Abstract
The eye is an essential sense organ and drug delivery to the eye is a challenging task due to protective barriers that hinder drug penetration. Over 90 percent of treatments for eye diseases are topical, but frequent administration over extended periods can lead to toxicity and compliance issues. Over the years, extensive research has been aimed at developing drug delivery systems that enhance drug bioavailability at the target site while minimizing side effects. Innovative drug carrier systems have been researched and developed to extend retention time, decrease administration frequency, improve therapeutic efficacy, and ensure biocompatibility. In this article, we delve into the various ocular barriers affecting drug delivery and provide an overview of the utilization of biomaterials and nanotechnology in ocular drug delivery. We explore its applications in the treatment and management of various diseases affecting the anterior segment of the eye.
Keywords
Introduction
The eye, a vital organ of the human sensory system, plays a crucial role in vision. Ocular diseases are widespread across the world; affecting nearly every individual at least once during their lifetime. Forecasts suggest a sharp escalation in the global demand for eye care services in the coming decades, presenting a substantial challenge to healthcare systems. According to the 2019 World Health Organization (WHO) report, approximately 2.2 billion individuals globally suffer from vision impairments, of which at least one billion cases remain unattended. 1
Etiological factors and implications for anterior segment.

Conventional drug delivery approaches and their limitations.

The foremost region of the eye is referred to as the anterior segment encompassing parts of the eye like the conjunctiva, cornea, sclera, aqueous humor, iris, pupil and lens (Figure 1). Anterior segment diseases include conditions such as dry eye, glaucoma, conjunctivitis, cataract, and corneal opacity, among others.
3
Layers of the tear film (A), layers of the cornea (B), the anterior segment of the eye (C), anatomy of the eye (D) [Created using BioRender.com].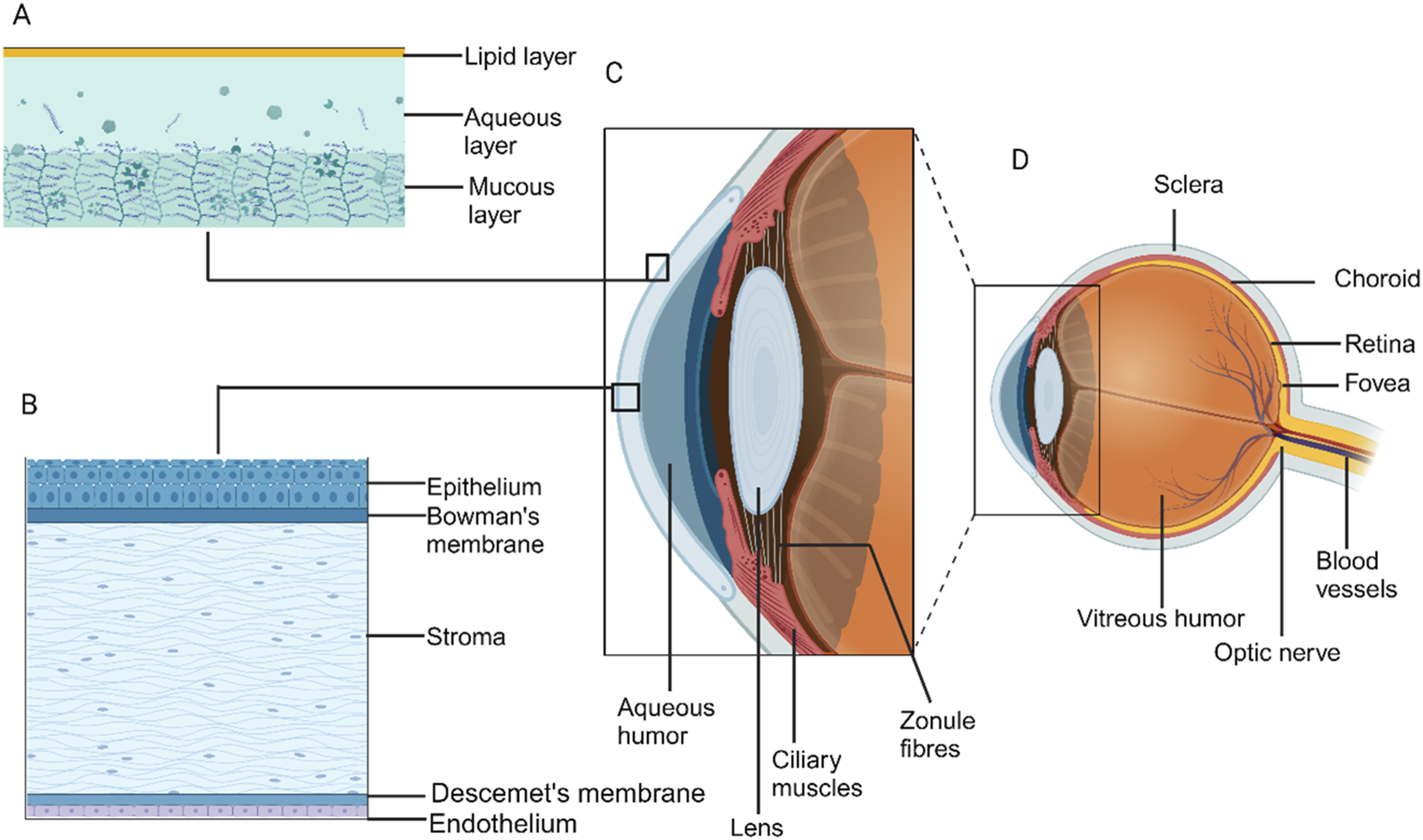
The eye is shielded from internal and external factors by an array of barriers, broadly categorized as dynamic, metabolic and static (Figure 2).
4
The successful delivery of drugs to the disease sites within the anterior segment of the eye necessitates overcoming one or more of these ocular barriers, depending on the chosen route of administration.
5
Conventionally anterior segment diseases are resolved mainly using topical (around 90%) application of medication but sometimes need intracameral or subconjunctival injections. Conditions like cataract require surgical intervention.
6
It must be mentioned that only 5% of the drugs permeate through the layers of the eye on topical application.7,8 Other routes like intracameral, intrastromal and subconjunctival injections require the aid of medical professionals for administration. Complications associated with invasive routes may include side effects such as endothelial cell loss, cystoid macular edema, toxic anterior segment syndrome (TASS), endophthalmitis, among others.
9
Frequent and prolonged administration of ocular medications significantly affects patient compliance. This is especially so when patients require assistance with administration thus influencing the treatment efficacy. Barriers in ocular drug delivery [Created using BioRender.com].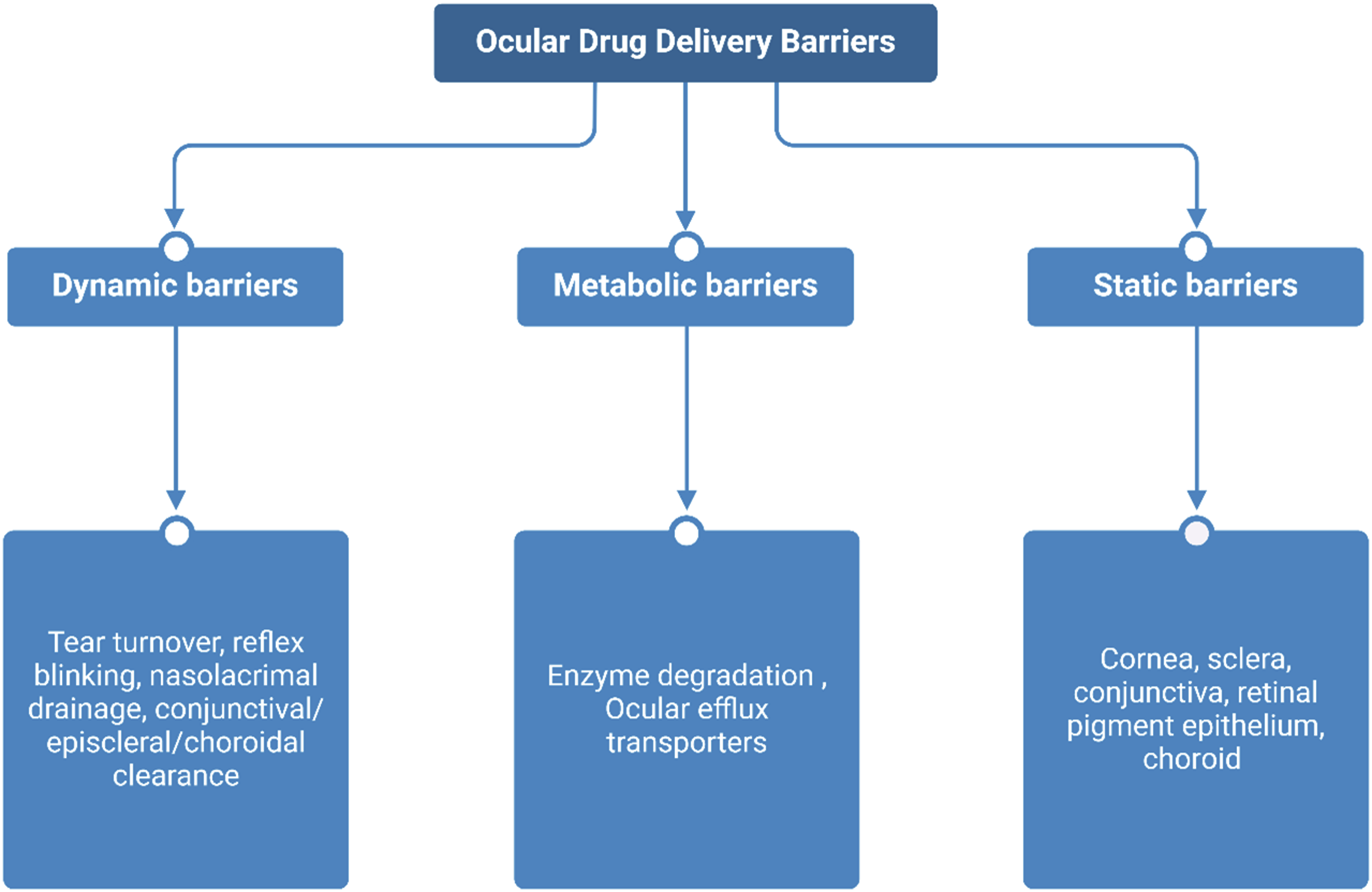
The therapeutic efficacy of a drug can only be achieved if appropriate drug concentrations reach the intended site. Given the various ocular barriers, it was necessary to devise a more efficient drug delivery system, which can reduce toxicity and frequency of application, improve bioavailability, minimize irritation and ocular tissue damage.5,6,9,10 Efforts were also directed towards enhancing drug solubility and permeability into ocular tissues. 9 Various methods to improve drug delivery have been tested over the years. In 1945, Swan proposed a method to improve the contact time and penetration of drugs into the eye using water-soluble synthetic polymer like methyl cellulose. 11 Nanotechnology-based alternative methods for drug delivery have been evolving since the 1960s. 12 The use of nanotechnology for drug delivery and diagnosis has led to the rise of the field called nanomedicine. 13
Biomaterials play a crucial role in drug delivery by enabling cyclic dosing, controlled release of hydrophilic and hydrophobic drugs, and sustained therapeutic effects. 14 Because of their capacity to degrade, they have become attractive options for ocular drug delivery systems (DDS). This allows for precise and continuous release of drug at the intended location by their controlled degradation. Furthermore, the intrinsic qualities of biodegradable materials-like their biocompatibility, flexibility, and formulation versatility-align well with the special demands imposed by the structure and function of the eye. They enhance drug stability, prolong ocular residence time, improve bioavailability, reduce dosing frequency, and enable targeted delivery, overcoming ocular barriers. 15
The biodegradation process involves the breakdown of materials via enzymatic or nonenzymatic mechanisms. One clear benefit of utilizing these materials to produce nanocarriers is the elimination of the need for surgical removal post clinical application. The physicochemical characteristics of these materials, including their molecular weight, charge, hydrophobicity, and functional group composition, render them suitable materials for a diverse array of applications. 16
The biomaterials are used for a variety of applications including preparation of nanoparticles (NPs), lens, intra ocular lens (IOL), implants and preparation of artificial corneas. 17 The drug delivery can be achieved by either formulation method or physical force-based techniques. The formulation-based technique uses chemical enhancers, prodrugs and carrier-based drug delivery like using contact lens, nano/microparticles, punctal plugs, hydrogels, amniotic membrane and so on.3,18,19 These systems act as drug reservoirs enabling a sustained drug release for a period which can be manipulated during the preparation of these carriers. This in turn prolongs the drug retention in the eye, protects the drug from degradation, reduces toxicity and frequency of application.9,10 In the recent years, this concept has evolved to provide targeted drug delivery and gene therapy. 20 In physical force technique, power generating devices are used to deliver energy such that the barriers are breached, and drug transport is enhanced. These include the use of electrical fields, sonophoresis and microneedles. But with the ocular tissues being very sensitive, the use of external energy source can also damage the eye if not controlled properly. 18
