Abstract
Introduction
Viral severe acute respiratory infections (SARI) are a major cause of intensive care unit (ICU) admission, with a significant burden and mortality. Comparative clinical data of patients admitted to the ICU with virus infections other than SARS-CoV-2 or influenza virus (IV) infection are limited. Therefore, this study investigates patient characteristics, clinical outcomes, and ventilation parameters of ICU patients admitted with SARI caused by Respiratory syncytial virus (RSV), Human metapneumovirus (HMPV), IV, or Parainfluenza virus (PIV).
Methods
A retrospective cohort study was conducted of patients with SARI admitted to the ICU of the Spaarne Gasthuis, a Dutch secondary teaching hospital, between 2017 and 2023.
Results
277 patients were included, with RSV (n = 51), HMPV (n = 40), IV (n = 142), and PIV (n = 44) infections respectively. Pre-existing hematological malignancies were more common in RSV patients. No significant differences were found in length of hospital stay or ventilation parameters across the respective virus groups. Median duration of ICU stay was four days (IQR 2-7). Bacterial co-infections, pulmonary infiltrates and a higher ROX-index were more common in patients with noninvasive ventilation (NIV) failure. Hospital mortality rates were not different between the groups; RSV (25.5%), HMPV (15%), IV (24.6%), and PIV (20.5%).
Conclusion
This study analyzed ICU patients with SARI caused by HMPV, RSV, IV, or PIV, revealing four key findings: high ICU, hospital and 1 year mortality rates with no differences and similar mechanical ventilation parameters between the groups, risk factors for NIV failure linked to prolonged ventilation, and co-morbidities associated with severe disease.
Keywords
Introduction
Following the COVID-19 pandemic, there has been a resurgence of common respiratory pathogens contributing to community-acquired pneumonia (CAP). 1 While bacterial pathogens remain prominent, a substantial proportion (18-30%) of severe CAP and severe acute respiratory infections (SARI) is attributable to viral etiologies. 2 Viral respiratory infections therefore represent a significant cause of intensive care unit (ICU) admissions, with a substantial impact on patient morbidity and mortality. Among critically ill adults presenting with respiratory infections, viral pathogen prevalence ranges from 17% to 53% depending on seasonality and geographical area. 3 Besides the now endemic SARS-CoV-2, frequently detected respiratory viruses in critically ill populations are influenza virus (IV), respiratory syncytial virus (RSV), human metapneumovirus (HMPV), and human parainfluenza virus (PIV). 4
These respiratory virus infections, due to their high incidence, impose a significant burden on morbidity and mortality as well as on healthcare facilities. Influenza has been associated with a reported 30-day mortality rate of 5–7% in hospitalized patients, while RSV has a mortality rate of 7–8% in older patients, increasing to 55% in high-risk individuals.5–8 A recent meta-analysis demonstrated that hospitalization was required in 24% of the elderly patients with acute RSV infection and admission to the ICU was necessary in 5%, increasing to 24% in high-risk patients. 9 It remains unclear whether seasonality and prevalence have shifted permanently from pre-COVID-19 patterns, which could significantly impact the burden on healthcare institutions.
The course of the disease can range from mild to severe infections, which may eventually lead to acute respiratory distress syndrome (ARDS).10–12 Several risk factors have been identified as predictors for the development of complicated SARI, such as: age over 60, chronic obstructive pulmonary disease (COPD), chronic heart disease, chronic kidney disease, long-term care facility residence and being immunocompromised. Early identification of high-risk patients facilitates prompt treatment and may lead to improved clinical outcomes. However, most of these data have been generated for influenza patients.5,6,8,13–15
Treatment primarily consists of supportive care, including intravenous fluid administration and supplemental oxygen therapy. During ICU admission, patients may require high-flow oxygen therapy (HFOT), non-invasive ventilation (NIV) or invasive mechanical ventilation (IMV).3,10 Additionally, most patients receive antibiotics to treat or prevent secondary bacterial infections or as part of selective decontamination of the digestive tract (SDD). Influenza virus infections may be treated with antiviral medications such as oseltamivir or baloxavir marboxil.16,17 These antiviral medications are effective when administered early in the infection to prevent (ICU) admission; however, their effectiveness decreases once patients become symptomatic. For RSV, only the off-label use of ribavirin in high-risk patients has been documented and more recent use of RSV monoclonal antibodies (palivizumab or nirsevimab) in infants. 18
The primary rationale for studying the impact of IV, RSV, HMPV, and PIV infections is that these contribute significantly to the burden of disease associated with severe CAP and SARI, leading to substantial morbidity and mortality worldwide. Data on patients admitted to the ICU with viral SARI, like those regarding mechanical ventilation, and mortality rates beyond the commonly reported 90-day survival and mid to long term prognosis remain limited. Comparative data on mechanical ventilation across different viral infections are essential for a better understanding of disease progression and more targeted treatment strategies. Clinical data on patient characteristics could help identify risk factors associated with complicated clinical course and challenging ventilation scenarios. Additionally, data on the disease course of various viral pathogens are essential for optimizing treatment options, implementing targeted prevention and risk group vaccination and developing new antiviral and immune modulating therapies. Therefore, the aim of this study is to investigate patient characteristics, course of disease, ventilation parameters and (long term) mortality rates among those admitted to the ICU with acute infections caused by IV, RSV and HMPV or PIV.
Methods
Study Design
A retrospective observational cohort study was conducted at the Spaarne Gasthuis Haarlem/Hoofddorp, The Netherlands. The study included all patients aged 18 years and older who had been admitted to the ICU with SARI and detection of IV, RSV, HMPV, or PIV between January 1, 2017, and December 31, 2023. Hospital admission related to IV, RSV, HMPV or HPIV infection was defined as the detection of one of these viruses in a respiratory sample using PCR-based diagnostic tests (as described below) up to 2 days before or during admission. Patients were excluded if their ICU admission was unrelated to the respiratory infection. This study was approved by the Local Institutional Review Board of the Spaarne Gasthuis, the Netherlands (2024.0082) on October fourth, 2024.
