Abstract
The application of hydrogels for anti-cancer drug delivery has garnered considerable interest in the medical field. Current cancer treatment approaches, such as chemotherapy and radiation therapy, often induce severe side effects, causing significant distress and substantial health complications to patients. Hydrogels present an appealing solution as they can be precisely injected into specific sites within the body, facilitating the sustainable release of encapsulated drugs. This localized treatment approach holds great potential for reducing toxicity levels and improving drug delivery efficacy. In this study we developed a hydrogel delivery system containing polyamidoamine (PAMAM) dendrimer and polyethylene glycol (PEG) for solubility enhancement and sustained delivery of hydrophobic anti-cancer drugs. The three selected model drugs, e.g. silibinin, camptothecin, and methotrexate, possess limited aqueous solubility and thus face restricted application. In the presence of vinyl sulfone functionalized PAMAM dendrimer at 45 mg/mL concentration, drug solubility is increased by 37-fold, 4-fold, and 10-fold for silibinin, camptothecin, and methotrexate, respectively. By further crosslinking of the functionalized PAMAM dendrimer and thiolated PEG, we successfully developed a fast-crosslinking hydrogel capable of encapsulating a significant payload of solubilized cancer drugs for sustained release. In water, the drug encapsulated hydrogels release 30%–80% of their loads in 1–4 days. MTT assays of J82 and MCF7 cells with various doses of drug encapsulated hydrogels reveal that cytotoxicity is observed for all three drugs on both J82 and MCF7 cell lines after 48 h. Notably, camptothecin exhibits higher cytotoxicity to both cell lines than silibinin and methotrexate, achieving up to 95% cell death at experimental conditions, despite its lower solubility. Our experiments provide evidence that the PAMAM dendrimer-mediated hydrogel system significantly improves the solubility of hydrophobic drugs and facilitates their sustained release. These findings position the system as a promising platform for controlled delivery of hydrophobic drugs for intratumoral cancer treatment.
Introduction
Cancer remains a leading cause of mortality in the United States, necessitating the continuous exploration of effective treatment strategies. Currently, chemotherapy and radiation therapy constitute the primary approaches for cancer treatment. However, these therapies are associated with severe side effects and limited delivery efficiency, highlighting the clinical need for enhancing the targeted delivery of therapeutic drugs to cancer sites. In recent years, hydrogels have emerged as promising platforms for drug delivery due to their superior biocompatibility, significant physical properties, and sustained drug release profiles. These advantages position hydrogels favorably over conventional drug delivery approaches which have the drawbacks of systemic toxicity and requirement of repeated dosing.1–4 Among various kinds of hydrogels for drug delivery, in situ forming hydrogels have gained great interests because they can be delivered to the desired sites through injection, enabling controlled release of the encapsulated drug molecules. The convenience, diversity, and versatility of in situ forming hydrogels have propelled their consideration as intelligent drug delivery systems in various biomedical fields, including tissue regeneration,5,6 cancer,7–9 wound healing,10,11 and antimicrobial or antifungal12,13 treatments. However, challenges persist in hydrogel-based drug delivery, particularly in solubilizing hydrophobic drug molecules, as the hydrophilic nature of hydrogels may not be inherently compatible with such compounds. Therefore, the development of hydrogel systems capable of enhancing the solubility of hydrophobic drugs is crucial for advancing the technology of hydrogel-based drug delivery in medical fields.
