Abstract
The objective of this study was to coat negatively charged polymer brushes covalently onto the surface of thermoplastic polyurethane (TPU) using a simple conventional surface free-radical polymerization technique. The coated surfaces were assessed with contact angle, protein adsorption, cell adhesion and bacterial adhesion. Bovine serum albumin (BSA) and bovine fibrinogen (BFG) were used for protein adsorption evaluation. Mouse fibroblasts (NIH-3T3) and Pseudomonas aeruginosa (P. aeruginosa) were used to assess surface adhesion. Results show that the TPU surface modified with the attached polymer brushes exhibited significantly reduced contact angle, protein adsorption, and cell as well as bacterial adhesion, among which the negatively charged polymers showed the extremely low values in all the tests. Its contact angle is 5°, as compared to 70° for original TPU. Its BSA, BFG, 3T3 adhesion and P. aeruginosa adhesion were 93%, 84%, 92%, and 93% lower than original TPU. Furthermore, the TPU surface coated with negatively charged polymer brushes exhibited a hydrogel-like property. The results indicate that placing acrylic acids using a simple surface-initiated free-radical polymerization onto a TPU surface and then converting those to negative charges can be an effective and efficient route for fouling resistant applications.
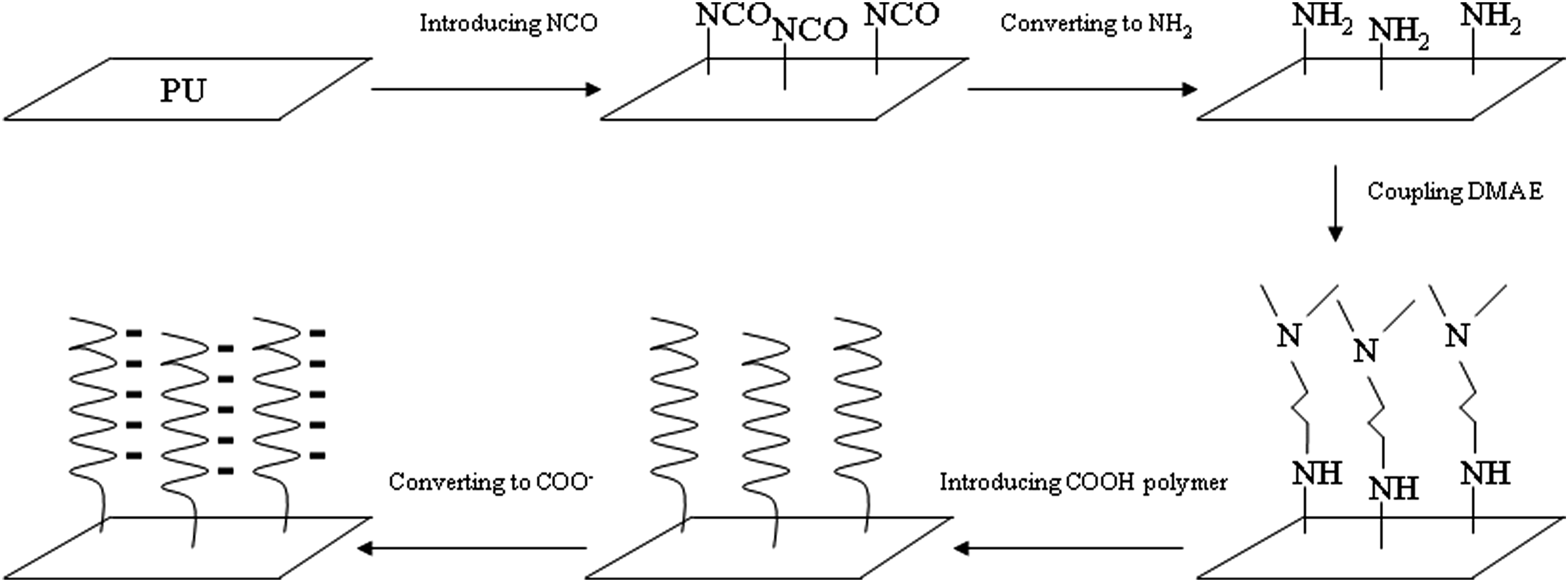
Introduction
Polyurethane (PU), due to its biocompatibility and unique elastic mechanical properties similar to blood vessels, has been successfully used as popular biomaterials, particularly in cardiovascular and urological biomedical areas for a long time.1–3 Its applications include but are not limited to catheters, artificial vascular grafts, dialysis pumps, etc.1,2 However, there exist two main issues that limit the use of polyurethane materials in certain medical areas: (1) its inherent hydrophobicity when it is in contact with blood and/or body fluid 4 and (2) association with bacterial contamination or infection when bacteria attach and proliferate on surfaces. 5 The hydrophobic surface can lead to nonspecific protein adsorption,6–8 cell adhesion9,10 and bacterial adhesion.11,12 There are numerous reports on polyurethane surface modification for decreased protein adsorption and cell adhesion.13–24 Some are focused on making entire hydrophilic or amphiphilic polyurethane polymers before forming a medical device 24 but some are focused on surface modification of the formed medical device.13–23
Generally speaking, interface between medical implant device and body fluid or tissue play a key role in biocompatibility of medical device. There are two major types of implant applications, one requires integration and the other requires antifouling. For dental and orthopedic applications, interface between implant device and tissue requires integration on physical, chemical and cellular levels, meaning that material must bond or integrate to tissue.1,25–27 On the other hand, in the case of applications such as cardiovascular and ophthamological medicines, interface between device and tissue or surrounding body fluid demands non-fouling or antifouling. In other words, no or minimum proteins or cells or even bacteria are allowed to attach to medical device.1,28–31 While biomaterial implants or devices such as PU graft are used inside the body, their surface is the main target for cells, proteins and other biological species.25,27 In addition, their surface is also likely contaminated or infected with bacteria or biofilm.25,28,29 It is well known that most synthetic polymeric materials are susceptible to cell, protein and bacterial attachment when being implanted, due to their hydrophobic nature. 1 To enhance survival rates of implanted biomaterial devices in service and reduce potential protein, cell or bacterial adhesions, surfaces of biomaterials must be treated to minimize potential protein, cell and bacterial attachment.1,32
To make an anti-fouling surface or reduce cell and protein attachment, surface needs to be modified at least to meet one of the following conditions 33 : (1) extremely hydrophilic, (2) bearing charges which can against charges carried by proteins or cells and (3) bearing lipid-like molecules such as liposome or water-soluble protein molecules such as albumin. To prevent or minimize bacterial contamination or biofilm formation, two strategies are often applied: (1) providing a highly hydrophilic surface which can minimize bacterial attachment and thus reduce the risk of bacterial contamination or biofilm formation,32,34 and (2) making an antibacterial surface which contains an antibacterial agent or a polymer that can either inhibit bacterial growth or kill bacteria.1,35
To find wider application for PU to better serve patients who need these medical devices, PU surface must be better modified. To modify surface of PU, its surface ought first to be activated. There are several ways to activate PU surface among which high energy irradiation including γ-radiation and electron beam, ultraviolet, plasma, ozone treatment, etc. have been extensively studied and applied. 36 However, most of them are either very expensive or show a limited surface area that can be treated.1,36 The wet chemistry surface treatment is another way to activate the surface.36,37 The main concern for this technique is the integrity of the surface after surface activation, because many chemicals or solvents used for the activation are organic and very polar, which may damage the surface. 36 PU has no direct reactive functional groups which can be easily modified to form chemical bonds and make polymers coated on the surface. The indirect functional group, urethane group can be activated through a urethane hydrogen group.33,38 Our prior research has demonstrated that we can successfully activate the PU surface without damaging the surface, using a non-destructive strategy.38,39 To coat a hydrophilic polymer onto the activated surface via a chemical surface modification, three strategies have been applied40,41: (1) attaching a preformed polymer to a surface through pendant reactive groups from the polymer backbone via a multi-point surface reaction, resulting in a polymer chain laying down on the surface38,40,41; (2) attaching a preformed polymer end-capped with a reactive group to a surface via a single point surface reaction, resulting in a polymer chain extending from the surface,40–42 and (3) coupling an initiator to a surface, followed by introducing a polymer onto the surface by in situ polymerization of monomers, resulting in a polymer chain growing from the surface.40,41 The pitfall for (1) is that surface coverage may not be ideal because steric hindrance between polymer chains may limit access of larger polymer molecules to the surface. The disadvantage for (2) is similar to that for (1), i.e., steric hindrance limiting accessibility of high molecular weight polymers, thus leading to insufficient surface coverage. The potential pitfall for (3) is that the procedure requires initiators to be linked to the surface before growing polymers but length or molecular weight of polymer chains may not be easily controlled. In this report, we would like to use the third strategy to grow a polymer on PU surface. Currently the majority of reports related to surface-initiated polymerization have been focused on atom transfer radical polymerization (ATRP) and reversible addition-fragmentation chain-transfer polymerization (RAFT).36,40,41 However, these advanced techniques are rather complicated as compared to conventional free-radical polymerization. The former requires toxic copper as a catalyst and the reaction to be carried out under an oxygen-free environment36,40 whereas the latter requires synthesis of a new dithioester RAFT agent that needs to be designed in consideration of monomer to be used and reaction temperature.36,40 Furthermore, both may also require very polar organic solvents to help attach the synthesized initiator onto surface, because the ATRP initiators contain bromine/chloride and RAFT agents contain aromatic phenyl rings that are very hydrophobic but polar.36,40,41 The above techniques may cause some surface damage more or less after surface modification although the issue has rarely been discussed in most published reports. In contrast, initiator and co-initiator used in conventional free-radical polymerization techniques may provide an easy and simple route for synthesis and efficient handling. It is our hypothesis that growing a polymer onto PU surface using a newly synthesized surface co-initiator (reducing agent) via a covalent linkage would provide a novel, easy and efficient route for growing polymer brushes on PU surface. We proposed to use a non-destructive strategy, i.e., reaction in water or a water mixture, to attach a conventional free-radical co-initiator onto PU surface.