At the Regional Public Health Laboratory Kennemerland, respiratory samples were analyzed using RT-PCR to detect IV, RSV, HMPV, or HPIV with a multiplex PCR (RespiFinder 2Smart/RF2S, PathoFinder B.V. Maastricht, The Netherlands). The RF2S kit detects 20 viral and 4 bacterial targets simultaneously. 19 Alternatively a point-of-care test at the emergency department was performed using GeneXpert (cartridge based nucleic acid amplification test) for IV and RSV or the ID NOW system (Abbott Rapid Diagnostic, Scarborough, ME, USA) for IV detection. Samples consisted of nasopharyngeal/oropharyngeal (NP/OP) swab, sputum, tracheal aspirates or bronchial alveolar lavage (BAL).
Data Collection
Data were retrieved manually from the patients’ medical records by two researchers (SB and RF). Collected data consisted of demographic information, comorbidities, clinical characteristics, ventilations parameters, treatment, date of admission and discharge and mortality. Mortality was categorized into ICU mortality and hospital mortality. Co-infection was defined as the detection of an additional viral, fungal, or bacterial pathogen in sputum, NP/OP swabs, tracheal aspirates, or BAL samples using Polymerase Chain Reaction (PCR) or culture-based techniques, in conjunction with the treating physician's assessment of the case as a co-infection rather than co-detection or colonization, and subsequent treatment of the patient for the identified pathogen. Immunosuppressant use was limited to systemic immunosuppressants. Antibiotic treatment included the use of selective decontamination of the digestive tract (SDD). Data on long term mortality were retrieved from the Dutch Central Bureau of Statistics (CBS).
Data Analysis
Patients with co-detections of the included viral groups were excluded from the analysis to avoid bias from the presence of two pathogens. There were no multiple ICU admissions related to any of the included pathogens per patient during the study period. Descriptive statistics were used to describe the incidence rate for the respective causative agent. Demographic and clinical parameters, outcomes, and additional test results are presented as categorical data with percentages, normally distributed continuous variables as mean (± SD), and non-normally distributed continuous variables as median (interquartile range [IQR]). Comparisons between groups were performed using chi-square for categorical variables, one-way ANOVA for normally distributed continuous variables or Kruskal-Wallis test for not normally distributed continuous variables. An overall p-value < 0.05 was considered statistically significant. A post hoc analysis was conducted to determine which of the four groups differed significantly. Pairwise comparisons were performed among all four groups, applying an adjusted significance level of 0.0083 to account for multiple testing