Abstract
This study measures the incidence and prevalence of idiopathic intracranial hypertension (IIH) in children in Wales and assesses risk factors and health care utilisation. We identified 177 children (1-15 years) with IIH diagnosed between 2003 and 2017. Incidence increased from 1.6/100 000 per year (2003) to 3.5/100 000 per year (2017). Sixty-three percent of the cases were girls, and 87% were diagnosed between 7 and 15 years. Children in the most deprived quintile were more likely to develop IIH compared to children in the least deprived quintile (odds ratio [OR] = 1.7 [95%CI = 1.1-2.9]). Obese and overweight children were more likely to develop IIH compared with non-overweight children (ORs = 21 [95%CI = 9-51] and 5.1 [95%CI = 1.9-14]). Children with IIH had higher hospital admission rates than children without IIH (rate ratio = 3.9; 95% CI 3.6-4.3). The increasing incidence, association with BMI and deprivation, and increased health care utilisation in children with IIH are relevant findings to health care professionals and policy makers.
Idiopathic intracranial hypertension (IIH), also known as pseudotumor cerebri or primary intracranial hypertension, is a condition characterised by raised intracranial pressure of unknown cause.1–3 People with IIH are at risk of headaches, visual disturbance, and visual loss.4–6 Other symptoms can include back and neck pain, nausea and vomiting, diplopia, tinnitus, and dizziness.2,4 IIH is diagnosed in the presence of papilloedema, a raised cerebrospinal fluid (CSF) opening pressure (generally greater than 28cmCSF when measured in the left lateral position), normal CSF constituents, normal brain imaging, and the exclusion of potential secondary causes of raised intracranial pressure such as venous sinus thrombosis, thyroid disease, certain medications, stroke, and traumatic brain injury.2,3,7–9 Rarely, IIH can be diagnosed without papilloedema if these criteria are present with a unilateral or bilateral abducens nerve palsy. 7
IIH primarily affects obese women of childbearing age, but can affect children. 1 In children, IIH can be divided into pre-pubertal- and pubertal-onset groups. 2 IIH in pubertal children mainly affects obese girls; however, in pre-pubertal children with IIH there is a higher proportion of males.2,10–13
A large prospective study in the United Kingdom identified 185 cases of IIH in children between 2007 and 2009 with an incidence of 0.7/100 000 per year. 13 The incidence of IIH in children found in other studies include 0.5/100 000 per year, 0.6/100 000 per year, and 0.6/100 000 per year in regions of Germany, USA, and Canada, respectively.10,14,15 There have been several recent large epidemiologic studies of IIH in adults.5,6 They have shown that in adults, the incidence of IIH is increasing significantly in the United Kingdom; IIH is associated with socio-economic deprivation (even when adjusted for BMI); and health care utilisation is higher in individuals with IIH.5,6 It is not known whether the same is true for children.
We identified children with IIH in Wales over a 15-year period (2003 to 2017). We measured trends in the incidence and prevalence of IIH, measured health care outcomes, and assessed risk factors for developing IIH.
Method
We used routinely collected Welsh health care data within the Secure Anonymised Information Linkage (SAIL) databank.16–18 SAIL has data from a range of sources including primary care data (general practitioners [GPs]) for 80% of the Welsh population and secondary care data for the whole Welsh Population (total population 3.1 million, SAIL population 0-15 years old = 400 000).
We identified children with a diagnosis of IIH in primary and/or secondary health care data using the Read code F282 (Benign intracranial hypertension; primary care) and International Classification of Diseases, Tenth Revision (ICD-10) code G932. (Benign intracranial hypertension; secondary care). We excluded children with possible secondary causes of raised intracranial pressure such as hydrocephalus (including post-traumatic hydrocephalus), venous sinus thromboses, stroke, tetracycline antibiotic treatment, Chiari malformations, and neoplasms (Supplementary Tables e-1 and e-2).8,9,11,19 We also excluded the small number of children who died within 30 days of diagnosis given the possibility of a secondary cause of raised intracranial pressure in this group. Children with no socio-economic deprivation or primary care data, or who were not registered with a GP in SAIL 1 year before their diagnosis were also excluded.
The study period was January 1, 2003, to December 12, 2017. We defined children as being less than 16 years old given that children transition to adult services at the age of 16 years in the United Kingdom. Only children diagnosed with IIH within the study period were included in incidence calculations, but children diagnosed with IIH before January 1, 2003, were included in prevalence calculations for the time that they remained less than 16 years old within the study period.