Our research group has developed a series of polyamidoamine (PAMAM) dendrimer-mediated in situ forming hydrogels for tissue engineering.14–16 PAMAM dendrimers are synthetic star-burst polymers characterized by well-defined structures, non-toxicity, non-immunogenicity, and excellent water solubility.17–19 These unique properties have earned PAMAM dendrimers fame in a wide range of biomedical applications including imaging, cancer targeting and drug delivery.19–25 In addition, PAMAM dendrimers have been known for their superb capability to improve the solubility of hydrophobic drugs through electrostatic interactions or hydrogen bonding between surface functional groups of the dendrimer and drugs, or through encapsulation of drug molecules in hydrophobic pockets of their dendritic structures.26–28 Therefore, PAMAM dendrimer-mediated in situ forming hydrogels could be effective systems for sustained delivery of hydrophobic drugs. In this study, we chose three hydrophobic drug molecules, e.g. silibinin, camptothecin, and methotrexate, whose clinical applications have been hindered due to their low solubility in aqueous solutions. In the presence of vinyl sulfone functionalized PAMAM dendrimer at 45 mg/mL concentration, their aqueous solubility is increased by 37-fold, 4-fold, and 10-fold for silibinin, camptothecin, and methotrexate, respectively. By crosslinking of the same functionalized PAMAM dendrimer and thiolated PEG, fast crosslinking hydrogels are formed with a large payload of solubilized cancer drug encapsulated for extended drug delivery. Release studies reveal that these drug encapsulated hydrogels release 30%–80% of their loads in 1–4 days when immersed in water. MTT assays of J82 and MCF7 cells with different doses of drug encapsulated hydrogels after 48 h demonstrate cytotoxicity for all three drugs. Despite its lower solubility, camptothecin exhibits higher cytotoxicity than silibinin and methotrexate, resulting in up to 95% cell killing at experimental conditions. Collectively, our experiments highlight the remarkable ability of the PAMAM dendrimer-mediated hydrogel system to improve the solubility of hydrophobic drugs and sustain their release, making it a promising platform for controlled delivery of hydrophobic drugs for cancer treatment.
Materials and methods
Polyamidoamine dendrimer generation 5 with terminal amino groups (PAMAM G5-NH2) is obtained from Dendritech Inc. (Midland, MI, USA). The 8-armed thiolated polyethylene glycol (8arm-PEG-SH, 20 kDa) is purchased from NOF America Corporation (White Plains, NY, USA). Anti-cancer drugs, namely silibinin, camptothecin, and methotrexate (MTX), as well as chemical reagents including acetic anhydride, 2-iminothiolane hydrochloride (2-IT), triethylamine, acrylic acid, divinyl sulfone (DVS), solvents such as dimethyl sulfoxide (DMSO) and methanol are ordered from Fisher Scientific without further purification. MCF7 and J82 cells are acquired from ATCC. Sterilized 48 well plates and MTT kit with inserts are purchased from Fisher Scientific.
Synthesis of PAMAM dendrimer conjugates (dendrimers 2-4 )
The vinyl sulfone functionalized PAMAM dendrimer is synthesized in three steps with a procedure reported in our earlier publication. 14 A brief synthetic protocol is provided below. To ensure purity, all dendrimer species are purified with membrane filtration in a stir-cell under pressure followed by thorough washing with phosphate-buffered saline (PBS) buffer and water. The solid products are obtained by lyophilization of the aqueous solution of dendrimer products and characterized by proton nuclear magnetic resonance (1H NMR) spectrometry.
Synthesis of G5-Ac(70) (dendrimer 2 )
PAMAM G5-NH2 (868 mg, 0.0327 mmol) is dissolved in methanol (100 mL) in a 250 mL round-bottom flask. Triethylamine (290 mg, 2.863 mmol) is added and the reaction mixture is stirred for 5 min. A solution of acetic anhydride (234 mg, 2.290 mmol) in methanol (40 mL) is added dropwise with vigorous stirring. After addition, the resulting mixture is allowed to stir at room temperature overnight. Subsequently, the reaction mixture is transferred into a 600 mL stir-cell equipped with a 10,000 molecular weight cut-off (MWCO) membrane. The residue is extensively washed with PBS buffer (20 mL × 3) and water (20 mL × 3). The aqueous solution of G5-Ac(70) is lyophilized, yielding a solid product (951 mg, 100% yield). 1H NMR spectrum of G5-Ac(70) is displayed in Figure 1(b). The number of amine groups acetylated is deterimined as 72 by proton intergration of the acetyl methyl group at 1.8 ppm versus the total internal protons in dendrimer, following the method reported.