across six pairwise comparisons. Linear regression was used to evaluate the association with ICU admission duration, while logistic regression was applied to assess predictors of NIV failure. Univariate analyses for candidate predictors were conducted with a significance threshold of 0.3. A liberal p-value threshold of 0.3 was used in the univariate analysis to avoid excluding potentially important variables from the multivariable model, in line with recommendations for exploratory analyses. Candidate predictors included baseline characteristics, comorbidities, and results from laboratory and microbiological analyses. The commonly accepted rule of at least 10 events per variable were used to minimize the risk for overfitting and false positive associations in logistic regression. Subsequently, a backward selection strategy was applied, iteratively removing the least significant variable (ie, the one with the highest p-value) until only variables with a p-value < 0.05 remained in the model. Statistical analyses were performed using SPSS version 26.
Results
During the study period 2500 to more than 5000 tests were performed, per year. While the amount of tests and percentage positive remained relatively consistent, there was a sharp decline in 2020 and 2021 due to the COVID-19 pandemic. The temporal trends are shown in Figure 1 by the total number of tests performed per year, the total number of hospital and ICU admissions. Differences in the number of tests can be explained by the introduction of a point-of-care PCR at the emergency department including the identification of IV, RSV and SARS-CoV-2 and another point-of-care PCR including IV and SARS-CoV-2. In Table 1 the percentages of positive tests, hospital and ICU admission are summarized. The introduced point-of-care test did not include HMPV or PIV; these were assessed using an additional full respiratory panel and are therefore tested less often.
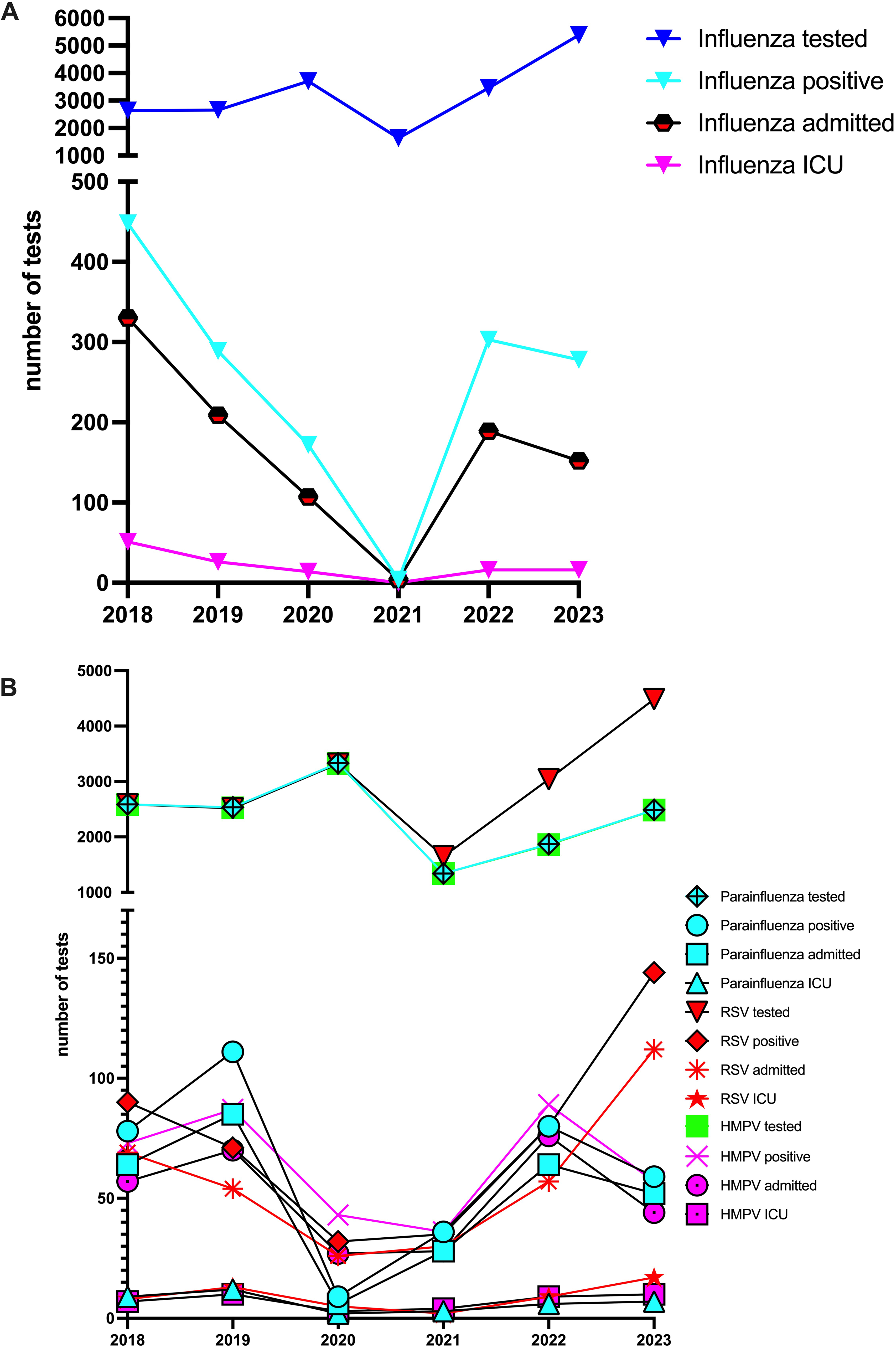
(A) Shows the total number of test performed per year, the total number of hospital admission and ICU admission for IV. (B) Shows the total number of test performed per year, the total number of hospital admission and ICU admission for HMPV, RSV and PIV. Abbreviations: Intensive care unit (ICU), Respiratory syncytial virus (RSV), Human metapneumovirus (HMPV).
Total Tests and Percentage Admissions Per Year.
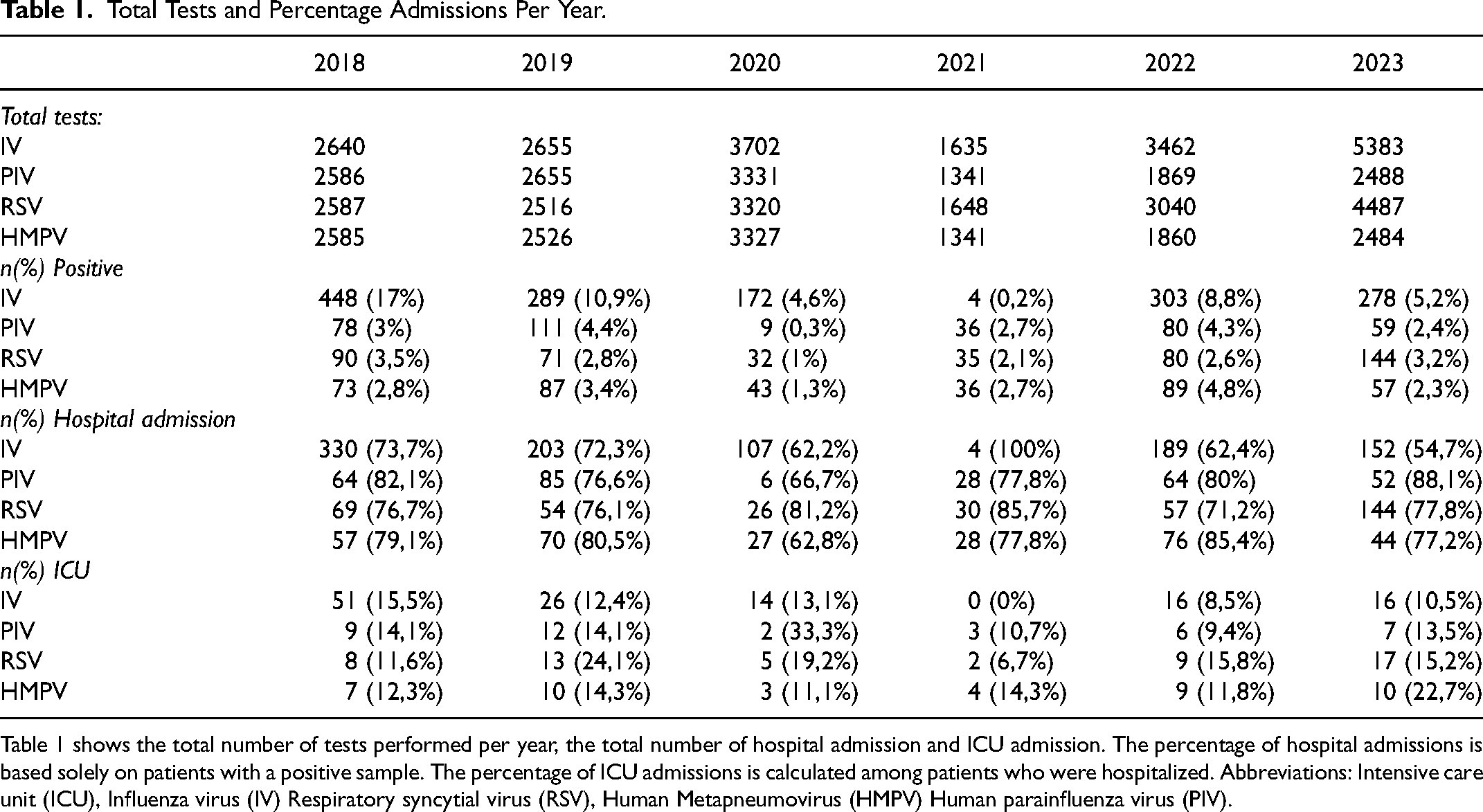
Table 1 shows the total number of tests performed per year, the total number of hospital admission and ICU admission. The percentage of hospital admissions is based solely on patients with a positive sample. The percentage of ICU admissions is calculated among patients who were hospitalized. Abbreviations: Intensive care unit (ICU), Influenza virus (IV) Respiratory syncytial virus (RSV), Human Metapneumovirus (HMPV) Human parainfluenza virus (PIV).