In the past 30 years researchers have also ventured into the application of 3D printing in drug delivery. 21 This innovative technology traces its origins to the early 1980s when Charles Hull introduced the first 3D printer, initially known as ‘stereolithography’. 22 In the pharmaceutical field, this technology enables the creation of drug carriers with unprecedented flexibility in both internal and external geometries, along with precise spatial drug distribution. 23 Its potential was notably recognized in 2015 when the Food and Drug Administration (FDA) approved Spritam®, the first 3D-printed drug, developed by Aprecia Pharmaceuticals. 24
This review provides a concise overview of the causes of the ocular barriers to drug delivery and examines the application of biomaterials as carriers in managing various anterior segment diseases.
Ocular barriers in conventional approach for anterior segment conditions
Topical
Drugs that are administered topically are typically made into eye drops, ointments and gels. The easiest, most practical, self-administrable, non-invasive, and most popular drug administration technique for treating anterior segment diseases is topical administration. 7 The blood-aqueous barrier, adverse effects due to injections, and first-pass metabolism that may occur with some systemically administered medications are all avoided by this local drug delivery approach.5,6 The above-mentioned factors and the ease of self-administration at home has led to topical application of drugs as one of the most common and favourable routes of administration of drugs for anterior segment diseases. 6 The drug molecules permeate through the cornea through paracellular and transcellular pathways. 25 But a matter that must be taken into consideration is that it has been reported that only 5% of the topically applied drug reach the aqueous humor.7,8 This is attributed to ocular barriers like tear film, layers of the cornea, nasolacrimal drainage and conjunctiva. Due to reflex blinking and fast tear turnover rate which is about 2 µl/min, there is significant wash off from the administered drugs.4,8 A drop of drug solution is about 50 µl, but the eye can only hold a maximum of 30 µl without spillage. 26 This leads to a substantial loss in the initial amount of drug that was administered. The corneal layers acts as a barrier to drug entry, depending on the properties of the cells in its various layers. The corneal epithelium acts as a limiting layer for the passage of hydrophilic agents while a hydrophilic environment is present in the stroma. The presence of sialic acid residues on the surface of the corneal epithelium gives it a negative charge which leads to better penetration of positively charged molecules across it.3,9,27 The corneal endothelium is also a lipophilic layer, but the movement of molecules is more dependent on its size than molecular nature. 28 The paracellular pores of the corneal epithelial cells have a porosity of approximately 1.8 nm and only molecules which are less than 500 Da can diffuse through these pores. 18 These properties of the corneal layers hinder the transport of both lipophilic and hydrophilic molecules. The presence of tight junctions in the conjunctiva hinders the paracellular transport of molecules larger than 40 kDa. 4 Another difficulty of topical route of administration is the drainage of hydrophilic drugs from the conjunctival sac through lacrimal drainage into the nasal cavities which is later eliminated systematically. 4 The movement of drug particles across the sclera is subjective to the size of the particle and hydrophilic molecules can diffuse across it more efficiently. 29 The presence of the negatively charged proteoglycan matrix on the sclera limits the diffusion of positively charged molecules due to binding effect. 18
This means, there is a need for higher concentration of the drug which must be administered at a higher frequency when applied topically. Common topically applied drugs include antibiotics, antifungals, antivirals, lubricants, anti-inflammatory and anti-glaucoma drugs. Prolonged application of drugs is not without possible side-effects and the overall recovery is affected due to non-compliance to the treatment regimen by the patients. 6
Intracameral
This route of drug delivery involves injection of drug directly into the anterior chamber of the eye. This method overcomes many of the barriers mentioned in topical route of administration. 20 It has shown to have drug levels in the aqueous humor that is 300–600 times more than in case of eye drops. 5 Drug groups applied through this route include antibiotics, steroids, anti-glaucoma, anaesthetics and so on. But it must be considered that due to the aqueous humor turnover, there is rapid clearance of the drug. Furthermore, commercial preparations for intracameral use are limited and lack regulatory approval. The rapid clearance of the therapeutic agents necessitates multiple injections. Intracameral administration of drugs is prone to side effects like toxicity to the endothelium of the cornea and TASS, should the drug formulations and dosing be incorrect.30,31 Other complications including corneal edema, retinal detachment and macular edema have been reported but are rare. 32
Subconjunctival
This route involves administration of drugs into the subconjunctival space. Sclera is easier to penetrate and has a surface area much higher than that of the cornea. 27 The administered drug will permeate through the sclera to reach the anterior segment. It can bypass the corneal barriers and the wash out due to tear turn over. 20 But the drawback of this method is the drainage of the applied drugs due to the presence of blood and lymphatic vessels in the conjunctiva where it circulates to various parts of the body. This means that very minor amount of the drug taken will be able to reach the anterior segment. 5 This is further limited due to the presence of blood-aqueous barrier because of which higher doses or increased frequency of administration of drug will be required to achieve therapeutic effect.
