teams received a handout of the training and were provided with various documents with information about local DVA services and ways (e.g., an app, documents) for reminding them to inquire about DVA with their patients. Knowledge, attitudes and skills training on mental health for DVA professionals. We noted previously the importance of system-level intervention, instead of exclusive focus on one element, in this case the CMH teams. To expand the intervention to the wider system, DVA professionals working in the municipality of Rotterdam also received four, three-hour workshops about mental illness during the intervention period. DVA professionals within the municipality of The Hague, however, declined participation (not to be confused with the CMH teams in The Hague which participated fully). The DVA professionals were not part of the randomization process. This workshop was inspired by the manual for mental health training for community health workers published by the World Health Organization (Richards et al., 2003). It consisted of three main themes: (a) information on mental illness: prevalence, definitions, and symptoms; (b) the organization of mental health care in the Netherlands, and (c) Dutch laws and legislation with regard to health care and mental health care, and communication and the exchange of information with professionals working in (mental) health care. Because of a nation-wide reorganization of the municipal public health service, participation could not be made mandatory. Provision and implementation of a direct-care referral pathway for victims of DVA. All CMH teams received the information and tools needed to implement a direct-care referral pathway in their daily working routine. This pathway contained the mandatory Dutch decision-making protocol on whether to refer (suspected) victims of DVA, as well as possible DVA institutions to refer to. The pathway was brought to the attention of the intervention teams in the following ways: it was included in the handouts that the intervention teams received after the training, recalled in bi-monthly newsletters, given to teams on a large poster, and it was available as smartphone app. The mental health institutions included in this study all had assigned a DVA advocate from within each CMH team. The main task of a DVA advocate is to remind colleagues to ask their patients about DVA and to act as a consultant regarding questions related to DVA. To improve the quality of referrals to DVA services and the communication and collaboration between mental health care and DVA professionals, we aimed to add a professional to all intervention teams.
Fidelity
Intervention fidelity for each team was assessed over the course of the intervention period using a prespecified fidelity scale. Fidelity was assessed by an independent researcher (KB). Information was collected over the course of the intervention period during routine visits (centered around support during training days, provision and restock of paper intervention material, and ad hoc consultancy on DVA knowledge, attitudes and skills, and updates on the referral pathway), and from a semi-structured interview by telephone with a representative of each intervention team at the end of the intervention period. The fidelity scales scored whether the five core components of the BRAVE intervention were adequately present. Each item could be scored with either yes or no (1 or 0). Component 5 consisted of one item which could be scored from 0 “not present” to 3 “weekly.” The components were as follows: (a) a DVA training course for mental health care professionals (3 items; e.g., “Every team has at least one manual on DVA”); (b) mental health training course for DVA professionals (3 items; e.g., “The training includes a short description of the major mental illnesses”); (c) identification of the components of DVA in routine CMH practice (6 items; e.g., “The training provided conversation techniques to enquire about DVA”); (d) integrated referral and care guideline (2 items; e.g., “An integrated referral and care guideline is available”), and (e) collaboration between CMH professionals and DVA-professionals (1 item; “A DVA professional joins our meetings at least once a month”). Maximum score (17points) reflects excellent intervention fidelity.
Fidelity scores ranged from 6 to 11, with a median score of 9.25 points. Overall, fidelity was limited by the absence of mandatory training of DVA professionals and of an integrated referral guideline between DVA services and CMH services as a result of a radical reorganization of all municipal public health services, including DVA services during the trial period. For the purpose of our analysis, a median split was used to dichotomize the intervention fidelity score of each team into high (n = 6) versus low (n = 6) fidelity.
Control Condition
Teams in the control condition did not receive a DVA training course or any additional information about DVA institutions or a referral pathway. They provided care as usual which consisted of outpatient care to patients with a severe mental illness. Clinicians in the control teams filled in the BRAVE survey at baseline, and after 6 and 12 months after the start of the intervention. A research assistant collected the completed paper surveys at the sites.
Measures
Primary Outcomes
The primary outcomes were the number of current DVA cases detected and the number of cases referred per team. To operationalize how many DVA cases were detected and referred, we searched through the EPFs of patients treated by each participating team during the 12-month period prior to the intervention, and at 6 and 12 months after the start of the intervention (a total of 24 months, divided into four times six months). The process of searching and extracting the outcome measures is described in full in the appendix (Appendix A). In short, an automatic search query was performed on the patient files and those flagged as potentially case of DVA were additionally vetted by two independent researchers (RR and HG) and categorized in either a detected current case of DVA or no DVA. Detected cases were then categorized as perpetrator or victim and type of violence (sexual, physical, material or emotional). Violence was stratified to physical violence if it was described in the EPF as a patient being, for example, slapped, hit or otherwise physically assaulted, this included misspellings and synonyms. Violence was marked as sexual if a patient was for example, raped, or otherwise sexually assaulted. Violence was marked as emotional if the EPF described a patient for example, being threatened or stalked. Violence was marked as material if a patient for example, was described to have been a victim of vandalism or financially exploited. A table with the words used in the search query can be found in the Appendix (Appendix A; Tables A.1 and A.2). For exploratory purposes, cases were also examined if they were referred to a DVA professional, externally discussed (discussed with a DVA service whether referral is necessary), internally discussed (discussed in a multidisciplinary setting whether referral is necessary) or no follow-up action was taken. Thus, detected cases were the sum of all cases in the patient files classified as such by the researchers and the number of DVA referrals was the sum of all follow-up actions. Refer to Appendix A for additional details on the procedure for searching, flagging, and categorizing cases and referrals. Furthermore, a flowchart depicting the operationalization process of the data extraction from the EPFs is also included in the Supplement (Figure A.2.).