The objective of this study was to synthesize a free-radical co-initiator (or reducing agent) derivative, attach the derivative onto TPU surface and grow hydrophilic polymer brushes on TPU surface at an environmental temperature, to achieve a goal that makes hydrophobic TPU to become the surface covered with a negative charged hydrogel network to minimize fouling.
Materials and methods
Materials
1,6-Diisocyanatohexane (DIH), dibutyltin dilaurate, potassium persulfate, 4-(dimethylamino)pyridine, 2-dimethylaminoethanol, succinic anhydride, N-hydroxysuccinimide, N,N’-dicyclohexylcarbodiimide, 2-hydroxyethyl methacrylate (HEMA), N-vinylpyrrolidone (NVP), 2-hydroxyethyl acrylate (HEA), N,N-dimethylacrylamide (DMA), acrylic acid (AA), methacrylic acid (MA), poly(ethyleneglycol monoacrylate) (PEGA), 2-methacryloyloxyethyl phosphorylcholine (MPC), sulfobetaine methacrylate (SBM), tetrabutylammonium bromide, acetone, tetrahydrofuran, sodium bicarbonate, sodium carbonate and calcium chloride were used as received from Sigma-Aldrich Co. (Milwaukee, WI) without further purifications. Tecothane (TT-1075D-M, medical grade), a thermoplastic polyurethane, was used as received from Lubrizol Co. (Wickliffe, OH).
Surface coating
Synthesis of succinimidyl 4-(N,N-dimethylaminoethyl)-succinate
A functionalized surface co-initiator, succinimidyl 4-(N,N-dimethylaminoethyl)-succinate (SDMAES), synthesized as shown below. Briefly, 2-dimethylaminoethanol and succinic anhydride were dissolved in tetrahydrofuran in the presence of 4-(dimethylamino)pyridine (catalyst). The reaction was run at 70°C for 5 h to give white crystalline product 4-(N,N-dimethylaminoethyl)-succinate (DMAES) with a yield of 89%, where the acquired 1HNMR data (ppm) are 11.2 (1H, on -COOH); 4.15, 2.52, 2.42, and 2.15 (2H, on -CH2−); and 2.20 (6H, on –CH3) (also see supporting document). Then to the above mixture, N-hydroxysuccinimide and N,N’-dicyclohexylcarbodiimide were slowly added. After completion of addition, the reaction was run at room temperature overnight, followed by filtering the water scavenger N,N’-dicyclohexylurea, removing solvent using a rotary evaporator, and drying in a vacuum oven. The obtained SDMAES was white paste material with a yield of 91%, where the acquired 1HNMR data (ppm) are 4.15, 2.72, 2.52, and 2.01 (2H, on -CH2−); 2.62 (4H, on -CH2−); and 2.20 (6H, on –CH3) (see supporting document).
Surface activation of PU
Surface activation of PU was conducted as shown below. Briefly, PU was dissolved in a solvent mixture of dioxane/tetrahydrofuran. The solution was poured in a glass dish and maintained at 40°C for 24 h. After recovering from the dish, the polymer film was cut to small circular specimens having a dimension of 10 mm in diameter and 0.5 mm in thickness. Then the specimens were transferred to a container containing 1,6-diisocyanatohexane and dibutyltin dilaurate in hexane. The reaction was conducted at 50°C for 2 h, 39 followed by washing three times with hexane and diethyl ether, to have isocyanate groups on the surface – PUNCO. The modified specimens were then placed in a container containing sodium carbonate solution at room temperature overnight to have amino groups on the surface – PUNH2 (see attached FT-IR spectra in supporting document).
Introduction of polymers to activated PU surface
Surface-initiated radical polymer synthesis on activated PU surface was conducted as shown in the following two steps. First, PUNH2 disks were placed in a container having SDMAES in a mixed solvent of acetone and water. After pH was adjusted to 8, the solution was stirred at room temperature for 1 h, followed by washing with distilled water three times. Second, the washed disks were placed in a container containing a monomer such as acrylic acid (AA) and potassium persulfate (a water-soluble free-radical initiator) in distilled water. The container was then degassed with a vacuum and purged with nitrogen back and forth for three times, followed by keeping at room temperature for 2 h and then washing with distilled water three times before analysis or evaluation. Nine hydrophilic monomers including HEA, HEMA, NVP, DMA, AA, MA, PEGA, MPC and SBM were evaluated.
Characterization
The synthesized functional derivative and precursor were characterized with Fourier transform-infrared (FT-IR) spectroscopy and proton nuclear magnetic resonance (1HNMR) spectroscopy. FT-IR spectra were acquired on a FT-IR spectrometer (Mattson Research Series FT/IR1000, Madison, WI). The 1HNMR spectra were obtained on a 500 MHz Bruker NMR spectrometer (Bruker Avance II, Bruker BioSpin Corporation, Billerica, MA) using deuterated dimethyl sulfoxide as solvent.
Contact angle measurement
Surface contact angle of the tested sample was measured with a contact angle goniometer (Kina SL200B, USA). Briefly, the specimens were washed with ethanol and de-ionized water several times and dried in a vacuum oven with a drying agent at 37°C for 24 h before testing. Measurement was conducted at 23°C via the sessile drop method using a 2 μL deionized water drop. 43 Three specimens were used to measure at three different locations for each specimen to obtain a mean value of contact angle.
Protein adsorption determination
The protein adsorption on PVC was carried out using the bicinchoninic acid (BCA) protein assay. Both bovine serum albumin (BSA) and bovine fibrinogen (BFG) were used for evaluation. Briefly, PVC and modified PVC specimens (12 mm in diameter) were soaked in distill water overnight to allow a swelling equilibrium, followed by placing in a well of 24-well tissue culture plate containing 500 μL BSA protein solution (2 mg/mL) and incubated at 37°C for 2 h. 25 Then the specimens were rinsed three times with 500 μL distill water to remove unattached BSA. The rinsed specimens were placed in a new well containing 500 μL of sodium dodecyl sulfate (1.0 wt%) solution and incubated at 37°C for 2 h to strip the BSA adsorbed on the surface if any, followed by performing BCA assay. Finally, the BSA absorbance was measured using a microplate reader at 562 nm, following the published BCA protocol online from Fisher Scientific Inc. The adsorbed BSA was calculated based on the calibration curve from standard BSA. BFG protein adsorption was measured similarly. The original PVC specimen was used as control. Each material or sample was measured in triplicate to obtain a mean value.
