Abstract
Introduction
Historically, small intestinal bacterial overgrowth (SIBO) has been characterized by an increase in the number and/or alteration in the type of bacteria in the upper gastrointestinal tract (GI). 1 However, over the past decades, our understanding of SIBO has evolved as more knowledge has come to light concerning the intestinal microbiome and its bidirectional interaction with the immune system, the digestion process and metabolism, as well as the gut-brain axis. There is a growing interest in the relationship between the intestinal microbiome and disease. Intestinal microbiome imbalance has been confirmed to be related to several gastrointestinal diseases, such as irritable bowel syndrome (IBS), inflammatory bowel disease, and gastrointestinal tumors. 2 Accumulating data suggest a strong association between IBS and SIBO, as bacterial overgrowth may be part of the IBS pathogenesis.3–5 Subsequently, the treatment of SIBO with nonabsorbable antibiotics has improved symptoms in patients with IBS, 3 as well as celiac disease.6–8 Recently, an association between SIBO and inflammatory bowel disease has also been described. 9 Moreover, SIBO has been investigated as a predisposing factor for spontaneous bacterial peritonitis (SBP) in cirrhotic patients.10–12 Bacterial translocation and hematogenous spread are considered the underlying mechanisms. Furthermore, cardiovascular disease, hypertension, and diabetes mellitus have all been associated with intestinal flora imbalance. 2 Additionally, bariatric surgery has also been associated with gut dysbiosis. Modification of normal gut anatomy after bypass bariatric procedures predisposes to bacterial stasis and consequent development of SIBO, with a reported incidence of 43%. 13
Several diagnostic tests for SIBO are currently available, such as microbial culture in duodenum/jejunum fluid aspirates, urinary excretion tests using chemically synthesized bile acid conjugates, as well as breath tests. 14 Among them, breath tests, most commonly hydrogen (HBT) and/or methane breath tests (MBT), are inexpensive, noninvasive, and safe, with a minimal lag time between testing and result which can easily be performed at the patient's bedside. The rationale behind these tests is based on the principle that carbohydrate fermentation by gut flora is the only source of hydrogen and/or methane in the body. Carbohydrate substrates, such as glucose and lactulose, are metabolized when exposed to gastrointestinal microbes following the consumption of a predefined amount, producing hydrogen and methane. These gases diffuse into the systemic circulation and are exhaled through the lungs, where they can be collected and analyzed. 9
Currently, SIBO is considered a common pathological state of intestinal dysbiosis with a higher prevalence than previously thought. This apparent increase in prevalence has, in part, been attributed to the existence of readily available diagnostic tests. Its development is associated with the disturbance of the normal homeostatic mechanisms controlling the enteric bacterial population.1,15 Bacterial overgrowth occurs when intestinal stasis provides coliform bacteria with the opportunity to proliferate locally. Two conditions that most commonly predispose to bacterial overgrowth are diminished gastric acid secretion and small intestine dysmotility. 15 Both of which may occur in critically ill patients. The inhibition of acid secretion via histamine type 2 receptor antagonists (H2RAs) or proton-pump inhibitors (PPIs) may predispose to SIBO, although the effects of H2RAs and PPIs on breath test results have been contradictory. 16 Furthermore, anatomical alterations and impaired immunity are also well-described risk factors. The relationship between gut bacterial overgrowth and critical illness has been well described. 17 Critical illness and its associated interventions have a detrimental impact on the gut flora, which plays an important role both in health maintenance and recovery by mediating local and systemic immunity. The use of antimicrobials, though fundamental and lifesaving in septic patients, is associated with significant and often long-lasting alterations in the gut flora, which has been linked to poor outcomes in critical illness, raising concerns about the overuse or misuse of antimicrobials in this setting. 18 On the other hand, gut dysbiosis in critically ill patients and its subsequent effect on the brain-gut, lung-gut, and heart-gut axis, among others, is a growing field of interest, but evidence concerning pathophysiology and underlying mechanisms is still scarce. 19
The primary goal of our study was to assess the prevalence of SIBO in mechanically ventilated, critically ill patients. Secondary objectives included investigation of the effect of SIBO on intensive care unit (ICU) length of stay (LOS), ventilator-associated pneumonia (VAP), and all-cause in-hospital mortality rate in critically ill patients. To our knowledge, this is the first study to employ a modified HBT in mechanically ventilated critically ill and initially SIBO-“naïve” adult ICU patients.
