Abstract
Acutely elevated intracranial pressure (ICP) may have devastating effects on patient mortality and neurologic outcomes, yet its initial detection remains difficult because of the variety of manifestations that it can cause disease states it is associated with. Several treatment guidelines exist for specific disease processes such as trauma or ischemic stroke, but their recommendations may not apply to other causes. In the acute setting, management decisions must often be made before the underlying cause is known. In this review, we present an organized, evidence-based approach to the recognition and management of patients with suspected or confirmed elevated ICP in the first minutes to hours of resuscitation. We explore the utility of invasive and noninvasive methods of diagnosis, including history, physical examination, imaging, and ICP monitors. We synthesize various guidelines and expert recommendations and identify core management principles including noninvasive maneuvers, neuroprotective intubation and ventilation strategies, and pharmacologic therapies such as ketamine, lidocaine, corticosteroids, and the hyperosmolar agents mannitol and hypertonic saline. Although an in-depth discussion of the definitive management of each etiology is beyond the scope of this review, our goal is to provide an empirical approach to these time-sensitive, critical presentations in their initial stages.
Keywords
Introduction
Elevated intracranial pressure (ICP) is a common and potentially life-threatening condition encountered in a wide array of neurological pathologies. It is associated with increased mortality1,2 and poor neurologic outcomes3,4 in traumatic brain injury (TBI), and decreased survival in intracerebral hemorrhage 5 and acute ischemic stroke. 6 Normal ICP in adults is generally defined as an ICP between 5 and 15 mm Hg. 7 It can be understood fundamentally as an equilibrium in the partial pressures of fluids, namely, arterial blood, venous blood, and cerebral spinal fluid, in addition to the largely incompressible brain parenchyma within the fixed volume of the cranium, as described by the Monroe-Kellie Doctrine. 8 If the volume of one of these or additional components (such as a neoplasm) increases, venous blood and cerebrospinal fluid are displaced out of the cranium to maintain equilibrium. In a state of elevated ICP, this compensatory mechanism may become dysfunctional, and neurologic injury may result through 2 mechanisms: hypoperfusion/ischemia and herniation. Ischemic injury is the result of decreased cerebral arterial blood flow secondary to insurmountable ICP and dysregulated vascular compensation. 7 Herniation is the process by which brain parenchyma is displaced from one compartment to another (eg, uncal or transtentorial herniation), with subsequent compression of critical structures such as the brainstem.
Prompt recognition, monitoring, and treatment of raised ICP are essential in the management of critical intracranial pathologies including TBI, intracranial infections, malignancies, and neurovascular emergencies. The Brain Trauma Foundation recommends treatment of ICP over 22 mm Hg, as this is the threshold for increased mortality. 9 In 2020, the Neurocritical Care Society released guidelines for the treatment of acute cerebral edema, 10 as well as an updated algorithm for intracranial hypertension and herniation as part of their Emergency Neurological Life Support program. 11 However, given that this topic is a rapidly evolving area of research, many of these suggestions are generalized and lack practical guidance that may be easily employed in patient care. Particularly, in acute resuscitation of patients with unknown etiology or degree of elevated ICP, delays or errors in treatment can have deleterious consequences.
This review presents a pragmatic approach to the initial management of a patient with elevated ICP, with a focus on early recognition in the first minutes to hours of resuscitation. The recommendations made herein combine current evidence with expert consensus to provide empiric treatment for the undifferentiated patient with raised ICP, such as those presenting in the emergency department. A disease-specific approach or advanced therapeutic strategies implemented outside of this hyperacute phase are outside of the scope of this review and covered in various guidelines.9,12–15 There will be particular emphasis on rapid recognition, noninvasive treatment, pharmacologic therapy, and practical considerations for neuroprotective intubation.
Diagnosis
History and Physical Examination
As many patients with elevated ICP have altered mental status, history is often limited and there is equivocal evidence supporting its utility. Upon approaching a patient with suspected elevated ICP, one may gather a history of classic signs or symptoms, including headache, vomiting, and change in mental status; however, none of these findings are specific. 16 Despite this, history may provide important information in patients whose diagnosis is uncertain to clarify the etiology or likelihood of high ICP (eg, meningismus, trauma, or use of anticoagulants).