Cell adhesion test
Cell adhesion test was conducted following our published protocol. 20 3T3 mouse fibroblasts (NIH-3T3) were used to assess cell adhesion on the PVC surface. First, the modified and original PVC samples (12 mm) were placed in 24-well plate and soaked in 70% ethanol for 1 h under UV light inside the cell culture hood, followed by rinsing three times with PBS. For 3T3 cell adhesion, cells were cultured in a mixture containing high glucose Dulbecco’s Modified Eagle Medium (DMEM, Lonza), supplemented with 10% fetal bovine serum (FBS, Invitrogen) and 5 mg/mL streptomycin as well as 5 mg/mL penicillin (Invitrogen Inc., Singapore). After incubating at 37°C under a humidified atmosphere of 5% CO2 for 48 h, the cells were harvested by adding a 5.3 mM trypsin-EDTA (ThermoFisher Scientific) solution in PBS, followed by centrifuging at 1200 rpm for 5 min, removing trypsin. A cell suspension with a cell density of 5 × 104 cells/mL was prepared to seed each well with 1 mL of NIH-3T3 fibroblasts. Cells were placed for 48 h in a 37°C incubator with a humidified atmosphere of 5% CO2. Then the specimens were transferred to new wells and washed with PBS several times to remove non-adherent cells. Finally, the adherent cells on specimens if any were detached with trypsin and counted. At least three specimens were tested for each material to obtain a mean value. To be able to visualize the attached cells on the surface, the same experiment was repeated except that each specimen was transferred to a new well where the side of the attached cells was in contact with the bottom of the well. Phase contrast images were taken using an inverted microscope (EVOS FL, AMG, Mill Creek, Washington).
Bacterial adhesion test
The bacterial adhesion test was conducted following published procedures with slight modification. 20 The original and modified PVC specimens were prepared as disks with 6 mm diameter for the bacterial adhesion study. Each sample was placed in the bottom of a well of 96-well cell culture plate and sterilized with 70% ethanol for 10 s and washed with sterile PBS trice. Pseudomonas aeruginosa (P. aeruginosa) was chosen as the model for bacterial adhesion and a suspension of 108 CFU/mL was prepared in Luria broth (LB) Miller. Each specimen was cultured in 200 μL of P.A suspension and placed in a 37°C incubator. After 24 h of incubation, each sample was transferred to a new well, washed with sterile PBS trice, transferred to a sterile tube containing 200 μL sterile PBS, and sonicated in a water bath for 10 min. Serial dilutions were made for agar plate colony counting. Also, 90 μL of the detached bacteria suspension was mixed with a mixture of 0.5 μL of SYTO 9 (green, fluorescent nucleic acid stain, live) and propidium iodide (red fluorescent nucleic acid stain, dead) (1:1) in order to visualize cells using a fluorescence inverted microscope (EVOS FL, AMG, Mill Creek, Washington). At least three samples per material were tested to obtain the mean value.
Statistical analysis
