Abstract
Introduction
Around 20% of intensive care unit (ICU) patients undergo tracheostomy insertion and expect high-quality care concentrating on patient-centered outcomes including communication, oral intake, and mobilization. The majority of data has focused on timing, mortality, and resource utilization, with a paucity of information on quality of life following tracheostomy.
Methods
Single center retrospective study including all patients requiring tracheostomy from 2017 to 2019. Information collected on demographics, severity of illness, ICU and hospital length of stay (LOS), ICU and hospital mortality, discharge disposition, sedation, time to vocalization, swallow and mobilization. Outcomes were compared for early versus late tracheostomy (early = <day 10) and age category (≤ 65 vs ≥ 66 years).
Results
In total, 304 patients were included and 71% male, median age 59, APACHE II score 17. Median ICU and hospital LOS 16 and 56 days, respectively. ICU and hospital mortality 9.9% and 22.4%. Median time to tracheostomy 8 days, 8.55% open. Following tracheostomy, median days of sedation was 0, time to noninvasive ventilation (NIV) 1 day (94% of patients achieving this), ventilator-free breathing (VFB) 5 days (72%), speaking valve 7 days (60%), dynamic sitting 5 days (64%), and swallow assessment 16 days (73%). Early tracheostomy was associated with shorter ICU LOS (13 vs 26 days, P < .0001), reduced sedation (6 vs 12 days, P < .0001), faster transition to level 2 care (6 vs 10 days, P < .003), NIV (1 vs 2 days, P < .003), and VFB (4 vs 7 days, P < .005). Older patients received less sedation, had higher APACHE II scores and mortality (36.1%) and 18.5% were discharged home. Median time to VFB was 6 days (63.9%), speaking valve 7 days (64.7%), swallow assessment 20.5 days (66.7%), and dynamic sitting 5 days (62.2%).
Conclusion
Patient-centered outcomes are a worthy goal to consider when selecting patients for tracheostomy in addition to mortality or timing alone, including in older patients.
Introduction
Around 10% to 20% of intensive care unit (ICU) patients will undergo tracheostomy insertion. Patients with a tracheostomy often have a long duration of ICU stay and high mortality. 1 For the majority, survivors, these patients expect high-quality care focused on patient-centered outcomes including communication, swallow, and mobilization. There has been little research performed on quality of life of patients post-tracheostomy and recommendations have been made that future studies should do so. 2 Although studies have shown no mortality benefit from early tracheostomy, 3 others suggest a reduction in resource utilization including reduced ICU length of stay (LOS). 4 We have already demonstrated that introduction of a multidisciplinary tracheostomy team (MTT) has reduced time to decannulation. 5 We now want to ascertain if our tracheostomy patients are achieving the goals that are important to them—those that relate to communication, mobility, and swallow, de-escalation to lower dependency units allowing progression of rehabilitation and social mixing. Obtaining this information will help better delineate the benefits associated with tracheostomy use to aid in the decision-making process surrounding tracheostomy insertion and timing and identify areas for improvement in the care of patients who undergo a tracheostomy in the future.
Methods
This is a longitudinal retrospective observational study conducted in the Mater Misericordiae University Hospital (MMUH), Dublin. MMUH is a university-affiliated adult quaternary referral center with an 18-bedded mixed medical and surgical ICU, with 1200 admissions per year. MMUH incorporates the National Centre for Cardiothoracic Surgery (including heart and lung transplantation), pulmonary hypertension, adult extracorporeal membrane oxygenation service and the National Centre for Spinal Injuries, as well as all major specialties excluding neurosurgery.
All consecutive patients requiring tracheostomy insertion during their ICU stay from 2017 to 2019 were included in the study. Patients having a permanent tracheostomy prior to ICU admission were excluded.
Data was retrieved from the critical care electronic patient record (ICCA, Philips Healthcare), on patient demographics, ICU admission category, and severity of illness scores. The number of tracheostomies performed over the study period was recorded, including technique (percutaneous vs open) and time to tracheostomy insertion. Outcomes documented included ICU and hospital LOS, time to transition to level 2 care (high dependency), ICU and hospital mortality and discharge location. Patient-centered outcomes detailed included use of sedation, time to liberation from ventilation, time to restoration of speech, time to swallow assessment and time to mobilization. Information gathered during a patient's ICU stay was retrieved from the critical care electronic patient record. For outcomes post-ICU discharge, this was gathered from our post-tracheostomy MTT audit and recorded patient hospital disposition. Details of swallow assessment timing and results were obtained from our MTT records. The overall ICU mortality for the time period included was retrieved from intensive care national audit and research center Irish National ICU Audit quality reports.
In terms of patient-centered outcomes, when looking at use of sedation, we looked not only at days of sedation (total and post-tracheostomy insertion), but also the Richmond Agitation-Sedation Scale (RASS) score of our patients 1 day prior to tracheostomy and the time to RASS 0 post-tracheostomy to ascertain if deep sedation/agitation was preventing weaning. From a ventilation perspective, we used time post-tracheostomy to noninvasive ventilation (NIV) and ventilator-free breathing (VFB), defined as the first 24 h period free from positive pressure (either invasive mechanical ventilation or NIV), as markers of weaning progress. In our unit patients are transitioned to NIV once gas exchange is stable on relatively minimal settings on the ventilator, for example, an FiO2 of 40%, low positive end expiratory pressure (PEEP) and pressure support (PS) less than around 16 cmH2O. The time spent on NIV daily is increased gradually according to the patient's ability to tolerate it until they are established on NIV 24 h a day. To determine whether or not our tracheostomy patients would have been extubated soon after tracheostomy and may not have needed to undergo an invasive procedure, we used a post-tracheostomy surrogate for “fit for extubation.” This surrogate consisted of the time to achieve settings we believed would be consistent with extubation, namely a PS <10, fraction of inspired oxygen (FiO2) <40% with a good cough and expectorating secretions or cuff deflation. This surrogate affords a decision point of probable extubation, but not necessarily extubation success, where data would suggest a possible 10-20% extubation failure rate with a prolonged weaning patient cohort. 6 When assessing mobilization post-tracheostomy, we used the Chelsea Critical Care Physical Assessment Tool (CPAx) to assess physical morbidity. We chose this outcome measure as it is calculated on a daily basis from day 5 of admission on our ICU patients. It is also ICU specific, validated by specialist physiotherapists and has good internal and inter-rater reliability. 7 The specific parameters we chose, days post-tracheostomy to a dynamic sitting score of 2 (moderate assistance required), sitting to the edge of the bed score of 3 (minimal assistance) and sit to stand score of 3 (minimal assistance), were chosen following discussion with our physiotherapy colleagues on what represented meaningful progression outcomes for our patients.
The influence of tracheostomy insertion timing, dividing the group into early versus late tracheostomy (<10 days vs ≥10 days), was analyzed. This was to investigate any effect of timing of insertion on outcomes and resource utilization. Outcomes were also analyzed according to age being divided into younger (age 65 years or less) and older (age 66 years and older) subgroups.
