Abstract
Introduction
Paediatric intensive care units (PICU) present a myriad of complex and frequently dynamic challenges for patients, their families and healthcare providers. Overuse of analgesia and sedation in a PICU settings is associated with a high risk of developing PICU-acquired complications and poorer outcomes including delirium, prolonged length of stay, longer ventilation times, neuromuscular atrophy and weakness, post-traumatic stress disorder and increased levels of drug tolerance, dependence, and withdrawal syndrome.1–3
The goal of pain management in PICU settings is to improve patient comfort and reduce associated physiological stress responses while managing risks such as respiratory depression, addiction, haemodynamic instability, delirium, and end organ injury.2,4,5 Children admitted to PICUs experience on average >10 painful and/or distressing procedures per day.1,6–8 This number significantly increases for children who are mechanically ventilated. 1 In addition to pain and psychological distress the deleterious and often unavoidable effects of patient-ventilator dyssynchrony, patient care interactions and environmental factors in the PICU environment places critically ill children at high risk of sleep deprivation.9–11 Poor sleep quality and quantity characterised by increased sleep fragmentation and decreased quantities of slow wave sleep are commonly associated with PICU-acquired complications.3,11,12
Rationale
Guidelines currently recommend the adoption of strategies with emphasis on non-pharmacological options in all PICU patients. 3 In the absence of a commonly accepted definition, this review refers to Extended Reality (XR) as the collective term for Virtual Reality (VR), Augmented Reality (AR), and Mixed Reality (MR). XR interventions offer potential non-pharmacological solutions to challenges such as pain,13,14 sleep disturbances 15 and anxiety. 14 However, without a systematic assessment of the evidence, the widespread uptake of XR in PICUs is unlikely to occur. 16
A scoping review was undertaken to allow for a broad investigation into the use of XR within PICUs guided by the following research question:
How has XR been utilised within paediatric intensive care units (PICUs)? Purpose of using XR in PICUs What types of PICUs have XR technology been utilised? What are the characteristics of participants? What types of XR hardware and software have been utilised? What cleaning/infection control policies/procedures are utilised for XR in PICUs? What outcome measures are being used to explore/investigate the use of XR in PICU? What are the experiences of participants (patients, caregivers, and healthcare providers)? Have adverse effects been reported in the treatment of patients in PICUs?
The following sub-questions are utilised to provide direction and further analysis of identified articles:
Methodology
Protocol and Registration
A priori protocol was developed in line with recommendations by the Joanna Briggs Institute (JBI) and the Prisma extension for scoping reviews reporting guidance (PRISMA ScR).17,18 The protocol was registered prospectively prior to the initiation of screening with Open Science Framework and updated on the 05/10/2022 (DOI:10.17605/OSF.IO/HVCAE).
Eligibility Criteria
A pre-determined eligibility criteria was developed informed by the population (any population), concept (utilising XR technology) and context (within a paediatric ICU) framework. 18 Pre-determined definitions for the terms paediatric, intensive care facility, VR and AR, outlined within the protocol, guided eligibility criteria development and exclusion processes. To ensure a broad scope of the literature was undertaken no research methodology was excluded. Due to feasibility only articles published in English were included.
Search Strategy
A search strategy was developed with the assistance of a medical research librarian in line with the following three-step methodological approach outlined by the JBI: a) a preliminary literature search was undertaken in PubMed and Google Scholar, b) additional search terms were identified and search strategies translated with the assistance of a validated search engine translation software (Polyglot), 19 c) execution of final search strategies (Appendix 1).
Information Sources
Four databases (PubMed, EMBASE, PyshInfo and CINAHL) were searched on the 16/09/2022. Google Scholar was searched, and the first 100 articles were exported on the 11/10/2022. Results from database searches were exported into Endnote X9. 20
Selection of Sources of Evidence
Duplicate results were removed within systematic review accelerators validated deduplication software utilising the cautious algorithm prior to being manually reviewed. 21 Articles were screened by two authors (AG & SF) by title and abstract within Systematic Review Accelerators Screenatron. 21 Full-text screening was undertaken within Covidence with discrepancies identified and resolved by a third author (SG) within Covidence. 22
Data Charting Process
A draft extraction table was developed within Microsoft Excel prior to data extraction to align with the scoping reviews research questions. Final extraction of data was undertaken by two authors (AG & SF), before manually being compiled and discrepancies resolved.
Data Items
The extracted data items are available within the data extraction template (Appendix 2). Only information described within the manuscript and supplementary documents were data extracted. Information not provided was recorded as “not described”.
Critical Appraisal
Critical appraisal of the quality of the included articles was undertaken by two authors (AG & SG) utilising the mixed methods assessment tool (MMAT) 23 and guidance provided by Hong et al, 2018. 24 Discrepancies were resolved by a third author (OB). Data relating to the quality of intervention reporting was undertaken utilising the TIDieR checklist. 25
Synthesis of Results
Data pertaining to the frequency of countries, year of publications, setting, barriers, facilitators, safety considerations and cleaning and infection protocols were tabulated within Microsoft Excel, in alignment with the research questions.
Results
Selection of Sources of Evidence
Database searching led to the retrieval of 188 articles of which 33 were automatically removed as duplicates. Title and abstract screening of the remaining 155 articles led to the exclusion of 102 articles. Full-text screening of the remaining 53 articles was undertaken with substantial agreement between authors (Cohen's Kappa = .66). Following the exclusion of a final 37 articles, 16 articles were included within our scoping review. A summary of the retrieval and screening process with reasons for exclusion has been displayed within a PRISMA flow diagram (Figure 1).
