Abstract
Background and Purpose
Structuring scaffold with both osteogenic and angiogenesis capabilities is a challenge for bone tissue engineering. Powder structured Si-CaP materials have shown excellent osteogenic properties and induction of stem cell differentiation. Our research group have successful produced 3D printed Si-CaP scaffolds by DLP technology. This study aims to explore the angiogenic effects of SiO32- and Ca2+ released by 3D printed Si-CaP scaffold, and whether there is a synergistic effect between the two ions.
Methods
The 3D printed Si-CaP scaffolds were immersed in endothelial cell medium solution for 24 h. The Si, Ca ion released was detected by Inductively coupled plasma–optical emission spectrometry. We used detected data as a standard to prepare the simulated solution to investigate the effect of SiO32-, Ca2+ separately. Experiment was divided into control group, Si ion group, Ca ion group and Si + Ca ion group. We evaluated different ionic effect on HUVECs viability, proliferation, migration, gene expression, and tube formation on different groups.
Results
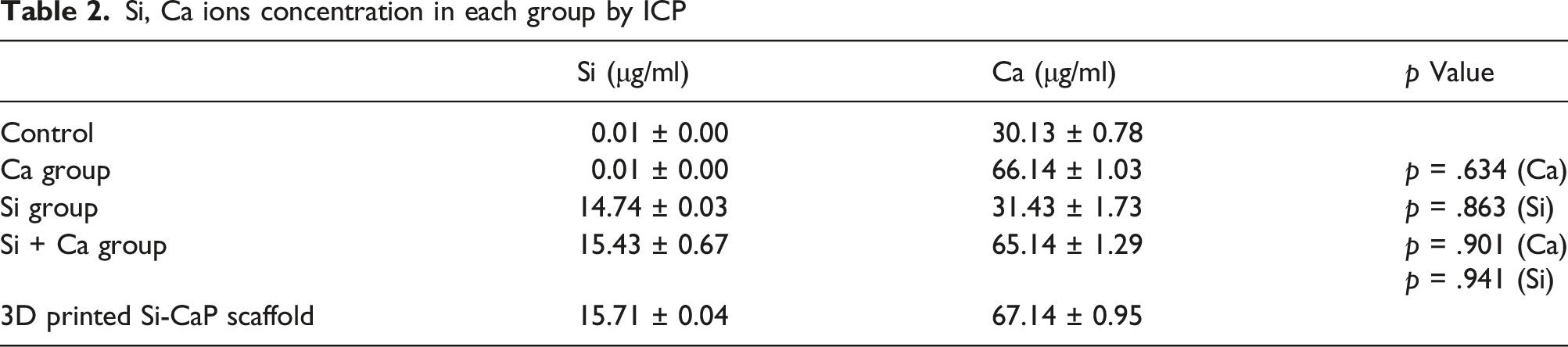
The concentration of SiO32- was detected as 15.71 ± 0.04 μg/mL, Ca2+ as 67.14 ± 0.95 μg/mL. Na2SiO3 and CaCl2 were used to prepare the simulated solution. There were no statistically difference between simulated solution from ion released by scaffold. Si + Ca group promoted the gene expression significantly compared with the control group, p < .01. Expression of vascular-associated protein in Si + Ca ion group was higher than that in Si ion group, Ca ion group and control group. Si + Ca ion group significantly enhanced endothelial cell on migration and tube formation assay.
Conclusion
The 3D printed Si-CaP scaffold can release effective physiological concentrations of Si, Ca ions. Si and Ca ions have a synergistic effect on promoting angiogenesis of HUVECs. 3D printed Si-CaP scaffold is promising in vascularized bone tissue engineering application.
Introduction
Trauma, infection, tumours and bone necrosis can result in severe bone defects. 1 Bone tissue engineering is a promising therapeutic approach to reach the regeneration of bone defects. 2 Tissue-engineered scaffold with angiogenesis capacity can provide nutrients and transport metabolic waste for cells in the centre of bone defects, thereby enhancing the osteogenic capacity of scaffold. 3 However, structuring tissue-engineered scaffold with both osteogenic and angiogenic capabilities is a challenge for bone tissue engineering.
Angiogenesis is the key to bone tissue repair. It involves the migration, proliferation, differentiation of endothelial cells and formation of a vascular network at last. 4 The newly formed vessels deliver growth factors, cytokines and progenitor cells to the center of damaged tissue. Current methods of vascularized scaffolds include ①using co-culture of various cell types ②external growth factor provision ③3D bio-printing ④ microfabrication ⑤ microfluidic techniques. 5 Although many researches have demonstrated some interesting methods to promote angiogenesis of bone tissue engineering, there is currently no simple, efficient method to enhance the angiogenic capacity of endothelial cells.
Si-CaP, a material has been proven the ability to exhibits excellent osteogenic properties and induces stem cell differentiation. 6 But powder shape Si-CaP showed poor ion release abilities and lacked a three-dimensional spatial structure conducive to angiogenesis. It is not favourable to completely unfold the excellent osteogenesis of Si-CaP in powder form. 7 With the advent of DLP 3D printing technology, our research group has successfully produced 3D printed Si-CaP scaffold. 8 DLP 3D printing technology greatly increased the porosity of the scaffold, enhanced adhesion ability of bone cells. This study will focus on the effect of SiO32- and Ca2+ released from 3D printed Si-CaP on the formation of blood vessels.