Normally distributed data were compared with Student's t-test. Categorical data were compared using Fisher's exact test. Data that was not normally distributed was compared using the Mann-Whitney U test. A two-tailed P value of <0.05 for each test was used for statistical significance. Data was analyzed in Microsoft excel and Prism 9 (GraphPad LLC).
Results
In total, 304 patients required tracheostomy insertion from 2017 to 2019. The demographic data, severity of illness scores, LOS, and mortality of our tracheostomy patients can be seen in Table 1. The median ICU LOS was 16 days with a median hospital LOS of 56 days. Of note the ICU mortality of our tracheostomy patients was 9.9% in comparison with 11.3% for our total ICU population over the same time period. Information on ICU admission category can be seen in Table 2. The majority of admissions were medical (69%), with the largest proportion of these presenting with a respiratory illness. In keeping with the hospital being the National Centre for Cardiothoracic Surgery, the preponderance of surgical admissions were cardiothoracic in origin.
Baseline Characteristics, LOS, and Mortality for Patients with Tracheostomy.
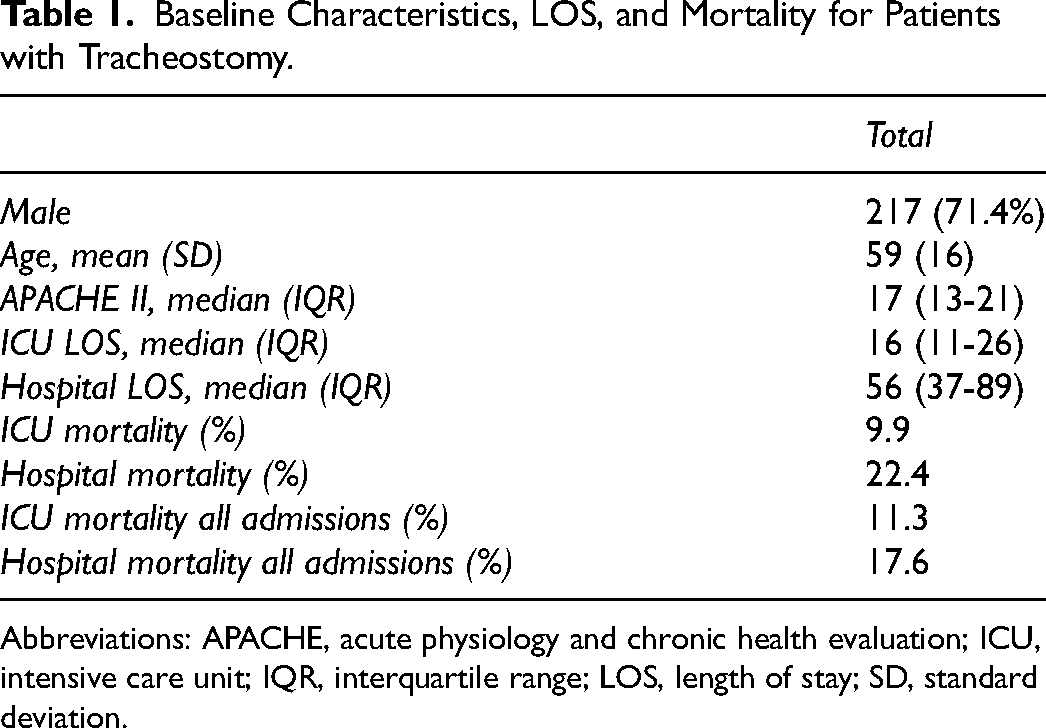
Abbreviations: APACHE, acute physiology and chronic health evaluation; ICU, intensive care unit; IQR, interquartile range; LOS, length of stay; SD, standard deviation.
Admission Categories for Patients Undergoing Tracheostomy Insertion During Intensive Care Stay.

In terms of the procedure itself, the median time to tracheostomy was 8 days (Table 3 and Figure 1). The majority were performed percutaneously in the ICU with a median stay in the ICU of 7 days following insertion.
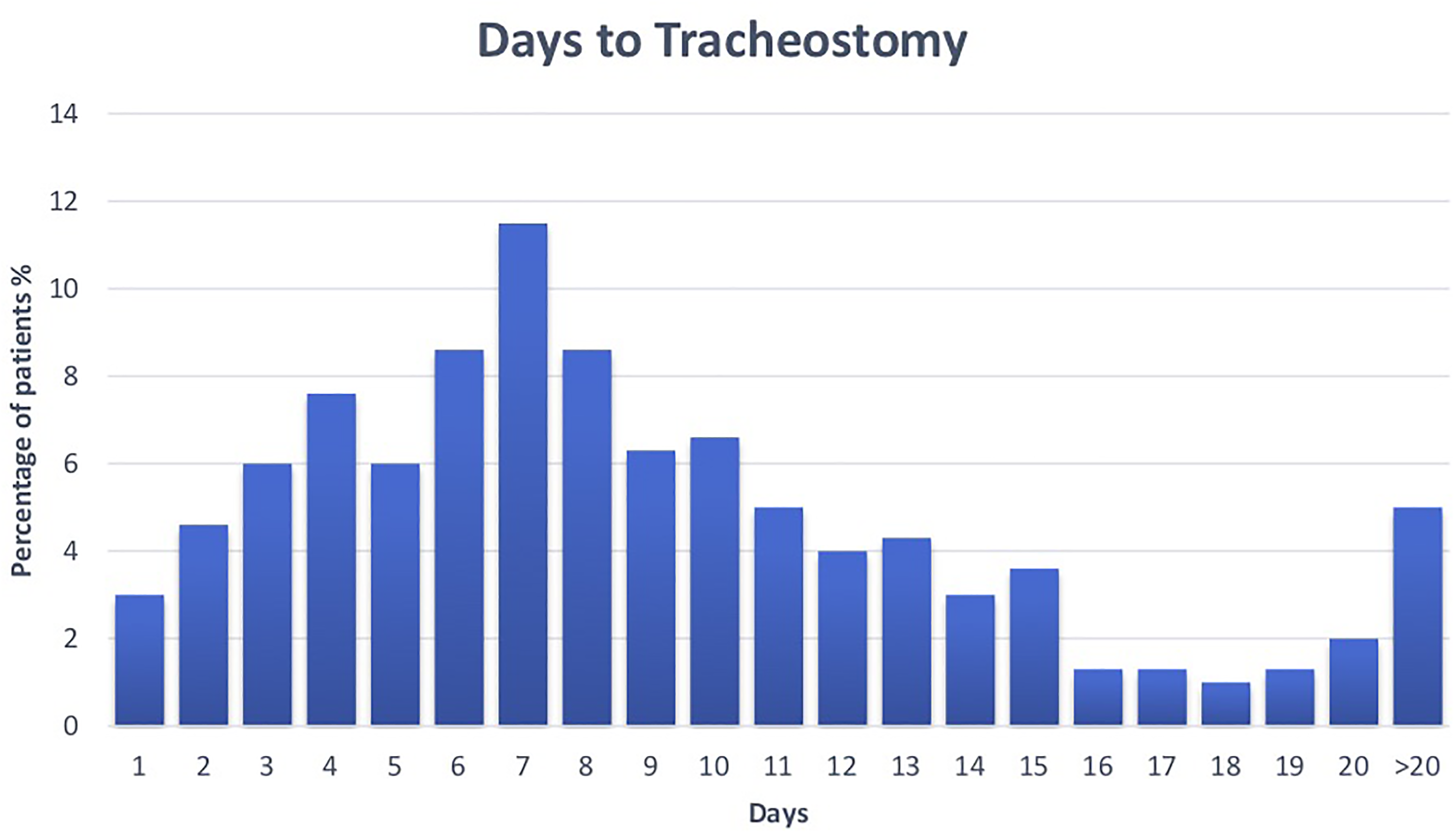
Timing of tracheostomy insertion following ICU admission in the total population. Abbreviation: ICU, intensive care unit.