A recent meta-analysis explored the utility of 3 common physical examination findings for raised ICP. 17 Although no single finding was diagnostic of the condition, pupillary dilation was found to be highly specific (85.9%) but not sensitive (28.2%). In contrast, a Glasgow Coma Scale (GCS) of 8 or below was quite sensitive (75.8%) but poorly specific (39.9%). There is evidence that the “Cushing triad” of hypertension, bradycardia, and irregular breathing may have a specificity as high as 96.9% 18 ; however, it is poorly sensitive.18,19 In extreme situations, uncontrolled ICP may lead to cerebral herniation syndromes, resulting in various findings including cranial nerve deficits and lateralizing neurological deficits. These are emergent conditions requiring prompt neurosurgical evaluation. In extremis or if left untreated, patients may develop brain death—the complete and irreversible loss of all brain function. 20 Given the importance of rapid intervention in elevated ICP, noninvasive management should be initiated based on convincing clinical history and physical examination findings and should not be delayed by further investigations.
Imaging
Computed tomography (CT) is a useful imaging modality for the evaluation of elevated ICP and should be used to supplement the history and physical examination when the clinical context permits. Especially, when the etiology is uncertain, convincing CT scan findings in conjunction with a history and physical examination may be sufficient to make the diagnosis of elevated ICP. 21 The most sensitive signs for elevated ICP on CT include basal cistern effacement (85.9%), midline shift (80.9%), and a Marshall class of at least 3 (80.6%). 17 The Marshall classification is a grading system using key findings on CT neuroimaging to prognosticate traumatic brain injuries. 22 In addition to specific signs of elevated ICP, CT may provide diagnostic clarification on the etiology of elevated ICP, allowing for planning of definitive management as initial resuscitative steps are taken. Conversely, magnetic resonance imaging (MRI) is rarely used in the evaluation of acutely elevated ICP. Although it has a similar utility as CT for ICP evaluation, MRI is relatively slow and costly, making it impractical in emergent situations. Imaging should not delay initial management steps in the context of a high pretest probability of elevated ICP. Although CT scans may provide valuable information and diagnostic clarification, noninvasive management should simultaneously be initiated in patients with a moderate probability of elevated ICP while arranging for imaging.
Point-of-Care Ultrasound
As point-of-care ultrasound (POCUS) becomes increasingly available, several techniques have gained popularity in the identification of raised ICP. The use of transcranial doppler ultrasonography relies on qualitative, noninvasive measurements of cerebral blood flow via bone windows in the cranium to estimate ICP using flow velocity and various other parameters. 23 However, transcranial doppler was shown to have limited utility with a relatively low area under the receiver operating characteristic curve (AUROC) (0.55-0.72 for pulsatility index, 0.85 for arterial blood pressure). 17 Likewise, another small study demonstrated pulsatility index to be a poor proxy for ICP estimate. 24
Optic nerve sheath diameter (ONSD) relies on the assumption that increases in ICP will be reflected in the dilatation of the optic nerve sheath through the communication of the intracranial and intraorbital subarachnoid spaces. Practically, it involves measuring the diameter of the optic nerve sheath posterior to the orbit with POCUS. Its utility is more encouraging (AUROC for diagnostic accuracy of 0.94 among 10 studies) 17 ; however, these studies used different cutoffs for diameter measurements, making comparison difficult. Another recent meta-analysis found ONSD to be highly sensitive (92%) and specific (86%) in the diagnosis of elevated ICP, with a positive likelihood ratio of 6.93 in non-TBI. 25 Despite these promising results, they should be interpreted with caution as their comparison was not exclusive to the gold standard of invasive ICP monitoring.
