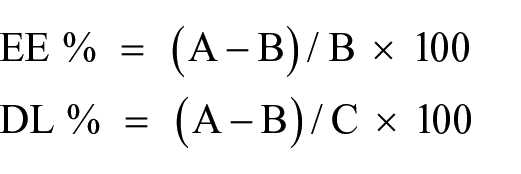Abstract
Peripheral neuropathies are associated with various detrimental complications, leading to life-debilitating disorders, including neuropathic pain. Hence, the current study aimed to incorporate naringin, a potential natural component, into chitosan nanoparticles (NPs) to ameliorate the complications resulting from chronic constriction injury (CCI) induced painful neuropathy. The prepared NPs had a particle size of 220 nm and PDI = 0.37, with relatively spherical morphology and zeta potential of +41.5 mV. The relevant analyses indicated the loading and high encapsulation efficiency of naringin into the NPs as well as a prolonged release of naringin. The anti-neuropathic evaluations of chronic constriction injury (CCI)-induced rats treated with naringin-loaded NPs (10 mg/kg) remarkably ameliorated hyperalgesia and cold allodynia. In addition, the treatment with naringin-loaded NPs led to improvements in sensory and locomotor impairment, as evidenced by changes in behavioral parameters such as reduced paw licking, increased rearings, and enhanced crossings. The NPs treatment significantly attenuated the elevated levels of nitrite and restored the reduced glutathione level in the serum of CCI-induced rats. Moreover, histopathological analysis exhibited regeneration of the sciatic nerve injury through reducing myelin degeneration, axonal swelling, and nerve fiber derangement. Therefore, these findings suggest that the naringin-loaded chitosan NPs has promising pharmacological activities for the treatment of neuropathic pain sufferers.
Introduction
Neuropathic pain, characterized by its long-lasting nature and lack of response to typical pain relievers, results from damage or disease of the somatosensory system, with rising universal incidence and roughly 3–17 % global prevalence rate.1,2 A diverse range of diseases and lesions, such as autoimmune/neurodegenerative conditions (e.g. multiple sclerosis and Parkinson’s disease), metabolic disorders (e.g. diabetic neuropathy), infections (e.g. HIV, leprosy, and shingles), vascular diseases (e.g. stroke), injuries (e.g. trauma and amputation), and cancer, are associated with neuropathic pain.3,4 Patients may exhibit various symptoms and signs clinically, depending on the anatomical localization or underlying cause. These manifestations are typically characterized by hyperalgesia (abnormal hypersensitivity to stimuli) and allodynia (nociceptive responses to non-noxious stimuli).5,6 If not controlled/treated, these sensory abnormalities may lead to persistent and severe pain, total numbness, eventually complete loss of sensation, limb disability and amputation. The underlying mechanisms of such pain have not been entirely identified due to their complex pathophysiology and heterogeneous etiology, comprising of the aberrant sympathetic–somatic nervous system interactions, the imbalance between excitatory and inhibitory somatosensory signaling pathways alterations in ion channels, disbalance in the production of various cytokines and increased levels of oxidative stress.4,7 –9
The pharmacological treatments of neuropathic pain are generally palliative to relieve the relevant symptoms using prescribing three-line medications that include gabapentinoids, tricyclic antidepressants and topical lidocaine as the first-line, opioid analgesics and tramadol as second-line, antiepileptic drugs and topical capsaicin as third-line.10,11 These treatment measures are performed to merely suppress symptoms, while efforts should be proceeding to achieve precisely mechanism-based treatments and efficient pharmacological approaches. Consequently, the majority of existing treatments have demonstrated moderate effectiveness and the potential for undesirable side effects, which restricts their utilization 2 ; therefore, alternative novel and effective therapeutic approaches are imperative for patients .
Nowadays, natural substances and their secondary metabolites have emerged as promising therapeutic candidates for treating neuropathic pain complications. Various plant-derived compounds such as capsaicin, curcumin, salicin and triptolide have been reported to provide pharmacotherapeutic activities against neuropathic pain.12 –15 These substances have preferred to synthetic drugs due to their less complication and fewer side effects as well as can confer several actions/effects concurrently such as anti-oxidant activity, anti-inflammatory, neuroprotective, modulation of pain management systems (opioids, serotonergic and cannabinoid systems) and ion channels. 15
Naringin, 4′,5,7- trihydroxyflavanone-7-rhamnoglucoside, is one of the main bioactive polyphenols derived from grape fruit and related citrus species. 16 Naringin has become of interest due to its therapeutic efficacy against diabetes, cancers, respiratory, cardiovascular and gastrointestinal, hepatic and neurodegenerative disorders through the relevant pharmacological effects for example, analgesic, anti-oxidant, anti-mutagenic, anti-inflammatory, anti-apoptotic, anti-arrhythmics and neuroprotective capacity.17,18 Naringin shows neuroprotection and the potential efficiency against neuropathic pain owning to inhibiting neuroinflammation through down-regulating free radicals/inflammatory mediators’ levels and upregulating antioxidant enzymes.19 –22 Along these, the molecular antinociceptive mechanisms of naringin have been verified in murine models through diminishing neuronal hyperexcitability by increasing the nociceptive threshold for activation and suppressing current of cation channels.17,23 On the other hand, naringin has a low water solubility and fast elimination rate, resulting in poor absorption through the gastrointestinal tract and hence poor bioavailability (⩾15 %).24,25
Developing novel drug delivery systems (DDSs) enhances the bioavailability and efficiency of herbal-derived medicines. Nanocarriers, delivery systems having nano-scale size and unique properties, have shown tremendous opportunities in DDSs, for instance, improving efficacy and enhancing absorption in a given tissue through encapsulation of therapeutics agents, protection against premature degradation, improving the solubility and pharmacokinetic parameters.26,27
One of the promising potential nanocarriers is chitosan nanoparticles (NPs). Chitosan is a polymer made up of random repeated units of N-acetyl-D-glucosamine and D-glucosamine, which are obtained through the deacetylation of the original polymer chitin.28,29 Chitosan NPs are non-toxic, biodegradable, biocompatible, and polycationic due to the presence of plenty of amine groups (–NH2) that facilitate the electrostatic interaction with negative cell membranes and biomacromolecules.26,27 In addition, chitosan NPs have presented anti-microbial, anti-oxidant, and mucoadhesive capabilities, which latter feature endows a prolonged contact time with the absorptive surface, modulating the integrity of epithelial tight junctions and thereby promoting the absorption. 28 Moreover, chitosan NPs as carriers could be incorporated with the therapeutic agent(s) for efficient and intact delivery.
As far as we know, there has been no research conducted to explore the utilization of chitosan NPs incorporating naringin for the purpose of treating neuropathic pain. Therefore, the current study intended to develop naringin-loaded chitosan NPs to effectively ameliorate the neuropathic pain complications related to sciatic nerve injury. Hence, naringin was incorporated into chitosan NPs and then characterized in terms of physicochemical properties. Then, the release pattern of naringin from the loaded NPs was assessed. Subsequently, the effectiveness of the formulations in relieving neuropathic pain was evaluated in a rat model with CCI of the sciatic nerve. Finally, the level of nitric oxide (NO) and glutathione (GSH) in the serum of rats was measured as biomarkers that can regulate and decrease the neuronal damage.
Materials and methods
Materials
Naringin (MW: 580%, purity ⩾ 95%) and chitosan (MW: 50–190 KD, 75%–85% deacetylated) were supplied from Sigma-Aldrich (Germany). Sodium tripolyphosphate (TPP) and acetic acid were obtained from Merck Company (Darmstadt, Germany). All other chemicals and solvents utilized in the study were of analytical grade.
