Abstract
Background
Elevated renin has been shown to predict poor response to standard vasoactive therapies and is associated with poor outcomes in adults. Similarly, elevated renin was associated with mortality in children with septic shock. Renin concentration profiles after pediatric cardiac surgery are unknown. The purpose of this study was to characterize renin kinetics after pediatric cardiac surgery.
Methods
Single-center retrospective study of infants who underwent cardiac surgery with cardiopulmonary bypass (CPB) utilizing serum samples obtained in the perioperative period to measure plasma renin concentrations (pg/mL). Time points included pre-bypass and 1, 4, and 24 h after initiation of CPB.
Results
Fifty patients (65% male) with a median age 5 months (interquartile range (IQR) 3.5, 6.5) were included. Renin concentrations peaked 4 h after CPB. There was a significant difference in preoperative and 4 h post-CPB renin concentration (4 h post-CPB vs preoperative: mean difference 100.6, 95% confidence interval (CI) 48.9-152.4, P < .001). Median renin concentration at 24 h after CPB was lower than the preoperative baseline.
Conclusions
We describe renin kinetics in infants after CPB. Future studies based on these data can now be performed to evaluate the associations of elevated renin concentrations with adverse outcomes.
Introduction
Vasoplegia and acute kidney injury (AKI) are potential complications after congenital heart surgery (CHS) with the use of cardiopulmonary bypass (CPB) and are associated with increased risk of morbidity and mortality.1,2 CPB is known to cause end organ dysfunction due to systemic hypoperfusion, reperfusion injury, and propagation of a systemic inflammatory response. 3 Another potential consequence of CPB is dysregulation of the Renin-Angiotensin-Aldosterone System (RAAS), a major contributor in the progression of vasoplegia in sepsis, cardiogenic shock, and other pro-inflammatory conditions.4-8 Disruption of RAAS homeostasis has been implicated in inflammation and distributive shock (including sepsis and post-CPB vasoplegia).9-11 Renin, an enzyme within RAAS, has been identified as a biomarker to predict significant morbidity and mortality in the adult CHS population.12,13 Serum renin concentrations in adults after CPB triple shortly after surgery and remain elevated in those who develop significant vasoplegia. 12
In pediatric patients, elevated renin concentrations have been shown to be predictive of severe persistent AKI, vasoplegia, and mortality in septic shock. 11 These findings highlight the important role of RAAS dysregulation in the propagation of a pro-inflammatory state. The kinetics of renin concentrations in the pediatric CHS population has not been defined. Steroids (dexamethasone, methylprednisolone) have been shown to reduce pro-inflammatory cytokines after CPB. 3 Other management strategies for postoperative vasoplegia include fluid resuscitation and traditional vasoactive medications. The vasoactive inotrope score (VIS) was also developed to provide a standardized form in which all vasoactive drugs are normalized to a whole number and summated to quantify the degree of vasoactive support.14,15 Newer agents, such as exogenous angiotensin II (AT-II), are not integrated into the VIS. An improved understanding of RAAS dysregulation after CHS could improve our knowledge of vasoplegia and use for targeted AT-II therapy in specific sub-phenotypes of children with elevated renin concentrations.16-23 To that end, the purpose of this study was to describe the kinetics of renin in the first 24 h after CPB.
Methods
This was a single-center retrospective study entitled “Renin Levels as a Predictor of Post-Operative Vasoplegia and Acute Kidney Injury in Patients with Congenital Heart Disease Undergoing Cardiac Surgery Utilizing Cardiopulmonary Bypass” which included analysis of samples and clinical data collection was reviewed and approved by the Cincinnati Children's Hospital Medical Center (CCHMC) Institutional Review Board (approved April 07, 2022, IRB#2011-2856) with a waiver of the need for additional informed consent. The study was performed in accordance with the ethical standards of the committee and with the Helsinki Declaration of 1975. The study was conducted from 2019 to 2022 using stored serum samples obtained from the CCHMC Heart Institute Biorepository. Infants whose parent or legal guardian did not consent to the biorepository and those undergoing cardiac surgery without CPB were excluded. Informed consent was obtained for biorepository sample collection. Study sample size was determined based on biorepository sample availability and resource allocation.