One-way analysis of variance (ANOVA) with the post hoc Tukey-Kramer multiple-range test was used to determine significant differences of each measured property or activity among the materials in each group. A level of α = 0.05 was used for statistical significance.
Results and discussion
Characterization
The synthesis scheme of SDMAES is shown in Figure 1. Surface activation and surface-initiated polymerization are also shown in Figure 1. Schematic diagrams for synthesis of SDMAES and preparation of PU surface coating: (a) synthesis of SDMAES; (b) preparation of PU surface coating.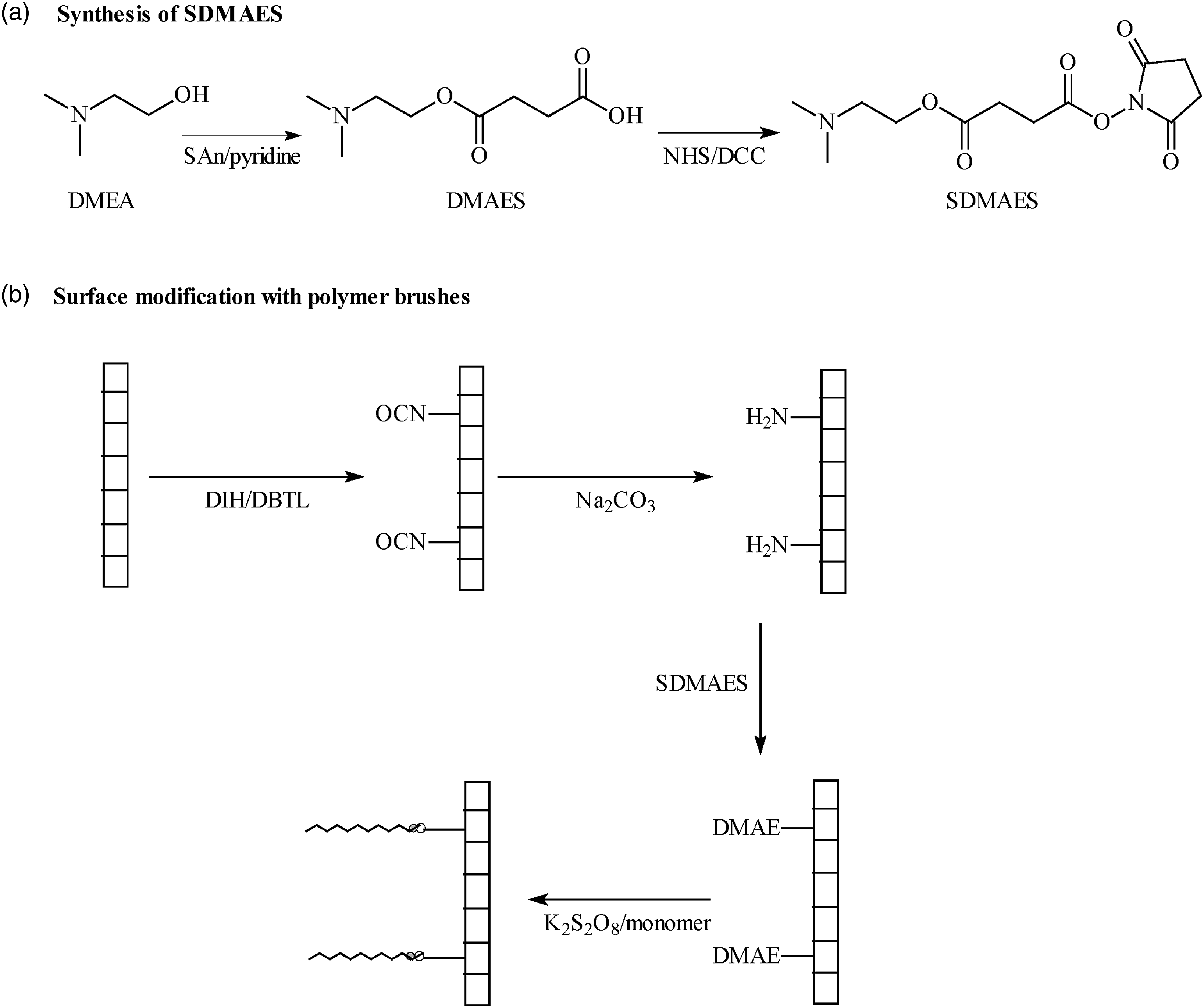
Figure 2 shows a set of FT-IR spectra for SDMAES synthesis: (a) DMAE, (b) SAn, (c) DMAES, (d) NHS and (e) SDMAES. By comparison of spectra (a), (b) and (c), two strong peaks at 1866 and 1783 cm−1 for anhydride group from SAn in spectrum (b) disappear from spectrum (c), indicating that anhydride have successfully reacted with hydroxyl group from DMAE in spectrum (a). One broad peak at 3700 to 1800 for newly formed carboxyl group and three new peaks (at 1730 for ester group, 1580 for amide I and 1469 for amine II) appear in spectrum (c), also confirmed the DMAES formation. By comparison of spectra (c), (d) and (e), the broad peak at 3700–2400 cm−1 for the carboxyl group from DMAES in spectrum (c) disappears in spectrum (e). two new peaks at 1818 and 1773 appear in spectrum (e) representing imide group from NHS and possible carbonyl group between SAn and NHS. The peak at 1730 for ester group from DMAES is found to shift to 1703. Furthermore, the peak at 3424 exists in spectra (a), (c) and (e) stands for the tertiary amine (dimethyl amino) group in DMAE and its derivatives. These changes confirmed the SDMAES formation. FT-IR spectra for syntheses of DMAE derivatives: (a) DMAE; (b) SAn; (c) DMAES; (d) NHS and (e) SDMAES.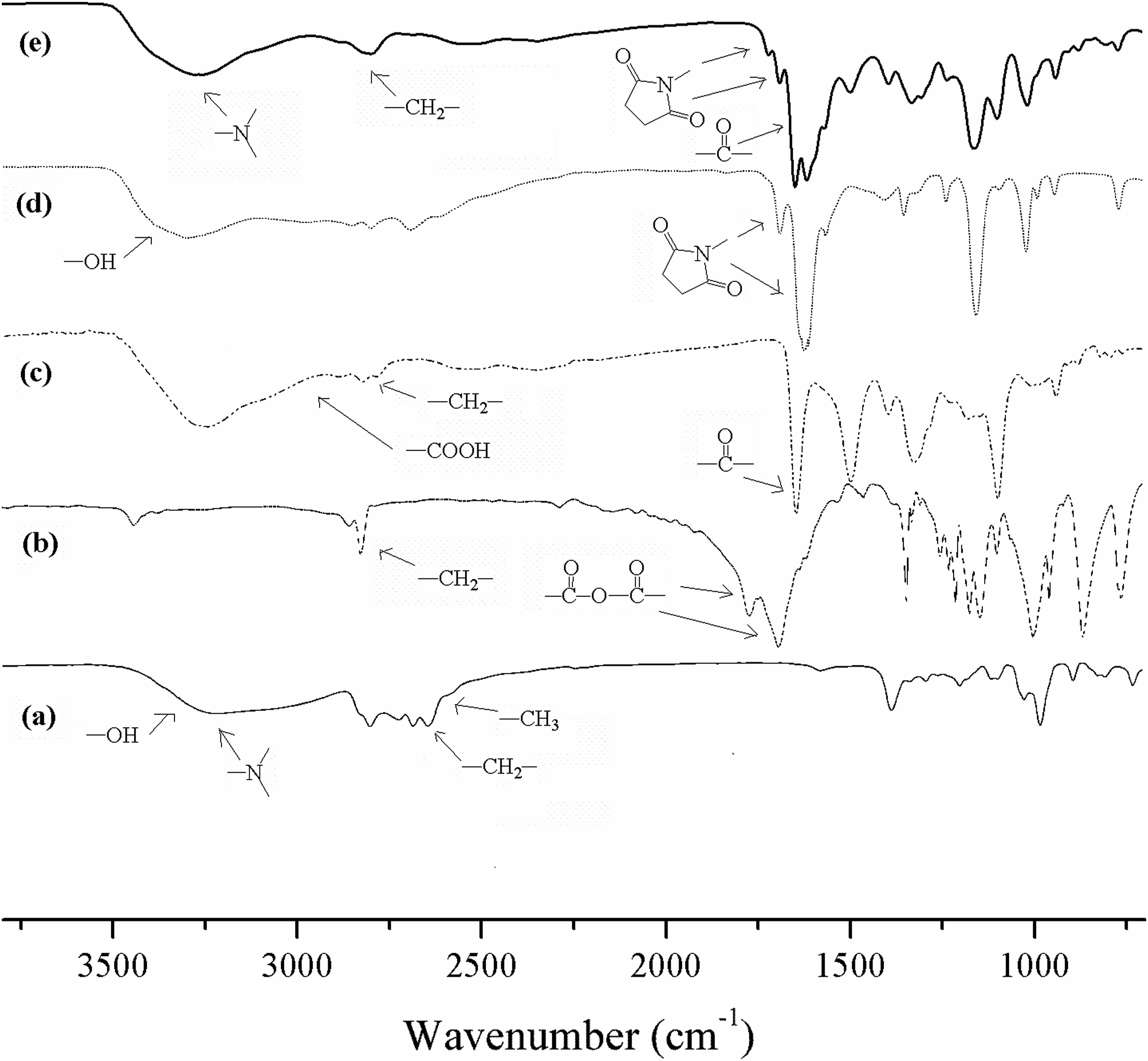
Figure 3 shows the effect of DIH concentration on contact angle of the modified PU surface. To determine the minimum DIH concentration needed to obtain the optimal surface contact angle, we examined the six DIH concentrations at 4, 8, 12, 16, 20, and 24 × 10−4 mmol/cm2. The result of contact angle in Figure 3 shows that no significant differences in contact angle were found before and after forming polymers on surfaces among different DIH concentrations, suggesting that the lowest concentration of DIH at 4 × 10−4 mmol/cm2 should be sufficient enough for making polymer brushes on surface to get a decent low contact angle value. Effect of DIH on contact angle of PU.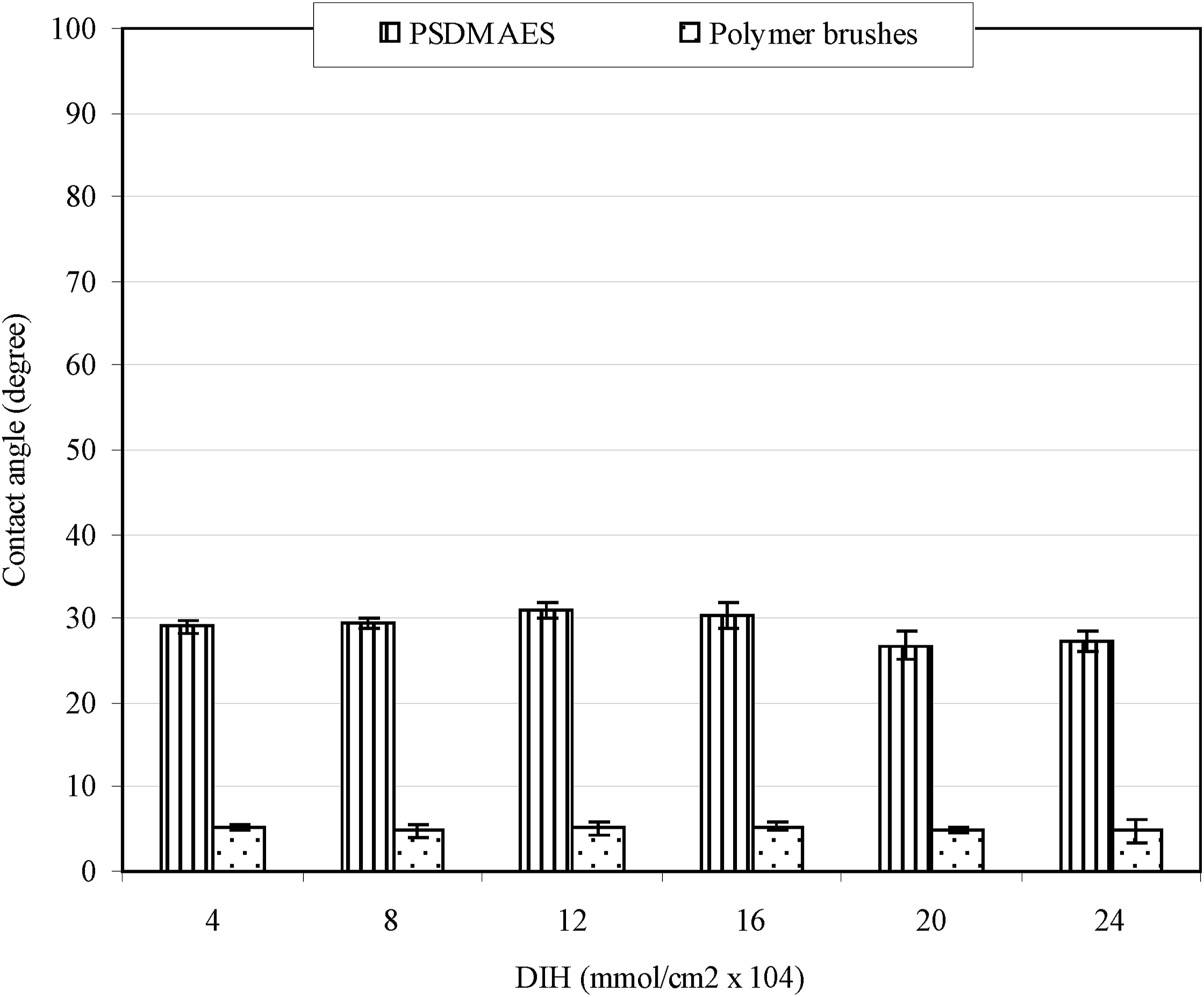