29
(a) Synthesis of vinyl sulfone functionalized PAMAM dendrimer and drug encapsulated hydrogels. (b)-(d) 1H NMR spectra in D2O for dendrimer 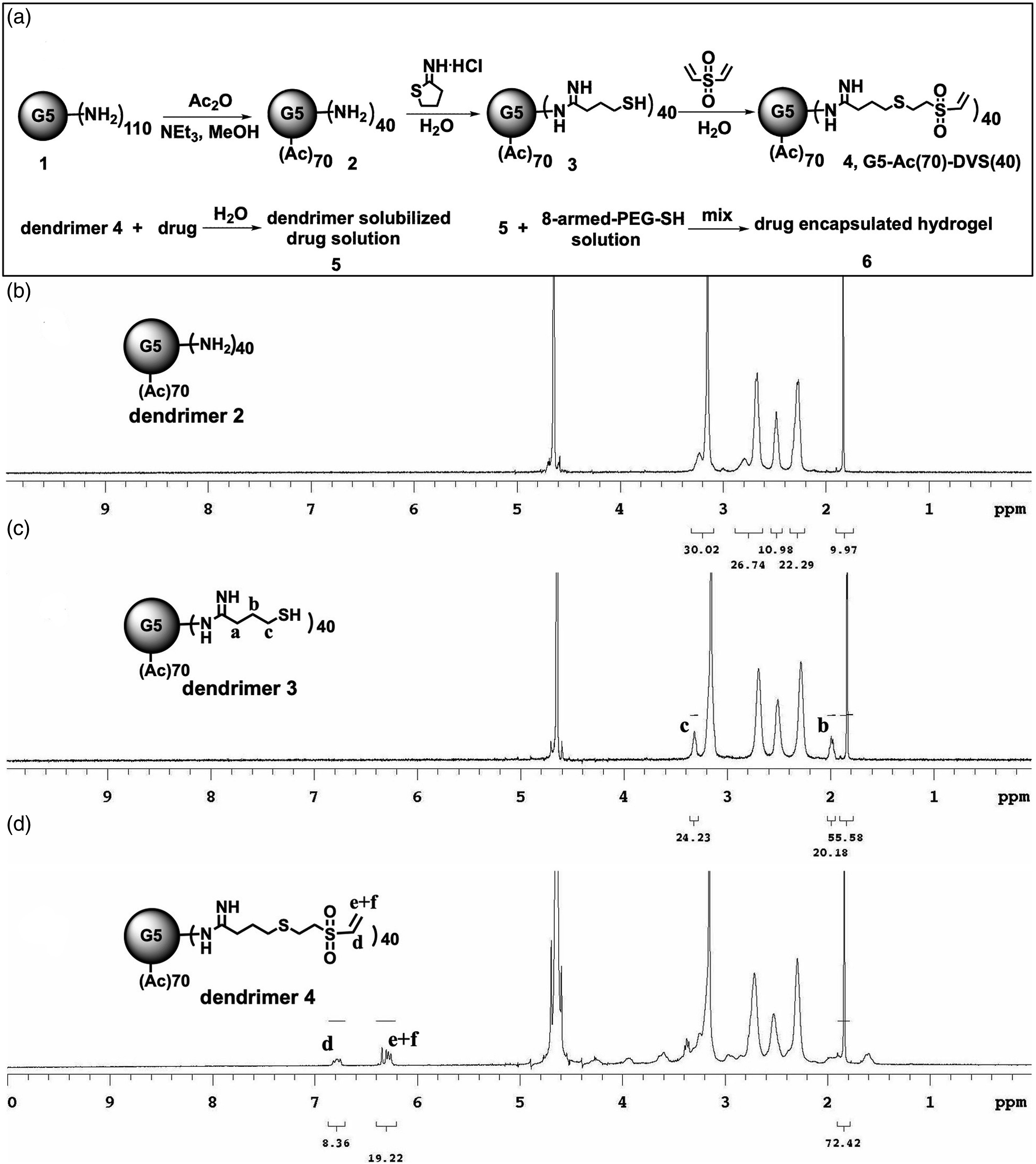
Synthesis of G5-Ac(70)-SH(40) (dendrimer 3 )
G5-Ac(70) (259 mg, 0.00,893 mmol) is dissolved in deionized water (20 mL). A solution of 2-IT (61.4 mg, 0.447 mmol) in deionized water (2 mL) is added dropwise. Nitrogen gas is bubbled into the solution for 2 min to remove the oxygen from media and flask. The reaction mixture is then allowed to stir at room temperature for 2 days. The resulting mixture is transferred to a stir-cell with a 10,000 MWCO membrane and is purified with the same procedure as for dendrimer
Synthesis of G5-Ac(70)-DVS (40) (dendrimer 4 )
Dendrimer G5-Ac(70)-SH(40) (155 mg, 0.00,484 mmol) is dissolved in deionized water (20 mL). DVS (114.3 mg, 0.968 mmol) is added to the dendrimer solution. Nitrogen gas is introduced to the mixture for 2 min to remove oxygen. The reaction is allowed to stir at room temperature for 3 days. The resulting mixture is transferred to a stir-cell with a 10,000 MWCO membrane and is purified with the same procedure as for dendrimer
Nuclear magnetic resonance (NMR) analysis of PAMAM dendrimers
1H NMR spectra of dendrimer species are recorded using a Varian 400 MHz spectrometer (Santa Clara, CA, USA) in D2O solvent.
