Abstract
Background
Post-critical illness cognitive dysfunction (PCICD) is a frequent and debilitating outcome among survivors of critical illness. Although sepsis has been associated with poor cognitive outcomes, its independent contribution remains unclear due to overlapping clinical factors. This study sought to characterize cognitive recovery trajectories over 12 months after intensive care.
Methods
In this single-center prospective cohort study, adult ICU survivors were assessed at 1, 3, 6 and 12 months post-discharge using telephone-administered Mini-Mental State Examination (MMSE) or Montreal Cognitive Assessment (MoCA-Blind). Total scores were standardized within instrument (z-scores). Linear mixed-effects models evaluated change in z-scores over time. Domain-specific analyses examined whether any cognitive domain was disproportionately impaired. Logistic regression estimated odds of cognitive impairment adjusting for time, sepsis status, test type, age, Charlson index, peak SOFA, and benzodiazepine exposure; complete-case analyses were used.
Results
Of 185 eligible patients, 84 (45%) completed at least one cognitive assessment. Standardized scores improved from 1 to 3 months (+0.40 SD; 95% CI 0.02-0.78; p = 0.04) and 6 months (+0.54 SD; 95% CI 0.10-0.98; p = 0.02), with a similar but non-significant rise by 12 months (+0.49 SD; 95% CI −0.05 to 0.95; p = 0.10). Adjusted odds of impairment declined at 6 (OR 0.25, 95% CI 0.12-0.55) and 12 months (OR 0.34, 95% CI 0.14-0.85) versus 1 month; the 3-month reduction did not reach significance (OR 0.48, 95% CI 0.23-1.04). Sepsis was not associated with impairment (OR 1.49, 95% CI 0.63-3.56). No single cognitive domain showed a significant longitudinal slope.
Conclusions
ICU survivors show measurable cognitive recovery over the first year—most prominently by 3–6 months—with reduced odds of impairment by 6 and 12 months. Sepsis did not independently alter recovery. These findings support early post-ICU cognitive follow-up and rehabilitation within the first six months after discharge.
Keywords
Introduction
As survival after critical illness improves, long-term cognitive sequelae have become a major public health concern. 1 Post-critical illness cognitive dysfunction (PCICD) encompasses persistent deficits in attention, memory, executive function, and processing speed that may last for months to years following discharge from the intensive care unit (ICU). These impairments are both common and disabling, limiting quality of life, independence, and return to work. Cognitive impairment after ICU admission is also taxing to patients and their families and carries an enormous societal cost estimated at $18 billion per year. 2 Risk is heightened in ICU-related conditions such as sepsis, shock, and respiratory failure, and is compounded by exposures including prolonged mechanical ventilation, sedative and steroid use, immobility, and comorbid disease.3,4
Meta-analyses estimate that nearly 50% of patients experience cognitive impairment within one month of ICU discharge, with 28.3% still impaired more than one year later. Honarmand et al similarly reported PCICD in ∼46% of survivors two years after discharge. 5 These findings underscore the need to map cognitive recovery trajectories, particularly in the first 3–6 months post-discharge when interventions may be most effective.
Sepsis, one of the most common ICU admission diagnoses, has been implicated in long-term PCICD. Sepsis-associated encephalopathy likely reflects convergent mechanisms—exuberant neuroinflammation (microglial activation and cytokine signaling), blood–brain barrier disruption, microvascular dysfunction, and altered neuroglial coupling—that impair network function and plasticity, plausibly yielding prolonged deficits in attention, memory, and executive function.6–8 One study reports up to a three-fold increased risk of cognitive dysfunction in septic versus non-septic patients. 9 Conversely, a multicenter cohort of mechanically ventilated patients found no significant difference in cognitive, psychiatric, or quality-of-life outcomes at six months between septic and non-septic survivors of similar illness acuity. 10 Thus, the independent effects of sepsis on cognitive decline remain uncertain.
Other clinical factors—including age, comorbidity burden, delirium, and educational level—also shape cognitive outcomes. Advanced age and higher Charlson Comorbidity Index scores increase risk, whereas higher education appears protective; patients with more than 12 years of education had up to a 95% lower odds of PCICD compared with those with six or fewer years. 11 Delirium is the most consistent modifiable predictor, independently associated with long-term decline, mortality, and prolonged ICU stay, with an estimated patient-level 30-day cumulative cost of $18 000.12–14 Benzodiazepine exposure, which increases delirium risk, has been identified as another potentially modifiable contributor. 12 Consistent with PADIS, modern ICU sedation strategies preferentially use propofol or dexmedetomidine over benzodiazepines, 15 paralleling practice trends showing declining benzodiazepine use and links to delirium and worse outcomes. 16 Nevertheless, benzodiazepines remain in selective use for specific indications—most commonly alcohol withdrawal or refractory agitation—and when non-benzodiazepine agents are contraindicated, poorly tolerated, or unavailable.
Despite growing evidence, important gaps remain. The longitudinal course of specific cognitive domains within global PCICD—particularly in septic versus non-septic patients—remains poorly characterized, and the influence of benzodiazepine exposure is not well understood. To address these gaps, we conducted a prospective cohort study of ICU survivors, assessed at 1 month, 3 months, 6 months, and 12 months post-discharge using validated cognitive tools. We examined recovery trajectories, stratified by sepsis status, to identify predictors of persistent or progressive impairment.