Procedure Type and Patient-Centered Outcomes for Patients Undergoing Tracheostomy in the Intensive Care Unit.

Values presented as medians. Percentages represent the proportion of patients attaining each variable. Abbreviations: CPAx, Chelsea Critical Care Physical Assessment Tool; N/A, not applicable; NIV, noninvasive ventilation; RASS, Richmond Agitation-Sedation Scale; SpV, speaking valve; TF NPO transferred nil per oral; VFB, ventilator-free breathing.
Data on patient-centered outcomes following tracheostomy can also be seen in Table 3. Sedation was promptly discontinued post-tracheostomy (Figure 2), with patients being lightly sedated prior to the procedure (median RASS −1) and achieving a state of alert and calm a median of 2 days following tracheostomy. The majority (94.4%) of patients were rapidly transitioned to NIV and progressed to VFB by a median of 5 days (72%). This is in comparison to a median of 7 days to the extubation surrogate. And 60% of patients had a speaking valve (SpV) placed at 1 week. There was data available for 279 patients regarding swallow assessment. This occurred at a median of 16 days from tracheostomy with 73% of patients having an assessment performed. Reasons for not undergoing swallow assessment included death and transfer prior to assessment. In terms of mobilization, as expected the first target achieved was dynamic sitting (median 5 days). The proportion of patients reaching each incremental target declined so that only 19% accomplished a CPAx sit to stand score of 3 prior to discharge to critical care.
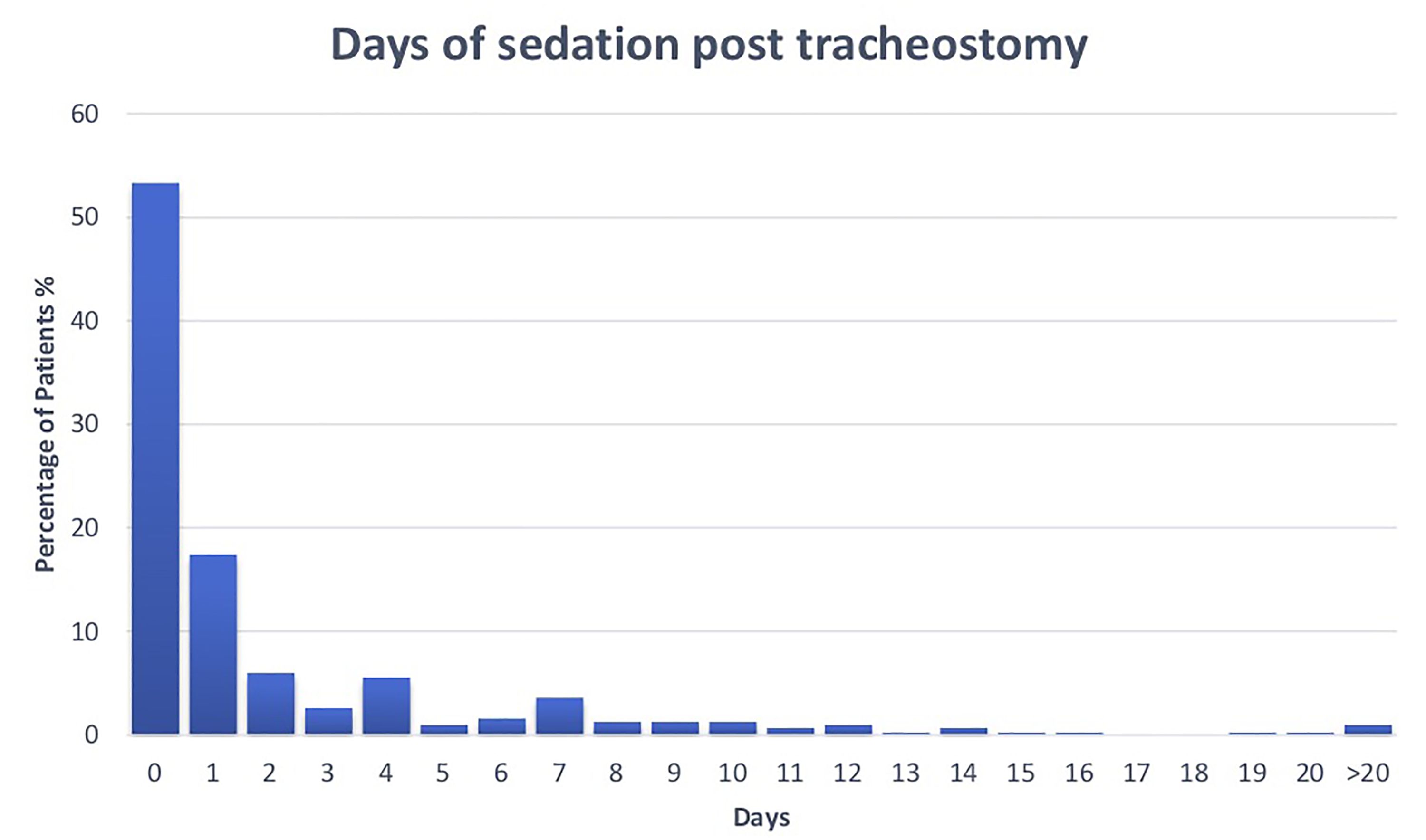
Proportion of patients discontinuing sedation on each day following tracheostomy insertion.
Analysis of tracheostomy timing on outcomes can be seen in Table 4. There was no difference in demographics, severity of illness, or mortality between the groups. There was a shorter ICU LOS in the early tracheostomy group (median 13 vs 26 days) and a faster transition to level 2 care (6 vs 10 days). There was a more rapid cessation of sedation in the early group (median total days of sedation 6 vs 12) and earlier weaning to NIV (median 1 vs 2 days) and VFB (median 4 vs 7 days). There was no significant difference in other outcomes between the early and late tracheostomy groups.
Comparison of Patient Characteristics and Outcomes for Early and Late Tracheostomy.
