Abstract
Background
Septic shock is a common cause of intensive care unit (ICU) admission and one of the major causes of death among ICU inpatients. Septic shock is characterized by systemic vasodilatation and vascular leakage arising from systemic inflammation induced by serious infection. An essential step in the management of patients with septic shock is to increase systemic and regional/microcirculatory flow. 1 Increasing arterial blood pressure with vasopressors when patients are hypotensive improves the input pressure that drives organ perfusion. Norepinephrine is the first-choice vasoactive drug for patients with septic shock because it maintains vascular tension and achieves target mean arterial blood pressure (MAP). 2 A double-blind randomized controlled trial revealed that norepinephrine administration at the beginning of sepsis with hypotension resuscitation was associated with a higher shock control rate by 6 h than the standard treatment. 3 The norepinephrine dose was based on the MAP that was determined to safeguard organ perfusion. However, excessive norepinephrine dosing is associated with the risk of extreme vasoconstriction, tissue hypoperfusion, and increased mortality. 4
The peripheral vascular bed is the first place where blood flow is sacrificed and the place where it is finally perfused. The peripheral perfusion index (PI) reflects pulsatile flow, which demonstrates the ability of circulation to provide blood perfusion to the tissue. 5 The greater the pulsatile flow, the greater the pulsation intensity and the PI value. Therefore, tissue perfusion can be reflected by PI, which acts as an ongoing monitor of local blood flow fluctuations. 6 Since norepinephrine affects blood flow, we aimed to determine whether norepinephrine use during the management of patients with septic shock affects the PI and patient outcomes.
Methods
Patient Sample
Using the administrative database of Peking Union Medical College Hospital, we performed a retrospective study of patients with septic shock who had undergone Pulse index Continuous Cardiac Output (PiCCO)-Plus cardiac output (CO) monitoring. All adult patients within 24 h after the onset of severe sepsis or septic shock who were admitted to the Department of Critical Care Medicine of Peking Union Medical College Hospital between January 2014 and December 2018, who required only a single vasoactive agent (norepinephrine) during the first 24 h after PiCCO initiation, and who underwent mechanical ventilation for resuscitation, were eligible for the study. Patients who were younger than 18 years or who were admitted to the intensive care unit (ICU) for <24 h were excluded. The Institutional Research and Ethics Committee of Peking Union Medical College Hospital approved this study (No. S-K980). All patients were involved in the study based on the voluntary principle, with informed consent obtained from all patients. We maximized the protection of the interests of the patients and did not cause harm to any patients.
PI Measurement and Hemodynamic Monitoring
The PI of the finger was continuously measured using an IntelliVue MP70 monitor (Philips Medical Systems, Boblingen, Germany). The MP70 system calculates the PI as the ratio between the pulsatile and nonpulsatile components of light reaching the light-sensitive cell of a pulse oximetry probe. The ambient temperature of the room was maintained at approximately 23 °C to 25 °C (climate controlled).
The thermodilution CO was measured by injecting 15 mL of 0.9% saline at 0 °C using PiCCO-Plus (PiCCO system: Pulsion Medical System, Munich, Germany). Three COs that were within 10% of each other were obtained and averaged. Global hemodynamic variables, such as CO and global end-diastolic volume index (GEDVI), were recorded simultaneously.
Simultaneous basic blood gases were obtained from arterial and central venous catheters (placement of a central venous catheter in the superior vena cava was confirmed by chest radiography). Blood gas samples were collected anaerobically in 3 mL heparinized syringes (PL67BP; BD Diagnostics, Plymouth, UK) and analyzed using blood gas bedside machines (GEM Premier 3000, model 5700; Lexington, MA, USA or ABL90: Radiometer, Copenhagen, Denmark).
Data Collection
Data regarding basic clinical characteristics were collected, including age, sex, Acute Physiology and Chronic Health Evaluation II (APACHE II) scores, Sequential Organ Failure Assessment (SOFA) scores, comorbidities, primary site of infection, blood temperature, serum white blood cell count, continuous renal replacement therapy, and 28-day prognosis after PiCCO initiation. Hemodynamic parameters such as central venous pressure (CVP), heart rate (HR), MAP, CO, GEDVI, superior vena cava oxygen saturation (ScvO2), norepinephrine doses, lactate, and PI at T0 and T24 were observed. T0 represents the time when PiCCO monitoring was started, and T24 represents the time 24 h after PiCCO initiation.