Methods
Study Design and Setting
A single-center, prospective cohort study was conducted in the medical and surgical intensive care units (ICUs) of an academic medical center from September 2020 to August 2024. Consecutive adult patients (≥18 y) who survived an index episode of critical illness and were discharged alive from the ICU were screened. Sepsis and septic shock were defined by Sepsis-3 criteria.17,18 Exclusion criteria were: (1) pre-morbid severe neuro-cognitive disorder prohibiting assessment, (2) non-English speakers, and (3) refractory delirium precluding follow-up.
Cognitive Assessments
Cognitive function was evaluated via telephone interviews at 1 month, and again at 3, 6, and 12 months after ICU discharge using two standardized instruments. Initially, we administered the Mini-Mental State Examination (MMSE; score range 0-27; Appendix A). In September 2022, we transitioned to the blind/telephone Montreal Cognitive Assessment (MoCA v8.1 Blind/Telephone 19 ; score range 0-22). The transition from MMSE to MoCA-Blind was motivated by methodological advantages inherent to the MoCA-Blind instrument. Unlike the MMSE, the MoCA-Blind encompasses more complex cognitive tasks, including an abstraction component, making it more sensitive for detecting subtle cognitive deficits frequently observed among ICU survivors. Additionally, the MoCA-Blind version used in this study was specifically designed to facilitate telephone administration, allowing for consistent, remote follow-up assessments. Both tools assessed orientation, attention, delayed recall, and language; however the MMSE additionally included an immediate recall task, whereas the MoCA-Blind incorporated an abstraction component. The MoCA-Blind assessment was administered by a trained/certified research coordinator.
We described baseline characteristics for participants completing no cognitive assessments versus ≥1 assessment (among those alive for at least one follow-up window) and, separately, for those completing exactly one versus ≥3 assessments (among those alive for ≥3 windows). Follow-up was censored at death to reduce survivorship bias.
Benzodiazepine Exposure
Given the observational nature of this study, benzodiazepine exposure was converted to lorazepam equivalents (LE) using route-specific factors: intravenous (IV) midazolam = 0.5×, oral (PO) midazolam = 0.1×, lorazepam (IV/PO) = 1×. The IV factor reflects RCT-based equipotency (lorazepam ≈2× midazolam) and published ICU conversion tables 20 ; the PO factor conservatively accounts for midazolam's low, variable oral bioavailability and relatively stable oral lorazepam bioavailability, with additional variability in enteral absorption during critical illness.21–23 To assess robustness to alternative published ratios, we performed a sensitivity analysis using IV midazolam = 0.33×.24,25
Statistical Analysis
All analyses were conducted using Python v3.11, pandas v2.2, statsmodels v0.14, and scikit-learn v1.5 libraries. Raw MMSE (0-27) and MoCA-Blind (0-22) totals were standardized to within-instrument z-scores (score minus the instrument's sample mean, divided by its sample SD) so both instruments contribute on a common scale. All subsequent analyses were performed on these standardized values.
To characterize the unadjusted cognitive recovery trajectory, a linear mixed-effects model with restricted maximum likelihood estimation was implemented. Standardized cognitive z-scores served as the dependent variable, modeled as a function of assessment time points (categorical: 1, 3, 6, and 12 months), cognitive test type (MoCA-Blind vs MMSE), and their interaction term. Primary models used subject-level random intercepts. We explored random slopes by visit; when these failed to converge, we retained the intercept-only structure.
To examine potential differences in recovery trajectories between septic and non-septic patients, we conducted a sepsis-stratified mixed-effects analysis, substituting cognitive test type with sepsis status (yes/no) and incorporating a visit-by-sepsis interaction.
For odds-of-impairment models, impairment was defined a priori as MoCA-Blind < 19 or MMSE < 22. We defined impairment using instrument-appropriate thresholds for non-visual administration: MoCA-Blind (22-point scale) was classified as impaired at <19, consistent with published guidance that ≥19/22 is “normal.” 26 For MMSE administered without visual items (“MMSE-blind”), using <22 preserves the conventional MMSE impairment boundary on the reduced scale and is consistent with descriptions of blind/telephone MMSE adaptations. 27 When both instruments were available we prioritized MoCA because it is generally more sensitive than MMSE for detecting milder/executive deficits, improving detection of subtle post-ICU impairment.
We fit GLM–logit models with subject-clustered robust standard errors, adjusting for visit (1, 3, 6, and 12 months), sepsis status, test type (CA-Blind vs MMSE), age, Charlson index, peak SOFA, and benzodiazepine exposure (days of use and total lorazepam-equivalent dose). Analyses were complete-case without imputation.
Analyses used all available observations at each visit (unbalanced panels). For covariate-adjusted models, we performed complete-case analysis; no imputation was used. Visualizations, including line plots with 95% CIs, boxplots, waterfall plots, and forest plots, were generated using seaborn v0.13.
