Abstract
Previous reports have described a spectrum of clinical phenotypes in patients with
The
Methods
The Pediatric Epilepsy Outcome-Informatics Project was implemented on January 1, 2016, by the Children's Comprehensive Epilepsy Program at the Alberta Children's Hospital with a vision to provide comprehensive clinical care, quality improvement, and research that leads to improved outcome and education for all stakeholders in a manner that optimizes societal resources. 7 The project includes utilization of standardized clinical note with common clinical data elements, point of care data entry, near-time data analysis, and availability of outcome dashboards. The clinical note was used as standard of care by epileptologists and nurses in the Neurology service at the Alberta Children’s Hospital at clinical encounters as part of patient's electronic health records. Clinical data elements included seizure description (duration, frequency, semiology, epilepsy diagnoses), treatment (antiseizure medications, side effects, nonmedication therapies), and psychological comorbidities. The patient data are then aggregated, analyzed, and then presented as population- and patient-specific dashboards by Data and Analytics (Alberta Health Services) and is available to clinicians at the point of care.
Using the outcome dashboard, we identified 4 epilepsy patients with
Interpretation of electroencephalography and report generation were performed by epileptologists from the Alberta Children's Hospital and is in accordance with the rigorous standards set forth by the Canadian Society of Clinical Neurophysiologists. Classification of seizure types was in keeping with the recent updated classification of epileptic seizures from the International League Against Epilepsy (ILAE). 8 Interpretation and classifying genetic variants were performed as per the American College of Medical Genetics and Genomics (ACMG) guidelines. 9
Case Series
Case 1 (N1)
Patient N1 is a 10-year-old female of Lebanese descent, and she was born to a 36-year-old mother via cesarean section at term. She was seen by pediatric neurology at 4 months of age, and her head circumference was around 40th to 50th percentile. General examination revealed bilateral scalloping of the temples, brittle hair, protuberant large tongue, and externally rotated and flat ears. Neurologic examination showed significant axial hypotonia, appendicular hypertonia, and normal deep tendon reflexes. Other medical conditions include bilateral Duane syndrome, sleep-disordered breathing, systemic hypertension, nephrocalcinosis, hypocalcemia, and gastroesophageal reflux. Developmentally, she did not speak any words and was nonambulatory. She is able to roll from prone to side, sit independently, and take steps with support. She can hold objects but does not have clear hand preference. She expresses discomfort with facial expression and sounds. Detailed neuropsychological assessment was not performed because of lack of patient's ability to engage with the testing. Research whole exome sequencing performed at the University of Calgary revealed a
A surveillance routine electroencephalography (EEG) performed at the age of 13 months revealed rare right temporal spikes and intermittent right posterior slowing. EEG at 3 years of age showed independent spikes recorded in the left and right parietal head regions that were activated in sleep, as well as infrequent generalized epileptiform discharges. Photoparoxysmal response was detected, most prominently at 11-Hz stimulation inducing generalized spike wave discharges. This was blocked by orange, red, blue, and green filters. Her first epileptic seizure was at 3 years 5 months of age and was described as focal preserved consciousness seizure with clonic of the face. She later developed frequent generalized myoclonic seizures. Other event types included staring spells and laughing spells that were deemed nonepileptic (behavioral) because of lack of ictal correlate on the EEG. Semiologically, the laughing spells did not resemble gelastic seizures. She was started on levetiracetam and clonazepam. At 6 years of age, EEG showed frequent multifocal and generalized discharges with epochs of spike wave activation in sleep. The patient eventually developed side effects to both levetiracetam (withdrawn, hallucination) and clonazepam (sedation), and the family declined to continue antiseizure medication. EEG performed at 9 years of age showed frequent generalized epileptiform discharges with photoparoxysmal responses most prominently at 18-Hz stimulation.