UV-Vis calibration of cancer drugs
Precisely measure about 2 mg (to 0.00,001 g) of a drug, e.g. silibinin, camptothecin, and methotrexate, and individually dissolve in ethanol (300 μL). Transfer the drug solution to a 100 mL volumetric flask and fill the flask with deionized water to 100 mL. In case the drug is not readily soluble, the solution is gently warmed up to solubilize the drug and then cooled to room temperature while ensuring the drug remains soluble. The resulting drug solution is mixed evenly and diluted to varied concentrations for absorbance measurement using a Shimazu UV-1650PC spectrometer. The absorbances are recorded at the corresponding maximum absorption wavelength for each drug (325 nm for silibinin, 367 nm for camptothecin, and 301 nm for methotrexate). These absorbances are then plotted against the concentrations of the dendrimer to yield a linear calibration curve.
Drug solubility enhancement with PAMAM dendrimers
The solubility enhancement of each drug is evaluated at different dendrimer concentrations. Different aqueous solutions of PAMAM dendrimer
Synthesis of PAMAM-PEG hydrogel encapsulated with cancer drugs
Each drug, namely silibinin, camptothecin, and methotrexate, is solubilized in 30 mg/mL dendrimer
UV-Vis measurement of released drugs in PBS
The drug encapsulated hydrogel pellet is immersed in 15 mL distilled water. The concentrations of drug released from hydrogels at 10 min, 30 min, 1 h, 3 h, 8 h, 1 day, 3 days, 5 days, and 7 days are measured with UV-Vis spectroscopy using the calibration curve in Figure 2(a). The drug release profiles are depicted in Figure 4.
MCF7 and J82 cell culture
The MCF7 and J82 cells are obtained from ATCC and cultured in T75 flasks using minimum essential media (MEM from Gibco) supplemented with 10% fetal bovine serum (FBS), and 1% pen-strep. The cells are maintained at 37°C in a CO2 incubator with a humidified atmosphere of 5% CO2 and 95% air. The growth media is refreshed every other day. Upon reaching approximately 80% confluency, the MCF7 and J82 cells are harvested from the T75 culture flask for future experiments.
Preparation of anti-cancer drug in hydrogel matrix
The hydrogels are prepared under sterile conditions. The anti-cancer drugs used in this experiment are silibinin, camptothecin, and methotrexate. Stock solutions for drug encapsulated hydrogel formation are as follows. The concentration of PAMAM dendrimer
MTT assays of MCF7 and J82 cells
The viability of MCF7 cells is assessed using the MTT assay. A brief procedure is described as below. MCF7 cells are seeded in four 24 well plates after reaching 80% confluency at a density of 5 × 104 cells and 500 μL fresh MEM media per well. After 2 days of culture, the media in each well is replaced. The drug encapsulated hydrogel pellets prepared earlier are carefully transferred into the insert in each well and are immersed in the media. As a control to each hydrogel with different amount of anti-cancer drugs, an equivalent volume of the drug-dendrimer
Results and discussion
Commercial PAMAM dendrimer G5-NH2 bearing 110 amino terminal groups is selected for hydrogel preparation. The synthetic scheme is illustrated in Figure 1. The numerical values indicated outside the parentheses of each functional group in the scheme represent the numbers of functional groups that are intended to be attached to dendrimers. These values are not optimized and can be adjusted according to the specific requirement of each application. The synthesis starts with a partial acetylation of PAMAM G5-NH2 using acetic anhydride, resulting in dendrimer
The selected model anti-cancer drugs for our study are silibinin, camptothecin, and methotrexate. Silibinin, also known as silybin, has demonstrated significant tumor suppressive activities in a number of cancers by promoting cell apoptosis. 30 Camptothecin is a naturally occurring compound with high activity towards several cancer cell lines. 31 Methotrexate is one of the most frequently used anti-cancer drugs to treat a number of cancers or severe and treatment-resistant autoimmune diseases. 32 However, the application of these drugs is restricted by their low water solubility.31,33,34 Therefore, it is of great importance to develop platforms to deliver those hydrophobic drugs not only in a controllable manner but with enhanced solubility.