Preparation of naringin-loaded chitosan NPs
Chitosan NPs containing naringin was synthesized by the ionic gelation method. 30 Firstly, an acetic acid solution (1% (v/v)) of chitosan at 2 mg/mL concentration was stirred overnight at room temperature. Then, the prepared dispersion was filtered through a 0.4 μm pore size membrane filter to remove undispersed polymers. Next, naringin (30 mg) was added to a 2% w/v TPP solution (20 mL). After mixing well, the naringin solution was added to the chitosan dispersion at the ratio of 1:5. Last, the mixture was stirred continuously for 60 min to form a suspension of naringin-loaded chitosan NPs. Finally, the mixture was centrifuged at 16,000g for 1 h at 4°C, the sediment (the obtained NPs) was collected, resuspended in deionized water, and freeze-dried for further experiments. Similarly, the blank chitosan NPs (without naringin) was fabricated following the above process without adding naringin.
Particle size, morphology and zeta potential determination
After centrifuging the prepared nanoparticle mixture, the obtained NPs were resuspended in deionized water and subjected to analysis. The particle size and polydispersity index (PDI) of blank chitosan NPs and naringin-loaded NPs were measured by a Malvern Zetasizer (Malvern, UK). Also, the zeta potential value of the prepared NPs was determined by electrophoretic light scattering (Zeta sizer, Malvern, UK) at 25°C, 150 V. Additionally, the morphology of the NPs was visualized using scanning electron microscopy (SEM, DSM 960A, Zeiss, Germany). The microscopy observation was performed at an accelerating voltage of 25.0 kV, and the samples were sputtered with a thin layer of gold.
Encapsulation efficiency and drug loading measurements
The NP suspension was centrifuged to separate the supernatant, which was then used to evaluate the encapsulation efficiency (EE%) and drug loading (DL%) of naringin in the loaded NPs. The concentration of naringin in the supernatant, which represents the unencapsulated naringin, was measured based on the concentration-absorption calibration curve of naringin using a UV-Vis spectrophotometer (Cecil Instruments Ltd., UK) at 282 nm. 31 The experiment was repeated three times. EE % and DL % were calculated according to the following formula 32 :
Where A represents the total mass of naringin used, B represents the mass of unloaded naringin, and C represents the total mass of nanoparticles obtained.
Infrared spectroscopy analysis
FTIR analysis was conducted to clarify the naringin incorporation and the interactions between the prepared NPs ingredients. The experiment was performed using an FTIR spectrophotometer (Shimadzu IRAffinity IS) at room temperature from 400 to 4000 cm−1.
In vitro release study
The rate of naringin release from naringin-loaded NPs was determined through in vitro testing using the dialysis bag method. 33 Initially, a dialysis membrane bag with a molecular weight cut-off of 12,000 Da (Spectra/Por®) was filled with 10 mg of naringin-loaded NPs. The membrane bag containing the NPs was then placed in 100 mL of a release medium composed of a mixture of phosphate-buffered saline (PBS) and methanol in a ratio of 9:1. The release medium had a pH of 7.4. The system was subsequently incubated at a temperature of 37°C in a shaker, with gentle agitation at 133 g. At predetermined time intervals, 1 mL of the medium was collected and substituted with an equal volume of fresh medium to maintain sink condition. The amount of released naringin at each determined time was quantified spectrophotometrically at 282 nm from the obtained standard curve (absorbance vs concentration). The experiment was repeated three times. The graph of the cumulative release percentage of naringin from the NPs versus time was plotted.
Animal experiments (in vivo studies)
All the animal experiments were conducted under the Ethic Committee for Animal Experiments and were in accordance with the institutional guidelines (Approval ID: IR.KUMS.REC.1400.559). Eight-week-old male Wistar rats, with an average weight of 240 ± 20 g, were provided unrestricted access to food and water. They were housed in controlled conditions of 25°C temperature, 60%–80% relative humidity, and a 12-hour light/dark cycle. The animals underwent intraperitoneal (IP) anesthesia prior to surgery using an 80:10 mL/kg combination of ketamine and xylazine.
Neuropathic pain resulting from peripheral nerve lesions was induced by a CCI of the rat sciatic nerve. 34 After deep anesthesia, the surgical region was clean-shaven and sanitized with ethanol 70%. CCI was induced on the left leg by making an incision on the skin and cutting through the connective tissue between the gluteus superficialis and biceps femoris muscles to expose the sciatic nerve. Then, four loose silk ligatures are tied around the sciatic nerve at intervals of 1 mm to merely occlude but not stop epineural blood flow. At last, the incision was closed by suturing the muscles and skin. Following the procedure, the animal was given 24 h to recover before starting the pain hypersensitivity tests and locomotor assessment. As sham group, the incision procedure was done and sciatic nerve was separated but not ligated.
Forty-two rats were randomly divided into seven groups, each containing seven rats. The rats in these groups were treated every 3 days over a period of 2 weeks following sciatic nerve injury. The following administration methods were applied:
Group 1: Sham, only treated with water as vehicle (IP injection); group 2: CCI received compression injury without treatment; group 3: Positive control, CCI treated with standard drug gabapentin (GBP, 30 mg/kg, IP injection); group 4: CCI treated with naringin (10 mg/kg, IP injection); group 5: CCI received naringin-loaded NPs (5 mg/kg, IP injection); group 6: CCI received naringin-loaded NPs (10 mg/kg, IP injection); and group 7: CCI received naringin-loaded NPs (15 mg/kg, IP injection).
Pain hypersensitivity assessments
To assess the impact of treatments on the heat pain threshold in rats, the hot-plate test (HPT) was conducted during the entire 2-week period following CCI. This test provides an objective and measurable analysis of behavioral responses. Thirty minutes after receiving the treatments, the rats were placed in a cylindrical enclosure with a metallic plate as the floor. Each animal was then positioned on the heated surface of a hot plate, maintained at a temperature range of 50°C–56°C. The latency time, which refers to the duration it took for the rat to either lick its paw or jump off the hot plate in response to the heat-induced pain, was measured and compared among the different groups.
Acetone drop test (ADT), which records the aversive responses triggered by evaporative cooling, was used to measure cold allodynia in CCI rats, separately on at least three rats from each group in order to not interfere with the HPT results. In this regard, the experiment was initiated 30 min after the morning injection. The animals were placed inside a glass cylinder with a mesh floor. A drop of acetone (100 μL) was applied to the mid-plantar area of the hind paw, causing the skin to cool down to a non-painful temperature range of 15°–21°. 35 The animals were observed for a duration of 20 s, and their response to the acetone stimulus was evaluated using a 6-grade scoring system. The scoring system was as follows: 0 indicated no response, 1 indicated a startle response without paw withdrawal, 2 indicated a brief withdrawal of the paw, 3 indicated a prolonged withdrawal lasting between 5 and 30 s, 4 indicated either a prolonged withdrawal or repeated flicking of the paw, and 5 indicated repeated flicking of the paw accompanied by paw licking. 36 The procedure was repeated thrice with a 5-min break between each test.