Case 2 (N2)
Patient N2 is a 20-year-old girl, older sibling of N1. Neurologically, she has global developmental delay, and examination revealed dysmorphia, hypotonia, and normal deep tendon reflexes. Her medical diagnoses included closed ventricular septal defect, short stature, clubfeet, hypercalcinuria, nephrocalcinosis congenital cataracts, retinal detachments, and glaucoma. At 17 years of age, she sits independently and requires a walker to ambulate. She has no clear hand preference. She uses an eye-gaze, “yes-no” communication device. She understands 1-step commands. Her clinical findings are in keeping with Basel-Vanagaite-Smirin-Yosef syndrome, and she shares the same homozygous
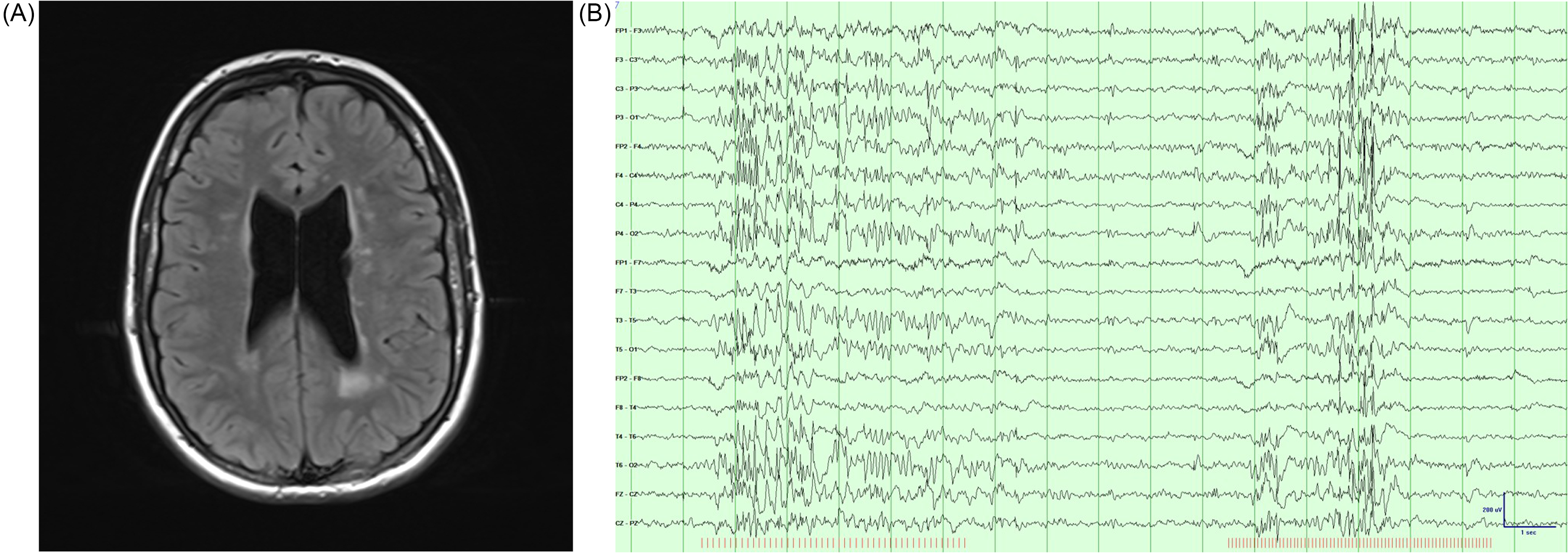
(A) Axial FLAIR Image Above the Level of the Basal Ganglia of Patient N2 Demonstrating Patchy Periventricular FLAIR Hyperintensity. (B) Epoch of EEG of Patient N2 at 11 Years of Age Demonstrating Robust Photoparoxysmal Response. Photic Stimulation at 10 Hz and 16 Hz Produced Occipital Predominant Diffusely Distributed Polyspikes and Spikes. Sensitivity 10 μV/mm; Time Base, 30 mm/s. FLAIR, Fluid-Attenuated Inversion Recovery.
Onset of epilepsy was at 3 years of age. Prolonged focal impaired consciousness seizures and generalized tonic-clonic seizures were documented. Other event types included episodic laughter and screaming that did not have EEG ictal correlate, and thus these were deemed nonepileptic (behavioral) in nature, similar to her sister (N1). The patient was started on levetiracetam. EEG demonstrated focal left and right parietal and generalized epileptiform discharges as well as a photoparoxysmal response at multiple frequencies (Figure 1B). Although the seizures were initially controlled on levetiracetam, the family declined to continue antiseizure medications after the age of 17 years because of adverse effects. This led to a recurrence of monthly generalized tonic-clonic seizures.
