Abstract
Introduction
Adult men are at increased risk of developing and dying from serious infections and sepsis compared to women, with differential effects by age group, era, and region.1–3 Multiple mechanisms have been proposed for this difference, with differential immune activation and pathogen clearance between men and women related to sex hormone levels, the X chromosome, immune activation, and other factors.4–10 In children, sex differences in sepsis susceptibility have been shown to be age-dependent.11–14 Infant and adolescent males have an increased risk of mortality in some studies with no difference in prepubertal children.11,15 It is not clear whether male children have an increased risk of adverse outcomes, including mortality or persistent decline in health-related quality of life (HRQL), after community-acquired septic shock, or if age plays a role in differential susceptibility to adverse outcomes.
The Life After Pediatric Sepsis Evaluation (LAPSE) was a multicenter prospective cohort study that characterized survival and HRQL in children for 12 months following community-acquired septic shock. 12 Many children in LAPSE had persistent decline in HRQL at 3 months after septic shock, which was mediated by acute organ failure severity. 13 The NIH has made the consideration of sex as a biological variable in research design, analysis, and reporting a research priority. 14 We performed this secondary analysis of the LAPSE cohort to evaluate for sex-based differences in the clinical course and outcomes of community-acquired septic shock in children of different age groups. Our primary hypothesis was that male children would have an increased risk of persistent decline in HRQL or mortality compared to female children and that this effect would differ by age group, with the highest risk in male infants and adolescents. Our secondary hypothesis was that male infants and adolescents would have increased acute severity of illness compared to female infants and adolescents.
Methods
Design and Setting
This was a secondary analysis of the LAPSE cohort. The design, methodology, and primary results of the LAPSE trial have been previously reported. 16 Briefly, LAPSE was a multicenter, prospective, cohort study of children with community-acquired septic shock. LAPSE was performed at 12 academic pediatric intensive care units (PICU) in the United States, recruiting patients from January 1, 2014, to June 30, 2017. The study was approved by the Institutional Review Board (IRB) at all sites and the University of Utah Data Coordinating Center. Parental permission was obtained for all participants. This secondary analysis used only de-identified data collected for the primary study, and additional IRB approval was not required.
Participants
The LAPSE trial enrolled children who were >1 month and <18 years old with community-acquired septic shock. Eligible children had documented or suspected infection within 48 h of hospital admission; presence of 2 or more systemic inflammatory response syndrome criteria, 17 at least 1 of which was abnormal temperature or leukocyte count; cardiovascular dysfunction with requirement for vasoactive or inotropic support; and pulmonary dysfunction with invasive or noninvasive ventilation. For this secondary analysis, sex was determined by parental report. Children were stratified into age groups of <1 year old, 1 to <13 years old, and 13 to <18 years old.
Data Collected
In the primary study, baseline (reflecting prehospitalization status), hospitalization, and long-term mortality and HRQL data were collected. Baseline data included demographics, functional status, HRQL, immunocompetency status, and chronic comorbid conditions. Demographic data included age, sex, race, ethnicity, household income, parental education, and health insurance type. Chronic comorbid conditions were defined using the Pediatric Medical Complexity Algorithm, which categorizes patients into no chronic conditions, noncomplex chronic conditions (lifelong but episodic and with periods of health and not progressive), and complex chronic comorbid conditions (involving 2 or more body systems, progressive and life-limiting, technology-dependent, or active malignancy) using ICD codes. 18 For this analysis, patients were categorized as none/noncomplex chronic comorbid conditions and complex chronic comorbid conditions. Baseline functional status was assessed using the Functional Status Scale™ (FSS), 19 which measures function across 6 domains (mental status, communication, feeding, motor, sensory, and respiratory). Immune competency was assessed by medical record review using a predetermined list of diagnoses as previously described. 16
Hospitalization data included severity of acute organ dysfunction, treatment intensity, and evidence of hyperinflammation or immune suppression. We quantified overall severity of organ dysfunction using the Pediatric Logistic Organ Dysfunction (PELOD) scale-2. 20 PELOD is a validated composite measure of pediatric organ dysfunction that is correlated with mortality. Composite organ dysfunction was described using the sum of PELOD, with data collected daily for the duration of ICU admission or truncated at 28 days, whichever came first. The total score for the ICU admission was summed to derive 28-day PELOD, which has been previously validated as a measure of composite organ dysfunction. 21 We described severity of shock using the maximum and sum of the vasoactive-inotropic scores (VIS, assessed twice daily) over the first 72 h of PICU admission, 22 VIS is a numeric representation of all administered hemodynamic support and is correlated with shock severity and mortality. We also described the duration of hemodynamic support. We described treatment intensity as the need for ECMO, continuous renal replacement therapy, endotracheal intubation, or central venous line placement. We also reported hospital and PICU length of stay.
