Abstract
Ultra-high molecular weight polyethylene (UHMWPE) can be made radiopaque for medical imaging applications through the diffusion of an iodised oil-based contrast agent (Lipiodol Ultra Fluid). A similar process is used for Vitamin E incorporated polyethylene which provides antioxidant properties. This study aimed to investigate the critical long-term properties of oil-infused medical polyethylene after 4 weeks of accelerated thermal ageing. Samples treated with an oil (Vitamin E or Lipiodol) had a higher oxidation stability than currently used medical grade polyethylene, indicated by a smaller increase in oxidation index after ageing (Vitamin E + 36%, Lipiodol +40%, Untreated +136%, Thermally treated +164%). The tensile properties of oil treated polyethylene after ageing were significantly higher than the Untreated and Thermally treated controls (p<0.05) indicating less mechanical degradation. There was also no alteration in the percentage crystallinity of oil treated samples after ageing, though the radiopacity of the Lipiodol treated samples reduced by 54% after ageing. The leaching of oil with time was also investigated; the leaching of Lipiodol and Vitamin E followed the same trend and reached a steady state by two weeks. Overall, it can be concluded that the diffusion of an oil-based fluid into polyethylene not only increases the oxidative and chemical stability of polyethylene but also adds additional functionality (e.g. radiopacity) providing a more suitable material for long–term medical applications.
Keywords
Introduction
Ultra-high molecular weight polyethylene (UHMWPE) is one of the most common bearing surfaces used in joint replacements. The long-term properties and durability of polyethylene play a significant role in the outcome of the surgical procedure.1,2 Around 1% of orthopaedic procedures fail within the first 10 years of implantation, 3 and most of the material-related failures can be associated with oxidative degradation of the polyethylene. Oxidised polyethylene has a higher brittleness which makes the polymer more prone to crack and wear. 4 Polyethylene wear particles can also trigger bone loss (osteolysis) and lead to issues such as mechanical implant loosening which often require revision surgery.1,5
Polyethylene has a limited X-ray attenuation making it difficult to see in a radiograph. 6 As a result of its limited X-ray attenuation, clinical evaluation of components and the early diagnosis of failure are challenging. To address this limitation, in our laboratory we developed a radiopaque polyethylene which contains an FDA approved oil-based contrast agent (Lipiodol Ultra Fluid). 7 Previous work identified an optimal treatment protocol which maintained the mechanical and physical properties of the polyethylene; 8 however, it is critical to assess how these properties will change over the long-term in an in vivo environment.
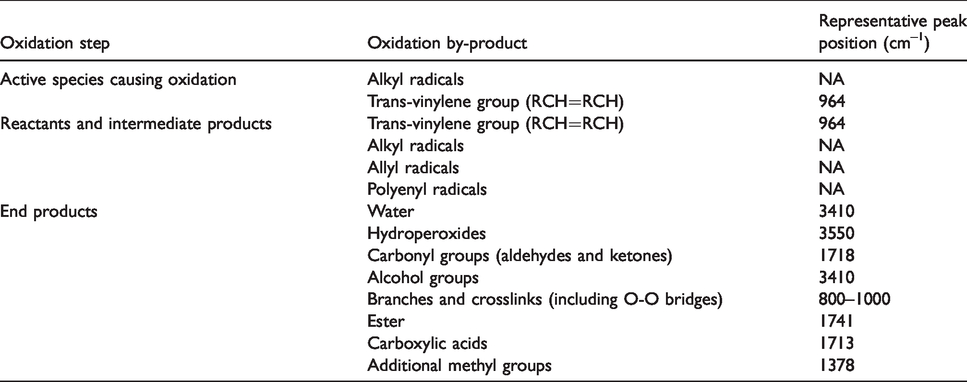
The most common mechanism of polyethylene degradation in vivo is oxidation. Oxidation of polyethylene can be explained by Bolland’s cycle. 9 In this cycle, the alkyl radicals present in polyethylene (due to the polymerisation and processing) react with oxygen to form peroxy radicals, which are able to attack other polymer molecules to form hydroperoxides and regenerate the original alkyl radicals. Hydroperoxides will eventually decompose to form more stable oxidation products (ketones, second aryl-alcohols, carboxylic acids and esters, see Table 1), which can be quantified using Fourier Transform Infrared Spectroscopy (FTIR). 4
It takes months or years to reach a detectable level of oxidation at ambient and body temperature 12 ; this is due to the slow diffusion of oxygen. It takes up to 120 days for an oxygen molecule to move 1 mm below the surface of a crosslinked polymer and for an uncrosslinked sample with 52% crystallinity 100 days is required for oxygen to travel 1 cm (diffusion coefficient 10–7 cm2s–1). 13 Consequently, an accelerated ageing protocol is required to experimentally assess the propensity of a polyethylene to oxidation in a reasonable time-frame. 14
All attempts to accelerate the rate of oxidation of polyethylene to date have either increased the diffusion flux of oxygen at the implant surface, or increased the kinetic rate of oxidation. Both of which can be achieved either by increasing the oxygen concentration at the boundaries (the partial pressure of oxygen in the atmosphere) or by exposing the sample to an elevated temperature. There are two commonly used standardised protocols of thermally accelerated ageing (ASTM F2003-02(2008)); in the first one the specimens are kept for 2 weeks in pure oxygen of 0.5 MPa pressure at 70∘C, in an air convection oven. In the second method, all specimens are kept at 80∘C at atmospheric pressure for 4 weeks in an air convection oven. Both protocols replicate five years of natural polyethylene ageing in vivo.12,15,16 Recent studies have developed even more aggressive accelerated ageing methods, which make use of squalene to more closely replicate in vivo oxidation. 17
Many clinical and laboratory studies have shown an increase in the crystallinity and a reduction in ductility and toughness of polyethylene after ageing, 12 hence it is crucial to also evaluate the mechanical properties and the percentage crystallinity of the aged polymer. Furthermore, it is unknown if the oil content can diffuse out of the polyethylene with time and consequently remove/reduce the intended functionality, such as radiopacity or antioxidant properties.
The aim of this study was to investigate long-term changes in the physical, thermal, chemical and tensile properties of Lipiodol-infused polyethylene, as compared to antioxidant vitamin E infused polyethylene which is used commercially for joint replacement implants.
Materials and methods
The experiments studied the longevity of oil-infused polyethylene by examining the thermal properties (melting point and crystallinity), oxidative stability, mechanical stability, and oil-diffusion stability (leaching) of the polyethylene. For the radiopaque Lipiodol treated polyethylene the radiopacity was also quantified before and after ageing (Figure 1).