Locomotor activity assessment
Open-field test (OFT) was used to quantitatively inform about animals’ anxiety-like behaviors and locomotor impairments. After the treatments, three CCI-induced rats were subjected to OFT every 3 days over 2 weeks. The used PFT apparatus consisted of a field (1 m2) divided into 16 same squares surrounded by high walls. Each animal was placed at the center of the field and given 5 min to freely explore. During this time, relevant behaviors such as crossings (counting the number of times the animal’s legs crossed the lines of a square), rearings (counting the number of times the animal stood on its hind legs), and paw licking (counting the number of times the animal licked its paw) were counted and recorded. 37 After the end of each use, the field was disinfected with 70% ethanol so that the test conditions were the same for all rats.
Determination of involved markers
The measurement of serum glutathione (GSH) secretion was performed using a colorimetric assay employing the sulfhydryl reagent 5,5′-dithio-bis(2-nitrobenzoic acid) (DTNB). On the 14th day, blood samples were collected from the anesthetized rats via the retro-orbital vein, and the serum was separated and transferred into heparinized containers. The separated plasma was then combined with DTNB, leading to the formation of a yellow derivative called 5′-thio-2-nitrobenzoic acid (TNB), which was measured at a wavelength of 412 nm using photospectroscopy. 38
Likewise, the serum level of NO was quantified with a colorimetric assay through Griess reagent (sulfanilamide and N-naphthyleth-ylenediamine) that measures nitrite (NO2–) as one of the stable metabolites of NO. Initially, the Griess reagent (500 μL) was exposed to the serum samples (100 μL), which resulted in a purple azo product. Then, the optical absorption of samples was measured at 546 nm. The concentration of nitrite in the samples was determined by using a standard curve, and the results are expressed in micromoles per milliliter of serum. 39
Histological assessment
The animals were sacrificed 14 days after injury by an excessive dose of anesthetics (a mixture of ketamine: xylazine, 100 mg/kg: 20 mg/kg). Then, the injured sciatic nerve (about 10 mm long) was removed, transcardially fixed with 4% paraformaldehyde in PBS and stored at 4°C overnight. The removed tissues were processed in a tissue processor (Didban sabz DS2080/H; Iran) and subsequently embedded in paraffin. Next, the tissues were cut with an ultra-microtome to obtain semi-thin sections with 5 μm-thick and placed on the slides. Three sections from each group were stained with Hematoxylin and Eosin staining (H&E) and photo-captured by a light microscope.
Statistical analysis
The mean and standard error of mean (Mean ± SEM) were reported for each experiment, which was carried out in triplicate. Utilizing OriginPro 2020 and Graph Pad Prism 9, all data and graphs were analyzed. The normality test in this study was carried out using the Shapiro–Wilk method. A p-value of < 0.05 was considered to indicate the statistically significant between the groups. As a results, ns indicates p > 0.05, * indicates p < 0.05, ** indicates p < 0.01, *** indicates p < 0.001, and **** indicates p < 0.0001.
Results and discussion
Particle size, morphology and zeta potential
The resultant blank chitosan particles (without naringin) were found to have a particle size of 219.8 nm with a PDI value of 0.33 (Figure 1(a)), thus specifying the homogeneous nano-scale particle size having narrow distribution. The naringin loading into chitosan particles resulted in forming NPs with 220.2 nm and narrow size dispersity (PDI = 0.372) (Figure 1(b)). These results indicated incorporation of naringin was not considerably altered the particle size and the NPs with proper size distribution was formed, which could confer a large surface area favorable for cellular interaction and internalization.40,41 Using more than 30 mg of naringin in preparing NPs caused a significant increase in particle size and changed their morphology; therefore, the higher amount was not used.
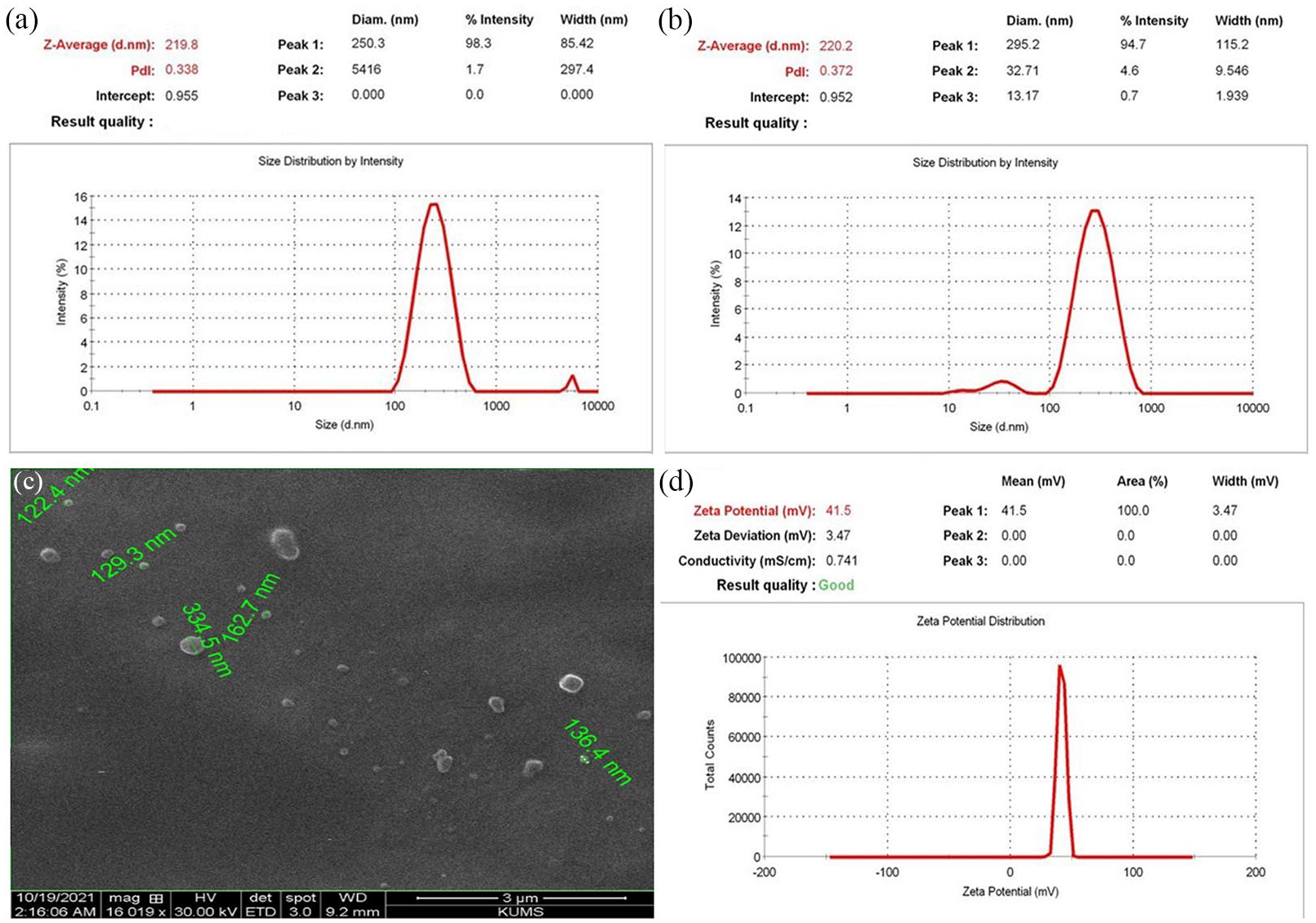
Particle size distribution of blank (unloaded) chitosan particles (a), and naringin-loaded chitosan particle (b) obtained from DLS technique. SEM micrograph (c) and zeta potential distribution (d) of naringin-loaded chitosan.
