Abstract
Keywords
Introduction
Sleep is integral to critical illness recovery yet most adults experience disrupted sleep during and after the intensive care unit (ICU) stay. 1 This sleep deprivation is extremely bothersome to both patients and their loved ones. A patient who is sedated is not necessarily sleeping. 2 Circadian alteration, frequent nocturnal interruptions, and reduced restorative sleep are common in the ICU. Poor ICU sleep is attributable to baseline (eg, increased age, comorbid conditions, and chronic poor sleep) and ICU (severity of illness, noise, pain, light, and care-related activities) factors, 3 and associated with delirium and increased Post-Intensive Care Syndrome. 4 The 2018 Society of Critical Care Medicine Clinical Practice Guidelines for Management of Pain, Agitation, Delirium, Immobility, and Sleep (PADIS) recommend the use of a protocolized approach to improve sleep that incorporates the simultaneous use of multiple non-pharmacologic components. 4 These sleep bundles have been shown to reduce delirium and improve sleep. 5 Although not recommended by PADIS, and despite questions regarding their benefit, medications with the potential to initiate or maintain sleep, or improve circadian entrainment, are often added to ICU sleep protocols.5–7
Medications are frequently initiated in the ICU to improve sleep yet the evidence to support their efficacy and safety has not recently been updated.8,9 The 2018 PADIS guidelines only considered evidence published prior to 2016. 4 Prior ICU sleep aid systematic reviews have focused on delirium occurrence and not measures of sleep improvement. However, the inter-relationship between delirium and sleep in the ICU remains poorly elucidated. 10 Moreover, the PADIS guidelines only evaluated dexmedetomidine, melatonin, and propofol; several agents with the potential to improve ICU sleep, 4 including dual orexin receptor antagonists and ramelteon, were not considered. Finally, the question of whether medication sleep aids augment non-pharmacologic sleep improvement interventions has not been rigorously reviewed. 11 We sought to address these evidence gaps by systematically reviewing and synthesizing evidence evaluating the use of medications focused on sleep improvement in adult ICU patients.
Materials and Methods
Protocol and Registration
This rapid systematic review and associated protocol were registered with the PROSPERO international prospective register of systematic reviews (Registration Number: CRD42022346818). This protocol did not require ethical approval.
Study Eligibility
We included randomized, controlled studies evaluating any pharmacologic intervention on a sleep-related outcome(s) in adults (≥18 years old) who were admitted to an ICU. Studies must have evaluated an outcome related to 1 or more of the following sleep parameters: duration, efficiency, stage, quality, or awakenings. We considered before-and-after cohort studies if they enrolled at least 20 patients in each arm. We excluded studies if a medication was being evaluated as 1 element of a multicomponent sleep bundle or where the medication was administered prior to noon. We also excluded all case reports and case series, and studies evaluating complementary medicines. Studies published in abstract form were included if there was no subsequent manuscript with the same dataset and only if our outcomes of interest were reported.
Search Strategy, Sources, and Study Identification
We performed computerized searches of Medline (Ovid), Cochrane Library (Wiley Online; includes Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials, Cochrane Methodology Register), and Embase (Elsevier). The searches were initially conducted on July 13, 2022 and rerun on October 3, 2022 by an experienced medical librarian (E.F.G.). Search strategies combined keywords and subject headings for the concepts of sleep induction, pharmacologic therapies, and ICU patients (Supplemental Data File). No filters or limits were applied. Hand searching was conducted in the reference sections of pertinent book chapters,12,13 and relevant studies were retrieved.
Study Selection
Study selection and data abstraction were conducted in Covidence. 14 Following the rapid systematic review process, 15 title and abstract screening was conducted by 2 authors (M.S.H. and P.R.L.) on 30 records. The remaining records were screened by 1 author with the second author reviewing excluded records. Full text screening was conducted by 2 authors (M.S.H. and P.R.L.) on 6 records. The remaining full texts were screened by 1 author with the second author reviewing excluded records. Disagreements in screening assessments were reconciled through discussion between the screeners, with further discussion by the author team, if needed. Interrater reliability was evaluated at both screening steps.
Data Extraction
Data of interest were collected from full texts by 2 authors (K.M.L. and D.V.). One author completed extraction, the second author checked for correctness and completeness of collected data. Author name, study size, mean age, percentage of patients on mechanical ventilation, type of study, type of ICU, standard of care used in both study arms, pharmacologic intervention, intervention dosing strategy, primary endpoint of the study (regardless of whether sleep-related), any sleep-related secondary endpoints, and safety parameters were collected. We also evaluated whether the study excluded patients with a history of sleep aid use or sleep-related problems prior to study enrollment.
Assessment of Risk of Bias
The Cochrane Collaboration Risk of Bias (RoB2) tool was used to assess methodologic quality of randomized studies and the Risk of Bias in Non-Randomized Studies of Interventions (ROBINS-I) tool was used for non-randomized intervention studies.16,17 One author (K.M.L.) completed the assessment and provided support statements as justification, a second (D.V.) verified the assessments based on the justifications and confirmed evaluation completeness. Disagreements were reconciled through discussion.
Data Analysis
Extracted data were categorized by medication intervention and then summarized by sleep-related outcome either subjectively (e.g., patient or nurse perception) or objectively (e.g., actigraphy, polysomnography). Studies were reported with an emphasis on randomized controlled trials (RCTs). We abided by the Cochrane guidance for conducting rapid reviews to determine if studies were similar enough to pool for analysis. 15
Results
Database queries and hand searching yielded 536 references. After duplicates were removed, 360 references remained for title and abstract screening (Figure 1). Of these, 33 references were subject to full text review. Ultimately, 16 references were eligible for data extraction and risk of bias analysis.
