Abstract
Complications of transcutaneous osseointegrated prosthetic systems (TOPS) focus on the metal-cutaneous interface at the stoma. Besides pain due to scare tissue as well as undefined neuropathic disorders, there is high evidence that the stoma presents the main risk causing hypergranulation and ascending infection. To restore the cutaneous barrier function in this functional area, soft-tissue on- or in-growth providing a vital and mechanically stable bio-artificial conjunction is considered a promising approach. In this study we assessed viability and proliferation of adult human dermal fibroblasts (HDFa) on modifications of a standard prosthetic titanium surface. Un-coated (TiAl6V4) as well as a titanium-nitrite (TiN) coated additive manufactured porous three-dimensional surface structures (EPORE®) were seeded with HDFa and compared to plain TiAl6V4 and polystyrene surfaces as control. Cell viability and proliferation were assessed at 24 h and 7 days after seeding with a fluorescence-based live-dead assay. Adhesion and cell morphology were analyzed by scanning electron microscopy at the respective measurements. Both EPORE® surface specifications revealed a homogenous cell distribution with flat and spread cell morphology forming filopodia at both measurements. Proliferation and trend to confluence was seen on un-coated EPORE® surfaces with ongoing incubation but appeared substantially lower on the TiN-coated EPORE® specification. While cell viability on both EPORE® specifications was comparable to plain TiAL6V4 and polystyrene controls, cell proliferation and confluence were less pronounced when compared to controls. The EPORE® topography allows for fibroblast adhesion and viability in both standard TiAl6V4 and – to a minor degree - TiN-coated specifications as a proof of principle.
Keywords
Introduction
Transcutaneous osseointegrated prosthetic systems (TOPS) have gained increasing interest as alternative to a conventional socket prosthesis.1–3 These systems consist of a bone-anchored, osseointegrated module, the “fixture”, which is press-fit implanted in the medullary cavity, and a skin-penetrating module, the “abutment”, which is attached to the fixture by an abutment screw. The basic concept is similar to the direct anchorage of dental implants in the jaw. 2 Benefits of this approach include improvements in tactile sensory perception and motion control,4,5 as well as prevention of socket-stump related soft tissue issues. This advantages face concerns regarding the soft-tissue-implant interface at the skin-penetration site or stoma respectively.5,6 The focus of the latter is on periprosthetic infection induced by biofilm formation and potentially leading to failure of the abutment as well as loosening of the osseointegrated module in case of a resulting ascending infection. Thus potentially pathogenic bacteria such as Staphylococcus aureus, coagulase-negative staphylococci, streptococci, and Enterococcus faecalis frequently colonize both localizations: the stoma as well as the medullary canal. However, despite high colonization rates, the indication for comprehensive revision surgery is much less frequent. 2 While some authors state that TOPS have no unpredictable high risk of periprosthetic infection if appropriate hygiene care is applied,5,7 other classify the permanent breach of the skin barrier at this contact area as a critical issue. 2 What is undisputed is that complications of TOPS are focussed on the stoma area 8 and infection prevention at this bio-artificial interface is considered to be the key for a successful routine application of such systems. 9 Restoration of the skin barrier therefore appears to be a promising approach. As various concepts to create a sealed soft-tissue-prosthesis interface have not proven successful in long-term situations, 9 creating a durable, vitalized and bacteria-proofed skin attachment to the soft-tissue penetrating abutment remains a challenge. Current material concepts of this interface area of TOPS include titanium 10 and cobalt-chromium-molybdenum alloys. 5 In order to evaluate the basic suitability of a TiAl6V4-based abutment component for fibroblast in-growth, we compared the impact of plain and additive 3D-manufactured topographies – in an un-coated as well as titanium-nitrite (TiN)-coated specification – on adhesion and cell-viability of adult human dermal fibroblasts.
Material and methods
Titanium sample specifications
Round TiAl6V4 disks of 15 mm diameter and 2 mm height were additively manufactured by electron beam melting (EBM) with each disc showing an upper surface topography divided into two halves. One half was plain, blasted and un-coated (specification 1, S1) as well as titanium nitride (TiN)-coated (specification 2, S2), while the second half provided a porous 3-D surface structure (EPORE®, Implantcast GmbH, Germany), characterized by a mean porosity of 61% ± 8%, a mean rod diameter of 360 µm and a Voronoi mesh densitiy of 1500 points/cm3 according to the manufacturer, in an un-coated (specification 3, S3) as well as an TiN-coated (specification 4, S4) design (Figure 1). According to the manufacturer the mean thickness of the respective cathodic arc deposition (PVD-arc) manufactured TiN layer reached 5.5 ± 1.5 µm. Scanning electron microscopy (SEM) image of an un-coated TiAl6V4 sample (a), as well as a TiAl6V4 sample coated with titanium nitride (TiN) (b). The upper half of the image shows a plain, blasted surface topography. The lower half of the image shows a porous 3-D surface structure (EPORE®, Implantcast GmbH, Germany).
Contact angle measurement
To asses the wettability of the surface specifications two un-coated and two TiN-coated plain TiAl6V4 samples were evaluated by contact angle measurement (OCA 15, DataPhysics, Filderstadt, Germany). Using water as liquid medium (density 0.998 g/cm3, viscosity 1 mPas, surface tension SFT 72.8 mN/m) three droplets per sample with a dosage volume of 5 µl and a dosage rate of 1 mL/s were applied according to the sessile drop method. Mean contact angle and standard deviation were calculated based on the resulting six measurement values per specification. The statistical analysis was performed using Microsoft Excel 2021 software, with differences among specifications being assessed via an analysis of variance followed by a two-tailed, unpaired t test and considered statistically significant when p ≤ .05.
