Abstract
Purpose
We investigated the association of age, sex, race, and insurance status on antipsychotic medication use among intensive care unit (ICU) patients.
Materials and Methods
Retrospective study of adults admitted to ICUs at a tertiary academic center. Patient characteristics, hospital course, and medication (olanzapine, quetiapine, and haloperidol) data were collected. Logistic regression models evaluated the independent association of age, sex, race, and insurance status on the use of each antipsychotic, adjusting for prespecified covariates.
Results
Of 27,137 encounters identified, 6191 (22.8%) received antipsychotics. Age was significantly associated with the odds of receiving olanzapine (P < .001), quetiapine (P = .001), and haloperidol (P = .0046). Male sex and public insurance status were associated with increased odds of receiving antipsychotics olanzapine, quetiapine, and haloperidol (Male vs Female: OR 1.13, 95% CI [1.04, 1.24], P = .0005; OR 1.22, 95% CI [1.10, 1.34], P = .0001; OR 1.28, 95% CI [1.17, 1.40], P < .0001, respectively; public insurance vs private insurance: OR 1.32, 95% CI [1.20, 1.46], P < .0001; OR 1.21, 95% CI [1.09, 1.34], P = .0004; OR 1.15, 95% CI [1.04, 1.27], P = .0058, respectively). Black race was also associated with a decreased odds of receiving all antipsychotics (olanzapine (P = .0177), quetiapine (P = .004), haloperidol (P = .0041)).
Conclusions
Age, sex, race, and insurance status were associated with the use of all antipsychotic medications investigated, highlighting the importance of investigating the potential impact of these prescribing decisions on patient outcomes across diverse populations. Recognizing how nonmodifiable patient factors have the potential to influence prescribing practices may be considered an important factor toward optimizing medication regimens.
Introduction
Clinicians are increasingly recognizing how external factors can impact patient health across a range of exposures and illnesses.1,2 Numerous nonmedical and medical factors influence exposures and outcomes for patients prior to arrival at the hospital and following their interaction with healthcare teams. These factors can influence interactions between patients and their medical team, though the degree to which they impact care is less understood. 3 The American College of Physicians has labeled understanding and addressing social factors that affect health outcomes to be a “pressing issue.” 4 It is therefore imperative that providers continue to work toward understanding how physiologic, social, and environmental factors interact to influence patient exposures and outcomes.
Among critically ill adults, antipsychotic medications are frequently initiated in management of acute agitation or delirium. In addition to large studies demonstrating no benefit,5-7 their use is associated with prolonged delirium duration, inappropriate medication continuation, and increased mortality.6,8-11 Use and choice of antipsychotic medications is often variable with differing prescribing patterns across patient groups and institutions.6,12,13 Given the lack of guiding pharmacologic data surrounding the use of these medications, this allows for significant opportunities to introduce provider and patient bias into their use.
We aimed to investigate the independent association of nonmodifiable factors including patient age, sex, race, and insurance status at admission on exposure to antipsychotic medications in critically ill adults. Additionally, we sought to describe the duration of antipsychotic medication use across subgroups including continuation at hospital discharge.
Materials and Methods
Study Design and Participants
Following local institutional review board approval (no. 171671) we performed a retrospective review of the electronic health record (EHR) for adult (≥18 years) patients admitted to Vanderbilt University Medical Center surgical, trauma, medical, or cardiovascular intensive care units (ICUs) from January 1, 2014 to December 31, 2016. Patients with home antipsychotic prescriptions and those administered < 1 mg of haloperidol (as is common practice for nausea prophylaxis) were excluded. Current practices at our institution during study interval aligned with ABCDEF bundle 14 practices emphasizing nonpharmacologic practices (ie, reorientation, provision of eyeglasses and/or hearing aids, sleep hygiene, removal of catheters and restraints, and mobilization as able) for delirium prevention with medications administered only for symptom control of acute agitation or hyperactive delirium. The ABCDEF bundle is a multicomponent, evidence-based model to improve outcomes for critically ill patients by assessing and treating pain, both awakening and breathing trials, choice of appropriate sedation, delirium monitoring and management, early mobility and exercise, and family engagement.