Methods
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the local research ethics committee (Decision Number 207) as a prospective noninterventional study and was conducted at the ICU of “G. Hatzikosta” General Hospital of Ioannina, Greece. The ICU of “G. Hatzikosta” General Hospital is an adult 8-bed mixed medical and surgical unit with more than 150 admissions per year. Written informed consent was obtained from the legal guardians of all participants. Clinical trial registration: NCT05765994.
Subjects
All patients initially assessed for eligibility were critically ill patients in need of mechanical ventilation with an expected ICU LOS and duration of mechanical ventilation of ≥48 h. Exclusion criteria included abnormal HBT on the day of ICU admission (Day 1); recent (up to 4 weeks prior to ICU admission) antimicrobial therapy or patients already receiving antimicrobial therapy on admission; recent (up to 4 weeks prior to ICU admission) use of gastroprotective medication, probiotics, and prokinetics; known gastrointestinal disease prior to admission; history of abdominal surgery or recent abdominal surgery resulting in planned or unplanned ICU admission; and patients already hospitalized or institutionalized before ICU admission. For reasons of homogeneity, the HBT was performed only in mechanically ventilated patients. Death or extubation before protocol completion constituted the exclusion criteria after enrollment. A flowchart of patient selection is shown in Figure 1.
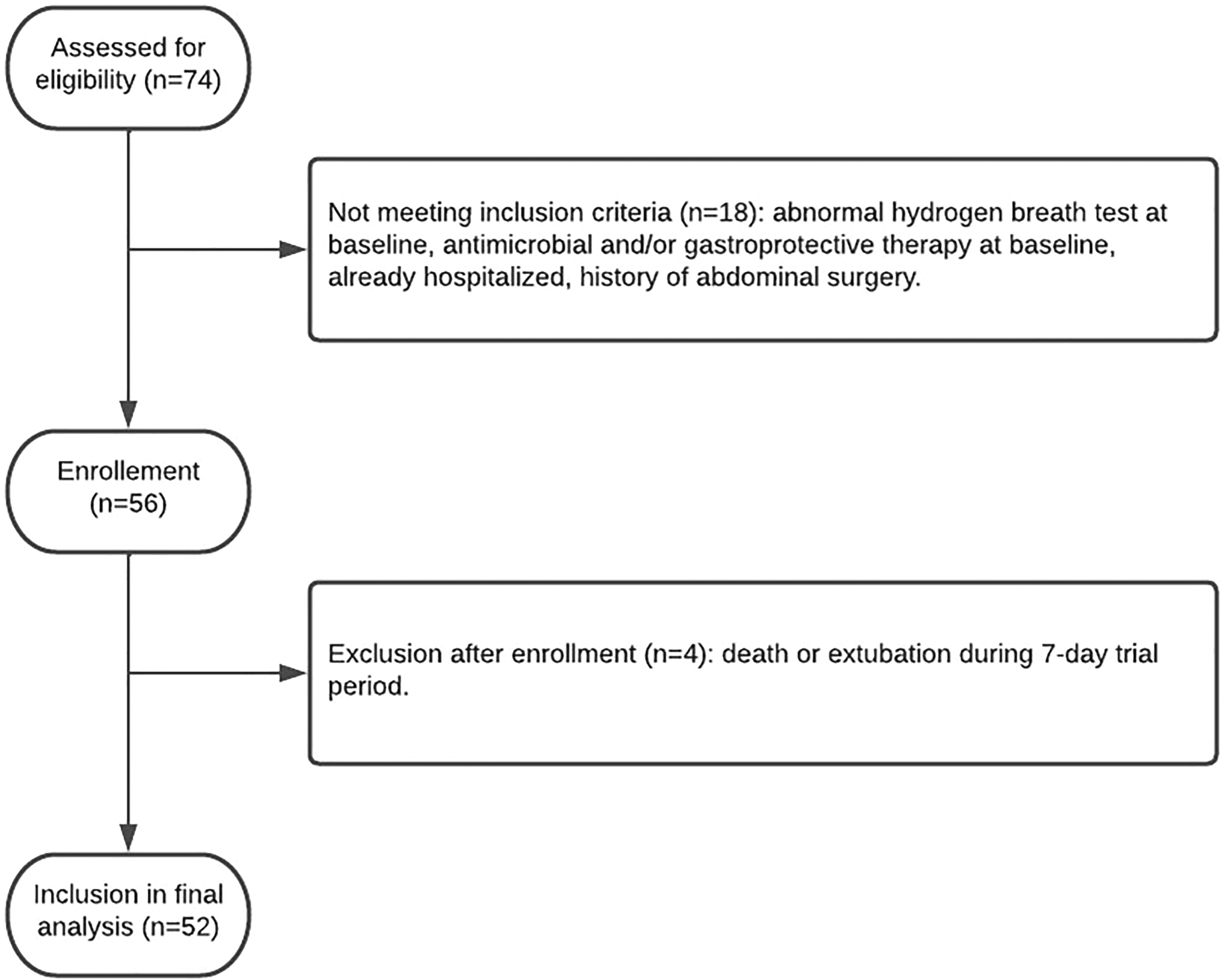
Flow chart of patient selection and recruitment.