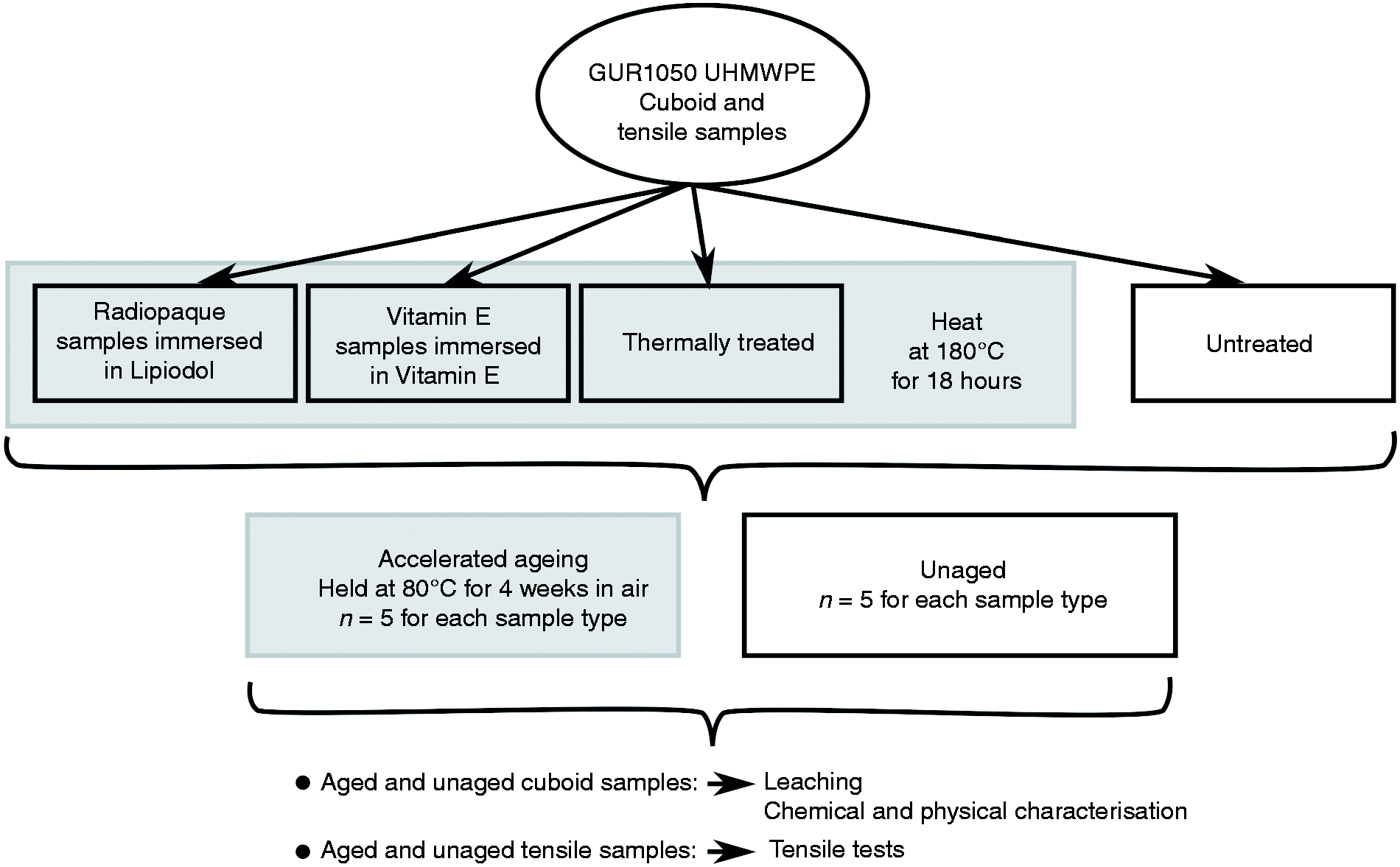
A schematic of the experimental procedure.
Sample preparation
The polyethylene used in this study was 4 mm thick sheet moulded un-irradiated GUR 1050 UHMWPE with an average molecular weight of 5.5 to 6.0 million g/mol, provided provided by Celanese, Inc (Oberhausen, Germany). GUR 1050 sheets were cut to small cuboids of 10 mm by 10 mm by 4 mm, and tensile test specimens with the geometry specified in ISO 572 Annex A, type 1AB. 18
Four different types of samples were used in this study (Table 2), with five sample repeats of each type. Radiopaque samples were immersed in 25 ml of Lipiodol (Guerbet, France) at 105∘C for 18 hours to create Radiopaque polyethylene.19,20 The second group of samples were immersed in 25 ml of Vitamin E (L-α–tocopherol, Sigma-Aldrich, Kent, UK) at 105∘C for 18 hours to create the Vitamin E samples. Thermally treated samples were treated at the same elevated temperature and time (105∘C for 18 hours) without the presence of any oil; and finally the Untreated control samples were not treated with temperature or any oily fluid.
Treatment conditions and additives used to prepare the four different sample types analysed.

NA: not applicable.
At the end of the treatment time, the samples were allowed to cool down to ambient temperature and wiped with a lint-free tissue to remove any excess oil from the surface.
Accelerated ageing
The accelerated ageing protocol used in the current study was in accordance of ASTM F 2003–00. 14 All the prepared samples were kept in the oven for 4 weeks at a constant temperature of 80 ± 2∘C 12 and the material properties were assessed before and after ageing.
Leaching
In vitro leaching of oil was evaluated in two different solutions: saline solution and Dulbecco’s Modified Eagle’s Medium (DMEM); both purchased from Sigma-Aldrich (Kent, UK). Penicillin-Streptomycin (Sigma-Aldrich, Kent, UK) was added to the DMEM solution (0.2% v/v) to prevent any bacterial growth. Each sample was located in a separate well (n = 5) and immersed in 10 ml of one of two solutions. The solutions were topped up every two days to mitigate the effect of evaporation. All the samples were located in an incubator (Incu-50S, SciQuip, UK) with a control temperature of 37∘C. The leaching was quantified from the gravimetric changes.
Crystallinity measurement
Differential scanning calorimetry (DSC 250, TA Instrument, Delaware, United States) was used to determine the melting point and the degree of crystallinity of UHMWPE (n = 5). Approximately 2.0 mg of each sample was taken from the outer surface of the samples to be analysed with DSC. The pan was crimped with an aluminium cover. The testing processed from 20∘C to 180 ∘C at a heat flow of rate 10∘C/min under a nitrogen purge. TA Instruments Universal software (Version 4.5) was used to analyse the data and calculate the degree of crystallinity. The crystallinity was calculated by first integrating the endothermic peak from 50∘C to 160∘C to find the enthalpy of fusion of the sample, and then by normalising the enthalpy of fusion with the heat of fusion (289 J/g 21 )
Radiopacity measurements
All the Radiopaque samples were imaged using a µCT scanner (X Tec, XT H 225 ST, Nikon Metrology UK Ltd, Derby, UK) before and after the ageing process; scanning protocols were the same for all samples (162 kV, resolution 0.2 mm). Analysis of the CT data was performed using Simpleware ScanIP (Synopsys Inc., Exeter, UK release version 2017). Water and air were used for calibration to calculate the Hounsfield units (HU). 22
Gravimetric changes
The weight of all samples was measured using a digital scale (Model XP205, Mettler Toledo, Columbus, OH, USA) before and after the sample preparation treatment, the accelerated ageing, and the leaching experiment. The Lipiodol intake and leaching was calculated from this information. For the leaching investigation, the gravimetric change in the samples was measured at weeks 0, 1, 2, 4, and 8.