We collected baseline patient demographic information, admission characteristics, patient severity of illness, and daily hospital course data.
Baseline patient characteristics collected on ICU admission included age, sex, race (Caucasian, Asian, Black, or unknown), body mass index (BMI), and insurance status (public, private, or unknown). Patients with missing demographic data were classified as “unknown” in the respective category.
The severity of illness was quantified using the University Health Consortium (UHC)- expected mortality index, a previously validated model using administrative data (diagnoses, comorbidities, and procedural information) to predict mortality among hospitalized patients as well as the presence of shock diagnosis. 15 Shock was defined as a lactate >2 mmol/L with concurrent norepinephrine administration. Hospital course data included daily sedation assessments (using Richmond Agitation-Sedation Scale [RASS]), 16 delirium assessments (using the Confusion Assessment Method for the ICU [CAM-ICU]), 17 requirement of mechanical ventilation (yes/no), hospital and ICU length of stay, initial admitting ICU location (surgical, trauma, medical, or cardiovascular), and in-hospital mortality.
Outcomes
Our primary outcome of interest included the receipt of an antipsychotic medication. We restricted antipsychotic medications to haloperidol, olanzapine, and quetiapine as these medications accounted for >97% of all antipsychotics utilized within the cohort. We obtained daily antipsychotic administration data for patients within the ICU and receipt (not just prescription) of medication qualified as antipsychotic medication exposure. Patients who received ≤ 1 mg haloperidol on any given day were not classified as an antipsychotic exposure as this is frequently administered for nausea within our institution. All haloperidol doses > 1 mg on any given day were considered antipsychotic exposure days. Discharge medication lists were collected for all hospital survivors with new antipsychotic prescriptions at hospital discharge classified as medication continuation. Patients with missing prescribing data were excluded from analysis.
Statistical Analysis
In the event of repeat ICU encounters, we included patient characteristics from only their initial ICU visit. We described patient and hospital factors and antipsychotic prescribing practices using median values and interquartile ranges (IQRs) for continuous variables and frequency (%) for categorical variables.
We performed 3 separate logistic regression analyses to evaluate the independent association of age (years), sex (male/female), race (Black/Asian/Indian/Unknown), and insurance status (public/private) on administration of each antipsychotic medication (olanzapine, quetiapine, haloperidol) – adjusting for UHC mortality, shock (yes/no), mechanical ventilation (yes/no), median RASS, presence of delirium (yes/no), ICU type (medical, surgical, trauma, cardiovascular), ICU length of stay (in days), and admission year (2014, 2015, or 2016). Nonlinear regression splines were used to model the effects of age on receipt of each antipsychotic medication outcome. Odds ratio and 95% confidence interval were reported, statistical significance for logistic regressions was assessed using Wald-type tests. For the effect of age (nonlinearly distributed), odds ratios comparing the 75th (69 years) with the 25th (43 years) centile of age, with 95% confidence intervals, were reported together with p values representing the overall statistical significance across the entire range of age for each outcome of interest.
We considered P-values < .05 as statistically significant. Due to the descriptive and exploratory nature of this study, no statistical power analyses were implemented a priori. We used R version 3.6.1 for all analyses.
This article adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Results
A total of 27,137 ICU patient encounters were included, containing 6191 (22.8%) encounters that were prescribed a new antipsychotic medication. The median age of the total cohort was 58 [43–69] years. The majority of patients were men (16,571; 61%), Caucasian (22,067; 81%), and had public health insurance (14,910; 55%). Patients were most frequently admitted to the medical and trauma ICU (8289; 31% and 8284; 31%, respectively) with a total in-hospital mortality of 8% (2127). In our cohort, 41% (11,188) of patients required mechanical ventilation and 18% (4935) had shock. Baseline patient characteristics by antipsychotic recipient status are described in Table 1.
Patient Characteristics Based on Receipt of Antipsychotic Medications.

