Abstract
Mesenchymal stem cells (MSCs) possess self-renewal ability, multi-differentiation potential and low immunogenicity, thus serving as an ideal choice for cell therapies. Ex-vivo expansion systems that have been developed to meet clinical demands are faced with two crucial barriers, limited quantity and stemness loss of expanded cells. Hence, it is crucial and feasible to construct microcarriers that can show high and specific affinity to MSCs, and support highly efficient cell expansion with minimal stemness loss. In this study, EPLQLKM (E7) peptides were modified onto gelatin microcarriers by poly (ethylene glycol) (PEG) linkers, which showed great antifouling ability against xenogenic components. The rat bone marrow-derived mesenchymal stem cells (rBMSCs) harvested from the E7-modified gelatin microcarriers achieved better cell attachment, stemness maintenance, viability, and multilineage differentiation potentials, especially those with a higher E7 density. Attributing to the promotion for cell adhesion, E7 functionalization increased the expansion efficiency of rBMSCs with improved quantity and quality simultaneously, thereby providing a novel strategy for scalable expansion to optimize the clinical performance of MSCs.
Introduction
Mesenchymal stem cells (MSCs) hold great potential in regenerative medicine due to their unique characteristics, including multipotency, self-renewal capacity, and minor immunogenicity.1 –3 Considering the low recruitment efficiency of endogenous MSCs, more focus is given to exogenous MSCs in the research of tissue repair. At present, limited quantity and replicative senescence of isolated cells are considered two crucial barriers during scalable expansion ex vivo.4,5 As for replicative senescence, it means that MSCs may undergo changes in phenotypes and multipotency, that is, stemness loss. 6
In comparison with conventional two-dimensional culture systems, 7 microcarriers not only provide biomimetic structures and biological functions for cell behavior regulation, but also serve as transportable substrates for cell delivery,8,9 which have gradually attracted extensive attention as a promising alternative to meet practical demands. As cell-substrate interaction has a profound impact on cell behaviors, multiple studies have reported the effects of various material characteristics on cell expansion.10 –12 Such extracellular physiochemical and biological cues, including chemical composition,13,14 mechanical properties,15,16 morphological structure,17,18 and so forth, regulate the spreading area, 19 aspect ratio, 20 and geometry21,22 of MSCs, and thereby playing critical parts in cell proliferation and stemness preservation. MSCs with larger spreading areas exhibit better proliferation and osteogenesis differentiation, of which the cytoskeleton is relatively stretched, and the actomyosin contractility is relatively higher. 23
There are two critical attributes of microcarriers, modest negative buoyancy and surface properties, that have to be considered to facilitate expansion efficiency.24,25 Among all forms of microcarriers, including microsheets, 26 microrods, 27 microfibers, 28 and so on, microspheres exhibit distinctive characteristics. When dispersed in the same volume of liquid, microspheres provide the maximum specific surface area for cell adhesion and nutrient diffusion, which promotes cell growth and proliferation. Additionally, microspheres can be readily suspended in culture media, avoiding sedimentation and enabling efficient handling, as well as easily prepared for large-scale culture and compatible with bioreactor systems, making them ideal for applications in tissue engineering and regenerative medicine.
According to previously studied microsphere products for cell culture, 29 various materials have been utilized as substrates of them, among which synthetic materials such as polyethylene, 30 polystyrene, 31 poly(lactic-co-glycolic acid), 32 as well as natural materials like agarose, 33 alginate, 34 and gelatin/collagen 35 are widely used. While these materials exhibit excellent mechanical properties, they may also lead to potential incompatibilities with tissues or cells. In this context, the incorporation of functional coatings, such as positively charged molecules36 –38 and Arg-Gly-Asp (RGD) peptides,39,40 is advantageous with emphasis on improvement of cell adhesion. Unfortunately, when it comes to MSCs, such biomolecules lack cell specificity, and even run a risk of inducing differentiation.41,42 To overcome these limitations, a peptide sequence EPLQLKM (E7) 43 with high and specific affinity to bone marrow-derived mesenchymal stem cells (BMSCs) has been discovered and used. Previous studies on the E7 peptide have highlighted its ability to selectively attract and adhere MSCs, which can further achieve less stemness loss.44 –47
Additionally, it is reported that xenogenic components, for instance, fetal bovine serum (FBS), not only introduce variability to cell culture media, 48 but also induce immunological response 49 and lineage variation.50,51 In recent years, numerous antifouling coatings have been developed, among which poly(ethylene glycol) (PEG)52,53 and its derivatives 54 are known for excellent hydrophilicity and biocompatibility, as well as their strong but flexible chains to increase the bioactivity of immobilized biomolecules. 55
Hence, it is crucial and feasible to construct microcarriers with regulated affinity molecules, which can show high and specific affinity to MSCs, and support highly efficient cell expansion with minimal stemness loss. In this study, microcarriers were manufactured from gelatin due to its remarkable biocompatibility. Subsequently, E7 peptides with different densities were modified onto the surface of PEGylated microcarriers by a click reaction between maleimide and sulfhydryl group. Since rat MSCs have similar characteristics to human MSCs in terms of morphology, phenotype, and differentiation potential, rat bone marrow-derived mesenchymal stem cells (rBMSCs) were seeded on the modified microcarriers to explore the effect of E7 on cell expansion efficiency. The relationship between E7 densities and the quantity and quality of expanded rBMSCs was further investigated.
Experimental section
Materials
Gelatin was purchased from Biofroxx GmbH (Germany). NHS-PEG-maleimide (NHS-PEG-MAL, Mw: 5000, purity >95%) was purchased from Ponsure Biotech, Inc. (Shanghai, China). E7 peptides (EPLQLKM, purity >98%) were customized by GL Biochem Ltd. (Shanghai, China).
