Abstract
Traditional point-of-care ultrasound (POCUS) training highlights discrete techniques, single-organ assessment, and focused protocols. More recent developments argue for a whole-body approach, where the experienced clinician-ultrasonographer crafts a personalized POCUS protocol depending on specific clinical circumstances. This article describes this problem-based approach, focusing on common acute care scenarios while highlighting practical considerations and performance characteristics.
Introduction
Point-of-care ultrasound (POCUS) has become nearly indispensable in the contemporary care of critically ill patients. It can improve the safety of bedside procedures, 1 enhance diagnostic acumen,2–4 and help monitor patient hemodynamics and response to therapies. 4 However, POCUS education still relies heavily on workshops and bedside training where discrete, individual ultrasound techniques are taught and practiced. While useful for teaching basic image acquisition, this manner of organ-specific ultrasound education can be challenging to apply in complex real-world patients with multisystem dysfunction.
Not long after the initial wave of individual POCUS techniques were created, a series of standardized scanning protocols followed.5,6 While an improvement on approaches based on isolated ultrasound techniques, these protocols still restricted the field of search to a single-organ system, more or less. More recently, experts have called for a move away from compartmentalized scanning and standardized protocols toward a more whole-body, multiorgan approach. 7 In this approach, the clinician, understanding which parts of the examination will add value and which can be omitted, crafts a personalized POCUS protocol depending on the clinical circumstances. This paper aims to describe how multiorgan POCUS can be integrated into a clinical workflow, describing a problem-based approach to common critical care scenarios while highlighting practical considerations and performance characteristics.
POCUS for Assessment of Shock
POCUS is a first-line assessment tool for determining the cause of undifferentiated hypotension. It has replaced more invasive techniques such as pulmonary arterial catheterization and is the current standard of care for the assessment of patients with shock. 8 This topic, however, has been extensively discussed elsewhere in the POCUS literature,2,9 and therefore we have focused our efforts on less commonly discussed clinical scenarios.
POCUS to Differentiate Causes of Respiratory Failure
For the patient with respiratory failure, multiorgan POCUS including cardiac, lung, and vascular ultrasound for deep vein thrombosis is standard and has been described in various protocols such as the BLUE, FALLS, and Volpicelli assessments.6,10 For pneumonia, 11 pneumothorax, 12 and interstitial syndromes 13 in particular, these protocols have an impressive accuracy, especially when compared to chest X-ray. The protocols mentioned above do not account for newer techniques like B-line assessment, 14 nor do they include advanced cardiac assessments to estimate left atrial pressure and diastolic function. Despite these omissions, determining the etiology of respiratory failure is one instance where a multiorgan POCUS assessment has been well established, and therefore we will not elaborate further.
POCUS to Identify the Source of Bleeding
An unidentified young woman arrives by ambulance to the resuscitation bay after having been thrown off her motorcycle at highway speed. She is awake and responsive, but profoundly hypotensive and tachycardic. She complains of pain to the right side of her chest and to her abdomen diffusely.
In scenarios such as this, where hemorrhage is suspected as the cause of shock, ultrasound can be helpful in identifying the source of bleeding. Individual POCUS skills, each developed in isolation and studied with varying degrees of rigor, can be combined in a systematic search. Recognizing the performance characteristics of each tool is important as some perform extremely well, some very poorly, and most somewhere in between.
It is unfortunately true that POCUS is rarely the best test for any individual hemorrhagic complication. In nearly all cases, computerized tomography (CT) performs better and eliminates problems related to operator dependence in image acquisition. CT does, of course, delay care, require transport to an unsafe location, and expose the patient to radiation. POCUS is therefore best applied in patients who are unstable and unsuitable for transport to CT, outside of the few ultrasound applications that perform as well as CT.
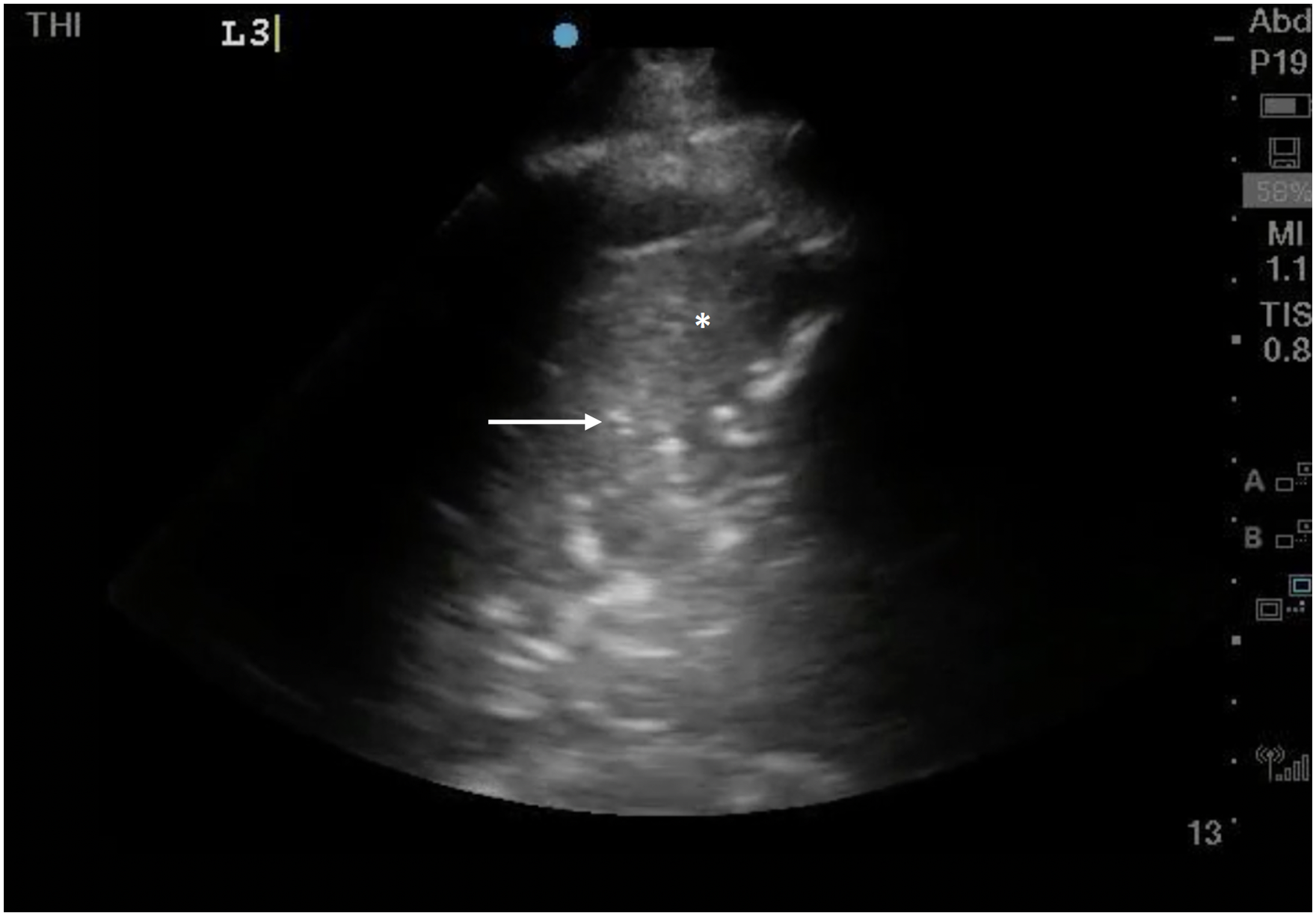
Ultrasound has been well proven to reliably detect blood in the pleural space (Figure 1, Video 1). Common clinical scenarios here include blunt or penetrating trauma, or hemodynamic deterioration in a postoperative patient. Drawing mostly from the extensive blunt trauma literature, ultrasound performs well indeed (sensitivity 0.67, specificity of 0.99 in one large meta-analysis 15 ), but attention must be paid to the mediocre sensitivity; smaller collections may be missed, and imaging may need to be repeated, especially in obese patients. The specific technique is well described elsewhere. 16 Important related injuries including pneumothorax 17 and rib fractures 18 are also reliably detected by ultrasound.
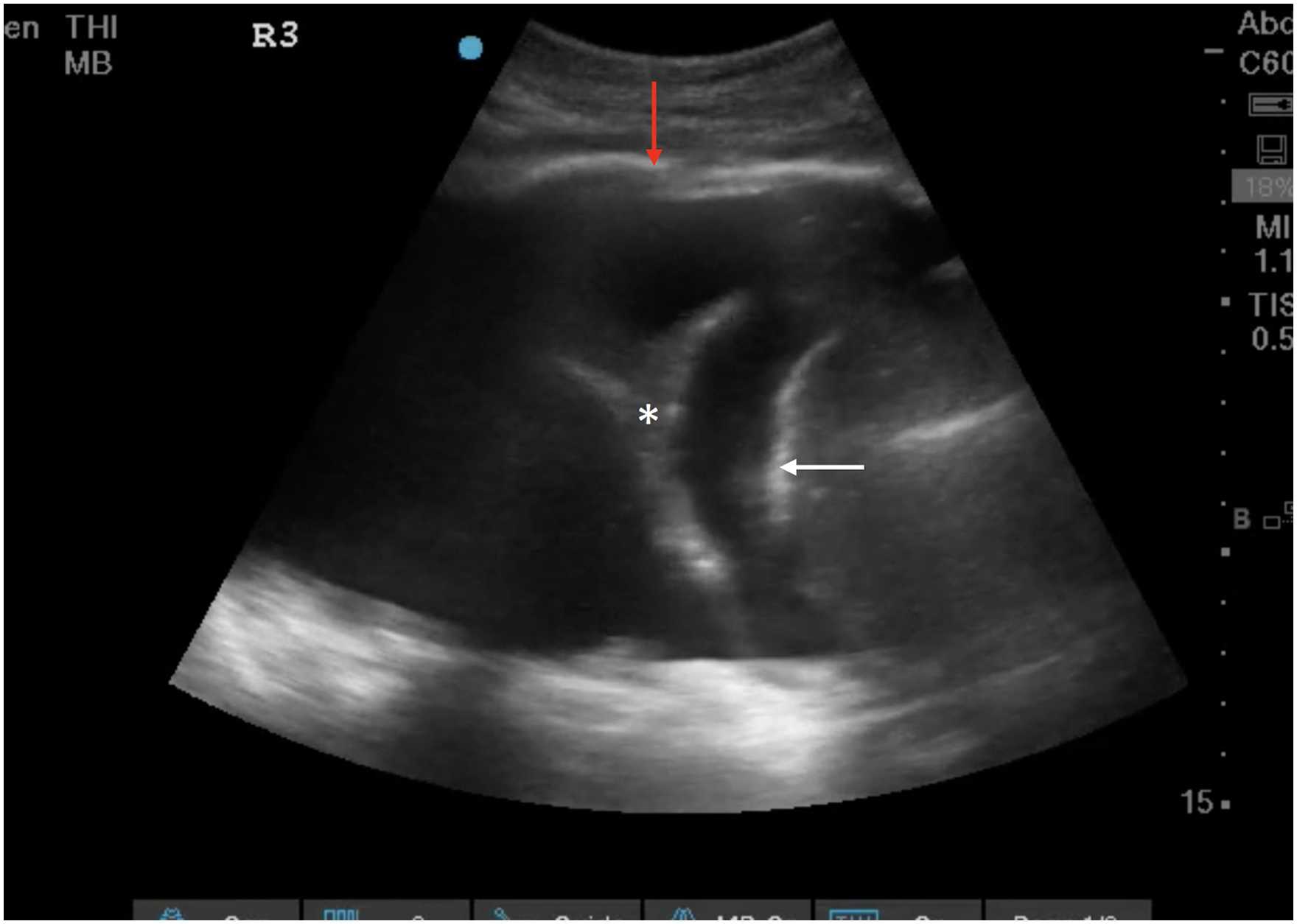
Large anechoic pleural effusion (anechoic space), bordered by the diaphragm (white arrow), pleura (red arrow), and containing hepatized lung (asterix*) in keeping with hemothorax in the right clinical context.