Patient demographic and clinical data were collected through the CCHMC Heart Institute Data Repository. Inclusion criteria for this study: (1) infants ages 1 to 12 months; (2) underwent cardiac surgery utilizing CPB; (3) enrolled into the CCHMC Heart Institute Biorepository with serum sample availability. Race and ethnicity data was obtained from the electronic health record and were assessed to characterize the CHS population at the institution. This study followed the use of the STROBE guideline.
The primary outcome was renin kinetics. Secondary outcomes included peak vasopressin dose and AKI incidence in the first 72 postoperative hours. Direct renin concentrations (pg/mL) were measured on serum samples at four time points: pre-bypass, 1, 4, and 24 h after initiation of CPB. Pre-bypass sample measurement was obtained within 48 h of surgery. Quantification of renin was performed in the CCHMC Clinical Nephrology Laboratory utilizing the DiazymeTM bioassay (Diazyme Laboratories, Inc., Poway, CA, USA). Vasoplegia was defined by peak vasopressin dose greater than 1 U/kg/min within the first 72 postoperative hours. Titration of vasopressin was determined at the discretion of the clinician. VIS was calculated as previously defined. 14 AKI was defined using the Kidney Disease Improving Global outcomes (KDIGO) serum creatinine and urine output criteria. Stage 1 AKI was defined as urine output (<0.5 mL/kg/hr for 6-12 h by rolling average) or serum creatinine criteria (1.5-1.9× greater than baseline or ≥0.3 mg/dL).24,25 Stage 2 AKI was defined as urine output <0.5 mL/kg/hr ≥12 h or 2.0-2.9× greater than baseline for serum creatinine. Stage 3 AKI was defined as urine output <0.5 mL/kg/hr ≥24 h (or anuria for ≥12 h) or 3.0× greater than baseline serum creatinine, increase ≥4.0 mg/dL, or requiring renal replacement therapy. Severe AKI was defined as patients with KDIGO stage 2 or 3. Pre-, intra-, and postoperative variables were extracted from the Pediatric Cardiac Critical Care Consortium (PC 4 ) database, a large multi-center data registry, and the local electronic health record. Surgical complexity was defined according to the Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery (STS-EACTS) Congenital Heart Surgery Mortality Category (STAT) score. 26
Statistical Analysis
Continuous variables were described using medians (interquartile ranges [IQR]) and categorical variables using frequencies (percent of total). Linear mixed-effects regression was used to describe the renin kinetics. Model covariates/fixed effects included categorical time (preoperative, 1-h post-CPB, 4 h post-CPB, 24 h post-CPB), age (days), gender, and CPB time (minutes). Subjects were modeled using a random effect term to account for the nesting of repeated measures within patients. The mean differences for renin concentrations according to the model covariates were obtained from the model parameters using the Satterthwaite approximation to the model degrees of freedom.
The logistic regression model evaluated the associations of renin concentrations with the presence of AKI (Stage 2 or higher). Ordinal regression model was used to fit the peak vasopressin dose. The ordinal regression model was used as it has a great robustness advantage for skewed continuous outcome variables while preserving power. 27 The model covariates included the renin concentrations at 4 h and CPB time. The results were reported as odds ratios (OR) with 95% confidence intervals (CI). For continuous variables, all reported OR were rescaled to reflect an increase from the 25th percentile to the 75th percentile (ie, interquartile OR). For peak vasopressin dose estimated via the ordinal regression model, an OR greater than one indicated a factor was associated with a higher peak vasopressin dose. In all analyses, P-values <.05 were considered statistically significant. All statistical analyses were performed using R version 4.3.1 statistical software (https://www.r-project.org/). Mixed-effects models were fitted using lmer function in the lme4 package (version 1.1-35.1). 28 Logistic regression models were fit using the lrm function in the rms package (version 6.7.1). 29
Results
Fifty patients were included in this study. A summary of demographics and clinical characteristics are included in Table 1, Supplemental Tables 1 and 2. Median age at surgery was 5 months (3.5, 6.5); patient sex was noted to be predominantly male (64%). Nearly all patients were planned surgeries (98%) with a near equal distribution of STAT categories except for STAT 1 and STAT 5 (18% and 6%, respectively). Median peak VIS was 6.5 (4.75, 8). Median duration of mechanical ventilation was 2 days (1.0, 2.5). Severe (Stage 2 + 3) AKI was present in 14% of patients. Overall, 40% of patients met criteria for Stage 1 AKI by serum Cr and 4% by urine output.