Intrastromal
In intrastromal route of drug delivery, the drug administration is made directly into the stroma which is the middle layer of the cornea. This route is mostly utilized for the delivery of antimicrobial drugs due to their poor penetration through cornea. 33 This method also helps to overcome the barriers observed in topical application of drugs and increase the bioavailability of the drugs. The main limitation of intrastromal injection, especially in cases of keratitis is the potential spread of infection through the cornea. Additionally, complications such as unintended anterior chamber entrance, intrastromal bleeding, and Descemet membrane detachment have been documented, although these are rare. 34
Systemic administration
This route of administration involves drug intake through oral, intravenous and intraperitoneal routes. This method is rarely preferred for ocular drug delivery due to lower bioavailability at target site and increased dosage of drugs that need to be administered to obtain therapeutic concentrations can risk in systemic toxicity.5,20 The blood aqueous barrier hinders transport of molecules due to the presence of tight junctions in the endothelial cells of the iris and non-pigmented cells of the ciliary body.
35
Although the ocular bioavailability of systemically administered drugs is less than 2%, there is a need for frequent administration of drugs, it is more widely accepted by patients compared to other routes (Figure 3).36,37 Nano drug delivery systems (A), Conventional routes of ocular drug delivery (B) [Created using Biorender. com].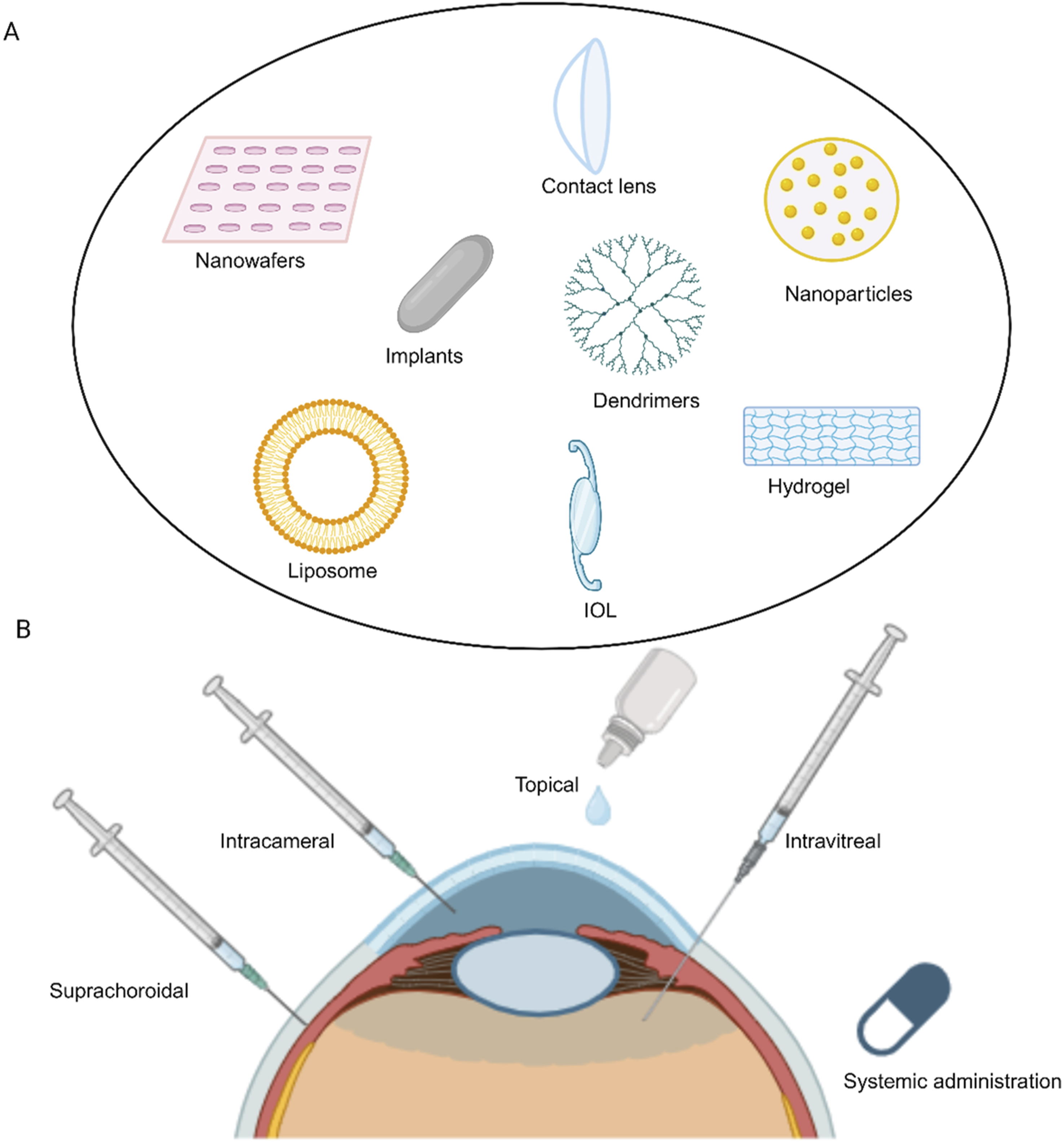
Nanotechnology based drug delivery systems
Biodegradable polymers
Because of their incredibly strong and difficult to break carbon backbones, biodegradable polymers frequently begin their breakdown at the end groups. 38 The biodegradability of a polymer seems to be higher when it has both hydrophilic and hydrophobic part than when it simply contains one of these configurations. The assessment of biodegradability can be determined using several general criteria. For instance, biodegradability is reduced when factors like size of crystalline domains, hydrophobicity, and molecular weights of macromolecules increase. 39
Key characteristics of biodegradable materials include, (a) produce nontoxic degradation products which are easily eliminated; (b) does not cause a prolonged inflammatory response; (c) have appropriate permeability and processability; (d) have appropriated mechanical properties for their intended use; and (e) have a degradation time that corresponds with their function.40–42
When drug encapsulated polymer DDS are used, the biodegradable polymers are broken down by the body into non-toxic components, causing sustained drug release. This makes it possible to deliver drugs without the requirement of manual removal. This eliminates the need for the implants to be surgically removed, which is especially relevant in the development of ocular implants. 43 Because of their biocompatibility, both synthetic and natural biodegradable polymers are widely used in drug delivery for variety of purposes like enhanced drug delivery, 3D printing and tissue engineering. They are more effective and cause less adverse effects by increasing the drug loading at the site of disease and lowering systemic drug concentration. 44 While nanotechnology is being utilized to generate many novel formulations, polymers are being used to improve and optimise the already existing formulations.
It is common practice to employ many polymers in a single composition. This is done to overcome the drawbacks that a particular polymer may have. For example, chitosan shows low elasticity and weak mechanical strength when incorporated into hydrogel, but a better hydrogel is created by adding another polymer like PVA. 45 Polyelectrolyte complexes (PECs) can be formed by several polymers. These arise from the interaction of a negatively charged polymer, like alginate, with a positively charged polymer, like chitosan, to form a cross-linked system. These complexes can later be used to create nanoparticles and used for drug administration. 46 Some polymers have characteristics to alter viscosity along with their mucoadhesive qualities. But excessive viscosity can irritate eyes, and it can respond defensively, which increases tear production, and the formulation will be eventually removed. 47
Some polymers are stimuli-responsive, which means that when their environment-such as temperature, pH, or pressure-changes, they can respond or release the drug. These “smart” polymers are used as in situ gelling systems. This allows the formulation to stay longer at the site, giving more time to penetrate the cornea.48,49 These bio responsive polymers can change in a variety of ways including, phase separation, permeability changes, and shape modifications. As a result, the drug can be released from the formulation as needed. 50
Surface area, hydrophilic and hydrophobic nature, molecular weight, glass transition temperature, melting point, elasticity and crystalline nature of the polymers significantly affect biodegradation process. Polyesters without side chains often degrade more readily than those with side chains. Molecular weight has a significant impact on biodegradability of the polymer since it dictates many of their physical characteristics. In general, a polymers biodegradability declines as its molecular weight increases. Moreover, polymer morphology has a significant impact on biodegradation rates. The degree of crystallinity is another important element influencing biodegradability since enzymes primarily target the amorphous regions of a polymer. This is due to the loosely packed molecules in the polymers amorphous portion, which increases its susceptibility to breakdown. However, molecules are packed closer together in the crystalline portion of the polymer, it is more resistant than amorphous portion. The enzymatic breakdown of the polymer is also significantly influenced by its melting temperature. A higher melting point of the polymer results in less biodegradation. Biodegradability decreases with increase in molecular weight, hydrophobicity, and crystallinity. A polymer with both hydrophobic and hydrophilic components is more biodegradable than those that have either one of them. Synthetic polymers made up of long repeating units are less likely to be crystallised and hence may be biodegradable.