Given the methodological limitations of the available studies, specifically the lack of a large, prospective trial with a gold standard comparator, there is limited high-quality evidence for the use of POCUS in the diagnosis of elevated ICP. In the hands of an experienced operator, POCUS findings may add additional information to other clinical signs of elevated ICP, but cannot be depended upon in isolation.
Invasive Intracranial Monitoring
Invasive intracranial monitoring remains the gold standard for measuring and diagnosing elevated ICP. 26 Invasive monitoring is typically done by placement of either an intraparenchymal sensor or external ventricular drain (EVD), with the latter having the advantage of permitting cerebrospinal fluid drainage if needed. Despite its widespread use, however, there is conflicting evidence that invasive ICP monitoring improves patient outcomes.27,28 The landmark BEST:TRIP trial found no improvement in neurologic outcomes or mortality with a treatment algorithm utilizing invasive ICP monitoring compared to an algorithm involving serial physical examinations and CT scans. 28 However, this trial did have important limitations including a lack of external validity (performed in Bolivian and Ecuadorian intensive care units where the standard of care may vary significantly from other countries, particularly in regards to pre- and post-hospital care) and a composite outcome that weighted mortality similarly to individual components of functional and cognitive tests. Although invasive ICP monitoring may provide valuable information on response to advanced management such as hyperosmolar therapy, a normal ICP reading in isolation is insufficient to rule out elevated pressure. 26 Various factors, including focal lesions, may prevent accurate measurement of ICP by monitors, making serial physical examinations and neuroimaging vital in acute resuscitation scenarios even if a monitor is in place. Additionally, the use of invasive ICP monitoring is not without risks. One review found the infection rate of EVDs to be as high as 22%. 29 EVD-related infections were associated with prolonged hospital stay, increased cost of care, and overall mortality. 30 In addition to other complications, including postprocedural hemorrhage and misplacement, one must also consider the potential harm from delaying other diagnostic and therapeutic interventions during monitor insertion.
Guideline recommendations for the routine use of invasive ICP monitors is specific to underlying etiology; for example, routine monitoring is not recommended for ischemic stroke 15 but is recommended for TBI. 9 A consensus statement by the International Multidisciplinary Consensus Conference on Multimodality Monitoring in Neurocritical Care recommends that ICP monitoring be incorporated in protocolized care for patients with acute brain injury in addition to imaging and clinical evaluation, with the indications and methods for monitoring tailored to the specific pathology. 31
Extrapolating these data to acute scenarios in which the underlying etiology may be unknown and competing management priorities exist, the decision to place an invasive ICP monitor needs to balance the potential benefits with its inherent risks. The information provided by the monitor may be particularly helpful in the hours, rather than minutes, following acute resuscitation to assess ongoing ICP fluctuations and response to therapy. Compared to an intraparenchymal monitor, the additional therapeutic benefit of fluid drainage with an EVD must be weighed by additional risks, particularly with insertion in the absence of hydrocephalus. Therefore, it may be reasonable to defer the placement of an invasive ICP monitor, while initial management steps are initiated, ensuring that serial clinical examinations and imaging studies are used to monitor ICP in the interim.
Management
Rapid assessment and management of elevated ICP is paramount in the prevention of secondary brain injury. Several organizations have released detailed recommendations for the management of specific etiologies of elevated ICP, including TBI,9,12 intracerebral hemorrhage, 14 subarachnoid hemorrhage, 13 and acute ischemic stroke. 15 More recently, the Neurocritical Care Society published guidelines on the management of acute cerebral edema. 10 Collectively, they lack a systematic approach to the acute resuscitation of critically ill patient with undifferentiated etiology of elevated ICP. Below, we will review the current body of evidence for various interventions, with the ultimate goal of reducing or stabilizing elevations in ICP while more definitive management for the specific underlying cause is arranged. We present a generally linear approach to management starting with the simplest methods and proceeding to more involved or invasive steps with less clear evidence of benefit. These management steps are summarized in Table 1.
Fundamental Management Principles in the Initial Empiric Treatment of Acutely Elevated Intracranial Pressure.