Secondary Outcomes
Clinicians’ readiness to manage DVA was assessed using the BRAVE survey at time points: baseline, 6 months after intervention and 12 months after intervention.
The BRAVE survey is a self-report questionnaire and consists of five sections (Ruijne et al., 2019): (a) respondent characteristics that are used to assess personal and professional information, (b) perceived skills to manage DVA, (c) clinicians’ perceived knowledge about DVA (d) clinicians’ factual knowledge on DVA, and (e) clinicians’ attitudes toward DVA. A total score is calculated by summing the Sections 2-5 (score range: 0-127. Internal reliability of the subscales was found to be sufficient (Section 5) to excellent (Section 2; Ruijne et al., 2019).
CMH Teams and Respondents’ Characteristics
Demographic and Professional Characteristics of the Teams (N = 24) and the Individual Respondents of the BRAVE Study (N = 214).
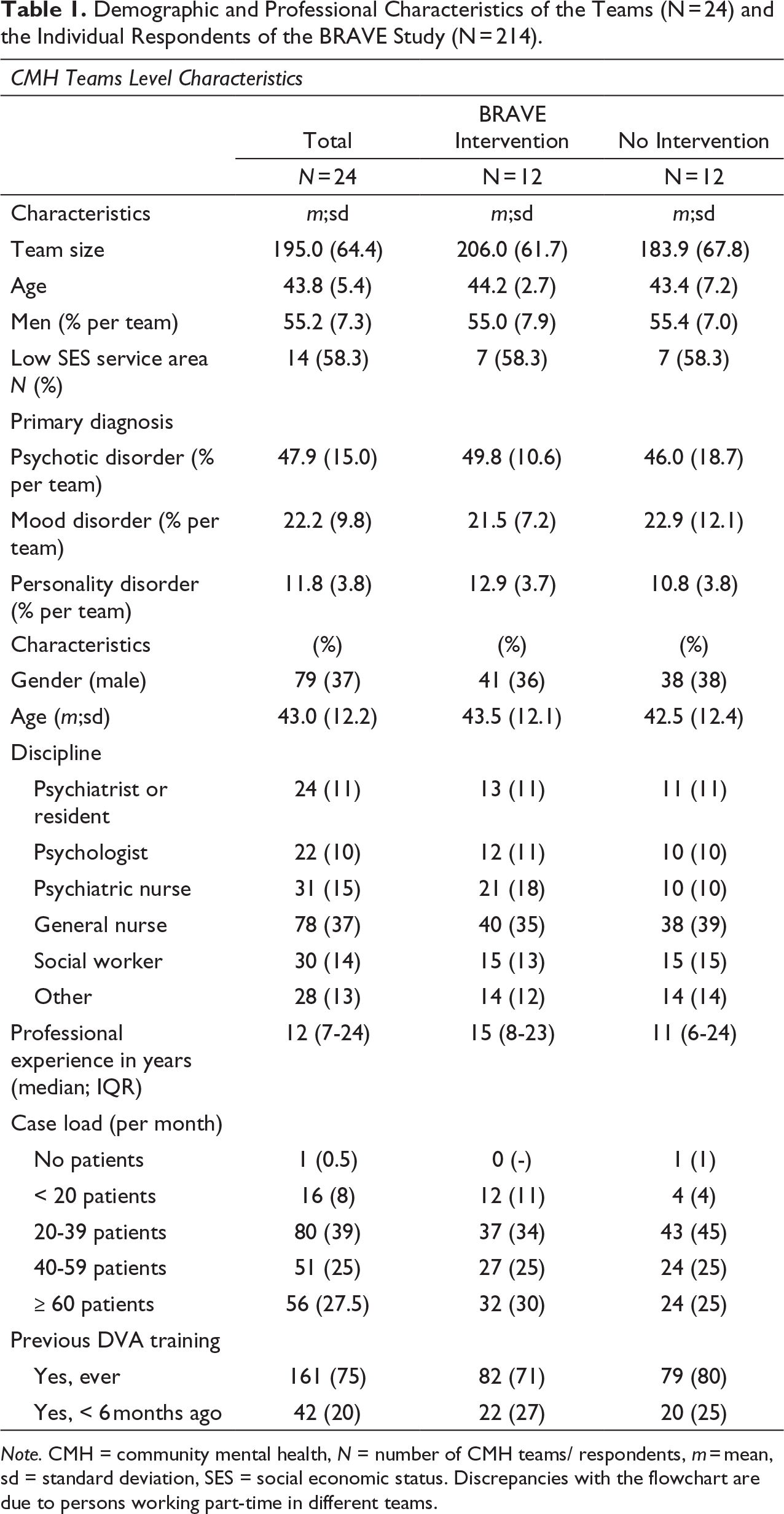
Note. CMH = community mental health, N = number of CMH teams/ respondents, m = mean, sd = standard deviation, SES = social economic status. Discrepancies with the flowchart are due to persons working part-time in different teams.