Ftir
A thin slice (approximately 200 µm) was sectioned from the surface of each cuboid sample using a sledge microtome. A Thermo Scientific spectrometer (Nicolet 560, Thermo Fisher Scientific, Waltham, MA, USA) was used to obtain the FTIR spectra in transmission mode. An average of 32 scans was taken for each section from 4000 to 600 cm–1. FTIR spectra was collected from each specimen within 24 hours of the start and end of accelerated ageing. Peaks to identify oxidation and crystallinity content were the interest of this study.
The oxidation index was calculated based on ASTM F2759–19. 23 The area under the carbonyl peak (1740 cm–1) was normalised against the peak from the methyl stretch at 1370 cm–1. Lipiodol contains iodine, and the presence of an electronegative group can shift the peak by 15 cm–1, 24 hence shifted peaks were used for the Radiopaque samples.
Tensile mechanical testing
Tensile tests were conducted at room temperature in accordance with ISO-527 18 using an electromechanical test machine (Model 5965, Instron, Norwood, MA, USA) at a rate of 50 mm/min. The tests were carried out with aged untreated UHMWPE, aged thermally treated UHMWPE, aged Vitamin E UHMWPE and aged Radiopaque UHMWPE. Five specimens per condition were tested to calculate the tensile modulus (E), 0.2% yield strain, ultimate tensile strength (UTS) and elongation at failure. The raw data were processed by a custom MATLAB script (R2017a, The MathWorks Inc., Nattick, MA, USA). The calculations were conducted as specified in the standards. 18
Statistical analysis
A Kruskal-Wallis test was used to compare the experimental results between different sample groups, and Mann-Whitney U tests were performed to compare the results of each sample group before and after ageing. IBM SPSS Statistics for Windows, version 25 (IBM Corp., Armonk, N.Y., USA) was used to perform the statistical analysis. The statistical differences in the above comparisons were considered significant at the p < 0.05 level.
Results
Accelerated ageing
The aged Radiopaque samples had lower surface radiopacity compared to the radiopacity of the unaged samples (Figure 2, 54 ± 8% reduction). In the unaged samples the maximum radiopacity observed on the surface of the samples and was 1060 ± 53 HU) and after ageing the surface radiopacity reduced to 600 ± 45 HU).
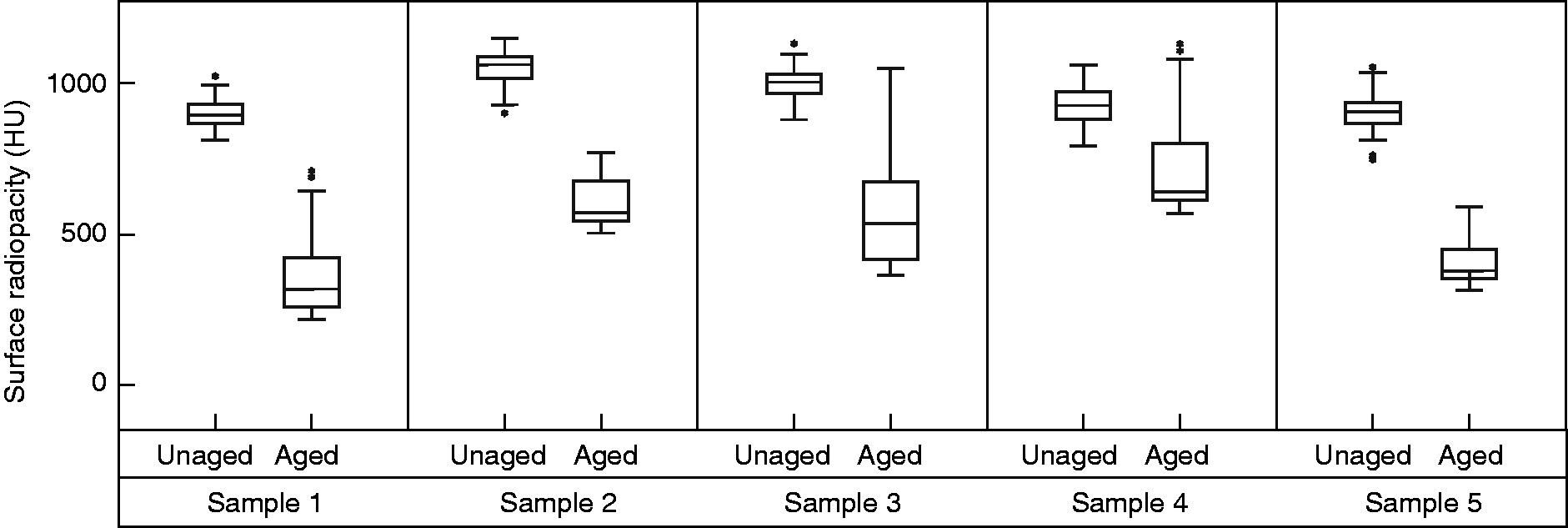
Box and whisker plots of the surface radiopacity to a 2 mm depth of the 5 Radiopaque sample repeats calculated before and after ageing.
The ageing process had no effect on the percentage crystallinity or melting point of any of samples (Figure 3); and all the p-values were between p = 0.055 to 0.51 using Mann-Whitney U tests. The ageing process also did not cause any visible colour change.