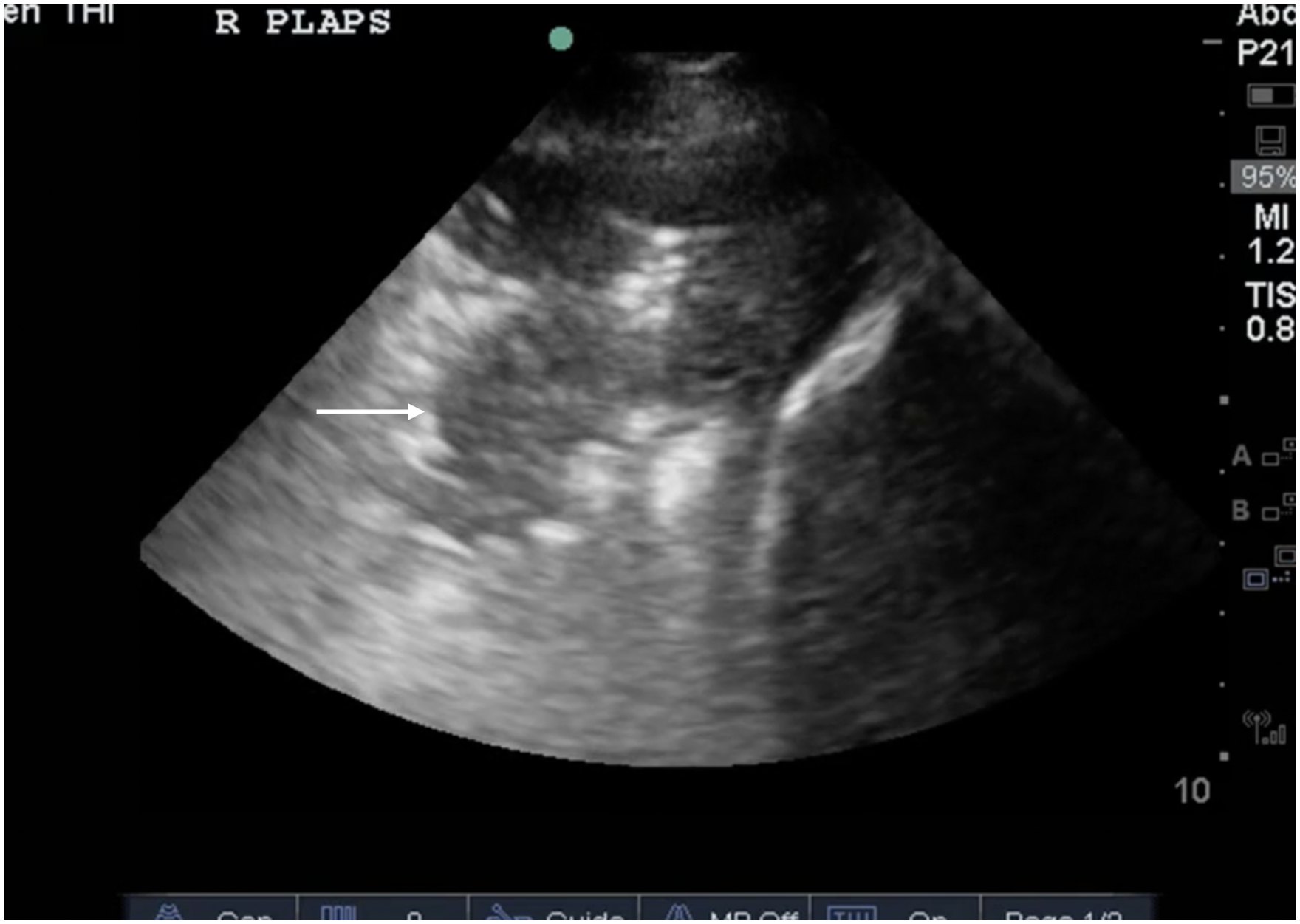
Detection of blood in the peritoneal cavity is very well studied and has good performance characteristics (sensitivity 0.75, specificity of 0.95 in one large meta-analysis 19 ), with most of the evidence in scenarios involving blunt abdominal trauma. The Focused Assessment with Sonography in Trauma (FAST) exam 5 is probably the best-known POCUS protocol of all time and has completely replaced diagnostic peritoneal lavage for this indication (Figure 2, Video 2). Because of its excellent specificity, a positive FAST exam in the correct clinical context obliviates the need for CT scan and should lead directly to operative intervention. Some limited evidence exists for the detection of the specific cause of intra-peritoneal bleeding, either splenic rupture 20 or liver laceration, 21 but CT remains the gold standard.
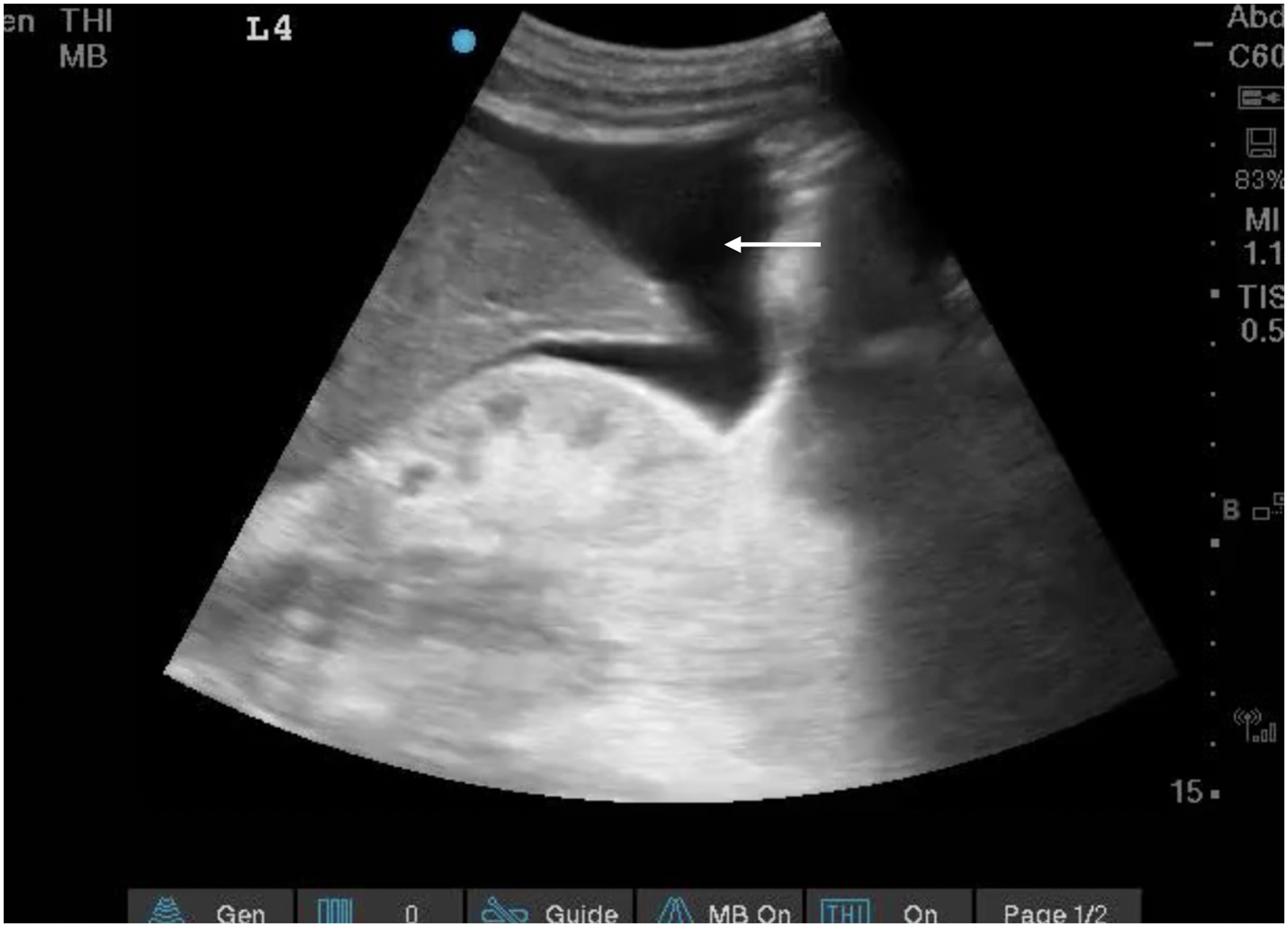
Positive FAST exam with fluid in the hepatorenal space and at the inferior tip of the liver (white arrow).