Moreover, the obtained electron microscopic imaging of naringin-loaded chitosan NPs is depicted in Figure 1(c). Spherical particles with smooth surfaces and relatively uniform non-aggregated morphology were observed in the SEM micrograph. Besides, the image confirmed the particle sizes between 100 and 300 nm, in accordance with the DLS findings.
In addition, the zeta potential of naringin-loaded chitosan NPs, as an indicator of particle stability and surface charge, was evaluated. As shown in Figure 1(d), the NPs had adopted + 41.5 mV electrical potential, which implies the high electrostatic stability of the prepared NPs. Also, the positive charge is possibly attributed to the residual -NH3+ in chitosan chains on the surface of NPs. 42
Drug loading and encapsulation efficiency
The EE % and DL % of naringin in the chitosan NPs were determined to be 94.4 ± 2.2% and 12.2 ± 3.6%, respectively. These findings show that the chitosan NPs have loaded with the appropriate content of naringin.
FTIR characterization
The FTIR analysis was performed to determine the samples’ functional groups and verify the extract’s presence in chitosan NPs. According to Figure 2(a), the spectrum of pure naringin showed characteristic sharp peaks at 1041 and 1089 cm-1 related to the C-O-C bonds of the ether as well as a peak at 1134 cm-1 could be attributed to the vibration of phenolic –OH groups. The peaks for C=C in aromatic cyclic groups, aromatic C=O stretching, and C-H bonds of the CH3 group were observed at 1517, 1645 and 2924 cm-1, respectively. Also, a broad peak around 3419 cm-1 appeared for the vibration of hydroxyl groups (OH). These FTIR results indicate entire matching with the naringin structure, consistent with previous investigations.43,44 In addition, the spectrum of chitosan NPs (Figure 2(b)) presents distinctive absorption peaks at 1068, 1238, 1431 and 1637 cm-1, indicating C-O stretching, C-N stretching, C-H bending, and N-H bending vibrational frequencies, respectively. The peaks at 2937, 3387, and 3562 cm-1 also appeared, matching C-H stretching, −OH and −NH2 stretching bonds in the chitosan structure.42,45 In the spectrum of naringin–loaded chitosan NPs (Figure 2(c)), the prominent bands of naringin and chitosan appeared in the loaded NPs. Further, the presence of distinct peaks corresponding to naringin in the spectrum of the loaded chitosan NPs indicated the successful encapsulation of naringin without any detrimental chemical interactions. However, the absorption band around 3398 cm−1 intensified, possibly due to hydrogen bonding between the naringin and the polymeric matrix, as well as the peak at 1641 cm-1 (N-H stretching) was attenuated, which is possibly attributed to the physical interaction between the positively charged amine groups of chitosan and the negatively charged hydroxyl groups of naringin.
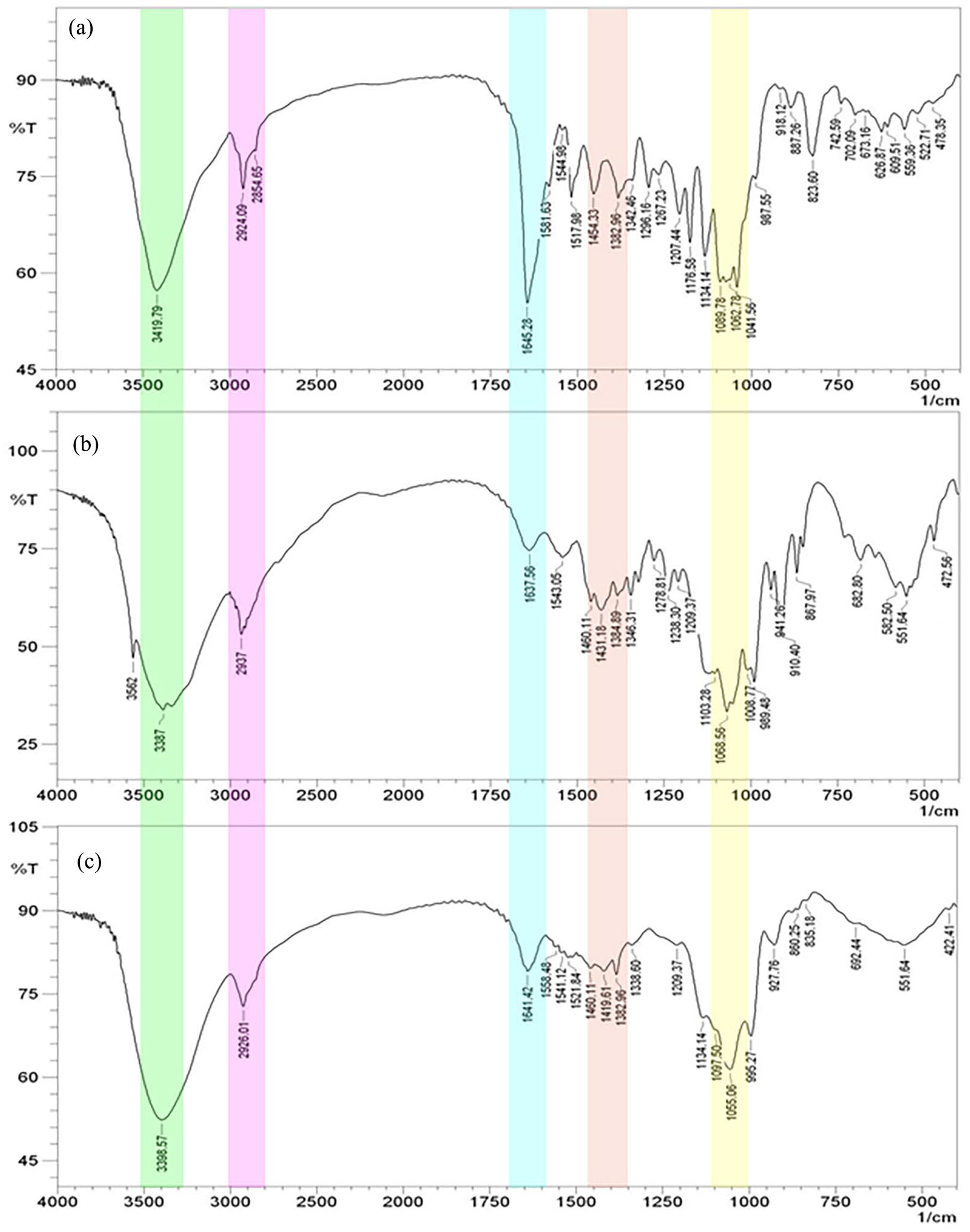
FTIR spectra of pure naringin (a), blank chitosan NPs (b), and naringin-loaded chitosan NPs (c).
In vitro release kinetics
To determine the release profile from the prepared NPs, firstly, the UV-Vis absorption spectrum of naringin was measured in different concentrations. As a result, the highest absorption (λmax) of naringin in PBS medium was observed at the wavelength of 228 nm (Figure 3(a)). Then, the standard curve of naringin concentration versus absorption was made in PBS at the obtained λmax (Figure 3(b)). The resultant standard curve of naringin indicate a good linearity with a high correlation coefficient (R2 = 0.92).