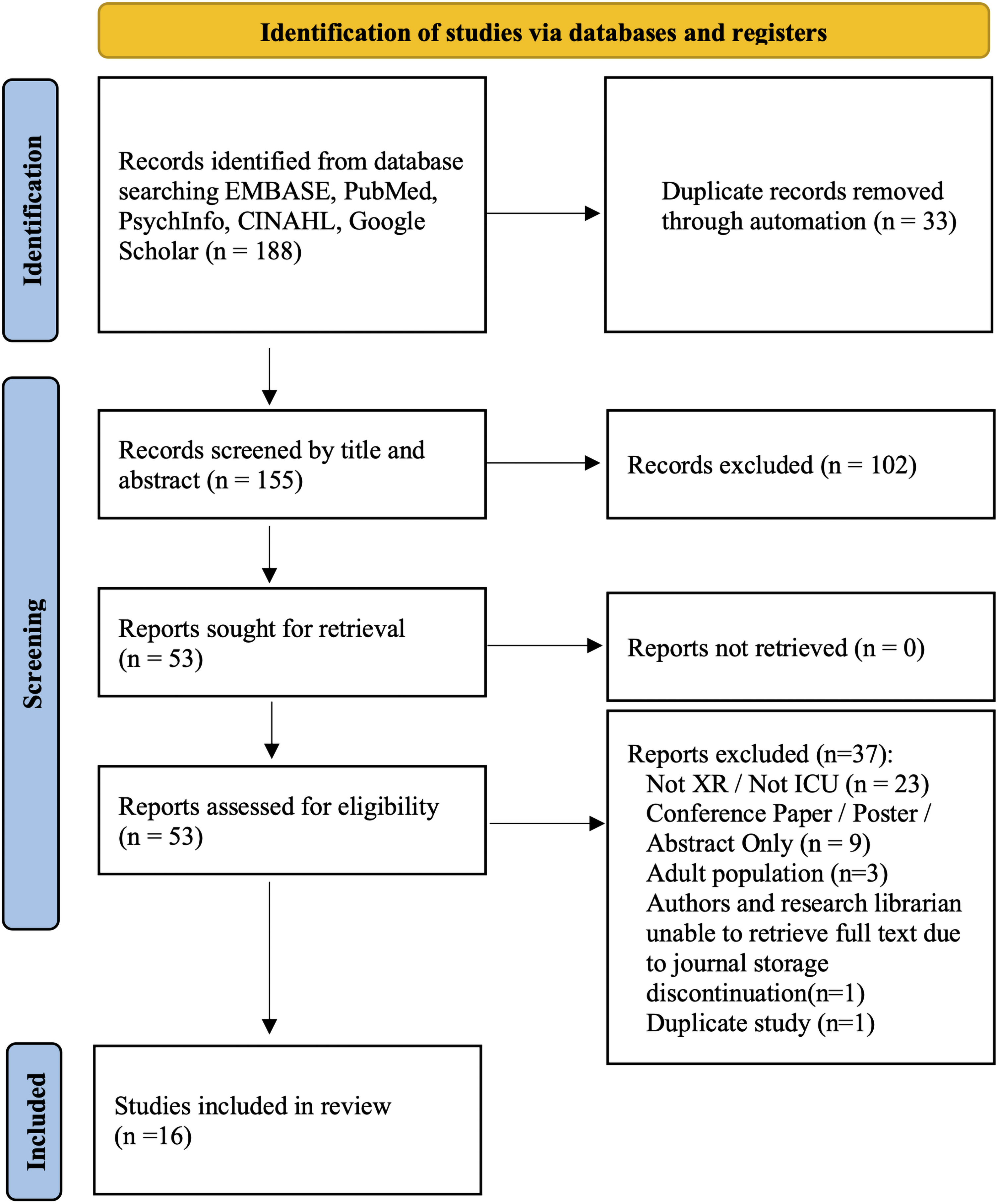
PRISMA flow diagram.
Critical Appraisal Within Sources of Evidence
The methodological quality of included articles was found to be of variable quality (Table 1).
Mixed Methods Assessment Tool (MMAT) Critical Appraisal Decision.
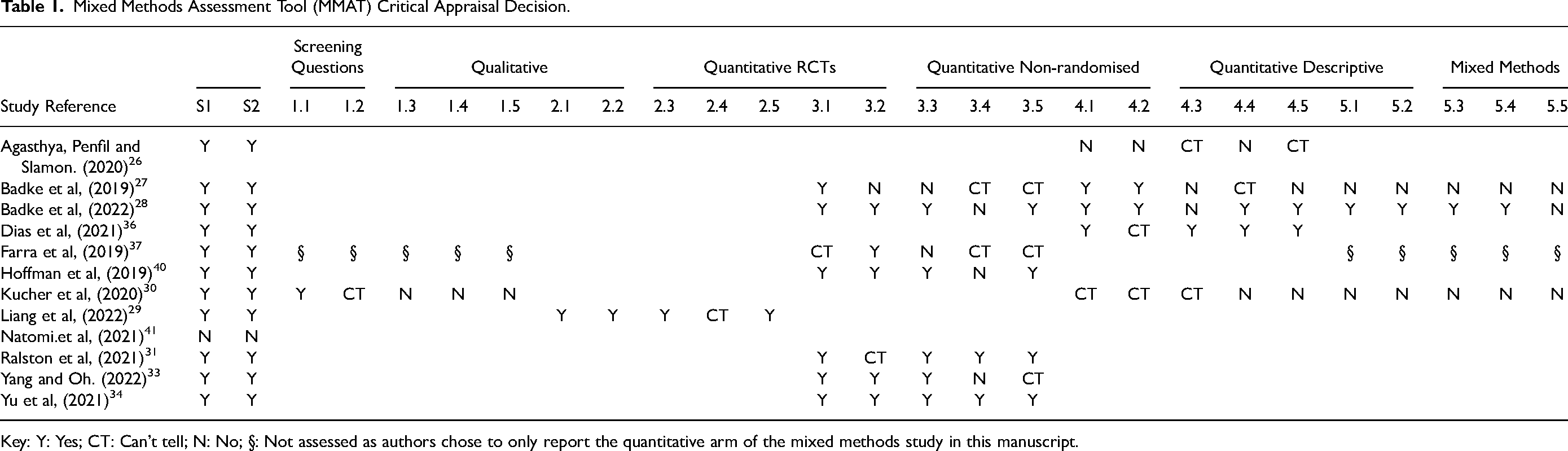
Key: Y: Yes; CT: Can’t tell; N: No; §: Not assessed as authors chose to only report the quantitative arm of the mixed methods study in this manuscript.