Statistical Analysis
The effect of the BRAVE intervention on the rate of DVA cases detected and referred over the four time points was estimated using a generalized linear mixed model with logit link and binomial distribution. Our model included intercept, allocation (BRAVE vs no intervention), time (as a continuous variable), and the interaction term of allocation with time. A significant (p < .05) time × allocation term was considered indicative of an intervention effect. The number of patients detected or referred during the intervention period was used as the nominator and the number of patients treated during this period was used as the denominator. We stratified the cases according to the type of violence (physical, sexual, emotional, and material), and whether the cases concerned victimization, perpetration or both. Since the use of a structured DVA form in the EPF is recommended during the BRAVE training, we assessed the rate of completed DVA forms. By means of sensitivity analysis, analyses were repeated excluding cases of violence occurring among roommates (i.e., sheltered housing), and adjusted for standardized baseline rate. We report the OR (odds ratio) of the time × allocation interaction term including 95% confidence interval and p-value, as well as the estimated rates at the end of the intervention period (at 12 months). Additionally, we plotted the estimated mean rates and standard errors over the full intervention period.
The effect of the BRAVE intervention on the clinicians’ readiness to manage DVA was estimated using a multilevel generalized linear mixed model. The model included intercept, allocation (BRAVE vs no intervention), time (three time points modeled as a continuous variable), and the interaction term of allocation with time. CMH team membership was included in the model as a level. A significant (p < .05) time × allocation term was considered indicative of an intervention effect. Assessments were weighted for the inverse of the number of teams a CMH professional was employed by, for example a CMH professional employed by two teams was analyzed as member of both teams with a 0.5 weighting factor each assessment. We assessed the effect of the BRAVE intervention on the BRAVE survey total score, and on the subscale scores independently. Sensitivity analyses were performed regarding the CMH clinicians who completed the training, for CMH clinicians who completed all three surveys, and adjusted for standardized baseline score. We report the unstandardized regression coefficient of the time × allocation interaction term including 95% confidence interval and p-value, as well as the estimated mean scores at the end of the intervention period (at 12 months). Estimated mean scores and standard errors are plotted over the full intervention period. Finally, we analyzed the detection and referral rates as well as the BRAVE survey total score stratifying teams with a high intervention fidelity versus teams with a low fidelity. Analyses were conducted using IBM SPSS Statistics Version 24.
Sample Size Calculation
A-priori sample size calculation had shown that by including 12 intervention and 12 control teams, we would be able to detect a three- to four-fold increase in detection rate (a coefficient of the time × allocation interaction term of 3.55) with a significance level of 5% and a power of 80%. Effect size was based on the previous LARA-study (Trevillion et al., 2014), ICC was assumed to be 0.03 (Feder et al., 2011). The calculation assumed an average of 200 patients treated per team.
Results
Primary Outcomes
Inter-rater reliability between researchers during the screening process was found to be very good (Kappa = 0.86 95% CI 0.79-0.97). As shown in Table 2 and Figure 1, we found no increase in the either detection of DVA victimization (regardless of the type of victimization i.e., physical, sexual, emotional or material victimization) or of DVA perpetration in the BRAVE intervention teams. Similarly, we found no increase in DVA referrals in the BRAVE intervention teams. No indication was found of a shift in type of follow-up action between control and BRAVE intervention teams (Figure A.3 and Table 3).
BRAVE Intervention Effect on Rate of Detection and Referral of DVA: Estimated Mean Rate and 95% Confidence Interval At 12 Months After Start of the Intervention and Accompanying Regression Coefficients of the Time × Allocation Interaction Term.