Patient Postoperative Characteristics and Outcomes.
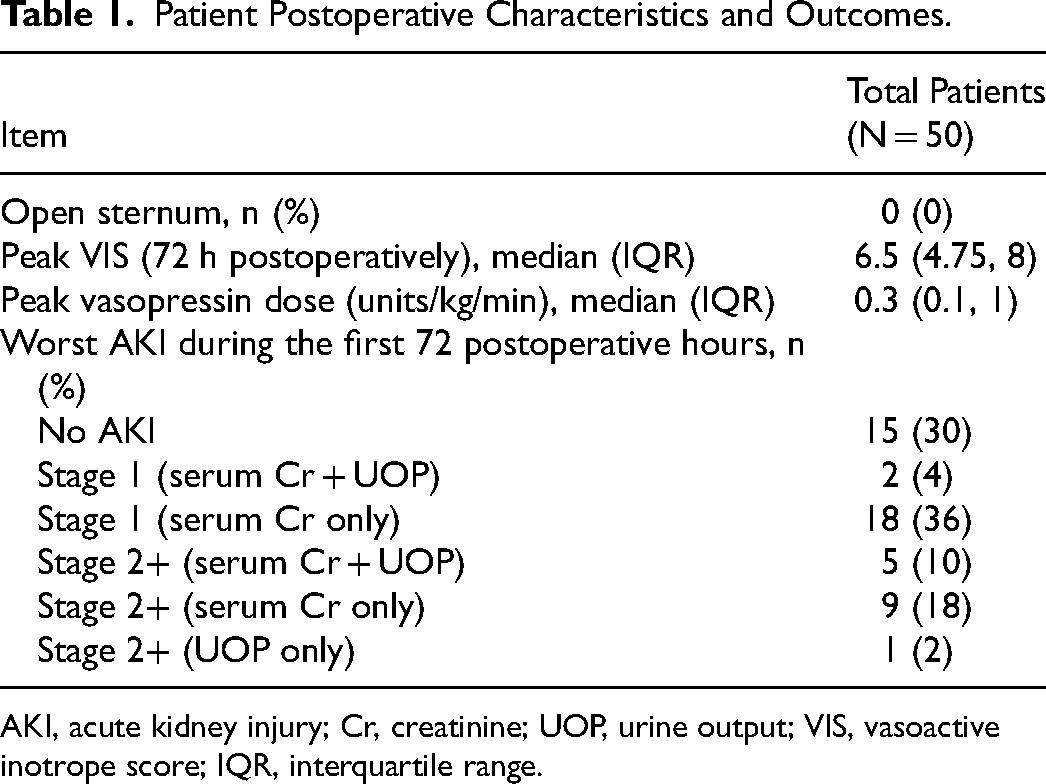
AKI, acute kidney injury; Cr, creatinine; UOP, urine output; VIS, vasoactive inotrope score; IQR, interquartile range.
The mean (95% CI) renin concentrations at each of the measured time points in the mixed model are summarized in Figure 1. Mean renin concentration peaked at 4 h after CPB. Mean renin concentration at 4 h was higher than the preoperative baseline (mean difference 100.6, 95% CI 48.88, 152.35, P ≤ .001). Nadir renin concentration was at 24-h after CPB and was lower than the preoperative baseline (mean difference −104.31, 95% CI −156.05 to −52.58, P ≤ .001) (Table 2).