Fabrication of E7-modified gelatin microcarriers
Prior to E7 functionalization, pure gelatin microcarriers were fabricated with emulsion method as previously described, 56 and sieved into a range of 300–500 μm. The filtered gelatin microcarriers were following immersed in a PBS (pH 7.4) solution of NHS-PEG-MAL (3 mM) at 37°C for 80 min, and then sequentially rinsed by absolute DMSO, ethanol, and PBS. After PEGylation, microcarriers were dispersed in reaction solutions at 37°C for 6 h with E7 concentrations of 0.75, 1.5, and 3 mM, respectively. Accordingly, E7-modified gelatin microcarriers were named E25, E50, and E100. Pure gelatin (N) and only PEGylated (P) microcarriers were used as controls.
Characterizations of E7-modified gelatin microcarriers
The E7 peptide-affinity assay was performed by flow cytometry (BD FACSCalibur with CellQuest software, BD Bioscience, USA). Briefly, rBMSCs, RAW264.7, and NIH3T3 cells were incubated with 1 nM FITC-labeled E7 for 1 h to allow cell binding and internalization, respectively. The cells were washed thrice with PBS, and then resuspended and fixed in 1% BSA before measurement. Cells incubated with 1% BSA for 1 h were used as the negative control.
To determine the sizes of E7-modified gelatin microcarriers, microcarriers were first dispersed in 37°C PBS overnight and then observed with a microscope (IX81, Olympus, Japan). The diameters of at least 200 microcarriers were measured with the Image Pro Plus software (Media Cybernetics).
The surface microstructures of E7-modified gelatin microcarriers were revealed by scanning electron microscopy (SEM, S-4800, Hitachi, Japan). The dispersion of E7-modified gelatin microcarriers in the culture medium was observed with a fluorescence microscope by the bright-field mode (BZ-X800, KEYENCE, Japan). The element compositions of N, S, and C were verified by SEM equipped with energy-dispersive X-ray spectroscopy (EDS).
Ninhydrin was utilized to test the quantity of grafted E7 peptides, on the base of the reaction with amino groups. E7-modified microcarriers were immersed in an ethanol solution of ninhydrin (0.1 M) at 80°C for 15 min, before determination by a microplate reader (infinite M200 Pro, ITecan, Switzerland) at 570 nm.
Cell culture
Animal experiments were performed according to the “Guidelines for Animal Experimentation” of the Institutional Animal Care and Use Committee, Zhejiang University. rBMSCs were harvested from Sprague-Dawley rats (6–8 weeks, male) and cultured in alpha-minimum essential medium (α-MEM, Gibco, USA) as the previous protocol. 57 All experiments were performed on cells at passage 2. The medium was supplemented with 10% v/v fetal bovine serum (FBS, Gibco, USA). Cells were incubated in a humid incubator maintained at 37°C in 5% CO2.
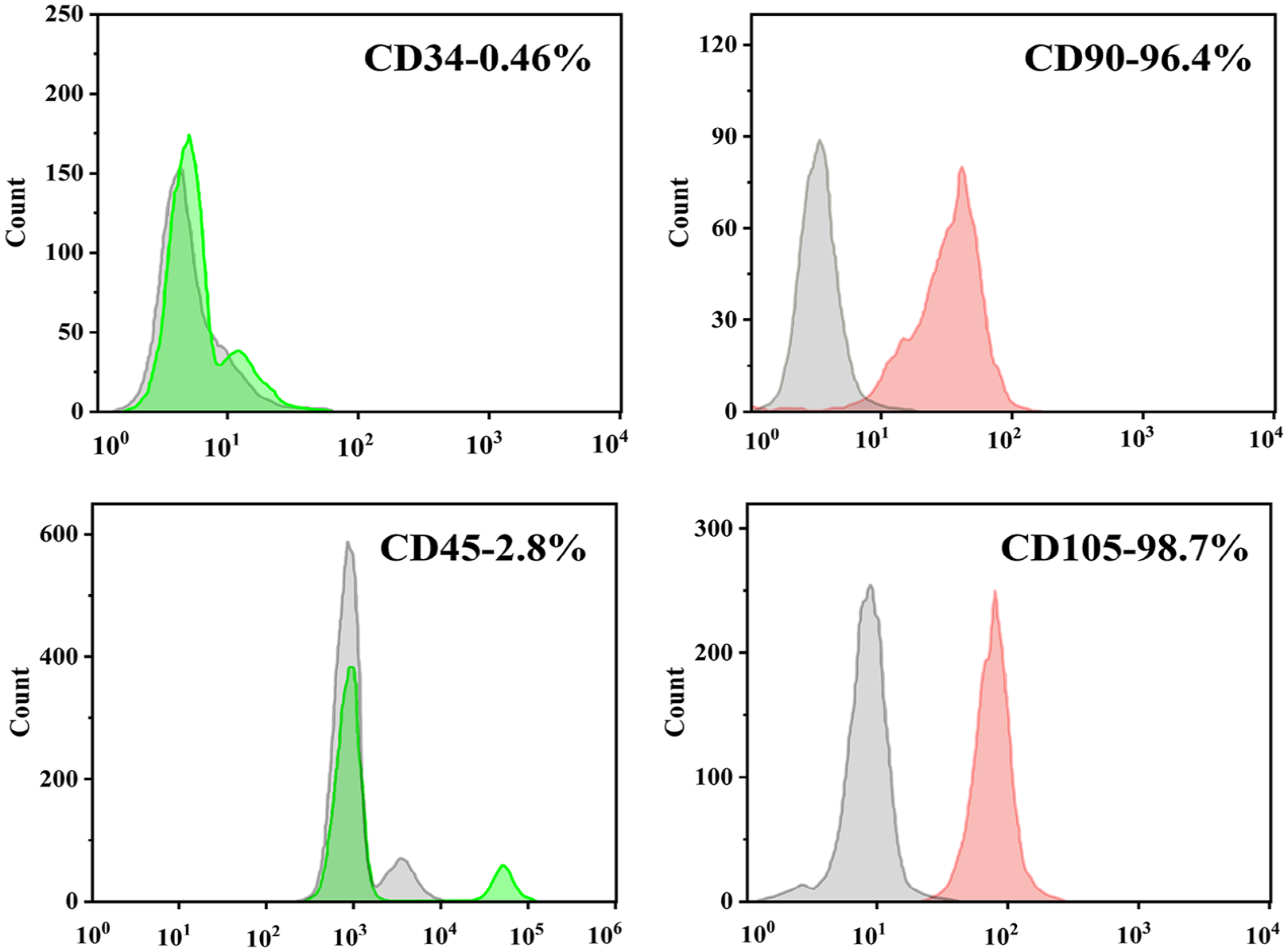
The specific surface antigen markers of rBMSCs at passage 2 were examined via flow cytometry (Figure 2). The antibodies for negative markers included CD34 and CD45, whereas the positive markers included CD90 and CD105. Cells (100, 000) were incubated with 1 µg of FITC-conjugated rat monoclonal CD34 antibody (Novus, USA), FITC-conjugated mouse anti-rat CD45 (BD Pharmingen, USA), PE-conjugated mouse anti-rat CD90 antibody (BD Pharmingen, USA), and PE-conjugated rat monoclonal endoglin/CD105 antibody (Novus, USA) for 1 h at 4°C, respectively. After three washes with PBS containing 1% BSA, the stained cells were subject to flow cytometry analysis.
