Abstract
This study evaluated the efficacy of the multiple sclerosis disease-modifying therapies, intramuscular interferon beta-1a (Avonex) and subcutaneous peginterferon beta-1a (Plegridy), using data from the United States Network of Pediatric Multiple Sclerosis Centers. In this retrospective analysis, 154 patients with multiple sclerosis were included who were treated with Avonex (n = 130), Plegridy (n = 23), or both treatments (n = 1) before the age of 18 years. After 3 months’ sustained use acclimation (“wash-in”), the probability of being relapse-free during the first year was 68.3% for Avonex-treated patients and 69.9% for Plegridy-treated patients; annualized relapse rates were 0.50 and 0.59, respectively. Both disease-modifying therapies demonstrated efficacy similar to that reported in adult populations. Despite the lack of formal approval for pediatric multiple sclerosis, these outcomes indicate that patients may benefit from treatment with Avonex or Plegridy. Understanding efficacy of specific disease-modifying therapies in pediatric multiple sclerosis is essential to making informed treatment decisions.
Although multiple sclerosis is more commonly diagnosed in patients 18 years of age or older, pediatric multiple sclerosis accounts for up to 10% of patients with multiple sclerosis.1–5 A growing body of literature has demonstrated that pediatric multiple sclerosis has distinct features compared to adult counterparts. Nearly all pediatric patients follow an initial relapsing-remitting course. 6 Patients with pediatric multiple sclerosis have an overall slower progression to irreversible neurologic disability; however, they reach this disability at an earlier age and have more frequent relapses.4,6–9 Early initiation of disease-modifying therapy is essential in these children and adolescents to help prevent accrual of neurologic disability.
A growing number of disease-modifying therapies using different mechanisms of action have been approved for use in multiple sclerosis. However, fingolimod remains the only US Food and Drug Administration–approved medication for use in pediatric multiple sclerosis in the United States. 10 Additionally, despite known differences between the clinical courses of pediatric and adult patients with multiple sclerosis, clinical trials and real-world safety and efficacy studies have been limited in pediatric compared to adult patients with multiple sclerosis. Clinicians are often left to extrapolate study findings in adults to the pediatric population when making disease-modifying therapy decisions and discussing options with families. Notably, in a large retrospective study of 307 children with subcutaneous (SC) interferon beta-1a, investigators reported tolerability and decreased relapse rates, highlighting a need to further evaluate different interferon beta-1a formulations for pediatric multiple sclerosis. 11
Historically, first-line therapy for pediatric multiple sclerosis has consisted of treatment with either glatiramer acetate or interferons.12–15 In recent years, there has been a trend of prescribing newer therapies early in the disease course; however, beta interferons remain a significant portion of disease-modifying therapy use in pediatric multiple sclerosis.16–18 Additionally, there may be co-morbidities or other considerations such as cytopenias, hypogammaglobulinemia, or co-existing immune dysregulation that may make disease-modifying therapies with lower risk of immunosuppressive effects preferred over therapies with higher risks related to immunosuppression for certain pediatric multiple sclerosis patients.
Intramuscular (IM) interferon beta-1a (Avonex) and IM/SC peginterferon beta-1a (Plegridy) are approved in the United States for the treatment of adult patients with relapsing forms of multiple sclerosis and have been shown to be effective in placebo-controlled, randomized trials and real-world evidence for this patient population.19–23 However, there are limited data regarding efficacy of these therapies in pediatric patients with multiple sclerosis.
Prior real-world data have demonstrated reduction of relapse rates in pediatric patients on injectables.24–26 Rates of adverse effects from beta interferons in pediatric patients have also been found to be similar to those of the adult population.24,27 Additionally, tolerability of interferons in the pediatric population has been previously described.27–29
We present a large retrospective analysis of pediatric patients with multiple sclerosis on Avonex or Plegridy. In this study, we summarize the demographic and clinical characteristics of patients with pediatric multiple sclerosis who have been treated with these medications and evaluate the effectiveness of these treatments based on clinical and radiographic outcomes.