Results
Cohort and Assessments
Among survivors eligible for ≥1 follow-up window, 131 participants completed ≥1 cognitive assessment and 24 completed none (Supplementary Table S1). Comparison between sepsis and non-sepsis cohorts is presented in Table 1. Compared with cognitive assessment non-completers, completers were younger and had lower comorbidity and acute-illness severity (lower APACHE II and SOFA), shorter hospital stays, more ICU-free days, and a lower proportion with sepsis (all |SMD| ≥ 0.20).
Clinical Features of Critically Ill Patients with and without Sepsis.
Among those eligible for ≥3 windows, 41 completed ≥3 assessments and 20 completed exactly one (Supplementary Table S2). Relative to the 1-assessment group, the ≥3-assessments group had higher illness severity (higher APACHE II and SOFA), longer hospital stays, fewer ICU-free days, higher Charlson index, and more sepsis (all |SMD| ≥ 0.20).
Overall Recovery Trajectory
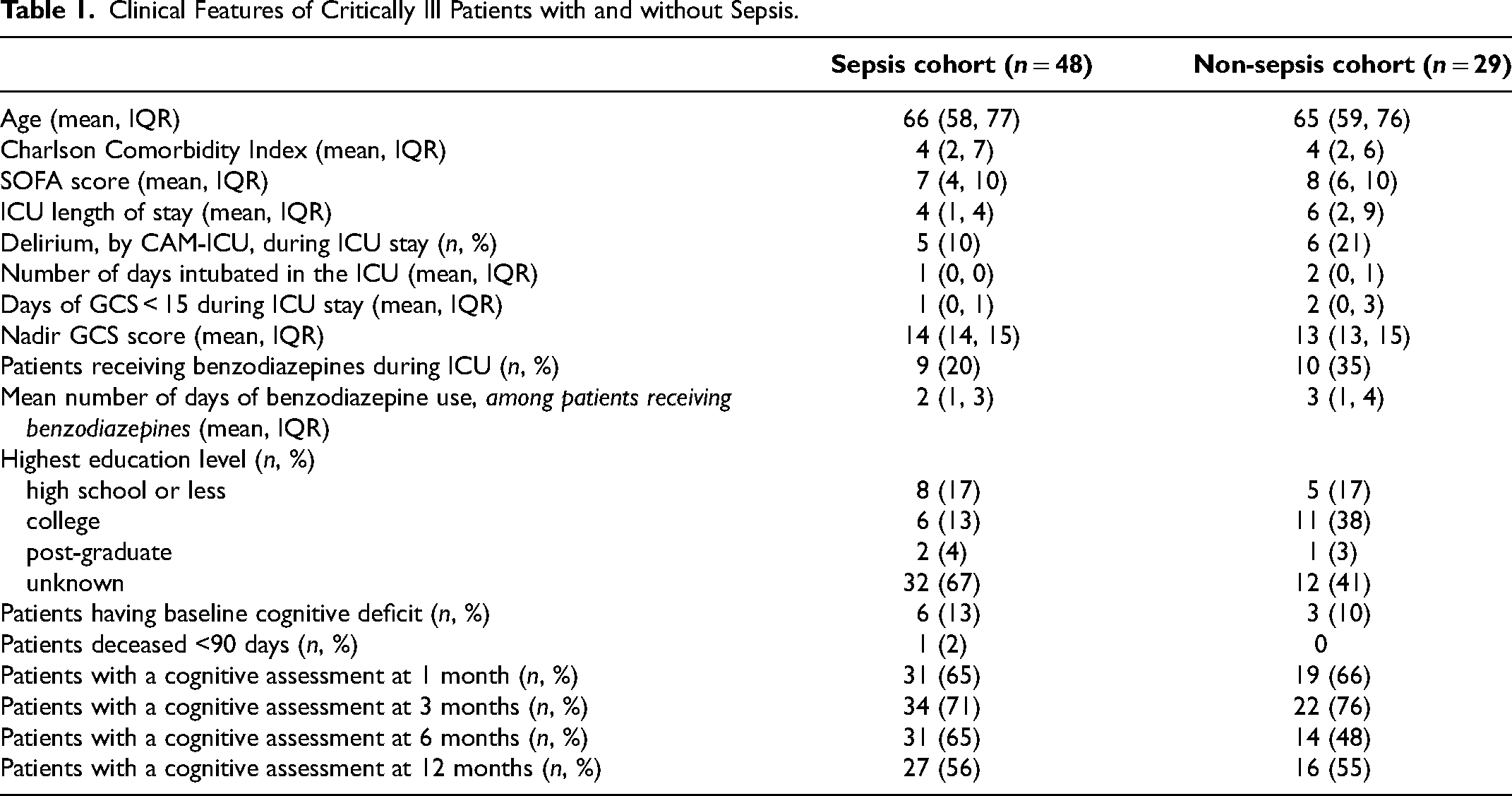
Standardized z-scores improved from 1 to 3 months (+0.40 SD; 95% CI 0.02-0.78; p = 0.04) and 6 months (+0.54 SD; 95% CI 0.10-0.98; p = 0.02), with a similar but non-significant increase by 12 months (+0.49 SD; 95% CI −0.05 to 0.95; p = 0.10) (Fig. 1A). Test type (MoCA-Blind vs MMSE) was not associated with z-scores (Fig. 2), and sepsis-time interactions were not significant, indicating comparable trajectories across instruments and sepsis status (Figs 1A–B, 3). These continuous improvements were paralleled by lower adjusted odds of impairment at 6 and 12 months (Table 2). Note that z-score models treated cognition continuously, whereas the logistic models used a thresholded definition of impairment.