HRQL was measured at baseline and 1, 3, 6, and 12 months after admission. Baseline data was collected by parental questionnaire when patients were enrolled in the study and measured prehospitalization HRQL. HRQL was measured using the Pediatric Quality of Life Inventory™ (PedsQL) 4.0 Generic Core Scales, 23 PedsQL™ Infant Scales, 24 or the Stein-Jessop Functional Status Scale-short form (FS-II-R). 25 The PedsQL™ assesses the physical, emotional, social and school functioning domains of HRQL. The PedsQL™ Infant scales assess physical symptoms and physical, emotional, social, and cognitive function. The FS-II-R short form is a measure of behaviorally based functional status applicable across the pediatric age span. The FS-II-R addresses communication, mood, energy, eating, sleep, responsiveness, and attention and may be more appropriate for children with severe developmental disability than the PedsQL™. Parents chose an age-appropriate PedsQL scale or the FS-II-R for their child based on which instrument they believed provided the most meaningful assessment for their child. All scales were completed by parent proxy.
We defined a clinically meaningful and persistent decline in HRQL 12 as a decrease of 10% from baseline at 3 months measured using the PedsQL™ or FS-II-R scale. The minimum clinically important difference (MCID) for the PedsQL™ is 4.5 points on the scale from 0 to 100. 23 The mean PedsQL™ score for previously healthy children is 82.3 ± 15.6 (mean ± SD) 23 and is 73.1 ± 16.5 for children with chronic diseases. A decline of 10% for previously healthy children would represent 8.2 points or nearly twice the MCID and 0.5 standard deviations below the mean. For children with chronic diseases, a 10% decline would be 7.3 points or about 1.5 times the MCID and 0.4 standard deviations below the mean. The MCID for the FS-II-R scale has not been reported. This scale is also scored from 0 to 100. The mean FS-II-R score for previously healthy children is 96.1 ± 8.2 25 and is 86.8 ± 15.7 for chronically ill children. A decline of 10% would therefore represent 9.6 points, or 1.2 standard deviations, for healthy children, and 8.7 points, or 0.6 standard deviations, for chronically ill children. We chose the 3-month time point to determine a clinically meaningful and persistent decline in HRQL because in the primary LAPSE trial, HRQL did not significantly change between 3 and 12 months although dropout was substantial. 16
Statistical Analysis
Prehospitalization characteristics for males versus females were summarized with counts and percentages for categorical variables and with medians and quartiles for continuous variables (Table 1). Subgroups included infants (<1 year), children (1–<13 years), and adolescents (13–<18 years). Differences between males and females were evaluated with Fisher's exact test and the Wilcoxon rank-sum test as appropriate. Measures of acute illness severity, treatment intensity, and length of stay were similarly summarized and compared between the three subgroups (Supplemental Material 1). 3-month mortality was presented and analyzed in each age group and overall (Table 2). Fisher's exact test was used to evaluate differences between males and females in each subgroup. The primary analysis was performed using the Cochran-Mantel-Haenszel test to pool differences between males and females across individual subgroups for an “overall” comparison of males versus females that is not confounded by age (Table 2). 1-month, 6-month, and 12-month mortality was analyzed analogously (Supplemental Material 2). Finally, a multivariable logistic regression model was created to assess the association of sex with death or persistent decline in HRQL at month 3 (Table 3) as a sensitivity analysis. The covariates for the multivariate model were selected a priori and included age, prehospitalization HRQL, immunocompromised status, and complex chronic condition. An interaction was included between age and sex to account for different associations between sex and persistent decline in HRQL within each age group. Analyses were performed using SAS 9.4 (SAS Institute, Cary, NC) with reported p values based on a 2-sided alternative and considered significant if <0.05.