Patients and Methods
Study Design
A multicenter, retrospective analysis of registry data from patients treated at United States Network of Pediatric Multiple Sclerosis Centers, which included 10 clinical sites, was conducted. Clinical data were prospectively obtained at clinic visits of patients enrolled between May 2011 and January 2023 with data logged in the Pediatric Multiple Sclerosis and Demyelinating Diseases database by February 2023. Demographics and clinical information from patients with pediatric-onset multiple sclerosis treated with Avonex or Plegridy were included. Patients were diagnosed according to the prevailing diagnostic criteria at the time based on evaluation of a pediatric multiple sclerosis expert (eg, McDonald 2010 or 2017 criteria). Data are managed by the Data Coordinating and Analysis Center at the University of Utah in Salt Lake City, Utah.
Study Population
The study population included individuals who used Avonex or Plegridy for any length of time before 18 years of age (Figure 1). Avonex dosing was most commonly 30 µg IM once a week, and Plegridy dosing was most commonly 125 µg SC approximately every 2 weeks. Patients were excluded if they had a recorded diagnosis at last entry before the data snapshot of primary progressive disease or a diagnosis other than multiple sclerosis. Patients with insufficient data were also excluded.
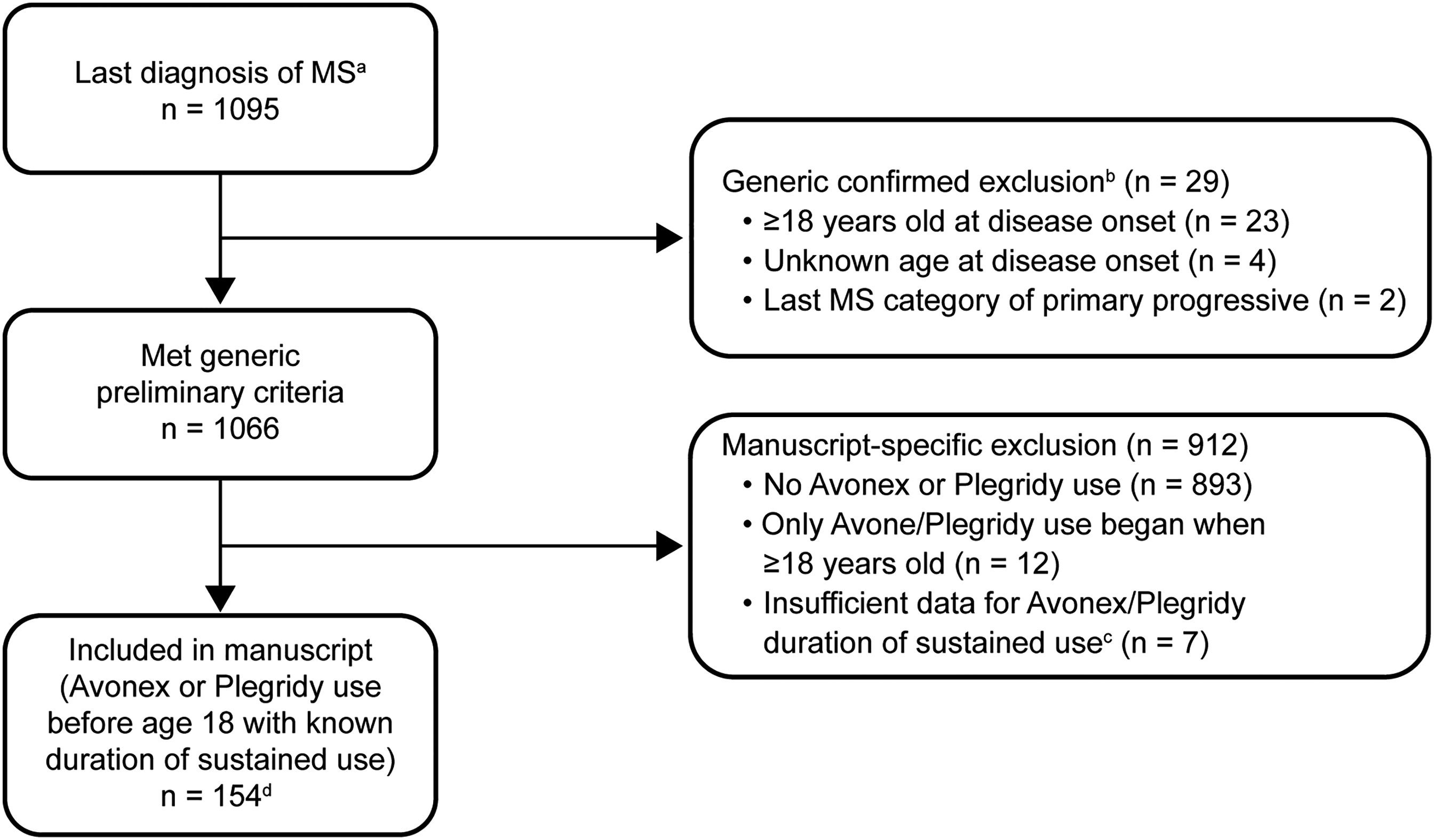
Participant-flow Diagram. aBased on PeMSDD database snapshot taken in February 2023 for participants at NPMSC member sites enrolled by January 2023. bMore than 1 may apply. cFor all uses that began before age 18 years, missing or inconsistent sustained start/sustained stop dates that would be used to determine sustained durations. dAvonex but no Plegridy, n = 130; Plegridy but no Avonex; n = 23; Avonex and Plegridy, n = 1. Throughout this article, if multiple uses were indicated, ignored any use with unknown duration due to missing/inconsistent sustained start/sustained stop dates. Abbreviations: MS, multiple sclerosis; NPMSC, Network of Pediatric Multiple Sclerosis Centers; PeMSDD, Pediatric Multiple Sclerosis and Demyelinating Diseases.
Follow-Up Period
Use of either of the focus disease-modifying therapies was considered sustained beginning on the day of its first use through the earliest of any of the following: last day of near-continuous use for the disease-modifying therapy, as indicated by no intermediate gap in use greater than 35 days; initiation of a different disease-modifying therapy; last day on which data were considered up to date. The acclimation period was defined by the first 90 days of Avonex or Plegridy based on general expert consensus regarding the need for time to assess efficacy of disease-modifying therapy, and is further supported by data suggesting the response to interferons stabilizes after 3 