Hydrogen Breath Testing Monitor
The hydrogen monitor Gastrolyzer/Gastro +™ (Bedfont® Scientific Ltd) was used for expired hydrogen (eH2) testing. This monitor measures eH2 in parts per million (ppm) but does not measure methane. Calibration was performed every 3 months according to the manufacturer's instructions using a standard gas sample containing 100 ppm eH2. Each test was performed within the stated operating temperature (15 °C–35 °C) and humidity range (25%–80% relative humidity).
Technique
A modified technique to obtain eH2 from ventilator tubes, as described by López et al, was performed. 20 A connection with a valve attached to a collecting bag was inserted at the end of the expiratory limb of the breathing circuit. According to the authors, this designed adaption is useful for collecting breath samples, and it does not alter the patient's ventilation, nor does it interfere with ventilator measurements.
Sampling and Measuring
All tests were performed after proper patient preparation (nil per os for 12 h). Lactulose was used as the substrate. Specifically, 20 g of lactulose diluted in 400 mL of water was administered to each patient through a nasogastric tube. Measurements were performed 0-15-30-45-60-90-120-180 min after lactulose administration. During measurements, the valve was kept open, and the collecting bag attached to the expiratory limb of the breathing circuit was filled with several patient exhalations. After valve closure and bag isolation, air was drawn using a 50 mL polyethylene syringe, which was connected to the valve system to prevent losses and/or contamination. The air was then blown into the breath hydrogen monitor via the D-piece™, and eH2 measurements were recorded.
The HBT was repeated at predetermined, protocol-based time intervals for all participants included in the study; first day of admission and third, fifth, and seventh day of ICU stay. The first day measurements served as a measure of eligibility, and patients exhibiting abnormal values of eH2 on Day 1 were excluded from the study.
Interpretation of Hydrogen Breath Testing Results
An increase of >20 ppm of eH2 from baseline within 90 min was considered an abnormal measurement, suggesting SIBO. Moreover, an increase of more than 12 ppm of eH2 from baseline within the first 30 min followed by a second peak within the next 15 min (double peak) was also considered an abnormal measurement, suggesting SIBO. The second peak, which is absent in healthy subjects, is the diagnostic criterium.
Data Recording
Age, sex, diagnosis, reason for ICU admission, detailed medical and surgical history, APACHE II scores, and length of ICU stay were recorded for all the participants. Legal guardians and patient attendants were thoroughly interviewed regarding any medical condition and medications used by the patients prior to ICU admission. Moreover, the doses of intravenous sedatives, analgesics, muscle relaxants, and vasoactive medications were recorded throughout the patients’ ICU stay. Details concerning enteral nutrition, such as the starting day, amount, and type of formula used, were also recorded. Daily bowel movements were recorded, as well as the number of episodes of vomiting and/or abdominal distention. Lastly, all participants were closely monitored for VAP development using daily chest radiographs, complete blood counts, and microbiological analysis of respiratory secretions.