Abbreviations: ETCO2, end-tidal concentration carbon dioxide; PaCO2, arterial partial pressure carbon dioxide; SBP, systolic blood pressure; SpO2, oxygen saturation.
If underlying etiology known, use specific guideline recommendations for perfusion target.
Initial Assessment and Patient Positioning
As with any unstable patient, one should begin by assessing the ABCs (Airway, Breathing, Circulation). Many patients with elevated ICP will have neurological dysfunction resulting in impaired ability to protect their airway or compromised ventilation. Therefore, it is important to anticipate and prepare for endotracheal intubation early (see section on “Neuroprotective Intubation” below). The patient must receive appropriate sedation and analgesia, as pain or agitation may cause further elevations in ICP. Possible treatments include rapid-onset, short-acting agents such as fentanyl and ketamine.
Targeting normal vital signs will mitigate secondary brain injury. This includes normotension (100-180 mm Hg; acknowledging there is limited evidence for a particular range in the undifferentiated patient, and that prevention of hypotension should remain the priority), normoxia (≥94% SpO2) and normothermia (36°C-37.8°C). Ideal blood pressure treatment utilizes rapid onset, short-acting and titratable medications such as labetalol for hypertension or norepinephrine for hypotension. Elevated ICP itself should not cause hypotension, so the presence of hypotension should prompt a thorough search for other causes. Regarding targeted temperature management, multiple guidelines recommend aggressively targeting normothermia using core-temperature monitoring and standard or advanced temperature-regulating devices.9,13–15 Due to the risk of severe adverse physiologic effects, therapeutic hypothermia is not recommended in the acute phase of treatment and is best reserved as a refractory or intraoperative treatment option for specific etiologies of elevated ICP, which is beyond the scope of this review.
Simple maneuvers to optimize patient positioning are widely recommended across guidelines and, given their low risk of harm, should be employed immediately for all patients with suspected raised ICP.9,31,32 The head of the bed should be raised to 30°, and the patient's head and neck kept midline to facilitate venous drainage from the cerebral vasculature. If the patient has an intracranial monitor in place, it is important to level and zero the device at the new position before proceeding with other treatments. Noxious stimuli such as tracheal suctioning should be minimized to avoid further elevating ICP.