Figure 4 shows effect of nine monomers on contact angle after being introduced onto PU surfaces. It has been found that all the polymer-modified surfaces exhibited significantly reduced contact angles, as compared PU (72.6). SDMAES also showed a much lower contact angle value (25.2) than original PU. That is because SDMAES is tertiary amine molecule, which is also very hydrophilic. In addition to amine groups generated on the PU surface, attaching SDMAES basically does not change surface hydrophilicity much. This may be why significantly low contact angle values were noticed. Among the surfaces modified with polymers, most of them did not present significant surface hydrogel properties, except for PNa. The modified surfaces were hydrophilic but water spread slow and did not completely cover the surface. On the other hand, PNa-modified surface showed water with instant absorption, fast spreading, and complete coverage on the entire surface. That is why the contact angle of PNa was hardly measurable (5.0). It is known that PNa is a salt form of polyacrylic acid, which carries negative charges on the entire polymer chain
44
in a buffer of pH = 7.4. Contact angle values of the surfaces coated with nine common hydrophilic polymer brushes.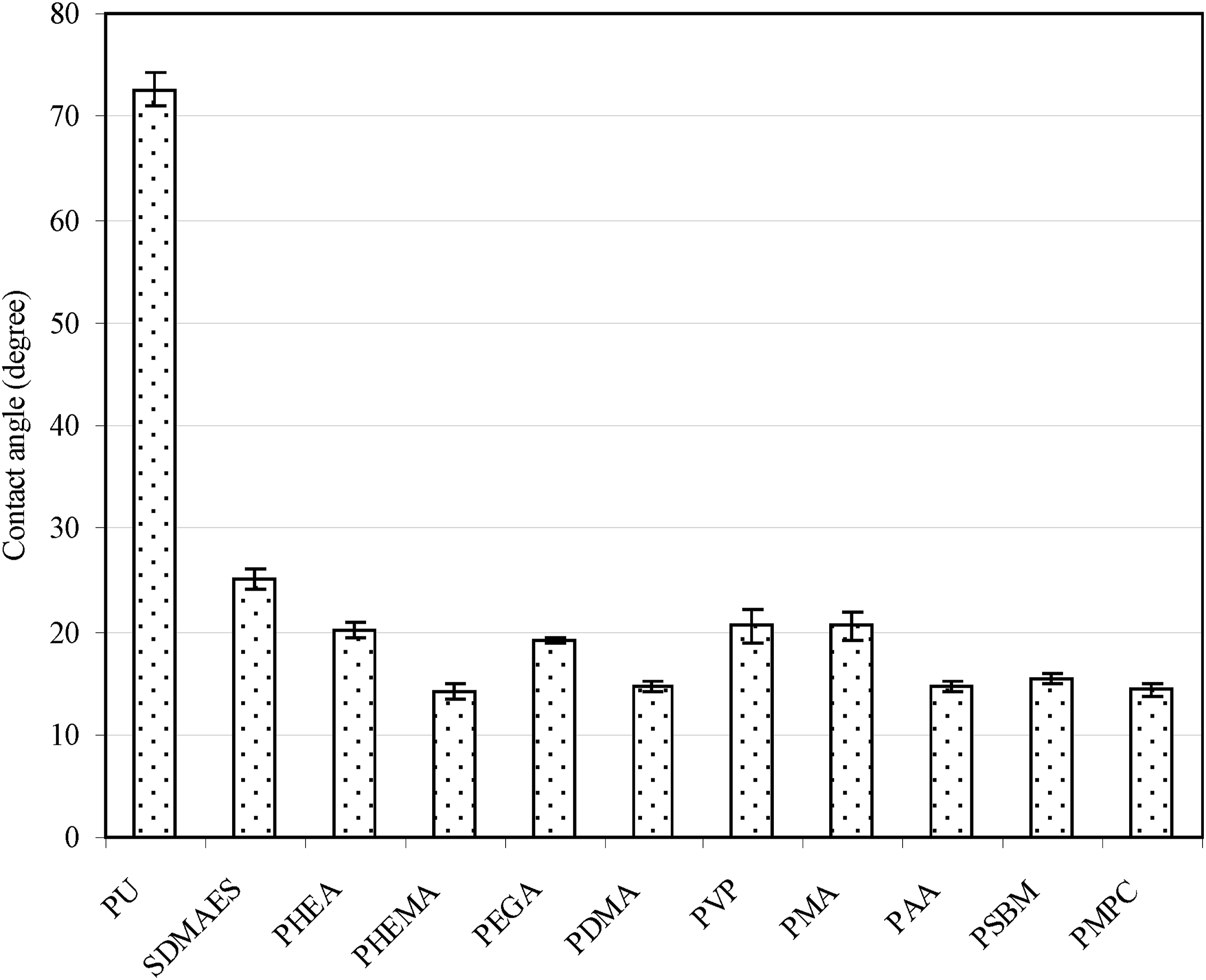
Figure 5 shows contact angle values of original PU and a group of surface modified PUs. The original PU showed the highest contact angle value, followed by PUNCO, PUSDMAES, PUNH2, PUPAA and PUPNa, where no significant difference was found between PUNH2 and PUSDMAES (p > .05). DMAE (dimethylaminoethanol) is a very water-soluble molecule. After being attached to PU surface through a coupling between NHS and amino groups, it might not change its hydrophilic nature of dimethylamino groups. That may be why no contact angle change was noticed. Due to polymer brushes formed on PU surface, PUPAA showed a significantly reduced contact angle (14.7). However, after introduction of salt formation to PAA, the contact angle was even reduced to a lower value at 5.5 (nearly zero) and surface behaved like a hydrogel with instant absorption, fast water spreading and complete water coverage on the surface. We have not seen this phenomenon since we started our surface modification research. Figure 5(b) shows a set of measured contact angle images on PU corresponding to the values shown in Figure 5(a). Apparently, Figure 5(b) (f) showed almost no contact angle that could be measured. During the measurement, it was observed that water was absorbed instantly spread immediately, and completely covered the entire surface, indicating that hydrogel was formed on the modified surface. It is worth to point out that when carboxyl acid groups on PAA are converted to their salt forms, PAA become negatively charged. These negatively charged polyanions not only lead to form a hydrogel but also are clinically important to cardiovascular applications. It is well known that most components in blood are negatively charged.45–51 Blood serum albumin, blood clot protein fibrinogen and hemoglobin are negatively charged because they each carry net negative charges on their entire protein.45–48 Blood platelets and red blood cells are negatively charged.49–51 Blood vessels such as arteries and veins contain negatively charged heparin sulfates and chondroitin sulfates on their inner walls.
33
Negative charges repel each other. That is why normally blood components never attach onto inner wall of blood vessels.