Control samples
Empty wells of a 24-well polystyrene plate (FalconTM, fisher scientific GmbH, Germany) were used as control.
Cell culture
Cell culture experiments were carried out using adult human dermato-fibroblasts (HDFa, GibcoTM, Thermo Fisher Scientific Inc, USA). Cells from frozen stock cultures were seeded in α-minimum essential medium (α-MEM, Pan Biotech GmbH, Germany), supplemented with 10% fetal calf serum (Merck, Germany), 1% L-glutamin (Sigma-Aldrich, USA) and 1% Penicillin/Streptomycin (Pan Biotech, Germany) utilizing a humidified incubator with 5% CO2 and 95% air at 37°C. Splitting was performed at near confluence (70%–80%) and cells from the fifth passage were included in the study.
Study design
Four sample specifications as well as empty wells of a 24-well polystyrene plate as control were seeded with 9,5*10^4 HDFa cells per well and incubated at 37°C under humidified atmosphere of 95% air and 5% CO2 for 7 days. For each measurement (d1 = 24 h after seeding, d7 = 7 days after seeding) as well as for each analysis procedure a separate 24-well plate had been used. A complete replacement of the culture media (α-MEM, Pan Biotech GmbH, Germany, supplemented with 10% fetal calf serum, Merck, Germany, 1% L-glutamin, Sigma-Aldrich, USA, and 1% Penicillin/Streptomycin, Pan Biotech, Germany) was performed after 24 h of incubation and subsequently every 48 h. Double triplets of the respective specifications were used for determination of cell viability utilizing fluorescein diacetate (Thermo Fisher Scietific Inc, USA) and propidium iodide (Merck/Sigma-Aldrich, St Loius, USA) (FDA/PI) staining. Controls were performed as triplet. For assessment of cell adhesion and morphology by scanning electron microscopy (SEM) doublets of the respective specifications and single determination of controls were used.
FDA/PI cell viability assay