Synthesis of Results
The majority of included publications were published since 2019 (Figure 2). Extended reality was utilised for the purposes of medical education and clinical interventions (Figure 3). Only three countries, The United States, China, and South Korea were represented. Articles were primarily of a quantitative (n = 7) or mixed methods (n = 6) design, with a case study, protocol and a cost analysis representing the remainder. No systematic reviews or meta-analysis met our eligibility criteria.
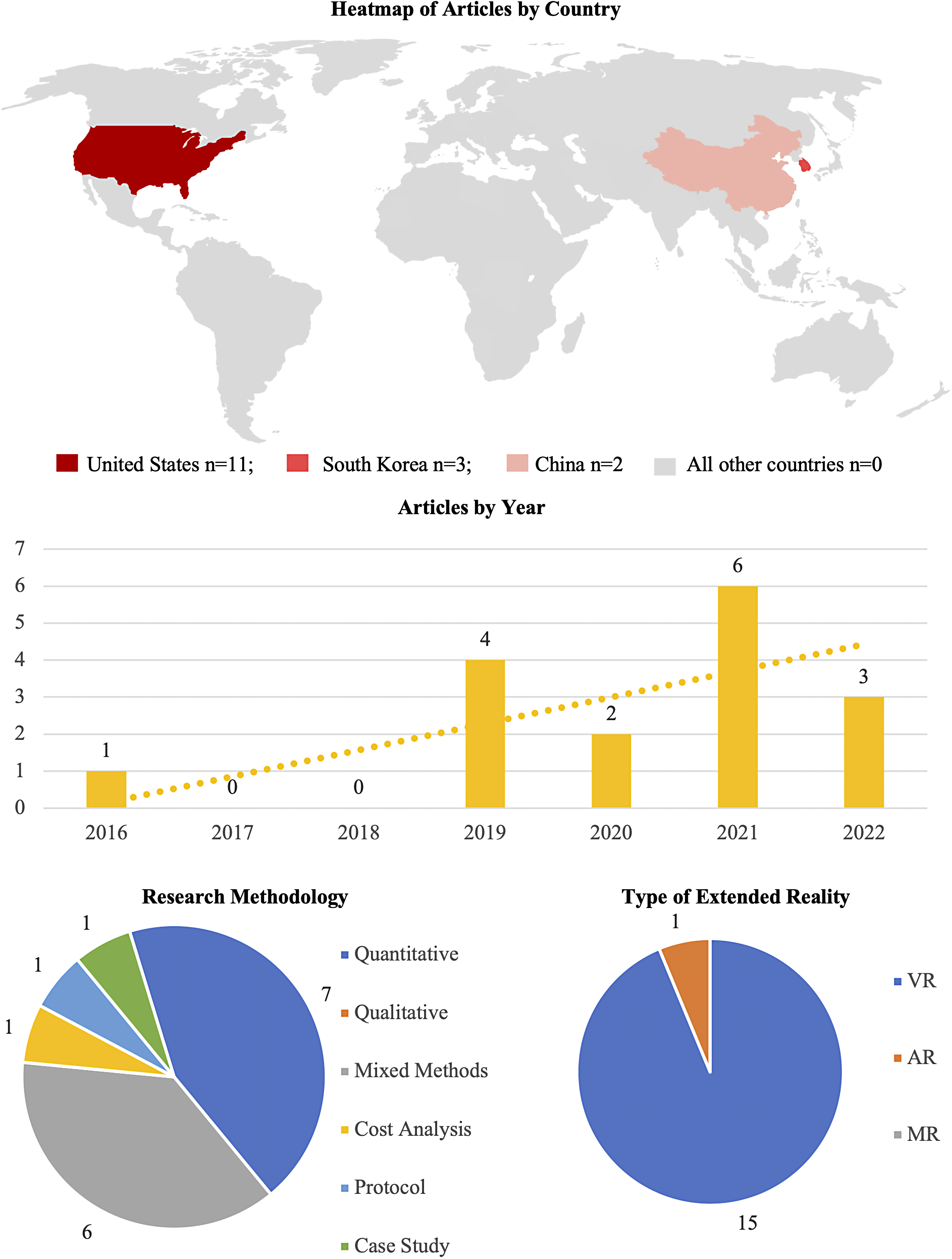
Articles by country, research methodology and type or extended reality.