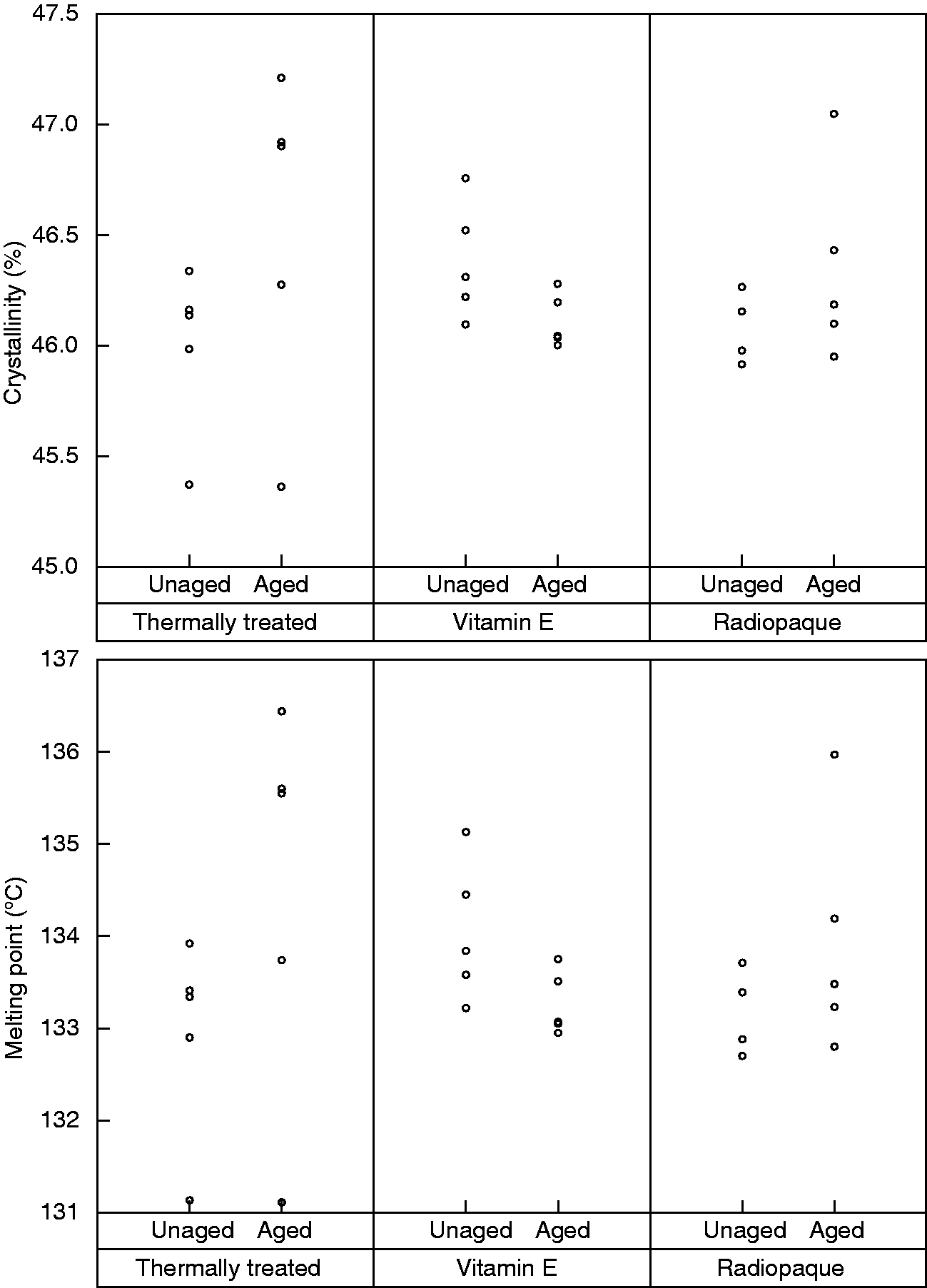
The DSC results indicated no significant (p > 0.005) alteration in a) crystallinity or b) melting point of any of sample group after ageing.
The oxidation index of all polyethylene samples increased after ageing. Before ageing, Vitamin E treated samples showed the lowest level of oxidation index (0.274 ± 0.036) while the Radiopaque samples had the highest amount of oxidation (0.796 ± 0.059). However, as Figure 4 shows the increase in the oxidation index of the Radiopaque samples after ageing was much lower than the Thermally treated and Untreated samples (p < 0.005). The oxidation index of the Untreated and Thermally treated samples increased by 50% and 115% respectively after ageing, and the oxidation index of the Radiopaque and Vitamin E treated samples increased by 36% and 24% respectively.
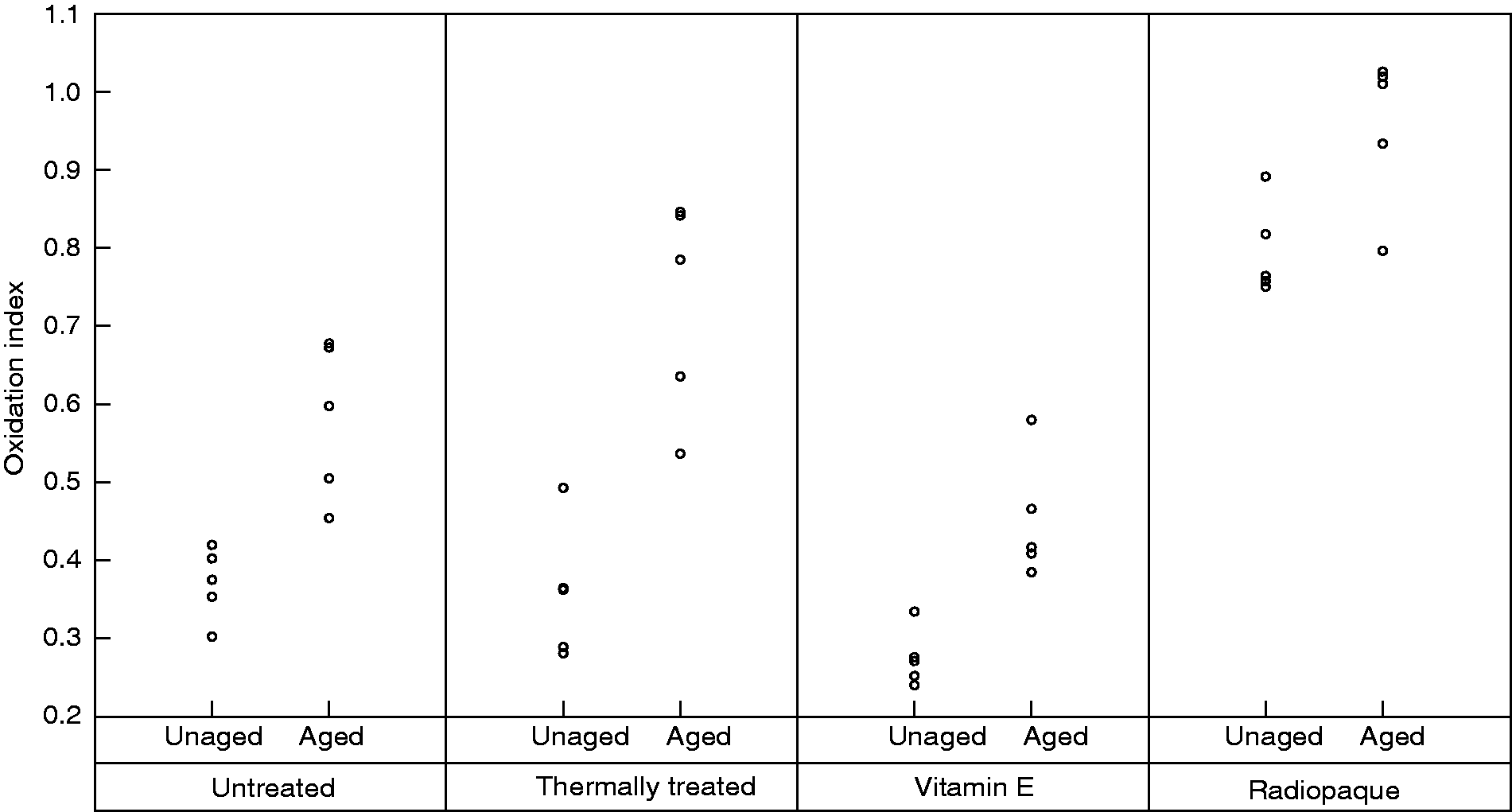
The oxidation index of all the samples increased after ageing. The lowest oxidation index was reported for Vitamin E infused samples whilst the highest was seen in the Radiopaque polyethylene samples.