Statistical Analysis
The Shapiro-Wilk test was performed to test for the normal distribution of continuous variables. The results for all quantitative variables are presented as mean and standard deviation (SD) or as median and interquartile range (IQR), according to normality. All qualitative variables are presented as absolute and/or relative frequencies. The t-test or its nonparametric equivalent, Mann–Whitney U test, was employed for comparison of continuous variables, as appropriate. Fisher's exact test was used to compare categorical variables. Multivariate logistic regression analysis was performed to identify the predictive factors for SIBO. A multivariate logistic regression model was constructed using the stepwise forward method.
All tests were two-tailed and statistical significance was set at 5% (P < .05). Data were analyzed using Stata™ (version 10.1 MP, Stata Corporation, College Station, TX 77845, USA).
Results
Patient recruitment was conducted over a 10-month period. Seventy-four consecutive patients were initially assessed for eligibility, and 52 patients were included in the final analysis (Figure 1). Participants were classified as SIBO positive or negative according to the results of the HBT on the third, fifth, or seventh day of ICU stay. A comparative assessment of demographic characteristics, APACHE II score, incidence of VAP, duration of ICU LOS, and all-cause in-hospital mortality was conducted.
The cohort was homogenous in terms of demographics, ICU diagnosis, reason for admission, and disease severity according to APACHE II score (P > .05; Table 1). The prevalence of SIBO was 36.54%. A SIBO-positive diagnosis was associated with prolonged ICU LOS (P = .033; Table 2). Moreover, all patients diagnosed with VAP were SIBO-positive (P < .001; Table 2). The all-cause in-hospital mortality rates were similar between the groups (P > .05; Table 2).
Baseline Cohort Description.
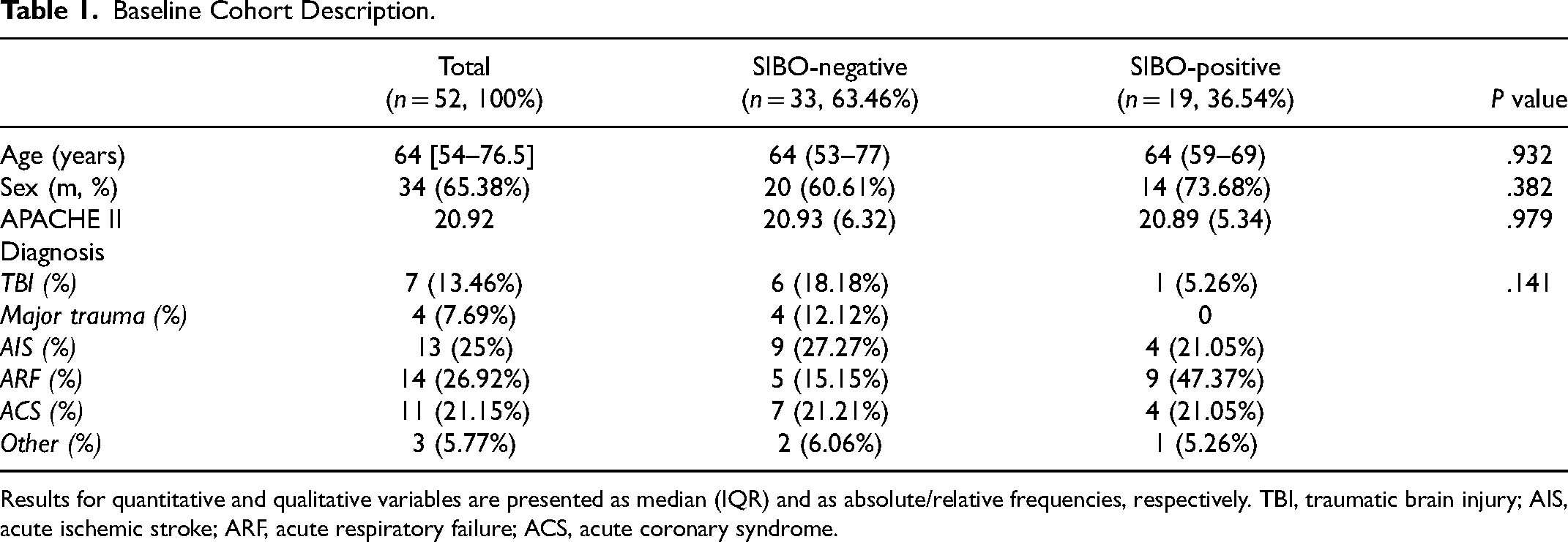
Results for quantitative and qualitative variables are presented as median (IQR) and as absolute/relative frequencies, respectively. TBI, traumatic brain injury; AIS, acute ischemic stroke; ARF, acute respiratory failure; ACS, acute coronary syndrome.