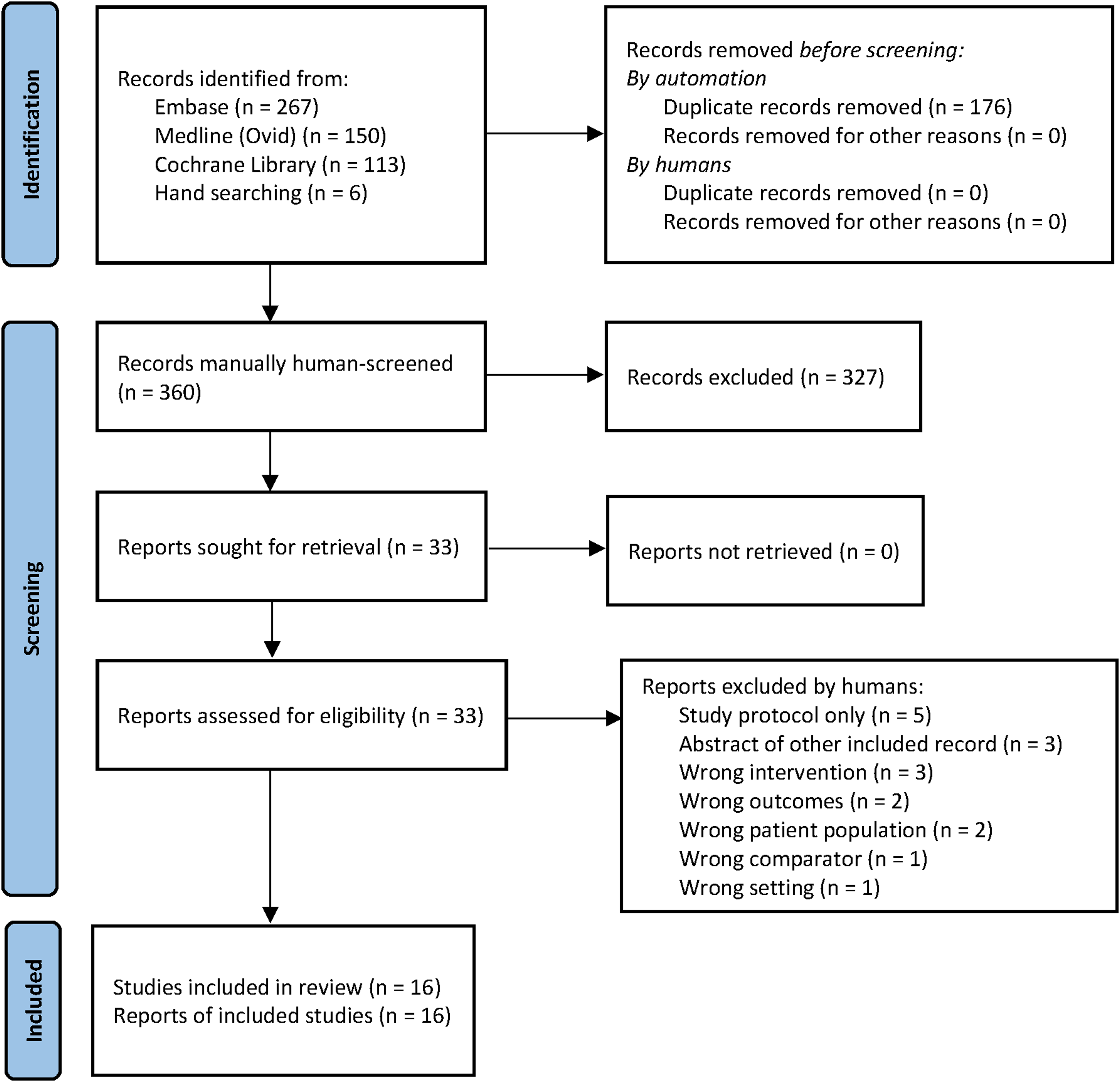
PRISMA 2020 flow diagram for new systematic review.
Summary of Included Studies
Of the 16 studies included for analysis, 12 (75%) were randomized trials and 4 (25%) were cohort studies.18–33 Publication years ranged from 1996 to 2022. Half of the studies excluded patients reporting sleep aid use prior to study enrollment. Among the 8 studies including invasively mechanically ventilated adults, per-study frequency of ventilation ranged 43% to 100%. No study evaluated an antipsychotic, a sedative-hypnotic agent (eg, zolpidem, zaleplon, eszopiclone), an orexin antagonist (eg, suvorexant), or antidepressant (eg, trazodone). The 2573 subjects enrolled in the 16 studies had a median age of 62 years, 2279 (88.6%) were admitted to a medical or surgical ICU, and 636 (24.7%) were invasively mechanically ventilated. Of these subjects, 1207 (46.9%) were allocated to the intervention group and 1366 (53.1%) to the control group.
Dexmedetomidine was evaluated in 7 studies (representing 505 [41.8%] of all total intervention patients), a melatonin agonist in 6 studies (5 melatonin, 1 ramelteon; 592 [49.1%] all total intervention patients), propofol in 3 studies (65, 5.4%), and a benzodiazepine in 2 studies (45, 3.7%) (Table 1).18–33 Due to the considerable variability in the method by which sleep-related outcomes were reported across studies, we were unable to analyze the pooled study data in aggregate. Safety events were reported in 12 (75%) of the included studies (Table 2).18–33
Characteristics of Included Studies.