There was no evidence of ketone (oxidation by-product) present in any of the FTIR spectra (peak at 1718 cm–1) before ageing, however after ageing the ketone absorbance peak was present in the all the spectra, indicating oxidation. Oxidation also leads to formation transvinylene groups and it was observed that the peak for transvinylene groups increased while the vinyl groups peak decreased. Table 3 summarises the peak changes in the sample groups before and after ageing.
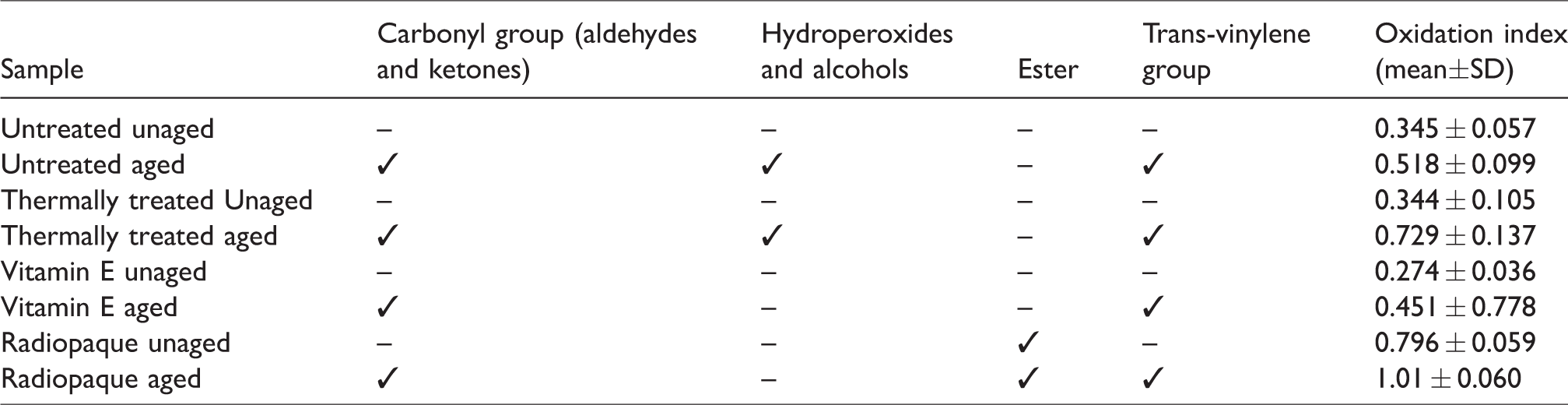
The peaks present in the FTIR spectra for each sample after and before ageing related to oxidation, and the oxidation index (OI) calculated as specified in ASTM F2012-01. ✓=peak present, SD=standard deviation.
The ageing process also led to a significant alteration in the tensile properties of samples. Oil free samples were oxidised and flaky after ageing, and the modulus was on average 84% lower than before ageing (Figure 5(a)), and the elongation at failure was 67% lower (Figure 6(a)). The yield strength of the Thermally treated and Untreated samples was higher than the rest of the samples (Figure 5(b)); however the difference was not statistically significant (p < 0.05). In terms of ultimate tensile strength (UTS) there was an increase in the UTS of the samples after the ageing process (Figure 6(b)). The UTS of the Radiopaque and Vitamin E samples was also higher than the Untreated and Thermally treated samples (p = 0.004). Finally the presence of the oily fluid preserved the ductility of the samples; the elongation at failure of the Radiopaque and Vitamin E samples was significantly higher than Untreated and Thermally treated samples after ageing.
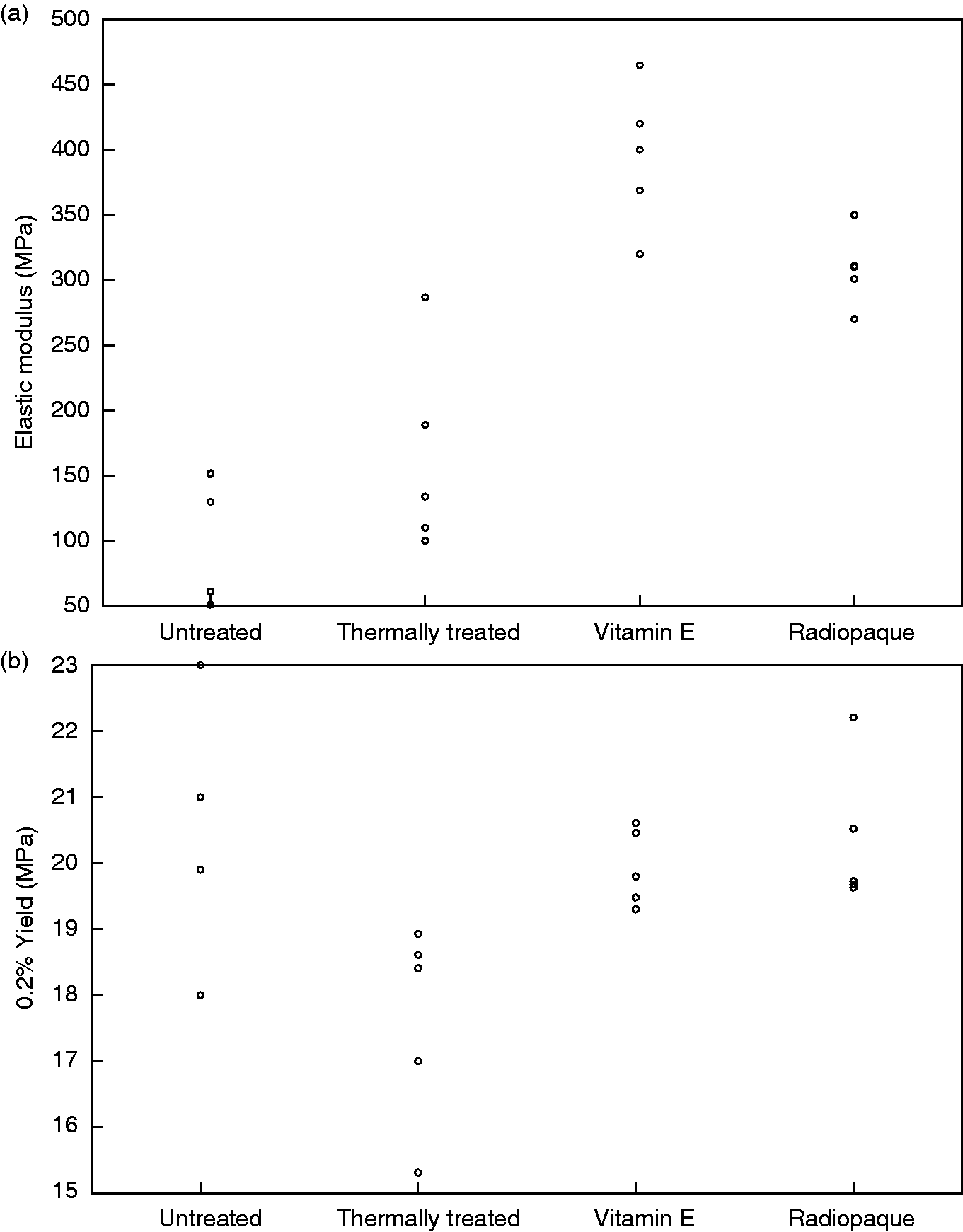
The elastic mechanical properties of oil treated polyethylene; (a) elastic modulus (b) and yield strength. The modulus and yield after ageing of the oil treated samples (Radiopaque and Vitamin E) were significantly higher than the thermally treated and untreated controls (modulus:p = 0.56, yield: p = 0.006).