Statistical Analysis
Descriptive analyses were performed. Abnormal outliers were defined as data with a standard deviation >3 based on the Pauta criterion. Such data were treated as missing values. The results for continuous variables with normal distributions are presented as mean ± standard deviation. Results for continuous variables without normal distributions are presented as median (25th-75th percentiles). For continuous variables, data were analyzed using the t-test or chi-square test, depending on the data distribution and number of variables. Multiple logistic regression models were used to measure the relative risk and 95% confidence interval (CI) for each factor to determine how they indicated a poor prognosis. Correlation analyses were performed using Spearman's test. Receiver operating characteristic (ROC) curves were constructed to compare the accuracy of the PI in predicting the prognosis of patients with sepsis. All comparisons were 2-tailed, and a value of P < .05 was required to exclude the null hypothesis. Statistical analyses were performed using the SPSS software (version 17.0; SPSS, Chicago, IL, USA).
Results
General Characteristics
During the study period (January 1, 2014 to December 30, 2018), 672 critically ill patients who underwent PiCCO monitoring during treatment were admitted to our department. The primary diagnosis in 423 patients was septic shock. Among them, 267 used only a single vasoactive agent (norepinephrine) during the first 24 h after PiCCO initiation. Patients who were admitted for <24 h (n = 36) or who were younger than 18 years (n = 24) were excluded from