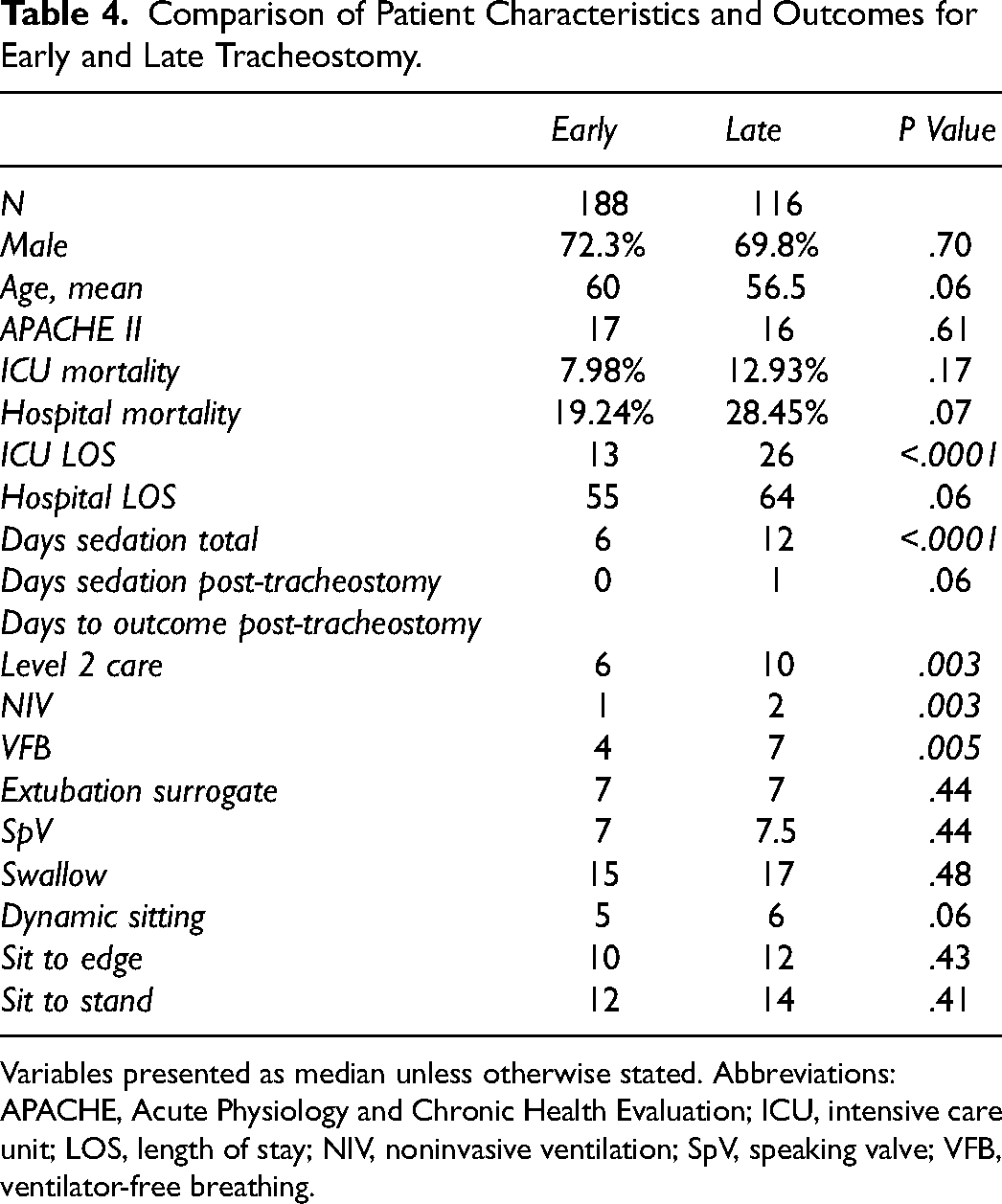
Variables presented as median unless otherwise stated. Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; ICU, intensive care unit; LOS, length of stay; NIV, noninvasive ventilation; SpV, speaking valve; VFB, ventilator-free breathing.
Evaluation of outcomes based on age category can be seen in Table 5. Mean age of older patients was 74 years versus 49 for the younger group. Patients who were older (≥66) had a higher Acute Physiology and Chronic Health Evaluation (APACHE) II score (19 vs 15) and a higher hospital mortality (36.1% vs 14.1%). They had a shorter total duration of sedation in comparison to younger patients (6 vs 8 days). Swallow assessment occurred later in the older subgroup (20.5 vs 15 days). Although the return to mobility was numerically longer in the older patients, this was not statistically significant. Two-thirds of older patients achieved VFB, placement of a SpV, swallow assessment, and dynamic sittings. And 50% of older patients passed their swallow assessment versus 58.5% of younger patients.
Comparison of Patient Characteristics and Outcomes by Age (Younger ≤ 65, Older ≥ 66 Years).
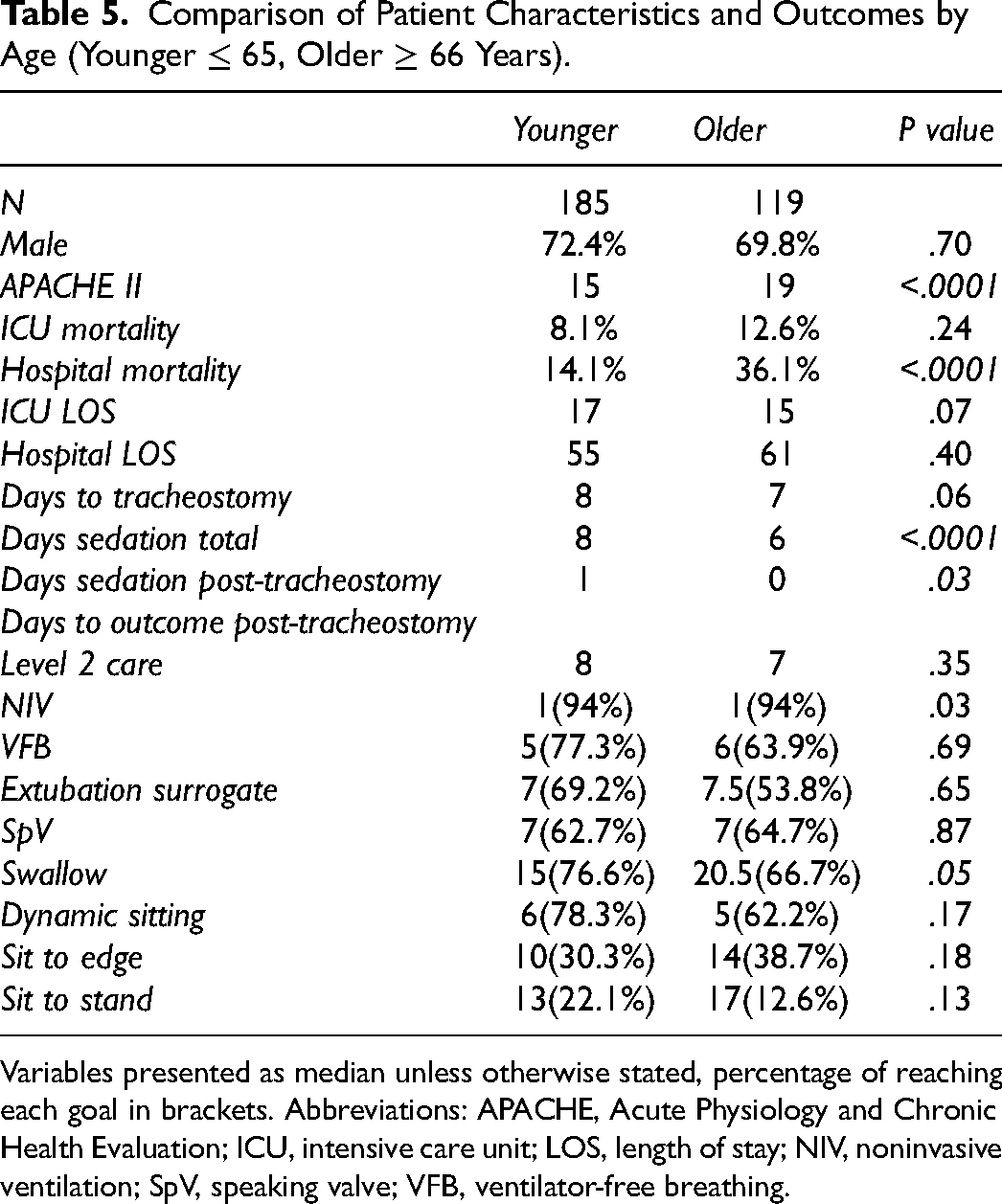
Variables presented as median unless otherwise stated, percentage of reaching each goal in brackets. Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; ICU, intensive care unit; LOS, length of stay; NIV, noninvasive ventilation; SpV, speaking valve; VFB, ventilator-free breathing.