Staying in the abdomen but moving to nontraumatic situations, there is a robust body of evidence supporting ultrasound in the detection of abdominal aortic aneurysms, both ruptured and unruptured. For unruptured aneurysm, ultrasound is highly accurate, and indeed a reliable method to follow aneurysm size over time 22 (Figure 3, Video 3). For patients presenting in shock or with severe abdominal pain where a ruptured aneurysm is suspected, POCUS has an excellent sensitivity and specificity 23 ; the specific technique is well covered elsewhere. 24
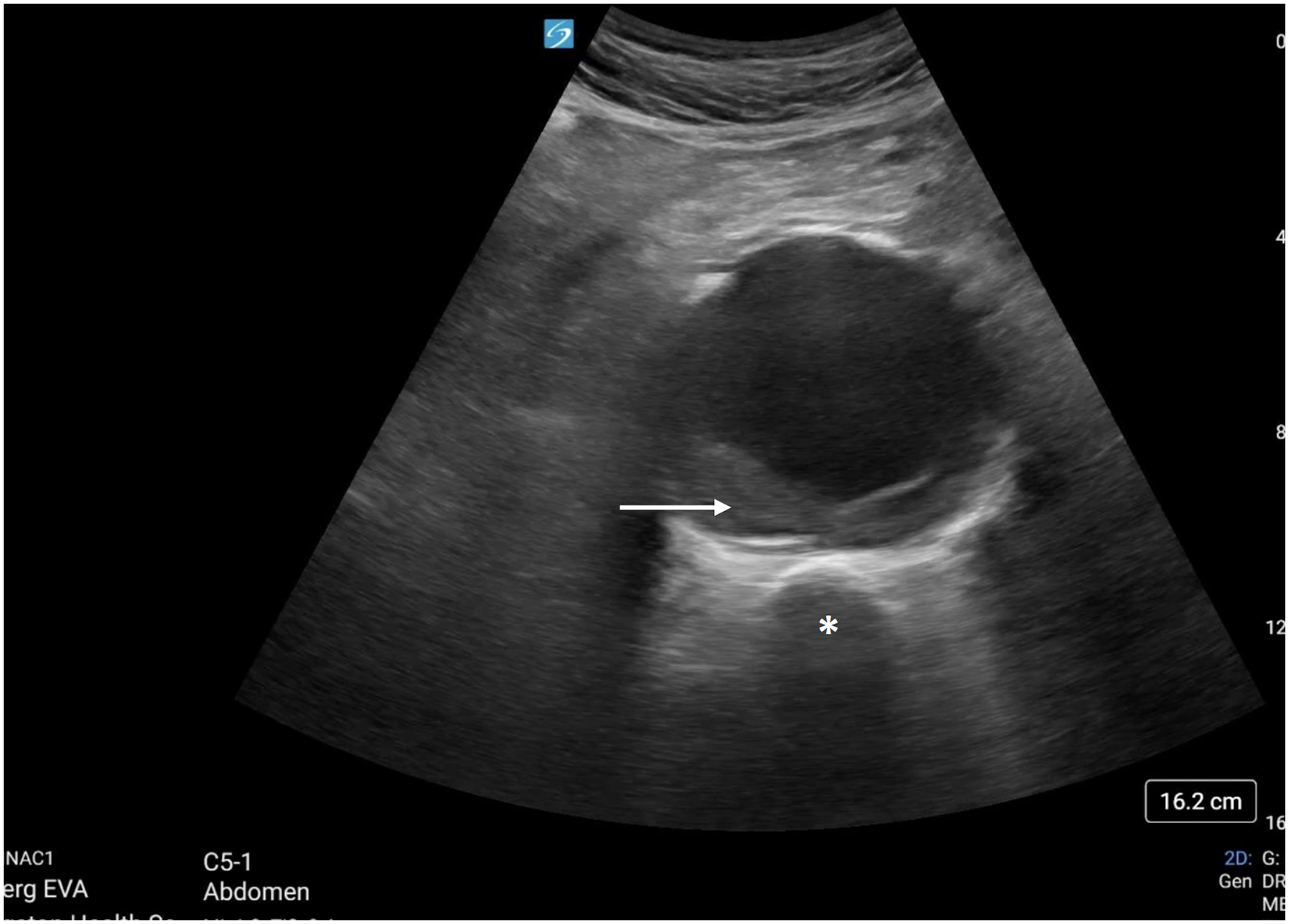
Abdominal aortic aneurysm with intramural hematoma (white arrow) seen overlying the vertebral body (asterix*).
Bleeding into the retroperitoneal space is usually a challenging clinical scenario. It can occur spontaneously, with minor trauma, or post-procedurally. Patients who are anticoagulated or coagulopathic are at higher risk, and the symptoms are typically nonspecific with no helpful physical exam findings. It would be ideal if ultrasound could help here, as a quick and easy test, but unfortunately, it cannot. While case reports have described the technique, 25 the performance characteristics are unknown but likely poor due to a combination of patient factors and a lack of easy ultrasound landmarks. CT remains the gold standard if the diagnosis is suspected.
The use of ultrasound in unstable pregnant patients will not be covered in detail here, but the tool has a major role to play. Confirming a viable intra-uterine pregnancy with ultrasound is the standard of care 26 when ectopic pregnancy is suspected. A ruptured ectopic pregnancy may present with intra-peritoneal bleeding, which will be detected via the FAST exam as described above. In later pregnancy, ultrasound plays a role in fetal monitoring 27 and in the investigation of important placental abnormalities such as placenta accreta or low-lying placenta 28 which may predispose to bleeding.
Beyond hemorrhage into the chest or abdomen, there are other very uncommon sites of catastrophic bleeding. Unfortunately, ultrasound is unlikely to offer any major benefit in any of these scenarios. Patients may bleed into a large compartment such as the thigh, for example. While ultrasound can detect large collections, the evidence base is very limited (and mostly focused on rectus sheath hematomas 29 ), the performance characteristics are unknown, and the diagnosis is generally clinically obvious. Similarly, patients can occasionally bleed massively into a joint (typically the hip, and almost always in the post-operative period). While ultrasound can, in theory, detect such a hemorrhage, 30 the diagnosis is generally obvious, and CT the superior test.
POCUS for Assessment of Sepsis
A 52-year-old person with cirrhosis is admitted to hospital with hepatic encephalopathy. Several days after admission they develop fever, leukocytosis, and worsening hypotension.
Sepsis is seen in all acute care medicine specialties and is a very common reason for admission to the intensive care unit (ICU). Timely diagnosis and source control are essential, but identification of the septic focus is not always straightforward. A significant subset of patients never have their source of sepsis identified, 31 and POCUS can be helpful in this regard. A multiorgan POCUS assessment of the lungs, abdomen, heart, and soft tissues resulted in a 25% improvement when compared to clinical examination alone for identifying the source of sepsis, as well as drastically improved time to diagnosis in one study, for example. 32
Not every organ system needs to be assessed with POCUS in every septic patient, every time. A thorough multiorgan assessment based on patient history, physical examination, and other investigations can be employed to identify the septic focus. Cardiac POCUS can be used first to confirm the likely cause of hypotension; a hyperdynamic left ventricle has been found to have a positive likelihood ratio of 5.3 for sepsis as a specific cause of shock. 33 For advanced practitioners, measuring an elevated left ventricular outflow tract velocity–time integral (LVOT VTI) and stroke volume in the presence of hypotension is also strongly supportive of a vasodilatory etiology.33,34 Cardiac ultrasound may also give clues as to the underlying etiology of sepsis; for example, a detailed assessment of cardiac valves may reveal the presence of endocarditis as evidenced by an iso- or hyperechoic mobile density (Figure 4, Video 4). Here POCUS should be viewed as a rule-in rather than a rule-out test, with a specificity of 94% in one study, but notably low sensitivity. 35
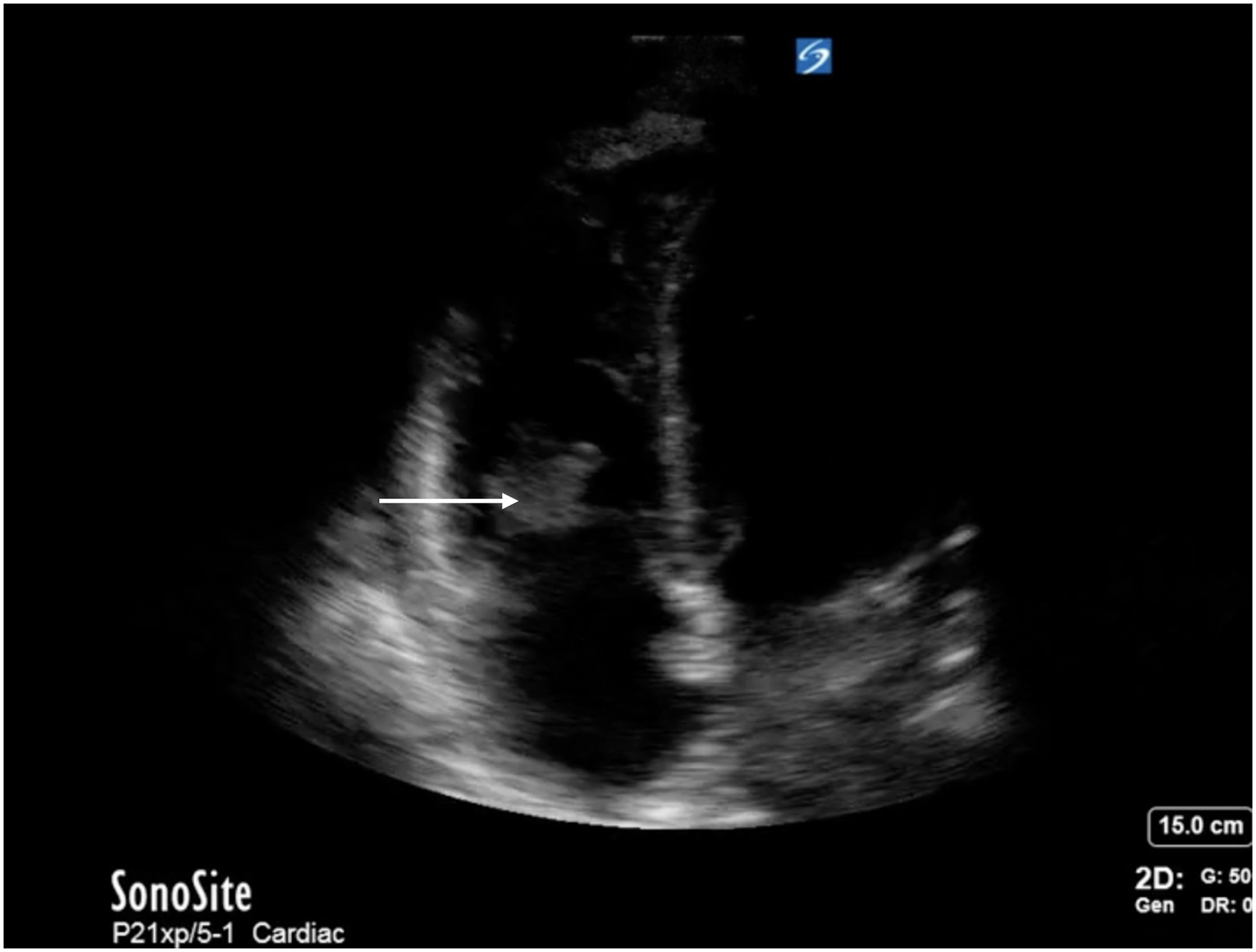
An apical four-chamber view of the heart with a mobile hyperechoic density is seen on the tricuspid valve (white arrow), supportive of infective endocarditis.