For the determination of the viable and necrotic cells, analysis was performed after incubation for 24 h and 7 days including three polystyrene controls as well as six un-coated and six TiN-coated TiAl6V4-samples respectively at each measurement. The culture medium was aspirated and samples as well as control-wells were washed three times with 700 µl phosphate buffered saline (PBS). Subsequently samples and controls were layered with 700 µl FDA/PI working solution, consisting of 3 µl fluorescein diacetate (4 mg FDA/ml acetone) and 10 µl propidium iodide stock solution (10 mg PI/ml bi-distilled water) in 20 mL PBS, and incubated at room temperature for 5 min. The working solution was aspirated and samples and controls were washed three times with 700 µl PBS and finally layered with 700 µl PBS.
Fluorescence-microscopy
Images of the FDA/PI stained samples and controls were taken with a motorized inverted epifluorescence microscope (Apotome AxioObserver.Z1, Zeiss, Jena, Germany), considering three regions of interest (ROI). These were located in the center of the plain, blasted topography (ROI A), at the interface of this same topography and the porous structured topography (ROI B), and in the center of the latter topography (ROI C) (Figure 2). To address the 3-D structure of the topography provided in ROI B and C, z-stacks using 30 µm steps (26 to 40 images per channel) were performed and imaged into one plane by orthogonal projections. Orthogonal projections of both channels (FDI- and PI-channel) then were merged to one resulting color composite per ROI B and C respectively. Controls in contrast were analyzed by merging one color composite of a respective central ROI from both, the FDA- and the PI-channel at both measurements. In total 78 color composites resulted from three images per sample (one image per ROI, three ROIs per sample, six samples per specification) and one image per polystyrene control (three controls) at the respective measurement. Each color composite was evaluated on a descriptive level considering live-dead ratio, cell distribution and cell morphology. Localization of the regions of interest (ROI) on the sample discs. ROI A was set in the center of the plain, blasted topography. ROI B was set at the transition of this same topography to the porous structured topography. ROI C was set in the center of the 3-D topography.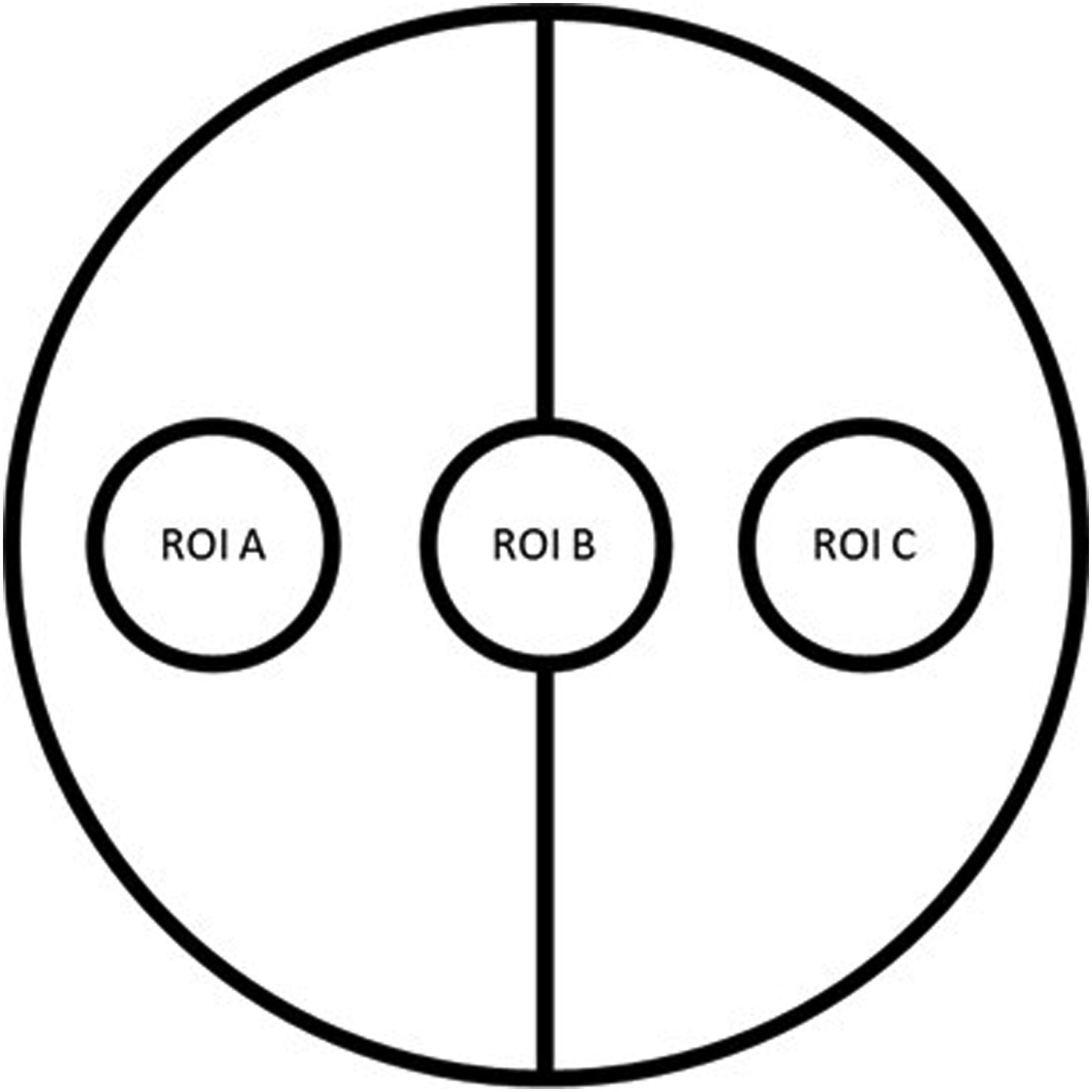
Scanning electron microscopy (SEM)