months of treatment.30,31 Most outcomes were defined beginning after 90 days of sustained use of the indicated disease-modifying therapy to better attribute outcomes to the indicated disease-modifying therapy rather than previous treatment(s) and only used subsequent observations if on continued sustained use of the disease-modifying therapy. Outcome follow-up was truncated when sustained use of the indicated disease-modifying therapy first ended, if not sooner.
Demographics, Clinical Characteristics, and Outcome Measures
Baseline characteristics included age at disease onset, age at initiation of disease-modifying therapy, sex, race, ethnicity, relapses prior to disease-modifying therapy initiation, Expanded Disability Status Scale scores, prior disease-modifying therapy use, and imaging characteristics. As this was a clinical registry, magnetic resonance imaging (MRI) scans were obtained with nonstandardized frequency per routine clinical care and at the discretion of the treating provider.
All relapse-related primary and secondary outcomes required at least 91 days of sustained use for the indicated disease-modifying therapy, and do not include events during the first 90 days. The starting day for time-to-event or rate outcome analyses was day 91 (ie, 90 days after the sustained use began). If the corresponding postacclimation “baseline” data were collected after day 91, then the starting day for those specific analyses was adjusted accordingly.
Relapses were defined as the appearance of new neurologic symptoms, or the return of previous symptoms, lasting more than 24 hours and occurring at least 30 days after a prior relapse/event, in the absence of infection, medication effects, trauma, or any other plausible explanation. 32 The primary outcome was years until first relapse, as defined above, during sustained Avonex or Plegridy use following the acclimation period. Events were counted as a distinct relapse if they started at least 30 days after the start of a prior relapse/event. Secondary outcomes assessed after the acclimation period while on sustained use of disease-modifying therapy included: annualized relapse rate; brain MRI-based time to first new/newly enlarging (N/NE) T2 lesion (this outcome used the first brain MRI during the postacclimation period to establish the baseline); brain MRI-based time to first gadolinium-enhancing (Gd+) lesion; time to disability progression, defined as an increase of ≥1 point in Expanded Disability Status Scale score compared to the first postacclimation Expanded Disability Status Scale score; and rate of change in postacclimation Expanded Disability Status Scale scores (change from first to last postacclimation Expanded Disability Status Scale scores divided by the years between these scores). Safety was not assessed, as the registry was not designed to capture these outcomes.