In this study, 3D printed Si-CaP scaffolds were prepared according to previous articles. 8 And its ion-releasing ablility was tested. In order to investigate the effect of SiO32- and Ca2+on blood vessel formation separately, SiO32-, Ca2+ were added to the endothelial cell medium to match the level of ions released from the scaffold. This study can demonstrate the advantages of Si-CaP materials in angiogenesis and clarify the specific effects of each ions on angiogenesis.
Methods
Manufacture of 3D printed Si-CaP scaffolds
As we introduced in our previous research, we synthesized Si-CaP and printed as we designed. 7 Briefly, Si-CaP powder Premixed with photosensitive resin (PEGDA, 3,3-dimethacrylic acid and polypropylene glycol), photoinitiator (2,4,6-trimethylbenzoyl)diphenylphosphine oxide (TPO) and dispersant (DCA-1228) to prepare different light-curableceramic slurries with solid phase ratios. Next, the CAD data were loaded in the molding control software, and the light-curable ceramic material was superimposed layer by layer using an ultraviolet laser. Third, once the layer was fully cured on the platform, a certain amount of liquid resin was used to form the next layer. These steps were repeated until a complete 3D printed scaffold was generated. 8
Detection of ion release from 3D printed Si-CaP scaffold carriers
Weigh approximately 30 mg of 3D printed Si-CaP scaffold carriers, wash them in deionised water, sonicate them for approximately 5 min to thoroughly clean the surface of the material and then dry the washed scaffolds in a blast box at 50°C. The treated scaffold material was immersed in a centrifuge tube with 10 mL of endothelial cell medium and set at 37°C. After 24h the supernatant night of the centrifuge tube was taken. The supernatant was filtered and the content was tested using dilute nitric acid solution configured to be weakly acidic, and the Si and Ca ions were quantified using Inductively coupled plasma–optical emission spectrometry (ICP–OES, iCAP 7400, Thermo Fisher Scientific)
Preparation of the simulated solution and ion detection
Preparation of SiO32- simulated solution
The Si ion content of the simulated solution was approximately 15.71 ± 0.04 μg/mL within 24 h of the test. Take a beaker and add 50 mL of deionised water, heat it continuously and add 1.71 g of Na2SiO3 to it and dilute it 10 times with deionised water. 9 Add 0.1 mL of the diluted solution to 5 mL of endothelial cell complete medium. After 24 h in a 37°C incubator and again the ion assay was performed
Preparation of Ca2+ simulated solution
The Ca ion content of the scaffold extract within 24 h of the assay was approximately 67.14 ± 0.95 μg/mL. As Ca ions are soluble, they were dissolved without heating. 50 mL of deionised water was added to a beaker and 0.26 g of Cacl2 was added. 0.1 mL of the diluted solution was added to 5 mL of endothelial cell complete medium. After 24 h in a 37°C incubator and again the ion assay was performed. Finally the ion concentration data of the three groups 3D printed Si-CaP scaffold carriers were compared statistically.
Subsequently, experiment was divided into control group, Si ion group, Ca ion group and Si + Ca ion group. The culture medium without any extracts of the Si-CaP was considered as a negative control.
Human umbilical vein endothelial cells (HUVECs) culture
HUVECs (Lonza Walkersville, In) were thawed and subcultured in 25-cm2 Corning® cell culture flasks with canted neck and vented caps following manufacturer's protocol (Technical Information, 2014). Endothelial cell growth media 2 (EGM-2; Lonza Walkersville, In) was used for the HUVECs' expansion, and the media was changed every 2 days until the cells reached 70% confluence. Cells from Passage three were used on all the designed experiments.
Cell viability
The effect of different ions on the growth and cell proliferation of HUVECs was determined using a standard colorimetric MTT assay (Gibco, USA). The cells were seeded in ECM in a 96-well plate for 24 h. Next,the culture medium was replaced with a simulated medium containing the different ions for 24h and 48 h. The cells containing MTT were incubated in a humidifified atmosphere of 5% CO2 at 37°C for 4 h.Terminate cultivation and carefully discard the culture supernatant in the well. Add 150ul of DMSO (Sigma-Aldrich, USA) to each well and shake for 10 min to fully dissolve the crystals. The absorbance of the violet-colored solution, which correlated to the viability of cells, was read using Synergy H4 Hybrid Multi-Mode Microplate Reader (Synergy HT, BioTek, USA) at 570 nm. The experiments were conducted three times.
Cell migration assay
HUVECs cells of the 3rd-5th generation were inoculated in 24-well plates at 5 x 104 per well and cultured until fusion. A 200 μL pipette tip was used to make a scratch and a prepared mock-up of each group was added to each well. The original edge was photographed and marked using an inverted light microscope. The cells that moved from the original wound edge over a 12 h period were counted from the photographs. Finally cell migration within the scratched area at different time points was analysed by Image J software. The number of cells migrating per unit area and the cell migration rate were calculated. The migration rate was calculated as: migration ratio = (original scratch area - final scratch area)/original scratch area. The experiment was repeated 3 times for each set of conditions.
In vitro tube formation assay
Matrigel matrix gel was removed from the freezer at −20°C and thawed, placed on an ice box and subsequent experiments were performed on ice. After curing, 2 x 104 cells per well were inoculated and subsequently incubated with different mimetic solutions. Images were taken with a light microscope at 0, 8 and 24 h into the cell culture as they were taken to assess the tubular shape of the HUVECs. The mean values of the tube formation parameters, including number of nodes, number of grids and tubule length, were counted for the three regions using Image J software.