Length of Stay, Antimicrobial Therapy, and Outcome.
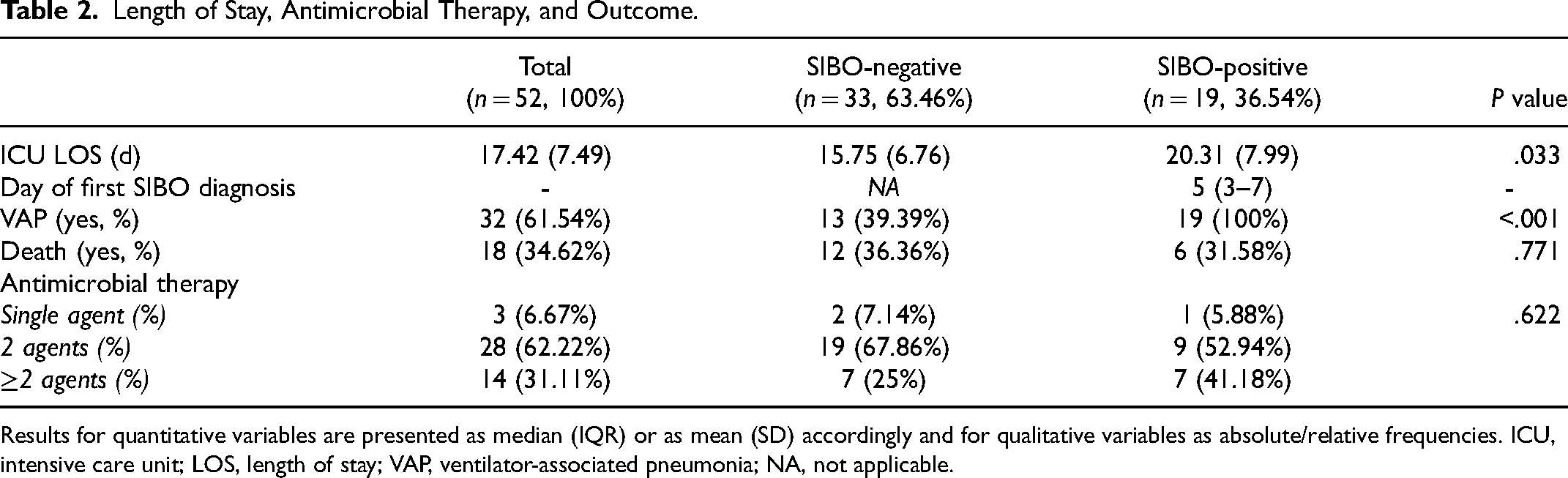
Results for quantitative variables are presented as median (IQR) or as mean (SD) accordingly and for qualitative variables as absolute/relative frequencies. ICU, intensive care unit; LOS, length of stay; VAP, ventilator-associated pneumonia; NA, not applicable.
Table 3 summarizes the results of the multivariate logistic regression analysis. SIBO served as the dependent variable, whereas sex, age, ICU LOS, APACHE II score, diagnosis of VAP, and antimicrobial regimen (as a categorical variable of 1, 2, or more than 2 agents) served as the independent variables in the stepwise forward procedure. Among the above-mentioned variables, only age was identified as a statistically significant independent predictor of SIBO (OR 1.08; P = .018).
Multivariate Logistic Regression Model of Factors Predicting SIBO.