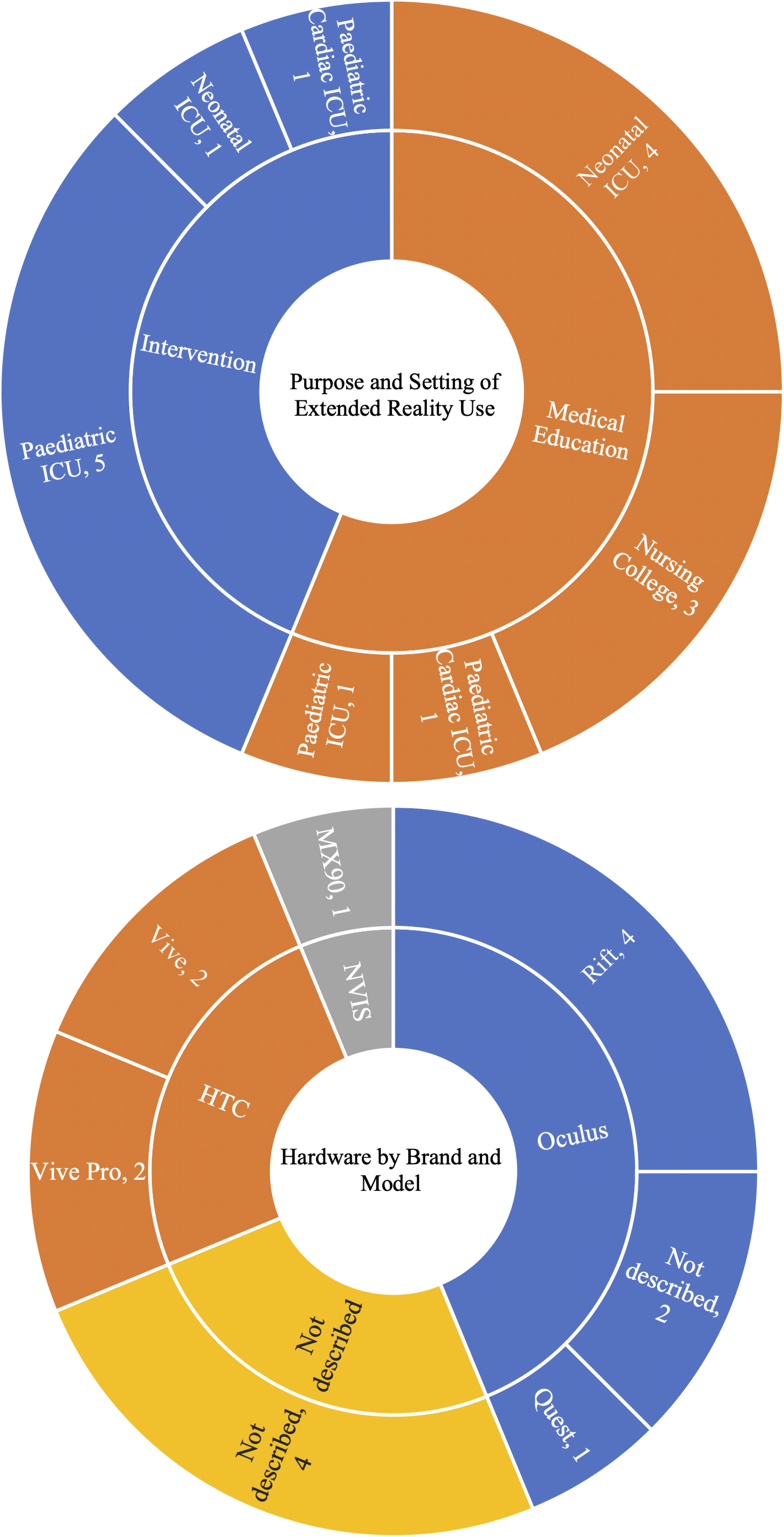
Purpose and setting of extended reality use and hardware by brand and model.
Types of Intensive Care Facilities
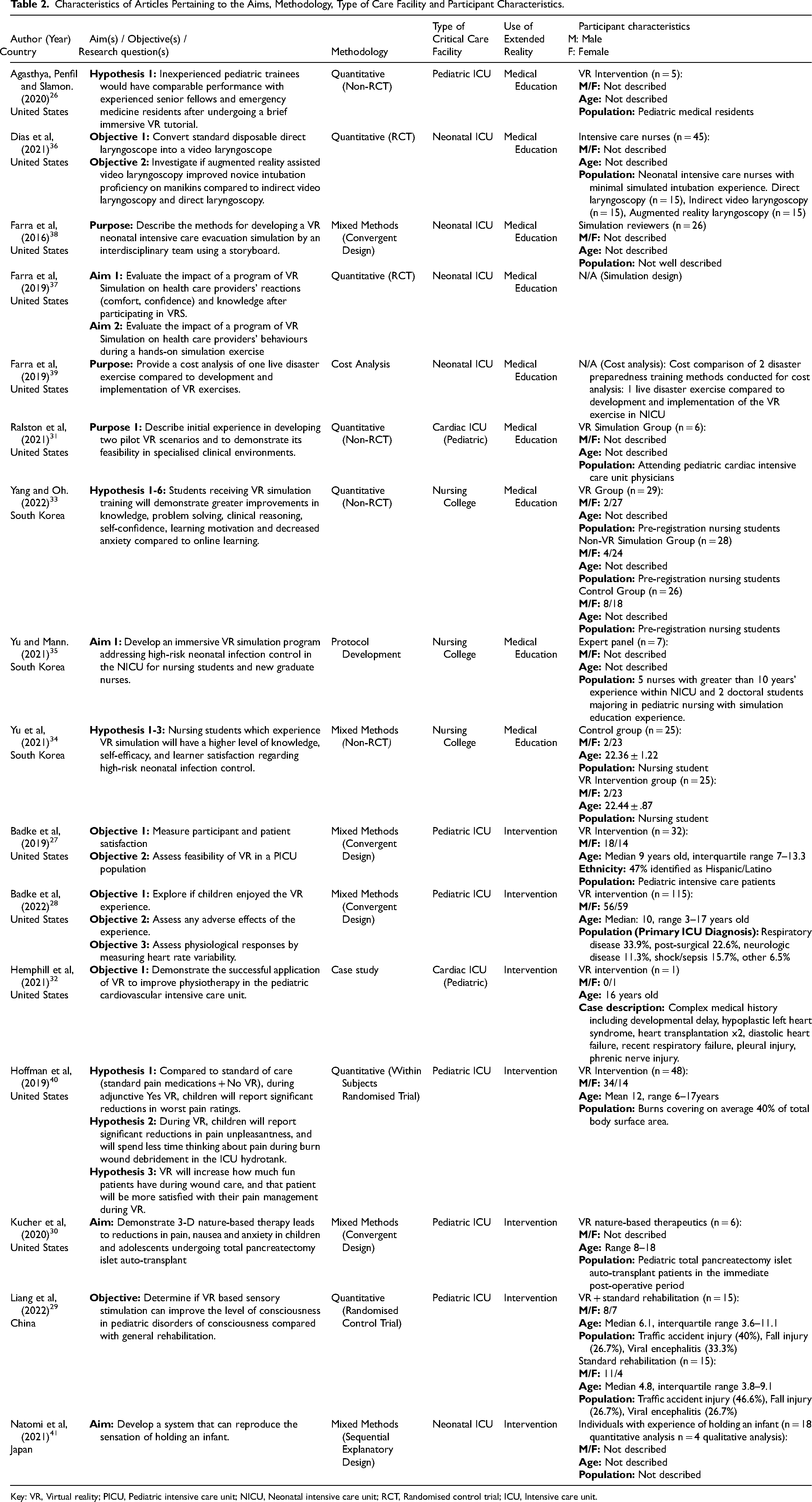
Three paediatric clinical settings; intensive care,26–30 neonatal intensive care and cardiac intensive care unit31,32 were described. Three articles utilised XR within a nursing college setting for the purposes of medical education.33–35 Neonatal intensive care was the most common setting for utilising XR for medical education.36–39 Comparatively, paediatric intensive care was underrepresented with only one article identified relating to medical education. 26 Conversely, PICUs represented the majority of intervention articles.27–29,40
Uses of XR in Medical Education
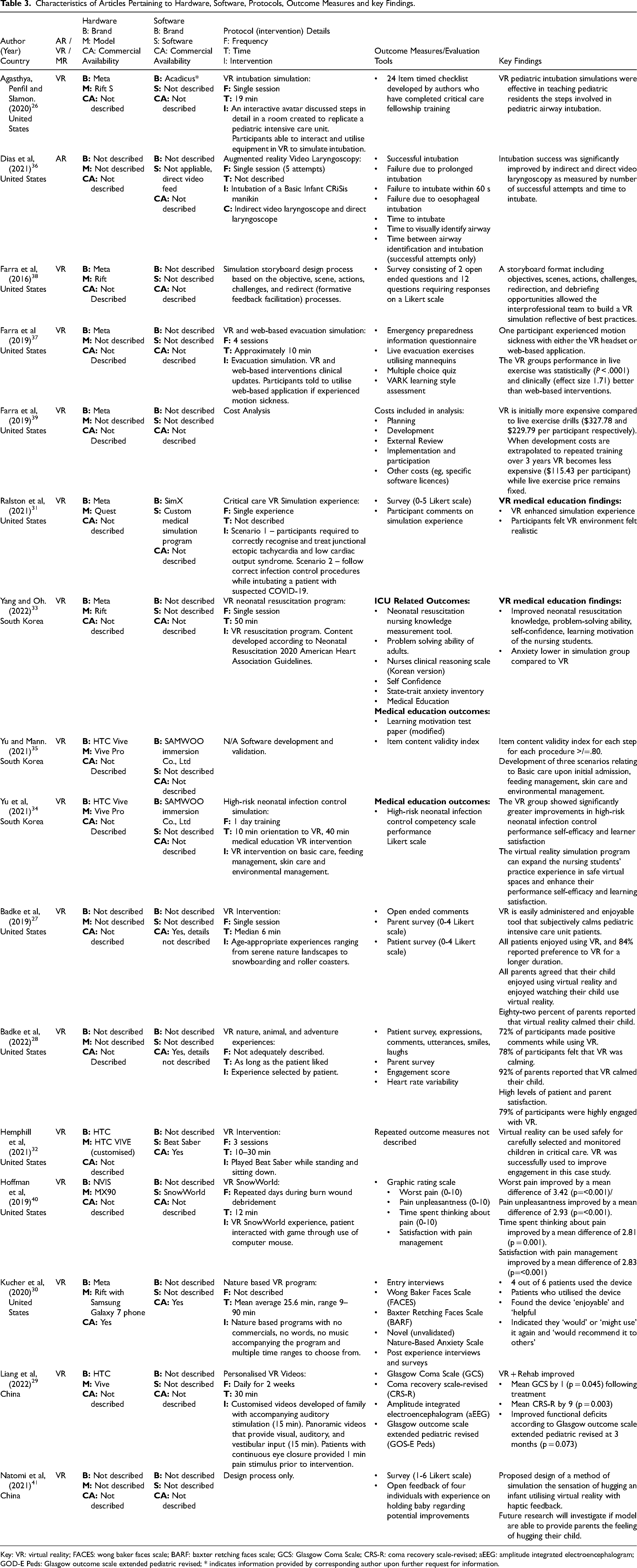
Nine articles were identified which utilised XR within medical education (Figure 3). The participant population was highly heterogenous including paediatric cardiac intensive care physicians, 31 paediatric medical residents, 26 neonatal intensive care nurses,35,36 and pre-registration nursing students33,34 (Table 2). No articles looked at the use of XR for patient and family education. Articles which used XR for the purposes of medical education pertained to four clinical skills: intubation,26,31,33,36 recognising and responding to a deteriorating patient, 31 neonatal ICU evacuation procedures 37 and high-risk neonatal infection control34,35 (Table 3). Healthcare providers reported that XR was easy to set up and required little space while improving learner satisfaction.26,31 However, haptic feedback within simulations of medical procedures was reported to be hard to reproduce accurately. 26 Farra et al, detailed the simulation design process, 38 implementation 37 and provided a cost analysis 39 of their simulation. No medical education articles utilised XR to assess performance or knowledge acquisition instead relying predominantly on surveys and expert examiners who evaluated performance (Table 3). Numerous benefits to the use of XR were noted (Table 4), primarily relating to learning outcome achievement and satisfaction, as well as areas of feasibility relating to cost and ease of delivery.
Characteristics of Articles Pertaining to the Aims, Methodology, Type of Care Facility and Participant Characteristics.
Key: VR, Virtual reality; PICU, Pediatric intensive care unit; NICU, Neonatal intensive care unit; RCT, Randomised control trial; ICU, Intensive care unit.
Characteristics of Articles Pertaining to Hardware, Software, Protocols, Outcome Measures and key Findings.
Key: VR: virtual reality; FACES: wong baker faces scale; BARF: baxter retching faces scale; GCS: Glasgow Coma Scale; CRS-R: coma recovery scale-revised; aEEG: amplitude integrated electroencephalogram; GOD-E Peds: Glasgow outcome scale extended pediatric revised; * indicates information provided by corresponding author upon further request for information.
Barriers and Facilitators Relating to Medical Education, Clinical Interventions, and Other Considerations.