Protein expression analysis
Western blot analysis was used to measure the protein expression of KDR and VEGF in HUVECs. Cells were cultured on different ion vulture medium for 24 h. After 24 h of incubation on different scaffolds, HUVECs were lysed to100 μL of RIPA lysate was added. The supernatant containing the total protein was collected after centrifugation. Total protein was collected after centrifugation and protein concentration was determined using the BCA kit. Protein concentration was determined using the BCA kit according to the manufacturer's instructions. Manufacturer's instructions. The samples were then subjected to SDS electrophoresis and transferred to polyvinylidene difluoride membranes (0.5 mm). difluoride membrane (0.45 μm, Millipore, USA). The membranewas blocked with 5% skimmed milk for 1 h and then incubated with the primary antibody overnight at 4°C. After three-times washes with TBST, the membrane was incubated with the secondary antibody for 30 min. Incubate with the secondary antibody for 30 min. The membranes were immersedin chemiluminescence reagent (ECL kit) and observed after immersion (ECL kit) and the relative intensity of the protein bands was calculated using ImageJ software.
Gene expression analysis
Primers used for qRT-PCR.

Statistical methods
The data obtained from the experiments were analysed using SPSS 25.0 statistical software, and the results were expressed in X ± S. One-way analysis of variance (ANOVA) was used to analyse the results of ion concentration, qRT-PCR, protein electrophoresis, and tubule formation for each group, followed by the Fisher post-hoc test. Independent t-tests were used to compare between the two groups, and p < .05 was considered statistically significant, and p < .01 was considered to have significant difference. The analysed experimental data were plotted using GraphPad prism nine software.
Results
Detection of ions released from 3D printed Si-CaP scaffold and simulated solution
Si, Ca ions concentration in each group by ICP
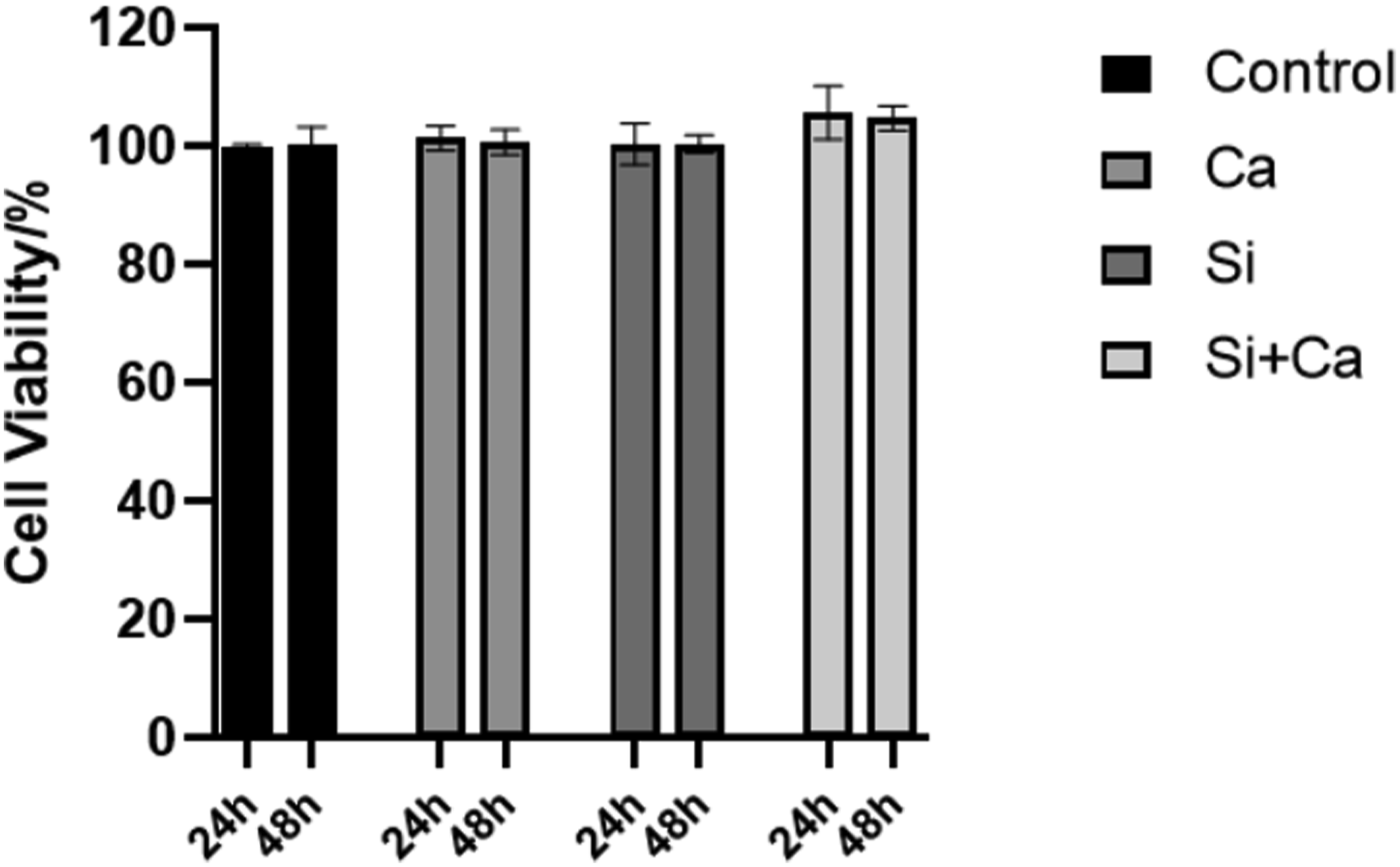
Results of MTT assay on different simulated solution. Each solution have no detrimental effect on the growth and proliferation of HUVECs.