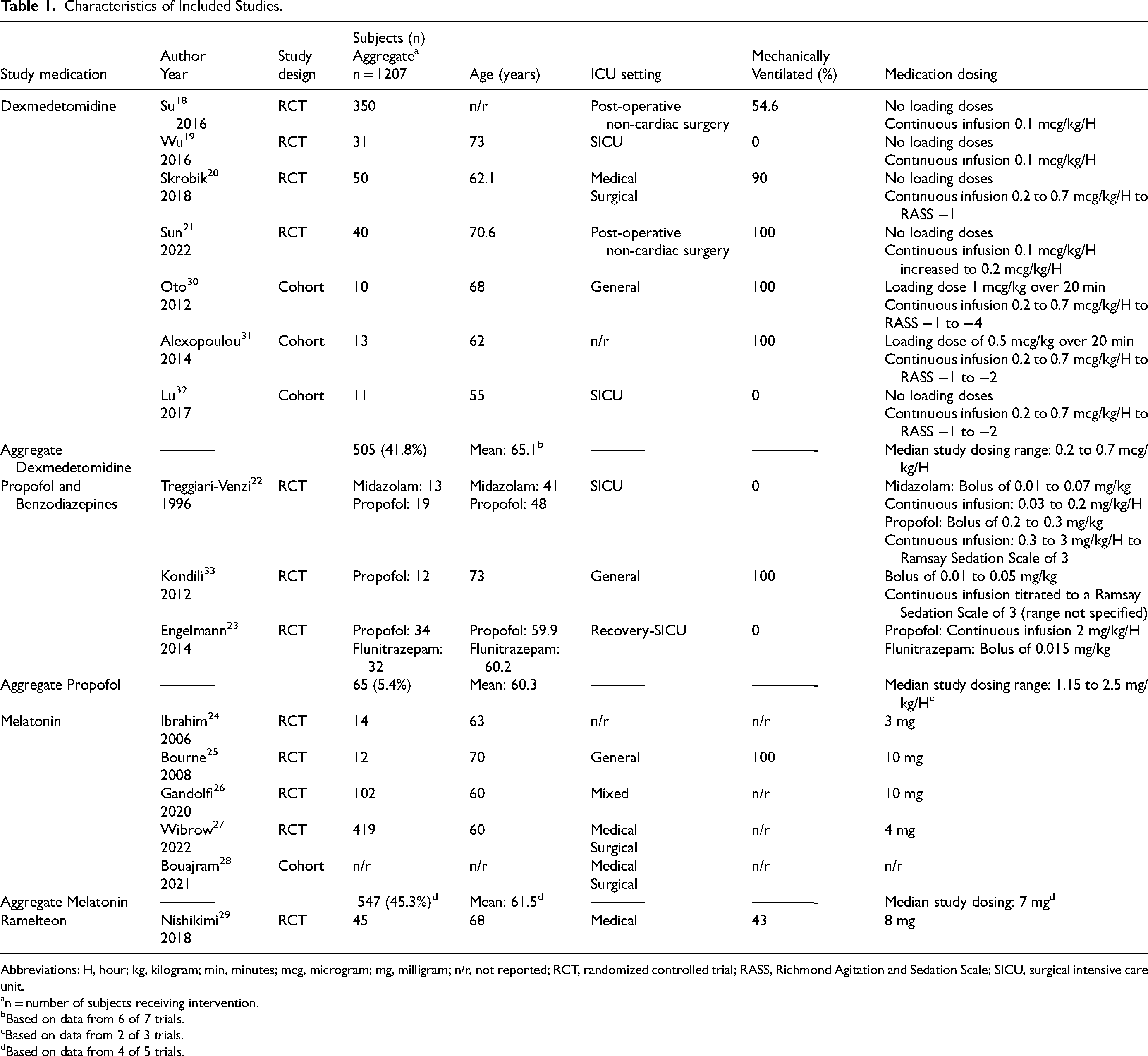
Abbreviations: H, hour; kg, kilogram; min, minutes; mcg, microgram; mg, milligram; n/r, not reported; RCT, randomized controlled trial; RASS, Richmond Agitation and Sedation Scale; SICU, surgical intensive care unit.
n = number of subjects receiving intervention.
Based on data from 6 of 7 trials.
Based on data from 2 of 3 trials.
Based on data from 4 of 5 trials.
Sleep-Related and Safety Outcomes.

Abbreviations: ABG, arterial blood gas; AEs, adverse events; AUC, area under the curve; BIS, bispectral index; bpm, beats per minute; BZD, benzodiazepines; CI, confidence interval; DBP, diastolic blood pressure; DEX, dexmedetomidine; flunit, flunitrazepam; H, hour; HAD, Hospital Anxiety and Depression; H:M:S, hour:minute:second; HR, heart rate; HTN, hypertension; ICU, intensive care unit; ITT, intention to treat; LFTs, liver function tests; LOS, length of stay; LSEQ, Leeds Sleep Evaluation Questionnaire; MAP, mean arterial pressure; MID, midazolam; min, minute; num, number; MV, mechanical ventilation; N1, stage 1 sleep; N2, stage 2 sleep; N3, stage 3 sleep; n/r, not reported; NRS, Numeric Rating Scale; NS, not significant; O2 sat, oxygen saturations; N/V, nausea/vomiting; OR, odds ratio; PaCO2, partial pressure of carbon dioxide; P:F, arterial oxygen partial pressure to fractional inspired O2; PSQI, Pittsburgh Sleep Quality Index; Prop, propofol; RASS, Richmond agitation sedation scale; REM, rapid eye movement; RCSQ, Richards-Campbell Sleep Questionnaire; RN, registered nurse; RR, respiratory rate; SAEs, serious adverse events; SBP, systolic blood pressure; SWS, slow wave sleep; TST, total sleep time; TV, tidal volume.
Non-pharmacologic sleep promotion protocol was utilized as a standard of care in both arms of study.