“Nonrecipients” include patients who did not receive antipsychotic medications. “Recipients” include those patients who did receive antipsychotic medications. Variables are presented as median [interquartile range] or count (%), as appropriate. Abbreviations: ICU, intensive care unit; UHC-expected mortality, University Health Consortium–expected probability of mortality, LOS, length of stay.
Overall, a majority of those administered antipsychotics were male (3898; 63.0%), Caucasian (5177; 83.6%), or receiving public health insurance (3767; 60.8%). Age, sex, race, and insurance status on antipsychotic prescribing practices are described in Table 2. The oldest age group (70 + years) analyzed had a longer median duration of antipsychotic prescription (3 [1,5] days) compared to the youngest age group (18-30 years; 2 [1,7] days). Median duration of antipsychotic prescription was longer for males (3 [1,7] days) compared to females (2 [1,5] days). Those of Caucasian race received the lowest median duration of antipsychotic prescription (1.5 [1,4.25] days) compared to Asian, Black, and Indian race (2 [1,6] days, 3 [1,6] days, and 3 [1,9] days, respectively). There was no difference in the median duration of antipsychotic prescription between insurance types (both 3 [1,6] days).
Antipsychotic Prescribing Patterns by Age, Sex, Race, and Insurance Status in the ICU.
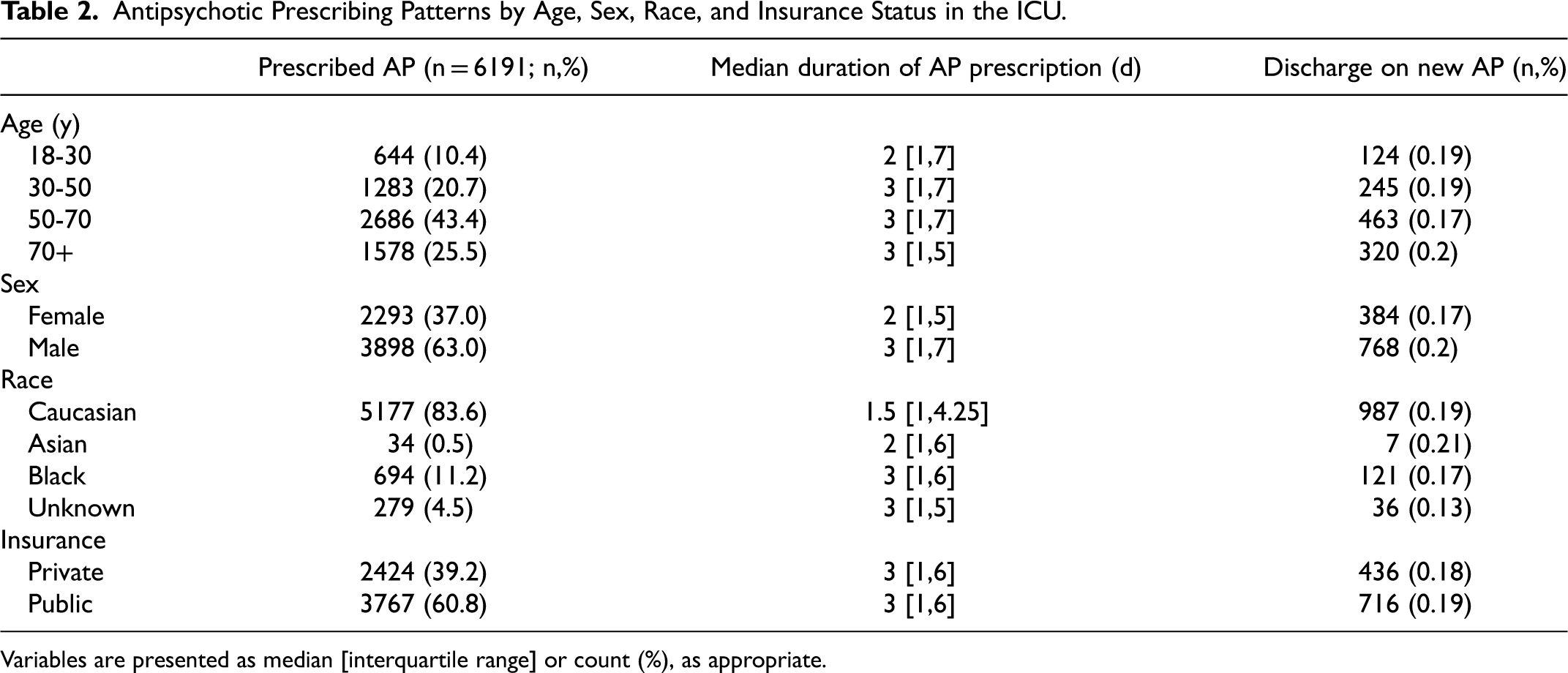
Variables are presented as median [interquartile range] or count (%), as appropriate.