The utility of lung ultrasound for the diagnosis of pneumonia has been well established, with excellent sensitivity and specificity. 11 Findings supportive of pneumonia include heavily consolidated (“hepatized”) lung, subpleural consolidation, dynamic air bronchograms, and the shred sign 11 (Figures 5 and 6, Videos 5 and 6). An assessment of the pleural space is important here; effusions, especially those with a complex sonographic appearance, should be considered for drainage where empyema is suspected. 16
Lung Ultrasound demonstrating hepatized lung (white asterix*) with hyperechoic dynamic air bronchograms (white arrow).
Lung ultrasound with shred sign: the interface between hepatized and aerated lung appears as a shredded, irregular line (white arrow). This is indicative of a nontranslobar consolidation.
Moving on to the abdomen, POCUS enables rapid and accurate evaluation of intra-abdominal free fluid. 36 Indicators of complex or exudative fluid such as fibrin stranding or cellular debris may prompt a diagnostic paracentesis (where POCUS can help with safe landmarking 37 ) or CT, if, for example, there is clinical concern for bowel perforation (Figure 7, Video 7). Biliary ultrasound is commonly employed for the identification of cholecystitis, 38 with typical findings including gallbladder wall edema, pericholecystic fluid, the presence of hyperechoic stones, and a sonographic Murphy's sign. The sensitivity of POCUS for these findings ranges from 70% for sonographic Murphy's sign to 93% for gallstones, and POCUS information can be valuable for surgical decision-making.38,39 The exam may also support more severe infections like cholangitis with the presence of a dilated common bile duct. 40 Other pathologies which may be evident on abdominal ultrasound include appendicitis, 41 diverticulitis, 42 liver abscess, 43 or portal venous gas 44 ; a more advanced skill set is required for any of these diagnoses, and POCUS is generally considered inferior to CT imaging here. Given the frequency of urinary sources of sepsis, renal applications should not be ignored. Where urosepsis is suspected or confirmed, a search for hydronephrosis (potentially caused by a septic stone) or perinephric abscess should be undertaken. POCUS performs very well here, 45 and the technique is straightforward.
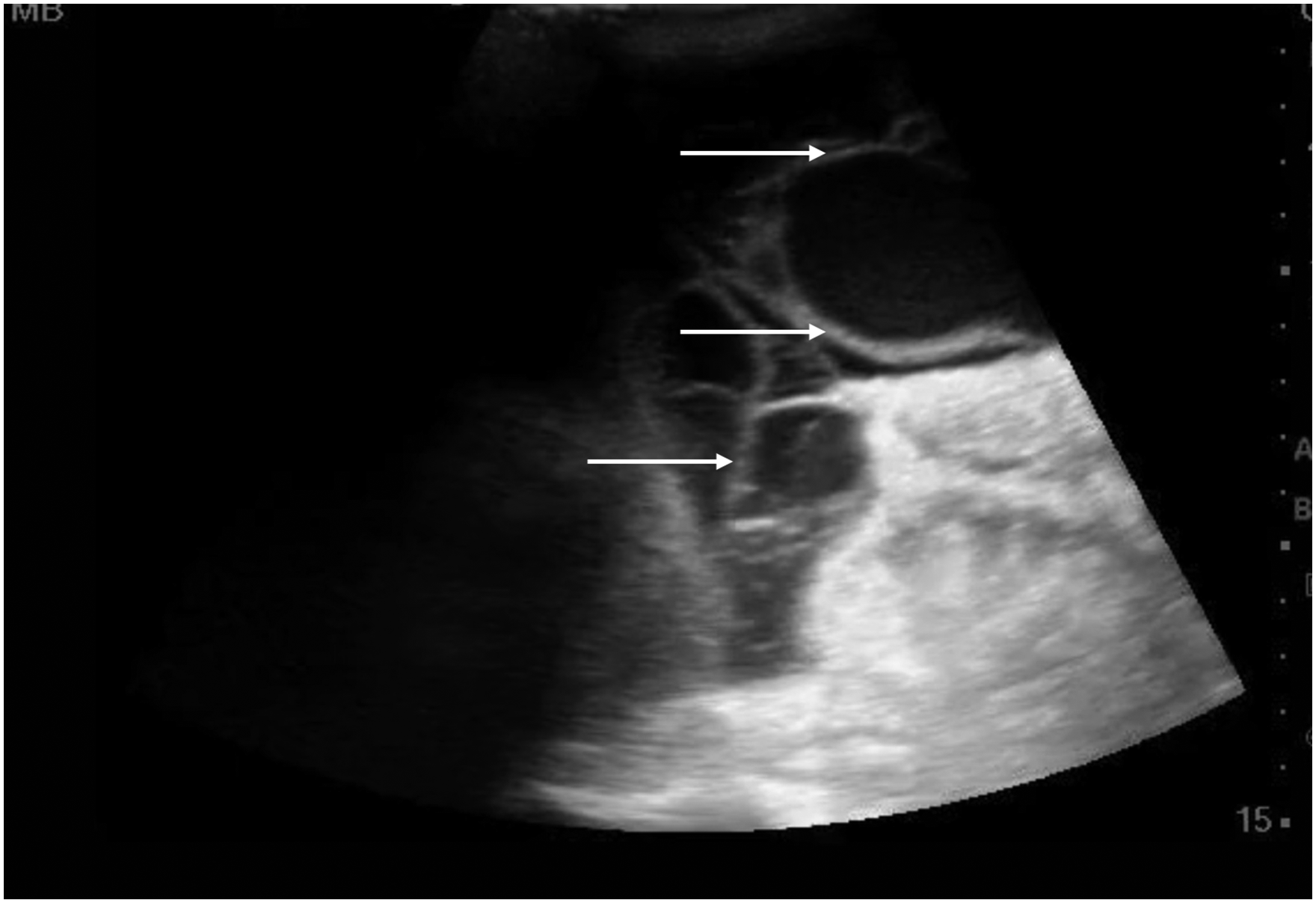
Complex, loculated ascitic fluid. Some (not all) loculations have been highlighted with white arrows.
Musculoskeletal and soft tissue ultrasound can help diagnose abscesses requiring drainage, cellulitis, and necrotizing skin and soft tissue infections. 46 The most sinister of these infections, necrotizing fasciitis, can be diagnosed by identifying fluid and air along the fascial plane (Figure 8, Video 8). Early data suggests that POCUS may have excellent test characteristics here, 47 but other imaging modalities remain the standard of care.
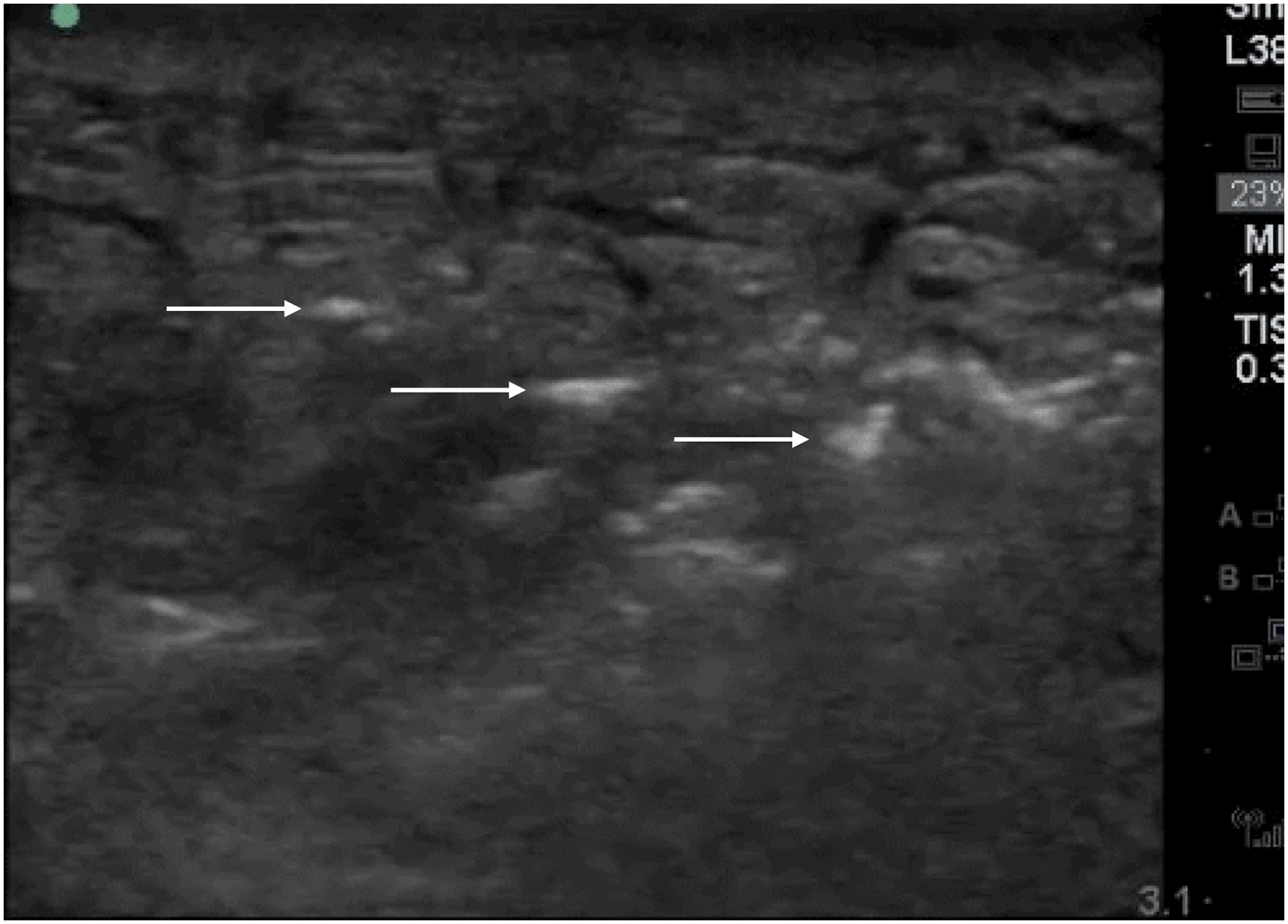
Soft tissue infection with cobblestoning (hypoechoic fluid surrounding the subcutaneous tissue and fat*) and air within the fascial plane that appears as linear hyperechoic densities (white arrows), consistent with necrotizing fasciitis.