In total 277 patients were included in this study; RSV (n = 51), HMPV (n = 40), IV (n = 142), and PIV (n = 44). Viral co-detections were identified in 5 patients, involving combinations of RSV/HMPV, RSV/IV, HMPV/IV, and IV/PIV, which were excluded from the analysis. Flowchart describing the inclusion process is provided in Supplemental Appendix B. The samples consisted of NP/OP swab (225 (81.2%)), sputum (43 (15.5%)) and BAL (9 (3.2%)) The median age was 71 years, with no significant differences observed between the groups. Of the total population, 50.2% was female. A significant difference in the distribution of male and female patients was found among the virus groups (p = 0.026), concerning more women in the HMPV group compared to the influenza group(67.5% vs 42.3%, p = 0.005). The demographic characteristics including co-morbidities of all groups are shown in Table 1.
A total of 238 patients (85.9%) had underlying comorbidities, with cardiovascular (151(53.5%)), diabetes (74(26.2%)), and COPD (109(38.7%)) being the most prevalent. The distribution of these comorbidities did not differ significantly between the virus groups, except for hematological malignancies, which were more prevalent in patients with RSV (19.6%) than in the other groups (HMPV 2.5%, Influenza 3.5%, HPIV 4.6%)(p < 0.0001)). Although no statistically significant difference was observed, a trend was noted for a higher prevalence of solid malignancies, dialysis use, and immunosuppressant use in patients with RSV compared to those with other viruses. Table 2 shows all the comorbidities for each virus group.
Demographic Clinical Characteristics of the Different Viral Groups.
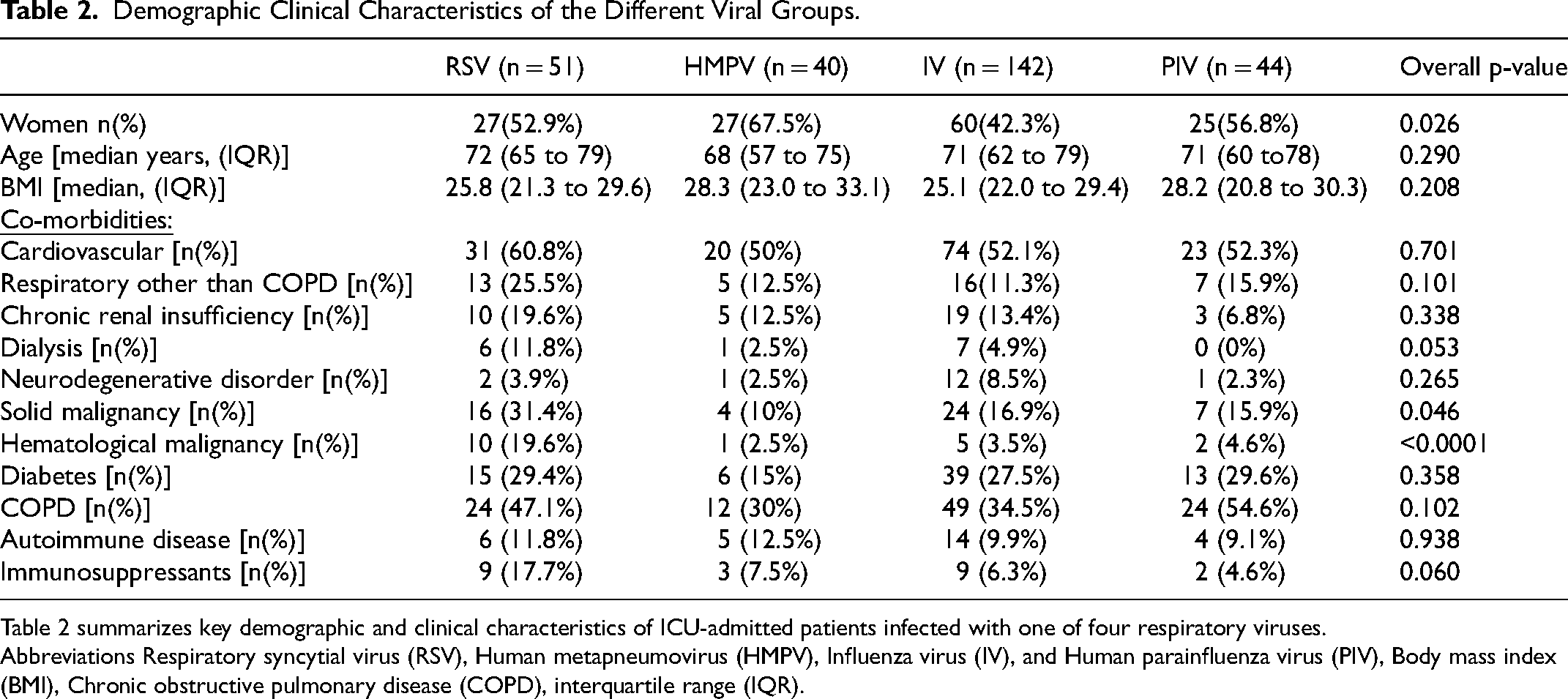
Table 2 summarizes key demographic and clinical characteristics of ICU-admitted patients infected with one of four respiratory viruses.
Abbreviations Respiratory syncytial virus (RSV), Human metapneumovirus (HMPV), Influenza virus (IV), and Human parainfluenza virus (PIV), Body mass index (BMI), Chronic obstructive pulmonary disease (COPD), interquartile range (IQR).
The duration of hospitalization did not differ among the four groups. The median length of hospital stay was 10(IQR: 6 to 17) days, while the median length of ICU stay was 4(IQR: 2 to 7) days. Overall ICU and hospital mortality did not vary significantly among the viruses, with an average ICU mortality ranging from 6.8% to 17.6%. All the data of duration of hospitalization and mortality are shown in Table 3.
Duration of Admission and Mortality for the Different Virus Groups.

Table 3 Shows the duration of admission, mortality and readmission stratified per viral pathogens. The one-year mortality percentages represent the patients who survived hospital admission. Overall mortality includes hospital and one year mortality.