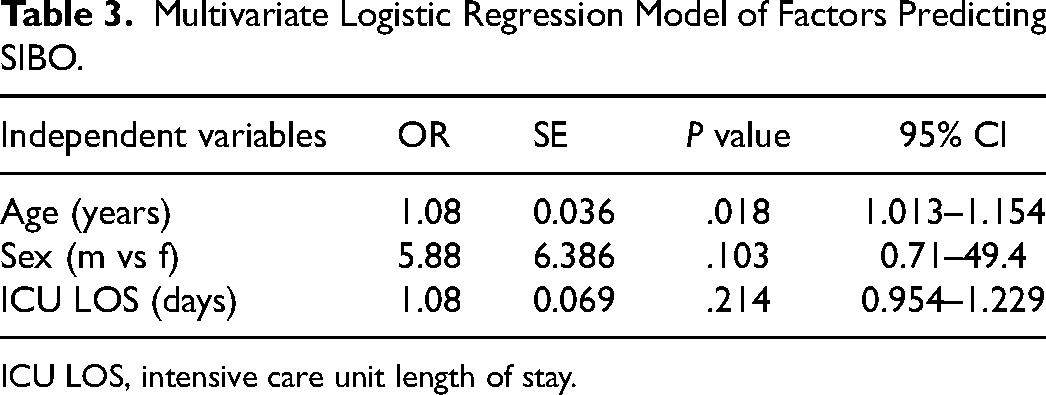
ICU LOS, intensive care unit length of stay.
Discussion
In this prospective observational study, we highlight a high prevalence (36.5%) of SIBO in critically ill mechanically ventilated ICU patients. SIBO diagnosis was based on a noninvasive modified technique for sampling exhaled air from the ventilator tubes and performing a standard ΗΒΤ. Although all-cause in-hospital mortality did not differ between SIBO-positive and SIBO-negative patients, SIBO was strongly associated with the presence of VAP and prolonged ICU LOS.
Currently, there are no universally accepted criteria for SIBO diagnosis. The gold standard for the diagnosis of SIBO is microbial culture of duodenum/jejunum fluid aspirates, 14 which is an invasive procedure that poses a potential risk to patients. On the other hand, urinary excretion tests using chemically synthesized bile acid conjugates have also been employed for SIBO diagnosis, but there is a paucity of evidence in literature concerning their use, especially in critically ill patients. 14 Alternatively, breath tests of hydrogen/methane gases after ingestion of carbohydrates have been widely used for the diagnosis of functional gastrointestinal disorders, including SIBO and carbohydrate malabsorption, as well as for measuring orocecal transit time (OCTT). 21 Several substrates have been studied, but none has been identified as being superior to the other. 15 The lactulose hydrogen breath test (LHBT) is the most widely used, 1 with the first report of an increase in hydrogen level after lactulose ingestion going back to 1972. 22 In our hospital, lactulose is the regular substrate to be tested.
Although HBT are widely used, their reliability and accuracy are controversial. According to Perrman et al, 23 elevated fasting expired hydrogen (>19 ppm) and methane (>10 ppm) are considered excellent predictors of overgrowth, being highly specific (>90%), but not sensitive (<30%). Corazza et al 24 reported sensitivities of 60% to 70% and specificities of 40% to 80% for glucose and lactulose breath tests, respectively. However, validation with microbial culture is limited. 25 Another study advocated that glucose and lactulose breath tests, when compared to culture of an endoscopic small bowel aspirate, have lower sensitivities (glucose 44%, lactulose 31%) but higher specificities (glucose 80%, lactulose 86%) than previously thought. 26
Although HBT are easy to perform, they are difficult to interpret. As a rule of thumb, a more than 10 to 12 ppm increase following glucose load27,28 and a more than 20 ppm increase following lactulose load 29 from baseline fasting hydrogen concentration during the first 90 min of the test have been accepted as proof of SIBO. Moreover, a double peak in eH2 has also been accepted as a proof of SIBO. This consists of an initial hydrogen peak prior to 90 min, followed by a fall of more than 5 ppm over 2 consecutive samples, and then by a second peak in eH2 as the substrate enters the cecum. 30 However, great diversity exists in what constitutes a positive HBT among clinical trials. These issues were finally addressed by the Hydrogen Breath Test—Rome Consensus 31 and the Hydrogen and Methane Breath Test in the Gastrointestinal Disease—North American Consensus 32 where specific criteria for SIBO diagnosis were set. An increase in eH2 of ≥20 ppm by 90 min during glucose or lactulose breath tests is considered a positive HBT for SIBO diagnosis, whereas a methane concentration increase of ≥10 ppm at any time constitutes a positive MBT. In our experiment, we expanded our diagnostic criteria and included the “double peak.” This is because, unlike glucose, lactulose is a poorly absorbed disaccharide that reaches the cecum. When fermented by the contaminating bacterial flora in the small intestine, there is hydrogen production, but LHBT may show 2 distinct hydrogen excretion peaks in individuals suffering from SIBO; the first “early” peak due to the small bowel microflora activity and the second “late” peak due to the colonic bacterial metabolism. 32