Cell migration experiment
In Figure 2 (a), it can be observed that both the Si and Ca ion groups were able to promote the migration of HUVECs compared to the control group. After processing by Image J software, the migration rate and the number of migrated cells were statistically different between the Ca ion group and the Si ion group compared to the control group, and there was no statistical difference between the two groups. Si + Ca group promoted the migration ability of HUVECs most significantly compared to other three group the control group, p = .00076; there was also a statistical difference compared to Si or Ca groups, p = .014. HUVEC migration in the scratch assay.(a) Images of HUVECs in each experimental group at 12 h. All white dashed lines indicate the width of the scratch gap at 0 h. The orange dashed line is the position of the last cell movement, magnification is 100×. (b) and (c) Quantitative analysis of the number of migrating cells and their migration rate after processing by Image J software, with statistical differences in each group compared to the control group. * indicates p < 0.05 compared to the control group, ** indicates p < 0.01 compared to the control group.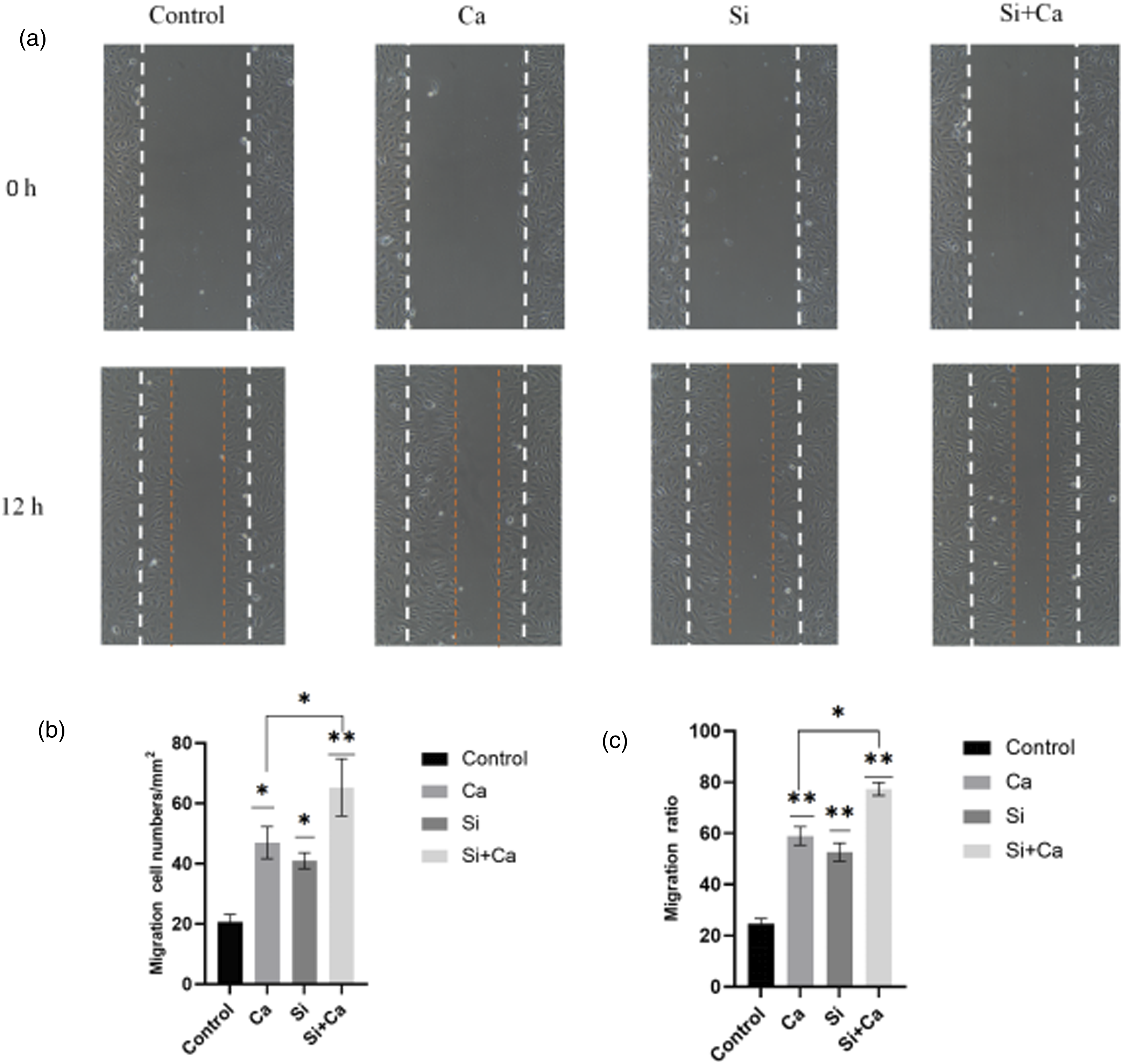
Tube formation experiments
Tube formation of HUVECs was tested on Matrigel matrix gel in the Si ion group, the Ca ion group and the Si + Ca ion group. 8 h showed no tube formation in the control samples, while tubule formation was shown in other three groups. The number of pores and the number of junctions were significantly superior in the Si + Ca ion group (p = .00013). The extent of tubule formation in the Si + Ca ion group at 8 h was not statistically different from that in the Si,Ca ion group at 24 h. The final Si ion group and the Si + Ca ion group generated two and 2.3 times the number of nodules, 1.9 and 3.1 times the number of mesh pores, and approximately 1.5 and 1.7 times the length of tubules compared to the control. Si ion and Ca ion groups shows statistically significant differences in the number of nodules and mesh pores, p = .028.