Subjective Sleep-Related Outcomes
A total of 12/16 (75%) studies reported subjective sleep-related outcomes; however, the tools used to subjectively evaluate sleep differed considerably across studies.18,20–29,32 The Richards-Campbell Sleep Questionnaire (RCSQ) was used in 5 studies.18,25–28 Less commonly, the Numeric Rating Scale (NRS) was used in 2 studies,18,19 and the Hospital Anxiety and Depression (HAD) scale, 22 Leeds Sleep Evaluation Questionnaire (LSEQ), 20 Little's Patient-Rated Sleep Quality, 27 Pittsburgh Sleep Diaries, 23 and Pittsburgh Sleep Quality Index (PSQI) 21 were each used in 1 study. Nursing assessment without a specific tool was utilized in 3 studies.24,25,29 These subjective strategies used a diverse set of parameters to assess sleep and are described in Supplemental Table 1.18,20–29,32
Dexmedetomidine
Four nocturnal dexmedetomidine RCTs evaluated subjective sleep quality.18–21 The first RCT evaluated 700 postsurgical adults admitted to a surgical ICU for a short stay after non-cardiac surgery who were randomized to receive dexmedetomidine (dosed at 0.1 mcg/kg/H) or placebo starting the day of surgery. 18 Dexmedetomidine (mean duration of use 14.95 h) was associated with decreased incidence of delirium in the first 7 postoperative days (dexmedetomidine 79 [23%] versus placebo 32 [9%], P < .0001). Quality of sleep on postoperative days 1 through 3 (as evaluated using the NRS) was significantly better with dexmedetomidine (dexmedetomidine mean 4 vs placebo mean 2, P < .0001).
The second RCT evaluated dexmedetomidine 0.1 mcg/kg/H administered for 15 h from 1700 the day of surgery until 0800 the morning after surgery in 76 adults ≥65 years old after non-cardiac surgery. 19 Subjective sleep quality per NRS was evaluated at 0800 on days 1, 2, and 3 after surgery. On day 1 after surgery the most notable difference between dexmedetomidine and placebo sleep quality was observed with patients receiving dexmedetomidine reporting better sleep quality (1 dexmedetomidine vs 3 placebo, P = .005). There were no significant differences in sleep quality on days 2 and 3.
The third RCT evaluated a 2130 to 0600 infusion of dexmedetomidine 0.2 to 0.7 mcg/kg/H titrated to a RASS of −1 in a population of 100 delirium-free, mixed medical-surgical ICU adults (90% were mechanically ventilated; most were receiving continuous midazolam or propofol). 20 A greater proportion of dexmedetomidine patients remained delirium free (80% vs 54%; RR 0.44; 95% CI [0.23-0.82]; P = .006). The average daily LSEQ evaluation scores (able to be evaluated on 70%-74% of study days in placebo vs dexmedetomidine) were not different between the 2 groups. When LSEQ results were stratified by individual domain, dexmedetomidine-treated patients felt less tired the day after study enrollment (P < .05).
The fourth RCT evaluated dexmedetomidine 0.1 to 0.2 mcg/kg/H (started the day of recruitment and throughout mechanical ventilation up to 3 days) in 80 mechanically ventilated adults after non-cardiac surgery. 21 Neither the median RCSQ score, the individual RCSQ scores assessing awakenings and sleep quality on the night of surgery, nor the individual RCSQ scores between postoperative days 0 and 7 were different between the dexmedetomidine and placebo groups. At 30 days, PSQI results were also not different. Objective sleep outcome data from this study are discussed in the Objective Outcomes section.
Benzodiazepines and Propofol
One open, comparative, prospective, and randomized study compared the effect of midazolam versus propofol on sleep quality using the HAD scale in 32 non-elderly SICU patients. 22 Sleep quality was found to be poor and not different between the 2 groups. Since the publication of this trial in 1996, contemporary data have linked daily ICU benzodiazepine exposure to increased delirium, reducing the external validity of this dataset.22,34
When propofol (2 mg/kg/H) was compared to flunitrazepam (0.015 mg/kg) in 1 RCT enrolling 66 postsurgical adults transitioning quickly through the surgical ICU, propofol was associated with fewer awakenings (0 propofol, 3 flunitrazepam, P < .001), a shorter awakening duration when they occurred (0 min propofol, 15 min flunitrazepam, P < .001) and better overall sleep quality (median score 2 propofol, 3 flunitrazepam P < .001). 23 Improved subjective metrics associated with propofol were further corroborated by improved bispectral index (BIS)-reported sleep architecture (see the Objective Outcomes section of this paper).
Melatonin
Four RCTs and 1 observational study were identified.24–28 The first RCT compared melatonin (3 mg at 2000 for at least 2 days) with placebo in ICU 32 tracheotomized patients. Nocturnal sleep, diurnal sleep, incidence of agitation, and a requirement for rescue sedation were used as estimates for sleep quality. 24 Despite achieving melatonin plasma concentration levels 1000-fold greater in the melatonin group, no differences were observed for any sleep outcome between melatonin and placebo.
The second RCT compared melatonin 10 mg nightly to placebo for 4 ICU nights in 24 mechanically ventilated adults. 25 Sleep efficiency was subjectively evaluated by the bedside nurse every 1 h during nights 3 and 4 and sleep quality was evaluated by the nurse in the morning after night 3 and 4 using the RCSQ. None of these outcomes were found to be statistically different between the melatonin and placebo groups.
The third RCT evaluated the effect of melatonin (10 mg at 2000 for 7 days) to placebo on sleep quality in a mixed population of 203 cardiovascular and postsurgical ICU adults. Patients were evaluated with the RCSQ each morning for 7 days or until hospital discharge. 26 The overall RCSQ was not different between the 2 groups. However, melatonin, when compared to placebo, was associated with improvement in 2 RCSQ sub-categories: sleep depth and overall sleep quality. Compared to placebo, melatonin was not associated with a longer duration of nocturnal sleep (or reduced delirium).
The fourth RCT compared the use of melatonin (4 mg at 2100 for 14 days or ICU discharge) with placebo in 841 delirium-free mixed medical-surgical adults. 27 Melatonin compared to placebo did not improve ICU sleep quality (as evaluated with the RCSQ) or recollection of ICU sleep quality (as evaluated using the Little's Questionnaire) in the post-ICU period. Melatonin, compared to placebo, was not associated with a reduction in delirium, regardless of the patients’ ICU baseline risk. Combined evaluation of data from the melatonin and placebo groups reinforces the persistence of disrupted and curtailed sleep during ICU admissions.