The composition of the intestinal microflora is influenced by several factors, such as age, dietary habits, immunological factors, etc. 33 Older age has been identified as a risk factor for SIBO in patients with Crohn's disease. 9 In our cohort of critically ill patients, age was also identified as a statistically significant independent factor predicting SIBO (OR 1.08; P = .018). Nineteen of our participants (36.54%) were SIBO-positive, 3 were under the age of 50 years (15.79%), and 16 (84.21%) were older. Several studies have reported a high prevalence of SIBO among the elderly; Lewis et al 34 reported a SIBO prevalence of 14.5%, Parlesak et al 27 15.6%, and almost 3 times greater than that in younger adults, whereas Mitsui et al 35 found a SIBO prevalence of 33% in disabled older adults. The prevalence of SIBO in our study was similar to that reported by Mitsui et al. Although age-associated decline in GI motility seems a reasonable explanation, there are studies failing to show this decrease in GI motility with age.36,37 The onset of new diseases (eg, diabetes), dietary changes that lead to malnutrition, and changes in gut immune function have also been implicated. 15 Moreover, elderly patients may be especially susceptible to SIBO because of both a reduction in gastric acid production and the consumption of a disproportionately large number of drugs that can cause GI hypomotility. 38
Of note, the prevalence of SIBO varies substantially in the literature and is influenced by the specific characteristics of the study population, as well as the diagnostic method used to detect or define bacterial overgrowth. 1 The prevalence of SIBO in IBS varies from 30% to 85%;25,39,40 in liver cirrhosis 41 and in celiac disease, 42 it is reported to be 50%, whereas in asymptomatic morbidly obese patients 17%. 43 To our knowledge, this is the first study reporting SIBO prevalence in critically ill mechanically ventilated patients.
VAP is the most frequent infection in mechanically ventilated patients, and upper intestinal tract colonization appears to be implicated in its pathogenesis. Critical illness drastically changes the dynamics of the aerodigestive tract, where bacterial translocation from the gut to lungs is enhanced. This is an inverted process compared to that in healthy subjects, where the oropharynx is the primary source of microbiota in the lungs and stomach. In critical illness, bacterial overgrowth in the stomach and small intestine becomes the primary microbiota source for the oropharynx and lungs.44–46 In septic patients, the lungs are dominated by gut-associated bacteria, and this shift in lung microbiota is now appreciated to be a long-lasting consequence. 47 Ecological analysis revealed the gut is the likely source of bacterial dysbiosis in the lung that can lead to VAP and ARDS.44,45 In our cohort of critically ill patients, we found a strong association between VAP and SIBO, and all patients diagnosed with VAP were also SIBO-positive (P < .001; Table 2).
Our study presents certain limitations. First, the results of the LHBT were not validated by microbial culture, which is the gold standard for diagnosing SIBO. Moreover, the follow-up period was short, breath tests were not repeated after ICU discharge and long-lasting consequences of our SIBO-positive participants were not investigated. Finally, death or weaning from mechanical ventilation within the 7-day trial period led to exclusion from the study, but the number of patients that were excluded after enrollment was small (Figure 1), and we believe that this does not have an impact on the estimation of the overall prevalence.
Conclusion
In conclusion, this study confirms the high prevalence of SIBO in mechanically ventilated, critically ill patients. The SIBO-positive patients were older, had a prolonged ICU stay, and all developed VAP, while the all-cause in-hospital mortality did not differ from that of their SIBO-negative counterparts. However, the clinical implications of our findings warrant further investigation.
Footnotes
Author Contributions
The study conception and design (AK, KB, AM
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