Diagnosis age was defined as the age at first diagnosis of IIH. When a child with IIH turned 16 years they were no longer counted in the prevalent IIH cohort for that year onward. Shunts and visual outcomes were still reported after the age of 16 years, whereas unscheduled hospital admission rates and prescriptions were only reported for children.
To validate the accuracy of our case ascertainment algorithm, we used a registry of confirmed paediatric IIH cases from a paediatric neuroscience centre (Morriston Hospital, Swansea). We anonymously linked 40 confirmed cases of paediatric IIH into the SAIL databank using the established split-file procedure.16,17 These cases had been diagnosed by a paediatric neurologist. We used these cases to calculate the sensitivity of our case ascertainment algorithm.
Incidence and Prevalence
We calculated the incidence and prevalence of IIH for each year in the study. We calculated prevalence by dividing the number of children with IIH on 30th June of each year, by the mid-year population for that year. We calculated incidence by dividing the number of children with a new IIH diagnosis (no previous IIH diagnosis) in that year by the mid-year population of children for that year. The mid-year population was defined as the number of children on 30th June registered with a GP contributing data to SAIL for at least 1 year.
Demographics
We recorded socio-economic deprivation using the Welsh Index of Multiple Deprivation (WIMD). 20 WIMD scores are calculated from different domains for small geographical areas with populations of around 1600 people. These areas are then ranked and divided into quintiles, quintile 1 being most deprived and quintile 5 being the least deprived areas.
Body mass index (BMI), defined as body mass in kilograms divided by the square of height in metres, was calculated from routinely recorded height and weight measurements in primary care taken within 1 year of diagnosis (±6 months from diagnosis date) for the IIH cohort. The most recent measurements taken before the age of 16 were used for the population without IIH. BMI was only calculated for children that had height and weight measured on the same day and was only available for a subset of the IIH cohort (29%) and population (21%). Aged-based BMI z-scores and BMI categories were calculated in R using the zscorer package and the World Health Organisation (WHO) classification of nutritional status.21–23
Health Care Outcomes
We recorded children who had CSF diversion surgery (Supplementary Table e-3), at least moderate visual impairment (Supplementary Table e-4), and prescriptions for acetazolamide, topiramate, or furosemide using prescription and diagnosis codes (Supplementary Table e-5). We created a control cohort with 3:1 matching on age (week of birth), sex, and WIMD quintile. Controls were given a study entry date corresponding to the IIH diagnosis date of the case to which they were matched. We calculated rate ratios for unscheduled hospital admissions in children with IIH without CSF shunts compared with controls, and children with IIH and CSF shunts compared to children with IIH without CSF shunts.
Statistical Analysis
We used a logistic regression model to calculate odds ratios for developing IIH for 2 groups: the entire cohort (177 children), and the cohort with BMI measurements (52 children). The features analysed were sex (female to male), socio-economic deprivation (most deprived quintile to other deprivation quintiles separately), and BMI category (for BMI cohort only). A diagnosis of IIH was the binary outcome variable. Socio-economic deprivation was regressed using the WIMD quintiles as a categorical feature for combined male and female and for each sex separately. BMI category was regressed as a categorical variable. In the regression model the population at risk were all children (all children with a BMI measurement for the BMI group) registered with a GP in Wales within the study period. We used R version 4.0.4.
Results
We analysed 10.3 million patient years of data, identifying 177 children with IIH between 2003 and 2017 (average incidence 3.0/100 000per year) (Supplementary Figure e1 and Table 1). Our case ascertainment method identified confirmed IIH cases with 90% sensitivity (36/40 cases identified). The incidence and prevalence of IIH in children in Wales in 2017 was 3.5/100 000per year and 17.5/100 000 respectively. This represents a significant increase when compared to the corresponding figures in 2003 (1.6/100 000per year and 6.5/100 000per year, P = .001 and P < .001) (Figure 1).