Natural polymers
Chitosan
Chitosan, a polymer produced from chitin, is frequently used in ocular drug delivery because of number of advantageous qualities: (a) mechanical and biodegradable characteristics that are easily controlled. (b) chemical side groups that can be attached to other molecules. (c) a lower level of foreign body response. (d) simpler processing conditions ,whereas synthetic polymers frequently require dissolution in harsh chemicals, chitosan will dissolve in water bases on pH. 42 Due to its cationic nature, the polysaccharide can interact with the negative charges present in the conjunctiva and mucus of the eye. Chitin is only soluble in acidic solutions, causing irritation of the eye, if it were put in the eye without totally neutralising it. As a result, several derivatives with better solubility profiles have been developed including galactosylated and thiolated chitosan.51,52
Among its many advantageous qualities are its biocompatibility, mucoadhesion, non-cytotoxicity, and biodegradability.53,54 Chitosan has been demonstrated to extend the retention period and enhance drug penetration through cornea by increasing the space between tight junctions. 45 Due to its intrinsic antimicrobial and wound healing qualities, chitosan is also being employed in a variety of new ocular formulations. 55
Chitosan and its derivatives are being used in various nano formulations like, nanoparticulate system for glaucoma, conjunctivitis, and immune related ocular degeneration.55–57
Hyaluronic acid (HA)
A naturally occurring polymer, HA is present in many parts of human body, including the eye. 58 It is a negatively charged polysaccharide. 59 Because it is mucoadhesive, biocompatible and biodegradable, it has been researched for application in ocular delivery systems. It is frequently combined with other polymers such as chitosan. 60 HA and chitosan can be used to deliver ocular gene therapy to corneal and conjunctival cells. 61 Like chitosan, HA is frequently altered, and its derivatives are used in tissue engineering and other medical applications in addition to drug delivery. Because of its mucoadhesive and viscosity modifying qualities, it has been utilised to extend the retention period of numerous ocular ingredients. 59
It is possible to functionalize HA to change its characteristics in a formulation. These functionalized HA were combined to prepare hydrogels which could be loaded with a drug directly or with drug containing liposomes. These provided sustained drug release. Therefore, in addition to the biocompatible and biodegradable qualities of HA, it can be used to optimise drug distribution to eye resulting in notably better pharmacokinetic properties.62,63 Micelles made from HA have recently been demonstrated to both decrease the rapid clearance rate and improve penetration of lipophilic drugs like dexamethasone. 59
Alginates
In ocular formulations, alginates-naturally occurring anionic polysaccharides-are primarily utilized for their gelling properties, which enable viscosity adjustment. They are easily processed in water and are relatively non-toxic and noninflammatory. Because of its increased permeability, biodegradability and biocompatibility, it is frequently used in ocular preparations. Nevertheless, it is highly vulnerable to enzymatic breakdown. To make sodium alginate perform as intended, it can be modified with other polymers, such as PLGA. 64
Sodium alginate is widely used because it is mucoadhesive and permits prolonged drug release, in addition to changing formulations viscosity. This helps in resolving some of the issues encountered by topical formulations.65,66 Chitosan coated alginate nanoparticles result in superior drug release profile compared to nanoparticles containing only chitosan. 67
Carboxymethylcellulose (CMC)
CMC has been demonstrated to have thermoresponsive qualities. Because of their capacity to alter viscosity, cellulose derivative polymers are frequently used in ocular treatments. It has been demonstrated that methylcellulose has the capacity to heal wounds in the eyes and to act as tear substitute.68,69 A polymeric membrane made up of sodium CMC and PVA was studied in ophthalmic formulation by Jain et al. PVA increased membrane stiffness, while CMC gave the composition its mucoadhesive and biodegradable qualities. 70
Synthetic polymers
Poly (lactic-co-glycolic acid) (PLGA)
It has been extensively studied for tissue engineering, drug delivery and biodegradable sutures mostly because of its biocompatibility, capacity to breakdown in an aqueous media, and sustained release profiles. A variety of techniques like solvent evaporation and nanoprecipitation, have been used to develop it into nanostructures. Copolymers such as PEG, can be used to modify PLGA to improve its properties. PLGA-PEG nanoparticles developed by Vasconcelos et al. demonstrated high entrapment efficiency, sustained drug release and low toxicity. 71 PLGA coupled to other polymers has shown promise in ocular drug delivery thus far, with uses ranging from enhancing drug penetration to increasing the residence period of nano systems.
Poly (ɛ-caprolactone) (PCL)
Due to its great biocompatibility, biodegradability and its capacity to improve drug release profile, PCL is widely employed in drug delivery. Compared to PLGA, PCL degrades slowly and has lower toxicity when formed into thin films and polymer solutions. 72 Its degradation rate depends on degree of crystallinity and molecular weight. Biodegradability of PCL can be increased by copolymerization with polyester. 42 It is used in systems like implants, where the drug is expected to be released gradually over a minimum period of 4 months. Studies on PCL have revealed that it increases the drug concentration in aqueous humor when combined with nanoscale drug delivery, such as nanoparticles. 73 Additionally, Da Silva et al. have described the creation of PCL nanofibers loaded with dexamethasone acetate could be given intravitreally. 74
Poly (ethylene glycol) (PEG)
PEG is a non-ionic, hydrophilic, synthetic polymer, used both alone and in combination with another polymer like PLGA to create DDS. It has been demonstrated that PEG improves mucoadhesive qualities of nanoparticle formulations. 71 Shi et al.’s study used diclofenac nanosuspension made of methoxy PEG-PCL and chitosan. Compared to conventional eye drops, this formulation showed higher concentration of the drug in aqueous humor, increased pre corneal retention time, and improved penetration of the drug across the epithelial barrier. 75 PEG has been utilised to increase permeability of nanoparticles, hence improving their performance. Additionally, it contributes to the stability of nanoparticles by preventing agglomeration. 76
Poly (2-hydroxyethyl methacrylate) (pHEMA)
Using contact lenses made of pHEMA-based hydrogel to deliver therapeutic medications to eyes is a promising way to treat intraocular conditions like glaucoma and uveitis as well as eye infections such as keratitis, blepharitis and conjunctivitis.77,78 Soft, flexible, and extremely oxygen permeable soft contact lenses made from hydrogels based on pHEMA offer patients comfort and safety while undergoing ophthalmic treatments. A tiny layer of tear is formed when a drug loaded pHEMA-based hydrogel lens is placed on the cornea, which allows the diffusion of the drug from the lens. 79 In vitro tests were carried out by Li and Chauhan in 2007 to administer timolol maleate using hydrogel contact lenses based on pHEMA. 80 According to them, the cornea will receive at least 20% of the drug present in the lens. When compared to conventional topical dosing, this bioavailability is significantly higher.
Nanoparticles and microparticles
NPs and microparticles can be made with a variety of materials like synthetic and natural polymers. The examples of polymers explored include PLGA, PLA, PCL, HA and chitosan. 81 These are biodegradable and biocompatible making them ideal for use in drug delivery systems. Their large surface area and accessibility for attachment of surface functional groups makes them a front runner for preparation of drug delivery systems. 5 It must be understood that the penetration of NPs across the cornea on topical application be dependent on various characters which must be taken into consideration during the preparation process. A size range of 50-400 nm is generally preferred for ocular drug delivery. 82 Smaller particles are preferred as they can penetrate the mucin layer of the eye and causes less irritation. 9 The size of the particles is preferred to be below 10 µm to aid penetration and avoid irritation on topical application. 10 Particles of about 100 nm are internalised by the corneal cells mainly through receptor-mediated endocytosis. 7 Most polymeric NPs will show a biphasic release with an initial burst release of the loosely bound drug followed by sustained release of drug over a period. NPs are known for their stability and most of the prepared formulations have shown to have high ocular tolerance. 10 They are often coated with other materials for mucoadhesion and improving retention time. 6 The metals like gold and silver are known for their anti-inflammatory, antioxidant and antibiotic properties and their NPs are known to easily penetrate the ocular tissues.83,84
Cell-penetrating peptides are positively charged sequences of less than 30 amino acids and are rich in arginine. These when incorporated on the surface of NPs enhance their penetration through the cornea using mechanisms like direct translocation or receptor-mediated endocytosis. 85
Contact lens
These are concave shaped, fragile and transparent devices initially used to correct refractive errors in the eye. Since first used in 1965 by Sedlacek, they have been extensively studied as carriers for drug delivery.86,87 The contact time between the drug solution and ocular tissues is about 1-2 min while using eye drops leading to almost 90% of the applied drug being lost. 3 The use of contact lens improves the bioavailability of the drug to up to 50% compared to while using eye drops. 88 Therefore, idea of using contact lens is to incorporate drugs into the contact lens to provide drug delivery with an increased retention time and do so without altering the lens optical and physical properties. 89 These can be self-administered by the patients. Commonly used materials to prepare these contact lens include silicone, 2-HEMA, polymethylmethacrylate and N-Vinylpyrrolidone. 87 The methods of incorporation of drugs onto the lens could be by soaking, inclusion of semi-circular rings containing drugs, NPs, molecular imprinting and so on.88–90 The use of chitosan in preparation of contact lens helps improve residence time of the drug as the NH2 group present in the chitosan binds strongly with the mucin anionic groups. 91 Some studies have used Vitamin E incorporated contact lens to extend the drug release from the lens while hyaluronic acid is used to hydrate and reduce irritation to the eye. 89 Contact lens prepared by soaking method have exhibited a short release period. Incorporation of NPs into contact lens help increase the retention time and penetration of the drug. This is possible because the drug is continuously released from the NPs and then into the lens matrix by slow diffusion.3,88