One observational cohort study, only available as a conference abstract, reported the effects of sleep medications on delirium in 285 medical and surgical ICU patients. 28 The trial analyzed 4 separate cohorts including melatonin, melatonin with supplemental sleep aids, alternative sleep aids alone, and no sleep medications. Sleep quality (as evaluated with the RCSQ) was not different between the 4 groups.
Ramelteon
One RCT compared the nighttime administration of ramelteon 8 mg with placebo (both given at 2000) until ICU discharge in 88 ICU adults (43% of whom were invasively ventilated). 29 The primary outcome, duration of ICU stay, was shorter in the ramelteon (vs placebo) group (4.56 vs 5.86 days, P = .028). Sleep evaluation was limited to the 57% of patients not intubated. Ramelteon (vs placebo) was associated with a statistically significantly reduced number of awakenings (0.80/night vs 1.31/night, P = .045) and a higher proportion of ICU nights with complete sleep (51% vs 30%, P = .04).
Objective Outcomes
Eight of the 16 studies identified studies included objective measures of sleep,19,21,23,25,30–33 5 utilized polysomnography (PSG),19,21,30,31,33 and 3 utilized BIS,23,25,32 1 of which also utilized actigraphy. 25 An overview of objective sleep assessment strategies is provided in Supplemental Table 1. 35
Dexmedetomidine
Five ICU dexmedetomidine studies (2 RCTs19,21 and 3 cohort30–32) where sleep was objectively evaluated were identified. A subset (n = 61) of patients enrolled in the Wu et al RCT (described above) were evaluated with PSG (31 dexmedetomidine, 30 placebo). 19 Dexmedetomidine increased the odds of achieving stage 2 (N2) sleep by 14.7% (95% CI: 0.0-31.9), P = .048. Improved sleep architecture was also noted with dexmedetomidine (vs placebo) as evidenced by a significantly longer sleep duration (213 vs 130 min, P = .028) and greater sleep efficiency (22.4% vs 15.0%, P = .033). In the second RCT evaluating sleep objectively, Sun et al (described above) successfully evaluated 68 of 80 total enrolled patients with PSG. 21 Patients in the dexmedetomidine (vs placebo) group tended to have a longer duration of sleep, higher sleep efficiency, lower percentage of stage N1 sleep, higher stage N3 sleep, and lower arousal index; however, none of these differences were statistically significant.
Among the 3 cohort studies, the first evaluated 10 adults who were invasively ventilated ≥48 h, where sleep was evaluated with PSG over 24 h before-and-after a single nocturnal infusion of dexmedetomidine (after initiation of a loading dose of 1 mcg/kg over 20 min at 2100, a continuous infusion of 0.2 to 0.7 mcg/kg/H titrated to a RASS goal of −1 to −4 was administered until 0600. 30 During a 15-h period on the day prior to dexmedetomidine therapy, the median total sleep time was only 1.7 h. During/after dexmedetomidine therapy, sleep efficiency increased from 11.3% during the day before dexmedetomidine to 52.3% at night with dexmedetomidine, median total sleep time increased from 1.7 to 4.7 h, and total sleep time localized to stage 2 (N2) sleep increased from 54 min to 188 min. However, sleep architecture remained fragmented after dexmedetomidine therapy with no patients achieving slow wave or rapid eye movement (REM) sleep.
The second cohort study evaluated sleep quality with PSG in 13 mechanically ventilated adults before and after a single-night nocturnal 2100 to 0600 dexmedetomidine infusion titrated to a RASS goal −1 to −2 (0.5 mcg/kg loading dose followed by a 0.2-0.7 mcg/kg/H infusion). 31 On night 2 (dexmedetomidine) compared to nights 1 and 3 (without dexmedetomidine), sleep efficiency was significantly greater (64.8% vs 9.7%/6.9%, P < .002), the sleep fragmentation index was significantly reduced (2.2 vs 7.1/7.2, P = .023) and a significantly higher proportion of patients achieved stage 2/N2 sleep (78.7 vs 39.2/47.5 min, P = .006). However, dexmedetomidine failed to achieve REM and slow wave sleep.
The last dexmedetomidine cohort study of non-ventilated surgical ICU adults compared 11 patients who received dexmedetomidine (0.2-0.7 mcg/kg/H from 2000 to 0800) with 9 patients who received no sedation. 32 Adequate sleep was defined a priori as a BIS score between 65 and 85. Dexmedetomidine (vs no use) was associated with a significantly longer sleep duration (8 vs 6 h, P = .001) and greater sleep efficiency (90% vs 78%, P = .008). Given the limitations BIS to evaluate sleep, sleep staging could not be evaluated.
Benzodiazepines and Propofol
One 48-h randomized crossover trial of 12 mechanically ventilated adults expected to require an assist control mode of ventilation for ≥48 h evaluated the effect of propofol on sleep architecture using PSG. 33 Investigators concluded that sleep fragmentation and irregular sleep progression was prevalent throughout the study cohort. Sleep fragmentation was not different between the propofol (vs control) groups (4.8 vs 8.1/h, P = .33); neither group achieved slow wave sleep. Propofol (vs control) use was associated with a lower proportion of patients who achieved REM sleep (0 vs 1.4%, P = .04).
The Engelmann et al trial (presented above) also objectively evaluated sleep using BIS where slow wave sleep, light sleep/REM sleep, and awake state were defined by BIS ranges of ≤74, 75 to 89, and ≥90, respectively. 23 Propofol (compared to flunitrazepam) was associated with lower BIS values in aggregate and was associated with a significantly increased duration in slow wave/deep sleep (2H:23 min vs 1H:23 min, P = .04) and a reduced duration of light/REM sleep (1H:44 min vs 2H:34 min, P = .004).