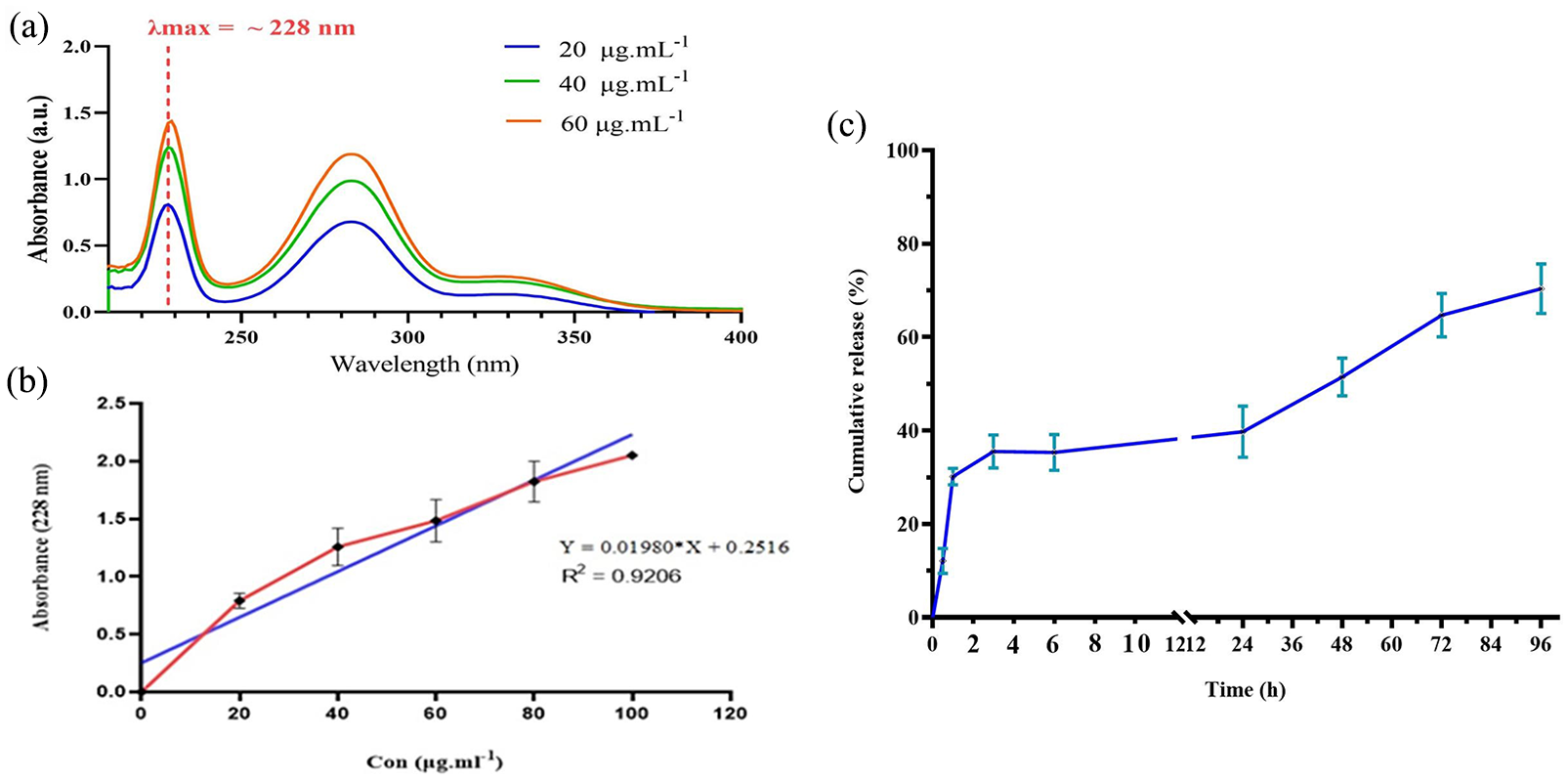
(a) UV-Vis spectrum of naringin at different concentration, (b) standard curve of naringin in PBS and (c) the in vitro release profile of naringin from the loaded NPs (n = 3).
Furthermore, the release profile of naringin from the chitosan NPs is depicted in Figure 3(c). The cumulative release of naringin was 12.1% at 0.5 h and 35.5% after 3 h, which indicate considerable burst release did not occur. While after 24 h and 48 h were observed nearly 42% and 53% cumulative release, respectively. In the following, 65% release of naringin content happened at 72 h and reached about 70% after 96 h so that more liberation was not obtained, possibly due to the drug’s low solubility in aqueous medium. 46 The resultant release profile indicates a biphasic pattern as a more release amount in the first day followed by a prolonged and sustained release profile.
Animal pain response examinations
To examine whether the treatments could improve CCI-induced pain behaviors, we scrutinized the pain threshold by HPT and ADT in the animals. The latency time to paw-licking in the different treated groups was depicted in Figure 4(a). The obtained results showed that in the sham group, the latency time did not change from the first day to the 14th day of the study, and no difference was observed at all time-points tested. Also, latency time in the animals of the positive group compared to the CCI-induced group increased significantly during the study period (p < 0.001). In addition, among varying doses of naringin-loaded NPs, the results showed that the group receiving the loaded NPs with a dose of 10 mg/kg had a better response roughly similar to the positive group response, as well as a significant rise in the latency time was observed compared to the CCI group during the study period (p < 0.001).
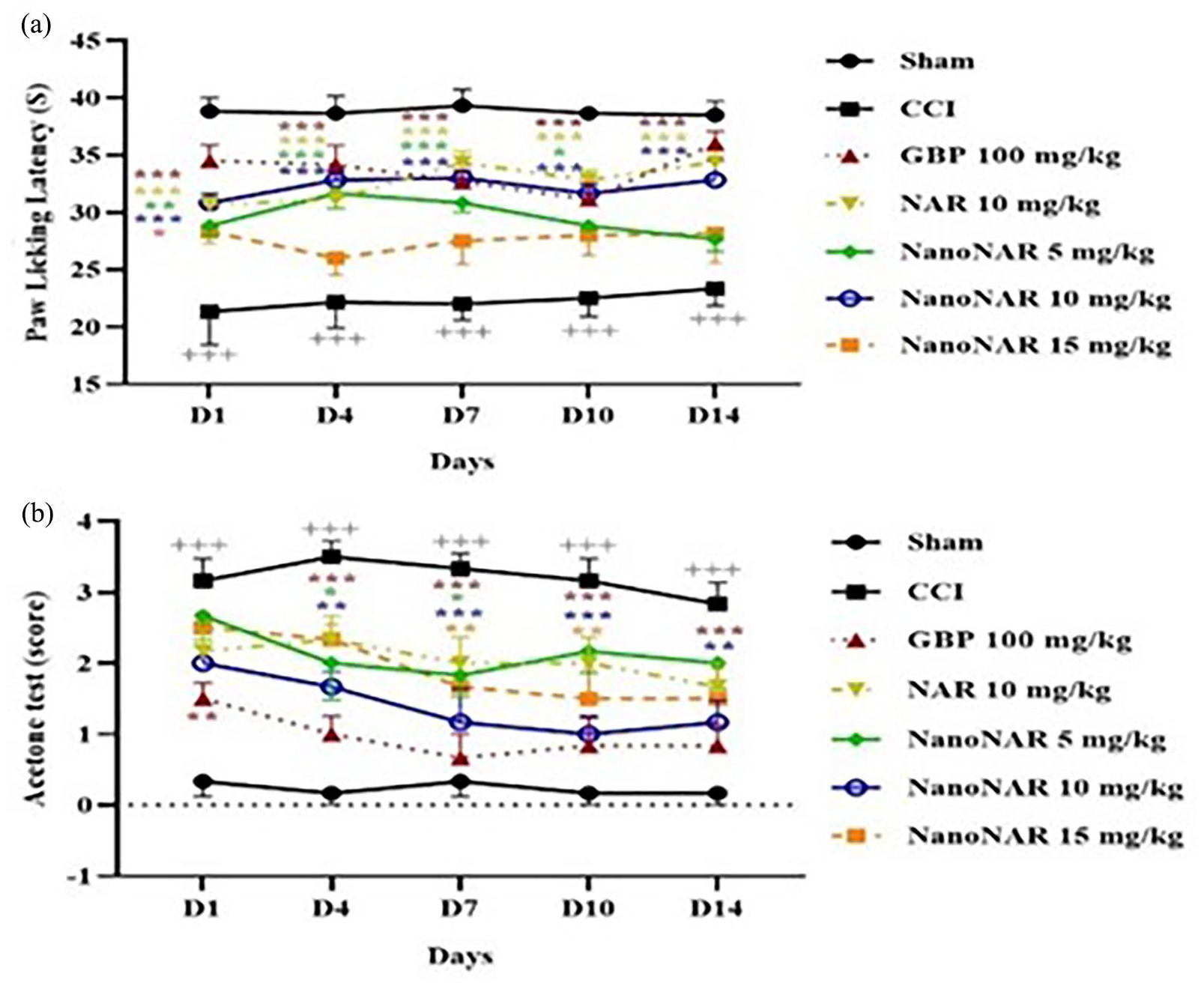
(a) The effect of different treatments on latency time to pain stimulus in hot plate test and (b) the obtained scores from cold stimulus in acetone drop test (+ indicates significant levels between sham and CCI groups, and * indicates the other groups relative to CCI group) (n = 3).