RAW264.7 and NIH3T3 cells were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China) and cultured under standard conditions.
Cell attachment and proliferation assay
Prior to cell culture, E7-modified gelatin microcarriers were sterilized by being immersed in 75 vol% ethanol overnight. Subsequently, all samples were washed at least five times with sterile PBS and placed in 24-well plates. rBMSCs were seeded onto microcarriers at a density of 10,000 cells/mL, with each well containing 1 mL culture medium and 10 mg microcarriers. After being cultured for 4, 8, and 12 h, respectively, cells were stained with calcein-AM (Yeasen, China) according to the manufacturer’s instructions and imaged under a fluorescence microscope (IX81, Olympus, Japan). The numbers of attached cells were measured using Image Pro Plus software. After 12 h, the microcarriers with cells attached were transferred to new 24-well plates and futher cultured for 1, 3, 5, and 7 days. Cell proliferation was defined as the cell count on day 1, 3, 5, and 7, respectively. On day 7, the cell loading capacity of microcarriers was observed with a fluorescence microscope by the bright-field mode (BZ-X800, KEYENCE, Japan).
The cell proliferation and viability was also characterized by CCK-8 assay. After being cultured for 1, 3, 5, and 7 days, the microcarriers with cells attached were transferred to new 24-well plates. CCK-8 solution was added to each well at a volume of 10% of the culture medium and incubated for 1.5 h at 37°C in a humidified atmosphere with 5% CO2. The absorbance of each well was measured at 450 nm using a microplate reader (MultiskanFC, ThermoScientific, USA). The OD value was considered to be proportional to the number of viable cells.
Cell morphology
Microcarriers cultured with rBMSCs for 5 days were fixed for 15 min with 4% paraformaldehyde solution and then dehydrated in ethanol solution with a series of concentrations of 30%, 50%, 70%, 80%, 90%, 95%, and 100 vol%, each for 10 min, respectively. They were subsequently dried for observation under scanning electron microscope (SEM, S-4800, Hitachi, Japan).
Moreover, microcarriers loaded with rBMSCs were fixed with 4% paraformaldehyde solution for 15 min at room temperature, rinsed with PBS for three times, permeabilized with 0.5% triton X-100 solution for 5 min, and then blocked with 1% bovine serum albumin (BSA) for 80 min. To visualize F-actin stress fibers, cells were stained with rhodamine-labeled phalloidin (Thermo Scientific, USA) overnight, and counter-stained for nuclei with 4,6-dithiol-2-phenylindole (DAPI, Sigma-Aldrich, USA) for 15 min. Residual reagents in between were removed with PBS to reduce the background. The dilution of all reagents was according to the manufacturer’s protocol. Eventually, the stained cells on microcarriers were examined under confocal laser scanning microscopy (CLSM, LSM-880, Zeiss, Germany).
Immunofluorescence staining
The expression of stemness markers (Stro-1 and CD166) of rBMSCs on microcarriers at passage 1, 2, and 3 was investigated by immunofluorescence staining. Harvested cells, after being filtered through a 70 μm sieve (BD Biosciences, USA) to remove microcarriers, were reseeded to new 24-well plates for 24 h at a density of 2 × 103 cells per well. Briefly, samples were fixed and blocked as mentioned before. Thereafter, they were incubated with primary antibodies mouse anti-Stro-1 (Invitrogen, USA) and goat anti-CD166 (R&D Systems, USA) at 4°C overnight followed by further incubation in the specified secondary antibodies FITC-conjugated rabbit anti-mouse IgM antibody (Thermo Scientific, USA) and Cy3-conjugated rabbit anti-goat IgG antibody (Jackson ImmunoResearch, USA) for 1 h. Following that, samples were counter-stained for nuclei with DAPI. The working concentrations of all reagents were in accordance with the manufacturer’s protocols. Imaging was performed with a fluorescence microscope (IX81, Olympus, Japan), and images were processed with Image Pro Plus software (Media Cybernetics).
Cell viability
After 7 and 14 days expansion, rBMSCs on E7-modified gelatin microcarriers were harvested, and stained with Annexin V-FITC apoptosis detection kit (Yeason, China) according to manufacturer’s protocols. Eventually, it was visualized using a flow cytometry (BD FACSCalibur with CellQuest software, BD Bioscience, USA) to distinguish viable cells (PI negative, Annexin V-FITC negative) from early apoptotic cells (PI negative, Annexin V-FITC positive) and dead cells (PI positive, Annexin V-FITC positive).
Differentiation potentials toward osteogenic, chondrogenic, and adipogenic lineages
To evaluate multilineage differentiation potentials after 7 days expansion on E7-modified microcarriers, rBMSCs were harvested and reseeded to new plates, and permitted to grow to confluence for 2 days in basal medium, which was followed by differentiation toward osteoblasts, chondrocytes, and adipocytes based on established protocols. For differentiation assay, SD rat mesenchymal stem cell osteogenic differentiation medium kit (ScienCell, USA), chondrogenic differentiation medium (ScienCell, USA), and adipogenic differentiation medium kit (ScienCell, USA) were prepared. Media were changed every 3 days, and analysis was performed after 14 days of differentiation by Alizarin Red, Oil Red O, and Alcian Blue staining, respectively. Imaging was performed with a fluorescence microscope (IX81, Olympus, Japan).