Prehospitalization Child and Caregiver Characteristics.
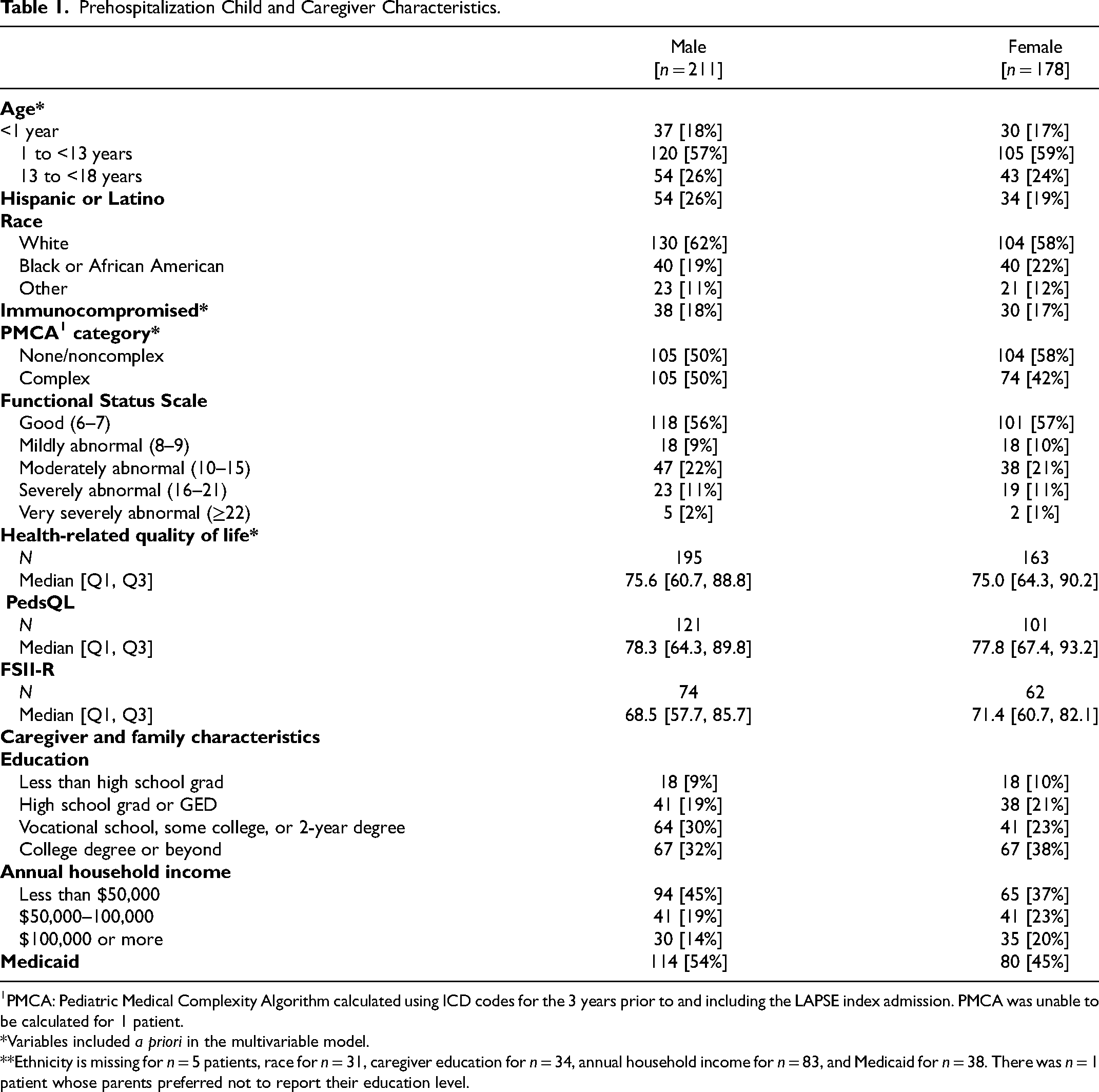
PMCA: Pediatric Medical Complexity Algorithm calculated using ICD codes for the 3 years prior to and including the LAPSE index admission. PMCA was unable to be calculated for 1 patient.