Some drawbacks of using contact lens are the need for frequent replacement of the used lens, some formulation methods may alter the properties of the lens and there have been cases where the patients have reported ocular infections. 89 These matters must be taken into consideration and tackled while preparing and using contact lens as drug delivery devices. The ion and oxygen permeability will be affected with decrease in water content which in turn will affect the durability of the lens. 88
Implants
Ocular inserts are solid dosage forms designed for administration in the conjunctival sac, lacrimal punctum, cornea and so on. These sustained-release systems minimize dosing frequency and reduce the risk of dosage form loss from the application site. 92 Polymers are frequently utilized in implantable devices because of their natural properties, including biocompatibility and biodegradability and are usually the main components of implantable devices. 93 Implants are solid drug delivery devices that can be inserted into regions of the eye like punctum, conjunctival sac, capsular bag and so on. These devices help to provide a long and sustained release of drugs with most of them designed to last for the whole treatment period on a single time application. This reduces the need for multiple administration, provide better bioavailability at target region and reduce systemic toxicity. 8 Over the years many different implants of various types, shapes and containing drugs either individual or in combination have been developed. Non- biodegradable implants will require the implant to be removed post treatment period because of which these days biodegradable implants are preferred. Polymers used for preparation of these include PLGA, PLA, polyethylene oxide, chitosan, etc.. 20 Polymers are favoured for creating ophthalmic implants because they possess biocompatibility, optical clarity, and mechanical resilience. 93 The fabrication methods commonly employed for the preparation of these polymer implants include solvent/melt casting, bench press and holt melt extrusion. These devices also provide more uniform drug distribution through the implant compared to others due to the processes associated with their preparation with adjustable drug dosages and is more suitable for scale up. 8 One of the first ocular implant to be marketed is the PLGA-dexamethasone intravitreal implant called Ozurdex®. The use of implants is not without certain potential risks like migration and need for removal of implant due to other complications like steroid induced glaucoma. 94
Other ocular inserts developed include punctal plugs for the delivery of drugs. 95 Punctal plugs are small biocompatible inserts roughly the size of a grain of rice that are widely used in the treatment of various eye diseases. Made from various polymers such as silicone, collagen, hydrophobic polymers, or hydrogel, these plugs are also referred to as punctum plugs or lacrimal plugs. They are inserted into the punctum or canaliculi of the lacrimal duct. 96
Presently, there are emerging studies employing 3D printing technology for the creation of drug delivery implants. 97
Intraocular lens (IOL)
Drug loaded IOLs are also being studied to be used as a drug reservoir. This can be especially effective in drug delivery to deal with post cataract complications as it does not require any additional steps during or post-surgery.98,99 Studies have been conducted to test the efficacy of IOL as a drug delivery reservoir for a variety of drugs. The drug loaded IOL can be prepared by soaking in drug solution, spray coating the IOL and modifications to the haptic. 100 The method of soaking or coating can be considered the simplest method to load the drugs, but they have often showed a shorter release time. 94 The other method involves making modifications or making extra attachments to the haptic where the attachment is the drug reservoir. But the problem with adding attachments onto the haptic of the IOL is that it might affect the insertion of the IOL during surgery as most prefer the use of foldable IOLs 94 Unlike in the cases of implant there is no risk of migration of the IOL. 101 Unfortunately, this category of drug delivery devices is yet to enter the market. 98
Nano-wafers
These are small circular/rectangular discs or membrane with drug loaded nano reservoirs and will eventually dissolve. 3 Common polymers used for their preparation include PVA, HPMC, CMC and PVP. 102 They are not affected by movement of the eyeball or blinking. Once placed on the eye and drug will gradually be released from the disc. 103 They can be easily applied onto the eye by the patient and does not require expert assistance for its application like contact lens. 104
Hydrogel
Hydrogels are network structures formed by crosslinking of hydrophilic polymers in water and are degradable. The commonly used polymers include polyvinyl alcohol, chitosan, and gelatin. 105 They are made of polymers that has good water holding capacity without getting disintegrated. They can hold up to 90% water by weight. 106 Some are capable of sol-gel transformation based on external stimuli and a capable of delivering a wide range of drugs from its crosslinked matrix for treatment of ocular conditions.27,107 The stimuli can be pH, temperature, light, or ionic strength. 105 Thermo-responsive hydrogels were the first to be studied and they undergo transition when exposed to a temperature above the lower critical solution temperature. The pH sensitive hydrogels are mostly formulated such that they are liquid at acidic pH and changes to gel when there is an increase in the pH as the surface of the eye has an almost neutral pH. 16 Their transformation to semi-solid or solid state from the original liquid state happens after application (in situ). Poloxamers®, xyloglucan and glycerol-2-phosphate are thermosensitive while Carbopol® and chitosan are pH-sensitive materials extensively employed in developing in situ gels for delivering drugs to the eyes. 16 Examples of ion-responsive materials include xanthan gum and alginate. These materials transition by generating ionic bonds inside the polymer in the presence of the cations present in the tear fluid. 108 Ideal characteristics of hydrogel include good adhesion to the ocular surface, sustained drug release, biocompatible and rapid gel transition. 109 The in situ forming gels are also often incorporated with other nanocarriers for improved drug delivery. Hydrogels also present some drawbacks, such as challenging sterilization procedures, restricted shelf life, potential harm to biopharmaceuticals from chemical cross-linking reactions, the risk of toxic effects from residues of polymerization initiators post-polymerization, and complexities in regulating degradation kinetics and drug release owing to water absorption during swelling. 110
Lipids as carriers
Lipids used as nanocarriers are often derived using low polarity solvents from animal or plant sources 4 and are biocompatible. These carriers include liposomes, niosomes, nanoemulsions, solid-lipid NPs and so on. Their structure is like cell membrane. This makes them biocompatible and enables uptake of both hydrophilic and hydrophobic molecules. 3 Emulsions prepared using two immiscible liquids that is stabilised using an amphiphilic surfactant with a size range of 20-500 nm are called nanosuspensions. They are kinetically stable systems but thermodynamically unstable. 82 Liposomes are small lipid vesicles with a central aqueous compartment surrounded by phospholipid bilayers. These nanocarriers exhibit stable adherence to the cell surface and charge on the carrier can also aid in the improvement of adhesion. 111 For example, the positively charged carriers are attracted to the surface of corneal epithelial and endothelial cells which have negatively charged moieties like sialic acid and mucopolysaccharides.112–114 Cholesterol and the presence of long hydrophobic phospholipid chains enhance the stability of the liposomes against degradation thereby controlling the drug release. 115 They can avoid ocular irritation on topical application as opposed to other micro sized particles and the certain emulsifiers used in the preparation also help to enhance the tear film aiding in management of conditions like dry eye. 116 But their application is restricted by its limited stability and short half-life in the body. 117 A cluster formed by the self-association of hydrated non-ionic surfactants in an aqueous phase with the addition of cholesterol or its derivatives results in the formation of niosomes. 16 They are similar in structure to liposomes and are often considered an alternative. 118 Although first reported to be used in the cosmetic industry in the seventies, it was discovered to have potential to be used as a carrier for drug delivery. Their non-ionic nature and absence of phospholipids makes them more stable, have lesser toxicity and better corneal penetration compared to liposomes. 46 Their ease of handling, storage and lesser production cost have also made them preferable for the pharmaceutical industry. 16 Colloidal dispersions made of solid-lipid molecules and surfactants/stabilizers where the drug is embedded in the lipid core or attached to its surface are called solid lipid NPs. 119 They are solid in ambient temperature but not in physiological temperature and their size ranges from 50 to 1000 nm. 69 They were introduced in the 90s as a substitute for polymeric NPs and emulsions. 120 The presence of solid matrix helps to prevent drug leakage which is otherwise seen other carriers like liposomes and nanoemulsions. The ease of scale up, sterilization and long-term stability make them ideal choice for commercialization.120,121
Dendrimers
These are polymeric hyperbranched star shaped macromolecules. First synthesized in 1980s, it soon found its potential in medical applications. 117 Their high-water solubility, surface functionalised groups and encapsulation ability make them a good choice to be used as nanocarriers. 122 They consist of a central core from which branches/dendrones project out. So far, over 100 dendrimer families with variations in their initiator cores, branching units, and terminal groups have been synthesized and evaluated. 123 The number of layers of branches corresponds to the generation the dendrimer. 124 Due to their tree-like branched structure and abundance of covalent connections, dendrimers have attracted considerable interest in the pharmaceutical industry for the delivery of drugs. The corneal permeation of dendrimers is dependent on its size and molecular weight. Commonly dendrimers in ocular drug delivery are poly(amidoamine) (PAMAM), polypropylenimines and phosphorous dendrimers. 81 In the recent years PAMAM with carboxylic and hydroxyl surface groups are preferred to create dendrimers for drug delivery. 3 It is the first of its kind to be commercialised. They have a size of about 3-12 nm and have also exhibited potential to cross the blood ocular barriers. 125 It has been reported that PAMAM dendrimers are cytotoxic, with toxicity increasing with each generation. But the near neutral surface charge of the hydroxy-terminated G4 PAMAM dendrimers allows them to be considered nontoxic as there is less non-specific retention and interaction with the tissues. 126
Management of ocular diseases using carrier based systems
Cataract and post-surgery complications
Recent publications in anterior segment disease management and polymer-based drug delivery systems.