Statistical analysis
Except where noted, experiments were carried with three replicates per trial. All data were presented as mean ± standard deviation (SD) in this study and analyzed by one-way ANOVA and post hoc Tukey’s test using p values to determine statistically significance.
Results and discussion
Characterizations of rBMSCs and E7-modified gelatin microcarriers
The characteristics of rBMSCs were examined by flow cytometry analysis. As shown in Figure 1, cells at passage 2 showed homogeneous phenotypes that were positive for CD90 (96.4%) and CD105 (98.7%). On the contrary, the cells were negative for other markers of the hematopoietic lineage, including the lipopolysaccharide receptor CD34 (0.34%) and the leukocyte common antigen CD45 (2.8%). These results indicate that the rBMSCs had a high purity and met the criteria for the further cell culture experiments.
Flow cytometry analysis of rBMSCs markers.
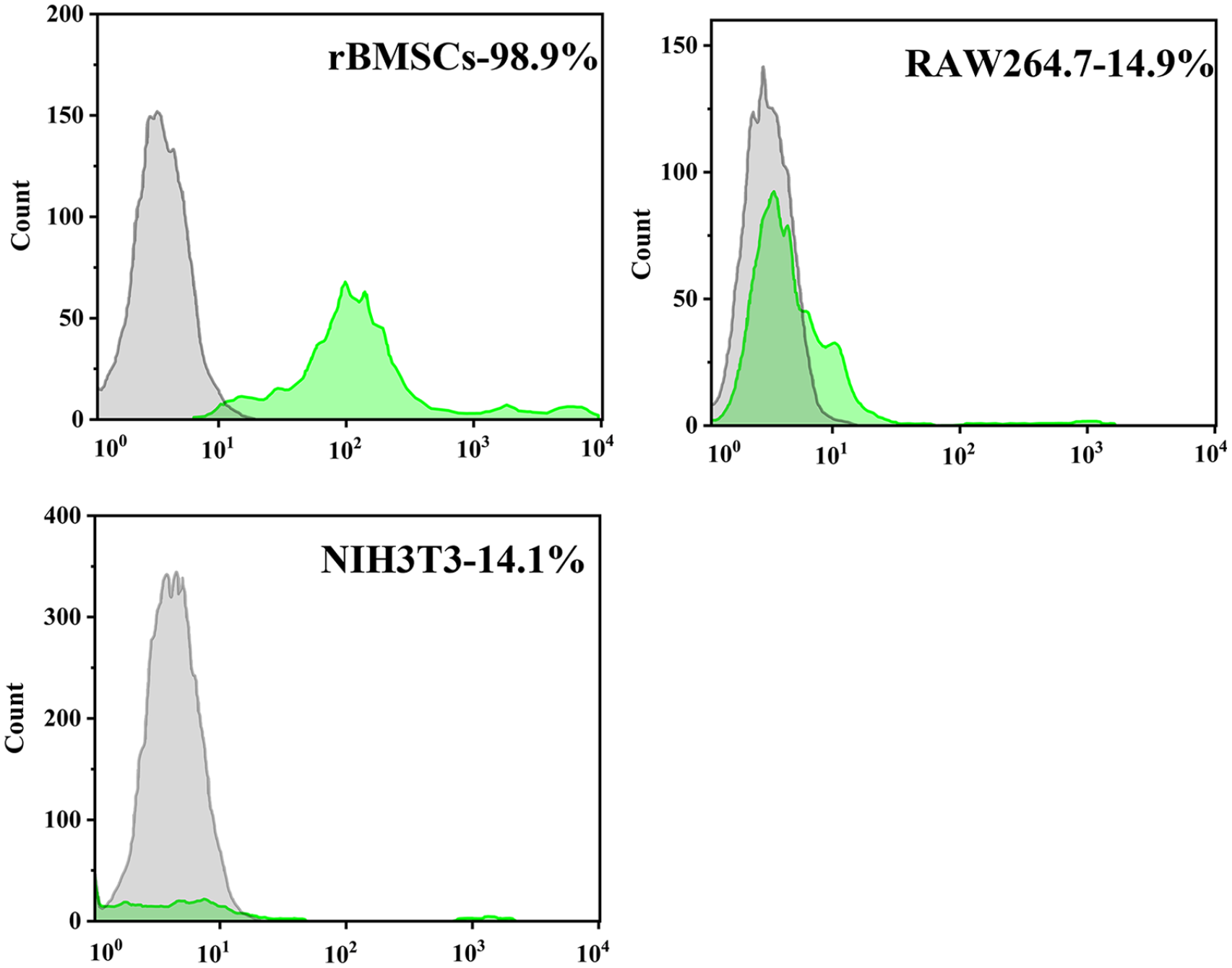
To determine the ability of customized E7 peptides to specifically adhere to rBMSCs, flow cytometry analysis was performed. As shown in Figure 2, the binding ratio of FITC-E7 peptides to rBMSCs was up to 98.8%, while the binding ratios to RAW264.7 and NIH3T3 cells were 14.9% and 14.1%, respectively.
Flow cytometry analysis of E7 peptide-affinity assay.
As shown in Figure 3(a), the diameters of E7-modified gelatin microcarriers at equilibrium swelling state mainly ranged from 150 to 550 μm, with an average of 380 ± 30 μm. The microcarriers showed round and smooth structures subjected to SEM (Figure 3(b)) and homogeneous dispersion in the culture medium under light field microscopy (Figure 3(c)). Owing to their structure and suitable hydrophilicity, the microcarriers were not prone to sedimentation and aggregation, and were easy to transfer, making them promising candidates for stem cell expansion. However, gelatin microspheres were also prone to deformation under the influence of pH, which might lead to the loss of cell support function, thus limiting their application in varying pH environments.