Melatonin
The Bourne et al trial (previously discussed) objectively evaluated sleep on ICU nights 3 and 4 using BIS, where a BIS score <80 was deemed to indicate good sleep. 25 BIS-generated sleep outcome measures evaluated included the sleep efficiency index and the sleep AUC, where a lower AUC indicated better sleep. Melatonin (compared to placebo) did not improve sleep efficiency (0.39 vs 0.26, P = .09) but was associated with significantly better-quality sleep, as indicated by a 7% decrease in BIS AUC with melatonin (AUC difference −54.23, P = .04). Also, there was no significant difference between melatonin and placebo with regards to an actigraphy-driven sleep efficiency index.
Adverse Outcomes
Only 6 of the 16 studies reported safety outcomes. Among the medications evaluated across these 6 studies (Table 2), dexmedetomidine was associated with the greatest number of safety concerns (bradycardia in 77/505 (15.2%) of total dexmedetomidine patients; hypotension in 142/505 (28.1%) of patients). The number of patients who required a clinical intervention for these hemodynamic effects (vs a reduction in the dexmedetomidine dose or its stoppage) was not generally reported.18–21,30–32 Dexmedetomidine is associated with bradycardia and hypotension and therefore should be avoided in patients with second- or third-degree heart block, bradycardia, or severe heart failure. Realizing these cardiac effects are usually dose-related and the dexmedetomidine ICU sleep improvement trials we identified administered a far lower infusion dose that studies evaluating dexmedetomidine for ICU sedation, 36 clinicians should continuously monitor hemodynamic parameters regardless of the dose administered or the indication for dexmedetomidine use. Benzodiazepines were associated with respiratory depression (3/45, 6.6%) and paradoxical psychological events such as confusion, dysphoria, and restlessness (5/45, 11.1%). Melatonin and ramelteon were associated with mild side effects such as headache in only 2/547 (0.03%) of patients. 25
Quality Assessment of Included Studies
Bias assessments are included in Table 3 (RoB2) and Table 4 (ROBINS-I). Seven out of 16 (43.8%) studies were assessed to have a low risk of bias, 5/16 studies (31.3%) had some concerns/moderate risk of bias and 4/16 (25%) had a high or serious risk of bias. Overall, all the studies included had some degree of bias with the most notable bias in the non-randomized control trials.
Risk of Bias Assessment—ROB2.
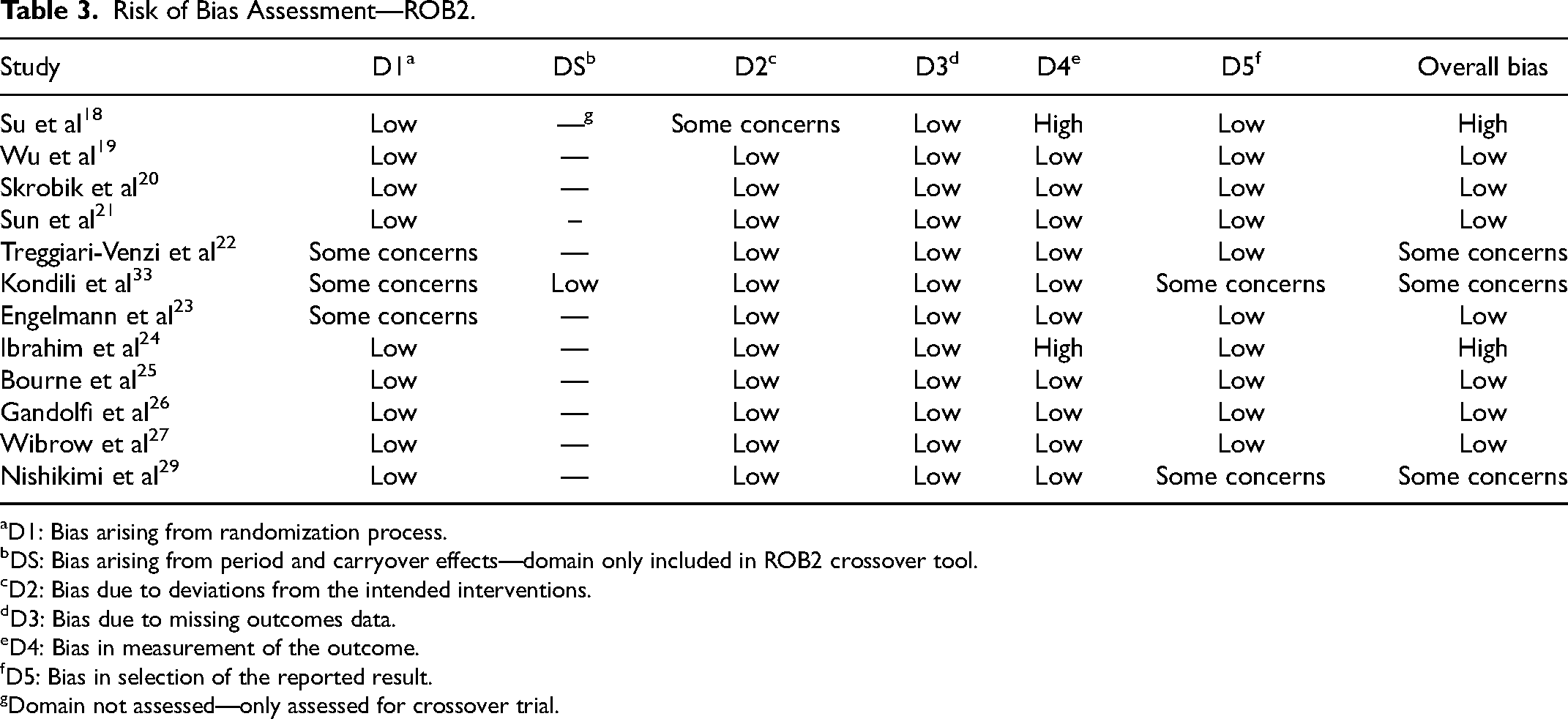
D1: Bias arising from randomization process.