Statistical Analysis
Demographic and baseline characteristics were summarized among those who ever used Avonex and among those who ever used Plegridy. Depending on what was deemed most pertinent, numeric variables were summarized with means, standard deviations, and/or various quartiles. Categorical variables were summarized using frequencies and percentages. For time-to-event outcome analyses, estimation of percentiles was based on Kaplan-Meier methods to allow for right censoring.
The duration of the sustained use of the indicated disease-modifying therapy (Avonex or Plegridy) was summarized, considering both the initial use and all combined use of the indicated disease-modifying therapy, after excluding any use with a missing or implied negative duration.
Relapse rates were estimated using a negative binomial model. Time-to-event outcomes were modeled using Kaplan-Meier estimation; a post hoc subgroup analysis was added for the primary outcome with subgroups defined by whether there was an event during the acclimation period. Rates of change in Expanded Disability Status Scale scores were summarized but not modeled.
Comparisons between Avonex and Plegridy were not intended for formal significance testing—a pragmatic decision given potential confounding variables and the possibility of overlap between the patients using either disease-modifying therapy.
Ethics Statement
The data were collected in accordance with The International Conference on Harmonization Guidelines on Good Clinical Practice, the ethical principles outlined in the Declaration of Helsinki, and all applicable local laws and regulations. The Pediatric Multiple Sclerosis and Other Demyelinating Diseases database was reviewed and approved by each participating center's institutional review board. 33
Results
Study Population
A total of 154 patients with pediatric-onset multiple sclerosis were included who were treated with Avonex (n = 131) or Plegridy (n = 24), (n = 1 of whom was treated with both) before the age of 18 years (Table 1). The majority of patients were female in both the Avonex- and Plegridy-treated groups (63.4% and 58.3%, respectively). The median age of disease-modifying therapy initiation was 14.8 years for Avonex and 15.8 years for Plegridy. Most patients had not been on any disease-modifying therapy prior to initiating use of Avonex (82.4%) or Plegridy (58.3%). For patients on a prior disease-modifying therapy, this was most commonly another injectable. Most had new or newly enlarging T2 and Gd+ lesions develop, as identified by brain MRIs, between the time of the baseline brain MRI and the time of initiation of treatment.
Baseline Demographics and Disease Characteristics of Patients in the NPMSC Registry Treated With Avonex and/or Plegridy.

Abbreviations: BMI, body mass index; DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; Gd+, gadolinium-enhancing; IQR, interquartile range; MRI, magnetic resonance imaging; N/NE, new/newly enlarging; NPMSC, Network of Pediatric Multiple Sclerosis Centers.
Among patients with disease duration of ≥1 year prior to use of the indicated DMT.
Among patients with a second demyelinating event prior to use of the indicated DMT.
Nearest EDSS had to be <6 months prior to initiating indicated DMT.
Injectable disease-modifying therapies include glatiramer acetate (Copaxone and Glatopa), interferon beta-1a (Avonex and Rebif), interferon beta-1b (Betaseron and Extavia), and peginterferon beta-1a (Plegridy).
Other disease-modifying therapies included cyclophosphamide and mitoxantrone.
Considered missing for those who did not have brain scans on ≥2 different days on or before first day of indicated DMT use.
Considered missing for those who did not have brain scans with gadolinium contrasts on ≥2 different days on or before first day of indicated DMT use.
Considered missing for those who did not have a brain scan with gadolinium contrasts at least once on or before first day of indicated DMT use.
Treatment Parameters
Median duration of the initial use of disease-modifying therapy was 1.1 years in patients who received Avonex and 0.6 years in patients who received Plegridy (Table 2). Avonex was first initiated in this cohort as early as 2001 (median 2012), whereas Plegridy initiations began in 2014, with a median start year of 2016. There were 28 patients (21.4%) with ongoing use of Avonex and 7 patients (29.2%) with ongoing use of Plegridy. In patients who at one point stopped near-continuous use of Avonex or Plegridy, it was uncommon to resume and have multiple periods of sustained use (9 patients and 0 patients, respectively). Although challenging to quantify precisely because of limitations of available data, provided reasons for stopping the first Avonex use, when provided, were most commonly because it was noted as being ineffective, with side effects and personal choice being two other prominent reasons. Provided reasons for stopping the first Plegridy use, when provided, were most commonly because it was noted as being ineffective or because of side effects.