qRT-PCR and Western blot experiments
Third generation HUVECs cells were co-cultured with each group of simulated solution until day 3 for cell collection operation. The HIF-1α gene expression in Ca ion group was statistically different from the control group, p = .027. The VEGF, KDR gene expression in Si ion group was increased compared to the control group, p = .033, while the HIF-1α expression was not statistically different. Gene expression of VEGF, KDR in Si + Ca ion group was significantly increased compared to the control group, p = .000023, and also statistically different compared to the Si ion and Ca ion groups, p = .0048; the gene expression of HIF-1α was also statistically different compared to the control group, p = .036. The gen expression of each group can be shown in Figure 2.
The protein expression of each group can be shown in Figure 3. The expression of VEGF protein and its receptor KDR, the most important protein in the vascularization process, was significantly higher in the Si ion group, the Si + Ca ion group and the scaffold carrier extract group, and the Si ion group had a stronger ability than Ca ion to promote angiogenesis at the gene and protein expression aspects, and the two ions could synergistically promote the expression of angiogenic genes and proteins. Tube formation assay. (a) At 8h the control group had only a few points of attachment and had not yet formed a complete closed mesh.Ca, Si ion group nodes are significantly more numerous, with a few closed mesh formations. The Si+Ca ion group already had the beginnings of a vascular network visible, and the number of nodes and closed pores was significantly higher.The vascular network of the Si+Ca ion group at 24h had a significant advantage in terms of number of nodes, number of closed meshes and length of tube formation (d), (c) and (d) are the statistical graphs of nodes, closed meshes and tubule lengths plotted by Image J software. * indicates p < 0.05 compared with the control group, ** indicates p < 0.01 compared with the control group.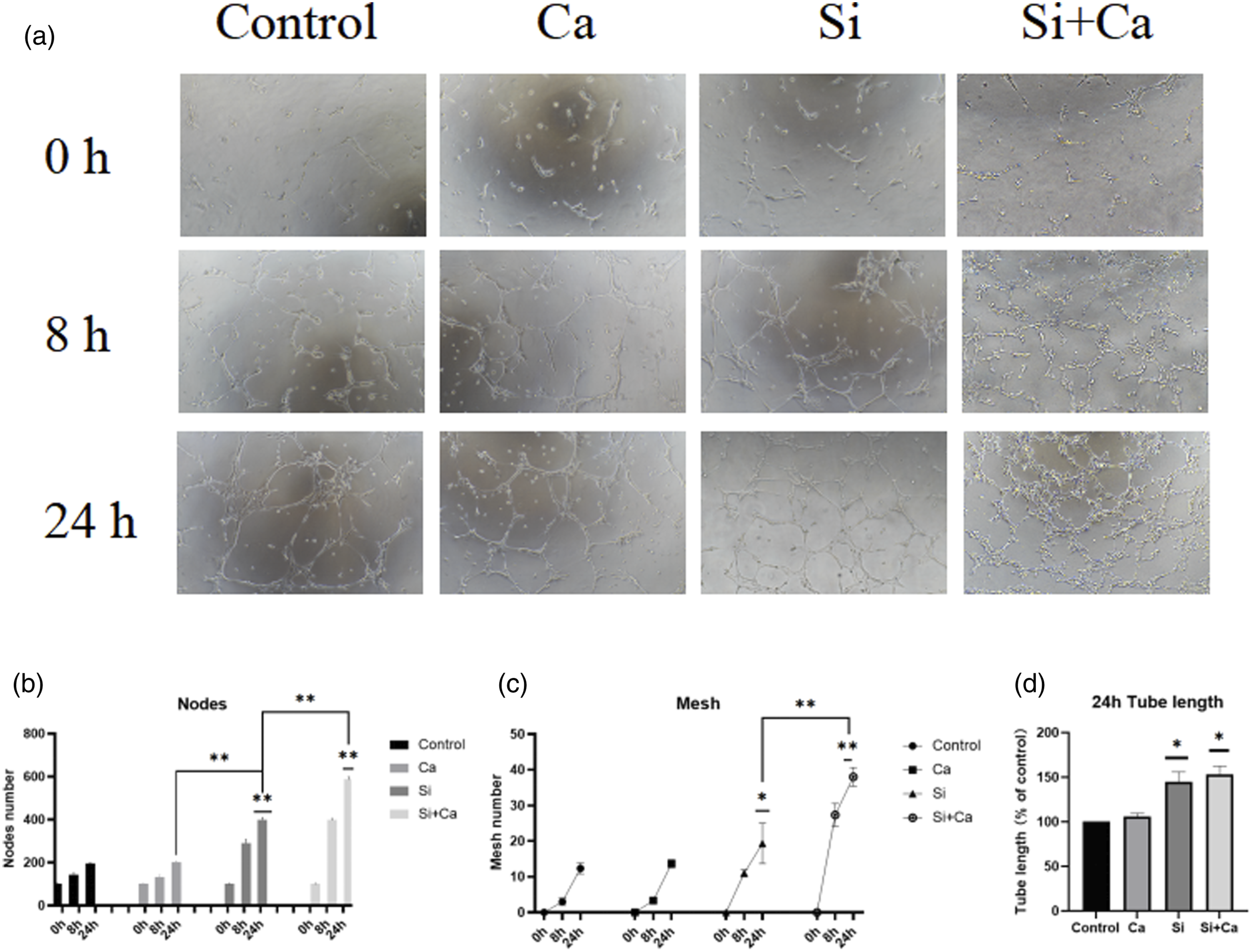
Discussion
Angiogenic effect of SiO32-
According to the process of chemical synthesis of Si-CaP [Si-Ca10(PO4)6(OH)2], in which the element Si partially replaces the element phosphorus in CaP. 6 In this experiment Na2SiO3 was added to simulate the SiO32-. There is no standard way of writing about the Silicon ion. In fact, as a 4-valent cation, Si4+ itself is extremely unstable in solution; in other words, a cation of 4-valent Si alone is almost non-existent in solution; the more stable form of Si present in solution would be the SiO32- ion. The reason why some articles do not express it in this way is probably because the mainstream detection means such as ICP–OES、Silicon electrodes, which detects the amount of Si present in this solution, does not prove that this Si element is present in the form of SiO32-, so many articles express it in terms of Si ions. 10
Among the studies of various silicon-containing materials, the release of SiO32- on endothelial cells to promote angiogenesis has been a hot issue in recent years. 11 Zheng et al. showed that high concentrations of Si can impair autophagy and induce apoptosis in HUVECs, and autophagy may play a protective role in HUVECs from apoptosis. Furthermore, the concentration of Si used in HUVEC should not exceed 1 mmol/L. 12 This study provides a therapeutic concentration range reference for the use of SiO32- in biomaterials. Li et al. used CS extracts to stimulate the expression of VEGF in HUVECs co-cultured with HDF and enhanced the expression of VEGFR-2, which in turn increased endothelial-type nitric oxide production and ultimately initiated angiogenesis. 13 During this process, the expression of endothelial calmodulin from HUVECs were up-regulated and calmodulin was concentrated at cell junctions to promote tube formation. SiO32- have been shown to play an important role in the induction of angiogenesis by silicate bio-ceramics. The effective silica ion concentrations was 0.7-1.8 μg/mL reported in Li and Chang’ studies. Silicate content in CS is 45%, The original CS extract needs to be diluted 64-128 fold to reach a physiological concentration suitable for the cell. However, such dilutions do not simulate the realistic environment of endothelial cells during in vivo angiogenesis.