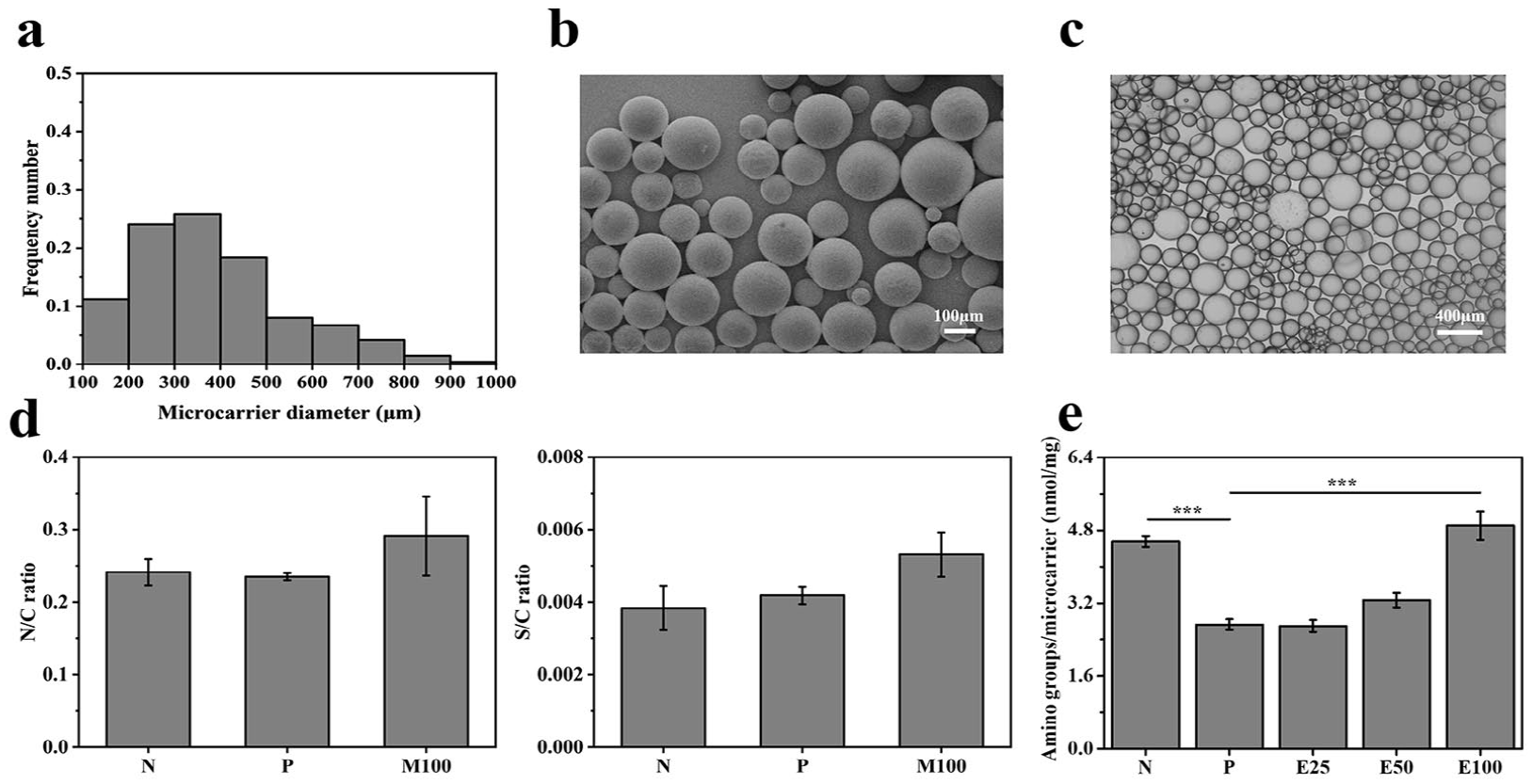
Characterizations of E7-modified gelatin microcarriers. (a) Size distribution at equilibrium swelling state, (b) SEM images, (c) bright field microscopy images, (d) EDS results, and (e) densities of amino group. Data are shown as mean ± SD (n = 3). ***p < 0.001.
The surface compositions of different microcarriers were characterized by EDS (Figure 3(d)). Nitrogen to carbon (N/C) ratios increased from 0.2 (N) to 0.3 (E100), while sulfur to carbon (S/C) ratios rose from 0.003 (N) to 0.005 (E100). As for E7-modified gelatin microcarriers, the raised N/C and S/C ratios could be attributed to the comparatively higher N/C and S/C ratios of E7 peptides than those of gelatin, which demonstrated the successful grafting of E7. The grafted E7 were further tested quantitatively on the foundation of ninhydrin reaction (Figure 3(e)). It was shown that the amino densities raised from 2.7 (E25) to 4.9 (E100) nmol/mg, along with the increased E7 concentrations. Thus, the grafting densities of E7 could be modulated by changing reactant concentration.
Cell attachment and proliferation behaviors
In order to analyze the impacts of E7 densities on cell attachment and proliferation behaviors, rBMSCs were seeded onto E25, E50, E100, respectively. At the early stage of expansion, we focused on cell attachment efficiency. The numbers of rBMSCs attached to microcarriers per well kept rising until 8 h after seeding in Figure 4(a). When it came to 12 h, cells had settled down. Moreover, microcarriers with various surface modifications supported different cell attachment efficiency. Owing to the antifouling PEG layer, the number of cells on P (0.7 × 104) decreased compared to those on N (1.0 × 104) 4 h after seeding. The cell number on E25 reached nearly 1.8 × 104 after seeding for 12 h, which was less than those of N (2.1 × 104) and E100 (2.1 × 104). Cell expansion behaviors were further evaluated following cell attachment. As shown in Figure 4(b), rBMSCs grown for 7 days on E100 (6.2 × 104) exhibited a higher cell number than cells on N (5.5 × 104), which was similar to the situation of 1 days.