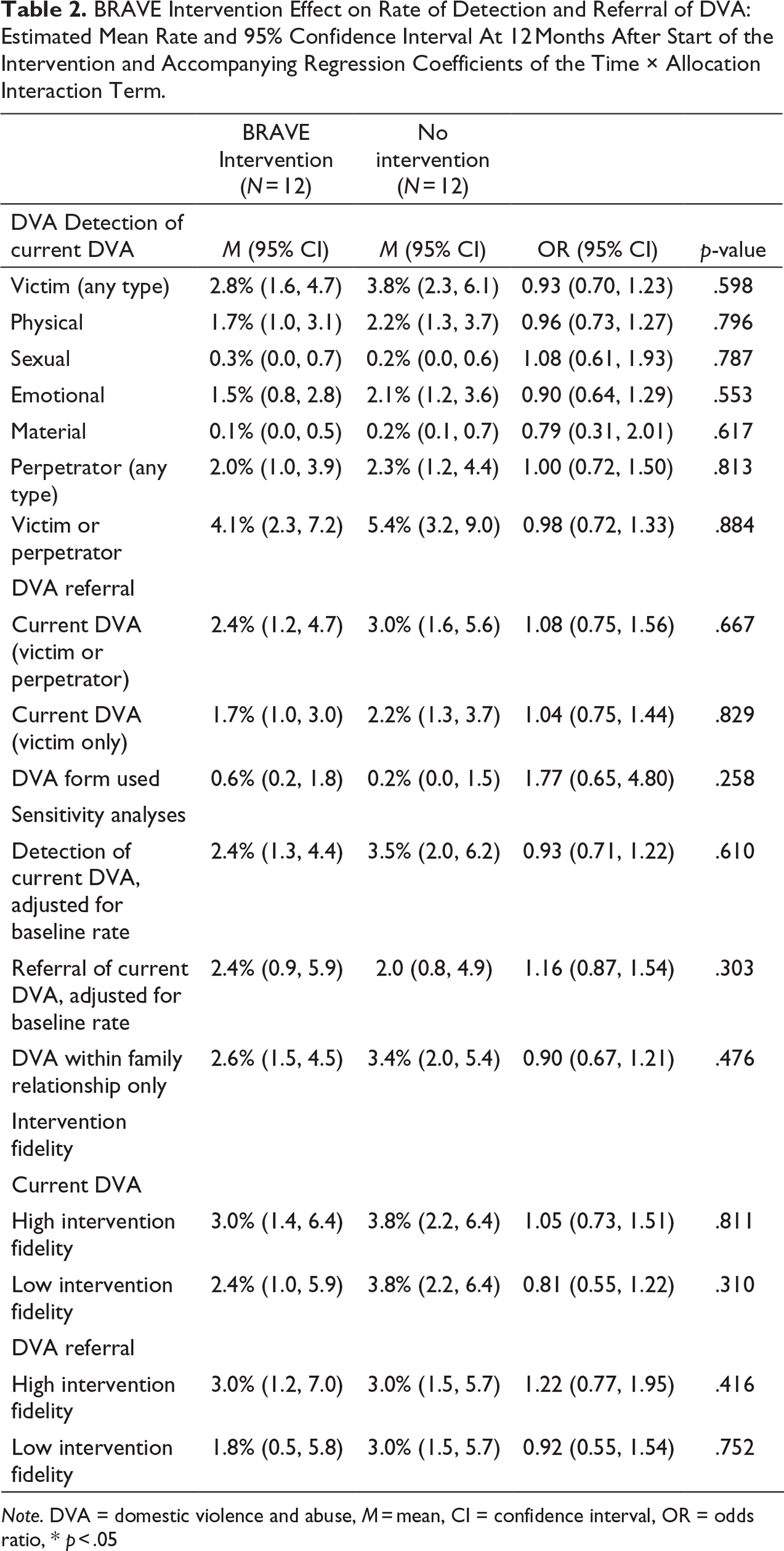
Note. DVA = domestic violence and abuse, M = mean, CI = confidence interval, OR = odds ratio, * p < .05
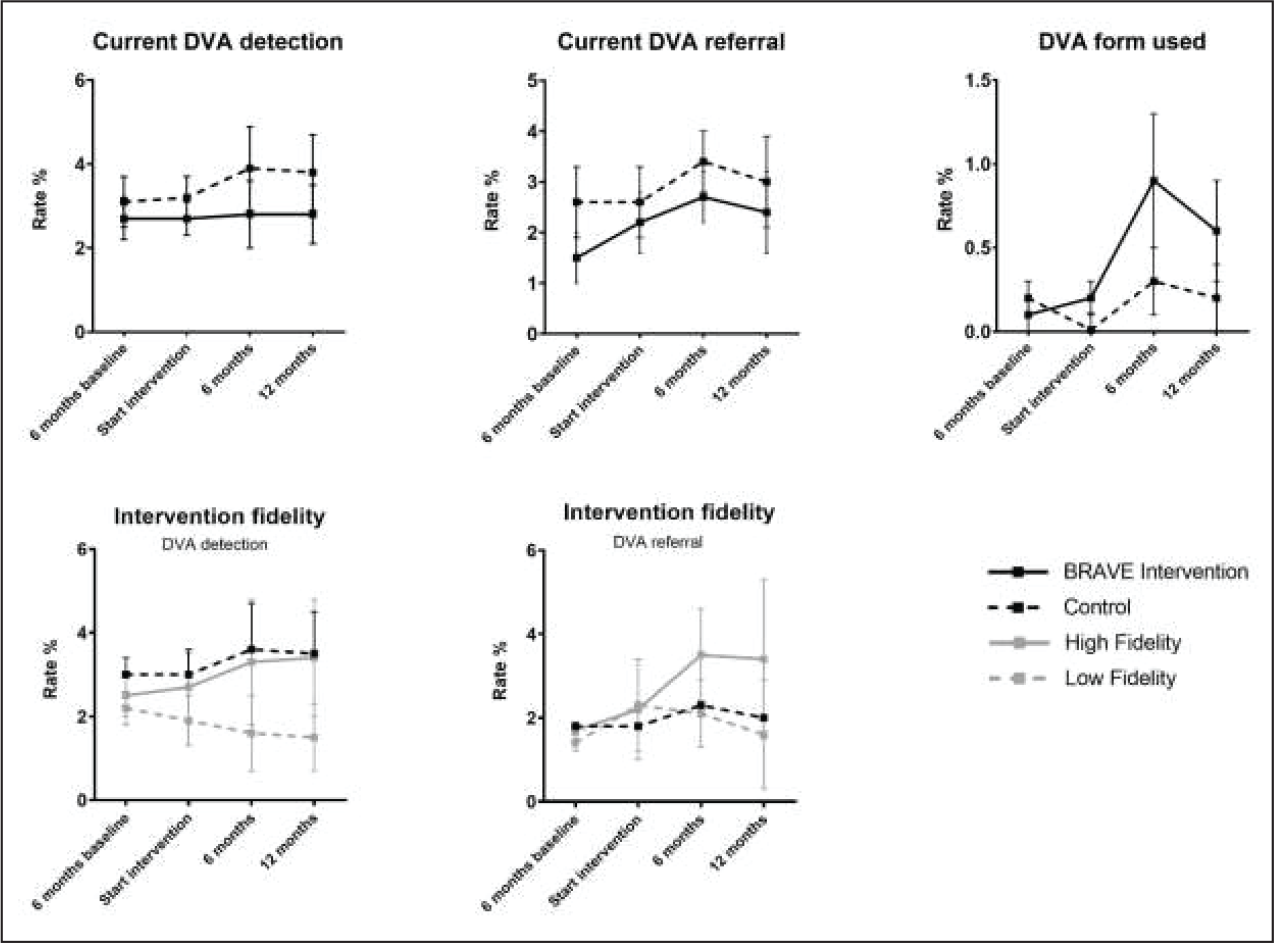
Note. Lines represent estimated means and standard errors.
Sensitivity analyses support the results of the BRAVE intervention (Table 2). The effect of the BRAVE intervention on DVA detection rate was slightly better in teams with better implementation of the intervention than in the teams where the implementation was less successful, although not significantly different than the control teams. Use of DVA forms was increased in the BRAVE intervention teams, but not significantly.
Secondary Outcomes
Table 3 and Figure 2 show the results of the BRAVE intervention on the clinicians’ readiness to manage DVA. As shown in Figure A.4, the overall response rate at all three time points was above 