DS: Bias arising from period and carryover effects—domain only included in ROB2 crossover tool.
D2: Bias due to deviations from the intended interventions.
D3: Bias due to missing outcomes data.
D4: Bias in measurement of the outcome.
D5: Bias in selection of the reported result.
Domain not assessed—only assessed for crossover trial.
Risk of Bias Assessment—ROBINS-I.
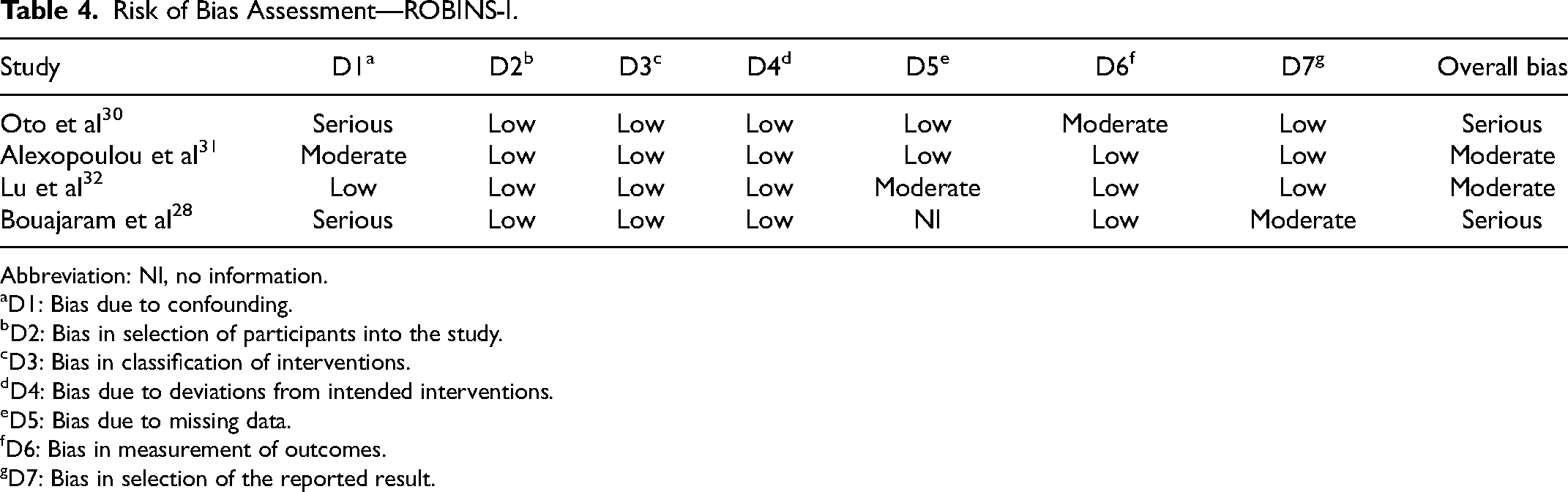
Abbreviation: NI, no information.
D1: Bias due to confounding.
D2: Bias in selection of participants into the study.
D3: Bias in classification of interventions.
D4: Bias due to deviations from intended interventions.
D5: Bias due to missing data.
D6: Bias in measurement of outcomes.
D7: Bias in selection of the reported result.
Discussion
In a review of 16 studies evaluating a medication to improve ICU sleep that enrolled 2573 patients, we found most studies (10/16, 62.5%) reported a significant improvement in at least 1 sleep-related outcome. The greatest sleep benefit appears to occur with dexmedetomidine, where 5 of 7 studies reported significant improvements in sleep efficiency, duration, and quality.18–20,31,32 Notably, most dexmedetomidine trials (4/5, 80%) demonstrated improved objectively-measured sleep outcomes.19,20,31,32 Melatonin agonists inconsistently improved any sleep outcome (3/6, 50% trials); only 1 trial objectively evaluated sleep quality.25,26,29 Propofol appears to adversely affect sleep architecture and as recommended in the 2018 PADIS guidelines should not be used to improve sleep. 4
Although we identified studies where dexmedetomidine, and to a lesser degree melatonin, appeared to improve 1 or more outcomes associated with improved sleep, the results of our review does not suggest that either of these agents should be routinely administered in the ICU solely to improve sleep until additional research where sleep improvement (vs delirium reduction) is the primary study outcome.18–20,25,26,29,31,32 Although safety was not rigorously evaluated in some studies,21,30 dexmedetomidine-associated hemodynamic effects were commonly reported (6/7, 86%).18–21,31,32 These safety concerns may occur more frequently outside a carefully-monitored controlled trial or when the administered dose of dexmedetomidine is higher (eg, around- the-clock sedation during mechanical ventilation).18–21,36
While ICU sleep and circadian disruption is an important therapeutic target for improving outcomes in critically ill adults, important technical and feasibility issues have limited research in this field. 11 The ICU sleep and circadian domains most related to patient-centric outcomes remain unclear, the natural history of sleep disruption in the ICU and post-ICU settings remains poorly elucidated, and longitudinal measures to rigorously evaluate sleep and circadian function that are feasible to use in a large number of patients have not yet been fully developed.11,35 Even patient-perceived sleep quality, the most commonly used sleep outcome in the studies in our review can only be evaluated in patients who are awake and without delirium. 35 Therefore, the evaluation of medications to improve individual domains of physiologic sleep and circadian rhythm remains at an early stage.