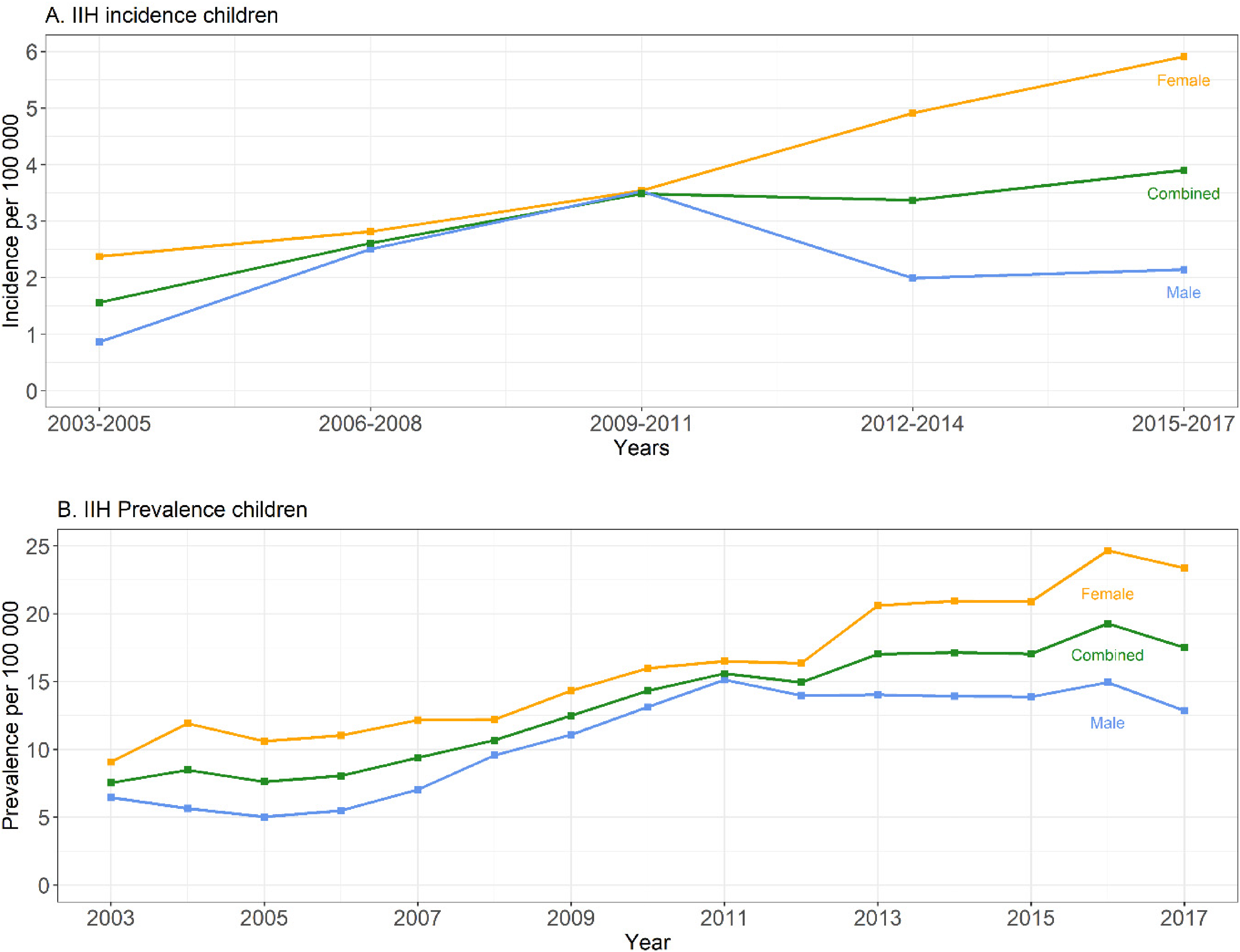
(A) The average annual incidence (per 100 000) of IIH in children in Wales across 3-year periods from 2003 to 2017 for male, female, and combined. The incidence was aggregated by 3 years to prevent possible reidentification of individuals through the disclosure of small numbers. (B) The annual prevalence (per 100 000) of IIH in children in Wales from 2003 to 2017. IIH, idiopathic intracranial hypertension.
Characteristics of the Paediatric Idiopathic Intracranial Hypertension (IIH) Cohort.