Recovery of Cognitive Scores, as Measured by MoCA and MMSE. (A) Standardized Total Score Recovery, Showing Mean z-Scores at 1, 3, 6 and 12 mo post-ICU, with 95% CI from a Categorical Time Mixed Effects Model. No Model Assumptions Other Than Independence. (B) Forest Plot for Fixed Effect Coefficients. Point Estimates and 95% CIs for Fixed Effects from the Continuous Mixed-Effects Model (Time, Tool, Time-Tool Interactions). The Vertical Line at Zero Denotes no Effect. (C) Spaghetti pot of Individual Patient Cognitive Trajectories. Gray Lines for Each Patient's z-Score Trajectory and Overlaid Colored Mean Trajectories ±95% CI. (D) Boxplot Distribution Over Time of z-Scores at Each Time Point (IQR ± 1.5 Times IQR, with Outliers). n (MoCA/MMSE): 1 mo = 34/16; 3 mo = 38/19; 6 mo = 27/19; 12 mo = 24/19.
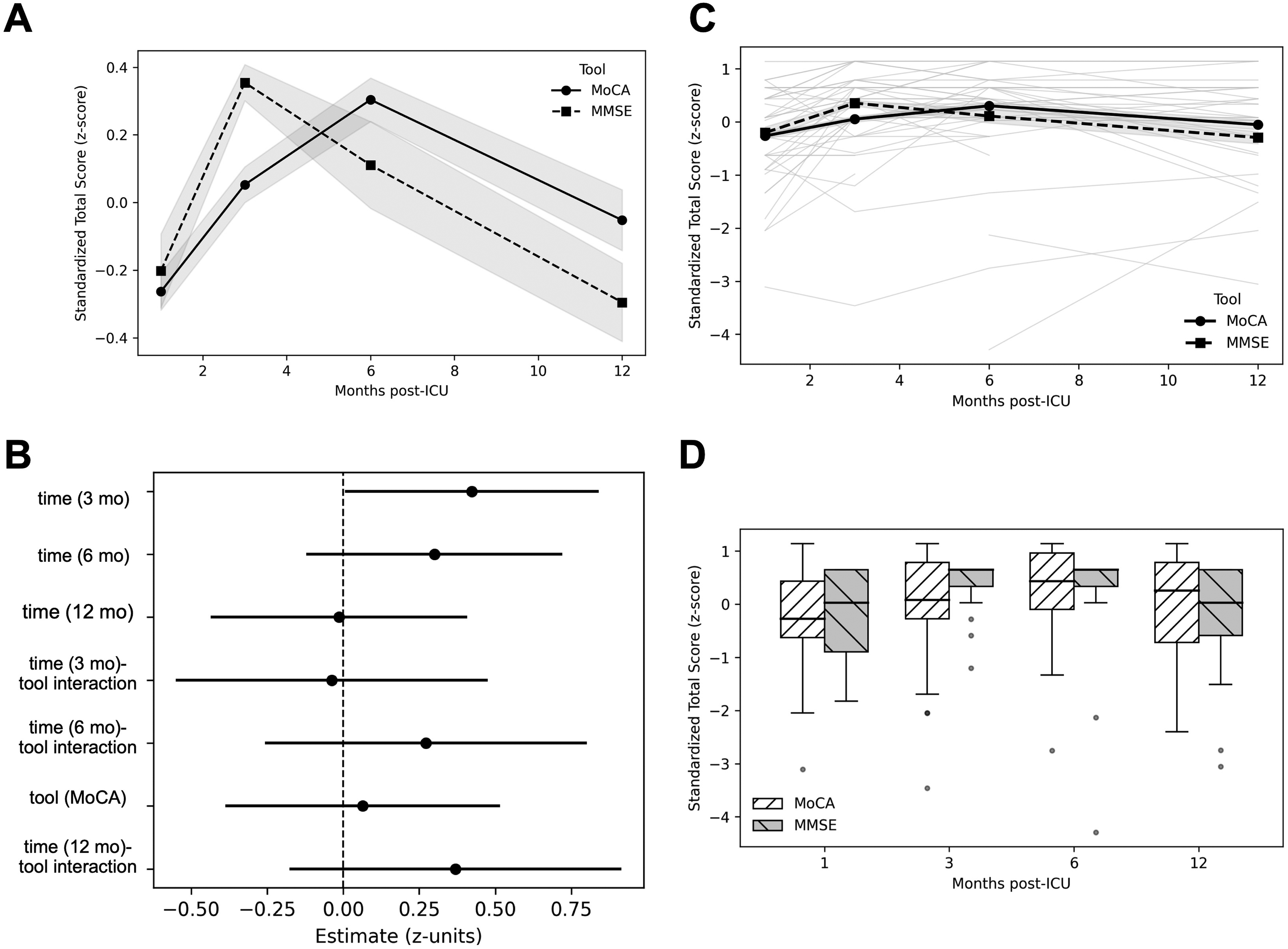
Estimated Marginal Mean Recovery Over Time. Model-Predicted z-Score Trajectories at 30 Days, 3 Months, 6 Months, and 12 Months—Derived from a Linear Mixed Effects Model That Treats Time/Visit as a Continuous variable. Solid Lines Represent the Estimated Marginal Means (EMMs) of Standardized Cognitive Scores per Month; Shaded Areas Denote 95% Confidence Intervals. Unlike Fig 1, which Plots Observed, Mean z-Scores at Each Discrete Time Point, This Panel Illustrates the Smoothed, Model-based Trend in Cognitive Recovery Across the Entire 12 Month Period.