*Variables included a priori in the multivariable model.
**Ethnicity is missing for n = 5 patients, race for n = 31, caregiver education for n = 34, annual household income for n = 83, and Medicaid for n = 38. There was n = 1 patient whose parents preferred not to report their education level.
Mortality and Persistent Decline in HRQL.

Cochran–Mantel–Haenszel test of general association, stratified by age.
Fisher's exact test.
Patients lost to follow-up for outcomes including HRQL are not included in denominators for percentages.
Multivariable Model of Predictive Baseline Factors for Persistent Decline in HRQL or Death at 3 Months in Children with Community-Acquired Septic Shock.
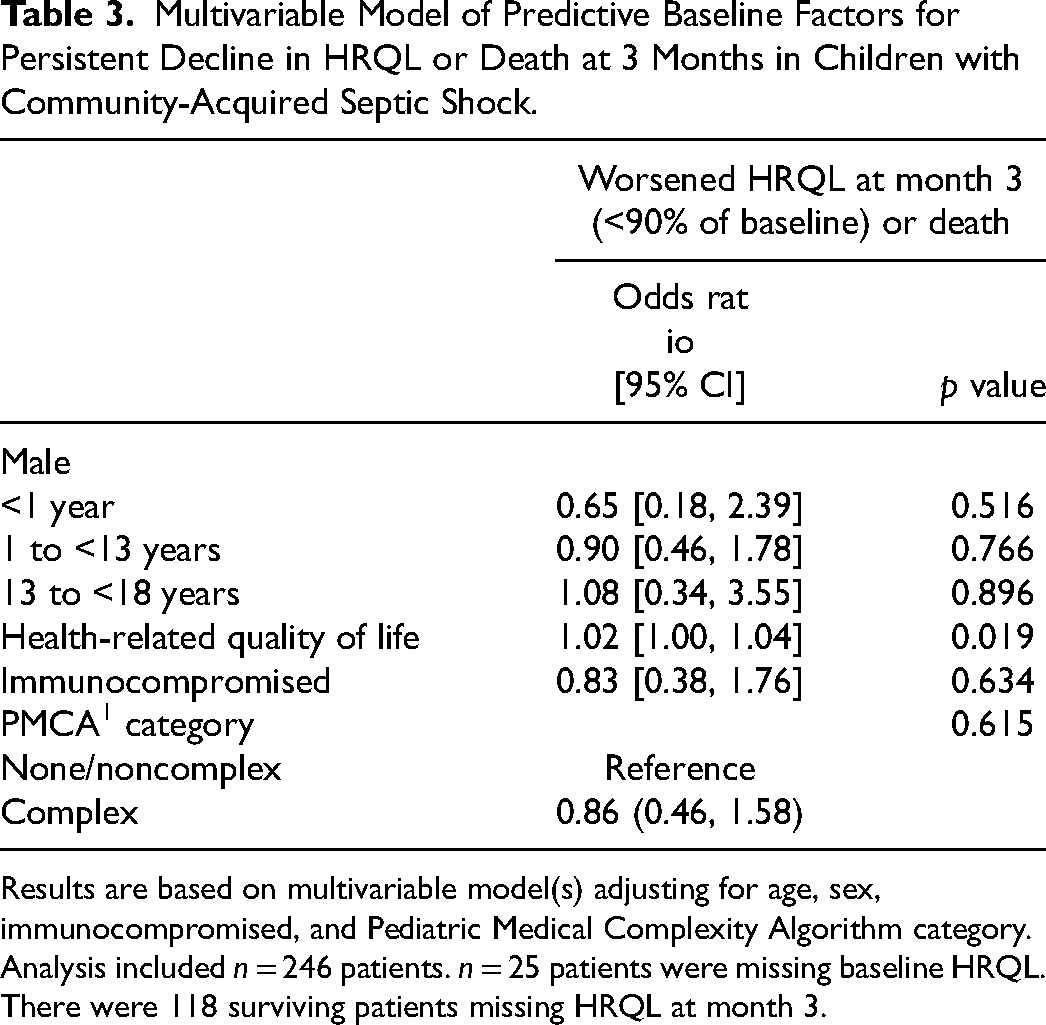
Results are based on multivariable model(s) adjusting for age, sex, immunocompromised, and Pediatric Medical Complexity Algorithm category.
Analysis included n = 246 patients. n = 25 patients were missing baseline HRQL. There were 118 surviving patients missing HRQL at month 3.
Results
Characteristics of the Cohort and Hospital Course
Of the 389 children in the LAPSE study, 54.2% (n = 211) were male. Baseline characteristics including race, ethnicity, caregiver education, and annual household income were similar between male and female children (Table 1). The age distribution was also similar for male and female children. Overall, 17.1% (n = 67) of the cohort were <1 year old, 57.8% (n = 225) were 1 to <13 years old, and 24.9% (n = 97) were 13 to <18 years old. A slightly higher proportion of males (105/211, 50%) than females (74/178, 42%) had a preexisting complex chronic condition. Baseline HRQL and functional status were similar between male and female children. Overall, 92.0% (358/389) of parents completed baseline HRQL data for their children, with similar proportions between male and female children (Table 1). There were no differences in acute inflammation, organ dysfunction, treatment intensity, or PICU or hospital length of stay between male and female infants, children, or adolescents (Supplemental Material 1).