this study. There were also some unreasonable values (n = 23), such as abnormal outliers, which were considered missing values and were abandoned. After exclusion, 184 patients were included in the study. The clinical characteristics of all the patients included in this study after ICU admission are shown in Table 1. Based on ICU mortality, we divided the patients into 2 groups: survivors (n = 144) and nonsurvivors (n = 40). Nonsurvivors were more likely to be females (P < .001), and they had higher SOFA scores (14 ± 3 vs 12 ± 4, P = .007) and higher APACHE II scores (28 ± 8 vs 24 ± 8, P = .014) than survivors. However, there were no significant differences in age, comorbidities, primary site of infection, or blood temperature between the groups.
The General Characteristics of the Patients Included.
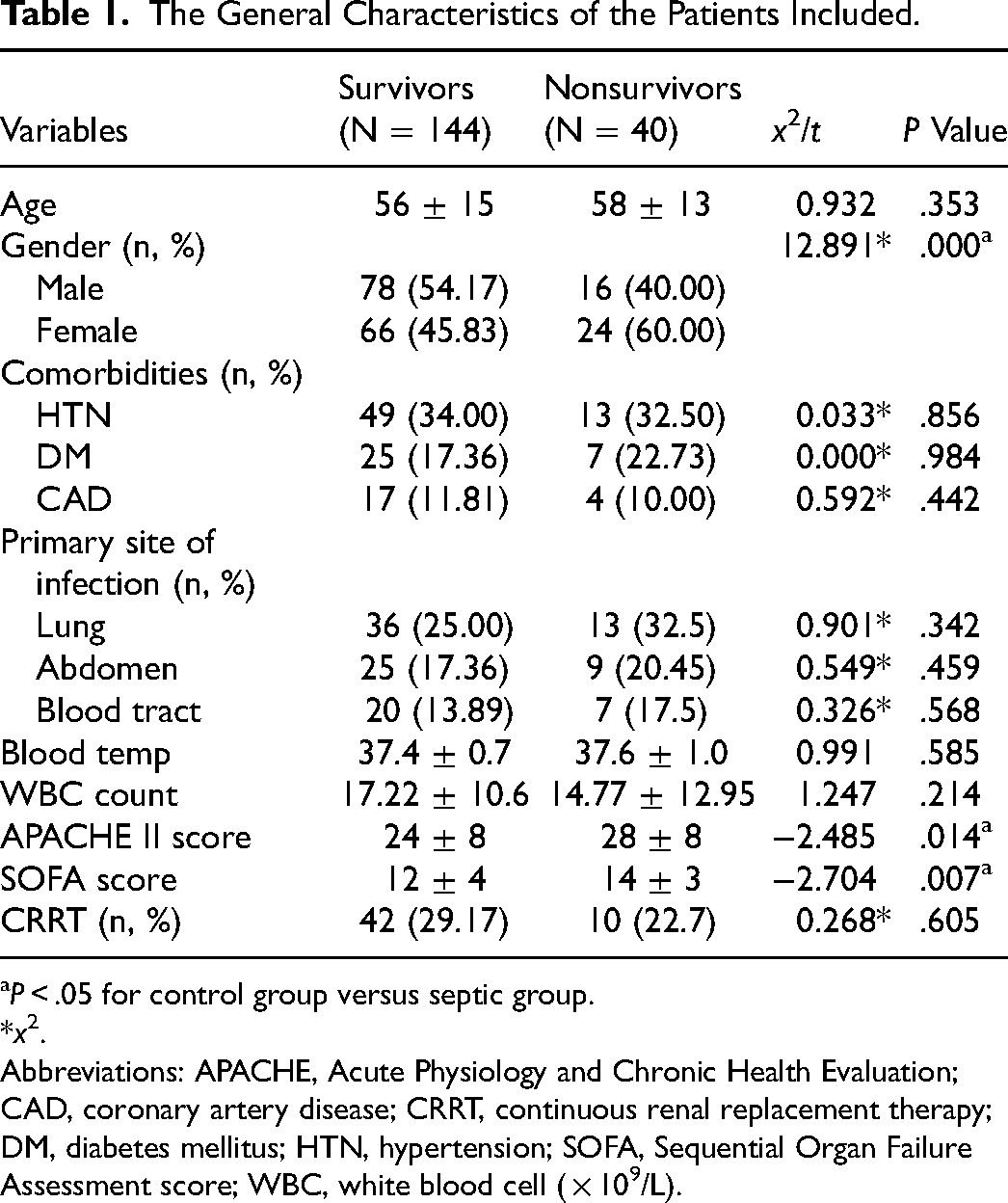
P < .05 for control group versus septic group.
*x2.
Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; CAD, coronary artery disease; CRRT, continuous renal replacement therapy; DM, diabetes mellitus; HTN, hypertension; SOFA, Sequential Organ Failure Assessment score; WBC, white blood cell ( × 109/L).
Hemodynamics and Circulation Perfusion Targets Between Different Prognosis Groups
There were no statistical differences in the systemic hemodynamic parameters such as CVP, HR, CO, GEDVI, and MAP at T0 and T24 in both groups (Table 2). PI (0.9 ± 0.8 vs 0.9 ± 0.7, t = 0.222, P = .824) and lactate levels (4.4 ± 3.9 vs 7.3 ± 6.6, t = −4.800, P = .089) were not significantly different between the 2 groups at T0, but ScvO2 of the nonsurvivor group was higher than that