In this research, we focus on the development of a PAMAM dendrimer-mediated hydrogel system for delivery of hydrophobic drugs. PAMAM dendrimers are a class of synthetic star-burst polymers that possess well-defined structures as well as desirable properties such as non-toxicity, non-immunogenicity, and excellent water solubility. Their active terminal functional groups allow for chemical modification and bioconjugation to various bioactive molecules, enabling their use in diverse medical applications including cancer targeting and treatment,35,36 drug delivery,21,37 gene delivery,38,39 photodynamic therapy,40,41 sensors,42,43 and imaging.44,45 Furthermore, PAMAM dendrimers have been recognized for their ability to enhance water solubility of hydrophobic drug molecules.26,27 We propose that a PAMAM dendrimer-mediated hydrogel system will not only increase the solubility of the selected drug molecules but also provide a sustained release for their delivery.
To quantitively determine the amount of drug released, a calibration curve of each drug is to be generated. A range of concentrations of each drug in PBS buffer is prepared, and their UV absorbance is determined on a UV-Vis spectrometer. Figure 2 displays the UV spectra of silibinin, camptothecin, and methotrexate, as well as the corresponding calibration curves. The absorbance measurements are recorded at 325 nm for silibinin, 367 nm for camptothecin, and 301 nm for methotrexate. (a) Calibration curves of drugs by UV-Vis (absorbances are measured at 325 nm, 367 nm, and 301 nm for silibinin, camptothecin, and methotrexate, respectively). (b) UV-Vis spectra of silibinin, camptothecin, and methotrexate in aqueous solutions.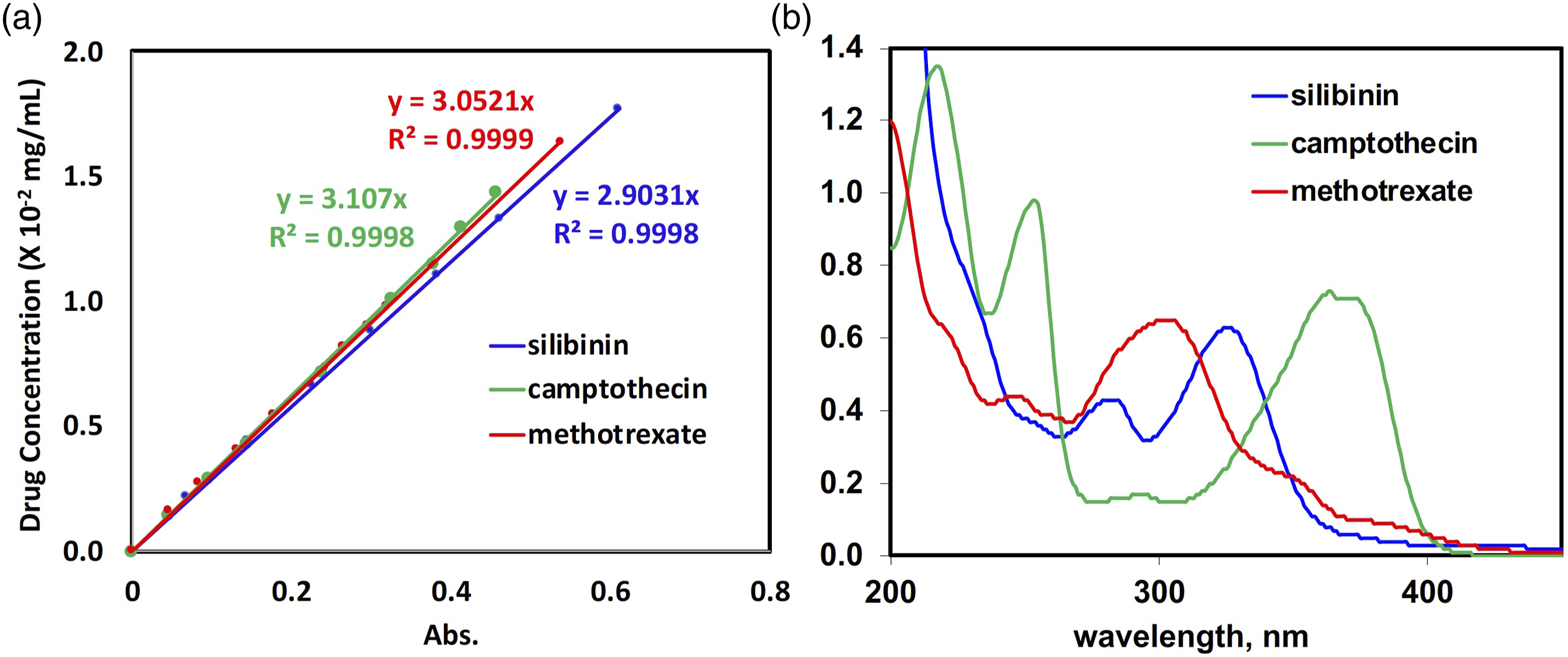
To investigate the potential of vinyl sulfone functionalized PAMAM dendrimer ( Drug solubility enhancement with dendrimer 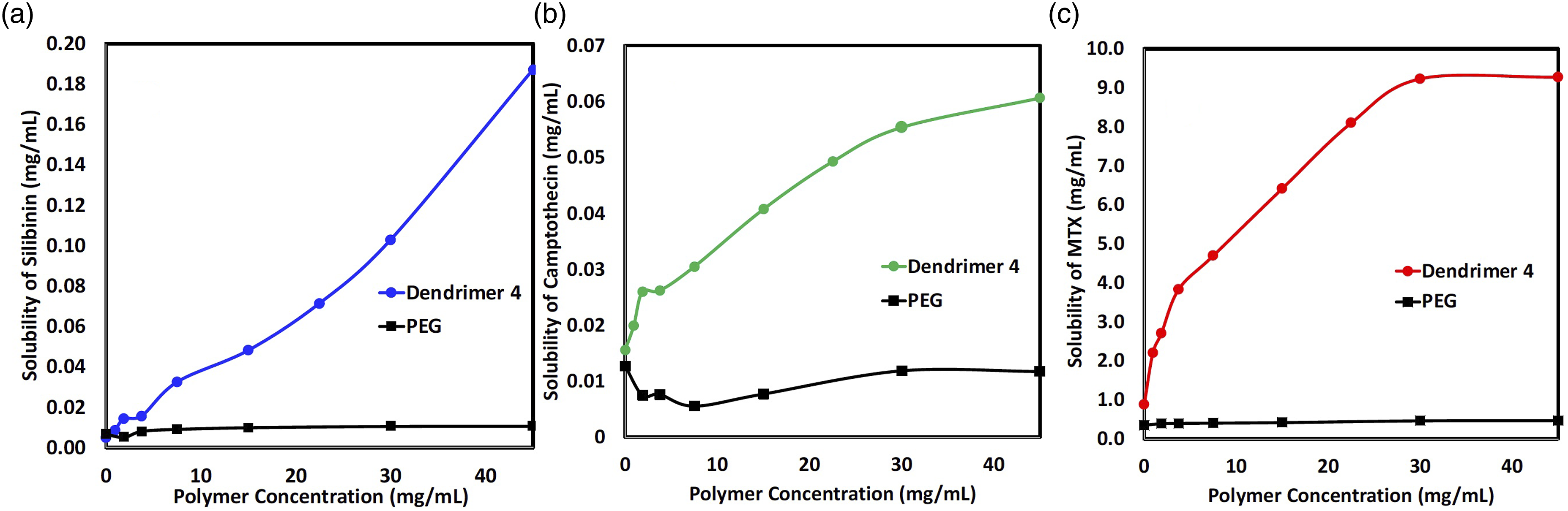