No barriers to clinical interventions or other facilitating considerations were reported in the literature. As a result, these cells have been intentionally left blank.
Uses of XR in Medical Interventions
Seven articles were identified which utilised XR for medical interventions (Figure 3.). Virtual reality was the only XR modality used as a medical intervention in PICU patients. Patient populations were inclusive of a wide range of primary conditions including respiratory, cardiac, and neurological disorders, burns, sepsis, and individuals admitted post-surgery. Patient populations included children between the ages of 3 and 18 who were either intubated or non-intubated and had varying levels of consciousness.
Virtual reality was most commonly used to improve psychological outcomes and decrease pain and discomfort. Hemphill et al, 2021, utilised the experience Beat Saber during physiotherapy to improve engagement in undertaking active proximal trunk strengthening exercises leading to improvements in independent sitting balance and reduced barriers to discharge. 32 Liang et al, (2022) utilised customised videos with accompanying audio from families to try to improve the level of consciousness in patients with low Glasgow Coma Scale scores. 29 A potential lack of engagement resulting from low levels of consciousness was addressed through the use of pain provocation prior to the initiation of a salient XR experience co-designed and produced with family members. Other interventions utilised VR to provide a wide range of experiences including nature-based scenes, fun experiences such as roller coasters and environments with therapeutic design elements such as SnowWorld.27–30,40 The number and length of interventions varied from single sessions to daily for two weeks, 6 to 90 min and were tailored by selecting age-appropriate experiences and allowing patients to select the length and content within the experience.27,28
Types of Hardware and Software Utilised Within Paediatric Intensive Care
Three brands of head mounted displays were identified: Meta (previously known as Oculus), HTC and NVIS (Figure 2). Articles utilising a Meta device either used a Rift,26,30,33,38 Quest 31 or failed to describe the model.37,39 Articles utilising a HTC device either used a Vive29,32 or Vive Pro.34,35 Hoffman et al 40 were the only authors to use NVIS MX90. Four articles did not describe the brand or model of the headset.27,28,36,41 Three articles detailed that software was commercially available.27,28,32 One article used the game Beat Saber. 32 Badke et al, used a commercially available nature-based video however, lacked specific software details enabling the experience to be identified further.27,28
Cleaning and Infection Control Procedures
Four articles described cleaning and infection control procedures (Table 5). Cleaning protocols consisted of disposable covers,34,35 utilising anti-bacterial wipes 29 and UV-C wands. 40 Hoffman et al, was the only article that indicated their cleaning protocol had been internally validated within the hospital. 40
Considerations Relating to Exclusion Criteria and Safety Protocols.

Outcome Measures to Explore/Investigate the Use of XR in PICU
Primary outcome measures were highly heterogenous (Table 3). No validated outcome measures were utilised to ascertain the level of ‘presence’ or cybersickness users experienced during exposure (Table 3). One paper measured nausea with the Baxter Retching Faces Scale. 30 The experiences of healthcare professionals, patients and caregivers were most often evaluated with surveys.27,28,30,31,38,41 No studies evaluated the experience of healthcare providers experiences providing XR interventions to patients.
Experiences of Patients, Caregivers, and Healthcare Providers
High patient satisfaction was reported by Kucher et al. 30 At times, the experience of paediatric patients was difficult to assess due to impaired consciousness or cognition. 29 Clinical judgements of the patients’ healthcare providers or caregivers were utilised in circumstances to therefore assess the patients’ experience.27–29 In line with existing literature, 42 XR interventions were well accepted by caregivers with 99% of parents in the article by Badke et al, 28 reporting they agreed or strongly agreed that they enjoyed watching their child during the intervention.
Safety Considerations and Adverse Effects
Healthcare providers reported that the use of VR in clinical interventions was safe (Table 5). Protocols for screening, monitoring, and ceasing VR interventions were highly inconsistent between articles or absent. Potential absolute contraindications or precautions were highlighted within the literature (Table 5). However, there was poor consensus as to what should be considered a contraindication. Virtual reality was reported to be safe and effective 28 with high patient satisfaction. 30 Clinicians monitored patients during interventions subjectively (signs of distress), objectively (heart rate) and through questions where appropriate. 28
Reporting Quality of Interventions
The reporting of interventions was poor within a number of domains (Table 6). No articles provided enough details regarding hardware and software to replicate the intervention. No articles fully described the training and experience level of staff with utilising VR with patients.
TIDieR Checklist.

Discussion
Summary of Evidence
To our knowledge, this is the first review to provide insights into the utilisation of XR within PICU, including a thorough exploration of the types of facilities, patient populations, the hardware and software, safety considerations, caregiver experiences, and adverse effects associated with the delivery of XR in PICU.
The paucity of research identified demonstrates that while research into XR in this clinical setting is increasing, widespread use has still not occurred. Identified articles concentrated on the use of XR for two key purposes; 1) for medical education and 2) to assist with clinical interventions to decrease pain and anxiety in a variety of paediatric intensive care settings.
Uses of XR in Medical Education
Extended reality use within medical education presents PICU staff an opportunity to learn and practice skills and procedures such as intubation which are otherwise challenging and expensive for PICU staff to gain sufficient experience and proficiency through traditional methods. 35 Difficulty in simulating realistic haptic feedback, however, remains a major barrier to the development of real-world proficiency. 26 Articles included in this review utilised XR equipment that has been superseded in recent years by technology able to more accurately provide realistic haptic feedback. As haptic technology becomes cheaper and more accessible the transferability of PICU specific procedural skills learnt within a XR environment to the real world may improve.
Use of XR in Medical Interventions
The diverse nature of patient ages, primary diagnosis and acuity within the PICU setting requires interventions to be able to be adapted to individual patient requirements and clinical goals. 43 Decreasing pain and psychological distress were the most common clinical goals identified. Research to date lacks the appropriate outcome measures and study designs to determine if improvements in patient pain and anxiety result in reduced requirements for sedatives and analgesics, improved sleep or reduces the rates of or severity of complications such as delirium. Moving forward it is important clinicians and researchers develop an understanding of XRs potential mechanisms in order to maximise the effectiveness of these interventions.