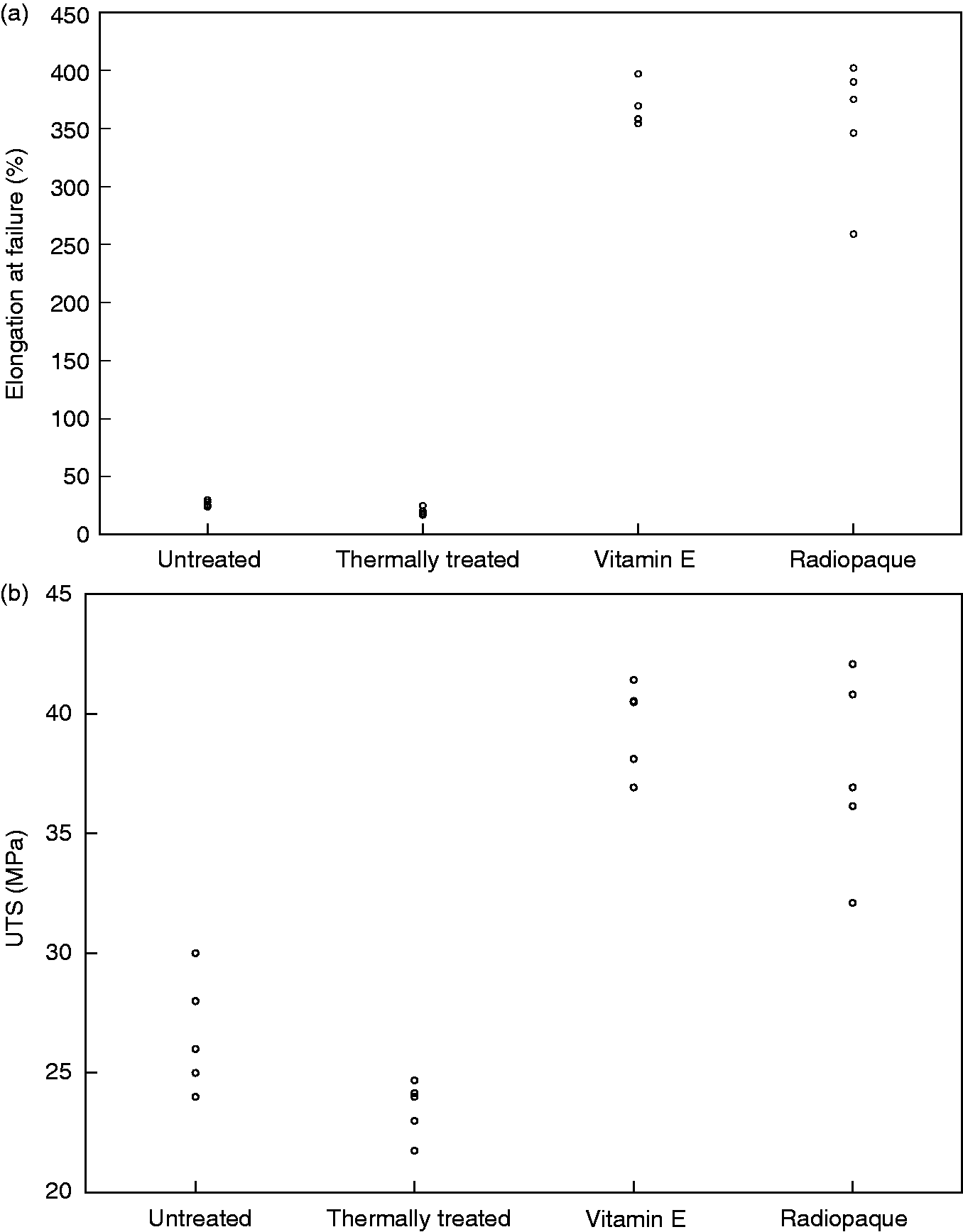
The post-yield mechanical properties of oil treated polyethylene; (a) ultimate tensile strength or UTS (b) and elongation at failure. The UTS and elongation at failure after ageing of the oil treated samples (Radiopaque and Vitamin E) were significantly higher than the thermally treated and untreated controls (UTS: p = 0.004, elongation: p = 0.002).
Leaching
Over the first 2 weeks of leaching there was a relatively large decrease in the weight of the both the Radiopaque and Vitamin E polyethylene samples (Figure 7). The average leaching rate for the Radiopaque samples in DMEM was 0.0022 ± 0.008 g/day in the first two weeks. Even though the plot suggests a slightly higher weight reduction for the Radiopaque samples (−0.03 g weight change compared to −0.02 g at 14 days), the difference was not statistically significant (p = 0.2 for DMEM). The weight change of the Radiopaque samples in saline was higher than the Vitamin E samples (p = 0.01). Table 4 shows the rate of leaching after two weeks and at 8 weeks for each sample group. There was no alteration in the weight of the control samples. After the second week, all samples had reached a steady state weight change of 0.02 ± 0.0001 g/day for both the Radiopaque and Vitamin E samples.