It can be seen clearly that the solubility of each drug increases with the concentration of dendrimer
The enhanced solubility of hydrophobic drugs in PAMAM dendrimers is attributed to surface absorption by electrostatic interactions or encapsulation of drugs in dendrimer cavities. Silibinin has been reported to strongly interact with dendrimers through static interactions on surface,
46
which explains its dramatic improvement in solubility in dendrimer
The drug encapsulated hydrogels are prepared according to the scheme in Figure 1. Each drug is saturated in a dendrimer Encapsulated drug release from hydrogels encapsulated with silibinin (a), camptothecin (b) and methotrexate (c).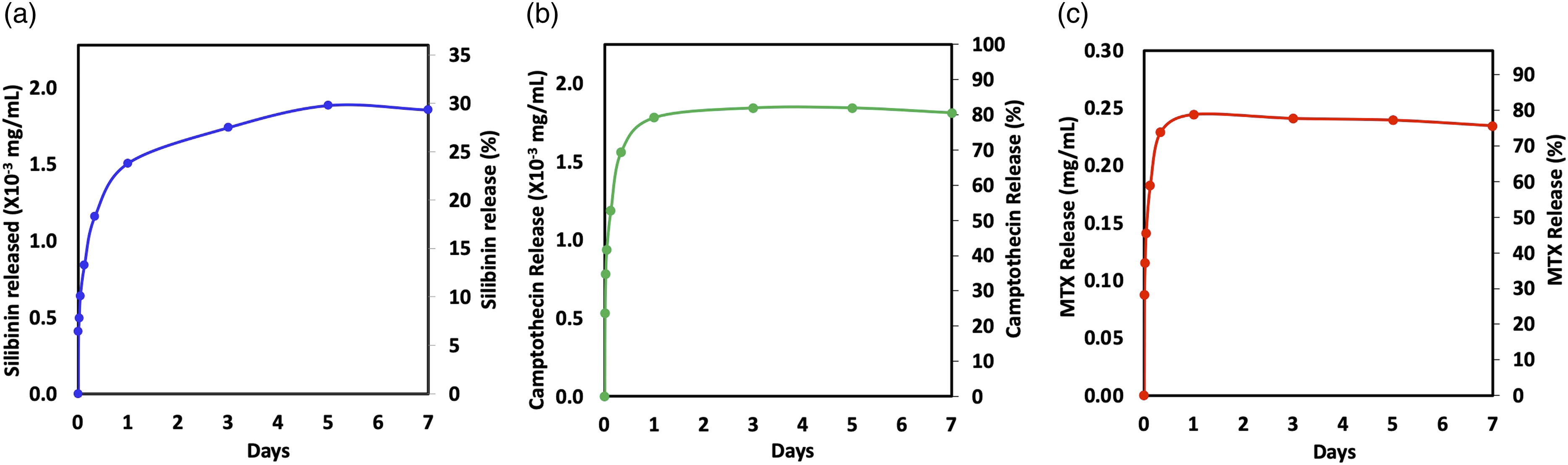
The cytotoxicity of drug encapsulated hydrogels is evaluated on J82 and MCF7 cell lines using MTT assay. The concentrations of drugs in 30 mg/mL PAMAM stock solutions are 0.095 mg/mL, 0.034 mg/mL, and 4.65 mg/mL for