33
The PAA sodium salts coated on the PU surface in this study should also be able to repel those proteins and cells if used in the above application. Contact angle comparison among PU and modified PU surfaces: (a) measured surface contact angle values; (b) photos of measured contact angles corresponding to those shown in (a).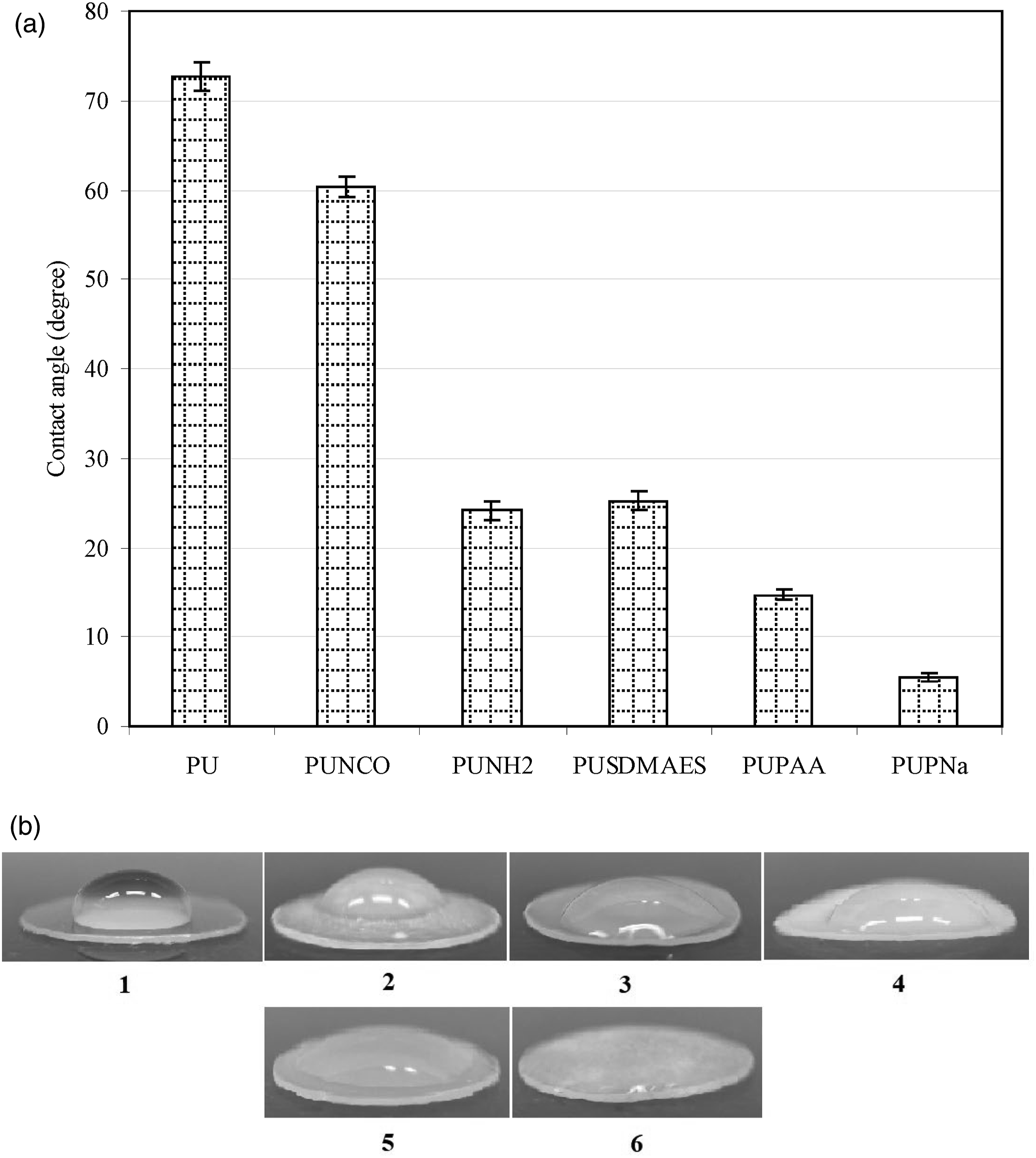
Evaluation
Biofouling is a common issue in implantable or in-body-use medical device applications.1,4,32 Unlike conventional medical supplies which are used outside the body, implantable medical devices being used in cardiovascular or body-fluid contact applications require low cell attachment and protein adsorption.1,3,4 This demands the implantable materials or devices to have an antifouling function, meaning that surface of the devices must be either hydrophilic or antifouling. In last several years, we have been working on PU surface modification with different approaches.38,39 However, none of them could completely diminish protein adsorption and cell adhesion. In this study, we adopted a strategy by growing polymers from the surface to obtain an antifouling surface. 36 Currently the majority of reports related to surface-initiated polymerization have been focused on atom transfer radical polymerization (ATRP) and reversible addition-fragmentation chain-transfer polymerization (RAFT).36,40,41 However, these two types of advanced polymerization synthesis are rather complicated as compared to conventional free-radical polymerization. The former requires toxic copper as a catalyst and an oxygen-free environment36,40 whereas the latter requires synthesis of a new dithioester RAFT agent that needs to be designed in consideration of the monomer and temperature.36,40 Furthermore, both may also require very polar organic solvents to help attach the synthesized initiator onto surface, because the ATRP initiators contain bromine/chloride and RAFT agents contain aromatic phenyl rings that are very hydrophobic but polar.36,40,41 The above techniques may cause significant surface damage after surface modification although it has rarely been discussed in most reports. In contrast, initiator and co-initiator used in conventional free-radical polymerization techniques provide an easy synthesis and efficient handling. In this study, we proposed to use a redox initiator system to help grow polymers onto PU surface. To achieve this goal, we synthesized a reducing agent derivative which can be covalently linked to PU surface. Then we covalently attached this derivative onto PU surface. Via a conventional redox free-radical polymerization technique, we successfully grew polymers onto the PU surface to form hydrogel-like polymer brushes. Redox-assisted free-radical polymerization is very popular in conventional and biomedical areas. Benzyl peroxide-N,N-dimethylaminotoluene is a pair of room-temperature initiators commonly used in dentistry and orthopedics.52,53 With them, dentures and orthopedic bone cements become popular in service. In BPO-DMT, BPO (hydrophobic oxidizer) is believed to be a radical carrier to grow a polymer. 52 On the other hand, in a water-soluble peroxide redox system such as potassium persulfate, reducing agent is believed to be a radical carrier. 54 In a visible light initiation system such as dl-camphoroquinone (photo initiator) and 2-(dimethylamino)ethyl methacrylate (DMAEMA, co-initiator), DMAEMA is said to be a free-radical carrier. 52 The reducing agent DMAE that we proposed to use in this study has a similar structure to that in DMAEMA. However, we do not know if the DMAE attached to the PU surface or potassium persulfate is a radical carrier to grow a polymer. It needs to be proved. Since the surface-initiated polymerization by the proposed redox system is simple and easy to operate, as compared to other advanced techniques such as ATRP, RAFT, etc, this reaction may be proven to be eventually extended to a variety of biomedical applications if successful. Detailed results and discussion are shown below.
To find out which one of potassium persulfate (an oxidizer) and DMAE (a reducing agent) in this study is a free-radical carrier to grow polymers, we made a simple test by mixing potassium persulfate, acrylic acid, PU dicks having DMAE on the surface, and water in a container. After degassing and allowing polymerization for 2 h, we checked both solution and PU surface. By freeze-drying the solution and examining the PU surface, we found that there was no polymer formation in the solution after freeze-drying. Meanwhile, contact angle of the PU surface was dramatically decreased, indicating that the polymer was actually grown on the PU surface. It also indicates that DMAE is a free-radical carrier but not potassium persulfate.
Figure 6 shows the effect of the attached polymer brushes on protein adsorption (BSA and BFN). Both BSA and BFG are the proteins associated with human blood.
46
They both have negative net charges in physiological pH.45–47 Bovine serum albumin has been commonly used to evaluate surface protein adsorption.45,46 Regarding PU as 100%, the BSA adsorption was in the decreasing order of PU > PUPHEA > PUPAA > PUPCa > PUPNa > PUPNaCa > PUPMPC, where no significant differences were found among PUPCa, PUPNa, PUPNaCa and PUPMPC (p > .05). Hydrophobic surfaces have been reported to have higher affinity to proteins, cells and even bacteria.1,3 PU is a very hydrophobic polymeric material. HEA-modified PU showed a significant reduction in BSA adsorption as compared to PU (almost 63% lower than original PU), which is attributed to hydrophilic polymer formation on surface. PAA-modified PU showed an even lower BSA adsorption (83% reduction). On the other hand, the salt forms of PAA showed the lowest BSA adsorption (87 to 88% reduction). The explanation is similar to that discussed in contact angle test. The salt forms of PAA exhibit a strong hydrogel nature, which can help hold water and make the surface extremely hydrophilic, thus showing the lowest BSA adsorption. Finally, PMPC showed the lowest but a similar BSA adsorption to the PAA salts, because PMPC has zwitterions (positively charged N and negatively charged P) on its polymer backbone. In the case of BFG, exactly the same trend is observed but the values are a little bit higher than BSA. BFG is a protein which is closely related to platelet adhesion.
55
It has been reported that platelets in blood adhere to blood fibrinogen more than to blood albumin.