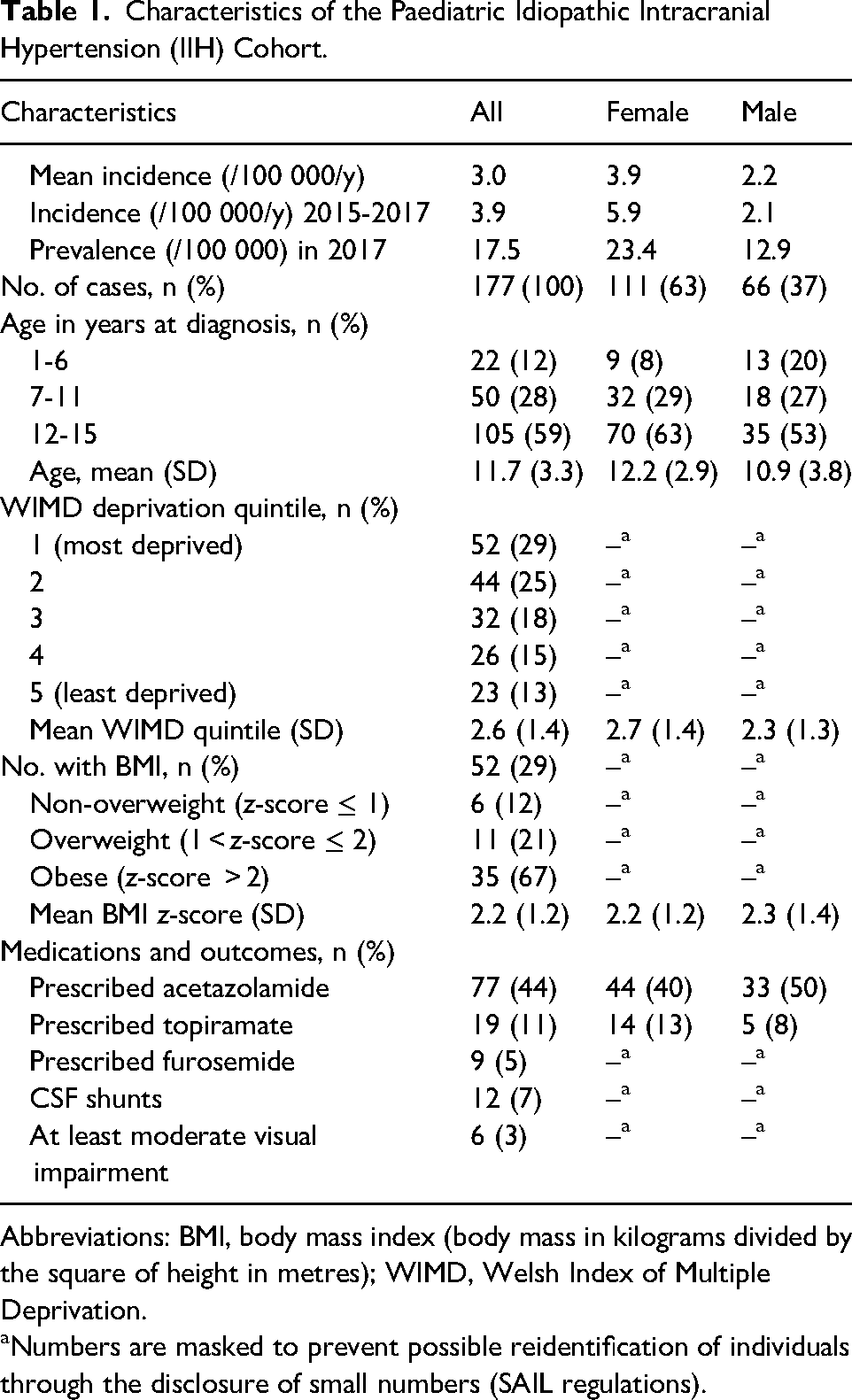
Abbreviations: BMI, body mass index (body mass in kilograms divided by the square of height in metres); WIMD, Welsh Index of Multiple Deprivation.
Numbers are masked to prevent possible reidentification of individuals through the disclosure of small numbers (SAIL regulations).
We found associations with IIH, female sex, deprivation, and BMI (Table 2, Figures 2 and 3). Three percent of the cohort (6 children) were recorded as having at least moderate visual impairment, a mean of 1013 days (33 months) after diagnosis (Table 1). Seven percent of the cohort (12 children) underwent CSF diversion procedures a mean of 404 days (13 months) after diagnosis. No children underwent bariatric surgery.