Neuroprotective Intubation
Intubation poses unique risks to the patient with elevated ICP, as stimulation via laryngoscopy may increase heart rate, increase blood pressure, and induce bronchospasm, whereas utilization of induction agents may induce hypotension, potentially compromising the patient's hemodynamic status. Therefore, these patients should be considered to have physiologically challenging airways. The procedure should be done by the most experienced provider to reduce the risk of prolonged attempt and subsequent hypoxia, as well as unnecessary stimulation from multiple attempts. The interventions discussed below are summarized in Table 2.
Approach to Neuroprotective Intubation of Patients With Acutely Elevated Intracranial Pressure.
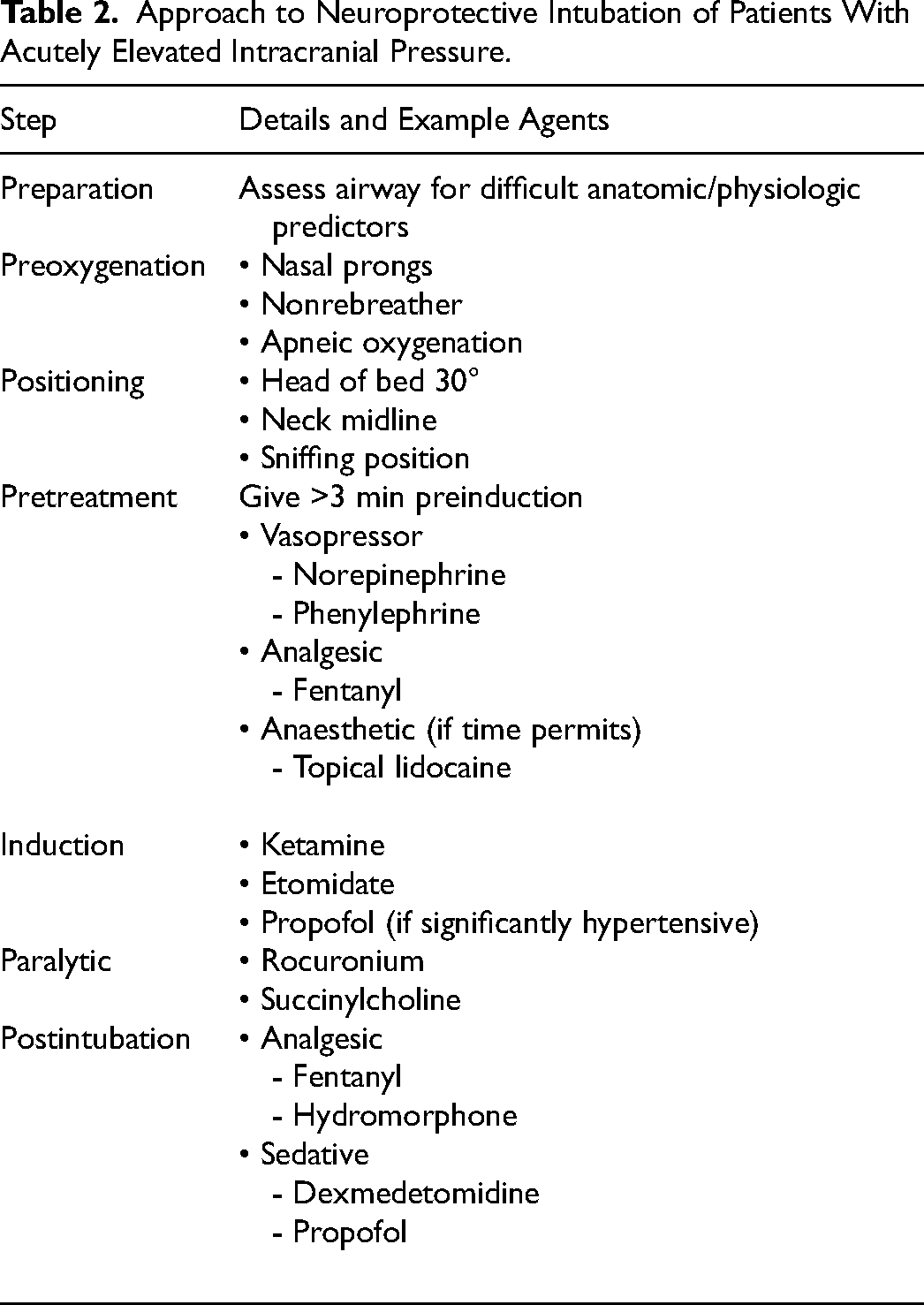
While the intubation equipment is being prepared, the noninvasive ICP management steps described above should be continued. During preoxygenation, patients should be pretreated with fentanyl at least 3 minutes before induction, as short-acting opioids have been shown to mitigate elevations in heart rate and blood pressure during rapid sequence intubation (RSI 33 ) as well as elevations in ICP during noxious procedures in brain-injured patients. 34 Hemodynamic changes should be anticipated with vasopressor therapy, which should be prepared and ready for immediate infusion.
The use of intravenous (IV) lidocaine for pretreatment in neuroprotective intubation was historically based upon a small trial of 20 patients who underwent elective neurosurgery for cerebral malignancy. 35 Since then, 2 systematic reviews of TBI patients undergoing RSI found no benefit of IV lidocaine for decreasing elevated ICP.36,37 A separate systematic review similarly found poor evidence that IV lidocaine is effective at attenuating ICP spikes nor improving patient outcomes. 38 Furthermore, a Cochrane review found that pretreatment using IV lidocaine in elective operations resulted in elevated odds ratios for bradycardia, hypotension, and other adverse events. 39 Other small studies have found similar results suggesting that IV lidocaine at doses of 2 mg/kg pretreatment 40 may induce hypotension and a drop in cerebral perfusion pressure. 38 Taken together, the evidence suggests that at best, IV lidocaine provides minimal benefit, and at worst, results in significant physiologic derangements.