of the survivor group (75.5 ± 10.0 vs 71.2 ± 12.1, t = −2.080, P = .039.) After treatment, at T24, the PI of the nonsurvivor group was significantly lower than that of the survivor group (0.5 ± 0.4 vs 1.5 ± 1.3, t = 5.809, P < .001). In contrast, the lactate level of the nonsurvivor group was significantly higher than that of the survivor group (7.5 ± 7.1 vs 4.0 ± 3.7, t = 5.809, P < .001) at T24. However, regarding the sublingual dose of norepinephrine indicators, we found significant differences between the 2 groups at T0 and T24.
Tissue Perfusion Indicator and Hemodynamic Index of Septic Shock at ICU Admission.
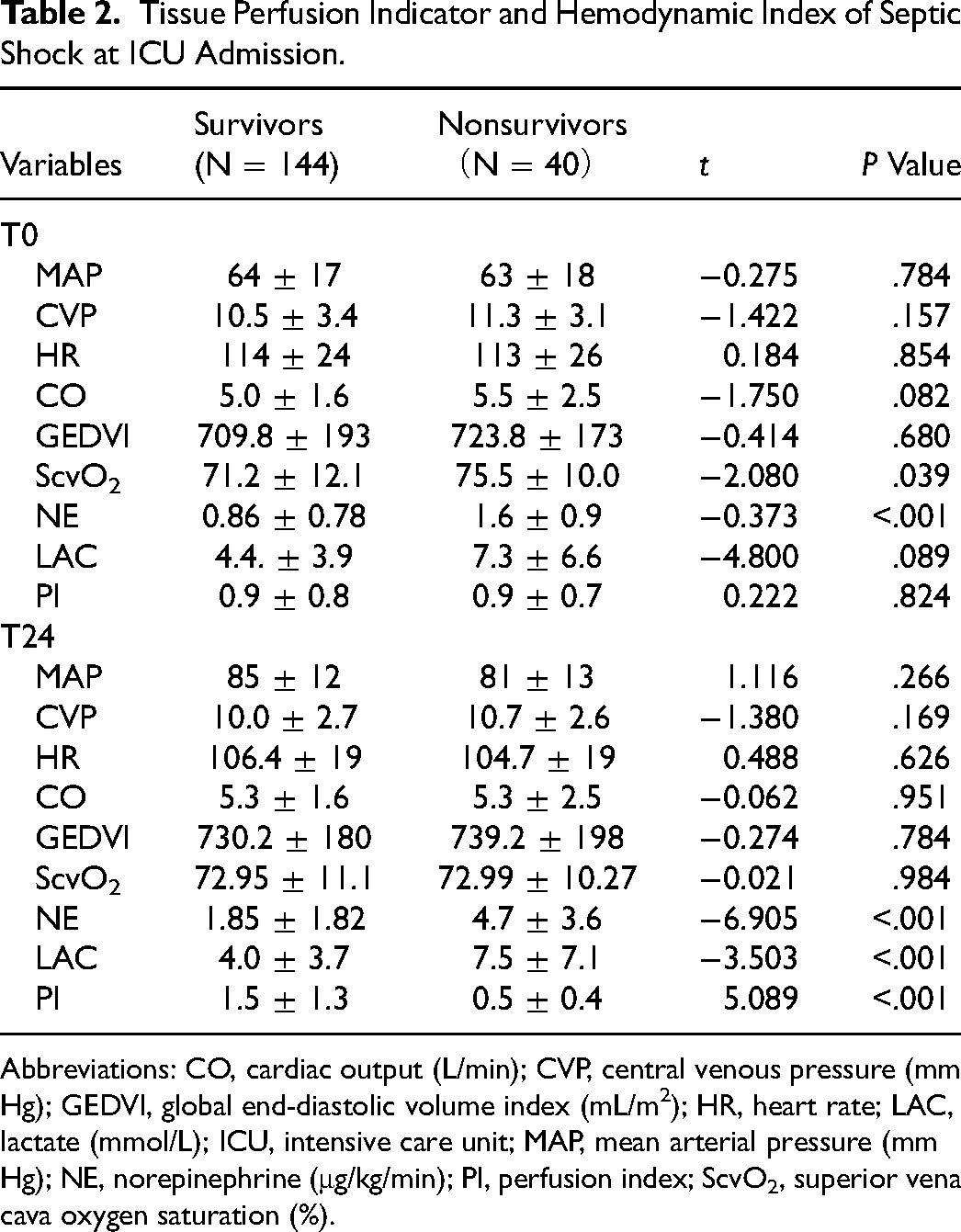
Abbreviations: CO, cardiac output (L/min); CVP, central venous pressure (mm Hg); GEDVI, global end-diastolic volume index (mL/m2); HR, heart rate; LAC, lactate (mmol/L); ICU, intensive care unit; MAP, mean arterial pressure (mm Hg); NE, norepinephrine (μg/kg/min); PI, perfusion index; ScvO2, superior vena cava oxygen saturation (%).
Risk Factors for ICU Mortality in Patients With Sepsis
A multivariate logistic regression analysis was used to examine possible risk factors for poor ICU prognosis (Table 3). Sex, APACHE II scores, SOFA scores, T0-ScvO2, T0-norepinephrine, T24-norepinephrine, T24-lactate, and T24-PI were considered in the model. T24-PI and T24-norepinephrine were the most significant in the regression equation (P < .05). The odds ratio of T24-PI and T24-norepinephrine were 0.146 (95% CI, 0.048-0.444) and 1.386 (95% CI, 1.119-1.317), respectively.
Multivariable Logistic Regression Analysis for Possible Risk Factors for ICU Mortality.