Mortality and Decline in HRQL
In the primary analysis, there were no differences between male and female infants, children, or adolescents in mortality or decline in HRQL at 3 months among those with available data (n = 246, Cochran-Mantel-Haenszel test p = 0.636) (Table 2). Overall, 41% (57/138) of males and 44% (48/108) of females had a persistent decline in HRQL or died. In analysis of individual outcomes, 10% (21/211) of males and 12% (22/178) of females died by 3 months (p = 0.454). Among survivors with HRQL data at 3 months (n = 203), 31% (36/117) of males and 30% (26/86) of females had persistent decline in HRQL (p = 0.861). Decline in HRQL or mortality was not different between males and females when these outcomes were considered at the other follow-up time points (Supplemental Material 2).
Multivariable Model
The multivariable model secondary analysis analyzing the association of factors present at hospital admission with the outcome of persistent decline in HRQL or death at 3 months is shown in Table 2. Covariates selected for analysis a priori include baseline HRQL, immunocompromised status, and medical complexity as well as sex and age. Children with a higher baseline HRQL were more likely to have persistent decline in HRQL or death at 3 months after admission (OR 1.02, 95% CI 1.00-1.04, p = 0.019). Infant (OR 0.65, 95% CI 0.18-2.39, p = 0.516) and adolescent males (OR 1.08, 95% CI 0.34-3.55 p = 0.896) did not have an increased risk of this adverse outcome compared to females. No other baseline factors were significantly associated with decline in HRQL or death at 3 months after admission.