Evidence supporting the use of topical lidocaine for pretreatment is also limited. A small trial of 21 patients found that instilling topical lidocaine through an endotracheal tube prior to suctioning was safe and effective at reducing stimulation-induced spikes in ICP. 41 Although this could indicate a blunting of sympathetic response induced by topical lidocaine, suctioning is notably different from endotracheal intubation and thus these results should be interpreted with caution. Topical lidocaine is used occasionally in neuroanesthesia for similar reasons; however, the evidence supporting it is largely confined to case reports and anecdotal experience. 42 Additionally, topical lidocaine takes 4 to 5 min to reach maximal effect, which may make it impractical in acute situations. 43 Based on this evidence, topical lidocaine may be helpful in situations where it does not delay intubation or jeopardize maintenance of normal ICP.
Induction should be done with a hemodynamically stable agent that does not induce hypotension or negative chronotropy, such as ketamine or etomidate.44,45 Historically, ketamine was avoided as a neuroprotective induction agent due to concerns about its potential to raise ICP. 46 More recently, large systematic reviews found little evidence supporting this effect on ICP 44 nor reduction of cerebral perfusion pressures in critically ill patients. 47 Similarly, a retrospective study of trauma patients undergoing RSI found no difference in mortality or patient outcomes between the use of ketamine versus etomidate for induction in the subset of patients with TBI. 45 Because of its vasodilating properties, propofol is best reserved for hypertensive patients but can also be used for normotensive patients provided hypotension is anticipated and treated with vasopressor therapy.
The paralytic of choice for neuroprotective RSI is succinylcholine or rocuronium, depending on institutional availability or provider preference. Concerns have been raised that succinylcholine may elevate ICP based on studies of elective neurosurgical patients48,49; however, subsequent studies have not demonstrated this nor have they shown an attenuation of elevation of ICP using pretreatment with a nondepolarizing neuromuscular blocking agent. 50 Succinylcholine is a safe option and has the potential benefit of decreasing time until repeat neurologic assessment due to its decreased duration of effect compared with rocuronium. Usual contraindications for succinylcholine, such as for patients with acute hyperkalemia, should be respected.
During endotracheal tube placement, particular care should be made for a gentle, nontraumatic insertion that minimizes glottic stimulation. Continuous analgesia and sedation following intubation should be maintained using opioids and either dexmedetomidine or propofol, titrated to clinical effect (as measured by a standardized sedation scale) and resolution of signs of elevated ICP. The 2019 Seattle International Severe Traumatic Brain Injury Consensus Conference (SIBICC) guidelines recommend considering a single dose of a neuromuscular paralytic agent in an adequately sedated patient to assess for efficacy in decreasing ICP and proceeding with an infusion if efficacious. 12
Hyperosmolar Therapy
Hyperosmolar therapy may reduce ICP via 2 primary physiologic mechanisms. Initially, a rheologic effect occurs via the expansion of plasma volume and reduction of blood viscosity, thereby improving cerebral oxygen delivery and resulting in cerebral vasoconstriction. 51 Secondly, the production of an osmolar gradient in the blood draws water from the extravascular (ie, brain tissue) into the intravascular compartment, thereby promoting venous drainage and reducing ICP. 51 Hypertonic saline (HTS) and mannitol have been widely used for the treatment of TBI for decades. 52 The 2020 guidelines from the Neurocritical Care Society support the use of hyperosmolar therapy in the management of elevated ICP from TBI, subarachnoid hemorrhage, intracerebral hemorrhage, and acute ischemic stroke based on demonstrated efficacy in several randomized controlled trials. 10
The effect of mannitol on ICP is thought to begin in minutes, peaking between 15 and 120 minutes and lasting a total of 1 to 5 hours based on sparse pharmacodynamic data.53,54 Notably, it has been shown to cause adverse effects including hypovolemia and renal failure.55,56 Although mannitol was formerly recommended as the first-line agent for hyperosmolar ICP therapy, 57 a meta-analysis found HTS to be more effective than mannitol at reducing ICP without the associated adverse effects. 58 A small RCT found that HTS had a faster time to onset and a more robust ICP reduction when mannitol failed to reduce ICP in TBI patients. 59
The NCS guidelines generally recommend the use of HTS over mannitol for the treatment of elevated ICP in TBI, subarachnoid hemorrhage, and intracerebral hemorrhage, 10 while acknowledging the absence of large, high-quality trials on the subject. The 2019 SIBICC guidelines recommend targeting serum sodium of less than 155 mEq/L and osmolality less than 320 mEq/L, with regular monitoring every 4 to 6 hours. 12 As an alternative to serum osmoles, ensuring a normal osmolar gap is a useful monitoring target when initiating mannitol therapy. 60 When initiating hyperosmolar therapy, a Foley catheter should be placed to monitor urine output.