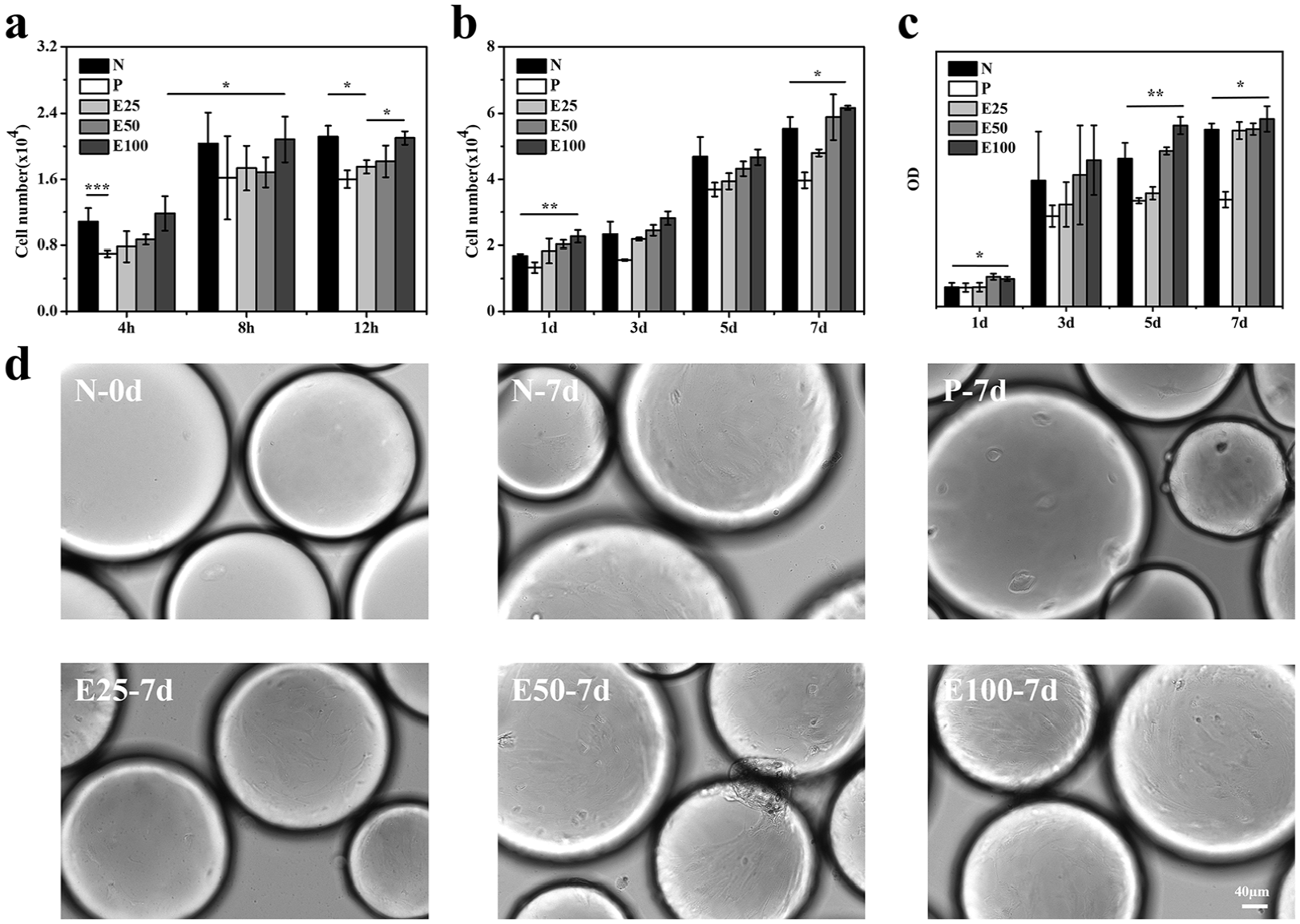
Effects of E7-modified microcarriers on rat bone marrow-derived mesenchymal stem cells (rBMSCs) attachment and proliferation behaviors of rBMSCs. (a) Quantification of cell attachment at 4, 8, and 12 h; (b) cell proliferation on different microcarriers for 1, 3, 5, and 7 days, respectively. (c) CCK-8 assay results of cell proliferation on different microcarriers for 1, 3, 5, and 7 days, respectively. (d) Representative bright field microscopy images. (All images were taken at the same scale.) Data are shown as mean ± SD (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001.
CCK-8 assay was conducted to evaluate the proliferation behavior of rBMSCs expanded on different microcarriers in situ. The results (Figure 4(c)) showed that the OD values of rBMSCs on E100 were higher than those on N at all time points, indicating that E7 peptide-grafted gelatin microspheres could support more cells on their surfaces, thus enhancing the cell proliferation rate. It could also be observed from Figure 4(d) that the number of cells expanded on microcarriers (N) significantly increased from 0 to 7 days and E100 achieved the highest cell density on 7 days.
Together, the results revealed that the E7 peptide grafting enhanced the cell affinity and interaction with the microspheres, resulting in higher cell proliferation rate than the unmodified (N) and PEGylated (P) microspheres. Since stem cells are typical adherent cells, E100, which had the highest E7 grafting density and could provide the most adhesion sites, exhibited the best performance in rBMSCs expansion.
Cell morphology
The cell morphology of rBMSCs after 5 days of expansion was visualized under SEM. It was observed from Figure 5(a) that the filopodia of rBMSCs grown on P reduced in comparison with those on N, such contractive morphology suggesting the efficient PEG blocking in preventing nonspecific cellular adhesion. Otherwise, the branched extremities of cell filopodia on E7-modified microcarriers may be attributed to E7, which acted as a specific ligand for the integrin on cell membrane. In addition, the spreading of rBMSCs was promoted on microcarriers with increased E7 densities, especially on E100, demonstrating that E7 enhanced the adhesion between rBMSCs and microcarriers.

Cell morphology after being cultured for 5 days on different microcarriers. (a) Representative SEM images. (All images were taken at the same scale.) (b) Representative CLSM images show rBMSCs cultured on E100. Cells were stained for F-actin (red) and nucleus (blue).
To further observe the promotion effects of E7, a typical CLSM image of rBMSCs on E100 was shown in Figure 5(b). With the help of DAPI (nuclei, blue) and rhodamine-labeled phalloidin (F-actin skeletons, red), most of the fully spread cells clearly represented spindle shapes with well oriented and aligned cytoskeletons.