Assessing one polystyrene control as well as two un-coated and two TiN-coated TiAl6V4-samples respectively at each measurement, for SEM preparation seeded titanium samples as well as polystyrene controls were rinsed two times with PBS and then fixed with a fixative solution containing 2% glutaraldehyde and 4% paraformaldehyde in 0.1 M sodium-cacodylate buffer for 2 h at 37°C and 5% CO2. After final rinsing and dehydration through a graded series of ethanol, samples and controls were vacuum-dried. SEM analysis (Crossbeam 540, Zeiss, Jena, Germany) was then performed considering three ROIs in case of sample analysis and one central ROI in case of polystyrene controls respectively. The polystyrene controls were additionally sputtered with a 5 nm mixed layer of platinum and palladium using a Leica EM ACE 600 sputter coater to ensure the conductivity of the samples. Each ROI was assessed using 30x, 100x, 300x and 1000x magnification. In total 104 SEM images resulted from 12 images per sample (four images per ROI, three images per sample, two samples per specification) and four images per polystyrene control (one control) at the respective measurement. Each SEM image was evaluated on a descriptive level considering morphology, adherence, proliferation, and distribution of cells.
Results
Wettability
Contact angle measurements of plain un-coated TiAl6V4 surfaces displayed a mean of 98.49° (standard deviation 2.4°) and proved statistically significant higher than values of plain TiN-coated samples (p < .001) with a mean of 84.02° (standard deviation 3.27°) (Figure 3). Contact angle measurements of un-coated (S1) and TiN-coated (S2) plain TiAl6V4 samples. Statistically significant differences, p < 0.001.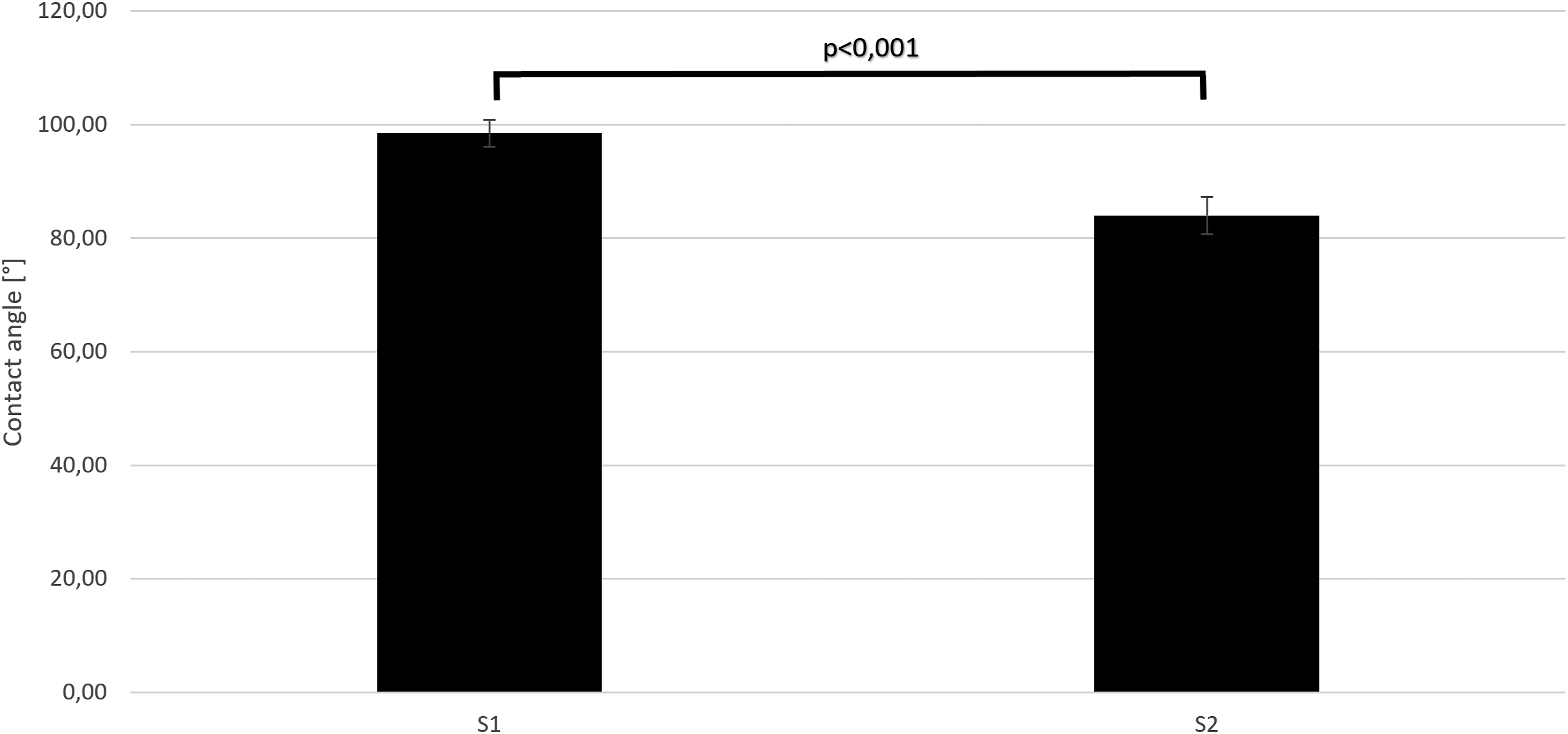
Fluorescence microscopic imaging
Cell viability, morphology and proliferation
24 h (measurement 1) and 7 days (measurement 2) after seeding all four TiAl6V4-specifications revealed live-dead ratios similar to the polystyrene controls while proliferation and distribution patterns of viable cells respectively showed a different appearance, more pronounced in the 3D-porous specifications and at measurement 2 when compared to control. Cell morphology appeared - similar to control – elongated and spindle-shaped at measurement 1 but revealed a different appearance from the controls at measurement 2.