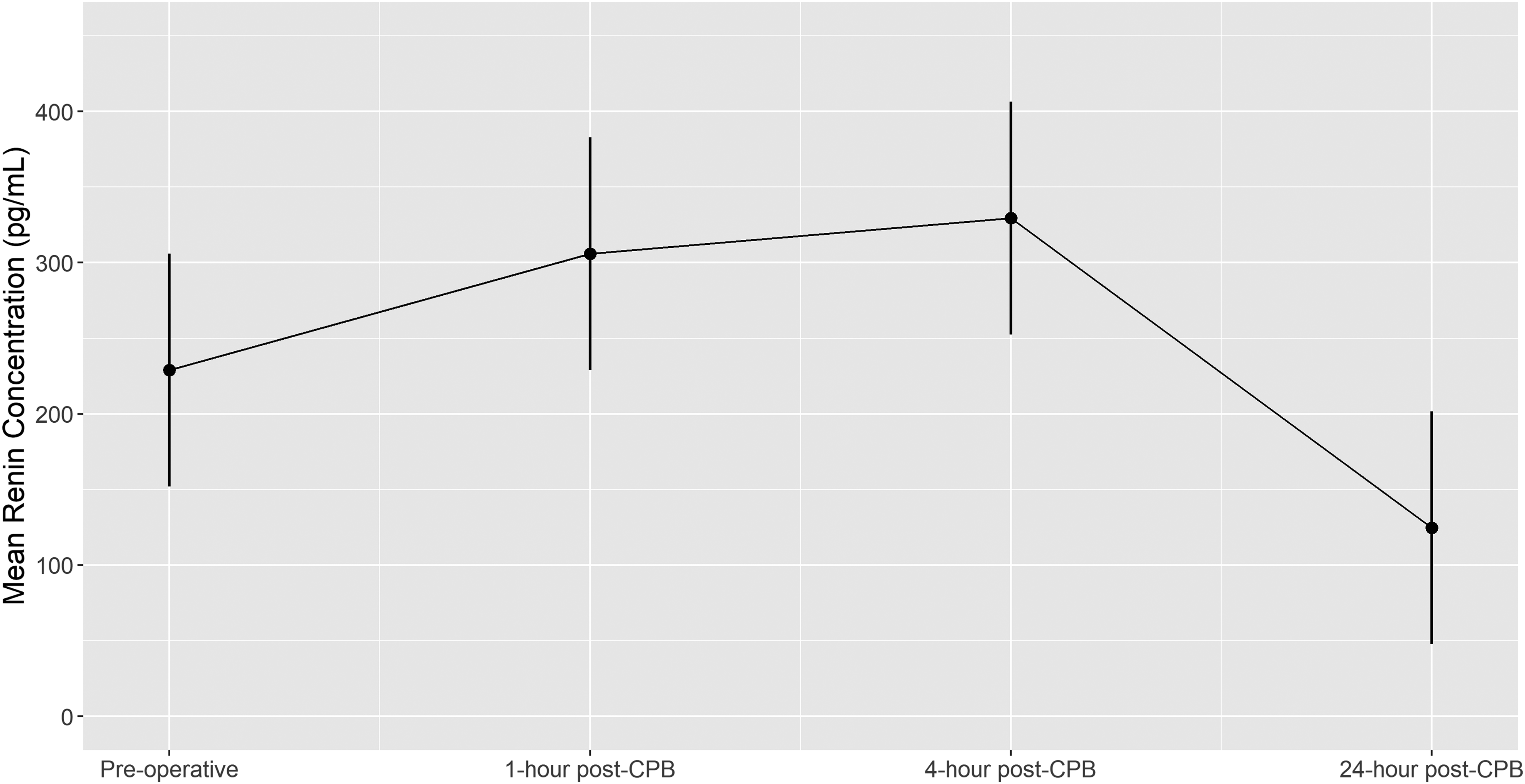
The predicted mean renin concentration trajectory over time (preoperative, 1-h post-CPB, 4 h post-CPB, and 24 h post-CPB) from the mixed model with random subject-specific intercepts, adjusted for age (days), gender (male vs female), and CPB time (mins) at the most frequent or median level. Error bars denote 95% confidence intervals. CPB, cardiopulmonary bypass.
Effects of Time, Age, CPB Time, and Sex on Serum Renin Concentration Using Linear Mixed-effects Model.