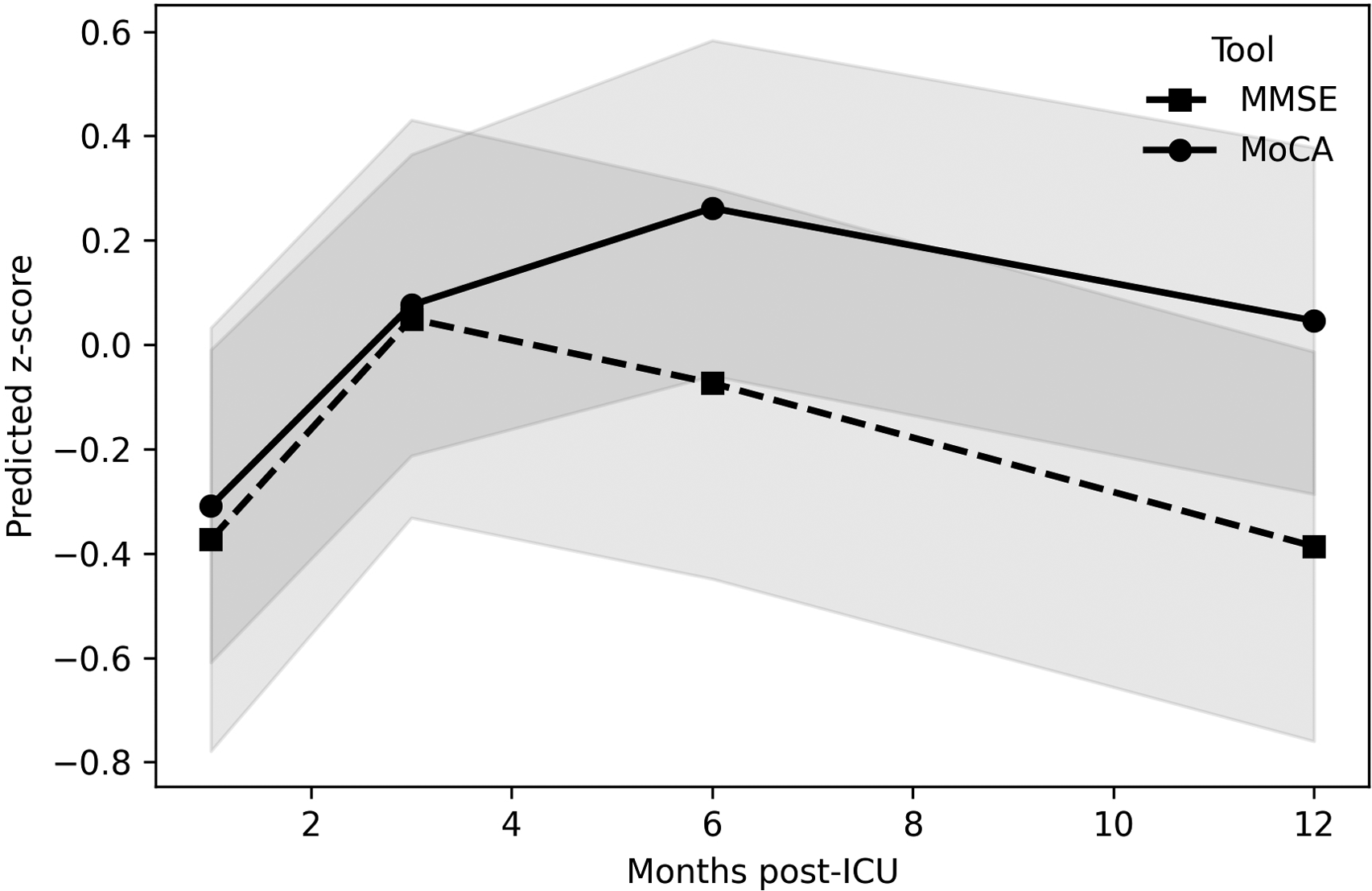
Cognitive Recovery in Septic Versus Non-Septic Patients. Mean z-Scores ±95% CI at 1, 3, 6 and 12 Months. n (non-Sepsis/Sepsis): 1 mo = 33/49; 3 mo = 36/53; 6 mo = 29/43; 12 mo = 27/37.
Adjusted Odds of Cognitive Impairment by Follow-up Visit.