Discharge disposition of patients who underwent tracheostomy can be seen in Table 6. About 40.9% of survivors (31.6% of the total population) were discharged home from hospital. A further 30.6% were transferred to another acute hospital, 11.9% to a rehabilitation hospital and 10.2% to another facility including nursing home (8.9%), hospice (0.4%) or no fixed abode (0.9%). When stratified by age, those in the oldest category (≥66) had the lowest proportion of patients being discharged home from hospital (18.5%; Figure 3). Over one-third of tracheostomies (38.5%) were performed in this age group (Figure 4).
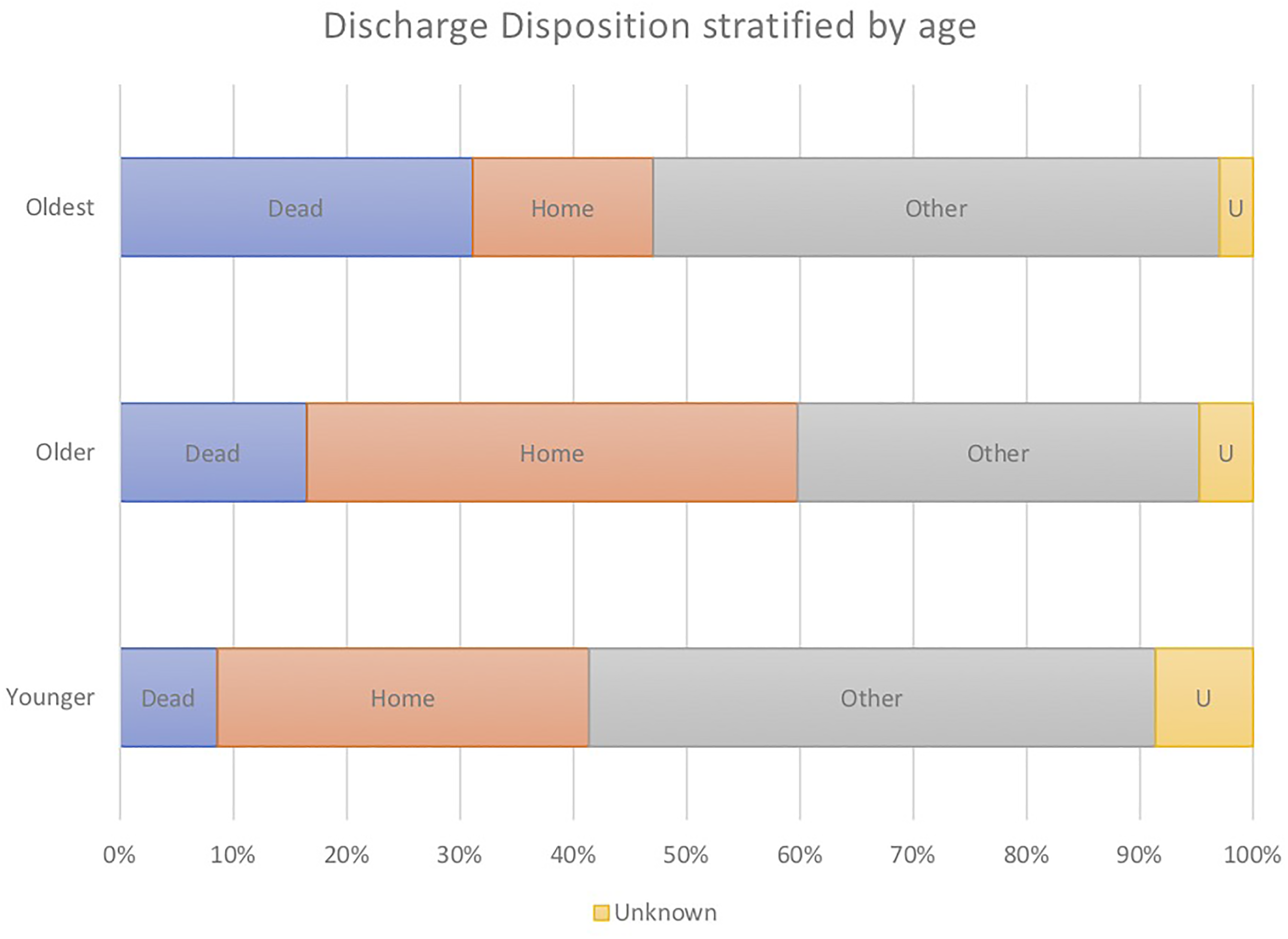
Hospital discharge disposition for patients with tracheostomy stratified by age. Oldest ≥66, older 45 to 65, younger ≤44 years. Abbreviation: Other—other acute hospital, rehabilitation, nursing home, hospice, no fixed abode.