At measurement 1 living cells were visible on polystyrene controls in an unordered distributional pattern as well as in spots of confluence covering the entire area of the respective ROI. Only minor amounts of death cells occurred. Living cells were spindle-shaped and elongated. In areas of confluence they partially appeared in parallel arrays (Figure 4(a)). Aside of these spots cell density appeared considerably reduced. HDFa cells were seeded on polystyrene controls and incubated for one day (a) and seven days (b). A live/dead staining was used to determine the cell viability. The fluorescence imaging with a magnification of 5x displays living cells in green and dead cells in red.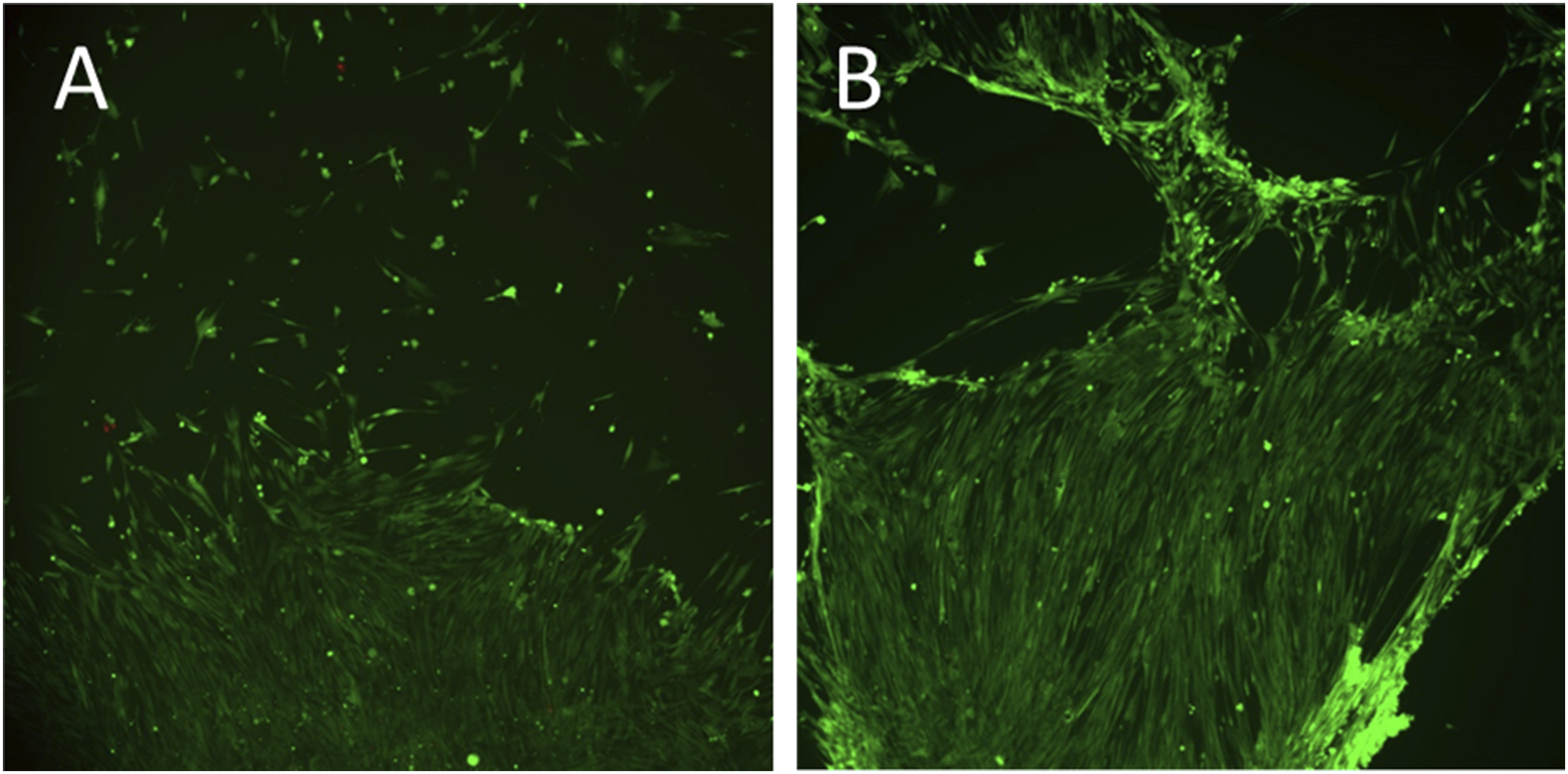
At this measurement cells on both plain specifications (S1, S2) showed distribution patterns, cell morphology and live-dead ratios similar to the cell-cluster-free areas of the control. Cell density nearly reached the appearance of these cluster-free areas of the control but revealed substantially reduced cell amounts when compared to the described confluent spots of the latter (Figure 5(a)–(b)). HDFa cells were seeded on sample discs and incubated for one day. A live/dead staining was used to determine the cell viability. The fluorescence imaging with a magnification of 5x displays living cells in green and dead cells in red on the plain topography specifications S1 (a) and S2 (b) as well as on the 3-D porous topography specifications S3 (c) and S4 (d), presented for the latter as orthogonal projections.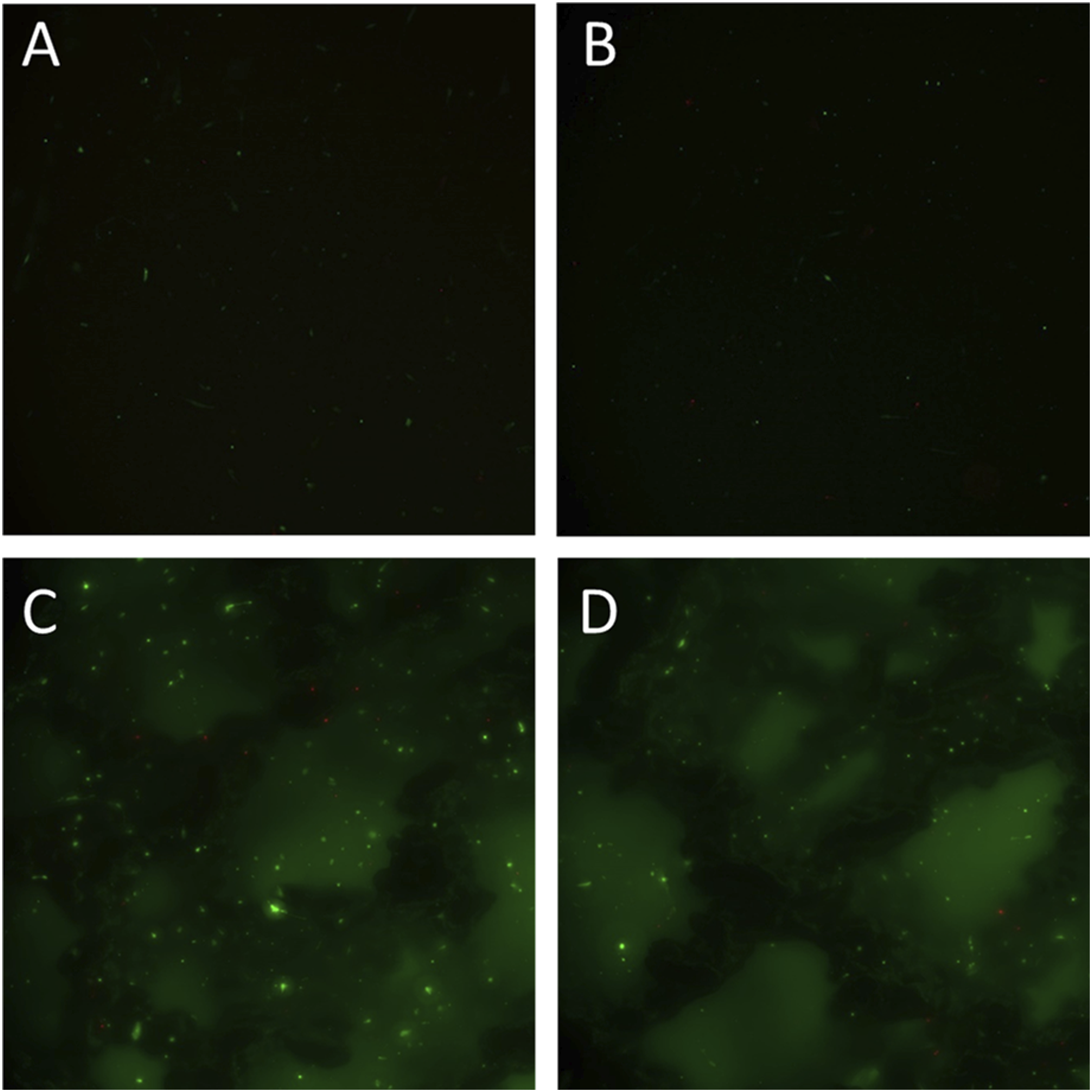
All areas of both 3-D porous specifications (S3, S4) were covered with cells, which revealed live-dead ratios similar to S1 and S2 and control as well as an unordered distributional pattern without confluent spots. As in controls cells appeared spindle-shaped and elongated and showed adherence on bridges and pillars as well as onto the plain bottom of the pores (Figure 5(c)–(d)).