Furthermore, cold allodynia was evaluated by ADT for the analysis of the latency to hind paw licking or withdrawal. As shown in Figure 4(b), the reaction to the cold stimulus in the sham group were the same (no response) at all time-points tested without significant differences. After CCI induction, the animals reacted to the stimulus and a significant difference was observed compared to the sham group in all the examined days (p < 0.001). Free naringin (10 mg/kg), naringin-loaded NPs at concentrations 5 and 15 mg/kg showed fewer responses than the sham group. At the same time, a greater reduction in the responses was observed in 10 mg/kg naringin-loaded NPs and GBP-treated groups that a significant difference was observed compared to the CCI group (p < 0.001).
According to the screening of thermal hyperalgesia/allodynia, the analgesic effects of 10 mg/kg naringin-loaded NPs were prominent and nearly similar to GBP; however, these effects were observed with much lower concentrations (10vs 100 mg/kg). Naringin has been reported as a novel analgesic candidate for pain relief, exhibiting antinociceptive effects, possibly due to the stimulation of KIR3 channels and the dopaminergic system, as well as the inhibition of transient receptor potential vanilloid member 1 channels.17,47 Moreover, the effects observed with naringin-loaded NPs could be attributed to the combined characteristics of chitosan NPs and the continuous release of naringin,48,49 along with the neuroprotective effects of naringin due to its antioxidant, anti-inflammatory, anti-apoptotic, and analgesic activities.17,50,51
In following, the anxiety- and depressive like behaviors were evaluated via OFT in the animals by using the three parameters. Regarding the parameter of the number of licking times (Figure 5), in the sham group, no difference was identified during the study period, and the number of licking times was remarkably low. After the induction of neuropathic pain (CCI group), the number of licking times increased considerably, and a significant difference was observed compared to the sham group (p < 0.001). Although free naringin led to a decrease in the licking numbers, the groups treated with different doses of naringin-loaded NPs showed more reduction, especially receiving the amount of 10 mg/kg the NPs with a significant difference compared to the CCI group (p < 0.001).
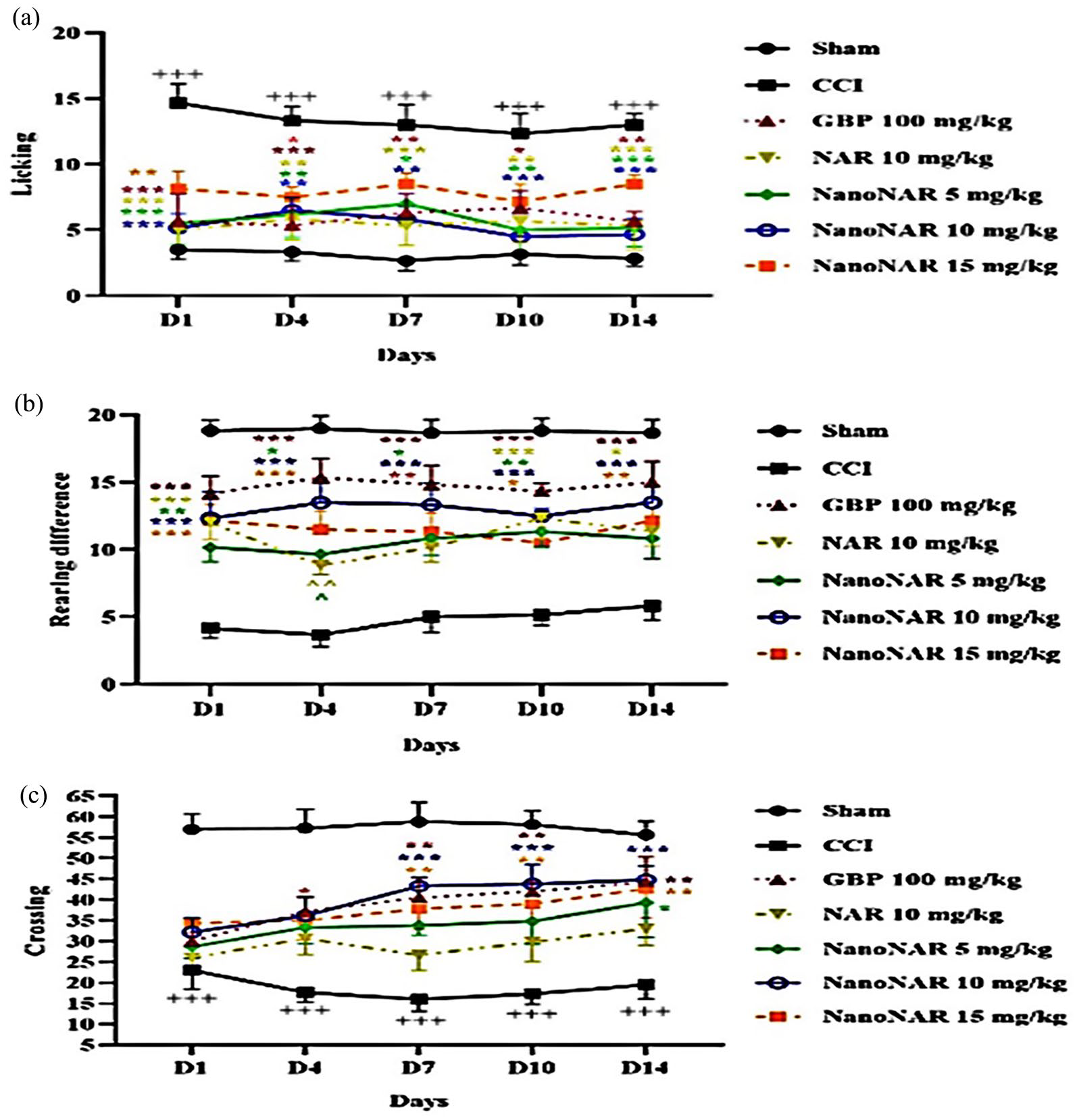
Results of open field test: (a) the number of licking, (b) rearing time, and (c) crossings during tested time-points tested (+ indicates significant levels between sham and CCI groups, and * indicates the other groups relative to CCI group) (n = 3).