Case 3 (N3)
Patient N3 is a 5-year-old girl, born at term to a mother with gestational diabetes. Both parents are of Lebanese descent. She had a history of bilateral congenital cataracts, diagnosed at 9 months of age. She had a moderate atrial septal defect and bilateral metatarsus adductus type 1, which were managed conservatively. Developmentally, she is able to ambulate independently with bilateral ankle foot orthoses at 2.5 years of age. Fine motor skills were normal. She communicates by babbling and responds to her name. She appears social with adults.
At 2 years of age, she had a bilateral tonic-clonic seizure—unknown whether focal or generalized in the context of a urinary tract infection. EEG showed frequent generalized epileptiform discharges and independent multifocal epileptiform discharges as well as photoparoxysmal response elicited between 9 and 30 Hz that was blocked by blue, red, and green filters. Four months after her first seizure, she had an unprovoked focal impaired consciousness seizure. She was seizure-free since levetiracetam was started.
Brain MRI showed subtle T2 hyperintense signals within the subcortical white matter in the left frontal lobe and diffuse scattered prominent perivascular spaces bilaterally.
Patient N3 has a second cousin who has homozygous variants in the
Discussion
In this case series, we described 3 patients from 2 unrelated Lebanese families in Calgary, Alberta, with the same
A Comparison of the Clinical Features of the 3 Basel-Vanagaite-Smirin-Yosef Syndrome Patients in This Report and Those Previously Reported in the Literature.
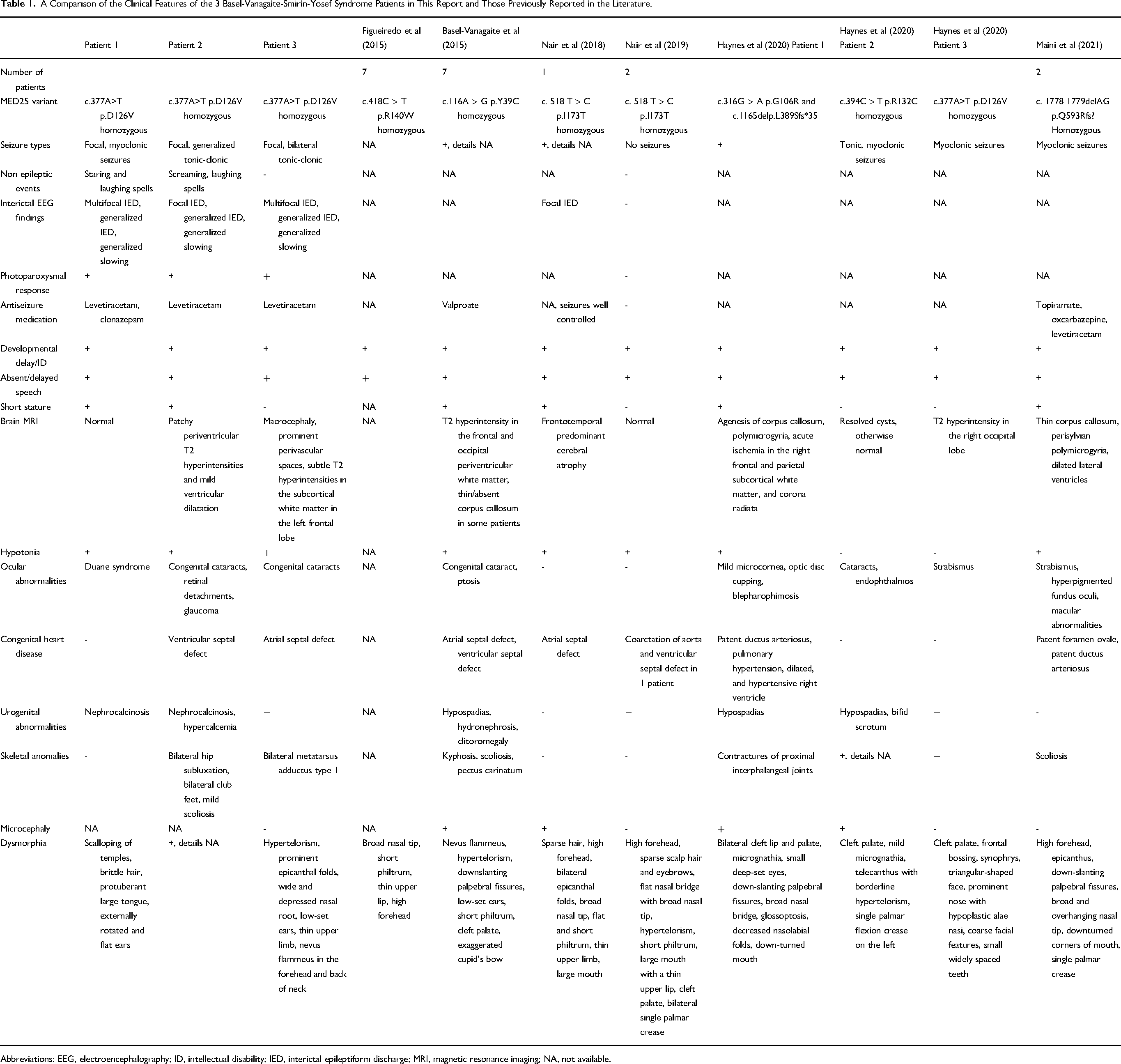
Abbreviations: EEG, electroencephalography; ID, intellectual disability; IED, interictal epileptiform discharge; MRI, magnetic resonance imaging; NA, not available.
The epilepsy phenotypes between the 3 patients were remarkably similar. Onset of seizure was between 2 and 3 years of age, and seizures were both focal and generalized in onset. In terms of seizure control, levetiracetam seemed to be very effective in the case of N3, but unclear in the case of N1 and N2 because of lack of adherence to antiseizure medications. EEG in all 3 patients demonstrated multifocal and generalized discharges and photoparoxysmal responses. Photoparoxysmal response or photosensitivity refers to the occurrence of spikes or spike and wave discharges during intermittent photic stimulation.