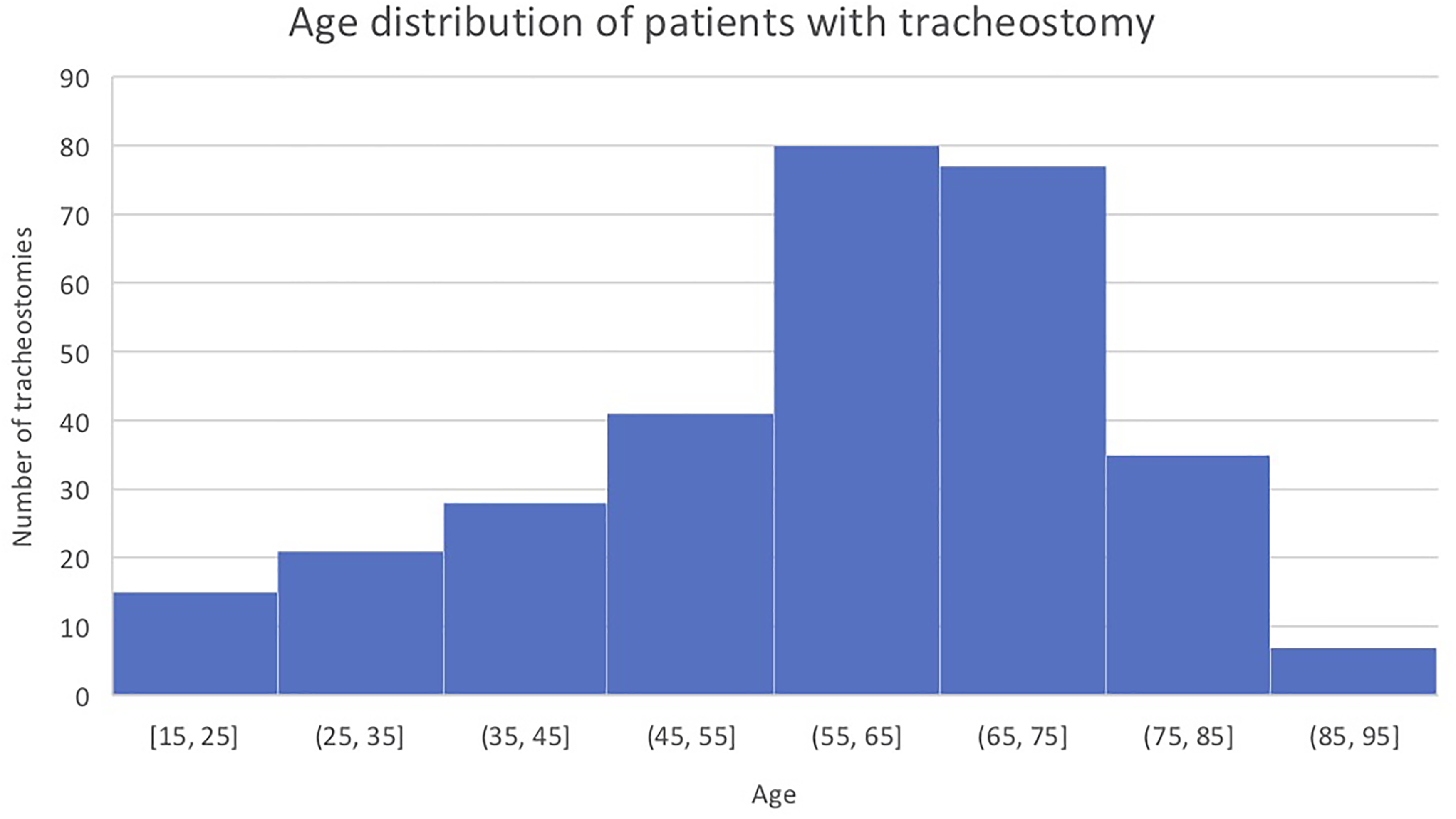
Age of patients at the time of tracheostomy insertion.
Discharge Disposition for Hospital Survivors Stratified by Age (Younger ≤ 44, Older 45-65, Oldest ≥ 66 Years.
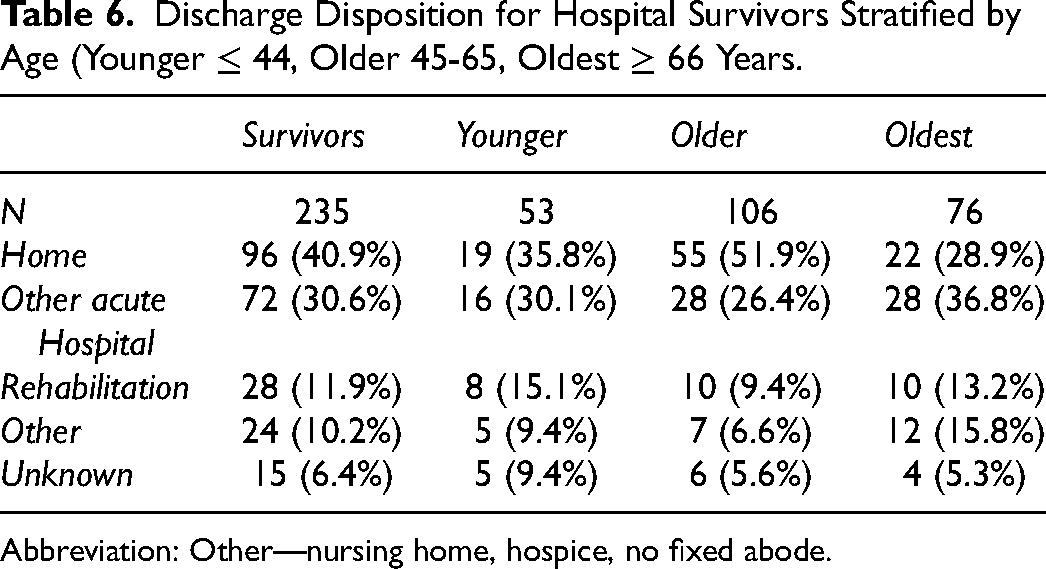
Abbreviation: Other—nursing home, hospice, no fixed abode.
Discussion
We have looked at outcomes for our tracheostomy patients in a university-affiliated teaching hospital over a 3-year period from 2017 to 2019. Around 10% of our patients require tracheostomy insertion (around 100 patients per year in a unit with 1200 admissions annually), in keeping with previously published data.1,8,9 The majority of these are performed percutaneously, as is the trend internationally. 3 In our own center, we have documented that the proportion of percutaneous tracheostomies has increased over time in comparison with the open technique from 80% to 85% in 2009 to 91% in our current study. 5 Considering timing of tracheostomy, the procedure was performed at a median of 8 days from ICU admission, consistent with previous data from our own center. 5
The median LOS in ICU was 16 days in our tracheostomy population, and 56 days for hospital LOS. This is similar to the expected values expressed in the UK Guidance for Tracheostomy Care, 1 but notably longer, particularly for hospital LOS, than those in secondary analysis of the LUNG-SAFE study (11 and 24 days for ICU and hospital LOS, respectively) and in Mehta's paper for the 2012 data (26-day hospital LOS).9,10 A potential explanation for this could be the differing patient cohorts included in the studies. 9 It may also be a reflection of ICU capacity, with Ireland having the lowest rate of ICU beds per head of population, at 5 per 100,000, in the latest report available from the Organisation for Economic Cooperation and Development (OECD) on ICU capacity, meaning an unwell patient cohort, with a median APACHE II score of 17, requiring prolonged ventilation are admitted to our unit. 11 This underlines that a tracheostomy is not performed lightly in our center. Despite a relatively long ICU LOS, once tracheostomy was performed, patients were discharged to a level 2 area at a median of 7 days. This is in keeping with previous results from our center and emphasizes the MTT in enhancing and progressing patient care. 5
The ICU mortality was 9.9% in our patient population. Although we could not statistically compare them, this is slightly lower than the mortality for our ICU patient group as a whole (11.3%). The converse is true when it comes to hospital mortality, with our tracheostomy population having worse outcomes than the ICU patients as a whole (22.5% vs 17.6%). Our ICU and hospital mortality are noticeably lower than those in the study by Young et al. 3 For our patients, an a priori decision is made that the patient has either an uncertain prognosis or is more likely to survive. Our lower mortality may signify improvements in care, as there has been a downward trend in hospital mortality over time in our patients, looking at previously reported figures from our own center. 5 The increase in mortality seen at hospital discharge is in keeping with that found in the LUNG-SAFE secondary analysis. 9 In this article, they suggested that although tracheostomy improves short-term survival, it has no impact on long-term outcomes. In fact, although there was a reduction in hospital mortality in Mehta's study, this may signify only a change in location of death to long-term care facilities. 10 We would argue that a hospital mortality of 22.5% for ICU patients with tracheostomy is consistent with the 90-day mortalities of recent sepsis studies.12,13
The Young et al’s paper and other studies are often misquoted as reasons not to perform a tracheostomy, either early or late, contending that neither has an impact on mortality. However, we contend that mortality should not be the index for appropriateness of the procedure. Outcomes that are important to the patient should be considered during the decision-making process and the significance of the potential return to vocalization, swallow, and increased mobility following tracheostomy should not be under-appreciated. The value to the patient is premised on appropriate patient selection, with a particular focus on the patient with longer term ventilation needs and a predicted prolonged ICU stay, associated myopathy, deconditioning and prolonged rehabilitation requirements.