At measurement 2 progressive cell proliferation became obvious on controls resulting in extended spots of cellular confluence with only marginally occurring isolated living or dead cells and live-dead ratios similar to measurement 1. Nearly cell-free areas in unordered distribution were localized between the confluent spots creating a reticular appearance of the cellular layer with narrow as well as broad connecting areas (Figure 4(b)).
Both plain specifications S1 and S2 in contrast revealed an approximately completed confluence with a considerably more elongated cellular morphology when compared to day one. Cells appeared predominantly in parallel, partly overlapping arrays and with live–dead ratios comparable to controls (Figure 6(a)–(b)). HDFa cells were seeded on sample discs and incubated for seven days. A live/dead staining was used to determine the cell viability. The fluorescence imaging with a magnification of 5x displays living cells in green and dead cells in red on the plain topography specifications S1 (a) and S2 (b) as well as on the 3-D porous topography specifications S3 (c) and S4 (d), presented for the latter as orthogonal projections.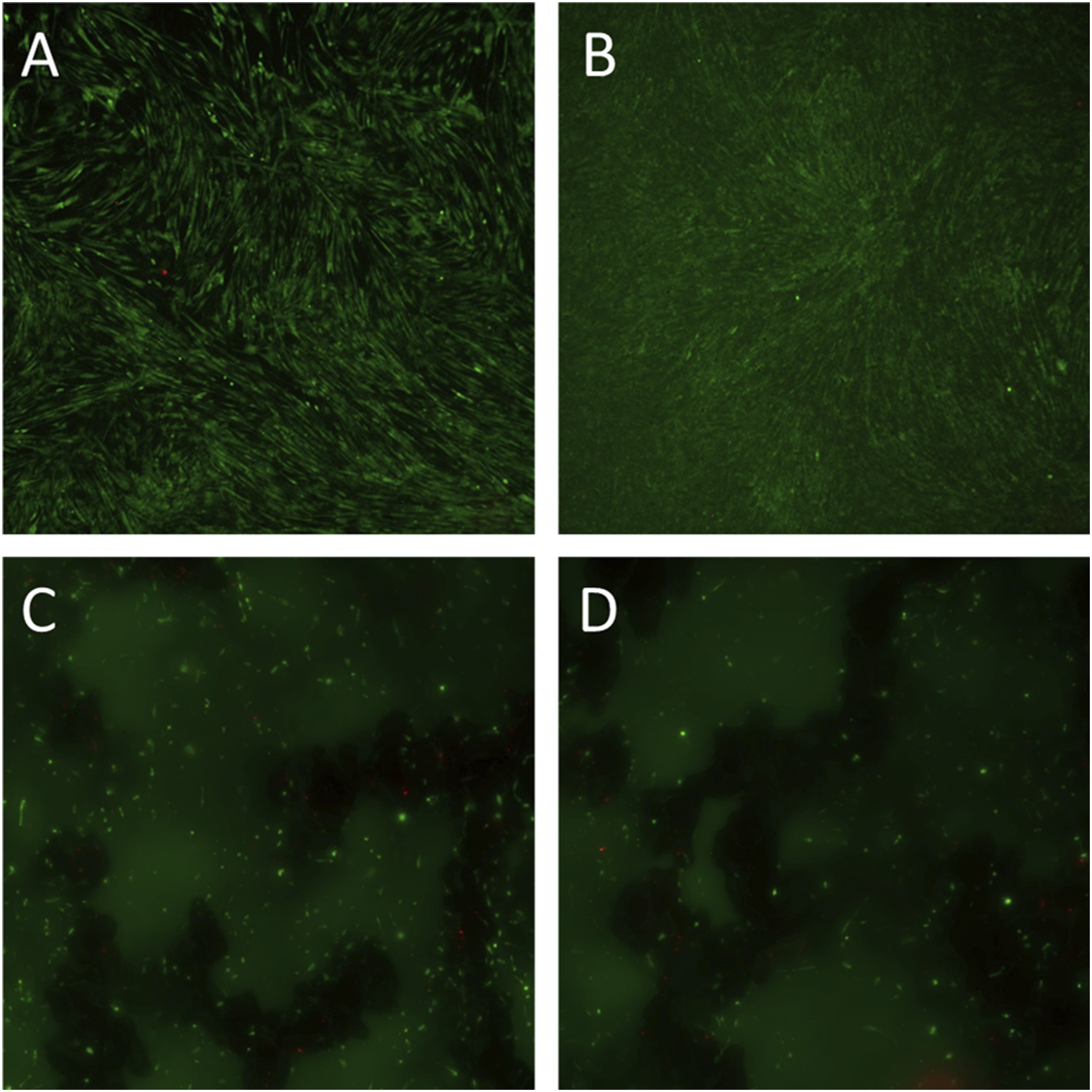
On the uncoated 3-D porous specification S3 cells had also proliferated but to a much lesser degree when compared to the plain specification and controls and without the occurrence of confluent spots. Cells appeared in linear arrays with a tendency to beginning confluence alongside the structural elements of the matrix but did not bridge macropores. Compared to day one and controls, live-dead ratios remained nearly unchanged. (Figure 6(c)).
On the TiN-coated porous specification S4 cells revealed distribution patterns and live-dead ratios similar to S3 and controls but showed reduced to no tendency to build linear arrays of beginning confluence. While proliferation became obvious, the overall cell count appeared to be reduced when compared to the un-coated porous specification at this measurement (Figure 6(d)).