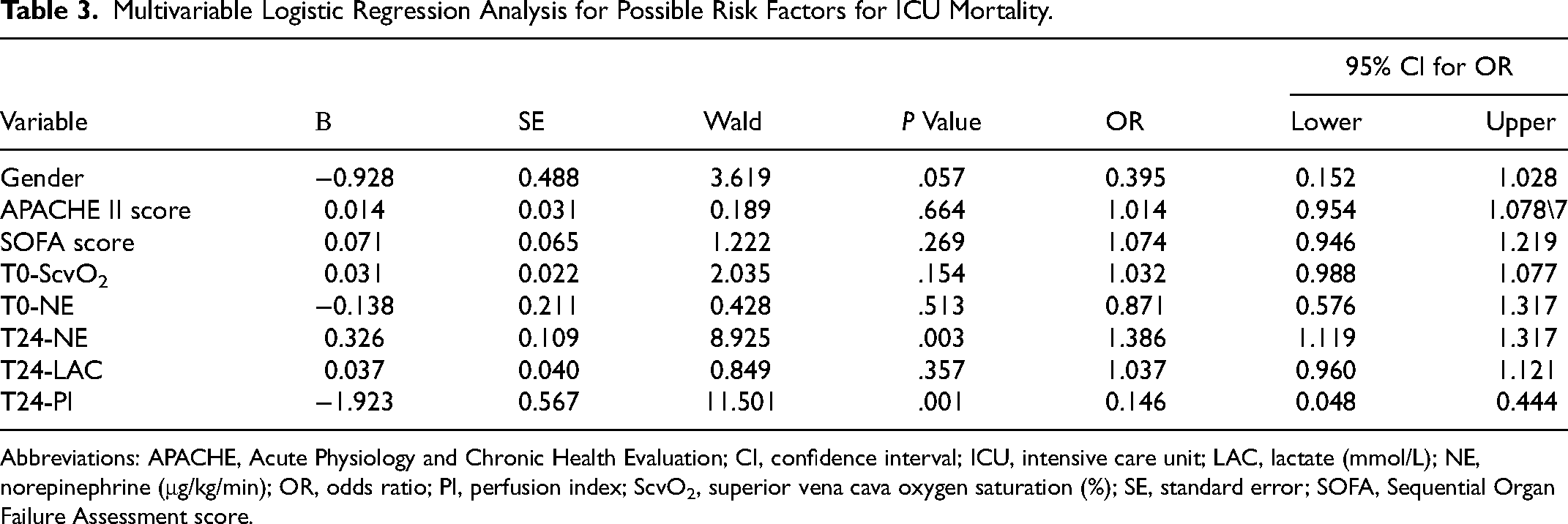
Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; CI, confidence interval; ICU, intensive care unit; LAC, lactate (mmol/L); NE, norepinephrine (μg/kg/min); OR, odds ratio; PI, perfusion index; ScvO2, superior vena cava oxygen saturation (%); SE, standard error; SOFA, Sequential Organ Failure Assessment score.
Receiver Operating Characteristic Analysis
The ROC curve of T24-PI for predicting ICU mortality is shown in Figure 1. The area under the ROC curve was 0.847 (95% CI, 0.782-0.912). The optimal cutoff value of the T24-PI for predicting ICU mortality was 0.6, with a sensitivity of 77.1% and a specificity of 80%. This cutoff value was used for all further analyses.
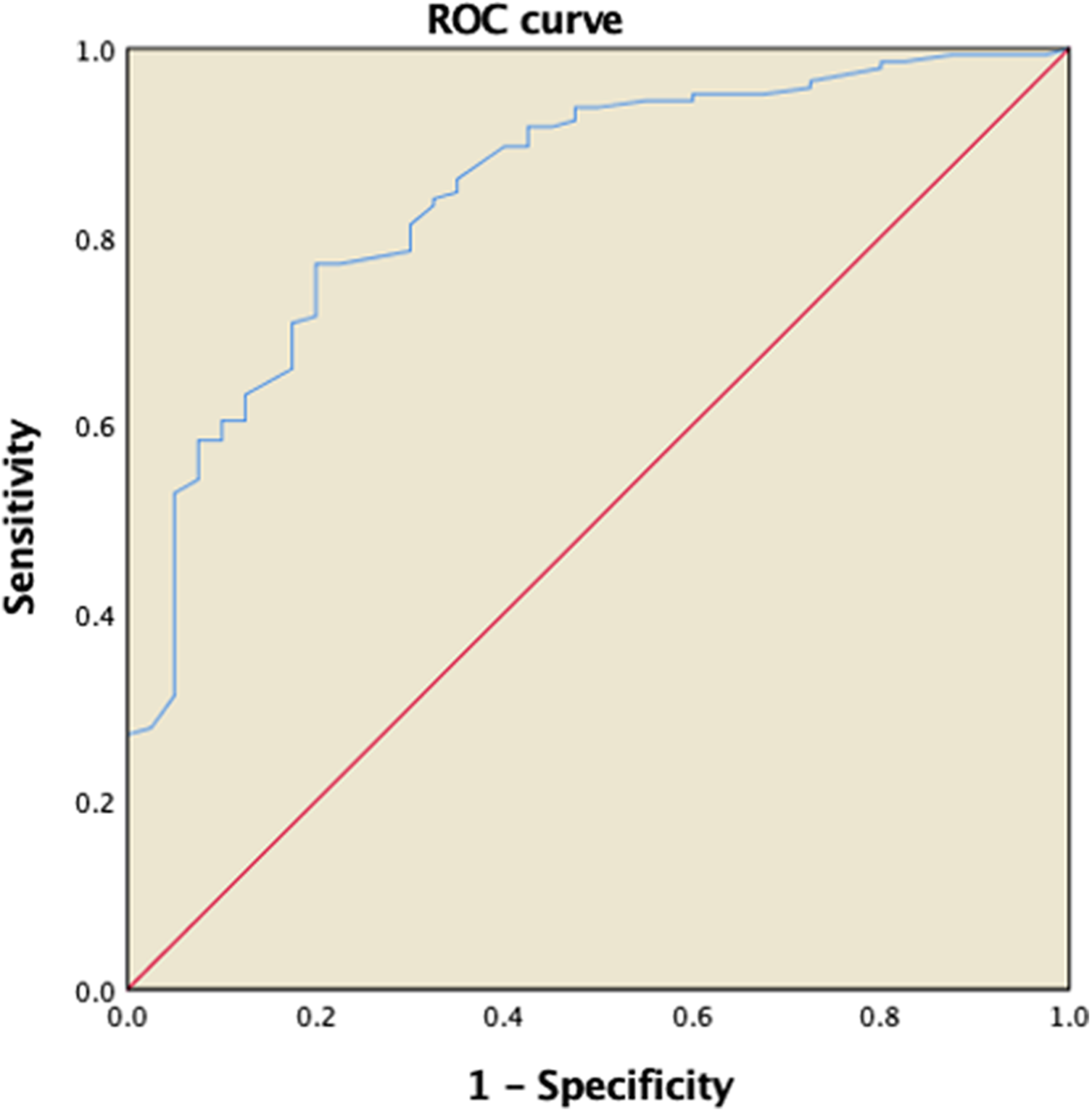
ROC curve using PI for predicting ICU mortality. Abbreviations: ICU, intensive care unit; ROC, receiver operating characteristic; PI, perfusion index.
Hemodynamics and Circulation Perfusion Targets at T24 Between the Different PI Groups
Based on the optimal cutoff value of the T24-PI, we divided patients into groups with PI ≥ 0.6 (n = 125) and PI < 0.6 (n = 59). There were no statistically significant differences in systemic hemodynamic parameters such as age, CVP, HR, CO, GEDVI, and MAP at T24 between the PI ≥ 0.6 and PI < 0.6 groups (Table 4). However, APACHE II scores of the PI < 0.6 group were significantly higher than those of the PI ≥ 0.6 group (28 ± 8 vs 24 ± 7, t = −3.145, P = .002). The lactate level of the PI < 0.6 group was significantly higher than that of the PI ≥ 0.6 group (6.9 ± 6.6 vs 3.6 ± 3.5, t = −4.396, P < .001) at T24. The PI < 0.6 group showed a significantly higher sublingual dose of norepinephrine indicators than the PI ≥ 0.6 group (3.6 ± 3.3 vs 2.0 ± 1.8, t = −4.423, P < .001).
Hemodynamic Index and Tissue Perfusion Indicator of Septic Shock at T24.
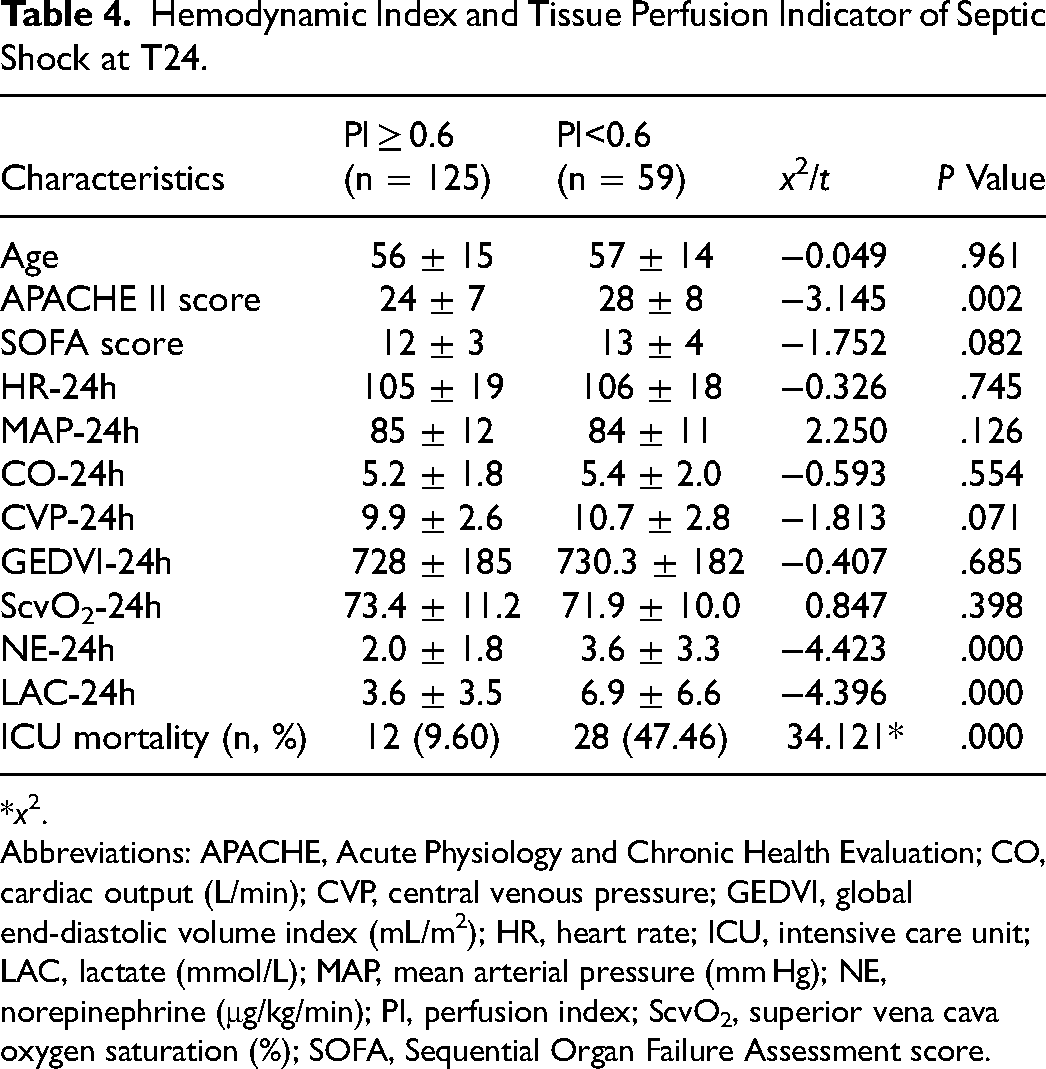
*x2.
Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; CO, cardiac output (L/min); CVP, central venous pressure; GEDVI, global end-diastolic volume index (mL/m2); HR, heart rate; ICU, intensive care unit; LAC, lactate (mmol/L); MAP, mean arterial pressure (mm Hg); NE, norepinephrine (μg/kg/min); PI, perfusion index; ScvO2, superior vena cava oxygen saturation (%); SOFA, Sequential Organ Failure Assessment score.
Correlation Analysis Between PI and Risk Factors
The PI did not correlate with APACHE II scores (r = −0.131, P = .076). In contrast, the PI was strongly negatively correlated with norepinephrine (r = −0.344, P < .001) and lactate (r = −0.291, P < .001).
Discussion
Our study showed that a higher PI is a protective factor and that using a higher dose of norepinephrine is a risk factor for the prognosis of critically ill patients with septic shock. Using a higher dose of norepinephrine can reduce the peripheral blood flow, which is reflected in the PI. A higher PI was associated with a lower dose of norepinephrine, which tended to improve the 28-day prognosis of patients with septic shock.
To our knowledge, this is the first study to explore the relationship between PI and norepinephrine in patients with sepsis after resuscitation. PI, defined as the ratio of the pulsatile to nonpulsatile components of the pulse oximetry plethysmograph, is used as a simple and accurate indication of changes in digital blood flow.7,8 Changes in the finger PI result from blood volume pulsation, dispensability of the vascular wall, and intravascular pulse pressure. 9 PI has been shown to reflect changes in peripheral circulation perfusion and central hypovolemia, both of which are derived from photoelectric plethysmographic pulse oximetry signals. 10 Some studies have shown that PI can be used as a predictor of early adverse respiratory neonatal outcomes after elective cesarean delivery. 11 It has been suggested to be a reliable and early indicator of regional block success and is known to increase because of the effects of the autonomic blockade during spinal anesthesia. 12 In patients with septic shock, PI is related to 6 h lactate clearance. 10 Some studies have used an abnormal PI of <1.4 as a potential trigger to start treatment. In our study, we found that the cutoff value of the PI was <0.6 for predicting ICU mortality in patients with septic shock, resulting in a sensitivity of 77.1% and a specificity of 80%.