Previous studies have shown that when rBMSCs were cultured in vitro, cells in good condition showed a spindle shape with clear intercellular demarcation, and when the cells proliferated to the point of cell fusion, the overall arrangement of the cells was slightly spiral or radial. As can be seen in Figure 4(d), the cells on E100-7 days presented a spindle-shaped and spiral arrangement, which again confirmed the important effect of high-density E7 peptide grafting on the regulation of cell morphology.
Stemness of harvested rBMSCs
The effects of E7 on the stemness of harvested rBMSCs after expansion were evaluated by immunofluorescence staining subsequently (Figure 6). When compared to N, E7-modified microcarriers significantly promoted the expression of stemness markers, Stro-1 and CD166. Particularly, cells from E100 yielded pronounced expression of those two markers.
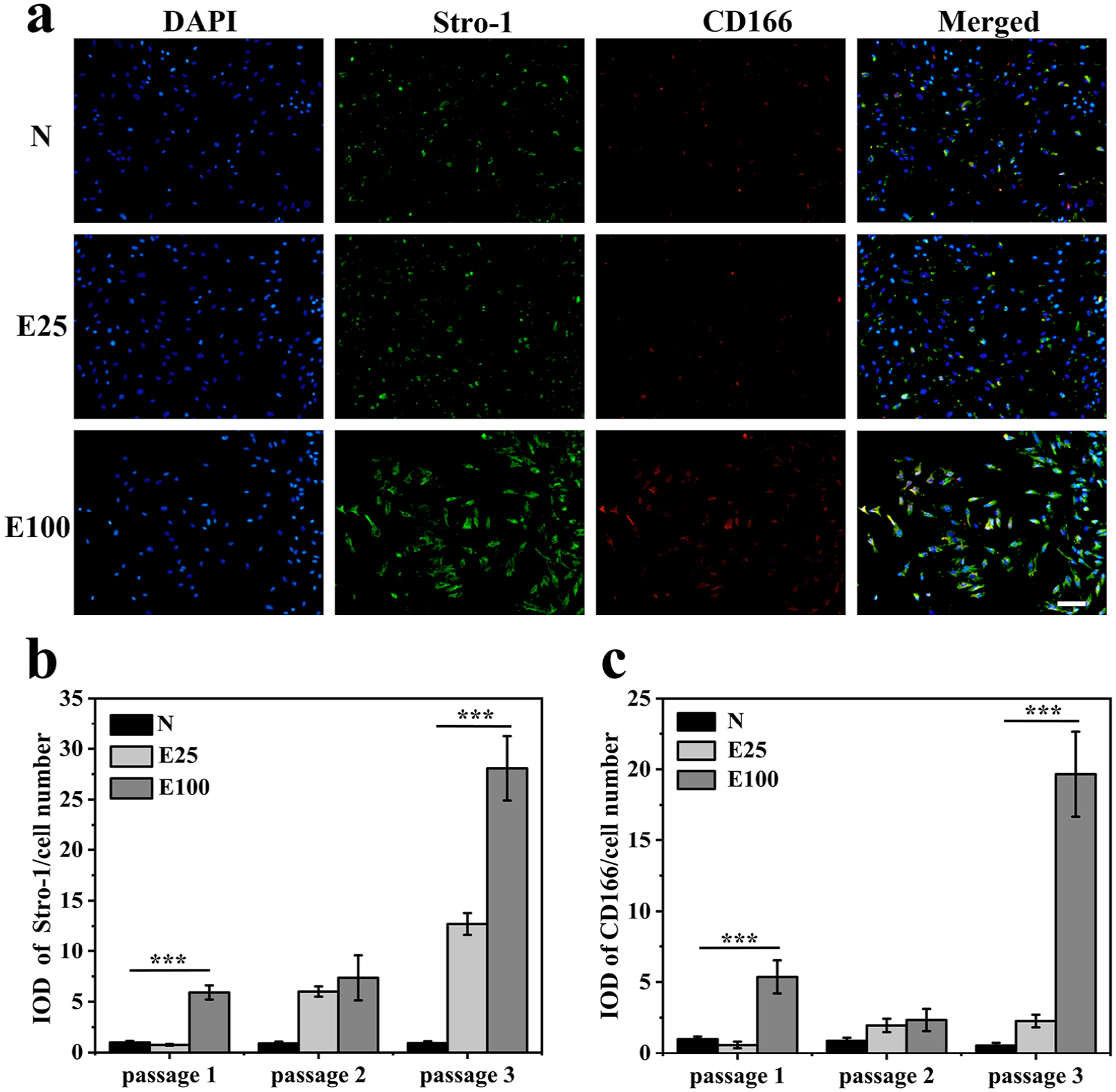
Effects of E7-modified microcarriers on rBMSCs stemness maintenance. (a) Immunofluorescence staining of stemness-specific markers (Stro-1, CD166) of cells at passage 1. The merged images show counterstaining for Stro-1 (green), CD166 (red) and nucleus (blue). Scale bars = 100 µm. (All images were taken at the same scale.) (b) Stro-1 intensities and (c) CD166 intensities of cells at passage 1, 2, and 3 normalized to those of cells on N at passage 1. Data are shown as mean ± SD (n = 3). ***p < 0.001.
Furthermore, the results of cells at passage 1, 2, and 3 were analyzed quantitatively by the corresponding intensities of Stro-1 and CD166 expression, normalized to those of cells on N at passage 1. For regions of interest, threshold levels were set to select positively stained areas and calculate the integrated optical density (IOD). Each total integrated density was then normalized to cell number. It was revealed from the analysis that with successive passages, the expression of stemness markers of cells on N gradually decreased, while that of cells on E100 increased significantly. The normalized Stro-1 intensity of cells expaned on E100 at passage 1 was about five folds higher than that of N, while CD166 intensity was nearly four folds higher. After three successive passages, the normalized Stro-1 and CD166 intensity of cells on E100 was even up to nearly 30 and 35 times that of cells on N respectively.