Once the source of sepsis has been identified and treatment initiated, ongoing management of the septic patient can also be assisted with POCUS. When used routinely as part of ICU rounds, POCUS has been associated with shortened length of stay and duration of mechanical ventilation, 48 possibly related to use of a more fluid-restrictive strategy. A discussion of POCUS-assisted fluid management is complex and beyond the scope of this paper. 49
POCUS in Renal Failure
A 68-year old male with a history of heart failure and benign prostatic hypertrophy presents to the Emergency Department with a 3-day history of malaise, shortness of breath, and decreased urine output. On presentation, he is found to be hypotensive and mildly hypoxic. Initial bloodwork reveals a serum creatinine of 480
POCUS can be extremely valuable in the evaluation of the patient with renal failure. Working within the traditional “pre-renal/renal/post-renal” algorithm, multiorgan POCUS can be employed in conjunction with traditional parameters, with a complexity that will depend on the operator's skills and experience. 50
The lowest-hanging fruit in the POCUS exam for acute kidney injury (AKI) is the exclusion of post-renal causes. The kidneys can be examined for signs of hydronephrosis, the bladder for urinary retention, and, in patients with an indwelling Foley catheter, POCUS can confirm or refute correct balloon placement (Figures 9 and 10, Video 9 and 10). Renal and bladder ultrasound for obstructive uropathy demonstrates stellar characteristics, with sensitivities and specificities of 86-100%.51,52
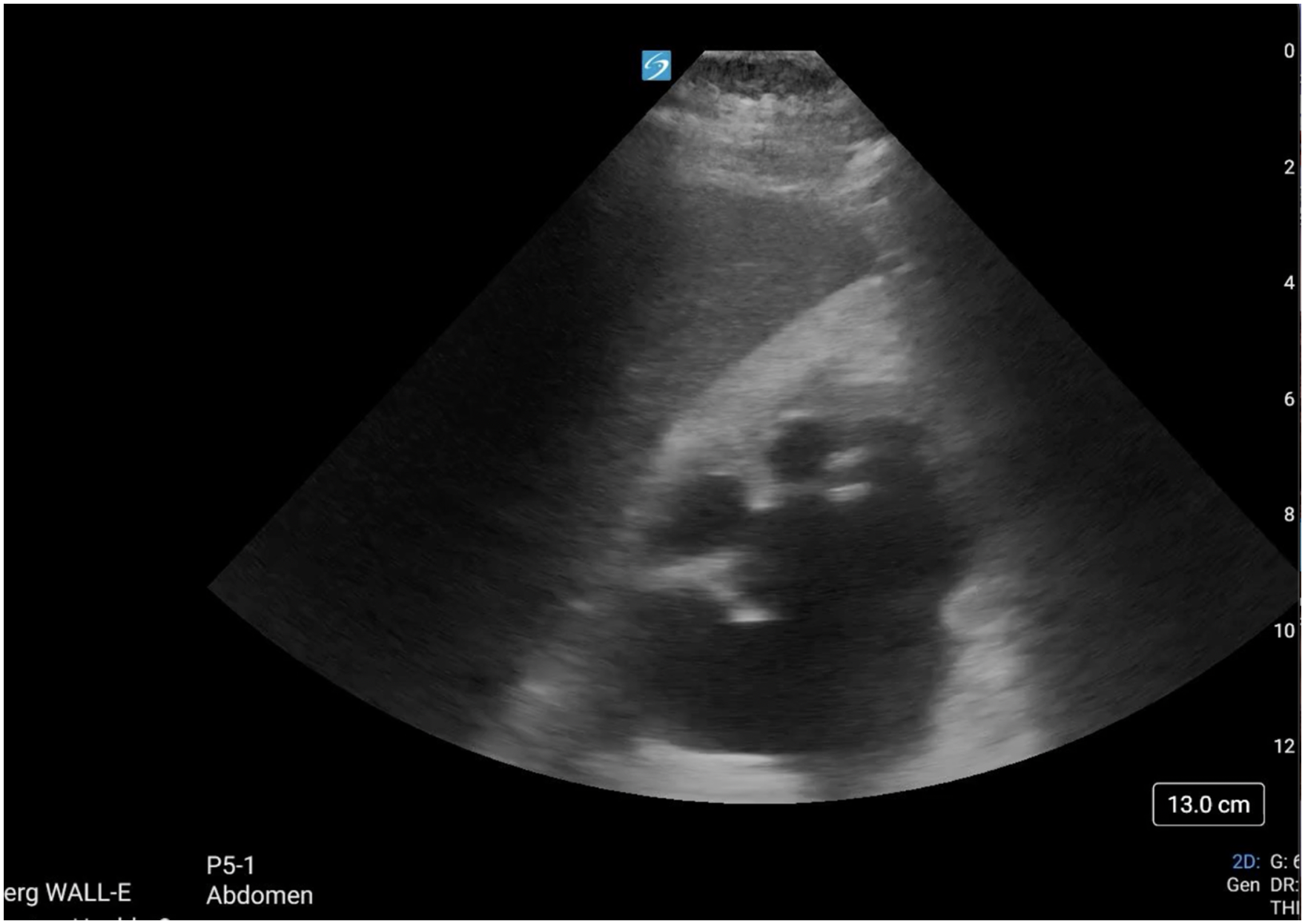
Dilated, anechoic renal pelvis and calyces, consistent with moderate hydronephrosis.
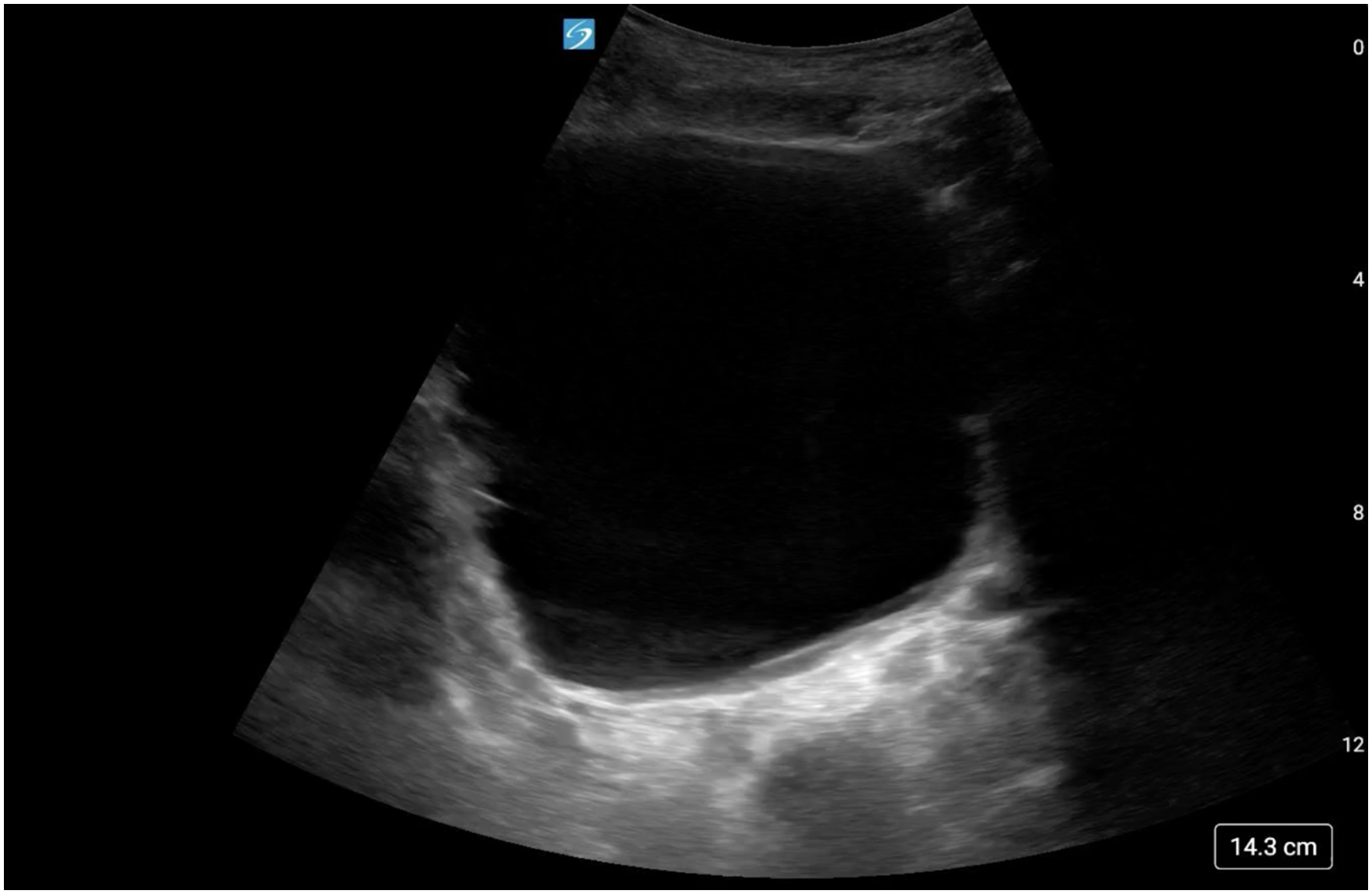
Fluid-filled distended bladder.