In our study, a higher norepinephrine dose was a significant risk factor for critically ill patients with septic shock. Norepinephrine is both an alpha 1 and beta 1 agonists and is therefore able to increase vascular tone and contractility. 13 Recent guidelines recommend norepinephrine as the first-line vasopressor for septic shock. 14 Recent studies have revealed that norepinephrine does not alter perfusion in the kidney or gut.15,16 However, because septic shock results from a variable combination of decreased venous return, myocardial depression, and decreased vascular tone, the location of norepinephrine during the initial resuscitation is not straightforward. Although the cause of shock and treatment with norepinephrine were not predictive of death when high doses of the drug were deemed necessary, rescue treatment with high-dose norepinephrine was futile in patients with severe disease and metabolic acidemia. 4 Norepinephrine-induced vasoconstriction may aggravate internal organ ischemia, leading to patient deterioration. 17 Adverse cardiac events occur in 48.2% of surgical ICU patients with cardiovascular failure and are associated with morbidity and mortality. The extent and duration of catecholamine vasopressor therapy are independently associated with, and may contribute to, the pathogenesis of adverse cardiac events. 18
In the application of norepinephrine, attention should be paid not only to the effect of norepinephrine on arterial contraction but also to its effect on microcirculation perfusion. The patients in our study who received a higher dose of norepinephrine and had a lower PI had worse outcomes. Moreover, a relationship between high doses of norepinephrine and PI has been demonstrated. In other words, a high dose of norepinephrine can decrease peripheral perfusion by affecting the peripheral blood flow. The administration of norepinephrine in patients with severely hypotensive septic shock is associated with an increase in CO. 19 In a series of 14 patients with septic shock, correction of severe hypotension with norepinephrine administration resulted in an increase in MAP from 51 ± 3 mm Hg to 79 ± 7 mm Hg, and norepinephrine had positive effects on renal function in patients with sepsis. 20 These results suggest that norepinephrine improves tissue perfusion when used to correct severe hypotension, although evidence is limited. However, most clinical trials have evaluated the short-term effects of vasopressors, and some of these beneficial effects may vanish over time. Under healthy conditions, both norepinephrine and vasopressin decrease microvascular perfusion. 21 Accordingly, the net effect of increasing the perfusion pressure on tissue perfusion may depend on the balance between the potentially beneficial effects on organ blood flow and the negative impact on microvascular perfusion. Therefore, we need to focus on the causal interaction between norepinephrine and PI, especially when the PI is lower than 0.6 on the first day of septic shock. Vasopressive catecholamines may be associated with excessive vasoconstriction, which may result in impaired tissue perfusion even when perfusion pressure is restored. 22 However, we should titrate a high dose of norepinephrine based on the PI level to some extent, which means that we should set a hemodynamic target based on the patient's PI. The PI can be used as a reference indicator to determine the dose of norepinephrine required to achieve circulatory protection.
Some limitations of our study need to be acknowledged. First, this study was based on data from a single center; therefore, additional clinical prospective cohort research and practice are required for confirmation. Second, we have not compared PI to other indicators such as lactate. We think we can do a deeper research in the future. Finally, our study is an observational one; significant correlations between microcirculatory hypoperfusion and changes in mortality rates do not prove causality. Because this is a single center, observational study, our results should be interpreted with caution.
Conclusions
Based on the analysis of a large set of data on patients with sepsis, we found that a higher PI is a protective factor and that the use of a higher dose of norepinephrine is a risk factor for the prognosis of critically ill patients with septic shock. Using a higher dose of norepinephrine and a lower PI resulted in a worse prognosis of patients in our study, and these 2 variables had a causal interaction; using a higher dose of norepinephrine can reduce peripheral blood flow, which is reflected in the PI. A higher PI is associated with a lower dose of norepinephrine, which may improve the prognosis of patients with septic shock.
Footnotes
Abbreviations
Authors’ Contributions
XW and DL conceived and designed the study, interpreted data, and helped draft the manuscript. CW participated in the study conception and design, recruited patients, collected data, performed the statistical analysis, interpreted the data, and drafted the manuscript. HZ participated in patient recruitment, data collection, technical support, and contributed to the critical review of the manuscript. CZ contributed to the critical review of the manuscript. All the authors read and approved the final manuscript.
Ethics Approval and Consent to Participate
The Institutional Research and Ethics Committee of Peking Union Medical College Hospital approved this study for human subjects (No.S-K980).
Consent for Publication
All authors agree to publish in this journal.
Availability of Data and Material
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