Distraction, or the allocation of cognitive resources away from the painful stimulus, is commonly cited as the primary mechanism of VR-mediated pain relief.44,45 However, modulation of stress responses with XR has the potential to influence pain perception and tolerance through the overlap of spinal and supraspinal neural substrates of stress and pain.46–48 Further research is currently required to determine if highly engaging and distracting experiences (eg, roller coasters) or relaxation experiences are more protective against negative outcomes associated with various common PICU scenarios and procedures.
Visual cues resulting from witnessing a caregiver's emotional reaction or identifying a potential threat (eg needle) can also modify a child's experience of pain.49,50 Extended reality may therefore assist in modifying pain by providing alternate visual input and occluding the child's vision of potentially distressing visual cues. Additionally, healthcare provider interaction with the patient may reduce sensations of presence altering clinical effectiveness. Clinicians and researchers need to improve reporting to clarify intervention procedures and assist in determining how interacting with patients while using XR or preventing patients from viewing painful stimuli (eg, needle) with the use of XR modifies procedural anxiety and/or pain.
The nature and context of the visual input are also of potential importance. 44 The experience SnowWorld40,51–53 is an example of how XR experiences can be tailored to improve outcomes associated with clinical goals. Research in pain science relating to colours and the contextualisation of information relating to pain suggests the depiction of the blue and snowy landscape within SnowWorld40,51–53 is of clinical significance. 54 Moreover, VR has the potential to indirectly reduce a child's experience of pain by reducing the deleterious effects of sleep deprivation on subjective ratings of pain, their pain threshold and activation within their somatosensory cortex.55,56
Hardware Considerations
The hardware and software utilised within the identified literature were poorly reported impacting on the potential for research to be translated into clinical practice. To enable the wider integration of XR into PICUs hardware must be designed carefully to overcome potential barriers. Devices made of easily cleanable materials that do not incorporate Velcro into the design of the headstrap and minimise unnecessary cracks and crevices will assist healthcare providers in ensuring adequate infection control in a way that allows for efficient and effective cleaning protocols. Additionally, hardware must be able to fit a wide range of age groups. Headstraps that are easily fitted and comfortable in a supine position may also provide benefits within an intensive care environment to prevent unwanted cervical movement or disruption of attachments. Due to poor reporting and a lack of use of new XR hardware, it is unclear if more recent XR hardware is appropriate for use within the PICU. Researchers and clinicians should endeavour to utilise new technology to ensure research can be translated effectively into clinical practice.
Cleaning and Infection Control Policies and Procedures
Cleaning and infection control protocols were poorly reported in some articles preventing replication of interventions. Where reported infection control practices utilised antibacterial wipes and UV-C in line with the findings of a recent survey of VR hygiene practices by Hoeg and Lange., 2022. 57 Strict adherence to infection control measures are important in preventing hospital associated infections and associated increases in morbidity, mortality and healthcare expenditure. 58 Hoffman et al, 2019 40 described the use of a UV wand. Clinicians and researchers should consider that UV-C wands may be unsafe with the FDA reporting some disinfection wands emitting 3000 times the safe limit of international standards. 59 Additionally, Roberts et al, 2022 found UVC failed to provide adequate disinfection. 60 Further research is therefore required to adequately detail if current UV-C technologies can effectively clean XR devices. This will be an important step in the development of new industry evidenced-based regulations and cleaning protocols which meet healthcare standards.
Participants Excluded from Participation in Studies
Participants were excluded from VR interventions according to four main categories: Pharmacological considerations, medical history, active medical conditions, and practical considerations.27–29,32,40 None of the included studies in this review featured AR interventions. Contrasting viewpoints exist within the literature pertaining to the need to exclude patients with a medical history (eg, epilepsy), 32 and active medical conditions such as neurological conditions.27–29,40 Hemphill et al, excluded individuals with a history of epilepsy 32 despite existing literature suggesting VR use is not a risk factor for individuals (including children) with a history of epilepsy or photo paroxysmal responses.61,62 Similarly, patients with recent neurological injuries were at times excluded from trials featuring VR.27,28 Liang et al,(2022) reported no serious complications requiring medical intervention or an escalation of care in this population. 29 The exclusion criteria identified within this review (Table 5) may assist clinicians and researchers in developing screening and exclusion protocols for use in PICU. To ensure future screening and exclusion protocols are appropriate and not overly restrictive it is recommended clinicians critically analyse the literature and utilise clinical reasoning, informed by an understanding of the psychological and neurophysiological impacts of VR.
Adverse Effects and Outcome Measures
Consistent with the wider literature adverse effects reported within included articles were rare, mild, transient in nature and related to symptoms of cybersickness (eg, nausea, dizziness) or neck discomfort. 28 Despite experiencing “mild discomfort” no patients requested the discontinuation of their intervention in a cohort of 115 PICU patients. 28 While all the mechanisms behind cybersickness have yet to be fully elucidated, research has demonstrated that reducing conflicts between sensory (vestibular, visual tactile etc) and predictive modelling systems is important. The users’ position (eg, supine, sitting or standing) may therefore contribute to any adverse effects. 63 Thoughtful design and thorough reporting of intervention protocols may assist in understanding if the adverse effects reported so far in the literature can be mitigated through the use of simple strategies such as progressive habituation, reducing positional and vestibular-ocular conflicts, and using fans. These would then need to be evaluated to ensure feasibility in a PICU setting. Validated outcome measures examining XR specific factors such as ‘presence’ and cybersickness need to be standardised and used for future research within PICU.64,65 Due to a lack of formal outcomes no conclusions regarding the experiences of clinicians utilising XR with patients can be drawn at this time. Qualitative approaches may assist in future to provide insights into patients’ experiences and the effect of XR interventions on clinicians’ decision-making.