silibinin, camptothecin and methotrexate, respectively. These drug-dendrimer MTT assay of J82 and MCF7 cells with drug encapsulated hydrogels. The top panels are J82 cells treated with silibinin (a), camptothecin, (b) and methotrexate (c) released from different amounts of hydrogels after 48 h. The bottom panels are MCF7 cells treated with silibinin (d) camptothecin (e) and methotrexate (f) released from different amounts of hydrogels after 48 h. The hydrogels are formed by mixing equal amount of drug-dendrimer 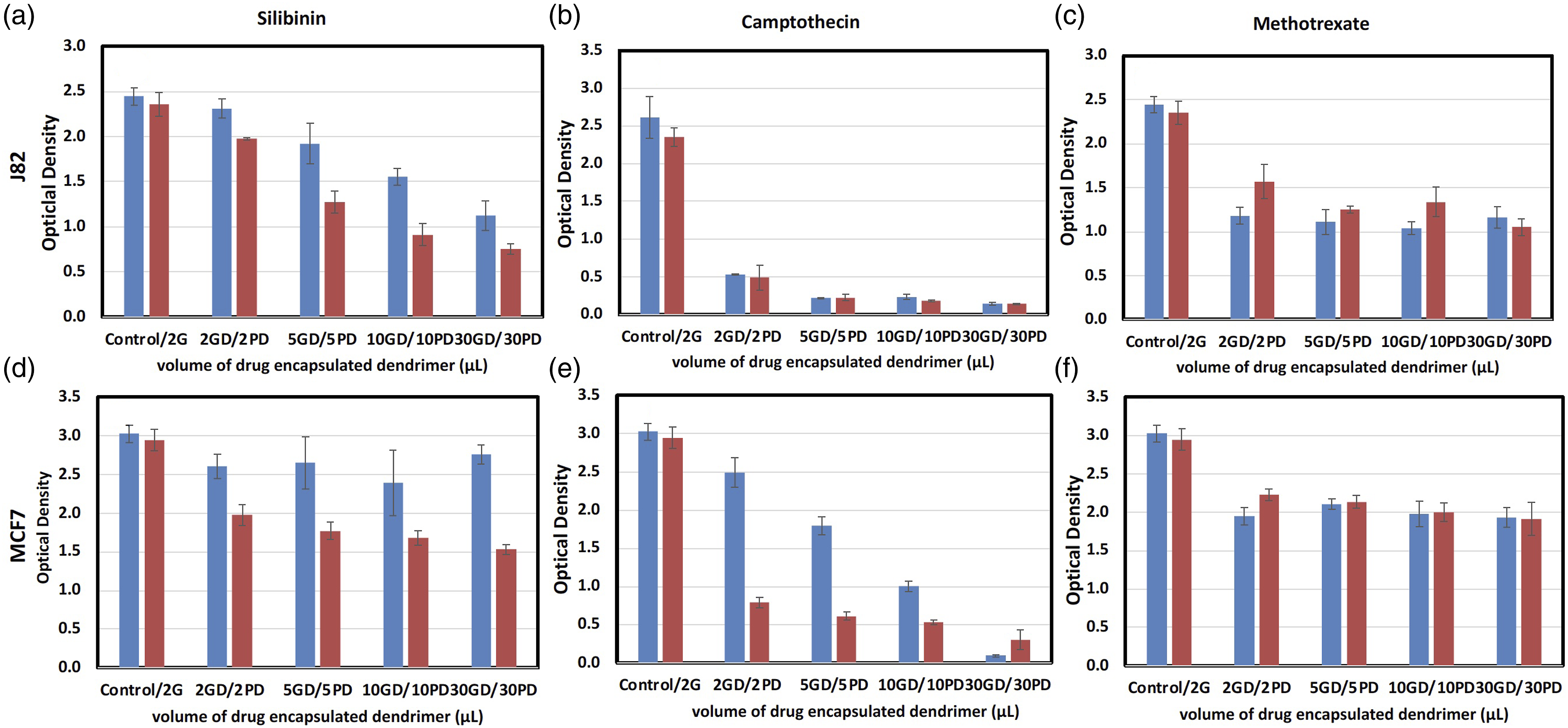
Conclusions
A hydrogel system incorporating vinyl sulfone functionalized PAMAM dendrimer and thiolated PEG is developed for solubility enhancement and sustained delivery of hydrophobic anti-cancer drugs. In the presence of functionalized dendrimer
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported in part by the NSF and SC EPSCoR/IDeA (MADE in SC) Program under GEAR CRP 20-GC02.