This finding consistently demonstrated the efficacy of E7 to lessen stemness loss during ex vivo expansion of rBMSCs. Considering the results of stemness (Figure 6) and cell morphology (Figure 7) together, cells on E100 showed better spreading morphologies, which indicated relatively stretched cytoskeletons and relatively higher actin contractility, thereby in favor of higher stemness than those of N. It showed consistence with previous reports focusing on the correlation between cell spreading areas, cytoskeleton tension, and stemness. 58 Thus, it is an effective strategy to modulate the E7 density on microcarriers to maintain stemness of rBMSCs.
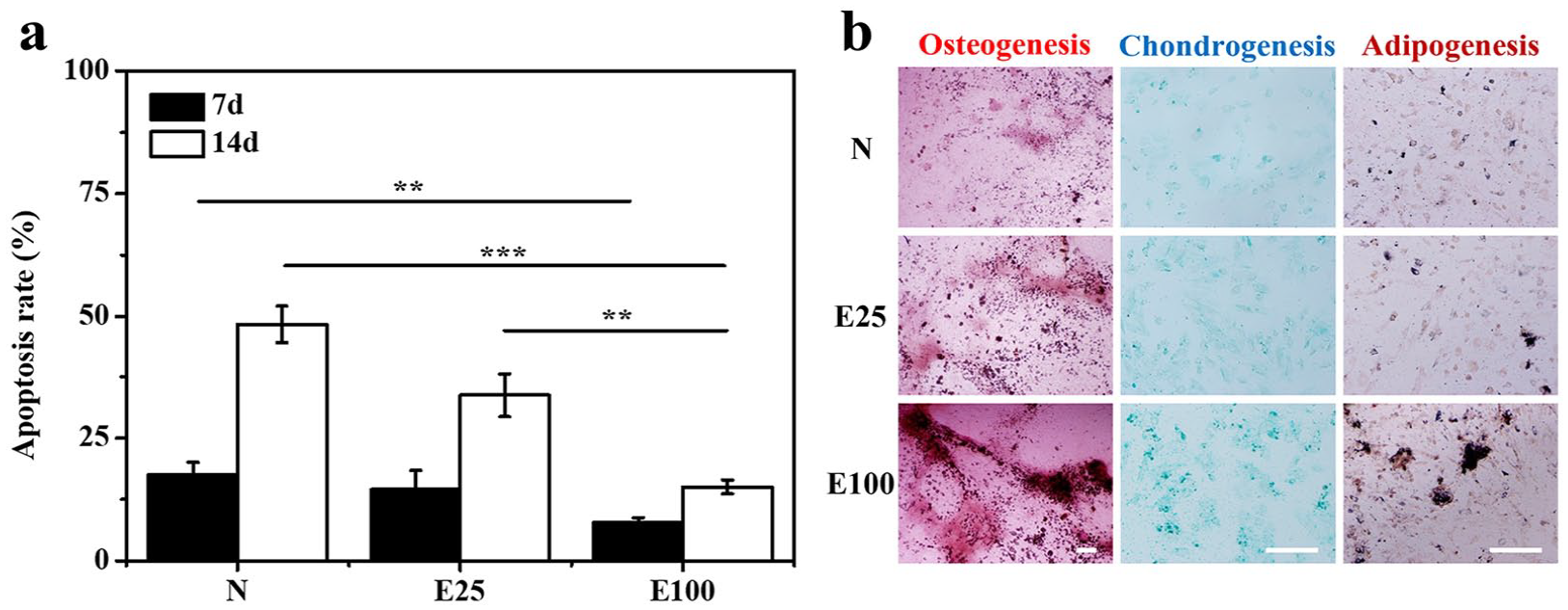
Cell viability and differentiation potentials. (a) Cell viability of rBMSCs harvested from N, E25, and E100 was determined by apoptosis ratios. (b) Cell differentiation was induced for 14 days and assessed by staining for osteogenesis (Alizarin Red), adipogenesis (Oil Red-O), and chondrogenesis (Alcian blue). Scale bars = 200 µm. Data are shown as mean ± SD (n = 3). **p < 0.01, ***p < 0.001.
Cell viability and differentiation potentials
Cell viability was assessed based on the ratios of apoptosis cells (Figure 7(a)). After culturing for 14 days, the apoptosis ratios of rBMSCs on N, E25, and E100 were 48.4%, 33.7%, and 15.0%, respectively. Compared with the control, apoptosis ratios of cells seeded on E100 were distinctly lower at all time points, and even E25 had less apoptosis cells than N. Hence, E7 might stimulate the viability of rBMSCs.
As for differentiation potentials, rBMSCs harvested after 7 days were investigated by inducing differentiation toward osteogenic, chondrogenic, and adipogenic lineages (Figure 7(b)). Mineral deposition was confirmed as a late-stage marker of osteogenesis and major calcium deposition could be detected by Alizarin Red staining. 9 In particular, obvious calcium deposits were observed on E100. Chondrogenic differentiation was detected by Alcian Blue staining and observed mainly on E100. With regard to adipogenic differentiation, Oil red O staining showed the presence of lipid vacuoles. The number of lipid vacuoles in cells on E100 exhibited a sharp increase, thus confirming the robust adipogenic differentiation. These results taken together suggested that E7-modified microcarriers could enhance multilineage differentiation potentials, while E100 was optimal with the highest E7 density.
Conclusion
The gelatin microcarriers modified with defined densities of E7 peptides served as cell culture substrates to expand rBMSCs ex vivo. Compared to pure gelatin microcarriers, cells cultured on E7-modified gelatin microcarriers achieved effective expansion, with improved quantity and quality simultaneously. The harvested cells showed better cell attachment, stemness maintenance, viability, and multilineage differentiation potentials. In this work, additional E7 improved attachment functionality of rBMSCs, which supported cell stemness after expansion, thereby suggesting that E7-modified gelatin microcarriers are a relevant platform for ex-vivo expansion to enhance the performance of clinical therapeutic applications involving MSCs.
Footnotes
Acknowledgements
This work was carried out in the Key Laboratory of Macromolecular Synthesis and Functionalization, Department of Polymer Science and Engineering, Zhejiang University
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research Program of China [2017YFA0104900].