56
Therefore, considerable reduction of BFG adsorption on the modified PU surface may be clinically important in prevention of cell surface adhesion. Although we did not have data for platelet adhesion due to limited access and resource, BFG data can be indirectly used for predicting the reduced risk for potential platelet adhesion. The result from BSA and BFG suggests that direct formation of polymer brushes on PU surface plays an important role in prevention of cell adhesion as compared to post grafting polymer on PU surface, because the former can give better surface coverage than the latter due to steric hindrance effect. It also indicates that hydrogel formation on modified surface is critical to minimize protein adsorption. Furthermore, negative charges from PNa may also play an important role in repel protein adsorption since both BSA and BFG are negatively charged.45–47 Protein adsorptions on original and modified PU surfaces.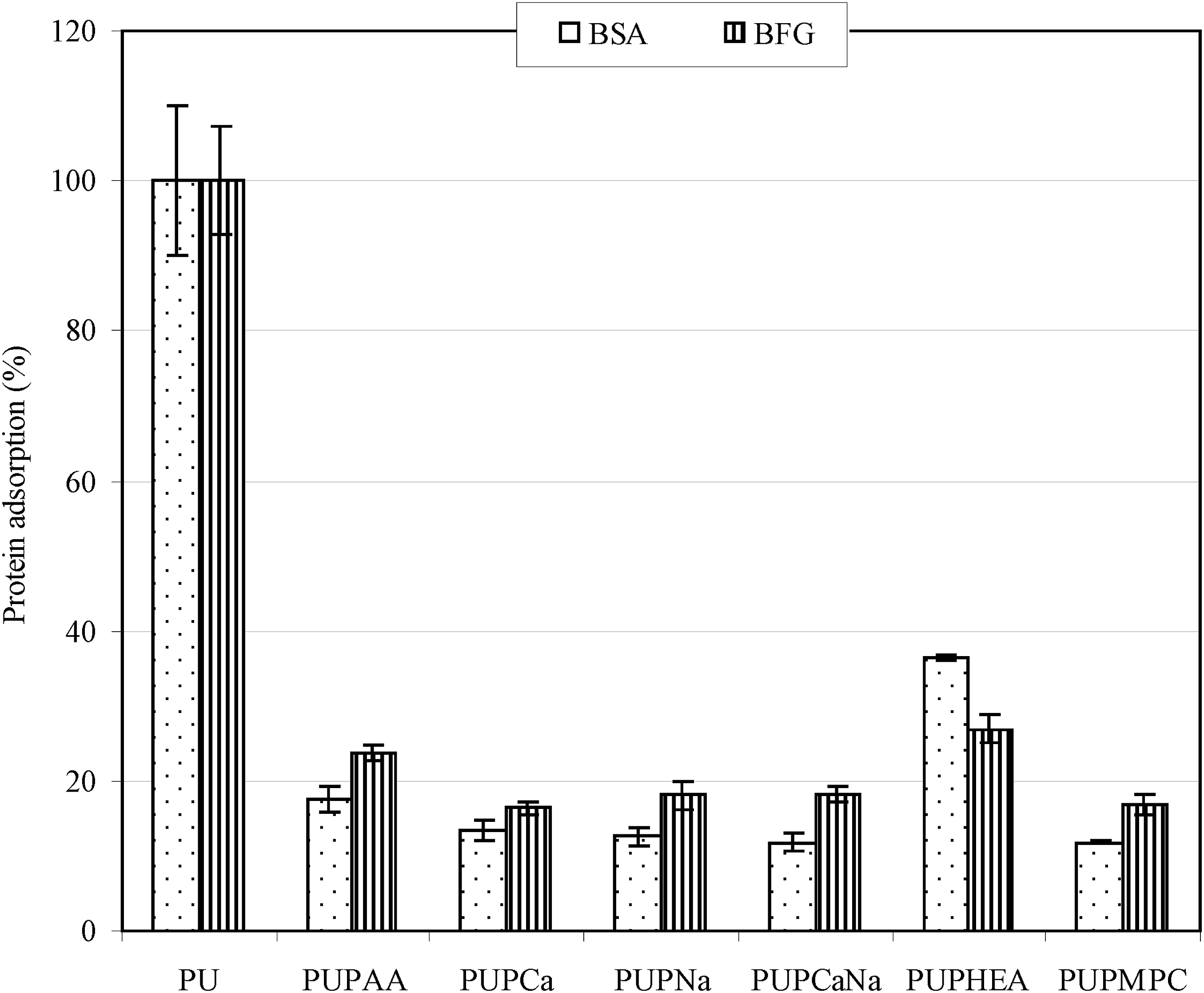
Figure 7 shows the effect of the attached polymer brushes on 3T3 mouse fibroblasts adhesion to the PU surface. The cell adhesion was in the decreasing order of PU > PUPHEA > PUPAA > PUPNa > PUPCa > PUPNaCa > PUPMPC, where no significant differences were found among PUPAA, PUPNa, PUPCa and PUPNaCa (p > .05). Again, hydrophobic surfaces have shown to have higher affinity to proteins, cells and even bacteria.1,3 Cell adhesion is highly dependent upon the matrix protein adsorption.
57
Therefore, cell adhesion usually shows the same trend as protein adsorption does.39,42 PU is very hydrophobic, attracting more matrix protein for adsorption and thus showing higher 3T3 cell adhesion. HEA-modified PU showed a significant reduction in cell adhesion as compared to PU (almost 70% lower than original PU), due to its hydrophilic nature. On the other hand, PAA and its different salt forms were statistically the same in cell adhesion (7 to 11%). We have noticed that PAA showed higher values in both contact angle and BSA adsorption, as compared to its salts (PNa, PCa and PCaNa). The difference between contact angle/protein adsorption and cell adhesion may be attributed to the difference in solution. We used distilled water as a medium to test the contact angle and protein adsorption of the samples whereas we evaluated the cell adhesion on samples in a cell medium which contain both sodium and calcium in 7.4 PBS buffer. In fact, any weak organic acids would automatically convert to their salts when they are added to a cell culture medium. That is why no difference is observed in cell adhesion test. The strong cell repulsion shown on PNa-modified PU surface can also be attributed to negative charges carried on coated PNa chains, since fibroblasts are negatively charged.
58
Same charges against or repel each other. Finally, PMPC showed nearly no cell adhesion (1%) on its modified PU surface. The zwitterion in MPC is the most resistant to cell adhesion. The result suggests that both negatively charged and zwitterion bearing polymer modified surfaces can significantly resist cell adhesion as compared to the surfaces bearing non-charged hydrophilic polymers such as HEA. It also suggests that direct formation of polymer brushes on PU surface plays an important role in prevention of cell adhesion as compared to post grafting polymer on PU surface, because the former can give better surface coverage than the latter due to steric hindrance effect. 3T3 cell adhesions on original and modified PU surfaces.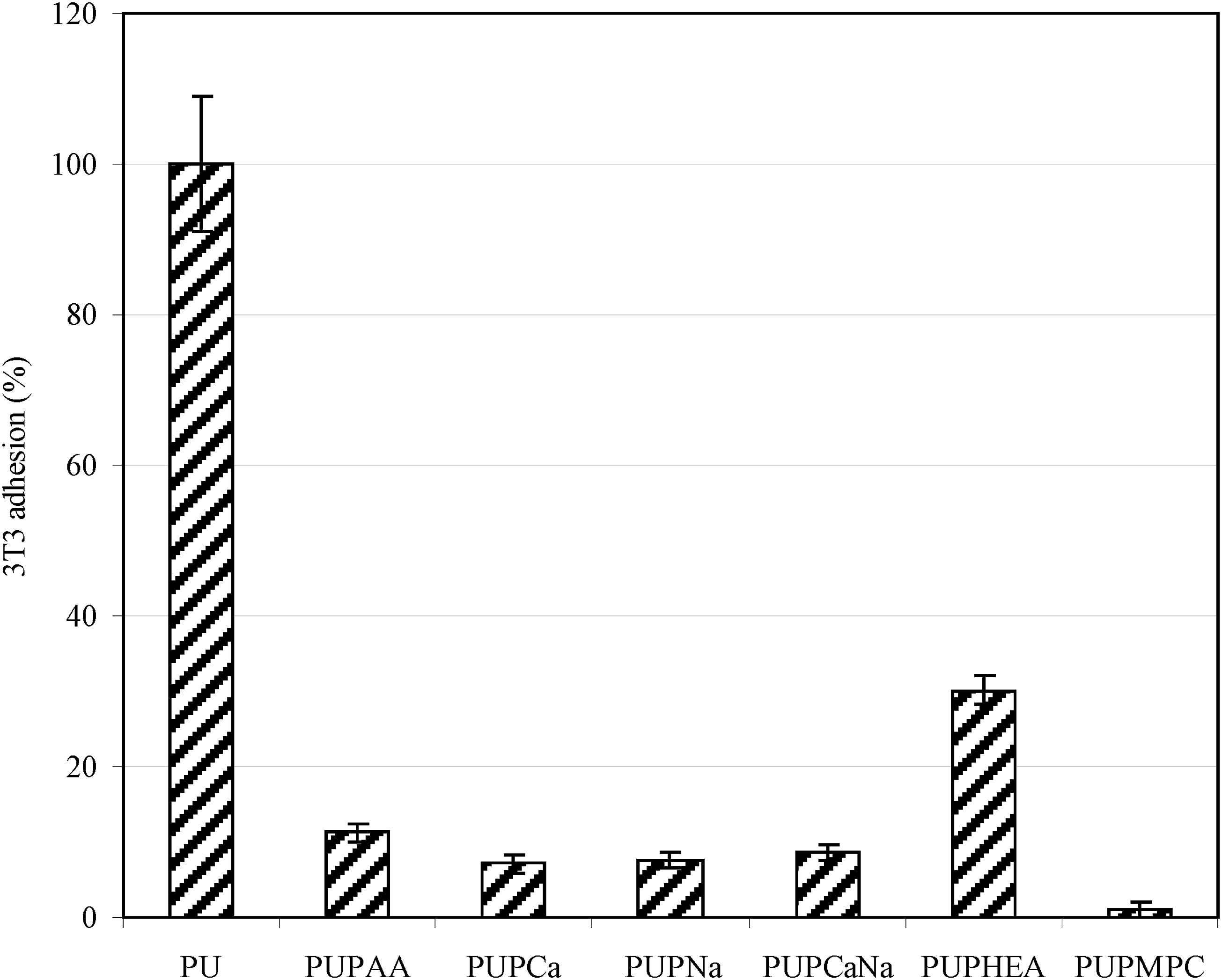
Figure 8 shows the effect of the attached polymer brushes on P. aeruginosa adhesion to the PU surface. The bacterial adhesion was in the decreasing order of PU > PUPHEA > PUPAA > PUPCa = PUPNa = PUPNaCa > PUPMPC, where no significant differences were found among PUPCa, PUPNa and PUPNaCa (p > .05). Except for PU, PUPAA and PUPMPC, all the other modified PU surfaces are made to have negatively charged surfaces, which all showed nearly no bacterial adhesion on the surface. It has been noticed that the surface charges of bacteria are always negative except below pH 2, due to the fact that all bacterial surfaces are rich in phosphate and carboxyl groups.