The experimental results showed that the concentration level of SiO32- released from the 3D printed Si-CaP scaffold can effectively promote angiogenesis in HUVECs. Different concentration control of SiO32- was not set in this study, we focus on the concentration released from the 3D printed Si-CaP scaffold. It should be pointed out that only considering the optimal ion release concentration of bone tissue engineering materials in terms of angiogenesis is one-sided. As a good biomaterial, the effect of this concentration on Ossification should also be considered. In the future, groups with different concentrations of SiO32- should also be set up to explore the optimal ratios of Si content in the scaffolds.
Angiogenic effect of Ca ions
As can be observed in Figure 2, Ca2+ and SiO32- have same ability in promoting cell migration. There is no statistical difference between these two groups. However, the promotion of cell motility is likely to be more important way for calcium ions to promote endothelial cell angiogenesis compared to gene and protein expression. 14 Oscar increases the release of Ca2+ in solution by regulating the silicon content of silicon-based calcium phosphate glasses. With the silicon content equaled to approximately 40%, Ca2+ release concentration of 2 mmol/L-3 mmol/L, the Ca2+ released allowed MSCs and endothelial cells to exhibit greater osteogenic and angiogenic capacity. 4 Aguirre seeded the cells on a matrix gel layer and cultured them with Ca2+ for 20 h to assess their tube formation capacity. The tube formation test was enhanced by induction, suggesting a positive role for calcium in angiogenesis. This phenomenon could be blocked by anti-CaSR antibodies, suggesting an association with CaSR-like receptors. 15 Ca2+ and VEGF enhanced cell migration ability suggesting activation of migration-related pathways. This suggests that calcium ions are likely to be a downstream pathway molecule for VEGF. Many materials have sought to be an effective tool for the regulation of calcium release, playing a key role in promoting angiogenesis. 16
In the present experiments, the results of the Ca2+ group were not as significant as those of the Si ion group, where the Si ion was a case of “nonexistence” to “existence” and the Ca2+ was a case of “less” to “more”. The gene expression of VEGF and KDR was only twice as high as in the control group but was not statistically different from the control group in the tube formation experiment. This may be explained by the fact that the ion assay shows that the endothelial cell basic medium itself contains a certain concentration of Ca2+, so the effect of Ca2+ alone is not so obvious and the exact effect of Ca2+ may be difficult to observe at the cellular level of the experiment. This is agreed with the findings in Oscar Castaño's experiments. 4 In Aguirre's experiments, no significant angiogenesis was observed with the addition of calcium ions alone, but the blocking effect of the adding anti-CaSR antibodies was significant. 15 Ca2+ may not be the primary stimulus for these events, however, it is an essential downstream effector molecule or signalling mediator. 17 The Ca2+ content in tissues of living organism is not evenly distributed as in cell cultures, and there is a relative lack of nutrition located at the centre of bone repair until the vascular network is formed, so adding Ca2+ to scaffold is essential. In the future we need to conduct further in vivo experiments in animals to further validate the effect of SiO32-, Ca2+ released from 3D printed Si-CaP scaffolds on the formation of blood vessels.