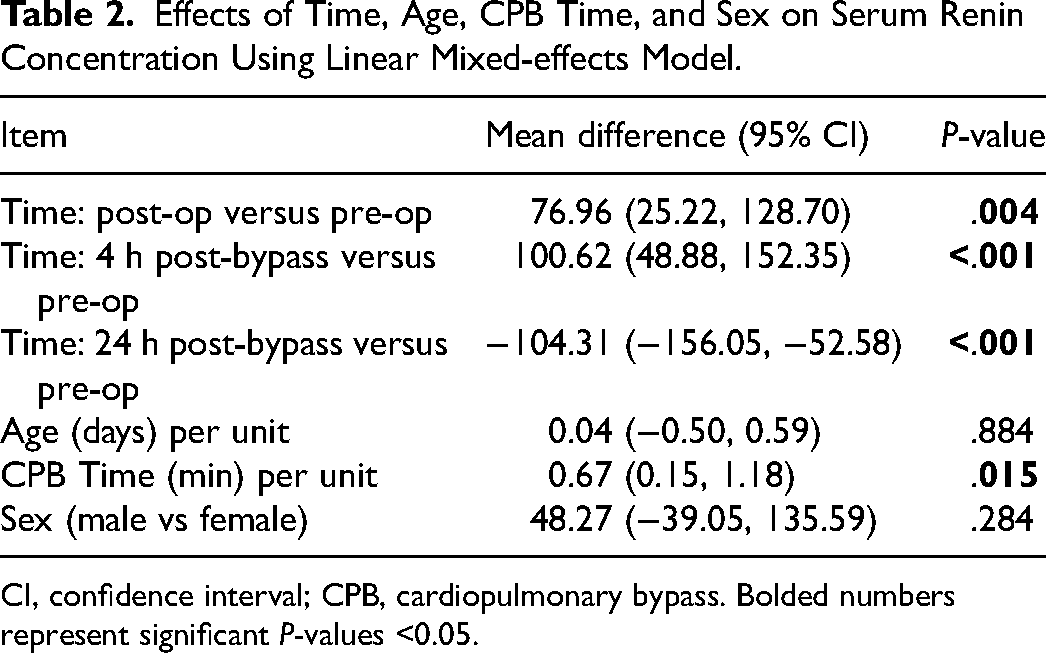
CI, confidence interval; CPB, cardiopulmonary bypass. Bolded numbers represent significant P-values <0.05.
Comparisons of renin concentrations with peak vasopressin dose as a continuous variable and the presence of AKI are summarized in Table 3. The multivariable regression models did not demonstrate associations between renin concentrations and peak vasopressin dose or AKI. Longer CPB duration was associated with a higher peak vasopressin dose (interquartile OR 3.58, 95% CI 1.53-8.37, P = .003). Figure 2 represents predicted outcomes as a function of renin concentration and CPB time from the regression models.
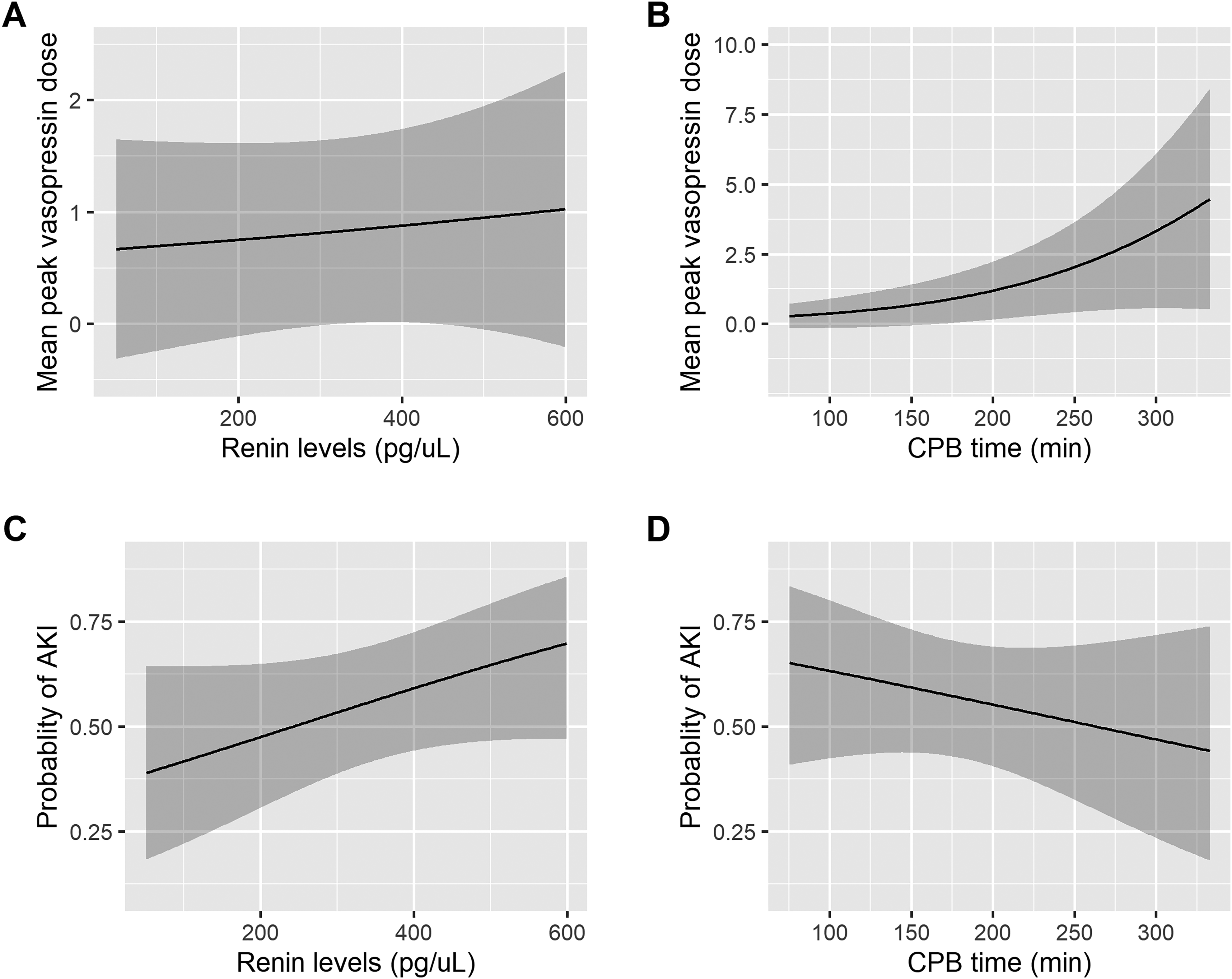
Predicted outcomes of mean vasopressin dose and severe AKI as a function of renin concentration (P = .637, P = .121) and CPB time (P = .003, P = .388) from the ordinal regression and logistic regression models. Shaded areas denote 95% confidence intervals. CPB, cardiopulmonary bypass; AKI, acute kidney injury.
Association Between Peak Vasopressin Dose and AKI.

Odds ratio (OR) and 95% confidence intervals (CI) are obtained by ordinal or logistic regression. ORs for continuous predictors scaled to reflect the interquartile range odds ratio (ie, reference = 25th percentile, contrast = 75th percentile). CPB, cardiopulmonary bypass; AKI, acute kidney injury; Cr, creatinine; UOP, urine output. Bolded number represent significant P-value <0.05.
Discussion
In this study, we described the changes in renin concentrations after cardiac surgery using CPB in infants. First, during the first 24 h concentrations peaked at 4 h and dropped below the pre-bypass baseline at 24 h after cardiac surgery. Second, there was no association between renin concentration and peak vasopressin dose or AKI. Interestingly, there were several patients who had elevated renin concentrations pre-bypass.
RAAS dysregulation is purported to occur after CPB. This is in part due to consequences of CPB with the introduction of foreign materials and microplastics into the bloodstream, mechanical stress on cells, tissue reperfusion, administration of blood products, and dramatic shifts in blood pressure and body temperatures. 