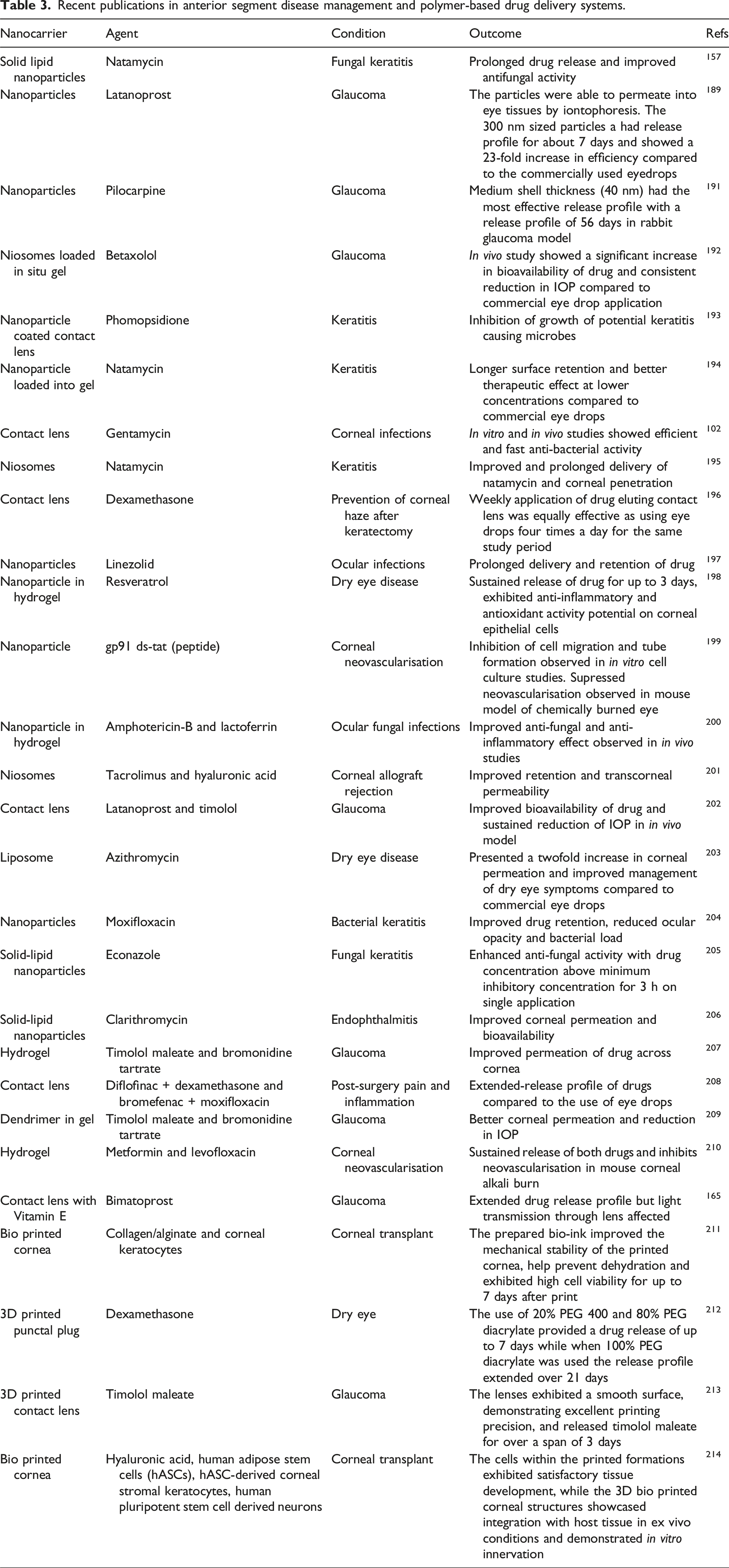
For post operative care of cataract surgery, a biodegradable PLGA implant containing an antibiotic drug called moxifloxacin and anti-inflammatory drug called dexamethasone was prepared by Subhash et al. When placed in capsular bag, it showed a sustained release of both dexamethasone and moxifloxacin for more than 30 days, demonstrating therapeutic efficacy. The release patter was like recommended dosing followed in clinical set up. 132
In another by Pinto et al., a dexamethasone containing biodegradable DDS was developed for post operative care of cataract surgery. The drug and PLGA was incorporated in a thin film which was attached to the haptic of IOL. This was able to release therapeutic dose of drug for over a period of 4 to 6 weeks, thereby overcoming the drawbacks of topical administration. 133
Dexycu® (Icon Bioscience, Inc., Sunnyvale, CA, USA) is a single dose intracameral treatment administered at the conclusion of cataract surgery. It utilizes Icon’s Verisome™ drug delivery technology to deliver a biodegradable, extended-release formulation of dexamethasone. 134 The system provides controlled delivery of dexamethasone for up to 21 days, though the release duration can be tailored, based on surgeon preference, to range from 1 week to over 9 months with a single injection. In early 2018, Dexycu became the first long-acting intracameral product approved by the FDA in the United States for managing inflammation associated with cataract surgery. 135 Currently, a silicone nepafenac punctal plug containing 204 µg of the drug is currently undergoing phase II clinical trials for the management of post-operative pain and inflammation. 107
New treatment strategies have been being investigated to prevent or stop the development of cataracts. Antioxidants, chaperons and NSAIDs are few of the agents known to help prevent and treat the condition. Initially oral administration of agents like ibuprofen and bendazac were used for delaying cataract formation. 128 But due to the high amount of drug that will be required to obtain therapeutic concentration, nanocarrier based systems were explored to increase bioavailability. One of the initial works were done by Romanelli in 1994 where various polysaccharide vehicles were analysed to deliver and improve the bioavailability of bendazac, a drug known to protect lens protein by glycation. 136 Antioxidants like curcumin encapsulated in PLGA have shown to have nine times better bioavailability than free curcumin on oral administration as per studies conducted for streptozotocin-induced diabetic cataract in rats.137,138
Corneal injury/scarring
Injury to the cornea can lead to fibrosis and scarring which will affect the corneal transparency.105,139 Keratoplasty is presently the only approved method for treating corneal scarring. 109 The obstacle is obtaining a suitable donor and the number of donors available compared the number of patients in need remains a challenge. 140 Post surgery complications and rejection of donor tissue are possible aftereffects of the surgery. Keeping these in mind new alternatives were explored like 3D printed cornea, biomimetic hydrogels, gene therapy and stem cell therapy.119,141–143 Hydrogels can be used to act as an artificial cornea substitute as well as load active compounds that aid in wound healing. 109 Metal NPs like gold and silver can be used as carriers for gene delivery due to their inert and non-toxic nature. 144 Xuan Zhao et al. used collagen as scaffold material into which complexes of gold NPs and miRNA 133b was combined to inhibit scar formation and repair the cornea in lamellar keratoplasty model on rabbits. The gold NPs-miRNA 133b complex inhibits the myofibroblast transformation, a process which leads to corneal scar development. 145 Bone morphogenetic proteins 7 (BMP7) is a growth factor which on administration has shown the ability to attenuate fibrosis. Polyethylenimine-conjugated gold NPs loaded with BMP7 gene on topical administration aids in targeted delivery into the keratocytes of the stroma of the cornea to modulate wound healing and inhibit fibrosis thereby reducing corneal scarring. 146 Mesenchymal stem cells are known for their ability to restore corneal transparency. These can be engineered for therapeutic applications.147,148 A promising option to create unique corneal scaffolds without using moulds is provided by 3D bioprinting, an emerging technology that enables digital manufacturing of cell-laden three-dimensional tailored constructs by layer-by-layer deposition of cells and biomaterials. 149 The bioink utilized for 3D bioprinting cells needs to be both biocompatible and non-toxic to enable subsequent testing in human subjects. 142 The long-chain PEG diacrylate and gelatin methacrylate blended hydrogels produced by 3D printing have been demonstrated to be cytocompatible, satisfy the needs of corneal implants in terms of promoting cell adhesion, proliferation, and migration, and have a high light transmittance. A bilayer corneal scaffold prepared using this hydrogel with one layer incorporated with corneal epithelial cells and the other with adipose-derived mesenchymal stem cells results in a biomimetic epithelium/stroma bilayer which has shown its efficiency in sealing of corneal defects and corneal regeneration. 116
Dry eye
Keratoconjunctivitis sicca or dry eye is a chronic condition that develops due to lack of tear fluid necessary to lubricate the eye. It can result in inflammation of cornea and conjunctiva. If not managed well can lead to corneal irritation and scarring. 20 The pathogenesis of the condition is still not fully understood. 115 Due to the chronic nature of this disease long term medication is required. 118 Treatment strategy mainly involves alleviation of the symptoms rather than a cure. Artificial tears and anti-inflammatory drugs are prescribed to patients as a remedy to this.9,120 There are liposomal eye drops in the market which are used to treat mild to moderated dry eye disease. 103 These eye drops are also known to improve the stability of the tear fluid. Lacrisert is an FDA approved hydroxypropyl cellulose insert used to treat moderate to severe dry eye condition. 121 Dual drug loaded gold NPs containing the drugs amfenac and catechin have shown reduced inflammation and reactive oxygen species compared to the application of cyclosporin as eye drops in the management of dry eye in rats. 150 Dexamethasone loaded carboxymethyl cellulose nano wafers have demonstrated that application of one disc on alternative days had a comparable effect to that of dexamethasone eye drops which were applied twice a day in controlling inflammation associated with dry eye disease. 104 Lactoferrin delivery using hyaluronic acid coated liposomes was found to be efficient in managing dry eye and its associated ocular inflammation. 151