When describing the continuation of antipsychotic prescriptions, we found similarities across all age groups listed in Table 2, with the oldest age group (70 + years) showing the highest proportion of continuation (320, 20%). Men were also shown to be more frequently continued on antipsychotic medications compared to females (768; 20% and 384; 17%, respectively). There was a trend showing that Asian race is more likely to be discharged on a new antipsychotic medication compared to unknown race (7; 21% vs 36; 13%). Continuation was similar between public and private insurance status (716; 19%, and 436; 18%, respectively).
Prescribing Patterns by Antipsychotic
Results from logistic regression testing the independent association of age, sex, race, and insurance status on administrations of olanzapine, quetiapine, and haloperidol are shown in Figure 1.
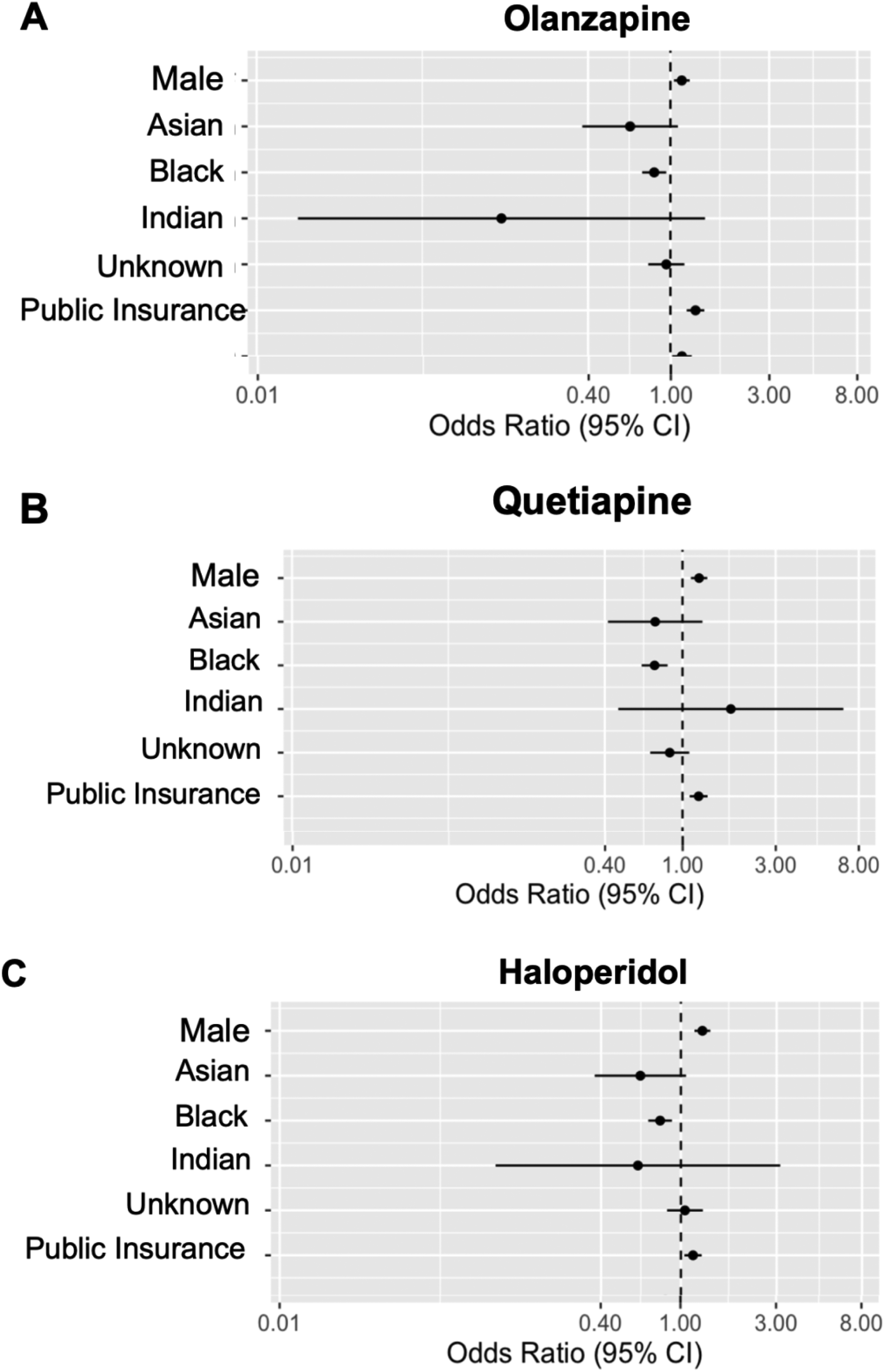
Odds of antipsychotic use by patient characteristics. Each figure is the result of a single model performed, adjusting for described covariates. (A) Age was nonlinearly associated with lower odds of receiving olanzapine (OR 0.99, 95% CI [0.86, 1.13], P < .001). Male sex was associated with increased odds of receiving olanzapine (compared to female sex; OR 1.13, 95% CI [1.04, 1.24], P = .0050). Black race was also significantly associated with olanzapine administration (compared to Caucasian race; Asian: OR 0.64, 95% CI [0.38, 1.09], Black: OR 0.83, 95% CI [0.73, 0.96]; P = 0.0086, Indian: OR 0.15, 95% CI [0.02, 1.47], Unknown: OR 0.96, 95% CI [0.78, 1.17]). Additionally, public insurance status was associated with increased odds of olanzapine administration (compared