In considering possible pre-renal etiologies of AKI, including both hypovolemia and venous congestion, POCUS is an indispensable tool. Recalling that renal perfusion depends on both adequate arterial inflow in addition to easy venous outflow with low resistance, both sides of this equation need careful consideration. Starting on the arterial inflow side, cardiac ultrasound can reveal evidence of left heart dysfunction and inadequate forward flow. This can be assessed subjectively by basic 2D views; or, for advanced users, via measurement of LVOT VTI 53 (Figures 11 and 12, Video 11). Particularly in critically unwell patients, the latter gives a more accurate assessment of real-time hemodynamics; where a low LVOT VTI may be supportive of inadequate forward flow as a driving factor behind renal dysfunction. For the detection of inadequate intravascular volume, the inferior vena cava (IVC) can be assessed. While this parameter may be useful at extremes—a very small, completely collapsing IVC in a spontaneously breathing patient may indicate low right atrial pressure—it is imperative to be aware of the many confounders and pitfalls of this exam54,55 (Figure 13, Video 12). The assessment of a patient's intravascular volume remains challenging, and while ultrasound may help to some degree it does not solve all related problems.
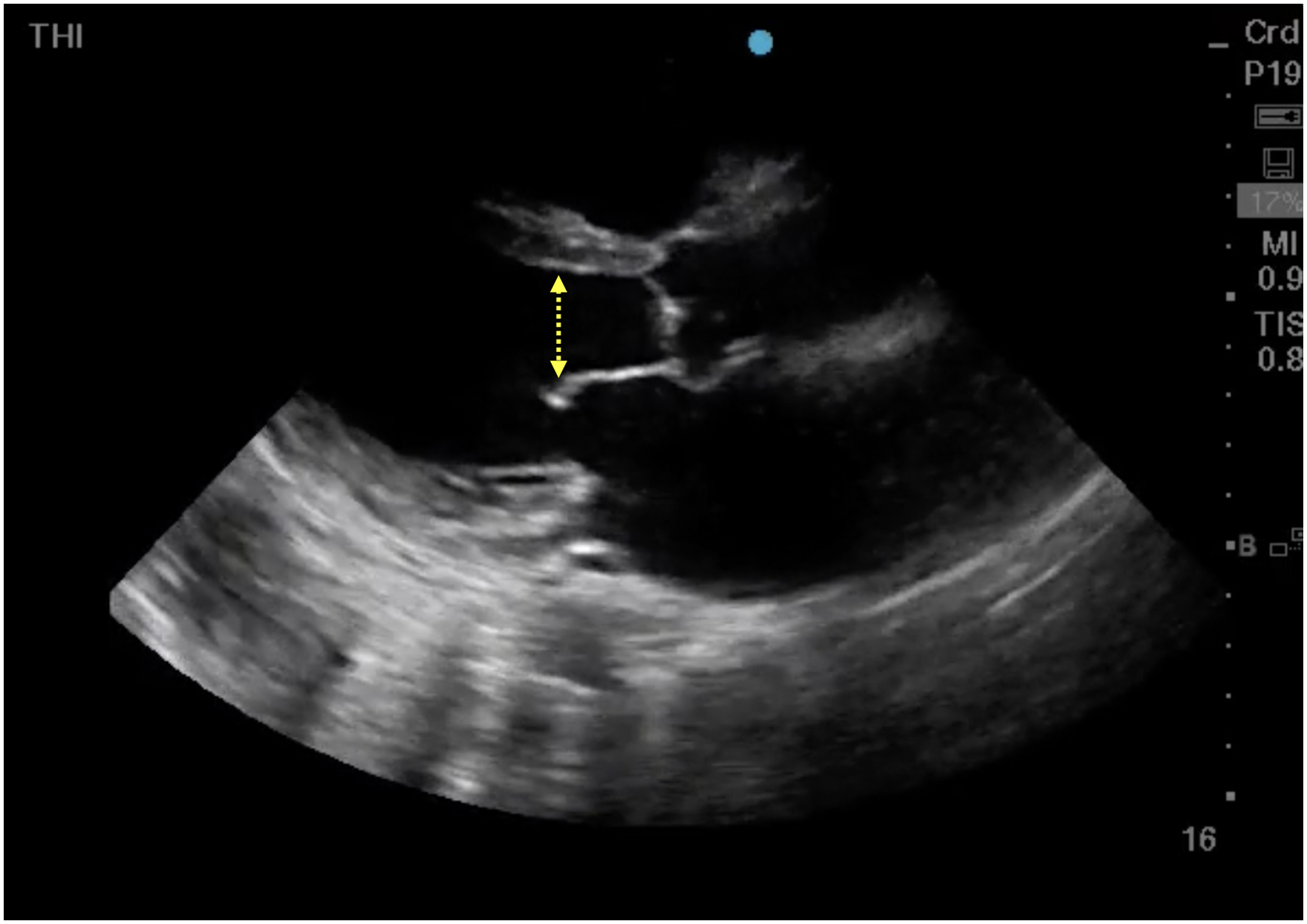
Parasternal long axis view demonstrating a severely reduced ejection fraction. In this still, image is represented by the increased distance between the anterior mitral valve leaflet and the septum (dashed yellow line).
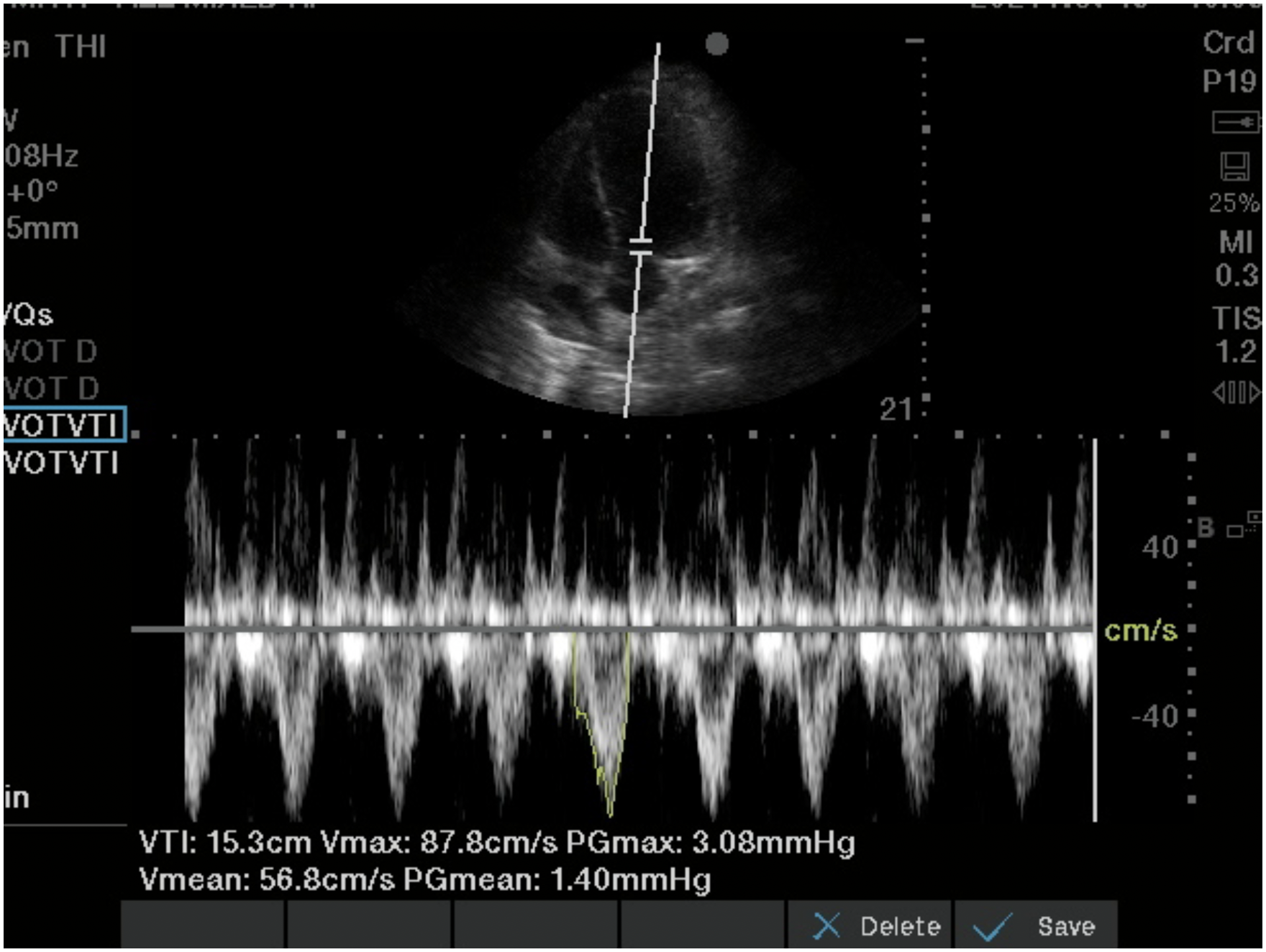
Spectral Doppler trace obtained from the left ventricular outflow tract in an apical five-chamber view. This trace demonstrates reduced left ventricular tract outflow tract velocity–time integral (VTI) of 15.3 cm (normal range 18-22 cm).