Endophthalmitis
It is referred to the inflammation of the aqueous vitreous humor of the eye. 84 Commonly caused by infection of organisms like Pseudomonas, E. coli and Streptococci. 106 It is a potential post-operative complication. Hydrophilic acrylic IOLs have proved to be good carriers in the delivery of antibiotics for the control of post-operative endophthalmitis with the significantly higher concentration of drug seen in the aqueous humor compared to free drug administration in in vivo model studies.152,153 The addition of HEMA hydrogel loaded with norfloxacin onto the IOL haptic that can be placed into the eye during cataract surgery has shown a release of the antibiotic for up to 4 weeks and controlled development of bacterial endophthalmitis with minimum toxicity. 154
Keratitis
It is associated with the presence of inflammation of the cornea which could be caused by either infectious or non-infectious factors. Patients experience pain, redness of the eye and reduced vision. Management of the condition is variable depending on the severity of the condition ranging from using antibiotic eye drops to be need for corneal transplants. 9 Corneal ulcers and scarring are a potential after effect of keratitis. 155 More prevalent in subtropical and tropical regions organisms causing fungal keratitis include Candida, Cladosporium, Fusarium, Aspergillus and Rhizopus. 156 These infections have a 25% incidence of causing perforations. 155 Natamycin is a broad spectrum anti-fungal drug which is currently the only FDA drug approved drug for fungal keratitis, but its poor corneal permeability is limits its therapeutic effect. The incorporation of natamycin into NPs help improve the corneal penetration and achieve a sustained delivery of the drug. 157 Considering the lack of anti-fungal agents that are safe and effective for clinical use anti-microbial peptides are being explored in the recent years. They are known for their potent broad spectrum antimicrobial activity and has shown potential to tackle the issue of drug resistance in microbes. They work by rapid disruption of the membrane in the microbes thereby preventing possible acquisition of drug resistance. 158
Conjunctivitis
It is a condition associated with the swelling of the conjunctiva. It is accompanied by increased tear production, pain and can be contagious. 117 Causative agent could be infection, allergy or exposure to certain chemicals and the treatment includes usage of topical anti-inflammatory agents and antibiotics in case of infectious condition.9,118 Allergic conjunctivitis is managed by using topical antihistamines. 117 A common cause for bacterial conjunctivitis is Staphylococcus aureus infection in the eye. 106 Hydrogel prepared using gelling agents poloxamer 407 and chitosan when incorporated with the antibiotic neomycin sulphate and anti-inflammatory agent betamethasone sodium phosphate has exhibited good tolerance during Draize and Hen’s egg test-chorioallantoic membrane (HET CAM) studies marking its potential to be used for the infection and inflammation associated with conjunctivitis. 159
Uveitis
Uveitis is an inflammatory condition in the eye involving the uveal tissues. It could arise due to several factors such as trauma, infection, inflammation and allergies. 118 The condition leads to the release of prostaglandins inducing vasodilation and increase in vascular permeability. This in turn causes a series of other events like pain, increased IOP and posterior capsule opacification. 81 The treatment strategy usually involves the use of anti-inflammatory agents. A single dose of pegylated liposomal formulations containing steroids like prednisolone and triamcinolone acetate when administered as subconjunctival injection as a have exhibited effective and sustained drug delivery in management of experimental uveitis in rabbit eyes. 160 Gold NPs have shown anti-inflammatory effect by attenuation of TNF-α levels and NF-κB levels when applied 2 hours post lipopoly saccharide injection in a uveitis model in rats. They have also shown that the gold NPs showed better anti-inflammatory effect than the application of topical prednisone eye drops in the study. 83
Glaucoma
Glaucoma is a multifactorial condition, and the pathogenesis is not well understood. One noticeable feature is the damage caused to the optic nerve due to increase in IOP. 161 Increase in IOP can also lead to damage of corneal endothelial cells. 162 There are two types of glaucoma namely angle-closure glaucoma and open-angle glaucoma. 105 The first one is caused by the blockage of the trabecular meshwork leading to increase in IOP while the latter is due to increase in IOP caused by reduction in the trabecular meshwork permeability. 163 This leads to increase in IOP and activation of oxidative stress related pathways. 164 The slow progression of the condition results in patients showing symptoms only once the condition has become rather severe. 161 Lowering of the IOP is the main treatment strategy for glaucoma. The drugs used for treatment has either of the two functions: improve drainage of fluid through trabecular meshwork or reduce aqueous humor production. The alternative strategy is surgical intervention to create a new drainage. Most avoid the surgical approach as its invasive and has more compilations. 109 Recently studies have been published reporting the use of nanocarriers like contact lens, nanoemulsions and hydrogels to improve the drug bioavailability and permeation across the cornea due to their longer residence time thereby providing a sustained reduction in the IOP. 165
Nanocarriers for drug delivery- marketed and under clinical trials
List of drug delivery devices in the market for anterior segment diseases.
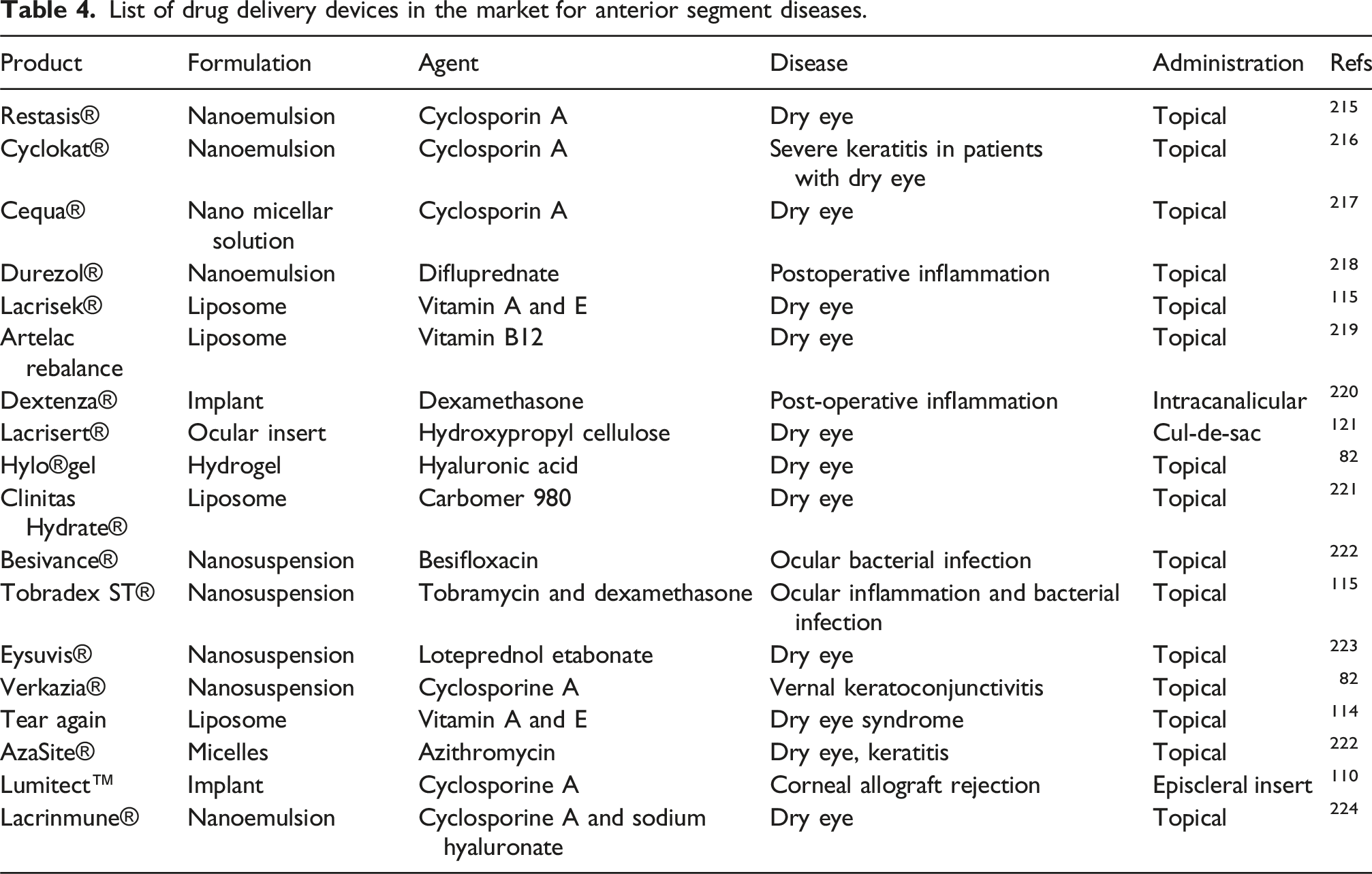
Surodex (Oculex Pharmaceuticals Inc., Sunnyvale, CA) is a biodegradable PLGA-based ocular implant designed to deliver a controlled dose of 60 µg of dexamethasone directly into the anterior chamber of the eye. This innovative device is used to manage inflammation following cataract surgery, providing a sustained release of medication over approximately 7 days. 170 By combining the biocompatibility of PLGA with a targeted drug delivery system, Surodex minimizes the need for frequent topical applications, enhancing patient compliance and post-operative outcomes. 171 Unfortunately, this study has been discontinued. But an issue noticed during the usage of implants like Ozurdex and Surodex was the migration of the implant to other regions from the site of placement. 172
Nano formulations under clinical trial for anterior segment diseases (Source: Clinicaltrials.gov).