Hyperventilation
Hyperventilation therapy relies on the principle that inducing cerebral arterial vasoconstriction via hypocapnia results in reduced cerebral blood volume and therefore decreases ICP. Although hyperventilation has been shown to rapidly reduce ICP, it has not been associated with improved patient outcomes in TBI.61,62 Nonetheless, it is strongly recommended by the NCS guidelines, with the rationale that the benefit of reducing harm from elevated ICP outweighs the risk of cerebral ischemia, which has only been demonstrated with prolonged employment. 10 In scenarios where hyperventilation is used, it is best employed as a temporizing measure for impending or active cerebral herniation until more definitive treatment (eg, neurosurgery) can be provided.
Initially, a low-normal PaCO2 between 35 and 38 mm Hg and end-tidal CO2 between 32 and 35 mm Hg (to account for dead space ventilation in mechanically ventilated patients with an endotracheal tube) should be targeted. If elevated ICP persists, the target may be lowered to 32 to 35 mm Hg PaCO2 and 28 to 32 mm Hg end-tidal CO2 for a maximum of 2 hours to mitigate ischemic brain injury.
Corticosteroids
There is little evidence to support the use of corticosteroids for several etiologies of elevated ICP. The landmark CRASH-1 trial found a higher risk of mortality within 2 weeks with IV corticosteroid use in 10 008 adult TBI patients. 63 Corticosteroid use has shown minimal benefit with increased rates of complications in intracerebral hemorrhage10,64 and ischemic stroke. 65
In the case of bacterial meningitis, the use of corticosteroids has not shown consistent improvement in patient mortality in several meta-analyses,66,67 although it does appear to reduce rates of sensorineural hearing loss. 68 In 2004, the Infectious Diseases Society of America released guidelines supporting the use of corticosteroids in adults with bacterial meningitis. 69 Although limited evidence exists on the use of steroids for elevated ICP caused by central nervous system tumors, there is some evidence it may be effective specifically for temporary symptomatic relief of the vasogenic edema caused by brain metastases. 70
Corticosteroids should, therefore, be reserved for patients with elevated ICP only in the context of bacterial meningitis or central nervous system tumors and not empirically in the patient with an undifferentiated underlying cause.
Conclusion
Acutely elevated ICP is a life-threatening condition that may result from a variety of neurologic disease processes. Early recognition depends on the findings of physical examination and neuroimaging modalities, with the addition of an invasive monitor if time and resources allow. Early treatment may need to be initiated before the underlying etiology is known, in which case specific treatment guidelines are less helpful. An empiric approach based on hemodynamic control and prevention of secondary brain injury can optimize the initial resuscitation while further investigations and definitive management are arranged. In cases where the patient's ability to protect their airway or respiratory status is compromised, neuroprotective intubation should be performed by the most experienced provider. Adequate analgesia and sedation can prevent abrupt increases in ICP, while hyperosmolar therapy and hyperventilation can be used for patients that continue to display signs of elevated ICP despite initial management. As future studies and trials elucidate the value of specific medications and interventions, clinicians must continue to rely on a multimodal approach to these complex, critical situations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