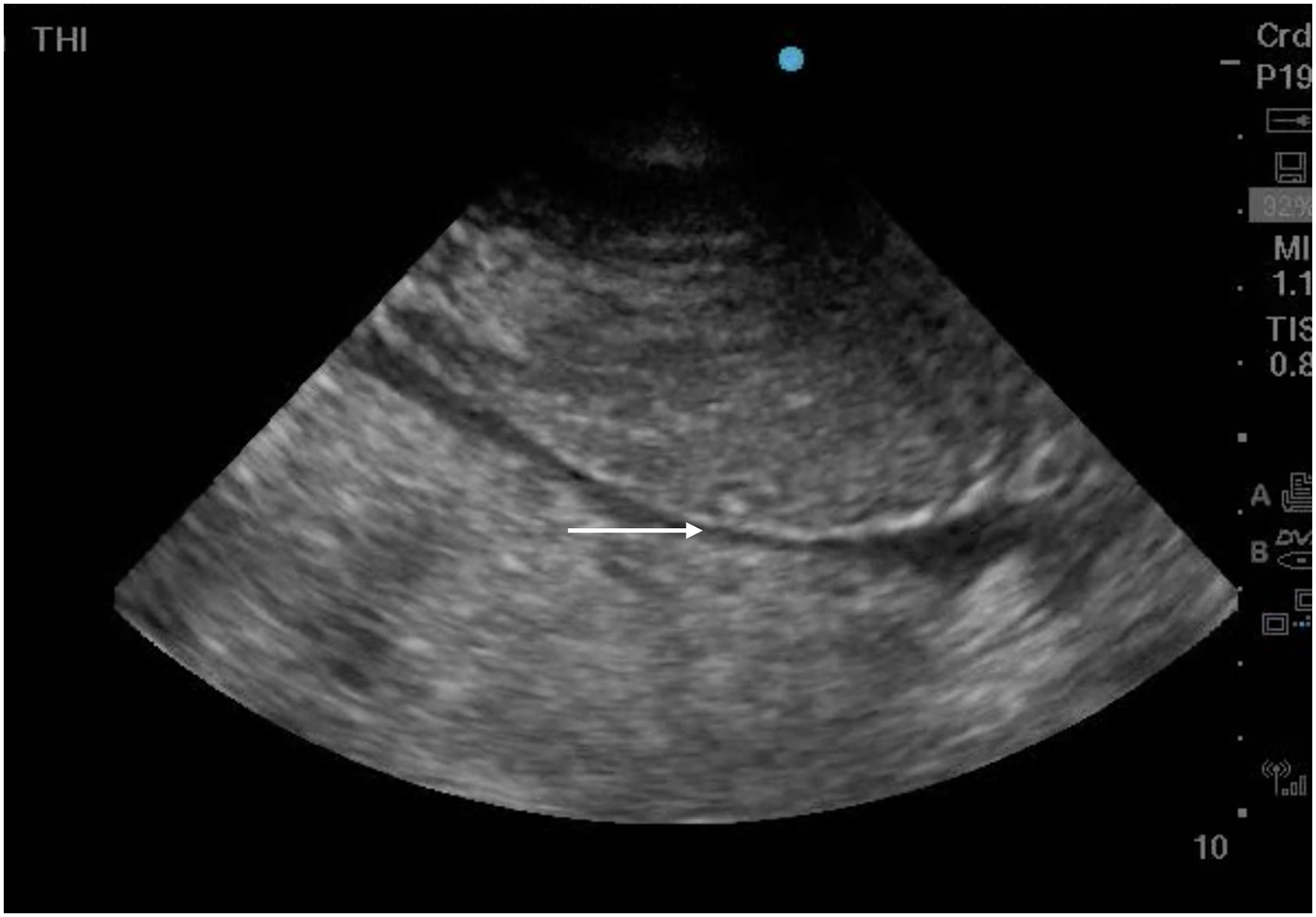
Small, fully collapsible inferior vena cava (white arrow) in the setting of low right atrial pressure.
The venous side of the renal perfusion equation, long ignored, has recently received significant attention. To detect potentially deleterious venous congestion, the hepatic, portal, and intra-renal veins may be interrogated with spectral Doppler. Each of these vessels has a characteristic Doppler profile under normal physiologic conditions, which changes predictably with increasing right atrial pressure (Figures 14–17). While a numerical scoring system, called a VeXUS score, may be used, 56 a recent excellent comprehensive review advocates for a standardized pattern-based description of the individual waveforms. 57 The evidence base behind this technique is still in evolution; increased venous congestion has been associated with worse renal outcomes in post-cardiac surgery patients and in general critically ill populations.56,58 However, as of yet there are no prospective studies looking at the use of these techniques to guide decongestion therapy. While there is a great deal of promise here, it must be approached with caution given the still-evolving evidence base as well as the technical challenges and pitfalls associated with these examinations. 59
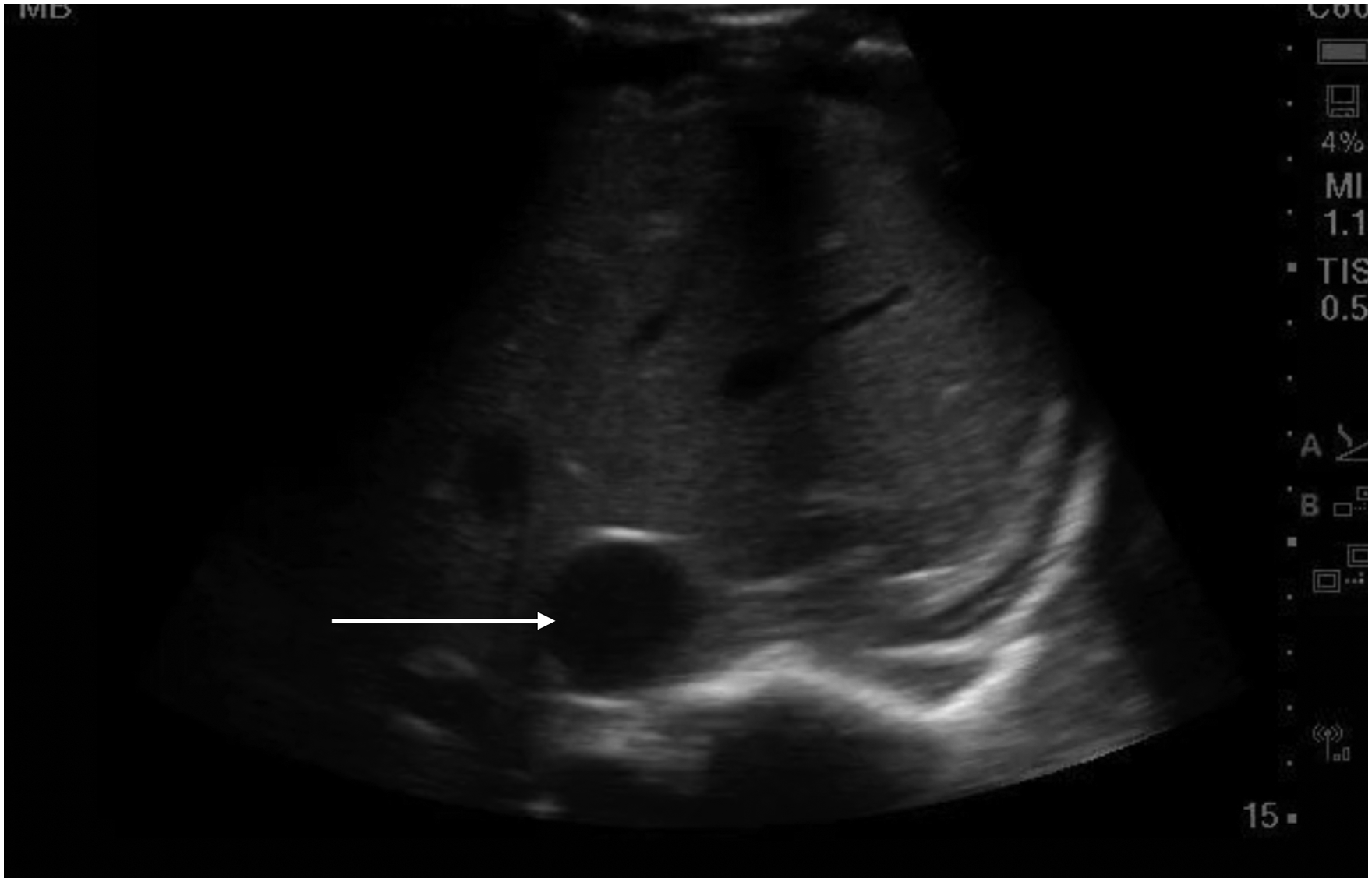
Short-axis view of a plethoric, spherical inferior vena cava (white arrow), a prerequisite for significant venous congestion.
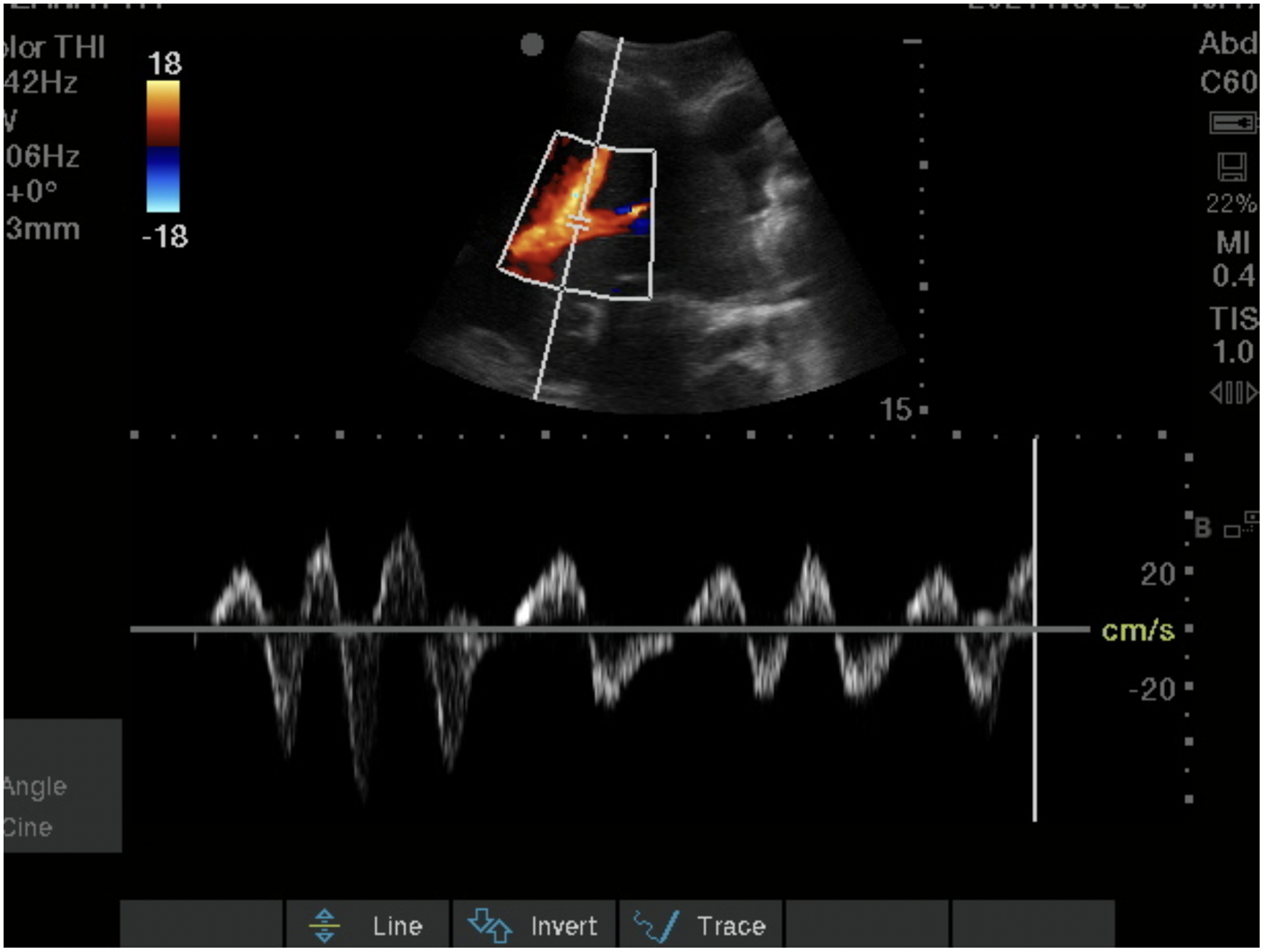
Spectral Doppler tracing from the hepatic vein shows systolic reversal (S wave reversal), consistent with severe congestion.