75%. The inflow and outflow of persons working in the included CMH teams during the follow-up period ranged from 12.5% to 22.1% for the intervention group and 15.5% and 15.7% for the control group. The BRAVE intervention resulted in a steeper and longer lasting increase in clinicians’ readiness to manage DVA. This effect was apparent from both the total BRAVE survey, and, separately, also on the subscales. We noticed an increase in readiness following the training, both in intervention and control clinicians. During the follow-up, skills, attitudes, and knowledge of the clinicians in the intervention teams continue to increase, while the skills and knowledge of the clinicians in the untrained teams stagnate after six months. Overall, effect sizes are small.
As expected, the results are more profound in the subset of trained clinicians. Results are similar for clinicians who completed the survey at all three moments, suggesting the absence of bias caused by the drop-out of various individual clinicians. Furthermore, we found that the impact of the BRAVE intervention on the BRAVE survey scores was similar for clinicians in the teams with high and low intervention fidelity. For information on the respondents: see A.4. and Table 1.
BRAVE Intervention Effect on Clinicians’ Readiness to Manage DVA: Estimated Means on Total Scale and Subscales of the BRAVE Survey At 12 Months After Start of the Intervention and Accompanying Regression Coefficients of the Time × Allocation Interaction Term.
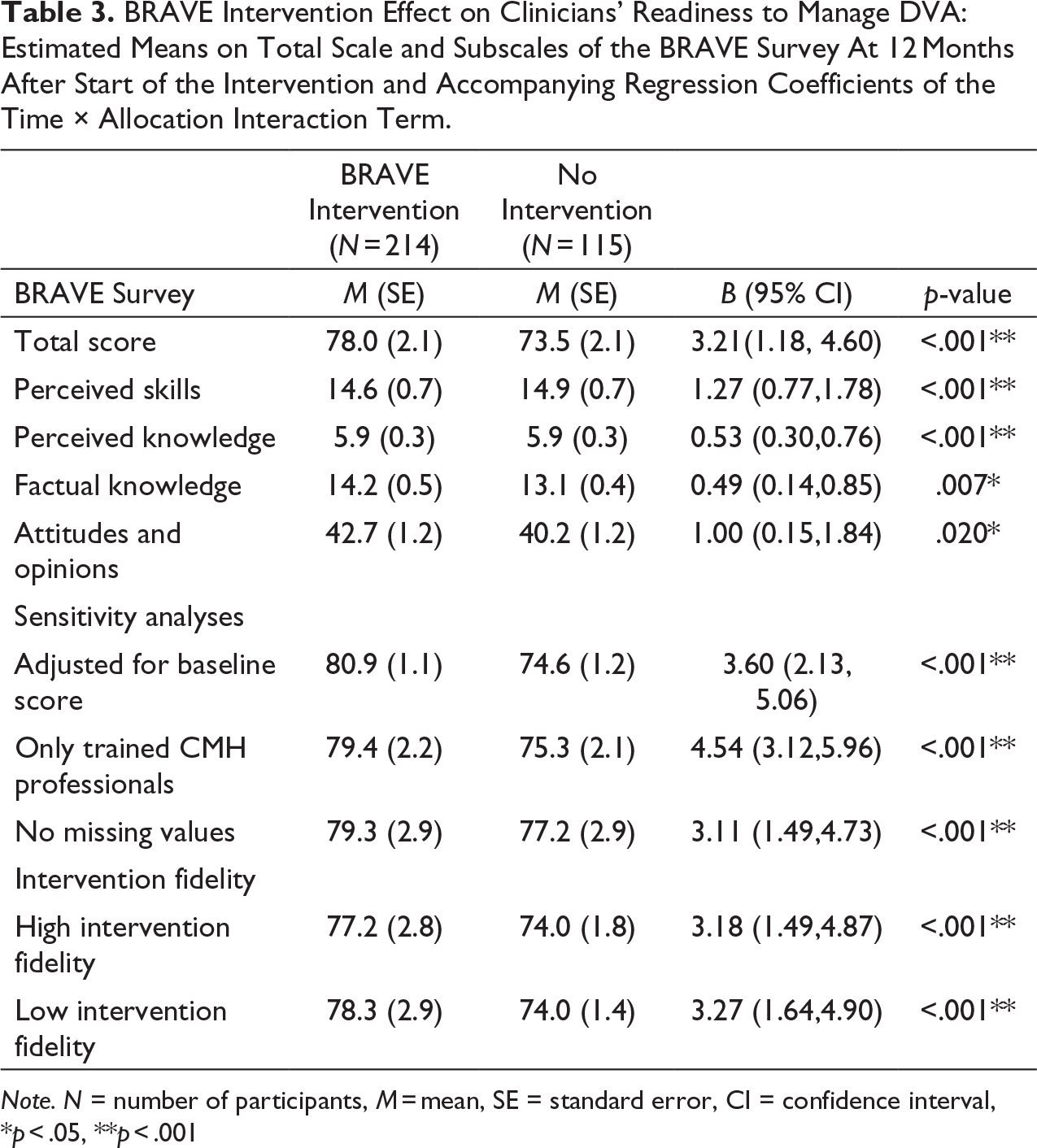
Note. N = number of participants, M = mean, SE = standard error, CI = confidence interval, *p < .05, **p < .001
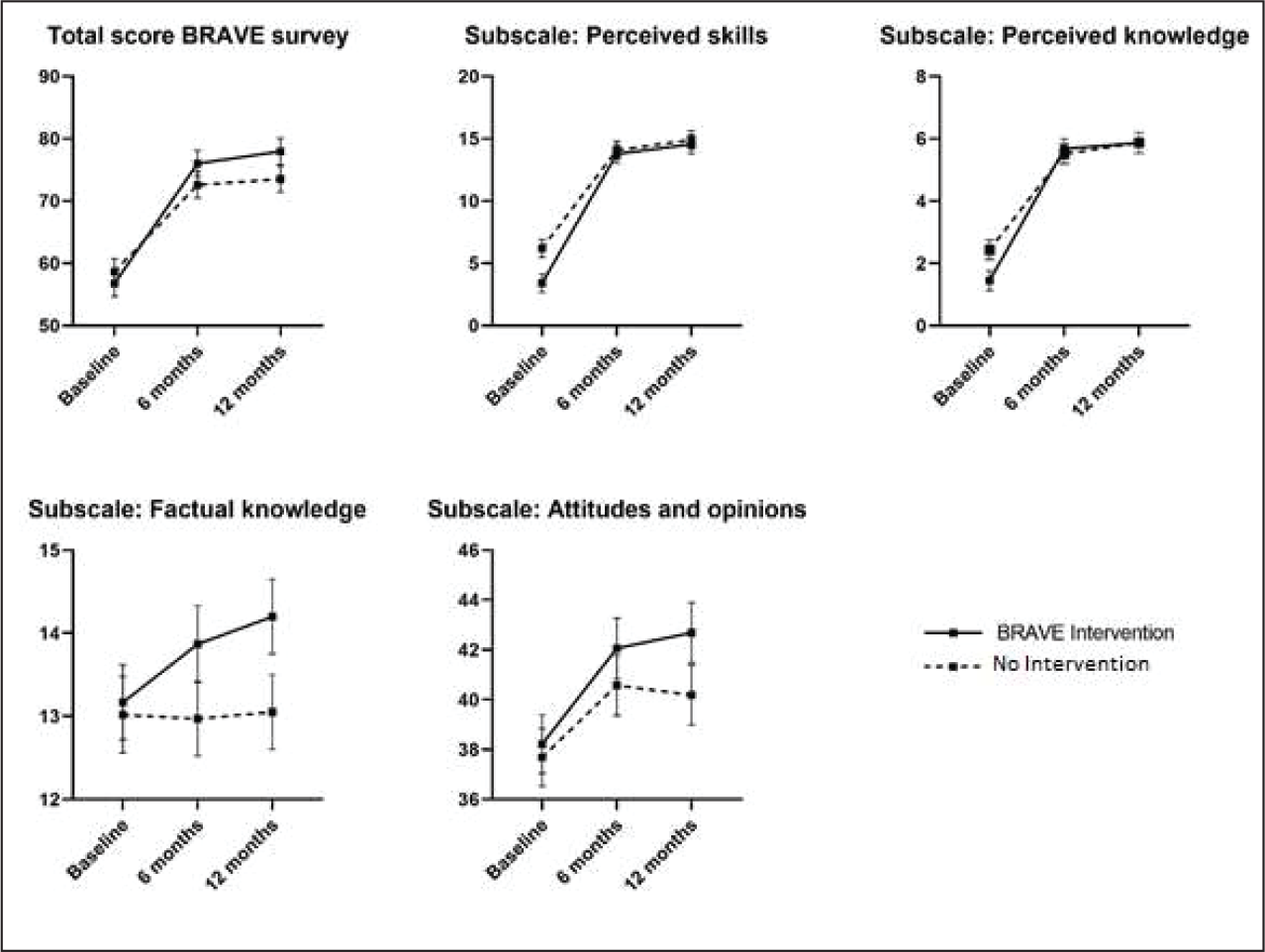
Note. Lines represent estimated mean scores and standard errors.