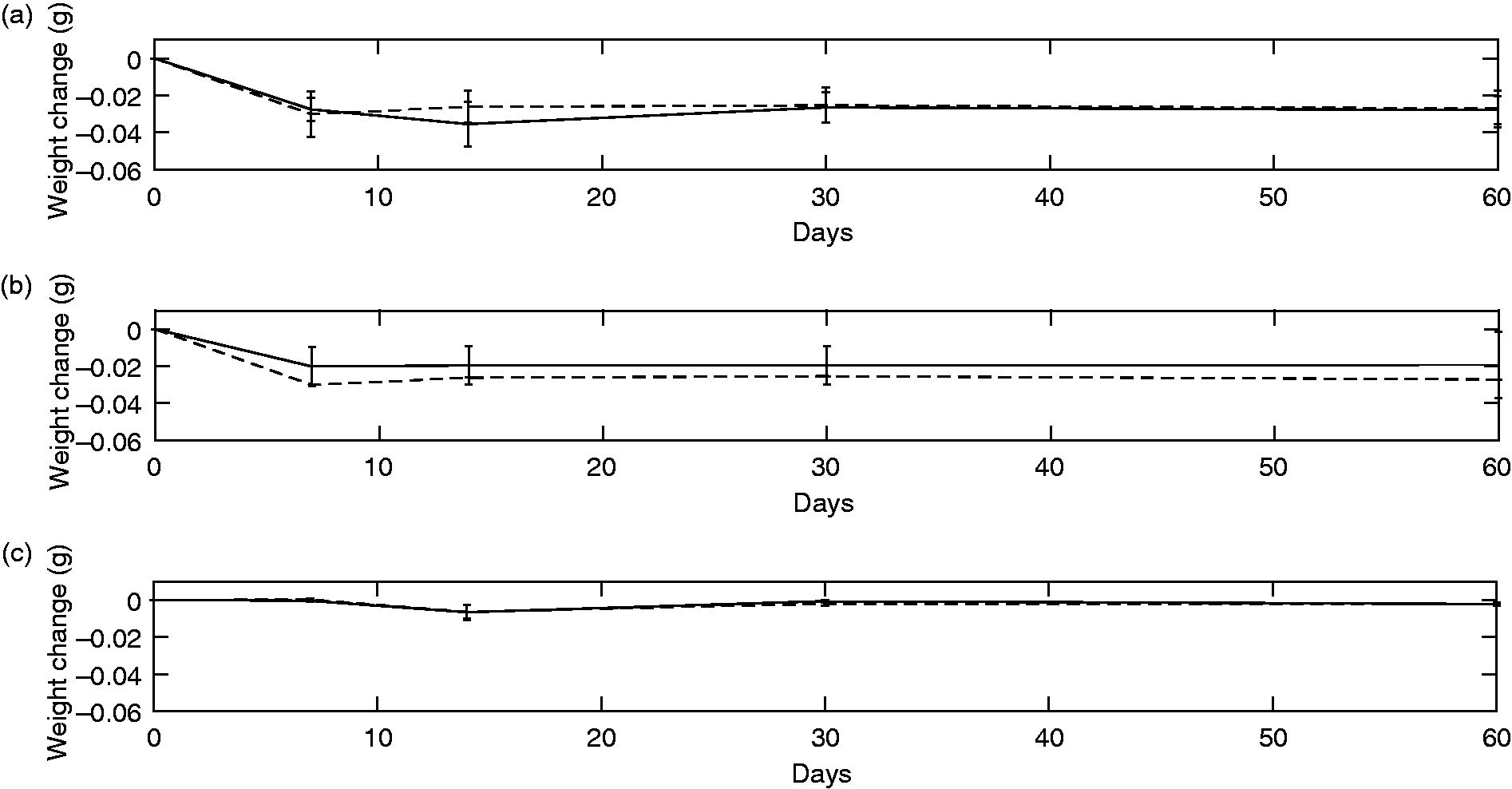
The leaching behaviour of (a) Radiopaque, (b) Vitamin E treated polyethylene samples, and (c) Thermally treated control samples, where the error bar represents the standard deviation. The Lipiodol leaching was slower than the Vitamin E leaching. The control samples were only treated thermally to ensure the the weight change was purely due to the leaching of the oil based fluid in the samples. The dotted lines represent results from samples held in DMEM and the solid lines indicate those in saline.
The average rate of leaching of Lipiodol or vitamin E out of the polyethylene samples at 2 and 8 weeks in saline or Dulbecco’s Modified Eagle’s Medium (DMEM). SD=standard deviation.

Discussion
This study aimed to evaluate the longevity of oil infused polyethylene (Lipiodol and Vitamin E) by examining how the thermal properties, oxidative stability, tensile properties, radiopacity and oil-diffusion stability might be affected by long-term implantation. The inclusion of an additive, such as Lipiodol for radiopacity or Vitamin E for oxidative stability, can improve the clinical performance of the polyethylene, but it is important to ensure (a) the functionality is maintained over the implant life and (b) the safety and performance of the material is not compromised.
Lipiodol infused radiopaque polyethylene has an enhanced X-ray attenuation due to the presence of an FDA approved oil-based contrast agent (Lipiodol). 6 This study showed that the surface radiopacity of aged radiopaque polyethylene samples was on average 54% lower than unaged radiopaque polyethylene. One possible explanation is that the ageing process allowed Lipiodol to diffuse further into the polyethylene away from the surface leading to a homogenised radiopacity and a lower surface radiopacity. The use of thermal treatment to cause homogenisation of oily fluids in polyethylene is deliberately performed for Vitamin E polyethylene to ensure the antioxidant properties protect the whole part. 25 Another possible explanation is that the high temperature led to the oxidation of Lipiodol compromising their functionality; there is insufficient data to support this idea however oxidation and degradation of contrast agents with temperature has been previously reported.26–28 Nuclear magnetic resonance (NMR) spectroscopy scans of aged and thermally treated Lipiodol (unpublished data) show aged Lipiodol contains a higher level of unsaturation compared with unaged Lipiodol; this may reduce the X-ray attenuation of Lipiodol. Unfortunately, the ageing protocol used in this study was designed to replicate the natural ageing of polyethylene and it has not been validated for Lipiodol.
The redistribution of radiopacity can be also associated with the effect of temperature on the crystallinity of polyethylene. 19 The diffusion of macromolecules in polyethylene is limited to the amorphous regions; the elevated temperature and ageing are both likely to cause a decrease in the crystalline regions providing more amorphous regions for Lipiodol to move away from the surface. 19 The degree of crystallinity is one of the most crucial properties of polyethylene. The crystalline content of polyethylene provides an additional resistance to mechanical deformation, so increasing the crystallinity reduces the ductility of the polyethylene. 29 Our previous studies showed that the Lipiodol treatment does not alter the degree of crystallinity of polyethylene. 19 The DSC results (accuracy of ±0.1∘C) from this study also confirmed no significant changes in the crystalline content of the aged samples (p > 0.05 for all the samples).