Abbreviations: Respiratory syncytial virus (RSV), Influenza virus (IV), Human metapneumovirus (HMPV), Human parainfluenza virus (HPIV) Intensive care unit (ICU), interquartile range (IQR).
Multivariable regression analysis revealed that a lower arterial oxygen partial pressure (PaO₂) to the fraction of inspired oxygen (FiO₂) (P/F ratio) at admission (B = -0.14, 95% CI [-0.023, −0.005], p = 0.002), bacterial co-infections (B = 1.899, 95% CI [0.432, 3.374], p = 0.012), pleural effusions on chest x-ray (B = 2.381, 95% CI [0.551, 4.210], p = 0.011), invasive mechanical ventilation (B = 2.826, 95% CI [0.759, 4.894], p = 0.008), and non-invasive ventilation failure requiring intubation (B = 3.608, 95% CI [1.045, 6.171], p = 0.006) were associated with prolonged ICU admission. In contrast, age was negatively associated with ICU stay duration (B = -0.069, 95% CI [-0.121, −0.017], p = 0.010)
During ICU admission most patients (63.9%) received NIV support, while invasive mechanical ventilation (IMV) was required in 28.2% of cases, with no significant differences between virus groups. During NIV, the highest FiO₂ levels were observed in influenza patients, as compared to those with PIV (64.3 (23.3%) versus 50.6 (21.3%) p = 0.003). Notably, 40 patients (22.5%) who initially received NIV later required IMV, indicating progression in respiratory support needs. The median duration of NIV ranged between 1 and 2 days, while the median duration of IMV ranged from 4 to 5 days, with no significant differences observed between virus groups. Table 4 shows duration and type of ventilation for the different pathogens.
Ventilation Parameters for Different Viral Groups.
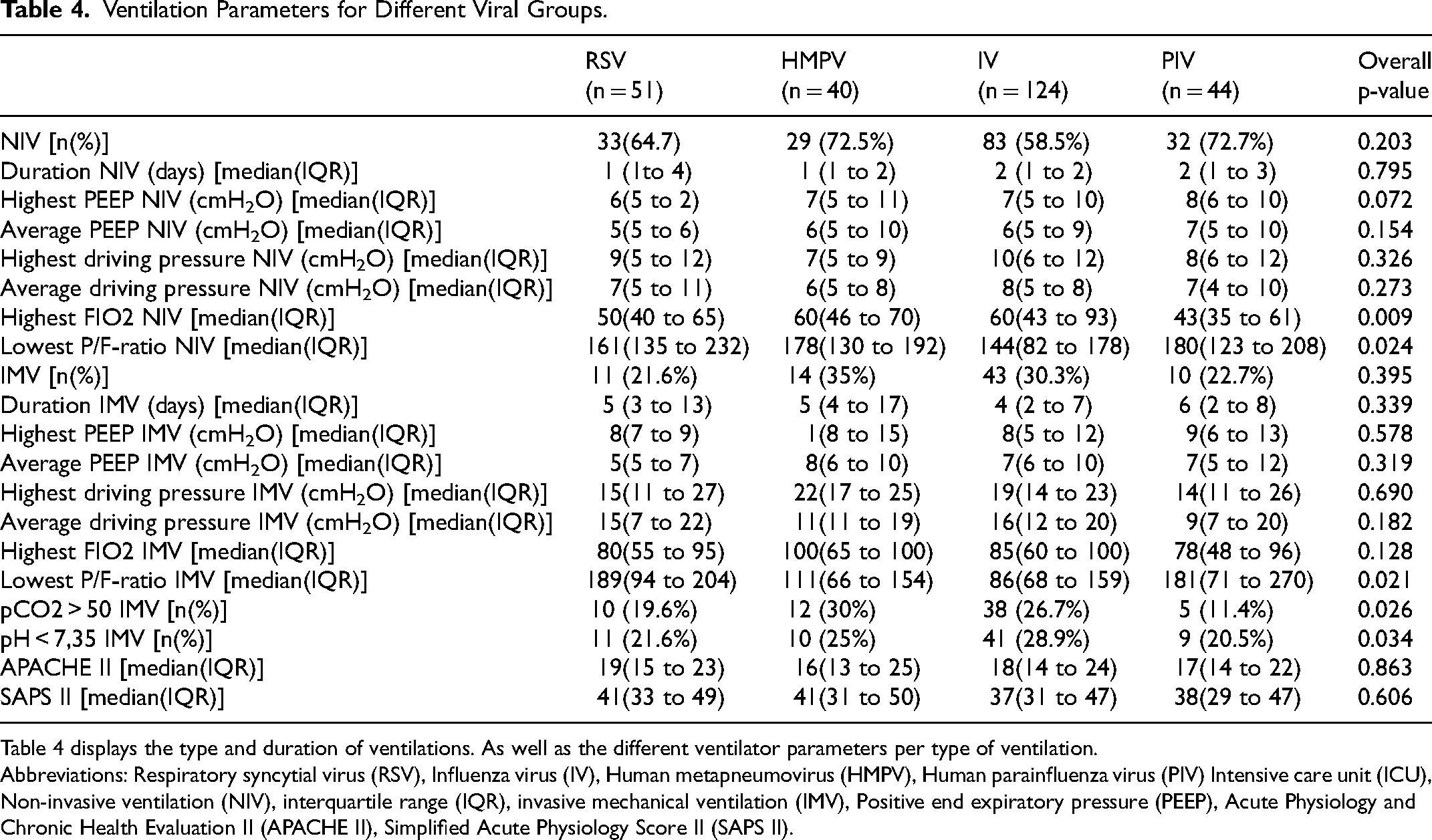
Table 4 displays the type and duration of ventilations. As well as the different ventilator parameters per type of ventilation.
Abbreviations: Respiratory syncytial virus (RSV), Influenza virus (IV), Human metapneumovirus (HMPV), Human parainfluenza virus (PIV) Intensive care unit (ICU), Non-invasive ventilation (NIV), interquartile range (IQR), invasive mechanical ventilation (IMV), Positive end expiratory pressure (PEEP), Acute Physiology and Chronic Health Evaluation II (APACHE II), Simplified Acute Physiology Score II (SAPS II).