Our focus on including only studies reporting sleep-related endpoints and not those only reporting delirium likely influenced our ability to fully evaluate the potential benefits of sleep medication use in the ICU. The relationship between ICU sleep quality, circadian rhythm normality, and delirium remains complex. 37 In 1 previously published systematic review evaluating the effect of ICU sleep medications on delirium and related outcomes (but not sleep quality), 6 of the 10 studies demonstrated significantly reduced delirium incidence, 4 demonstrated reduced delirium duration, and 2 showed a reduced ICU length of stay. 38 Although delirium (and its related outcomes) remain important patient-centered outcomes, it remains important to determine whether the administration of nocturnal sleep aid medications actually improves ICU sleep. 11
The frequent prescribing of medications that have limited data supporting their use for sleep improvement in the ICU (eg, antipsychotics, antidepressants, and benzodiazepines) 8 is concerning given these agents may be associated with increased delirium and important safety concerns, particularly when they are used on a long-term use or in the elderly.39,40 Despite these concerns, and a lack of data demonstrating they improve sleep,12,13 these medications are frequently newly-initiated in the ICU at night to improve sleep to treat agitation associated with delirium and continued after hospital discharge. 8
In our systematic review, we focused on the effects of pharmacological modalities on ICU sleep and therefore excluded studies evaluating both sleep aid medications and non-pharmacologic sleep improvement interventions. 4 Contemporary systematic reviews suggest (albeit with a generally weak level of evidence) the use of individual non-pharmacological sleep improvement modalities (earplugs, eye masks, and music) may reduce delirium (and improve sleep) in critically ill adults.41–43 The greatest benefit is thought to occur when non-pharmacologic interventions are combined as part of a multicomponent sleep promotion protocol, that is, “bundled care.” 44 However, only about half of the studies in our review employed a non-pharmacological sleep promotion protocol.18,21,23,25,29–33
Our review has limitations. Unlike prior pharmacologic sleep modality reviews that have used delirium as the primary outcome, 38 which can be easily measured and results in a dichotomous outcome, our review focused on sleep quality, for which multiple different objective and subjective methods exist for its measurement. 35 The relationship between different sleep assessment methods and the multiple domains by which “good sleep” in the ICU can be characterized remain an active area for research. 11 The quality of reporting for some trials was insufficient to permit definitive judgment for all risk of bias domains. While our review focused on RCTs, most of these studies were small and multiple potential sources of risk of bias were identified. Results from before-and-after cohort studies and RCTs cannot be compared. Sleep quality was not the primary study outcome for many included studies. Most studies evaluated subjective sleep quality; only 25% evaluated sleep quality objectively. Invasively mechanically ventilated patients, who are usually sicker, were poorly represented in most studies. Significant between-study heterogeneity precluded pooling for any outcome. The lack of individual patient data prevented subgroup analysis.
Our results highlight important areas for additional research. 11 Medications for sleep need to be evaluated in more patients at the greatest risk for sleep and circadian disruption (older adults, mechanically ventilated patients with a high severity of illness). The timing and duration of medication interventions relative to ICU admission and the presence of disrupted sleep or delirium needs to be carefully evaluated. A beneficial effect of nocturnal melatonin may occur in the absence of other interventions focused on improving circadian entrainment (eg, daytime light and mobility) or be detected without the use of sensitive measures of circadian function (eg, urine melatonin concentrations, day vs night heart-rate variability).11,45 Future RCTs should ensure that basic sleep interventions are employed as a “standard of care” bundle (eg, avoiding routine care at night unless medically necessary, turning off lights, limiting noise, retiming nocturnal medication administration) to support circadian entrainment and restore normal day-night physiology using environmental Zeitgebers in both intervention and control arms. 46 Sleep assessment remains a limiting factor for measuring sleep-related outcomes. 35 Only RCSQ has been validated formally for sleep assessment in ICU patients. In non-ICU patients, the Insomnia Severity Index and PSQI are validated tools, but application to the ICU environment would have practical challenges. 11 With the current lack of a feasible ICU sleep assessment method that can evaluate physiological sleep, circadian rhythm, and patients’ perception of their sleep, 35 future studies should consider incorporating at least 1 subjective (eg, RCSQ) and 1 objective (eg, actigraphy) sleep assessment methods. Ideally, both sleep quality and quantity should be measured to obtain the most comprehensive evaluation. 11 Our review suggests dexmedetomidine and melatonin agonists are the most promising pharmacologic candidates for future research.18–20,25,26,29,31,32 Suvorexant and other orexin receptor antagonists are also important candidates for ICU sleep research given they have been found to be beneficial for chronic insomnia, may help prevent delirium, and have a relatively benign safety profile.47,48
Conclusions
This systematic review of 16 trials including 2573 patients demonstrates that dexmedetomidine and melatonin are the most studied pharmacologic modalities used to promote sleep in critically ill adults. However, due to the high number of methodological issues identified across studies, including the variability by which potential factors affecting sleep quality were considered and the multiple different ways sleep quality was evaluated, we are unable to suggest the routine use of any pharmacologic modality to improve sleep in the ICU. Additional, well-designed RCTs, that carefully consider baseline patient risk for disrupted sleep and incorporate a robust non-pharmacologic sleep improvement bundle, are needed to elucidate the roles for medications to improve sleep in the ICU.
Supplemental Material
sj-docx-1-jic-10.1177_08850666231186747 - Supplemental material for A Rapid Systematic Review of Pharmacologic Sleep Promotion Modalities in the Intensive Care Unit
Supplemental material, sj-docx-1-jic-10.1177_08850666231186747 for A Rapid Systematic Review of Pharmacologic Sleep Promotion Modalities in the Intensive Care Unit by Mojdeh S. Heavner, Patricia R. Louzon, Emily F. Gorman, Kaitlin M. Landolf, Davide Ventura and John W. Devlin in Journal of Intensive Care Medicine
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.S.H., P.R.L., E.F.G., K.M.L., and D.V. have nothing to report. J.W.D. reports the following: Research funding—NIA, AHRQ, Sedana Medical; Consulting—Trevana Medical, Ceribell Inc., BioExcel Pharma.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