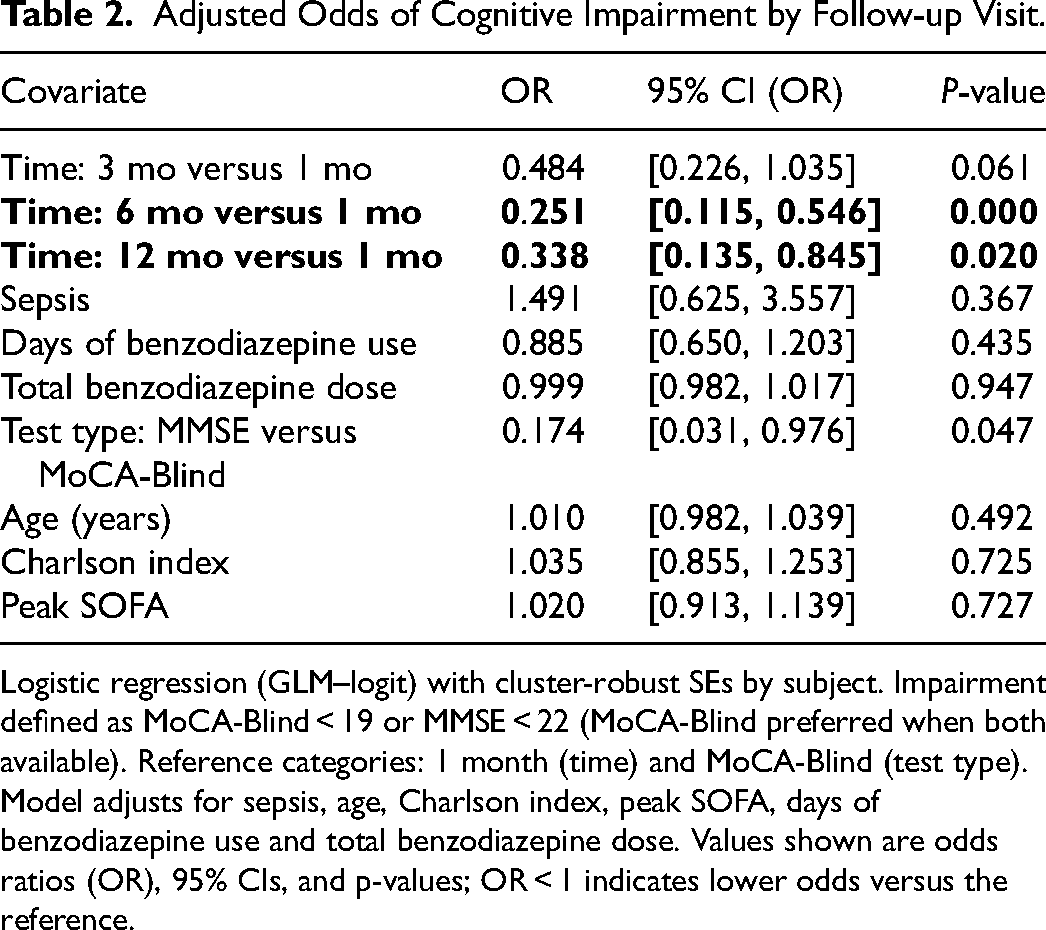
Logistic regression (GLM–logit) with cluster-robust SEs by subject. Impairment defined as MoCA-Blind < 19 or MMSE < 22 (MoCA-Blind preferred when both available). Reference categories: 1 month (time) and MoCA-Blind (test type). Model adjusts for sepsis, age, Charlson index, peak SOFA, days of benzodiazepine use and total benzodiazepine dose. Values shown are odds ratios (OR), 95% CIs, and p-values; OR < 1 indicates lower odds versus the reference.
Adjusted Clinical Predictors
Adjusted odds of impairment declined over time after ICU discharge (Table 2). Compared with 1 month, odds were lower at 6 months (OR 0.25, 95% CI 0.12-0.55; p < 0.001) and 12 months (OR 0.34, 95% CI 0.14-0.85; p = 0.020), with a non-significant reduction at 3 months (OR 0.48, 95% CI 0.23-1.04; p = 0.061). Sepsis was not associated with impairment (OR 1.49, 95% CI 0.63-3.56; p = 0.37). Neither days of benzodiazepine use (OR 0.89, 95% CI 0.65-1.20; p = 0.44) nor total benzodiazepine dose (OR 1.00, 95% CI 0.98-1.02; p = 0.95) independently predicted impairment. MMSE showed lower odds of impairment versus MoCA-Blind (OR 0.17, 95% CI 0.03-0.98; p = 0.047), consistent with instrument differences. Findings for thresholded impairment differ from our continuous z-score analyses, likely reflecting the dichotomization of scores and reduced complete-case sample.
Domain-Specific Trajectories
Modeled separately, no domain showed a statistically significant monthly change over 12 months (Fig. 4). Slopes (β per month, p): abstraction 0.039 (0.087), orientation 0.038 (0.095), attention −0.009 (0.701), immediate recall −0.005 (0.859), delayed recall −0.029 (0.339), language −0.003 (0.903).