Experiences of Patients, Caregivers, and Healthcare Providers
Positive experiences were reported when XR was used in both medical education and clinical interventions in the PICU setting indicating further research may be warranted.26,30,31,37 However, the identified research indicates that to date research has been limited to the experience of the child and the caregivers perception of their child's experience. No research was identified within this review that sought to evaluate if witnessing their child partake in positive XR experiences has a positive effect on caregivers experience and well-being. Additionally, in future further research should investigate how providing XR interventions affect healthcare provider workflow and decision-making.
Gaps and Future Recommendations
Optimal management of pain, delirium, sleep, anxiety and distress are key recommendations within critical care guidelines. 3 While VR has been utilised successfully to improve anxiety and pain in paediatric intensive care,27,28,30,40 no studies included in this review evaluated VRs effect on delirium or sleep as a primary outcome. None of the included articles utilised XR for the purposes of improving caregiver or healthcare professional health-related outcomes. The reporting quality of interventions was poor. The development of a customised TIDieR checklist specific for the use in XR may be of use to assist in standardising reporting in the future. Further research utilising head-to-head study designs, comparing various XR protocols are required to provide further insights into effectiveness of specific design parameters.
Limitations
Despite an extensive search strategy, a limited number of articles was identified within this scoping review. South Korea and China represent two of the three countries in which XR is currently being utilised within PICU. As a result, it is likely that this review has not identified articles written in their primary languages. Rapid increases in research within this field are likely to warrant further review within the near future.
Implications for Integration of XR in PICU
Virtual reality is a rapidly emerging tool for use in patients within the PICU setting. Its use as an education role in this setting is steadily increasing and is associated with positive results. While current evidence suggests VR is safe in PICU populations conjecture remains regarding the use of VR in some populations and in determining the specific role for this tool clinically. Currently, a lack of methodological rigour and head-to-head study designs prevents the identification of optimal VR prescription protocols.
Conclusion
While researchers, educators and clinicians are increasingly seeking to harness the advantages of XR to provide education and develop new interventions in PICU the use of XR in this clinical setting is still in its infancy. To support clinicians in exploring the potential application of XR applications clinical research needs to focus on increasing the understanding of XRs mechanisms, screening, and implementation strategies. To achieve this integrated, multidisciplinary research teams that include clinicians, researchers and industry partners are required. This collaboration will ensure that current gaps in policy, evidence and clinical expertise can be overcome, and that the expertise is available for continual refinement of technology and clinical protocols to provide meaningfully outcomes for PICUs patients and their families.
Footnotes
Acknowledgements
We thank Sarah Bateup Bond University Faculty Health Sciences and Medicine Librarian for assisting in development of search strategies and Sengio Fong for assisting with data extraction.
Author Contributions
Dr Adrian Goldsworthy was involved in the conceptualisation, protocol development, data extraction, data analysis and manuscript drafting and editing.
Dr Jasneek Chawla provided significant contributions to the data analysis, drafting and editing of the manuscript.
Dr James Birt provided significant contributions to the protocol development, data extraction, data analysis, and drafting of manuscript.
Dr Oliver Baumann provided significant contributions to the protocol development, data extraction, data analysis, and drafting of manuscript.
Dr Suzanne Gough was involved in the conceptualisation, protocol development, data extraction, data analysis and manuscript drafting and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1: Search Strategies
Appendix 2: Data extraction form

| Author(s) | Published Year | ||
| Title | |||
|
|
|||
|
|
|||
| Aim(s) / hypothesis / research question(s) of study | Aim 1: |
||
| Methodology | eg, Quantitative (randomised control trial) | ||
| Context / Setting | eg, Inpatient, Outpatient, Research facility | ||
| Use of XR |
|||
| Participant details / characteristics | Group 1: |
||
| Risk of Bias | |||
|
|
|||
| AR / VR / MR | |||
| Hardware | Brand: |
||
| Software | Company: |
||
| Protocol | Intervention 1: |
||
| Outcomes | Primary Outcomes: |
||
| Key Findings | |||
|
|
||||
| Brief Name | 1. Do the authors provide the name or a phrase that describes the intervention? | Yes complete |
||
| Why | 2. Do the authors describe any rationale, theory or goal of the elements essential to the intervention? | Yes complete |
||
| What |
3. Materials: Do the authors describe any physical or informational materials used in the intervention, including those provided to participants or used in intervention delivery or in training of providers, or provide information on where the materials can be accessed (online appendix or URL)? |
Yes complete |
||
| 4. Procedures: Do the authors describe each of the procedures, activities and/or processes used in the intervention, including any enabling or support activities? |
Yes complete |
|||
| Who Provided? |
5. For each category of intervention provider (psychologist and nursing assistant), describe their expertise, background and any specific training given? |
Yes complete |
||
| How? |
6. Do the authors describe the modes of delivery (face-to-face or by some mechanism, such as internet or telephone) of the intervention and whether it was provided individually or in a group? |
Yes complete |
||
| Where? |
7. Do the authors describe the type(s) of location(s) where the intervention occurred, including any necessary infrastructure or relevant features? |
Yes complete |
||
| When and How Much |
8. Do the authors describe the number of times the intervention was delivered and over what period of time including the number of sessions, their schedule, and their duration, intensity, or dose? |
Yes complete |
||
| Tailoring |
9. If the intervention was planned to be personalised, titrated, or adapted, do the authors mention so and then describe what, why, when and how? |
Yes complete |
||
| Modifications |
10. Do the authors mention if the intervention was modified during the course of the study, and if so, describe the changes (what, why, when, and how)? |
Yes complete |
||
| How Well |
11. Planned: If intervention adherence or fidelity was assessed, do they describe how and by whom, and if any strategies were used to maintain or improve fidelity, did the authors describe them? |
Yes complete |
||
| 12.(If above answer was yes) Actual: If intervention adherence or fidelity was assessed, did the authors describe the extent to which the intervention was delivered as planned? |
Yes complete |
|||
|
|
|||
| Excluded populations | |||
| Safety specific protocols / considerations relating to XR | |||
| Side effects / complications relating to XR | |||
|
|
|||
| Barriers to Implementation of XR | |||
| Facilitators to Implementation of XR | |||