Scanning electron microscopy
Cell morphology and adherence
At measurement 1 plain as well as 3-D manufactured topographies (S1 – S4) confirmed cell distribution patterns and morphologies of the controls as visualized by fluorescence microscopy (Figure 7(a)–(d)). Cells started to form small filopodia and to a lesser degree lamellipodia. However, on the 3-D-topographies (S3, S4) cells incidentally revealed a more polygonal or triangular shape with extended lamellipodia. At measurement 2 the plain surface specifications (S1, S2) revealed an increased progressive proliferation and a tendency to cellular migration when compared to controls, while on the 3-D-topographies (S3, S4) a reduced proliferation speed with a mixed morphology of spindle-shaped and polygonal or triangular shaped cells was noticeable indicating a more stationary, spreading appearance. Overall, the TiN-coated specifications (S2, S4) showed a considerably decreased cell density when compared to the un-coated specifications, controls and plain specifications in that order. SEM images of HDFa cells seeded on sample discs and incubated for one day. Cell behavior on the plain topographies of specification S1 (a) and S2 (b) as well as on the 3-D porous topography specifications S3 (c) and S4 (d) is displayed at 30x magnification.
While on polystyrene controls cell adherence and acquisition of the surface were the main focus at measurement 1, at measurement 2 the SEM analysis revealed progressive proliferation and cell distribution patterns similar to the data provided by fluorescence microscopy (Figure 8(a)–(b)). SEM images of HDFa cells seeded on polystyrene controls and incubated for one day (a) and seven days (b).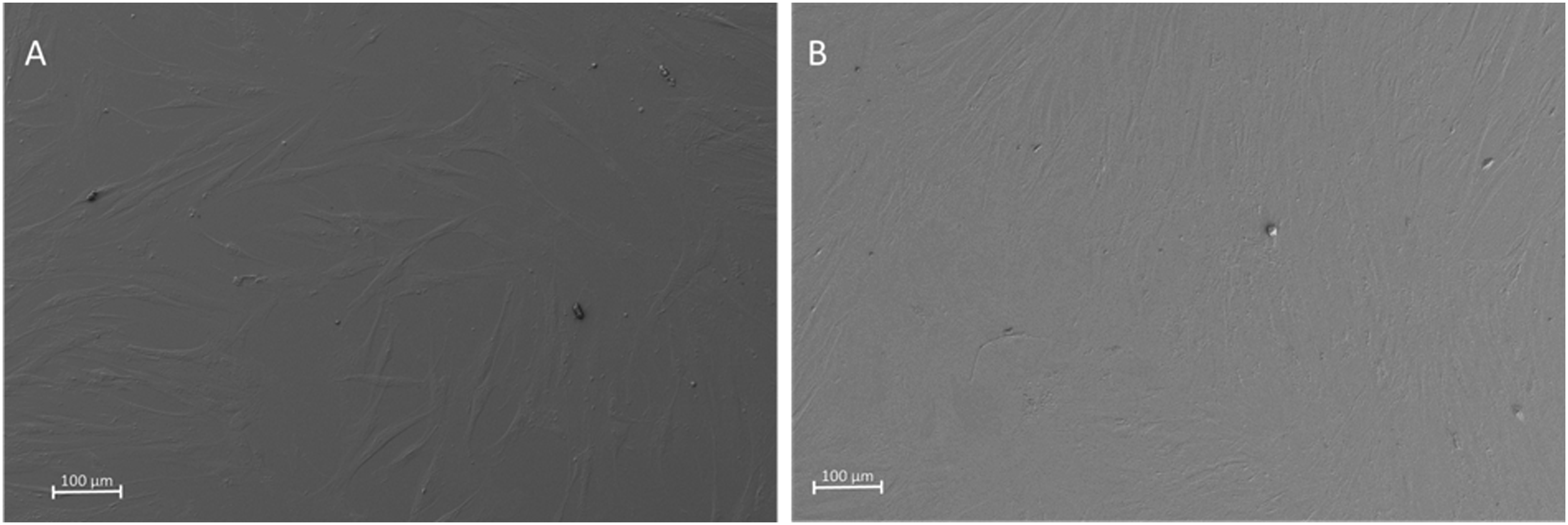
At measurement 2 fibroblasts on the un-coated plain TiAl6V4 surface (S1) were nearly confluent and appeared much more elongated when compared to day 1. Cells seeded on the plain TiN-coated surface (S2) at this second measurement also had nearly reached complete confluence and showed the similar increasingly elongated appearance but tended to reach not quite the cell density given by the S1 specification (Figure 9(a)–(b)). A-D SEM images of HDFa cells seeded on sample discs and incubated for seven days. Cell behavior on the plain topographies of specification S1 (a) and S2 (b) as well as on the 3-D porous topography specifications S3 (c) and S4 (d) is displayed at 30x magnification.
On the un-coated 3-D topography (S3) cells also had proliferated but to a much lesser degree. Their gross appearance was in parts similar to day one with a spindle-like morphology, while others revealed a polygonal or triangular shape with lamellipodia and filopodia. All cells on this topography adhered to the structured surface as well as bridged small cavities or gaps (Figure 9(c)).
The TiN-coated 3-D topography (S4) also revealed proliferation of fibroblasts but cell density appeared considerably decreased when compared to S3 at this measurement. While distribution patterns were comparable to the S3 specification, polygonal or triangular cells with lamellipodia and filopodia appeared only incidentally and in most cases in a bridging manner over gaps between the primary structure of the topography (Figure 9(d)).
Cell alignment occurred predominantly according to the rods and bridges of the topography following the longitudinal axis of the respective structural element but incidentally appeared also oriented according to micro-structural gaps within the structural elements.