Synergistic effects of Si, Ca ions
Expression of HIF-1α-the upstream initiation gene, VEGF the most critical protein in angiogenesis, was remarkable in the Si + Ca group (Figure 4). This was observed in in vitro matrix gel tube formation assays. The Si + Ca ion group formed a vascular network essentially equivalent to that of the other groups at 8 h, with a more significant difference at 24 h (Figure 3(a)). After analysis by Image J software, there was a significant increase in the total length of the points, circles, and vessels within the vascular network (Figure 3). . The Si + Ca ion group was able to rapidly form a vascular network, which could greatly improve the survival rate of osteoblasts/stem cells at an early stage and ensure the repair of the bone defect. Gene expression of VEGF (a), HIF-α (b) and KDR (c) in different groups. * indicates p < 0.05 compared with the control group,** indicates p < 0.01 compared with the control group.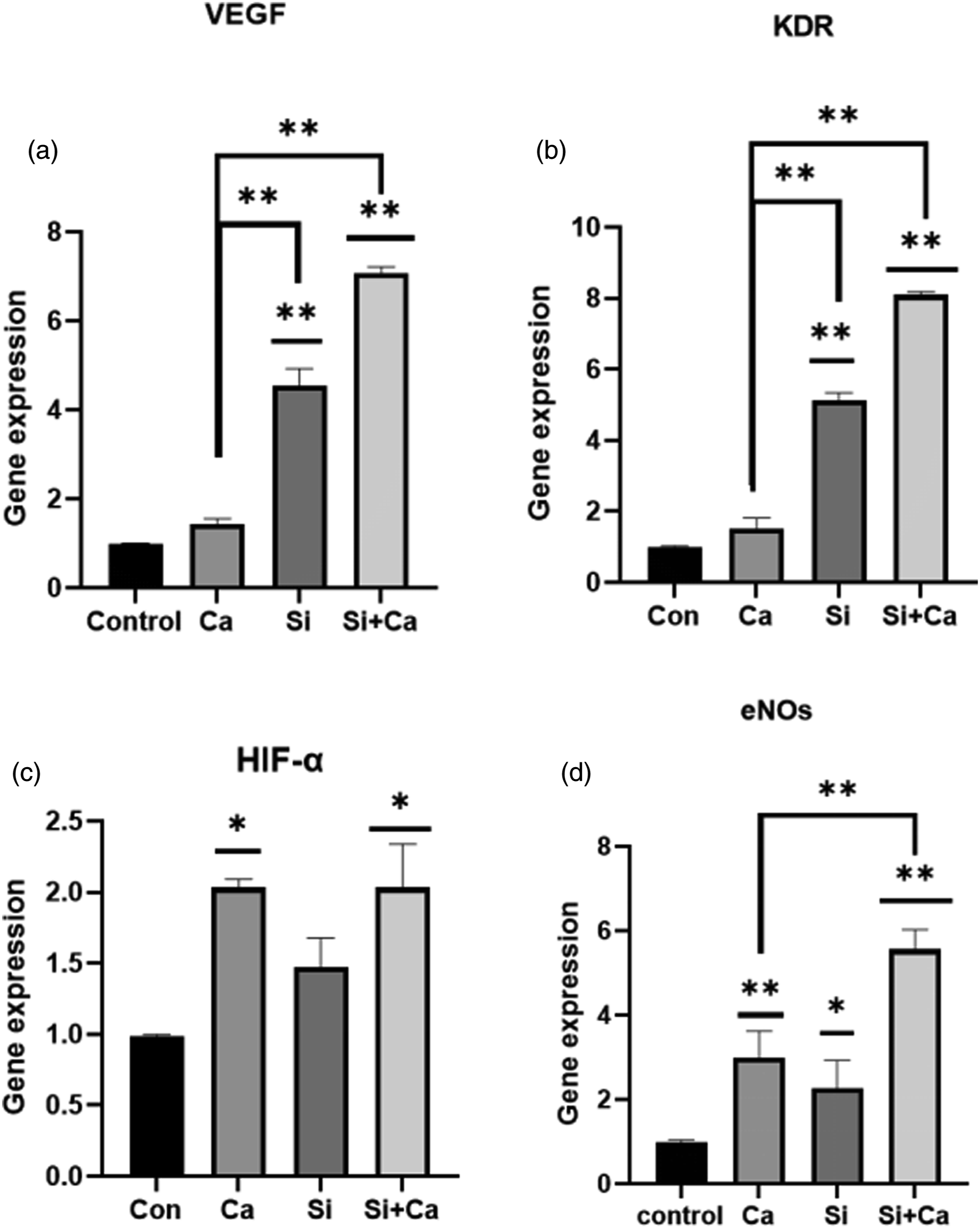
Figure 5 As the most important signal pathway in angiogenesis--HIF-1α.
18
Si ions replace Fe ions and change the phenotype of PHD2, allowing HIF-1α to combine with HIF-β, activating the HIF pathway and greatly increasing the expression of VEGF and KDR.
19
VEGF activates eNOs in endothelial cells, opening Ca ion channels in the cell membrane and endoplasmic reticulum, prompting endothelial cells to migrate and divide and proliferate
20
]. The increased expression of KDR allows the endothelial cells to amplify the pro-angiogenic effect of VEGF again, forming a cascade effect. Adding both ions to the scaffold material promotes the formation of the vascular network and provides sufficient ‘resources' for the pre-endothelial cell foundation work in the early nutrient-poor defect center. In previous studies of biomaterials, such as CS, which contain not only Si ions but also Ca ions in their extracts, the focus has been on the promotion of Si ions on blood vessel formation at different concentrations, neglecting the role of Ca ions in angiogenesis.