Patients who failed NIV, ie, requiring intubation and IMV, were compared with those who succeeded on NIV. They required a significantly higher fraction of inspired oxygen (FIO₂) at admission (80%) compared to those who succeeded (50%; p = 0.002) Similarly, the ROX score at admission was significantly lower in NIV failures (p = 0.039), indicating a higher risk for needing intubation. The incidence of multilobar infiltrates also differed significantly between the groups (p = 0.025), with 45.0% of NIV failures showing multilobar infiltrates compared to 26.4% of NIV successes. Co-infections were significantly more frequent in NIV failures (72.5%) than in those who succeeded on NIV (35.7%; p < 0.0001). Additionally, NIV failures had a longer median ICU stay of 10 days (IQR = 9) compared to 7 days (IQR = 8) for IMV patients (p = 0.037). Finally, cardiovascular comorbidities were more present in NIV success (63.6%) than in NIV failure (40.0%) (p = 0.008). Although not statistically significant, a higher percentage of patients with HMPV infection appeared to be present in the NIV-failure group compared to the NIV-success group. A logistic regression analysis of the entire population revealed that bacterial co-infection (OR = 3.935, 95% CI [1.794, 8.632], p = 0.001), lower pH (OR = 0.007, 95% CI [0.809, 0.987], p < 0.0001) and a lower ROX score (OR = 0.893, 95% CI [0.809, 0.987], p = 0.026) were associated with NIV-failure. All the differences in patients with NIV-success or failure are shown in Table 5.
Differences in Parameters Between NIV-Success and NIV-Failure.
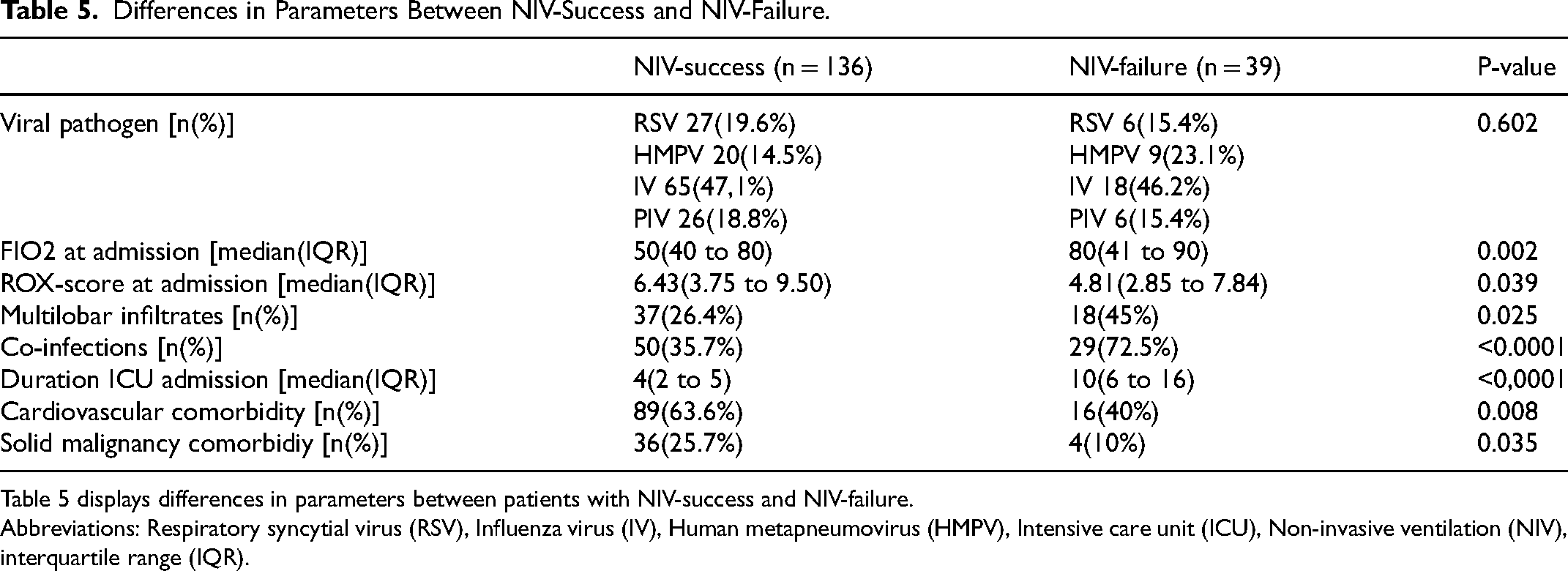
Table 5 displays differences in parameters between patients with NIV-success and NIV-failure.
Abbreviations: Respiratory syncytial virus (RSV), Influenza virus (IV), Human metapneumovirus (HMPV), Intensive care unit (ICU), Non-invasive ventilation (NIV), interquartile range (IQR).
Respiratory markers like pCO2 and pH did not significantly differ between virus groups after adjusting for multiple comparisons, suggesting a comparable respiratory impact across the groups. Additionally, all other ventilation data, including duration, peak pressures, mean driving pressures, positive end-expiratory pressure (PEEP) levels, and other relevant parameters for both NIV and IMV, showed no significant differences between the four groups infected with the respective viruses, indicating similar respiratory support requirements for these infections. All ventilation parameters are shown in Table 4.
Steroid treatment was administered to more than half of the patients in all virus groups. There was a significant difference in the amount of steroid use between the IV and PIV groups (p = 0.005). However, there was no difference in the daily dose of steroids between the groups. Prednisone was the most frequently administered steroid, with an average dose of 39.3 mg. Antiviral treatment was administered significantly more often to patients with influenza than to those with RSV. Specifically, 52.11% of influenza patients received antiviral therapy, compared to 13.73% of RSV patients. This reflects the more routinely use of oseltamivir in all critically ill influenza patients. Almost all patients (89.4%-97.5%) received antibiotic treatment. Reflecting the protocolized use of SDD in these patients. Data on steroid use is shown in Table 6.
Co-Infections and Treatment.

Table 6 Displays the amount of secondary and the most common secondary infection for the virus groups. Furthermore data on steroid use and the average daily dose of steroid.