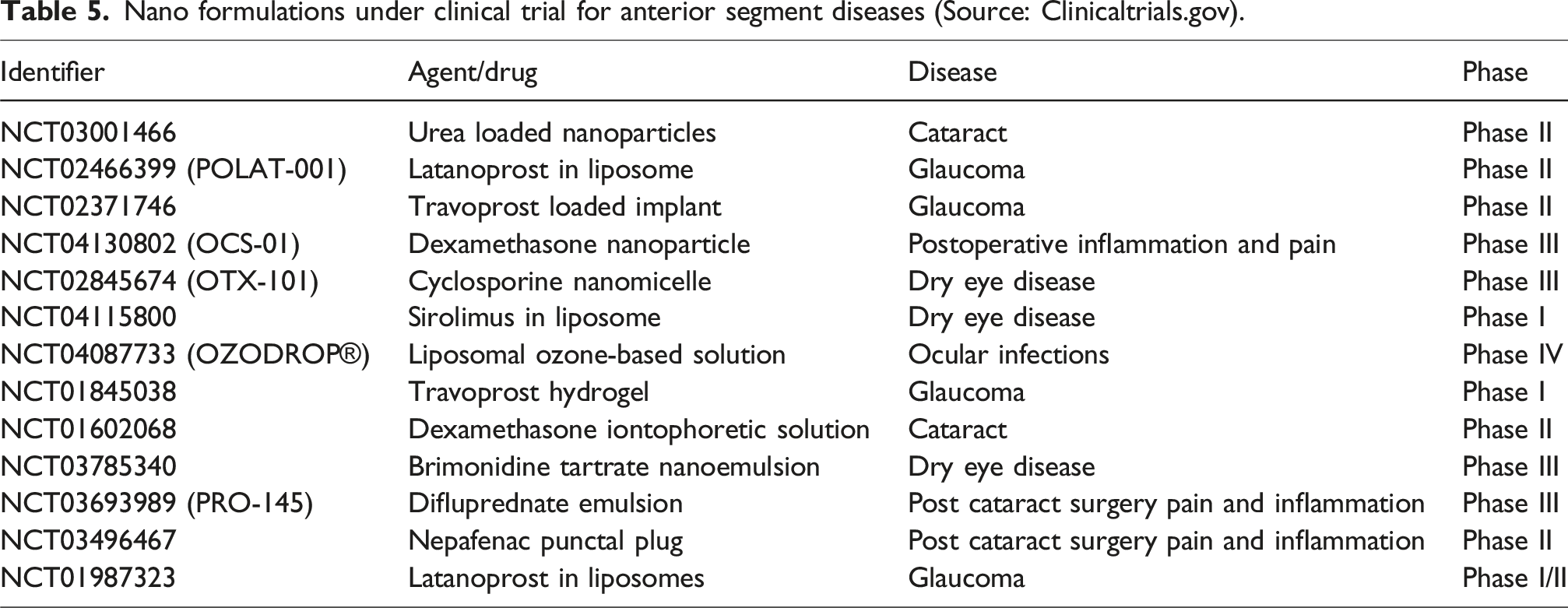
Conclusion
Eye disorders indeed pose challenges, impacting both finances and quality of life. While traditional treatments like eye drops and ointments are beneficial for conditions such as glaucoma, dry eye, and infections, they often face limitations due to anatomical barriers within the eye. These barriers hinder effective treatment, leading to insufficient care and recovery from these conditions. Moreover, compliance with traditional medicines, especially among geriatric and paediatric patients, becomes challenging due to the need for frequent administration and high doses to achieve therapeutic levels. Systemic absorption of the drug leading to systemic side effects is another problem.
Extensive research in ocular drug delivery aims to address challenges like invasive techniques, frequent application, short retention time, and ocular irritation are associated with conventional treatments. Optimal drug delivery to the eye, requires measures to overcome the ocular barriers, achieving therapeutic concentrations, and minimizing tissue damage or side effects. Nanotechnology has emerged as a promising approach in this field, showing potential in surmounting these conventional barriers. However, despite numerous studies on formulations and carrier systems, many have not entered the market or clinical practice. This may be attributed to reasons such as lack of suitable animal models or challenges related to mass production. Additionally, there can be discrepancies between the behaviour of nanocarriers in vitro and in vivo, further complicating their clinical translation. The choice of polymers for preparation of nanocarriers is also crucial and should fit the specific requirements of the application. The stringent requirements for approval of new drug delivery systems add to the challenges, necessitating significant time and effort for regulatory clearance. Most 3D-printed devices are currently in their initial development phases and await extensive clinical trials.
Despite the promising potential of 3D printing in the medical field, the regulatory framework for its use in pharmaceutical and medical applications remains incomplete. Although the FDA approved the first 3D Printed device, Spritam, in 2015, comprehensive guidance for 3D Printing applications was not issued until 2017. 173 Furthermore, the FDA has yet to establish official regulations specifically governing 3D printing technology. This regulatory gap poses significant challenges for the adoption of 3D printing medical devices and slows the clinical translation of 3D printed products to patients. The lack of clear guidelines also creates uncertainty regarding accountability in cases involving safety concerns with 3D printed products. As of now, discussions continue to develop a comprehensive regulatory framework to ensure the safety, efficacy, and consistent quality of 3D printed medical devices and pharmaceuticals. 174 In December 2017, the FDA published guidance titled “Technical Considerations for Additive Manufactured Medical Devices,” offering initial regulatory insights into the requirements for the use of 3D printing in medical applications. 175
Nonetheless, as 3D printing technology progresses and becomes more accessible, an increasing number of medical research teams are crafting implants tailored to the needs of the patients. Therefore, while nanotechnology holds promise for revolutionizing ocular drug delivery, several obstacles remain before successful implementation in clinical practice.
Future perspectives
The current focal point of research revolves around enhancing the existing state of ophthalmic care. Future inquiries should prioritize the engineering of nano formulations targeting the anterior eye segment. These formulations ought to prioritize safety by accommodating both small molecules and biologics, such as genes or peptides, showcasing reduced toxicity, heightened stability, and efficient drug delivery with improved pharmacokinetics and pharmacodynamics. Furthermore, formulating strategies should aim to co-encapsulate ocular active drugs and enzyme inhibitors, custom-designed to impede eye-specific enzymes, thereby facilitating ocular absorption and boosting bioavailability. This aspect holds promise as a focal point for potential research hotspots that could drive clinical transformation in the future. An ideal nano formulation should sustain consistently effective drug levels post a single application and demonstrate high bioavailability. Improving scalability and cost-effectiveness, especially in industrial settings, is also a critical priority. Collaboration between formulation scientists and clinicians is essential to identify and address specific barriers hindering the clinical translation of these systems. This collaborative endeavour aims to bridge the gap between laboratory innovations and practical clinical application. By 2028, the global ocular drug delivery market is anticipated to reach a valuation of approximately USD 27.02 billion, reflecting substantial growth from its 2021 value of USD 16.40 billion. This growth trajectory corresponds to a robust CAGR of 7.4% during the forecast period from 2021 to 2028. 176
Footnotes
Acknowledgements
We would like to express our sincere gratitude to Manipal Academy of Higher Education (MAHE), Manipal for providing the opportunity and resources to undertake and complete this review paper. We would like to thank the Director of Manipal Institute of Technology (MIT), Manipal for his encouragement and assistance during this endeavour. Our sincere gratitude extends to the Dean of Kasturba Medical College (KMC), Manipal, who has provided invaluable support.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Biotechnology Industry Research Assistance Council (BIRAC)–Biotechnology Ignition Grant (BIG) (BT/7908/BIG-15/19).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