to private insurance; OR 1.32, 95% CI [1.20, 1.46], P < .0001). Age, sex, race, and insurance status were all associated with olanzapine administration. (B) Age was nonlinearly associated with higher odds of receiving quetiapine (OR 1.02, 95% CI [0.88, 1.18], P = .001). Male sex was associated with an increased odds of receiving quetiapine (compared to female sex; OR 1.22, 95% CI [1.10, 1.34], P = .0001). Black race was associated with quetiapine use (compared to Caucasian race; Asian: OR 0.72, 95% CI [0.42, 1.27], Black: OR 0.72, 95% CI [0.62, 0.84]; P = .0004, Indian: OR 1.77, 95% CI [0.47, 6.67], Unknown: OR 0.86, 95% CI [0.68, 1.08]). Additionally, public insurance status was associated with increased odds of quetiapine administration (compared to private insurance; OR 1.21, 95% CI [1.09, 1.34], P = .0004). Age, sex, race, and insurance status were all associated with quetiapine administration. (C) Age was nonlinearly associated with lower odds of receiving haloperidol (OR 0.84, 95% CI [0.73, 0.96], P = .0046). Male sex was associated with increased odds of receiving haloperidol (compared to the female sex; OR 1.28, 95% CI [1.17, 1.40], P < .0001). Black race was associated with haloperidol use (compared to Caucasian race; Asian: OR 0.63, 95% CI [0.37, 1.06], Black: OR 0.79, 95% CI [0.69, 0.90]; P = .0006, Indian: OR 0.61, 95% CI [0.12, 3.13], Unknown: OR 1.05, 95% CI [0.85, 1.29]). Additionally, public insurance status was associated with increased odds of haloperidol administration (compared to private insurance; OR 1.15, 95% CI [1.04, 1.27], P = .0058). Age, sex, race, and insurance status were all associated with haloperidol administration.