We feel that the ability to rapidly wean sedation following tracheostomy is an important patient-centered outcome, as it reflects the ability of the patient to interact with their environment, caregivers, and family and to take an active role in their care and rehabilitation. A reduction in use of sedation with tracheostomy has been noted in previous randomized-control trials and meta-analysis.3,14 In our own population we were able to stop sedation on the day of tracheostomy for a large proportion of our patients. Including a measure of the depth of sedation enabled us to ensure that our patients were calm and alert following discontinuation. Deep sedation is associated with an increased risk of death, delirium, and delayed extubation in mechanically ventilated patients. 15 Exposure to psychoactive drugs, with benzodiazepines being of particular concern, has been identified as a modifiable delirium risk factor. 16 There is also concern that prolonged sedation could contribute to long-term cognitive impairment, particularly executive function. 17 Given that tracheostomy-facilitated discontinuation of sedation in our patients, this could potentially reduce the incidence of delirium and long-term cognitive impairment and have a positive effect on mortality.
The ability to transition from invasive ventilation to NIV is a patient-centered outcome from our perspective for a number of reasons. The first and foremost is that once patients are weaned from ventilation, they are free from the apparatus required for its delivery. This leaves them free to mobilize and interact with their surroundings and take a more active role in their rehabilitation. The second is that of resource utilization. Patients can be discharged to a high dependency level of care once established on NIV via a tracheostomy and may be discharged to a ward with follow up by our MTT once VFB is accomplished. This is in keeping with previous trials in ICU trauma patients and recent meta-analysis.4,18 There is a conflicting evidence in the literature as to whether tracheostomy reduces days of mechanical ventilation.3,4,14,18 In our own population, once tracheostomy was performed 94% of our patients were transitioned to NIV at a median of 1-day postinsertion, with VFB by day 5 in 72%. In comparison, it took a median of 7 days to reach our extubation surrogate and only 63% of patients reached this target suggesting that tracheostomy insertion potentially reduced days on mechanical ventilation with the patient-centered benefits of this as described above. It also implies that we are correctly identifying a group of patients who require prolonged ventilation in which to perform tracheostomy, many of whom have ongoing high ventilatory requirements or excess secretions and a poor cough as a component of their critical illness. It should be taken into consideration that our extubation surrogate does not equate to extubation success. An extubation failure rate of 10% to 20% in the prolonged weaning cohort is to be expected. 6
Restoration of vocalization represents one aspect of high-quality care for tracheostomy patients and is a key patient-centered outcome. According to the latest guidance document from the UK Faculty of Intensive Care Medicine on tracheostomy care, vocalization should be a daily goal of care and speech and language therapy (SLT) assessment should occur at the point when sedation is held, and ventilation being weaned. 1 It is well known that loss of the ability to communicate causes fear, anxiety, and powerlessness in tracheostomy patients.1,19 It can also contribute to depression and post-traumatic stress disorder. 20 Attempts at nonverbal communication can be frustrating and cumbersome for patients as they may not have the strength, energy, or co-ordination it takes to complete such tasks. This makes restoration of vocal communication a crucial goal of tracheostomy care. This is most often achieved with use of an SpV, which in addition to restoration of speech, has a number of other advantages including improved laryngeal sensation and function, improved smell and taste, and creation of a level of continuous positive pressure. In our patients, the median time to placement of an SpV was 7 days and this occurred in 60% of the population. In a paper looking at early targeted intervention for vocalization, return to phonation occurred at a median of 7 days in comparison to 18 days for those in the control group. 20 Our figures are reassuring when comparing to this trial. However, they measured return to phonation as the ability to count from 1 to 10 whereas we have used first placement of an SpV as a surrogate for speech. In a recent trial analyzing the impact of implementing quality improvement initiatives from the Global Tracheostomy initiative in 20 sites, time to first use of an SpV decreased from 14 to 7 days with implementation of standardized multidisciplinary care, educational initiatives, patient and family involvement, and tracking patient data. 21 Our institution has had an MTT in place since 2009, with weekly full team ward rounds, daily advanced nurse practitioner rounds and standardized equipment, protocols and guidelines for care of tracheostomy patients. We have previously shown that establishment of the MTT lead to a reduction in time to decannulation. 5 The MTT and early involvement of SLT with our ICU patients likely accounts for the short time to placement of an SpV. Our data give us a benchmark for further improvement, along with recording time to first SLT referral and consideration of 40% of the patients who did not meet this target. A proportion of this may be down to documentation but looking into other clinical, systems, or process reasons for this would help improve outcomes.
Return to oral intake is a significant goal for patients. It is well known that dysphagia is a common occurrence in tracheostomy patients. While this was initially thought to be due to the presence of a tracheostomy tube altering swallowing biomechanics, it is more likely that dysphagia is part of the underlying condition leading to tracheostomy insertion. 22 As with speech, there can be significant patient fear and anxiety surrounding swallow and diet with a tracheostomy in place. Return to oral intake is recommended as a daily goal of care for tracheostomy patients. 1 It has been highlighted that recognition of dysphagia needs to improve, with earlier referral to SLT an essential component in management, including use of functional endoscopic evaluation of swallow (FEES).1,23 The median time to swallow assessment in our population was 16 days, with 55% of those followed resuming oral intake following assessment. This is with an MTT in place and regular use of FEES in our center. In comparison, a retrospective review of tracheostomy patients in a tertiary referral center found a median of 10.5 days to resumption of oral intake, with 82% of patients commencing diet with a tracheostomy in situ. A small proportion of these resumed intake prior to cuff deflation. 22 The quality improvement initiative referred to above also found a significant reduction in time to first oral intake (decrease from a median of 26 to 9 days), with introduction of their initiatives. 21 In comparison to these papers, there is a significant delay to swallow assessment with a smaller proportion of patients resuming oral intake. At the time of our data collection, there was a 0.5 whole time equivalent SLT covering both ICU and ear nose and throat (ENT) tracheostomy patients and providing services to the outpatient department. This likely contributed to the delay in time to swallow assessment. In a paper by Sutt et al they found a return to swallow on average 7.4 days earlier with a tracheostomy versus endotracheal tube but commented that this was in a well-resourced department with several full-time allied health professionals. 24 Recent guidelines from the Faculty of Intensive Care Medicine and Intensive Care Society in the United Kingdom recommend 0.1 SLT staff per ICU bed. 25 Our data underlines the necessity of such a valuable resource. Resources in our own unit have improved in recent times with an increase in the number of SLTs dedicated to the ICU. Along with this, education and awareness of the importance of early referral to SLT and speech and swallow assessment has increased, and it would be interesting to compare our current data to that which we have here.