Abbreviations: Respiratory syncytial virus (RSV), Human metapneumovirus (HMPV), Influenza virus (IV), Human parainfluenza virus (PIV)
ICU-specific complications, such as bacterial or fungal co-infections were noted, but their incidence did not differ significantly between the virus groups. Notably, in each virus group, 40% or more of the patients experienced a co-infection. The most common co-infections were those with Streptococcus pneumoniae (10.85%), Haemophilus influenzae (7.75%), Pseudomonas aeruginosa (4.65%), and Staphylococcus aureus (4.65%). Patients with a co-infection had significantly more often infiltrates on x-ray (p = 0.003) and a significantly longer ICU stay compared to those without a co-infection (3 (IQR: 2 to 5) versus 5.5 (IQR: 3 to 10), p < 0.0001). Overall hospital stay duration and mortality showed little difference between these groups. Table 6 shows the bacterial co-infections per group.
To investigate whether specific pathogens are more prevalent in high-risk populations and whether ICU admission due to such infections could indicate frailty, we analyzed one-year mortality in these groups. The one-year mortality rate following admission was substantial. Among patients who survived hospital admission, the following numbers of deaths occurred within one year: RSV (7 patients, 18.4%), HMPV (6 patients, 17.6%), IV (14 patients, 13.1%), and PIV (4 patients, 11.4%). There were no significant differences between the virus groups (p = 0.757). The data on one year mortality are shown in Table 3. Overall mortality at one year, thus including hospital mortality was 39.2% for RSV, 30% for HMPV, 34.5% for IV and 29.5% for PIV.
Discussion
The recent COVID-19 pandemic highlighted the potentially disastrous trajectory and healthcare burden associated with viral CAPs. Advancing pathogen-specific management strategies is crucial for effectively addressing surges in cases and mitigating the peak strain on healthcare facilities. This study analyzed the clinical data and outcomes of 277 ICU-admitted patients infected with IV, RSV, HMPV, or PIV between January 1, 2017, and December 31, 2023. Notably, this is among the first studies to compare ICU-admitted patients across these viral pathogens with an in-depth analysis of all included patients.
Four principal findings of this study should be noted. First, although mortality did not differ between the groups, this cohort demonstrated that short and long term mortality remains substantial among patients admitted to the ICU for viral CAP. Second, the duration of ventilation and other ventilation parameters did not differ among the four groups infected with the respective viruses; however, certain risk factors for NIV-failure were identified and proved to be associated with prolonged duration of admission. Third, bacterial co-infections are common in patients with SARI, complicate treatment and duration of admission. Finally, certain comorbidities were more prevalent in patients with specific pathogens, suggesting that these may serve as risk factors for a more complicated disease course.
The considerable ICU and hospital mortality rates found in this study, ranging for ICU from 6.8% to 17.6% and for hospital from 15% to 25.5%, are consistent with the existing literature.20–25 Although reported mortality rates vary, our relatively high rate can be attributed to the population of relatively high percentages of critically ill ICU patients in this cohort. This observation suggests that once patients are admitted to the ICU with severe illness, the virus involved may exert less influence on the clinical trajectory and outcomes than factors like baseline health status and comorbidities. Furthermore, the absence of significant differences in the Acute Physiology and Chronic Health Evaluation II (APACHE II) score and Simplified Acute Physiology Score (SAPS II) among the virus groups reinforces this idea, as these scores reflect a comparable severity of illness and respiratory function at the time of ICU admission, irrespective of the viral infection involved. While these scoring systems are routinely utilized for prognostic assessment in critical care, their consistent values imply that the initial severity of disease is likely shaped more by individual patient characteristics and pre-existing health conditions than by the virus infection involved. Additionally, the one-year mortality of these patients was notable. More than 10% of patients who survived ICU and hospital admission died within one year post-admission, with no significant differences observed between the pathogens. In addition to the already high mortality during admission leading to 1-year mortality rates of 29.5–39.2%, highlighting the severe disease burden and the fragility of patients following ICU admission
The previous hypothesis that the virus involved may exert less influence on the clinical trajectory and outcomes than factors like baseline health status and comorbidities, is supported by our second key finding: the type and duration of respiratory support did not differ between the groups infected with the respective viruses. This suggests that once NIV or IMV is required, the course of disease and duration of ventilation are not influenced by virus specific factors. The ventilation parameters, such as type and duration of ventilation, found in this study are consistent with the existing literature.23,26 Additionally, our study found that NIV-failure patients had higher oxygen demand and lower ROX-scores at admission as an identifier for NIV-failure. Furthermore, infiltrates on chest x-ray and co-infections were associated with NIV-failure. This association has been reported earlier.27,28 Currently, the only definitive indications for NIV are acute exacerbations of COPD and asthma cardiale; therefore, cardiovascular disease was more common in the NIV success group. Patients with pneumonia and infiltrates on chest x-ray may benefit less from NIV than those without these conditions. Moreover, delaying intubation in these patients may be harmful, potentially leading to a more complicated disease course and prolonged duration of IMV compared to NIV patients who undergo immediate intubation. Although not statistically significant, a higher percentage of patients with HMPV appeared in the NIV-failure group compared to the NIV-success group. This observation suggests that HMPV infection may be associated with an increased risk of NIV failure. To our knowledge, this association has not been reported in the existing literature. Although Given the potential clinical implications, further prospective research is essential to investigate this relationship in greater detail.
A substantial number of bacterial co-infections were found in this cohort. Most frequently found co-pathogens were S. pneumoniae, H. influenzae, P. aeruginosa and S. aureus. These are commonly associated with secondary bacterial infections but may also have colonized the respiratory tract without causing major signs of infection, 29 which is common in patients with chronic pre-existing respiratory disease. The existing literature does not agree on the prevalence of bacterial co-infections in SARI patients with these pathogens. Our study reports a higher incidence of co-infections compared to previous research.30–33 This underscores the challenge faced by treating physicians in distinguishing co-infection from co-detection or colonization. Viral infections are known to promote pre-existing bacterial infections or bacterial superinfections, 34 and patients with co-infections are at higher risk of NIV failure as well as prolonged admission and ventilation durations. However, unnecessary antibiotic treatment can contribute to increased antibiotic resistance and the emergence of infections with multidrug-resistant bacteria. It is important to note that nearly all patients in this cohort received prophylactic use of SDD, consisting of ceftriaxone, colistin, tobramycin, and amphotericin.