Discussion
Contrary to our primary hypothesis, we found no difference between male and female children when accounting for age group in mortality or persistent decline in HRQL after community-acquired septic shock. This finding was consistent in the primary pooled univariate analysis and the secondary multivariable analysis. There was no difference in severity of illness based on acute organ dysfunction, treatment intensity, excessive inflammation, immune suppression, or severity of shock.
In adults with sepsis, data regarding sex differences in mortality and their relationship with age are conflicting. A large propensity-matched study found that men had a higher risk of mortality despite similar presenting severity of illness, 1 which was driven by reduced mortality in postmenopausal women compared to age-matched men. A more recent study using the MIMIC-III dataset 3 found an increased risk of in-hospital and 1-year mortality following ICU admission for sepsis in men, with 3% to 4% excess risk depending on the time point used for analysis. Other evaluations have found an increased risk of mortality in women with severe sepsis in the adult medical and surgical ICU population 26 and in women who develop nosocomial infection after ICU admission. 27
Differences in mortality between adult men and women after sepsis vary with age, 28 suggesting a possible role for sex hormones 29 in susceptibility to adverse outcomes in addition to many other factors. 3 Animal models have also shown sex hormone-independent differences with differential susceptibility to late infection attributable to changes in the microbiome 30 and genetic differences in early clearance and mortality. 31 Overall, adult and preclinical data suggest that sex differences in outcomes after septic shock are multifactorial, with important differences in immunity and pathogen clearance resulting in a heterogeneous effect of sex that differs by age, comorbid conditions, sex hormones, and many other factors. This heterogeneity may be one reason for our finding of no difference in mortality or HRQL between male and female children after community-acquired septic shock. Three recent systematic reviews28,32,33 concluded that the effect of sex on mortality after sepsis is unclear, with the most recent review concluding that there may be a small increase in 1-year sepsis mortality for men. 28 In our study, we found no difference in 1-year mortality between males and females; however, this may be related to the low overall mortality rate for children and loss to follow-up in the LAPSE study.
In children, prior data suggest that male infants and adolescents are at increased risk of death from septic shock, suggesting that some difference may be mediated by sex hormones. An analysis using data from the Virtual PICU Systems (VPS) database. 11 found a substantially increased risk of mortality following sepsis in male adolescents and young adults (16-21 years old) compared to females, with no differences observed in prepubertal children (2-7 years old). The VPS study was larger than our study, with 1020 subjects, and found a 2-fold increased mortality in male compared with female adolescents (11.8% vs 5.6%). We did not observe a difference in mortality between male and female adolescents in our study; however, our adolescent group was younger overall (13-<18 years) and may have included some prepubertal children. The LAPSE study also included a small number of adolescents (n = 88) and may have been underpowered to detect a meaningful difference in this population. Additionally, nearly one-third of the mortality in the VPS database occurred within 24 h of hospital admission. The VPS database is a registry including all PICU admissions from participating hospitals. Because of the prospective design of LAPSE and the need for parental permission to participate, children who died of refractory shock early in their hospital course were unlikely to have been enrolled. Thus, the VPS mortality data likely reflect a broader population of children who die from sepsis at any time during their hospitalization, whereas our LAPSE mortality data likely reflect the population of children with sepsis who survive their initial shock but die somewhat later during their course.
Among neonates, male sex is a risk factor for mortality in early- and late-onset sepsis, 15 with most patients dying within 72 h of sepsis diagnosis. This population of infants who die within 72 h of sepsis diagnosis was also unlikely to have been enrolled in the LAPSE study. An older study of children with sepsis and purpura (enrolled 1988–2006) 34 found no difference in mortality rate between prepubertal male and female children. Unlike the VPS dataset, acute illness severity was worse in prepubertal males. Overall, prior pediatric data suggests that mortality and illness severity may be higher in adolescent and infant males but is likely concentrated in patients who die early of overwhelming infection with refractory shock, a population unlikely to have been enrolled in the LAPSE study.