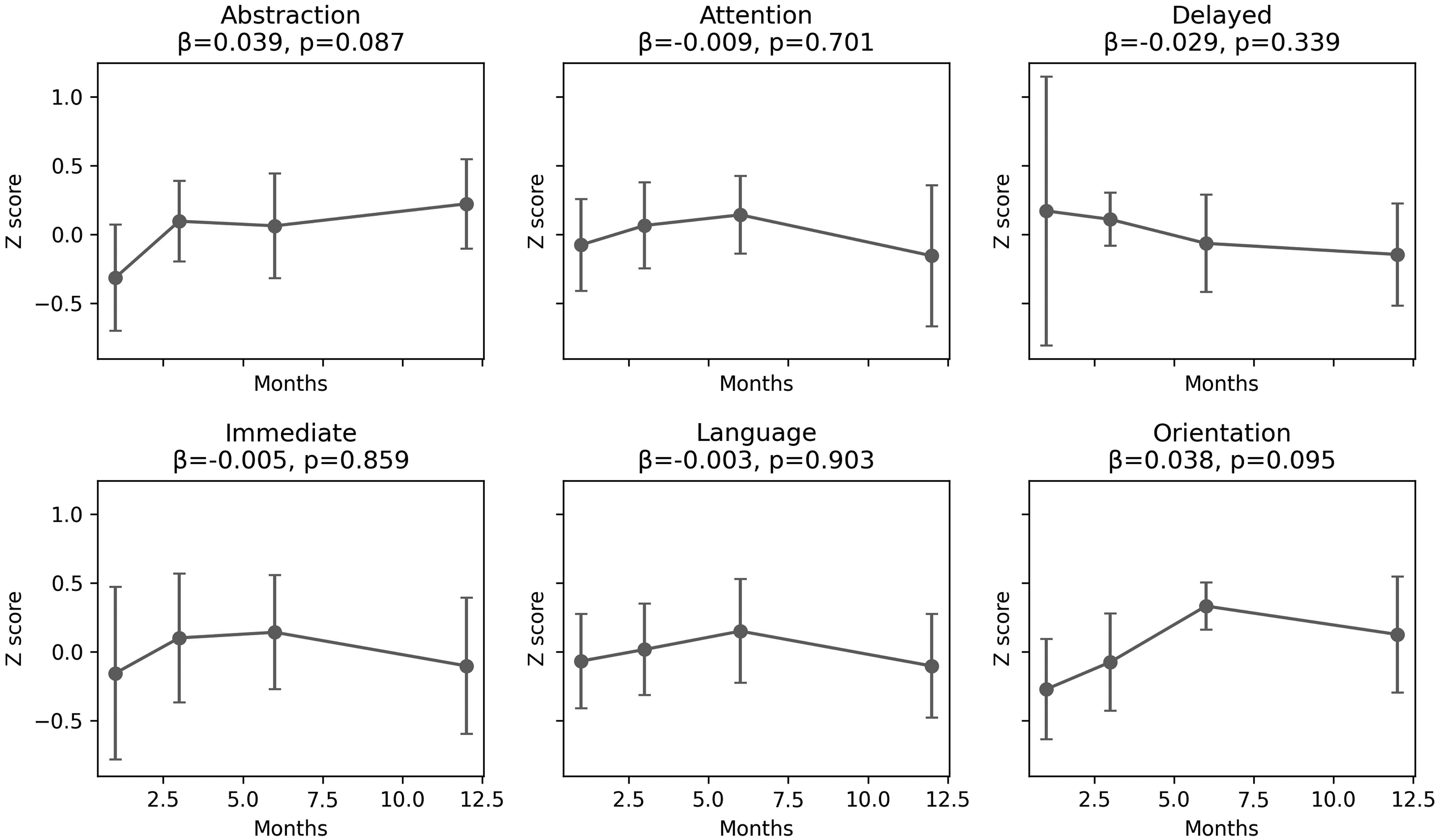
Small-Multiples of Cognitive Domain Recovery. Each Panel shows the Mean Standardized (z-Scored) Performance in One Cognitive Domain (Abstraction, Attention, Delayed Recall, Immediate Recall, Language, Orientation) at 1 mo, 3 mo, 6 mo, and 12 mo Post-ICU. Points Indicate the Mean; Vertical Bars Denote 95% Confidence Intervals (mean ± 1.96×SE). In Each Subplot Title, the Estimated Slope (β) and P-value for the Time Effect is Shown, Derived from a Linear Mixed-effects Model with a Random Intercept (or, Where the Mixed Model Did Not Converge, from Ordinary Least Squares). No Domain Exhibited a Statistically Significant Improvement Over the Study Period. Immediate = Immediate Recall; Delayed = Delayed Recall.