The analysis of the number of rearings, stands on its hind legs (Figure 5(b)), was shown a high rate at different time intervals in the sham group. After CCI induction, the rearing number reduced significantly compared to all other treatment groups. Also, GBP treatment significantly (p < 0.001) raised the relevant parameter compared to sham counterparts. The animals treated with naringin-loaded NPs (at 10 mg/kg) exhibited a more increment in the rearing number relative to the CCI group and free naringin at most time points.
Further analysis of the number of crossings of square lines (Figure 5(c)) revealed a significant reduction compared with all of the other treatment groups after CCI induction. This parameter was elevated in rats treated with free naringin during the investigation. While the naringin-loaded NPs resulted in more promotion in the crossing numbers, particularly at a concentration of 10 mg/kg, similar to GBP treatment. This response was statistically different from those observed in the CCI group (p < 0.001).
In the experiments, it was observed that the administration of naringin-loaded NPs (at a dosage of 10 mg/kg) resulted in stronger anti-anxiolytic effects compared to free naringin. These effects were similar to those observed with a dosage of 100 mg/kg of GBP. These behavioral improvements can likely be attributed to the restoration of locomotor activity, as indicated by changes in the number of paw licking, rearings, and crossings. Moreover, naringin-loaded chitosan NPs, owing to their high surface-to-area, positive surface charge and mucoadhesive properties, can adhere more effectively to cells and internalize efficiently, possibly loading to improve the recovery of locomotor functions and tissue repair following CCI. These findings are in accord with the earlier studies that demonstrated the capability of chitosan NPs containing various drugs to enhance neuronal injury recovery and improve locomotor function.52 –54
Inflammation markers studies
Changes in GSH levels and related metabolites are correlated with inflammation and pain-like behaviors in CCI induced neuropathy. The objective of measuring NO and GSH in this study was to gain insights into the pathophysiology of neuropathic pain. Oxidative stress, in particular, has detrimental effects on neuronal cells in neuropathic pain, leading to alterations in GSH and NO levels. By measuring these biomarkers in the serum of rats, we aimed to better understand and regulate the neuronal damage associated with oxidative stress. This investigation provides valuable information on the mechanisms underlying neuropathic pain and offers potential avenues for therapeutic interventions. As the determination of GSH in serum shown in Figure 6(a), CCI induction decreased glutathione levels and potentially raised oxidative stress, in line with earlier reports.55,56 GBP treatment significantly raised the reduced levels of GSH in the CCI-induced animals (p < 0.01). Additionally, both free naringin and naringin-loaded NPs at doses of 5 and 15 mg/kg were not able to statistically enhance the levels of GSH compared to the CCI group’s level. In contrast, naringin-loaded NPs at 10 mg/kg led to considerable promotion in the level of GSH as reflected by a significant increase in the respective component relative to the CCI group (p < 0.001) and naringin-loaded NPs at 5 mg/kg (p < 0.05). These findings imply that the administration of naringin-loaded NPs, particularly at a dosage of 10 mg/kg, could potentially exert a therapeutic effect on neuropathic pain by elevating the levels of GSH.
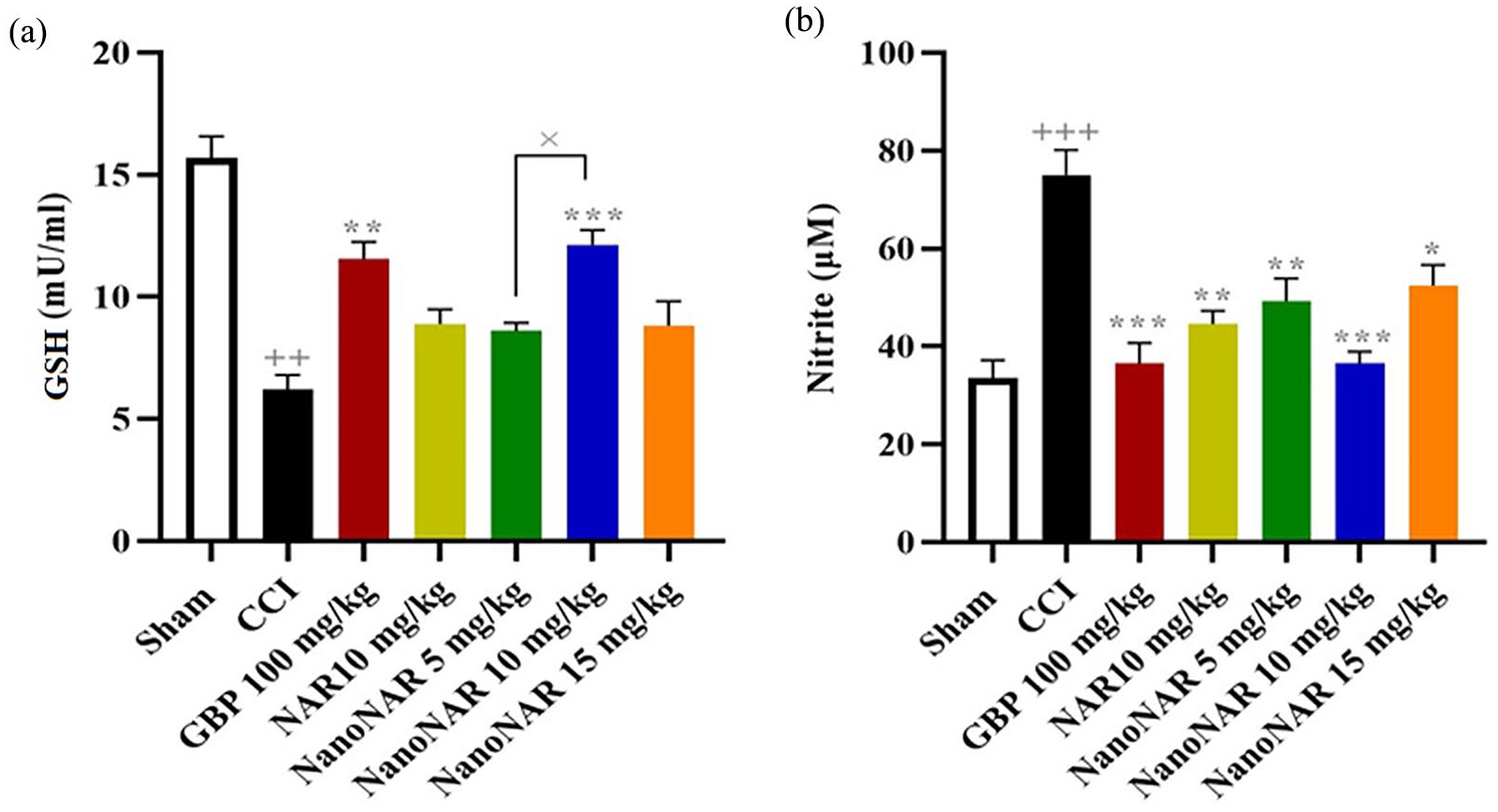
(a) The serum level of glutathione (GSH) and (b) Nitrite in rats treated with various formulations (+ indicates significant levels between CCI group and sham, and * indicates the other groups relative to CCI group) (n = 7).