A higher prevalence of hematological malignancies was observed in patients with RSV compared to those with the other viral pathogens. Additionally, there appears to be a trend for a higher prevalence of solid malignancies and other co-morbidities in this group. Importantly, testing policies did not differ between patient groups in our clinical setting; most of the patients were tested with a broad respiratory panel, which is not limited to high-risk patients or more severe cases. This finding aligns with existing literature, which indicates that RSV tends to be more aggressive and severe in individuals with compromised immune systems or underlying hematological disorders.35–37 These results underscore the importance of identifying high-risk patients who may benefit from vaccines or prompt antiviral therapies, such as oseltamivir or baloxavir, available for influenza. Furthermore, it is important to highlight this patient population, which may benefit from recently approved vaccines against RSV infection. These insights are crucial for adapting national vaccination strategies. A limitation of our study is the absence of influenza vaccination data, as this was not routinely recorded in local electronic health records. Additionally, the Netherlands lacks a centralized vaccination registry, making it difficult to retrospectively verify vaccination status. RSV vaccination is not yet implemented nationally, limiting any assessment of its potential impact. With the expected rollout of RSV vaccination, establishing comprehensive and accessible vaccination registries will be crucial for evaluating vaccine effectiveness and guiding public health policy.
For the interpretation of the data, it is important to consider the strengths and limitations of this study. The robust sample size of 277 patients, combined with the long study period of over six years, significantly strengthens the validity of our findings in the ICU setting involved. The comprehensive testing approach, which included a broad panel of 20 respiratory viruses and 4 atypical bacteria all tested early after hospital admission, reduces the likelihood of missing other pathogens that could contribute to SARI. These factors suggest that our results are likely generalizable to similar ICU settings and high-risk patient populations. Furthermore, a strength of this study is, despite its sample size, its in-depth analysis of patient files, rather than relying on automatic data extraction. As a result, the extracted data can be interpreted on a case-by-case basis. However, there are some limitations. First, as a retrospective study, it is inherently subject to biases, such as the potential misclassification of data and missing values in electronic health records. Even though we took steps to ensure data accuracy by performing in-depth case analysis, the retrospective design limits our ability to draw definitive causal relationships between variables. Second, the study was conducted in the ICUs of Spaarne Gasthuis, a large peripheral teaching hospital, which may limit the generalizability of the results to other healthcare settings with different clinical practices or patient populations. This focus on a single hospital means that the findings may not fully reflect the diversity of treatment approaches used in ICUs worldwide. Another limitation is the risk of selection bias, as we included only patients with a positive test result for one of the selected viruses. This approach does not necessarily confirm that the detected virus is the primary cause of the patient's condition, as it can be difficult to determine whether the identified pathogen represents an incidental finding rather than the actual cause of illness.. Finally, our focus on respiratory symptoms when testing for viral infections may have led to missed cases with extrapulmonary manifestations. For instance, if a virus like RSV presents with cardiac or neurological symptoms, or arrhythmias,38,39 Such cases may not have been tested for RSV and were therefore potentially overlooked. This selective testing approach could have led to an underestimation of the true impact and clinical burden of these viral infections, including their potential to cause complications beyond the respiratory system.In conclusion, this study provides a detailed analysis of patients admitted to the ICU due to SARI caused by IV, RSV, HMPV, or PIV. Four key findings were found. A high mortality rate was observed, which did not differ between the groups. Furthermore, no differences were found in mechanical ventilation parameters, although some risk factors for NIV failure were found and NIV-failure was associated with a prolonged duration of ventilation. Finally, risk factors and co-morbidities for a complicated disease course were identified. The results underline the need for clinical awareness and diagnostic testing algorithms to optimize treatment strategies. A prospective study will be necessary to ensure comprehensive testing of all patients and to fully investigate risk factors for disease progression, which would underscore the need for development and use of new vaccines and treatment strategies.
Supplemental Material
sj-docx-1-jic-10.1177_08850666251375938 - Supplemental material for Is Clinical Outcome Pathogen Related? Characteristics and Outcomes of ICU Patients with Severe Acute Respiratory Infections: Focusing on Respiratory Syncytial Virus, Human Metapneumovirus, Influenza Virus, and Parainfluenza Virus
Supplemental material, sj-docx-1-jic-10.1177_08850666251375938 for Is Clinical Outcome Pathogen Related? Characteristics and Outcomes of ICU Patients with Severe Acute Respiratory Infections: Focusing on Respiratory Syncytial Virus, Human Metapneumovirus, Influenza Virus, and Parainfluenza Virus by S. van der Bie, MD, R.C. Fluit, MD, T. Neijzen, BSC, S.M. Euser, PhD, E.C.M van Gorp, MD, PhD, J. Kalpoe, MD PhD, D. Souverein, PhD, D. Snijders, MD, PhD, J. Du Toit, MD, A.D.M.E. Osterhaus, DvM, PhD, D. Gommers, MD, PhD, S.F.L. van Lelyveld, MD, PhD, and M. Goeijenbier, MD, PhD in Journal of Intensive Care Medicine
Footnotes
Author Contributions
SVB, JK, DS, SE, DS, SVL & MG were responsible for study design.
SVB, TN, RF, DS, SE were responsible for data management.
SVB, TN, SE, DS, SVL, DS, JK & MG for first analysis.
EVG, AO, DG feedback and suggestions for analysis.
All authors contributed to manuscript drafting.
All authors have read and approved the final manuscript.
MG is the guarantor, had final responsibility for the decision to submit for publication an attests that all listed authors meet authorship criteria.
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Studies involving human participants were conducted in accordance with institutional guidelines and the declaration of Helsinki. This study was approved by the Local Institutional Review Board of the Spaarne Gasthuis, the Netherlands (2024.0082) on Oktober fourth, 2024.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was independently conducted by the authors. Funding for the study was provided by Moderna, Inc. The sponsor had no role in the design, execution, or interpretation of the data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