59
Gram-positive bacteria have negatively charged teichoic acids linked to either the peptidoglycan or the underlying plasma membrane. Gram-negative bacteria have an outer covering of phospholipids and lipopolysaccharides which impart strongly negative charges to their surface. Therefore, the negative charges on biomaterials surface no doubt favor biofouling resistance,
60
because same charges repel each other. The result shown in Figure 7 clearly supports this concept. PMPC with zwitterions also showed extremely low bacterial adhesion due to lipid like properties. P. aeruginosa adhesions on original and modified PU surfaces.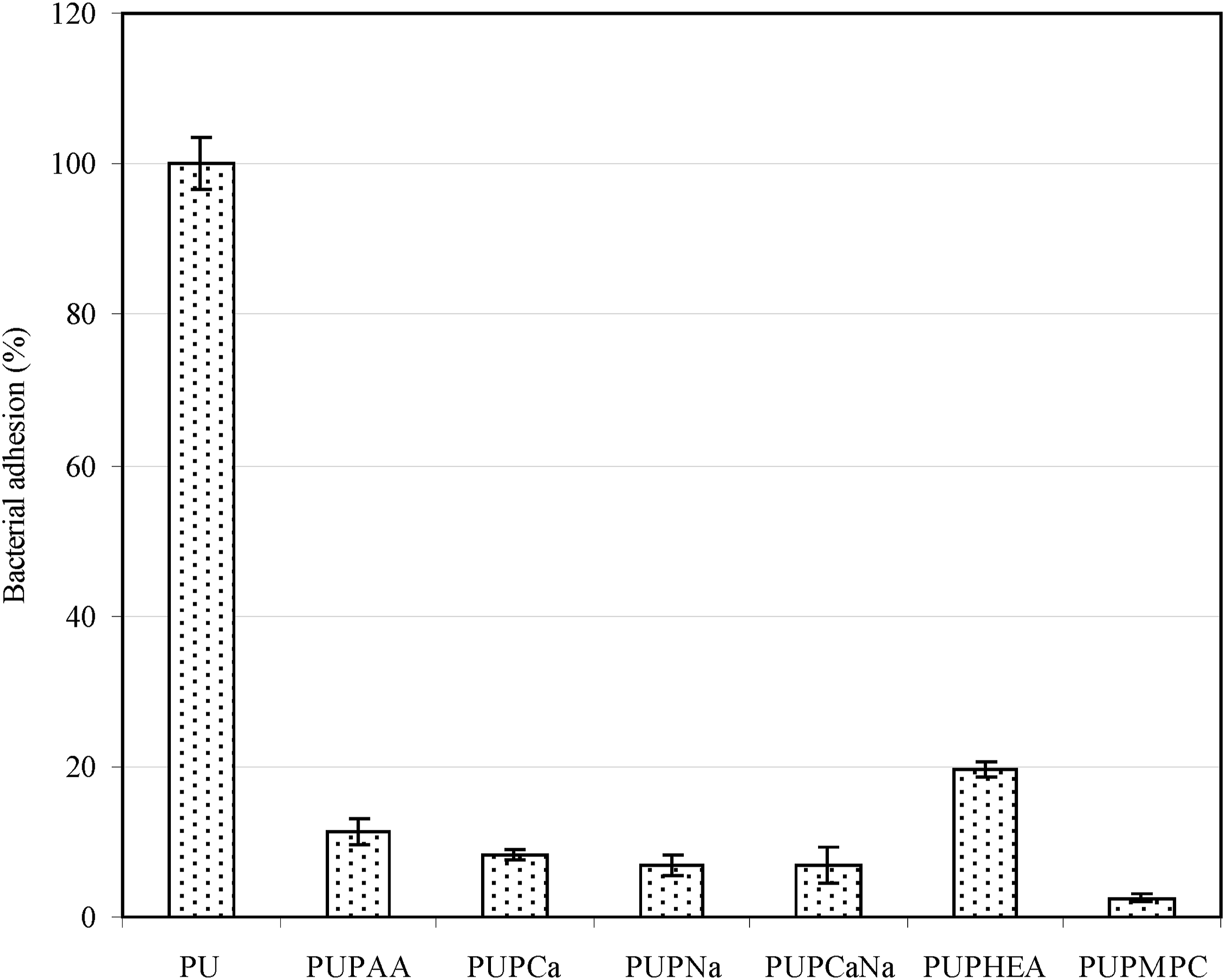
Figure 9 shows a set of photomicrographs of 3T3 fibroblasts adhesion and P. aeruginosa adhesion after incubating with original PVC and PVC surface modified with PNaCa. The images depict (a) 3T3 fibroblasts with PVC, (b) 3T3 fibroblasts with PVC surface modified with PNaCa, (c) P. aeruginosa with PVC, (d) P. aeruginosa with PVC surface modified with PNaCa. Apparently, Figure 9(a) shows that many cells are attached to PVC surface, whereas no cells are attached on the modified surface (Figure 9(b). In the case of bacterial adhesion, a lot of green bacteria are shown in Figure 9(c) but no bacteria are adhered to the modified surface (Figure 8(d)). This proves that the modified surface with hydrogel characteristics and negative charges is very fouling resistant. 3T3 cell adhesion and P. aeruginosa adhesion images after incubating with original PU and PU with PCaNa: (a) PU with 3T3 cells, (b) PU coated with PCaNa with 3T3 cells, (c) PU with P. aeruginosa, (d) PU coated with PCaNa with P. aeruginosa.
Conclusions
We have successfully synthesized a redox co-initiator derivative, used it to initiate a surface free-radical polymerization, and grow polymer brushes on the TPU surface. The surface modified with all the attached polymers exhibited significantly reduced contact angle, protein adsorption, and cell as well as bacterial adhesion. The surface modified with negatively charged polymers showed the lowest values in all the tests. Its contact angle is only 5.5° as compared to 73° (original TPU). Its BSA adsorption, BFG adsorption, 3T3 adhesion and P. aeruginosa adhesion were 88%, 82%, 92%, and 93% lower than original TPU. Furthermore, the TPU surface coated with negatively charged polymer brushes not only exhibited a hydrogel-like property but also showed an excellent biofouling resistance. These results indicate that surface modification of TPU using a simple and conventional surface free-radical polymerization technique with an induced hydrogel formation can be an affective and efficient route for antifouling applications, which holds great promise in preventing or reducing medical device-associated adhesion and infections. Future studies will focus on optimization of the polymers and other characterization/evaluation.
Footnotes
Acknowledgments
This study was partially sponsored by IUPUI BBRC Research Center Support. Mr. Rashed Almousa would like to thank the Deanship of Scientific Research at Majmaah University for supporting this work under Project Number R-2023-709.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the by the Majmaah University for supporting this work under Project Number R-2023-709.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