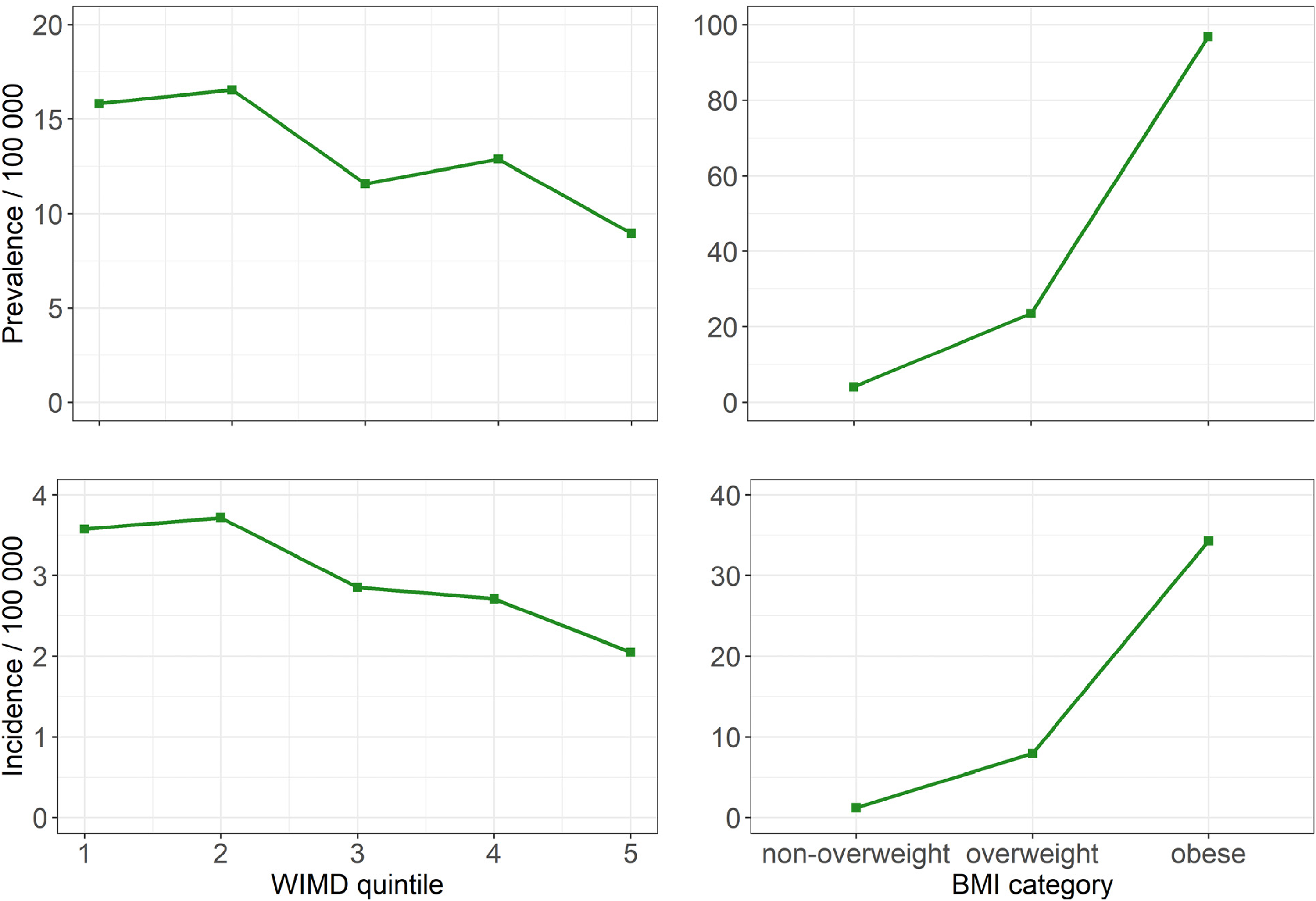
Average IIH prevalence and incidence (per 100 000) in children from 2003 to 2017 by WIMD deprivation quintile (1 = most deprived, 5 = least deprived) and BMI category. BMI, body mass index; IIH, idiopathic intracranial hypertension.
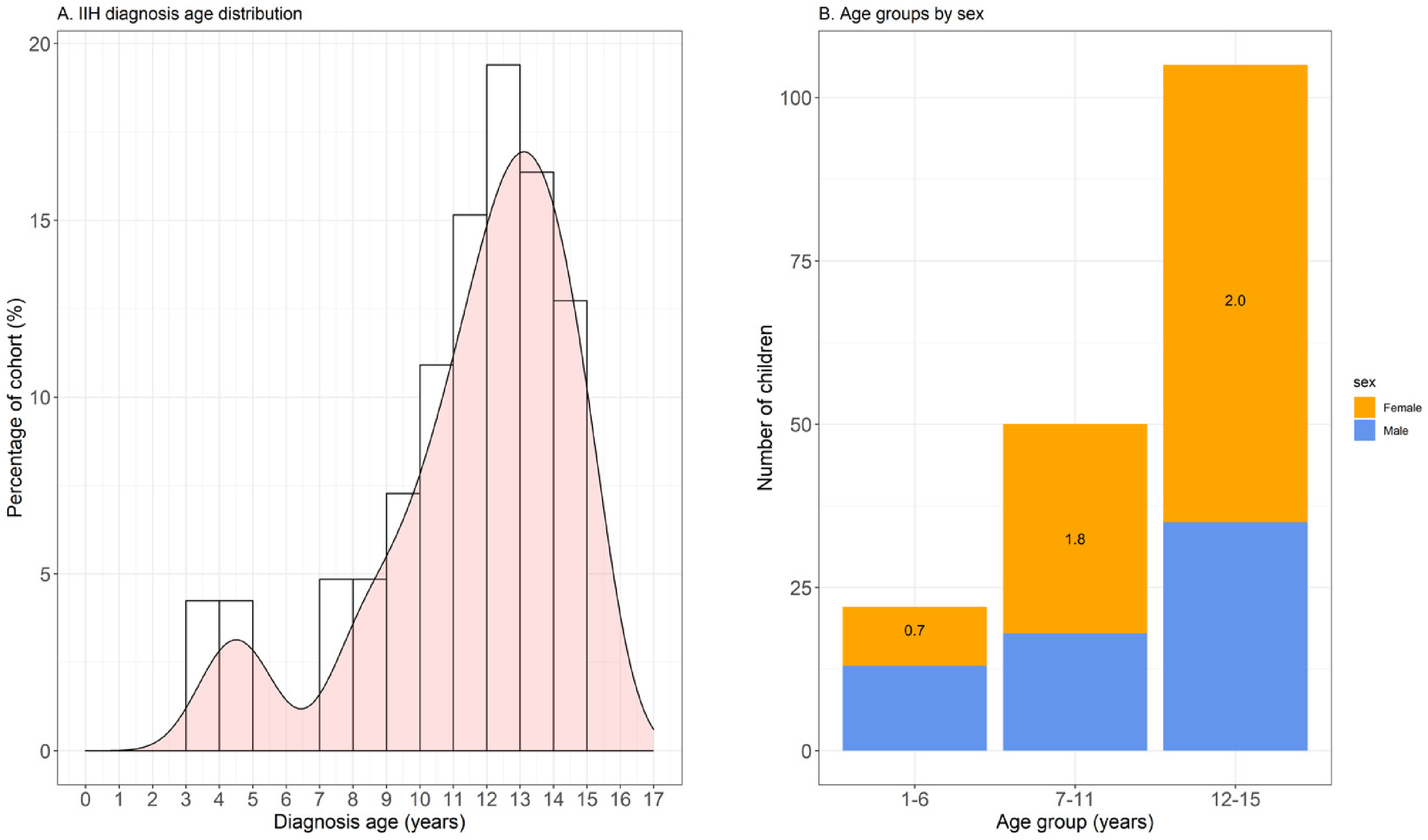
(A) Histogram showing the distribution of IIH diagnosis age. The bin width is 1 year. All bars that refer to less than 5 individuals have been removed to mask values that refer to <5 individuals (SAIL regulations). (B) Stacked bar chart showing the number of children with IIH split into diagnosis age groups and sex. The number on each bar is the ratio of female to male children in each age group. IIH, idiopathic intracranial hypertension.
Odds Ratios and 95% CIs for Developing Idiopathic Intracranial Hypertension as a Child by Sex and Deprivation Using a Logistic Regression Model.

Abbreviations: BMI, body mass index (body mass in kilograms divided by the square of height in metres); OR, odds ratio.
Not reported because of small numbers of children in deprivation quintile 5 with BMI readings. Cannot report total because of SAIL regulations.
Children with IIH had increased rates of hospital admissions (Supplementary Table e-6). The average number of admissions per year for controls, children with IIH without CSF shunts, and children with IIH with CSF shunts were 0.1, 0.4, and 0.8, respectively. Children with IIH without CSF shunts had 4 times the rate of unscheduled hospital admissions compared with controls (rate ratio: 3.9, 95% CI 3.6-4.3). Children with IIH with CSF shunts had twice the rate of hospital admissions compared to children with IIH without CSF shunts (rate ratio: 2.0, 95% CI 1.7-2.4).