Mobility has been recognized as an important patient-centered outcome following tracheostomy. 2 Functional disability is well described following critical illness. A study by Herridge et al 26 found that all patients reported weakness 1 week following ICU discharge, with 60% being unable to walk without assistance. The worst outcomes were seen in older patients with longer ICU LOS. This was the subgroup most likely to undergo tracheostomy. And 77% of these patients could not walk unassisted 1 week after ICU stay and had a 1-year mortality of 40%. 22 In our patients insertion of a tracheostomy-facilitated discontinuation of sedation enabling patients to actively participate in rehabilitation. As a result, the majority of patients with tracheostomy were able to sit with assistance 2 days earlier than when the extubation surrogate was achieved. A recent paper looking at patient-centered outcomes with earlier tracheostomy found that 88% of patients could perform out of bed exercises by ICU discharge. They also found that this occurred 6.2 days earlier with early tracheostomy. 24 Although there was no statistically significant difference in time to mobilization between our early and late tracheostomy groups, we measured these variables from tracheostomy insertion, this then suggests that with earlier tracheostomy patients are able to mobilize sooner. Insertion of a tracheostomy therefore accelerated patient rehabilitation and improved outcomes important to patients.
The ability to return home is highly valued by patients and their families. Reassuringly, the majority of our tracheostomy patients were discharged home (40.9%). Nearly one-third of patients were transferred to another acute hospital reflecting the fact that our hospital is a quaternary referral center and only 8.9% were discharged to a nursing home. This indicates that we are selecting a patient population with a positive trajectory. The discharge location diverges with Mehta's paper in which the discharge disposition of 71.9% of patients was long-term care in the recent period, with only 13.1% being discharged home. 10 This may reflect the differing structure of the healthcare system in the United State, with greater availability of long-term care facilities that can care for tracheostomy patients, shifting the burden of care for these patients. 10 Similar to Herridge's data, it was our oldest patients (≥66) who were least likely to be discharged home (28.9%). 26 This information can help inform discussion about selection of patients for tracheostomy in the future considering it from a patient-centered perspective.
As noted by Herridge et al in their paper, the oldest tracheostomy patients had the worst outcomes in terms of mortality and discharge home. 26 This is backed up by recent evidence from the CoVIP investigators who reported a 50.4% mortality in patients with COVID-19 over 70 years of age with a tracheostomy. 27 Another recent paper has found that, not only are mortality rates high in elderly tracheostomized patients, but that survivors had profound physical morbidity with a significant impact on their functional independence with greatly reduced ability to eat, speak, and ambulate. 28 The hospital mortality in our older patients (36.1%), although high, was lower than both these studies. In comparison to the COVID 19 disease in Very Elderly Intensive Care Patients (CoVIP) study, whose cohort also had an average age of 74 years, mortality was measured at 3 months rather than hospital discharge and included only patients with COVID-19. 27 In the paper by Lee et al, the average age was 81 years, perhaps explaining the higher mortality rate than our own. More importantly, in contrast to the paper by Lee et al, around two-thirds of our older patients reached VFB, had an SpV placed, swallow assessment performed (with 50% pass rate) and achieved dynamic sitting. 28 The older subgroup also received less days of sedation than the younger population allowing them to interact with their surroundings, caregivers, and family earlier. This reduction in sedation may also contribute to reduced rates of delirium and long-term cognitive impairment as mentioned above. This reinforces our viewpoint that mortality is not the only or most valuable point to be considering when making the decision to perform a tracheostomy. The significance to older patients of being able to communicate, eat, and interact with their loved ones should not be underestimated. Again, this is premised on appropriate patient selection, focusing on those who we feel are more likely survive and to require prolonged ventilation.
When analyzing our population by time to tracheostomy, there were a number of significant differences between our early and late groups. We found that early tracheostomy was associated with a shorter ICU LOS, transition to level 2 care, reduced duration of sedation and less days of mechanical ventilation. Although there are a number of studies that have not found any association with early tracheostomy and reduced LOS or ventilation,3,14 there are a number of recent publications that support our findings.4,18 A lower incidence of ventilator-associated pneumonia was also found in these papers.4,18 We did not find a mortality difference with early tracheostomy in our group, similar to the majority of previously published data, including a Cochrane systematic review whose findings were only suggestive at best that early tracheostomy was associated with reduced mortality. 29 We did not find a statistically significant difference in patient-centered outcomes with early tracheostomy. This is in contrast with the paper by Sutt et al which found that patients had return of voice, oral intake, and mobilization on average 1 week earlier with early tracheostomy. 24 This paper looked at time from endotracheal tube insertion to patient centric outcome, with tracheostomy insertion being an intermediate event. Multistate modeling was used to account for the varying time to tracheostomy insertion, rather than an arbitrary cut-off to define an early or late event which they felt would create bias. 24 This is in comparison to our own which used day 10 as the cut-off point for early tracheostomy and measured timing of patient-centered outcomes from the day of tracheostomy insertion. The variation in methods of statistical analysis may account for why no difference was found in our groups. We agree with the Sutt paper that patient centric outcomes are an important goal when considering tracheostomy insertion.
The limitations of our study are that it is retrospective in nature and performed in a single center with a heterogenous case mix. We have used a number of broad measures of ICU outcome including LOS and mortality and surrogates such as placement of an SpV to measure our patient-centered outcomes. We did not include a control group of ventilated ICU patients who did not receive tracheostomy, instead using a surrogate measure for extubation. Finally, we followed patients only to hospital discharge and disposition at this time and did not look at long-term outcome.
Conclusion
Patient-centered outcomes are a worthy goal to consider when selecting patients for tracheostomy. Mortality, which has been improving over time, is not the only, or most appropriate metric on which to base the decision and selecting patients with a positive or uncertain prognostic trajectory is more important than timing of tracheostomy alone. In our single center retrospective study, tracheostomy facilitated rapid weaning of sedation, ventilation, and progression to a high dependency level of care. Around two-thirds of patients with a tracheostomy achieved meaningful patient-centered outcomes in terms of communication, oral intake, and mobilization and a considerable number were discharged home. Although our oldest subgroup of patients had the highest mortality and were least likely to be discharged home, two-thirds of them also reached noteworthy patient-centered outcomes. This knowledge will help inform discussions on how best to use a scarce resource in patients who may go on to have a protracted stay in the ICU. It will also ensure we are considering outcomes important to patients when making these decisions surrounding their care and identify areas for future research in patients with tracheostomies in critical care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