Olanzapine
Age was significantly associated with odds of receiving olanzapine (P < .001). Compared with patients aged 43 years (25th percentile), patients aged 69 years (75th percentile) had a lower odds of receiving olanzapine (OR 0.99, 95% CI [0.86, 1.13], Supplemental Figure 1). Sex and health insurance were also associated with olanzapine administration. Male patients had 13% greater odds of receiving olanzapine (OR 1.13; 95% CI [1.04, 1.24]; P = .0050) comparing to female patients, and patients with public health insurance status had 32% greater odds of receiving olanzapine (OR 1.32; 95% CI [1.20, 1.46]; P < .0001) comparing to those with private insurance. Race was associated with olanzapine administration (P = .0177). Comparing to Caucasian race, Black race demonstrating a decreased likelihood of olanzapine administration (Black vs Caucasian: OR 0.83, 95% CI [0.73, 0.95], P = .0086).
Quetiapine
Age was associated with the odds of receiving quetiapine (P = .001). Compared with patients aged 43 years (25th percentile), patients aged 69 years (75th percentile) had a higher odds of receiving quetiapine (OR 1.02, 95% CI [0.88, 1.18], Supplementary Figure 1). Male sex and public health insurance were also significantly associated with quetiapine administration (OR 1.22; 95% CI [1.10, 1.34] P = .0001 and OR 1.21; 95% CI [1.09, 1.34] P = .0004, respectively). Race was associated with quetiapine administration (P = .0004). Comparing to Caucasian race, Black race demonstrating a decreased likelihood of quetiapine administration (Black vs Caucasian: OR 0.72, 95% CI [0.62, 0.84], P < .0001).
Haloperidol
Age was associated with the odds of receiving haloperidol (P = .0046). Compared with patients aged 43 years (25th percentile), patients aged 69 years (75th percentile) had a lower odds of receiving haloperidol (OR 0.84; 95% CI [0.73, 0.96], Supplemental Figure 1).
Sex and public health insurance were also associated with the administration (Male vs Female OR 1.28, 95% CI [1.17, 1.40], P < .0001; public insurance vs private insurance OR 1.15, 95% CI [1.04, 1.27] P =.0058, respectively). Comparing to Caucasian race, Black race demonstrated a decreased likelihood of haloperidol administration (Black vs Caucasian: OR 0.79, 95% CI [0.69, 0.90], P = .0006).
Discussion
We found patient factors including age, sex, race, and insurance status to be associated with antipsychotic medication administration in critically ill adults. Male sex and public insurance were associated with increased odds of receiving all antipsychotic medications analyzed (olanzapine, quetiapine, and haloperidol) after controlling for potential baseline and illness confounders, while Black race was associated with decreased odds of administration compared to Caucasian race. Age was nonlinearly associated with all antipsychotic medications investigated. Older age, male sex, and non-Caucasian race were also associated with a longer median duration of antipsychotic prescription. Together these findings help illustrate nonmodifiable patient factors that may influence provider administration and continuation of these medications to patients during acute illness.
Antipsychotic medication administration was chosen given its high prevalence of use and subjective administration across health care providers and institutions.6,12,13 Despite the lack of robust evidence supporting the role of antipsychotic medications in preventing or treating delirium, they are frequently administered to treat acute symptoms, particularly agitation, with subsequent inappropriate continuation even after the acute indication has resolved.5-7,18-20 Their use and subsequent continuation therefore present potential acute and downstream health impacts to patients as these medications are associated with significant negative patient outcomes.6,8-11 Given the subjectivity in the determination of threat, with demographic characteristics associated with differential assessments of threat, there is room for bias in antipsychotic prescribing practices for agitated delirium.21,22 Previous studies have demonstrated the impact of these biases on prescribing patterns of antipsychotics in the inpatient psychiatric ward, emergency department, and pediatric populations.23-26 Despite this, we are not aware of any previous studies investigating this relationship within the ICU.