Discussion
Complications of TOPS focus on the area of the skin-penetration-site with high impact on functionality of the prosthetic system and concomitant patient satisfaction. 8 Soft-tissue in-growth into the skin-penetrating prosthetic component (abutment) to re-establish the barrier function of the skin appears to be a promising approach to address this issue. As for current TOPS abutment components besides cobalt-chromium-molybdenum alloys primarily titanium alloys are used, in this study we evaluated a 3-D-manufactured TiAl4V6 topography (EPORE®, implantcast GmbH, Germany), originally intended for osseous in-growth, in an un-coated as well as a TiN-coated specification regarding its impact on fibroblast adhesion, proliferation, in-growth and basic viability in comparison to plain TiAl4V6 surfaces. Both the un-coated and TiN-coated EPORE® surface specifications revealed a homogenous cell distribution accompanied with a flat and spread cell morphology forming filopodia on day 1 and day 7 of incubation. Proliferation and a trend towards confluence were seen on un-coated EPORE® surfaces with ongoing incubation but appeared substantially lower on the TiN-coated EPORE® specification. Cell morphology as well as fast proliferation on the plain surfaces indicated a tendency towards migration and thereby to a lack of adhesion strength. This tendency was significantly less pronounced on the EPORE® topography, where cells tended to remain in place with slower proliferation and less migration activity. Overall while cell viability on both EPORE® specifications was comparable to plain TiAL6V4 as well as polystyrene controls, cell proliferation and confluence were less pronounced.
Limitations of this study focus on the descriptive character of the study design. Lacking statistical data increases the potential of an investigator bias impact. Furthermore additional evaluation of cell viability and metabolism as well as cell density would allow for a more in-depth assessment of fibroblast adhesion. Moreover the short incubation period does not allow for analysis of the tissue building potential of the adhered cells but provides information on the initial adhesion and proliferation behavior.
Titanium alloys are a widely used biomaterial providing osseointegration in orthopedic applications. This scope has been widened to dental implants which require both, osseointegration and soft-tissue on- or in-growth. 11 Along with advances in the development of additive manufactured interconnecting porous topographies, recent approaches further extended the field of application to pure soft-tissue integration.12,13 Against this background in this study we evaluated a 3-D-manufactured TiAl6V4 alloy topography, intended for osseointegration, which is applied in clinical orthopedic routine, with regard to its potential for fibroblast adhesion and proliferation. The live-dead ratios of all TiAl6V4-specifications at both measurements indicate that there are no reasonable cytotoxic effects caused by the titanium alloy at this early stage of cellular adherence. This is in line with the results of current in-vitro studies reviewed by Willis and co-workers (2021). They concluded that an unchallenged TiAl6V4 component, meaning in pH neutral environment without mechanical stress, has little to no cytotoxic effect on fibroblasts whereas impacts like corrosion or wear lead to increased cytotoxic effects on fibroblasts in-vitro when compared to commercially pure titanium due to the subsequent release of aluminum and vanadium particles. 14 To what extend this effect can be extrapolated to a clinical, i.e. in-vivo, setting, remains questionable.
To address this issue TiN-coating may be an option to mask the titanium oxide layer of the abutment, and improve the surface characteristics while maintaining titanium biocompatibility. 15 The coating is intended to enhance fibroblast cell proliferation, attachment and differentiation, as well as to reduce bacterial adherence and biofilm formation. 15 The latter appears to be a critical functionality especially in the initial healing stage of the implant-soft-tissue interface. Enhancement of cell proliferation by TiN-coating could not be confirmed in this study. The slightly increased wettability of TiN-coated samples had no positive effect on cellular capture of the surface as in fluorescence microscopy as well as in scanning electron microscopy the TiN-coated specifications revealed reduced cell density when compared to un-coated TiAl6V4 specifications. This is in contrast to other studies that revealed comparable or enhanced fibroblast cell density on TiN-coated titanium alloys when compared to un-coated titanium alloy specimens.15,16 Ritz and co-workers (2017) on the other hand detected no positive or adverse effects of TiN-coating on human gingival fibroblast behavior in-vitro. 17 With regard to the versatile potential of TiN-coatings this issue should be subject to further investigations.
Compared to 3-D-topographies and even polystyrene controls, the considerably higher proliferation rates on plain TiAl6V4 specifications indicate that fibroblasts adhere less strongly onto this surface specification 18 in favor of an increased tendency to migrate. This is underlined by the morphologic changes towards a significantly enhanced elongation of the cells while reaching nearly complete confluence. In an extended observation period we would expect the confluent cell layer on plain TiAl6V4 surfaces rather to detach as can be observed in a clinical setting in-vivo where soft-tissue attachment to a plain titanium alloy surface does not occur. On 3-D-topographies in contrast human dermal fibroblast adhered in a more stationary manner. The cells exhibited a spread polygonal or triangular-shaped morphology and formed filopodia and lamellipodia, which provided a more stable attachment. The biological behavior of cells is closely related to surface topography, roughness, hydrophilicity and texture of the respective implant material.19–21 Identical material composition of the plain as well as the 3-D-manufactured TiAl6V4 surfaces point out that the described morphological differences are due to the 3-D-topography. Tolksdorf and co-workers (2020) underlined that fibroblasts need a sort of irregular surface of synthetic or artificial materials for an uncomplicated integration process. 22 The random porosity and micro-as well as nanostructure of the 3-D-surfaces seem to be in line with this requirement and may be seen as promising baseline for the development of a vitalized, sealed implant-soft-tissue interface.
Conclusion
The EPORE® topography allows for fibroblast adhesion and viability in both standard TiAl6V4 and – to a minor degree - TiN-coated specifications as a proof of principle.
Footnotes
Acknowledgments
We thank the Stiftung Universitätsmedizin Essen for supporting this study. Furthermore, we thank the Imaging Center Essen (IMCES) for their technical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Stiftung Universitätsmedizin Essen.