Discussion
The average incidence of 3.0/100 000per year in our study is higher than most previous studies. Studies from the United Kingdom, United States, and Germany between 2007 and 2013 found the incidence of IIH in children to be 0.5-0.7/100 000per year.10,13,14 A 2005-2007 population-based study of hospital data in Israel found an average incidence of 1.8/100 000, which is similar to our average incidence for those years (1.6 and 2.6/100 000per year for 2003-2005 and 2006-2008). 24 One reason for this higher incidence may be that we have used population-level routinely collected data and may therefore have included more cases. Our data are also more recent and may reflect a true increase in the incidence of IIH in children, a trend that has been recently described in adults.5,6 The trend for increasing incidence of IIH may be due to an increase in obesity rates in children in Wales, an increase in routine funduscopy in opticians, and an increased awareness of IIH in general. 25
The sex and age distributions of our cohort (Table 1 and Figure 3) are in keeping with previous paediatric cohort studies.13,14,26 Sixty-three percent of our cases were female, and 87% were diagnosed at ≥8 years. Girls had a higher risk of developing IIH than boys (OR = 1.8, 95% CI 1.3-2.4) although there seems to be a higher proportion of IIH in male children when compared to adults.5,6,10,26
There appear to be 2 distinct groups of paediatric IIH based on age. The younger group accounts for a minority of the cases (12% diagnosed <7 years old), has a higher proportion of boys, and the children are less likely to be overweight. The older group is mostly female and has a higher proportion of overweight and obese children.10–13
A similar proportion of our cohort underwent CSF shunt surgery (7%) (Table 1) when compared with studies of adults (9%), and adults and children (6%-8%).5,6,27 It may be, however, that the number of shunt procedures performed is decreasing. 27 In addition, 3.3% of our cohort were diagnosed with at least moderate visual impairment, which is higher than in other cohorts studied (1.5%-1.9%).5,6,27
Forty-four percent of cases had at least 1 prescription of acetazolamide. The proportion prescribed acetazolamide seems low given that this medication has been shown to show modest benefits in a randomised controlled trial—the idiopathic intracranial hypertension treatment trial (IIHTT). 28 However, the IIHTT was carried out in adults, and guidelines recommend its use in adults with IIH. 29 Eleven percent of the children with IIH were prescribed topiramate and 5% furosemide. We did not have access to secondary care prescription data.
Obesity is an established risk factor for IIH in children.12,19,26 In our subgroup analysis of children with a BMI measurement in primary care (21% of the GP population), the odds ratio for developing IIH was 21 (95% CI 9-51, P < .001) in obese children and 5.1 (95% CI 1.9-14, P = .001) in overweight children. These ORs are higher than previous studies (ORs for obese children of 6.5-16, and 3.6 for overweight children). 19 Our results for obesity need to be interpreted with caution given that only a minority of children had their BMI recorded in primary care. There is a possibility of selection bias—children with weight issues are more likely to have their BMI recorded.
We found an association between socio-economic deprivation and IIH (OR = 0.58; 95% CI 0.35-0.95, P = .029 for least compared with most deprived children, Figure 3). Children in the most-deprived areas are 1.7 times more likely to develop IIH than children in the least deprived areas. Fifty-four percent of our cases came from the 2 most deprived quintiles, similar to adult studies.5,6 The association between IIH and socio-economic deprivation is well documented in adults; there is also some evidence of an association in children.5,6,14 Obesity in children is linked with socio-economic deprivation in Wales. 25 After adjusting for BMI, in the minority of children with BMI measurements (Table 2), there is no significant association between IIH and socio-economic deprivation. This would seem to suggest that the association between socio-economic deprivation and obesity could explain the association between IIH and socio-economic deprivation. However, the numbers are small in this subgroup, and this association is worthy of further analysis since there may be other factors with socio-economic deprivation that explain the link.
Children in the most-deprived areas are more likely to be overweight or obese, and obese children in the most-deprived areas have higher BMI z-scores than obese children in less deprived quintiles. This appears to be true for both the child population of Wales, and the IIH cohort (Supplementary Table e-7). Analysis of socio-economic deprivation in a paediatric IIH cohort with more complete BMI data is required to support this.
Children with IIH in our study had 4 times the rate of unscheduled hospital admissions compared with controls. Children with CSF shunts had even higher rates of hospital admissions. A recent large population study of adults and children with IIH showed an increasing number of hospital admissions because of IIH in England, but to our knowledge there have been no studies specifically looking at hospital admissions in children with IIH. 27 It is likely that a significant proportion of excess hospital admissions are due to the need for urgent inpatient investigation of papilloedema but are also explained by admissions for headache and concerns about vision loss. The 30-day readmission after primary CSF shunt placement may be as high as 41%, which can explain the higher hospital admission rate in the group of children with IIH and shunts. 27 Children with IIH and shunts have double the rate of unscheduled hospital admissions compared to children with IIH without shunts.
Strengths
We used population-level data over a 15-year period giving a relatively large number of paediatric IIH cases. Our method of identifying paediatric IIH cases in routinely collected data had a sensitivity of 90% when identifying 40 confirmed IIH cases from a paediatric neuroscience centre. We used detailed area-based socio-economic deprivation measures and identified a large, matched cohort when measuring hospital admission rates.
Limitations
We could not accurately measure the specificity of our case ascertainment algorithm. Our incidence and prevalence figures may be underestimates of true figures given that our case ascertainment algorithm was 90% sensitive. We also used routinely collected data, not research specific data and cases without an appropriate diagnosis code in their health data would not have been included.