3 Additional factors have been shown to be associated with elevated renin concentrations include male gender, elevated B-type natriuretic peptide levels, and worsened left ventricular systolic ejection fraction. Renin concentrations have been associated with postoperative outcomes in adult cardiac patients including duration of vasopressor therapy, ICU length of stay, and hospital length of stay. We did not investigate these specific outcomes in our study given the heterogeneity of the patients and low prevalence of vasopressin use in our study cohort.
While our study did not demonstrate any association between renin concentration and postoperative outcomes, this has been observed in other pediatric cohorts and different study designs. The lack of association could be in part due to the relatively even distribution of surgical complexity with an overall small cohort size which would not capture the CHS population most at risk for development of significant postoperative morbidity and mortality. Another consideration is the AKI definition utilized in this study which did not identify any patient with Stage 3 or persistent AKI indicating that these were transient changes because of ultrafiltration. 30 Stanski and colleagues demonstrated the relationship between serum renin concentrations in pediatric septic shock patients and outcomes in the ICU. Elevated serum renin and prorenin concentrations predicted severe persistent AKI and 28-day mortality. 11 The data from these studies also demonstrated, when compared with adult septic shock cohorts, the degree of elevation in renin and prorenin concentrations were much higher. Similar studies have hypothesized the utility of elevated renin concentrations as a method to identify patients with RAAS dysregulation.9,11-13,31
Elevated renin concentrations pre-bypass can suggest RAAS dysregulation due to systemic inflammation. Preoperative endotoxicity and inflammation has been described as a poor prognostic indicator of vasoplegia after CHS. 32 Elevated preoperative renin concentrations may corroborate with the previously described phenomena and direct us to identifying patients at risk for significant morbidity and mortality undergoing cardiac surgery with CPB. Understanding the utility of serum renin concentrations in the CHS population is important with its potential impact on patient outcomes including the use of vasopressin and postoperative AKI.