Discussion
This prospective longitudinal cohort study shows that standardized cognitive performance improved by 3–6 months and did not increase further at 12 months, while the odds of cognitive impairment were significantly lower at 6 and 12 months. These findings align with prior studies. 1 Impairment was global rather than domain-specific (no single domain's slope reached significance). Sepsis status did not independently affect the trajectory or magnitude of recovery. Recent research similarly suggests that sepsis alone may not be a strong independent predictor of long-term cognitive impairment after critical illness.10,28
In the impairment-based logistic model adjusted for age, comorbidity, illness severity, sepsis, and test type, benzodiazepine exposure was not independently associated with impairment: days of use OR 0.89 (95% CI 0.65-1.20; p = 0.435) and total dose OR 0.999 (95% CI 0.982-1.017; p = 0.947). Only 14 of 65 participants (22%) received any benzodiazepine, and days and total dose were highly collinear (r ≈ 0.93), which likely limited power and precision. These null findings should be interpreted alongside prior work linking benzodiazepines to delirium and later cognitive decline and the broader trend toward reduced ICU use of these agents. 16
Survivors, on average, improve most in the first 6 months; this is a practical window for screening and referral. By referral we mean timely linkage to post-ICU/geriatric cognitive rehabilitation—neuropsychology evaluation with feedback and care planning; occupational therapy and speech-language pathology targeting attention, memory, and executive function; compensatory strategy coaching and assistive technologies (eg, calendars, pill organizers, smartphone reminders); caregiver education; and, when appropriate, return-to-work/driving counseling and community-based programs. We found no independent sepsis effect after adjustment, suggesting survivorship care should be broadly applied following critical illness, regardless of sepsis status. Especially in older adults, cognitive rehabilitation should be prioritized on par with physical rehabilitation, which is often pursued more aggressively, because cognitive deficits strongly influence independence, medical adherence, and safety.
This study has several limitations. First, it was conducted at a single academic ICU with a modest sample size which may limit generalizability. The cohort may not reflect the diversity of case-mixes or rehabilitation practices seen in community or non-academic settings. The study is under-powered to detect subtle differences, particularly in interaction terms across subgroups.
Second, participant attrition and unbalanced follow-up introduce the potential for bias. While we employed linear mixed models that can handle data under the “missing at random” assumption, if sicker patients were less likely to complete cognitive testing, our estimates may understate the true cognitive burden. Moreover, data missingness may have been non-random: participants with greater cognitive impairment were likely underrepresented because they were less likely to answer or complete telephone assessments.
Third, we lacked formal cognitive baseline assessments prior to ICU admission. Although we standardized scores within each cognitive tool, we cannot determine whether individuals recovered to their premorbid cognitive level. This absence limits interpretation of whether changes over time reflect true cognitive decline or return to baseline function.
Fourth, missing covariates reduced the complete-case sample for adjusted analyses, and education had substantial missingness and was not included. Although our fully adjusted mixed-effects model incorporated age, comorbidity, illness severity, ICU length of stay, benzodiazepine exposure, sepsis status, test type, and selected neurological covariates, residual confounding by unmeasured factors (eg, delirium duration, socio-economic context) is still possible.
Fifth, we harmonized MMSE and MoCA-Blind by standardizing scores within each instrument (z-scores) rather than applying a crosswalk. This reduces uncertainty from deriving or using linking functions, but it assumes comparable variance structures across instruments and does not yield raw-score equivalence; our inferences are therefore about standardized levels and change, not absolute points.
Despite these limitations, our study adds to the growing literature on cognitive trajectories following septic and non-septic critical illness. Future research with larger, multicenter cohorts and more comprehensive covariate data is warranted to further explore these associations.
Supplemental Material
sj-docx-1-jic-10.1177_08850666251395999 - Supplemental material for Longitudinal Cognitive Recovery After Critical Illness: Trajectories in Sepsis and Non-Sepsis Survivors
Supplemental material, sj-docx-1-jic-10.1177_08850666251395999 for Longitudinal Cognitive Recovery After Critical Illness: Trajectories in Sepsis and Non-Sepsis Survivors by Ruhi Sahu, Ruth-Ann Brown and Anthony S Bonavia in Journal of Intensive Care Medicine
Footnotes
Acknowledgements
Not applicable.
Author Contributions
RS and ASB conceived the study. ASB designed the study. RB and RS curated the data, while RS and ASB performed formal analyses. RS and ASB prepared the figures and drafted the manuscript. All authors reviewed and edited the final version. ASB supervised the project and secured funding.
Consent for Publication
Not applicable.
Consent to Participate
All participants, or their legally authorized representatives (LARs), provided informed consent prior to study enrollment. Written consent was obtained whenever feasible. In situations where the LAR could not be physically present and timely enrollment was required, verbal consent was obtained by the study coordinator in accordance with IRB approval.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
This study was approved by the Penn State University Human Subjects Protection Office (IRB Protocol #15328, 7/30/2020). The studies were conducted in accordance with local legislation and institutional requirements. The study was also registered on clinicaltrials.gov (NCT03146546).
Funding
This study was funded by the National Institute of General Medical Sciences grant #K08GM138825 and #R35GM150695 (ASB), of the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
Appendix A: The Mini-Mental Status Examination (MMSE)
1. What is the year/season/date/day/month (score 0–5, Orientation Domain) _______
2. Where are you (country/State/Town/hospital or bldg/floor or street (score 0–5, Orientation Domain) _______
3. I am going to name 3 things and I want you to remember them. Fish, hook, shoe, green.
First, can you repeat these? (score 0–3, Memory - Immediate Recall Domain) _______
4. Can you count backward from 100 by 7 (stop after 5 answers, score 0–5, Attention Domain) _______
5. Can you spell “world” (as in, the world we live in) backward? (score 0–5, one point per letter, Attention Domain) _______
6. Can you remember the 3 things I had asked you to remember before (score 0–3, Memory - Delayed Recall Domain) _______
7. Can you repeat the following: “no if, and or buts” (score 0–1, Language Domain) _______
Total Mini-Mental Status Exam Score_________________(range 0–27)
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