Discussion
This study evaluated the effects of the BRAVE intervention on DVA detection and referral rates of clinicians working in CMH teams. We found no increase in detection and referral rates of DVA, despite an overall increase in knowledge, attitudes, and skills related to DVA. This suggests that the increase in knowledge, attitudes and skills after the training was not accompanied by changes in clinicians’ behavior.
A systematic review by Zaher et al. (2014) suggested that interventions for improving detection of DVA have a higher chance to be successful when they consist of several components namely, (a) multidisciplinary reinforcement sessions for (CMH) clinicians; (b) additional training for DVA advocates; (c) cues or alerts for (CMH) clinicians reminding them to be alert for possible DVA; (d) improved access to support and to referral websites for DVA; and (e) continuous audits and feedback. The BRAVE study was able to provide multiple training sessions, send out newsletters to the CMH clinicians, provide CMH clinicians with cues or reminders to ask their patients about DVA, and provide information about referral websites or services but did not have the other components described earlier. These missing components provide initial pointers at why the intervention did not increase DVA detection.
Earlier studies showed the estimated prevalence of DVA among SMI patients to be 35% (Kamperman et al., 2014). We found a detection rate of only 3% in the intervention teams and 4% in the control teams. These numbers suggest a large degree of structural underdetection of DVA in CMH teams, despite the BRAVE intervention. Systematic screening for other problems or diseases (e.g., breast cancer, prostate cancer) often does systematically increase detection rates (Siegel et al., 2020). Clinicians have many reasons to be hesitant to detect DVA in their patients such as safety concerns for both the victim as the professional, not finding an opportune moment to discuss DVA, a lack of available shelters, or because no effective intervention to stop the problem of DVA after detection, is currently available in the Netherlands (Ruijne et al., 2020).
Another possible explanation for the absence of an intervention effect might have been the significant inflow and outflow of clinicians working in CMH teams (ranging from 30% to 34% over the full intervention period). This might have diluted the effect of the training course at the team level. Additionally, differences in management objectives and reorganizations within and between teams might have led to inconsistencies in the following three aspects of these teams: DVA management, the professional responsibilities of the CMH clinicians, and the number of DVA advocates. This could have resulted in a lack of therapeutic engagement, which is a known barrier for patients disclosing DVA (Rose et al., 2011). Unfortunately, only 8 out of 12 DVA advocates in the intervention group were advocates during the whole intervention period, resulting in a low overall score on the fidelity scale.
Our main results are similar to most intervention studies on the management of DVA in health care (Zaher et al., 2014). However, a similar intervention conducted in general practices in the United Kingdom (Feder et al., 2011) did find a higher detection and referral rate of DVA in the intervention group. Like the BRAVE intervention, this intervention also focused on contact with the patient instead of mandatory screening, and also implemented a DVA training course for clinicians. There were two key differences however: whereas the patients in the BRAVE study had contact with several professionals working in a team, patient contact in general practices is usually limited to contact with the general practitioner and, the BRAVE study focused on SMI patients while general practices focus on the general population. Diffusion of responsibility (Beyer et al., 2017) is thought to make people feel less responsible for the outcome of group decisions, and may cause communication problems between CMH teams and DVA professionals. Such hampered communication could lead to prolonged victimization of DVA or more emergencies in DVA.
Strengths and Limitations
This study has a number of strengths. First, this study was conducted according to the study protocol: the CMH teams included in the study cover a large and diverse part of the Dutch population; during the intervention phase, none of the teams dropped out; both attendance rates and response rates were high (> 75%); and the study was conducted in a naturalistic setting which means that the results are valid both internally and externally. Second, the extraction of data was done automatically by an independent researcher. All data were analyzed by two independent researchers. In theory, it is possible that some cases of detected DVA were missed. However, the initial data extraction was very broad, thereby limiting the possibility of false negatives. All cases were thoroughly assessed according to the highest standards, and this was reflected in the high levels of inter-rater reliability. Third, as researchers we did not intervene with the daily practice of the teams, ensuring the intervention to take place in a realistic setting.