The influence of age and sex on outcomes within critically ill adults have previously demonstrated women, particularly older women, were less likely than men to be admitted to an ICU, receive mechanical ventilation, or invasive monitoring. 27 Our results align with these prior studies showing women were less likely than men to receive invasive or escalating pharmacologic therapies.28-30 Interestingly, despite receiving less invasive treatment, women experienced similar outcomes compared to men in these studies, suggesting these interventions may not be providing their intended benefit.29,30
Our findings demonstrated Black race was associated with decreased odds of antipsychotic medication administration and public health insurance was associated with increased odds of receiving medication. While the use of antipsychotic medications has not reliably demonstrated benefit to critically ill delirious patients as noted previously, much of this data was not present during the current study period. It is concerning that our findings echo those of other cohorts where Black race was independently associated with differential exposure to medications.31,32 Additionally, patients on primary public health insurance plans had increased odds of exposure to antipsychotic medications compared to privately insured patients. In investigating the influence of patient race on provider care and patient outcomes, there is additional evidence that disparities in care may be explained by variations in care across hospital systems and socioeconomic statuses more than individual patient race.33,34 Together these highlight study limitations that may impact generalization and interpretation of findings.
We have several strengths within this study that are highlighted by the large, granular, and robust dataset obtained. We were able to quantify not just prescription but receipt of antipsychotic medication to better define our primary outcome. Additionally, this study was conducted over a period where our institution practiced high ABCDEF bundle compliance across ICUs with guidance to perform nonpharmacologic interventions prior to administration of medications in the management of ICU delirium. Our study is limited by the single-center design that prevents us from comparing prescribing patterns across hospital systems. The 4 different ICUs included within this study, however, are staffed by different intensivists with varied training backgrounds that allow for a large and diverse representation across our center. Our patient cohort is representative of admissions received during the study period, which is primarily Caucasian. Smaller proportions of minority groups (Black, Asian, Indian) limit our ability to generalize findings within these patient groups. We were also unable to obtain granular data on patient income or socioeconomic status, history of psychiatric illness or abuse disorders, which may limit the interpretability of our findings. Data on adverse effects of antipsychotic medications was not collected. Additionally, critically ill patients often have complex and dynamic changes in care that influence individual patient and provider decisions. Our study is not designed to describe these individual factors but to provide data on trends in prescribing patterns across a large system over several years.
As we aim to improve our use of antipsychotic medications and minimize adverse medication exposure within the ICU, understanding the association of nonmodifiable patient factors on prescribing patterns is valuable to understand when these medications are used. Interventions aimed at optimizing medication regimens should consider provider prescribing patterns and potential implicit biases to improve optimization strategies.
Conclusions
Age, sex, race, and insurance status were all associated with administration of some or all antipsychotic medications. Recognizing the association of nonmodifiable patient factors on prescribing patterns is valuable to start understanding how these medications are used and the potential impact they carry on patient outcomes.
Supplemental Material
sj-docx-1-jic-10.1177_08850666231198030 - Supplemental material for The Association of Nonmodifiable Patient Factors on Antipsychotic Medication use in the Intensive Care Unit
Supplemental material, sj-docx-1-jic-10.1177_08850666231198030 for The Association of Nonmodifiable Patient Factors on Antipsychotic Medication use in the Intensive Care Unit by Jennifer Connell, Brittany McCann, Xiaoke Feng, Matthew S. Shotwell, Christopher G. Hughes and Christina S. Boncyk in Journal of Intensive Care Medicine
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CSB and CGH are consultants for Sedana Medical. The remaining authors declared no potential conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute on Aging, Society of Academic Associations of Anesthesiology & Perioperative Medicine, Foundation for Anesthesia Education and Research, National Institutes of Health (grant numbers R01AG053582, GM120484, HL111111).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