As survival after septic shock has improved in recent decades, the pediatric critical care community and families of children who have experienced critical illness have focused on measuring and improving HRQL after septic shock and other critical illnesses. 35 Understanding risk factors for adverse outcomes after septic shock requires inclusion of patient-centered outcomes besides mortality. The long-term burden of critical illness is substantial in children, with many survivors experiencing persistent decline in their HRQL as demonstrated in the primary analysis of the LAPSE trial 16 and other cohorts of children with other critical illnesses.36,37 Prior studies in adults have found a small or absent effect of sex on HRQL after sepsis.38,39 To our knowledge, this is the first study describing the effect of sex on persistent decline in HRQL after septic shock in children. We did not find a significant difference in this outcome between male and female children.
Limitations of this secondary analysis include a study sample selected for the primary LAPSE trial, which did not recruit children who died early after admission from refractory shock. Based on prior studies, children who die early in their course with refractory shock may be disproportionately male. The primary study was designed to detect a decline in HRQL after septic shock and not to evaluate sex-based differences in decline in HRQL and mortality. Therefore, it is possible that our secondary analysis may have a Type II error if a small difference was present. However, we were unable to identify even a trend in our data suggesting that infant and adolescent males have worse outcomes compared to females. Additionally, the original LAPSE study had a significant number of patients who were lost to follow-up at 3 months after enrollment. These patients were disproportionately previously healthy with higher illness severity. This is a potential source of bias in this secondary analysis. Finally, as a secondary analysis, our study was limited to variables and outcomes collected as part of the primary study. There may be additional predictive variables that are importantly different by sex in pediatric septic shock that were not collected.
The strengths of this study include the prospective, multicenter design of the LAPSE trial. Detailed information about baseline and hospitalization characteristics, as well as longitudinal HRQL, were collected. This allowed for thorough investigation of sex-specific differences in organ failure and treatment intensity as well as mortality and HRQL. The longitudinal nature of the LAPSE study facilitated robust characterization of patient-centered long-term outcomes stratified by age and sex after pediatric community-acquired septic shock.
Conclusions
In this study of children with community-acquired septic shock, health-related quality of life and mortality were not different between male and female children of any age. There were no significant sex-based differences across multiple measures of illness severity or treatment intensity. Given the conflicting prior literature in children and adults, this negative finding adds to the literature by suggesting that male and female children who survive early refractory septic shock do not have significant differences in short-term severity of illness or long-term outcomes. Differences based on sex in pediatric community-acquired septic shock may be more related to differences in infection susceptibility and early mortality than differences in outcome after initial resuscitation.
Supplemental Material
sj-docx-1-jic-10.1177_08850666231190270 - Supplemental material for Association of Sex and Age with Mortality and Health-Related Quality of Life in Children with Septic Shock: A Secondary Analysis of the Life After Pediatric Sepsis Evaluation
Supplemental material, sj-docx-1-jic-10.1177_08850666231190270 for Association of Sex and Age with Mortality and Health-Related Quality of Life in Children with Septic Shock: A Secondary Analysis of the Life After Pediatric Sepsis Evaluation by Andrew J. Prout, Russell K. Banks, Ron W. Reeder, Jerry J. Zimmerman and Kathleen L. Meert in Journal of Intensive Care Medicine
Footnotes
Acknowledgments
The authors would like to thank the Data Coordinating Center at the University of Utah for their logistic support.
Author Contributions
AJP, KLM, and JJZ have given substantial contributions to the conception or the design of the manuscript. RKB and RWR contributed to the acquisition, analysis, and interpretation of the data. All authors have participated in drafting the manuscript. KLM and JJZ revised the manuscript critically. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by grant R03HD104001 from the National Institute of Child Health and Human Development of the National Institutes of Health to Ron Reeder. The authors report no involvement in the research by the sponsor that could have influenced the outcome of this work.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