The WIMD is an area-based not an individual-based measure of socio-economic deprivation. Although the areas used in WIMD are small, it is still possible that children living in the same WIMD area have quite different individual levels of socio-economic deprivation. BMI measurements were only recorded in primary care for 30% of children with IIH, raising the possibility of ascertainment bias, which needs to be considered when interpreting results associated with BMI. We have also used BMI measurements ±6 months from diagnosis date. BMI measurements can change with time in children, and it is possible that the BMI measurements that we have used are significantly different from those at the time of diagnosis. We have used age cut-offs to group IIH cases rather than pubertal status using Tanner staging as in other studies. 12 Data to determine puberty status other than age are not available.
We did not specifically exclude demyelinating or infectious disorders when ascertaining cases of IIH or prescriptions of growth hormone in our primary analysis. A post hoc analysis of the data identified a total of 5 cases throughout the study period who had a diagnosis of a demyelinating disease, CNS infection, or prescription of growth hormone (see Supplementary Tables e8 and e9 for codes) a year before or a year after the diagnosis of IIH. These cases of possible secondary IIH represent 2.8% of the total cohort. This also needs to be balanced against the small number of true positive cases that we will have missed with our case ascertainment algorithm that was 90% sensitive.
Using routinely collected, anonymised data meant that we were able to look at relatively large numbers of patients; however, we could not assess the clinical features of individual patients, whether they were symptomatic or asymptomatic or what were the criteria used when deciding to perform CSF shunting.
Conclusions
In this large, population based, study we have found a higher incidence of IIH in children than previously reported. The incidence of IIH in girls has more than doubled from 2003 to 2017. Children who are obese or overweight as well as those in more deprived areas are more likely to develop IIH. Children with IIH have almost 4 times more unscheduled hospital admissions per year when compared with children without IIH. The increasing incidence of IIH, increased health care utilisation of children with IIH, and links with obesity and socio-economic deprivation are relevant findings to health care professionals and policy makers.
Supplemental Material
sj-docx-1-jcn-10.1177_08830738261436139 - Supplemental material for Paediatric Idiopathic Intracranial Hypertension in Wales: A Retrospective Observational Study
Supplemental material, sj-docx-1-jcn-10.1177_08830738261436139 for Paediatric Idiopathic Intracranial Hypertension in Wales: A Retrospective Observational Study by Russell Azad Khan, Huw Strafford, Beata Fonferko-Shadrach, Kathryn Bush, Robert Powell, Cathy White, Arron Lacey and William Owen Pickrell in Journal of Child Neurology
Footnotes
Acknowledgements
Our study makes use of anonymized data held in the Secure Anonymised Information Linkage (SAIL) system, which is part of the national e-health records research infrastructure for Wales. The authors thank the data providers who make anonymized data available for research.
ORCID iDs
Ethical Considerations
Our study had SAIL information governance review panel (IGRP) approval (ref 0695). The Research Ethics Service has previously confirmed that SAIL projects using anonymised, routinely collected data do not require specific UK National Health Service research ethics committee approval.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Russell Azad Khan–Drafted and edited the manuscript, performed statistical analysis, analysed the data. Huw Strafford–Provided supervision, assisted with data analysis, edited the manuscript. Beata Fonferko-Shadrach–Provided supervision, edited the manuscript. Kathryn Bush–Provided specialist advice, edited the manuscript. Robert Powell–Provided supervision, edited the manuscript. Cathy White–Provided specialist paediatric IIH advice, edited the manuscript. Arron Lacey–Provided supervision, assisted with data analysis, edited the manuscript. William Owen Pickrell–Designed, coordinated, and supervised the study, drafted and edited the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by staff funded as part of the Brain Repair and Intracranial Neurotherapeutics (BRAIN) Unit Infrastructure Award (grant UA05) and the Wales Gene Park. The BRAIN Unit and the Welsh Gene Park are funded by the Welsh Government through Health and Care Research Wales. This work was also supported by Health Data Research UK, which receives its funding from HDR UK Ltd. (HDR-9006) funded by the UK Medical Research Council, Engineering and Physical Sciences Research Council, Economic and Social Research Council, Department of Health and Social Care (England), Chief Scientist Office of the Scottish Government Health and Social Care Directorates, Health and Social Care Research and Development Division (Welsh Government), Public Health Agency (Northern Ireland), British Heart Foundation (BHF), and the Wellcome Trust.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data used in this study are not publicly available because proposals to access the SAIL Databank are subject to review by an independent Information Governance Review Panel (IGRP) to ensure compliance with data protection laws.
Supplemental Material
Supplemental material for this article is available online.