The oxidation index of all specimens increased after ageing. Oxidation leads to chain scission and formation of shorter chains 30 ; from a thermodynamic point of view, the shorter chains are able to re-arrange themselves into highly crystalline arrangements increasing in the degree of crystallinity, which contradicts the results from the DSC. The crystallinity can also be examined using FTIR; the peak at 989 cm–1 represents the crystalline content of polyethylene and the peak at 1305 cm–1 is an identification of the amorphous content of specimens. The FTIR spectra showed that while there was no change in the crystalline peak of the samples before and after ageing, a reduction in the peak at 1305 cm–1 indicates a reduction in the percentage of the amorphous phase. This suggests the amorphous regions were transformed into thinner, less perfect crystals, which could not be identified by neither DSC nor FTIR. 30 Previously it has been reported that even though oxidised retrieved polyethylene samples have increased percentage crystallinity, it is uncommon for thermally aged samples to show any significant increase in crystallinity.30,31
None of the current ageing protocols are able to fully reproduce the oxidative changes seen in naturally aged polyethylene. 32 Natural ageing of polyethylene is affected by multiple factors such as the concentration of free radicals, the duration of shelf storage, the surrounding fluid16,33 as well as many patient related factors such as lifestyle, level of activity or patients’ weight. Previous studies showed that after 4 weeks of accelerated ageing of non-irradiated polyethylene, there were some observations (e.g. ketone peak) which were not associated with long-term naturally aged explants. 34
Although this study showed signs of oxidation after ageing for all samples groups, oil-infused samples showed a superior oxidation stability compared to untreated controls. The increase in the oxidation index of the Radiopaque samples was almost half of the Thermally treated samples and Untreated samples (56% compared to 118%). The Radiopaque samples however had a high initial oxidation index; which may result from the aldehyde peak in Lipiodol which has a similar wavenumber to the peak used to calculate the oxidation index, potentially resulting in an over-estimation of the oxidation index.35,36 For a more comprehensive investigation, the peaks representing other oxidation products (e.g. ketone) can be checked. Ketone is one of the common products of oxidation and frequently was found in thermally aged polyethylene.32,34 None of the specimen groups (including the Radiopaque polyethylene) showed a peak for ketone before ageing, supporting the hypothesis that the high oxidation index of the unaged Lipiodol treated samples was not due to true oxidation but was as a result of a systematic error in the Oxidation Index calculation.
This study also evaluated the effect of oxidation on the tensile properties of polyethylene, the oil-free samples had significantly lower tensile properties (elastic modulus, yield, elongation at failure, p < 0.05) than any oil treated sample. For instance, the oil-free samples were very brittle and had an elongation at failure less than 50%, while this value for the oil treated samples was more than 300%. The oil free samples also had a lower modulus compared to the oil treated samples. Furthermore, many of the tensile properties of the Radiopaque samples were on average slightly lower than the Vitamin E samples, yet never statistically significant.
The leaching of Lipiodol was confirmed thorough a reduction in weight after the first measurement at week 2. After week two the rate reached a plateau suggesting minimal further leaching. This could be because fluid on the surface of the samples had diffused through to the centre, away from the surface, and was no longer able to diffuse out due to the very slow rate of diffusion. Neither DMEM nor saline fully represent the synovial fluid; saline does not contain the proteins or lipids and although DMEM has a similar protein content it has no lipid content. However, to our knowledge there is no cost-efficient fluid which fully represents synovial fluid. 37 The lipid content is of particular importance to oil-infused polyethylene as the carbonyl group of lipids could facilitate the diffusion of Lipiodol out of the polyethylene. 38
The ageing protocol used in the present study has been validated by comparing the density, oxidation and the mechanical properties of the artificially aged components with those sterilised and shelf-aged. 14 Most accelerated ageing methods have been developed to simulate shelf ageing caused by free radicals.39,40 This is because oxidative degradation is catalysed by the free radicals produced during sterilisation.4,41,42 Implants are normally sterilised and crosslinked using gamma irradiation. The material used in this study was un-irradiated and had not been treated with gamma irradiation. A study by Kurtz et al. suggested that any alteration in the manufacturing process (e.g. crosslinking) is likely to alter the oxidation mechanism. 31 Future work will evaluate the concentration of free radicals in the polyethylene prior to ageing in order to better understand the oxidation rate. 43
A limitation of this study, and the standard ageing protocols, was the lack of mechanical loading during ageing. The mechanical loading which will occur in vivo can itself can lead to chain scission and accelerate the oxidation via the formation of free radicals, which is the rate controlling step for oxidation.10,44 The lack of mechanical loading can also affect the leaching results as it may alter the diffusion pathway via alteration of the crystalline arrangement.45–48 Future studies should evaluate the effect of static and dynamic loading on the long-term properties of polyethylene.
Conclusion
This study examined the effect of oily fluids on the long–term properties and durability of polyethylene. The results confirmed that the presence of an oily fluid (Lipiodol and Vitamin E) in polyethylene improved the oxidative stability, tensile strength, yield, elongation at failure after ageing, when compared with Untreated and Thermally treated polyethylene. The improved chemical stability is favourable for long-term medical applications; however, the surface radiopacity of the Radiopaque samples reduced by 54% after 4 weeks of accelerated ageing. Although the Radiopaque polyethylene was still clearly visible on a CT scan (500 HU), the surface X-ray attenuation may not be adequate to monitor the clinical performance of a polyethylene joint replacement component through hard tissues over a long period of time (e.g for monitoring wear, creep or migration). It may be possible to mitigate the reduction in radiopacity after ageing through crosslinking, which has proven successful for Vitamin E polyethylene. In summary, oil treated polyethylene has shown a higher tensile and oxidative stability for joint replacement applications compared to standard polyethylene. This work has also highlighted a need for more sophisticated accelerated ageing standard tests, which are more representative of the impact of surrounding physiological fluids, mechanical loading, and the impact of oily additives within polyethylene.
Supplemental Material
sj-pdf-1-jba-10.1177_0885328220977353 - Supplemental material for Chemical stability of oil-infused polyethylene
Supplemental material, sj-pdf-1-jba-10.1177_0885328220977353 for Chemical stability of oil-infused polyethylene by Fedra P Zaribaf, Harinderjit S Gill and Elise C Pegg in Journal of Biomaterials Applications
Footnotes
Acknowledgements
Authors would like to acknowledge Celanese for providing the medical grade polyethylene, Mrs Clare Ball for their technical support, and Prof Michael Whittlesey from the Department of Chemistry (University of Bath) for access to FTIR.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the resesarch, authorship and publication of this article: This project was supported by the University of Bath through the University Scholarship Research Allowance funding scheme.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.