A limitation with regards to the implementation was a high turnover of personnel during the intervention phase. This might have diluted the effect of the BRAVE intervention during the year of follow-up. A possible limitation that could impact that could impact the experience of the individual patient is that detection and referral rates were aggregated on a team level. Thus, it was no longer possible to attribute personal characteristics, such as the patient’s gender to the detected and referred cases. However, results from sensitivity analyses on these characteristics did not suggest an alternative explanation for the found nonsignificance. Furthermore, since we assessed the primary outcome on a team level, the impact of individual differences in skills, knowledge, and attitudes of DVA and individual differences with regards to behavioral change over the course of the intervention were masked. The training course for CMH clinicians took place only once, which was apparently not enough to establish a change in behavior. Although we took care in adapting the training to the specifics of DVA in CMH patients—such as gender differences and cultural differences—the possibility exists that not all practical circumstances of clinical care were addressed. However, no omissions were reported by the attendants and the training course for CMH clinicians was highly appreciated by the attending clinicians and met the standards for courses on DVA of the World Psychiatry Association (Stewart & Chandra, 2016, 2017). Although the clinicians’ knowledge and attitudes toward DVA victims did significantly increase in our study, a repeated training course and more support from a DVA professional in CMH teams might be needed to change clinicians’ behavior and to increase DVA detection and referral rates.
Another limitation was the fact that the BRAVE survey was a self-report questionnaire, not assessing actual behavior of clinicians in discussing DVA with their patients (Ruijne et al., 2019).
Not being actively involved in the teams as a research team could be seen as a limitation, as professionals may forget to ask about DVA if they are not reminded on a daily basis. However, if we want the BRAVE intervention to be successful in increasing detection rates, the effect of the intervention must be independent of RCT participation.
Clinical Implications and Conclusion
Our trial was successfully conducted but unfortunately failed to increase the DVA detection and referral rate in CMH teams, which highlights the great complexity of handling DVA in mental health care and the difficulties implementing interventions. Our results also suggest that improvements in cognition and attitudes do not necessarily correspond with improvements in behavior and skills (Miller, 1990). Although the BRAVE intervention was successful in increasing the clinicians’ knowledge, attitude, and skills toward DVA, we suggest that to increase DVA detection rates, a change in behavior or routine while working with patients is needed. For future research we recommend including more obligatory elements in such interventions, for example the screening of all new patients during intake interviews or adding a prompt in the EPF that reminds CMH clinicians to consider DVA in their assessments. We also strongly recommend improving the collaboration and information exchange between DVA services and mental health care. A structured way of improving communication and collaboration could be to add a DVA professional or physician employed by the DVA services to CMH teams or to add a CMH clinician to DVA services, which could add flexibility and enables CMH teams to provide care aimed at the specific needs of the individual.
Our trial showed that a training program on DVA knowledge, attitude, and skills in CMH teams can increase knowledge, attitudes, and skills toward DVA. However, our intervention does not appear to increase the detection or referral rate of DVA in SMI patients.
Supplemental Material
Supplemental material for this article is available online.
Supplemental Material for Detection of Domestic Violence and Abuse by Community Mental Health Teams Using the BRAVE Intervention: A Multicenter, Cluster Randomized Controlled Trial by Roos Ruijne, Cornelis Mulder, Milan Zarchev, Kylee Trevillion, Roelvan Est, Eva Leeman, Willemien Willems, Mark van der Gaag, Carlo Garofalo, Stefan Bogaerts, Louise Howard, and Astrid Kamperman, in Journal of Interpersonal Violence
Footnotes
Acknowledgment
The authors would like to thank Harriet Gussinklo who was one of our raters of the electronic patient files. We would like to thank Femke Jongejan for her contribution in maintaining contact with the community mental health teams and retrieving all BRAVE survey data. We would like to thank Karima Baddi for assessment of the intervention fidelity scale and Katinka Lünnerman for her contribution to the design of this study. We also thank Milan Zarchev for reviewing this article. Lastly, we thank all participating community mental health teams for participating in the study.
Declaration of Conflicting Interests
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article:
This project received a grant from the Netherlands Scientific Organization: Grant number: 432- 13-803. The funder of this study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. RR and AM had full access to all the data in the study and had final responsibility for the decision to submit for publication.
LM. Howard reports grants from NIHR, grants from UKRI, personal fees from NICE Scientific Advice, during the conduct of the study; K. Trevillion’s funding during this work was from the National Institute for Health Research (NIHR) under its Programme Grants for Applied Research Programme (Grant Reference Number RP-PG-1210-12002) and is currently funded by the NIHR Policy Research Programme and the NIHR HS&DR Programme (Grant Reference Number 16/117/03); R. Ruijne, CL. Mulder, R. van Est, E. Leeman, W.Willems, M. van der Gaag, C. Garofalo, S. Bogaerts, AM. Kamperman have nothing to disclose.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