It is important to acknowledge our study limitations. The overall cohort size is small and likely underpowered to detect associations with secondary outcomes. We limited the population to infants (1 to 12 months), which may make our findings not generalizable to neonates and older children. We also used vasopressin dosing as a surrogate for vasoplegia based on institutional practice of initiating it as a second- or third-line vasoactive agent which is clinician dependent but ultimately captures patient clinical severity given no patients were on vasopressin as monotherapy in this study. It is also important to acknowledge with respect to Diversity, Equity, and Inclusion that our study contained a predominantly white population which is congruent with our center's surgical population, but the data may not be applicable across all race categories.
Our study demonstrated changes in serum renin concentrations in infants undergoing CHS with CPB. We were able to demonstrate a trend of mean renin concentrations within a small cohort which suggests RAAS disruption after CPB occurs and is temporary with rapid resolution. This differs from the adult CHS population in which renin concentrations typically peak at 24 h and remain elevated through 72 h post-CPB. 12 This information is valuable as a first step in investigating the utility of renin concentrations in the pediatric CHS population and the potential in identifying those who may suffer from post-CPB vasoplegia. While we failed to find any association between renin concentration with AKI or peak vasopressin dosing, the limitations and size of our study give enough reason to believe that future work should include a higher proportion of postoperative vasoplegia patients to better evaluate pattern changes in renin concentration in this patient phenotype. Identification and treatment of postoperative vasoplegia is critical. While steroids have been demonstrated to ameliorate postoperative inflammation, the risks of adrenal suppression, infection, and hyperglycemia cannot be underestimated. 3 Identifying RAAS disruption would allow the use of targeted therapies and avoid the deleterious effects of polypharmacy to treat refractory vasoplegia. AT-II is one such drug that is being investigated as a potent vasoconstrictor in the setting of vasodilatory shock.16,20,22,31 The use of AT-II has been studied in both the adult and pediatric population in septic shock and vasoplegia syndrome after adult cardiac surgery.16-19,33-35 Optimal dosing and timing of AT-II administration in children is not well established. Additionally, its utility in the pediatric CHS population is also unknown. Understanding the typical changes in renin concentration after CHS is essential in understanding who will benefit from targeted therapy following cardiac surgery.
We described the perioperative profile of serum renin concentration among infants undergoing cardiac surgery. While small in sample, we lay the foundation for larger studies examining a heterogeneous cohort of children that are sufficiently powered to detect associations between renin levels and clinically important outcomes.
Supplemental Material
sj-docx-2-jic-10.1177_08850666241268655 - Supplemental material for Kinetics of Renin Concentrations in Infants Undergoing Congenital Cardiac Surgery
Supplemental material, sj-docx-2-jic-10.1177_08850666241268655 for Kinetics of Renin Concentrations in Infants Undergoing Congenital Cardiac Surgery by Michael E. Kim, Katja M. Gist, Katie Brandewie, Huaiyu Zang, David Lehenbauer, David S. Winlaw, David L.S. Morales, Jeffrey A. Alten, Stuart L. Goldstein and David S. Cooper in Journal of Intensive Care Medicine
Supplemental Material
sj-docx-3-jic-10.1177_08850666241268655 - Supplemental material for Kinetics of Renin Concentrations in Infants Undergoing Congenital Cardiac Surgery
Supplemental material, sj-docx-3-jic-10.1177_08850666241268655 for Kinetics of Renin Concentrations in Infants Undergoing Congenital Cardiac Surgery by Michael E. Kim, Katja M. Gist, Katie Brandewie, Huaiyu Zang, David Lehenbauer, David S. Winlaw, David L.S. Morales, Jeffrey A. Alten, Stuart L. Goldstein and David S. Cooper in Journal of Intensive Care Medicine
Footnotes
Acknowledgements
We would like to thank the Heart Institute Research Core at Cincinnati Children's Hospital Medical Center for supporting the study and its personnel.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article. Internal funding was provided by the Heart Institute Research Core (HIRC) for the entirety of the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