Nitrite has been shown to play a complex role in neuropathy pain and is implicated in hyperalgesia effects as well as neuroinflammation through its interaction with oxidative stress.57,58 The analysis of nitrite levels in the serum revealed a significant increase in neuropathic rats with CCI-induced injury compared to the sham group (Figure 6(b)). Whereas, administration of GBP produced a steep reduction in the level of nitrite relative to the CCI group (p < 0.001). Besides, free naringin and naringin-loaded NPs at 5 mg/kg alleviated the level of nitrite, although this reduction was observed to be more significant for naringin-loaded NPs at 10 mg/kg compared with the CCI group (p < 0.001). These findings imply that naringin-loaded NPs at 10 mg/kg, similar to GBP, have potential therapeutic effects in reducing nitrite levels associated with neuropathy pain.
Histopathology studies
To examine changes in histological features, the cross-section of the sciatic nerve was subjected to H&E staining. Figure 7 presents the observed histological manifestations and the impact of the treatments. In comparison to the sham group, the CCI group exhibited disruptions in myelinated nerve fibers, swelling of axons, and the formation of nerve clefts. By contrast, GBP treatment could alleviate the abnormal architecture by reducing the degeneration of myelin sheath, axonal swelling and the disarrangement of myelinated nerve fibers. In addition, the staining of the sciatic nerve of rats treated with free naringin showed extended neuronal gaps, nerve fiber derangement and axonal abnormal swelling. While dose-dependent naringin-loaded NPs treatment ameliorated the CCI-induced histopathological alterations so that neural clefts diminished, axonal swelling was remarkably reduced, and the myelin sheaths regenerated in a dense arrangement. Notably, the most significant improvement in myelin sheath integrity and Schwann cell numbers was observed with a dosage of 10 mg/kg of naringin-loaded NPs.
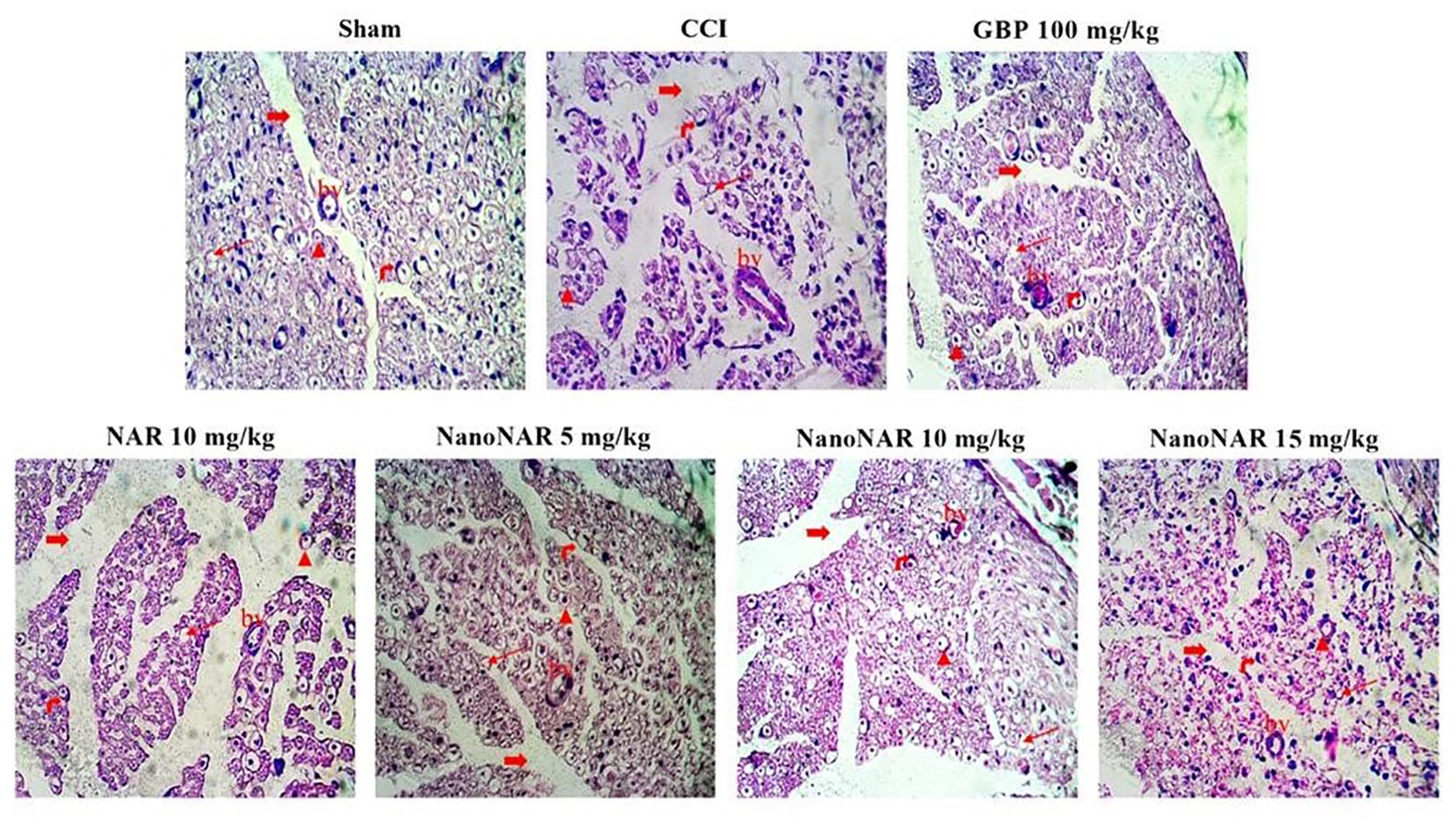
Histopathological results related to the studied groups (Thick red arrows: neural clefts (neuron gaps), thin red arrows: axonal abnormal swelling, right-side red arrows: Schwann cell nucleus, the red arrowhead: axon, BV: blood vessel).
These findings provide significant evidence that the naringin-loaded NPs possess neuroprotective effects. They demonstrate the ability of the NPs to restore damaged axons and myelin, suggesting their potential as a therapeutic intervention for the treatment of nerve injuries and disorders.
Conclusion
This study investigated the preparation and characterization of naringin-loaded chitosan NPs and their potential therapeutic effects on neuropathic pain. Preparation of the NPs specified the formation of NPs with proper size distribution with spherical morphology and uniform size. The relevant analyses exhibited high electrostatic stability and successful loading of naringin into the NPs. Also, in vitro release profile showed a prolonged release of naringin. In animal pain response examinations, administering naringin-loaded NPs at a dose of 10 mg/kg showed significant improvement in pain threshold compared to the CCI-induced group. The NPs also demonstrated the restoration of locomotor activity and anti-anxiolytic effects, as indicated by changes in the number of paw licking, rearings, and crossings. Inflammation markers studies revealed that the naringin-loaded NPs, increased GSH levels and reduced nitrite levels, indicating their potential therapeutic effects in reducing oxidative stress and neuroinflammation associated with neuropathic pain. Histopathology studies demonstrated improved histological manifestations of the sciatic nerve, including reducing myelin degeneration, axonal swelling, and nerve fiber derangement. The findings suggest that the naringin-loaded chitosan NPs possess neuroprotective and analgesic properties, making them a potential therapeutic intervention for neuropathic pain treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Kermanshah University of Medical Sciences, Grant No. 4000603.