10
Photoparoxysmal response is found in 0.6% to 30% of patients with epilepsies.
11
However, the pathogenesis of photosensitivity is still poorly understood.
12
Three susceptibility loci mapped to chromosome 6p21, 13q31, and 7q32, termed photoparoxysmal response 1-3, respectively, have been described.
12
Given that all 3 patients with the same
Epilepsy appears to be a common finding in the published literature cases of
Variants in other mediator complex subunits have been associated with genetic and neurologic diseases
18
These subunits include MED1, MED12, MED12L, MED13, MED13L, MED17, MED20, MED23, MED25, MED27, and CDK8. Intellectual disability appears to be a common feature among pathogenic variants in these genes. Pathogenic variants in
Conclusion
Epilepsy is a common finding in patients with Basel-Vanagaite-Smirin-Yosef syndrome. We revealed that the electroclinical characteristics of their epilepsy demonstrated remarkable similarity, such as the presence of multifocal and generalized discharges and photoparoxysmal response. This is the first detailed description of the epilepsy phenotype of Basel-Vanagaite-Smirin-Yosef syndrome patients. Systemic features overlapped with patients described in previous reports.
Footnotes
Ethical Approval
This case series does not require ethics review by the research ethics board (REB) according to the 2nd edition of the Tri-Council Policy Statement (TCPS2) of Canada. Consent for chart review and publication was obtained from the patients’ family.
Author Contributions
AC-HN wrote the first draft of the manuscript and made the necessary revisions. SD reviewed ethics requirement and revised the manuscript. AMI identified and provided expert advice on the
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AC-HN was funded by Alberta Children's Hospital Research institute (ACHRI) and Stollery Clinical Research Fellowship award. The Pediatric Epilepsy Outcome-Informatics Project was funded by the Alberta Children's Hospital Foundation, Department of Pediatrics at the University of Calgary, and the Alberta Children's Hospital Research Institute. AMI received funding from the Alberta Children's Hospital Foundation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