21
In this study, the role of both ions on vessel formation was investigated separately by simulated solution, which provides a theoretical basis for the design of vascularized scaffolds in the future.
22
Protein expression of VEGF (b), KDR (c) in different groups. * indicates p < 0.05 compared with the control group, ** indicates p < 0.01 compared with the control group.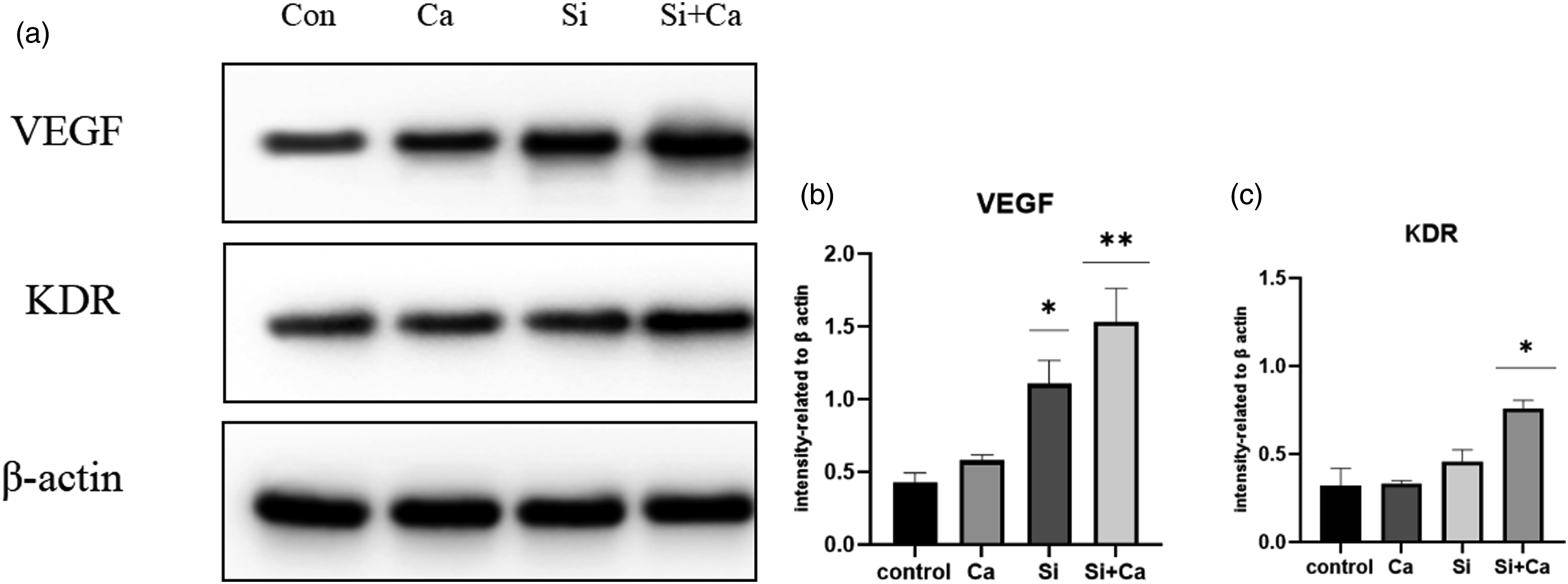
Advantages of 3D printed Si-CaP scaffolds
As many materials do not contain SiO32-, how to introduce Si into scaffold materials and the way it acts on cells is currently a major research hotspot for various new scaffolds. Duan Wei designed a micron-sized Si particle that can release silicate ions when its surface is released with water and can carry VEGF growth factor, which can effectively It can promote endothelial cell angiogenesis. 23 The release of extra Si microparticles and VEGF protein is under risk of temporary high local concentration. The release method of SiO32- by 3D printed Si-CaP scaffold is natural dissolution, controlled by a reversible chemical reaction of Si ion dissolution to stabilise the rate, avoiding the risk of localised high concentrations. The SiO32- contained in the bioglass material in Chang's study were too high for the original solution to be suitable for endothelial cell’s growth and required a 128-fold dilution to achieve an effective concentration. Many studies have shown that SiO32- have a negative effect on endothelial cells at concentrations above 1 mmol/L, with a significant decrease in proliferation and angiogenic capacity. 12 The SiO32−and Ca2+ released by 3D printed Si-CaP were percisely in an effective concentration and form.
Summary and outlook
In summary, this study revealed that 3D printed Si-CaP releases SiO32- and Ca2+,the ion concentrations matches the effective physiological concentration. Two ions have a synergistic effect in promoting the expression of angiogenic-related genes and proteins in HUVECs and the formation of vascular networks in vitro. However, the drawback of this experiment is that the grouping of different concentrations of SiO32−and Ca2+ was not set up. The optimal concentration of SiO32- and Ca2+ is not yet uniformly concluded. The present experiment illustrates the synergistic effect of SiO32−and Ca2+ released from 3D printed Si-CaP in promoting blood vessel formation, without an in-depth study of the specific mechanism. It needs to be further explored in future experiments.
Conclusion
The 3D printed Si-CaP scaffold can release effective physiological concentrations of SiO32−and Ca2+. SiO32−and Ca2+ have a synergistic effect on promoting angiogenesis of HUVECs. 3D printed Si-CaP scaffold is promising in vascularized bone tissue engineering application.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nature Science Foundation of Heilongjiang province of China (JQ2020H003).