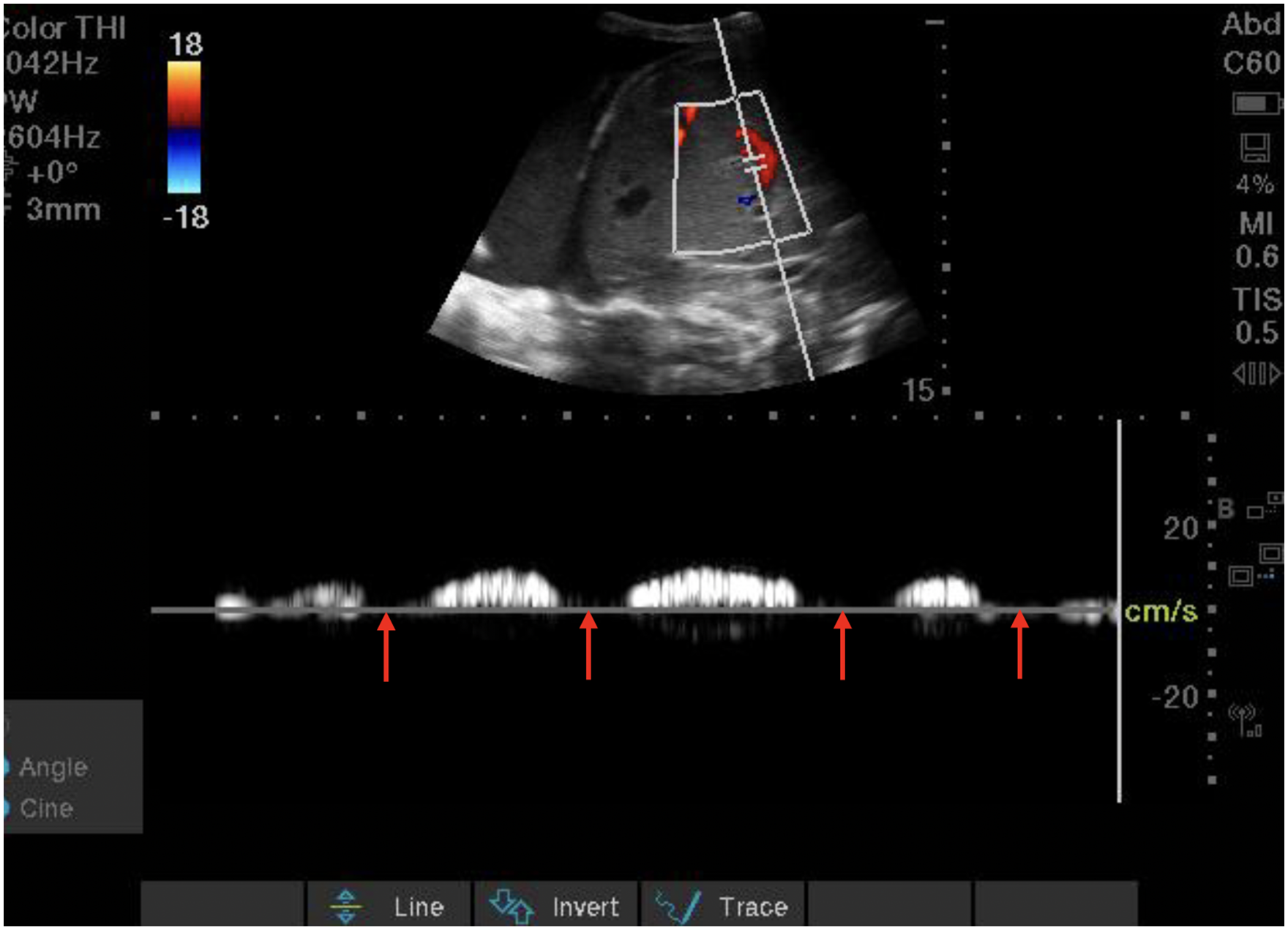
Portal venous Doppler tracing demonstrating 100% pulsatility (severe congestion). Red arrows highlight areas of absent diastolic portal venous flow.
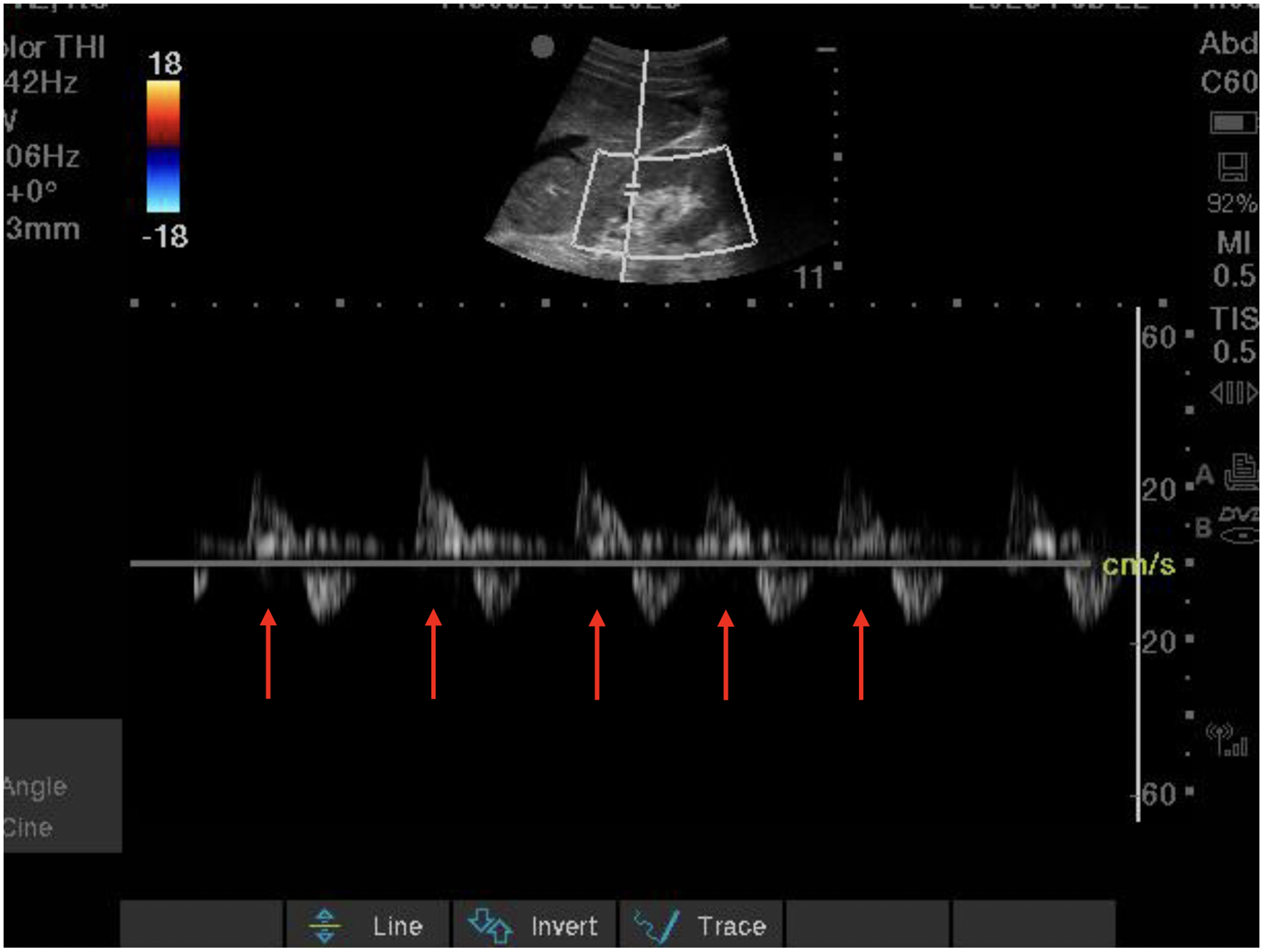
Intra-renal venous Doppler tracing (below baseline) demonstrating severely abnormal diastole-only flow. Red arrows highlight periods of complete absence of venous flow during systole.
In the evaluation of potential renal etiologies of AKI, ultrasound plays a much more muted role. While POCUS may be able to provide clues supporting an intrinsic renal cause, the value of ultrasound in these cases truly lies in ruling out pre- or post-renal etiologies; other nonultrasound parameters such as historical clues, urine analysis and microscopy, and laboratory values will be much more useful. Sonographic abnormalities in renal parenchymal disease may include abnormal renal size, increased cortical echogenicity, and prominent medullary pyramids. 60 However, in a recent study, none of these ultrasound parameters were helpful in detecting renal causes of AKI using multiorgan POCUS. 51 While some propose Renal Resistive Index as a useful tool in these cases, it is affected by a multitude of factors, including hemodynamics, cardiac disease, and underlying renal disease, so its value in this setting is limited. 61
Conclusion
The days of ultrasound as a simple tool for vascular access and compartmentalized single-organ assessments are behind us. POCUS can, and should, be used in a comprehensive, individualized, whole-body approach. As more advanced POCUS applications emerge and our understanding of physiology evolves, we have an increasing number of tools to draw from to help us make these complex assessments.
Within the plethora of POCUS applications discussed above, it takes an experienced clinician to know which parts of the assessment will add value, and which can be safely omitted. An understanding of the pitfalls, caveats, and test characteristics for each exam is essential; and of course, there is no protocol or article that can substitute for good clinical acumen. The beauty of POCUS lies in its scalability; and with an ever-expanding repertoire of techniques at our disposal, whole-body ultrasonography is more helpful than ever in the care of the complex critically ill patient.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