Treatment Parameters for Patients in the NPMSC Registry Treated With Avonex and/or Plegridy.
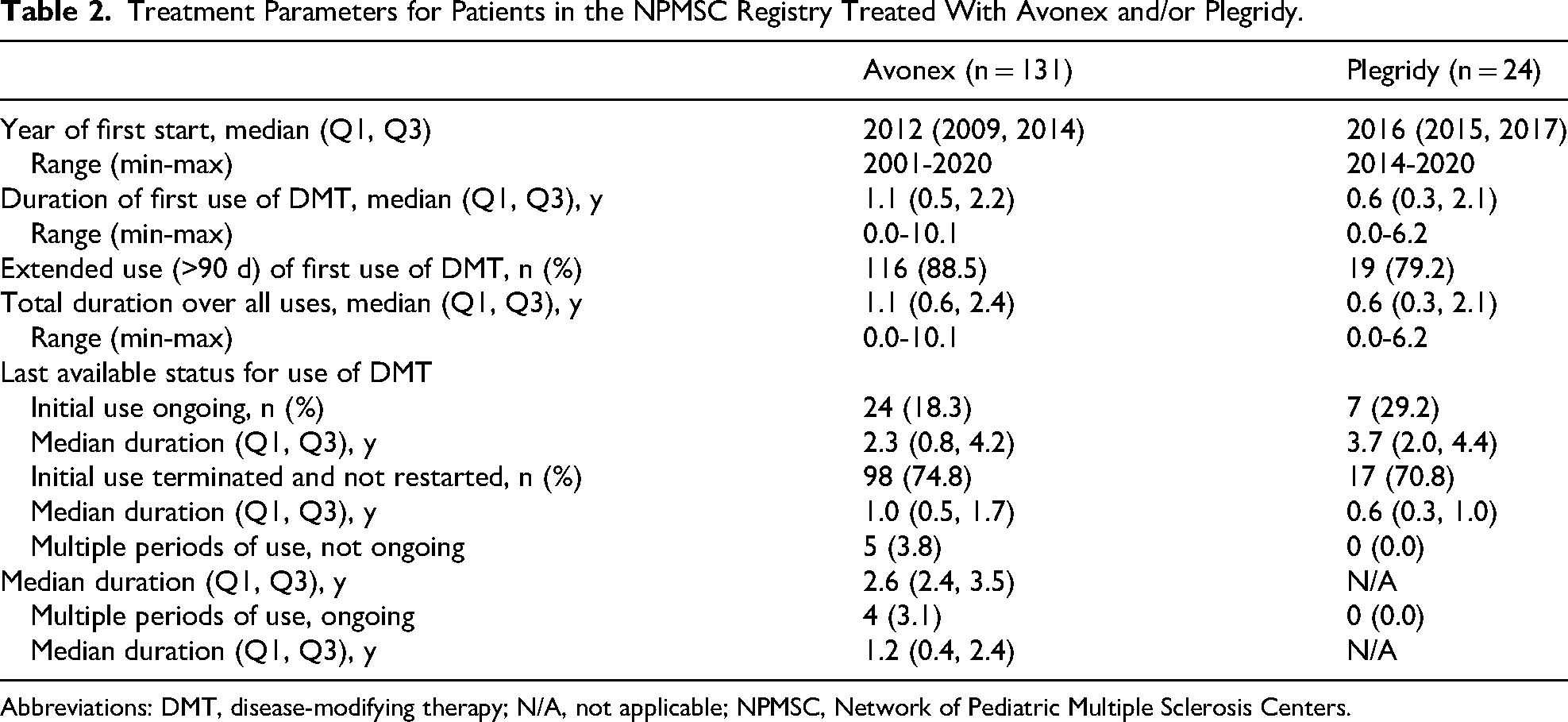
Abbreviations: DMT, disease-modifying therapy; N/A, not applicable; NPMSC, Network of Pediatric Multiple Sclerosis Centers.
Outcomes
During the first 90 days of therapy, 19.5% of Avonex and 10.5% of Plegridy patients had a clinical relapse (Table 3). After 3 months of use, the probability of being relapse-free during the next year was 68.3% for patients on sustained Avonex and 69.9% for patients on sustained Plegridy (Figure 2). The annualized relapse rate was 0.50 (95% confidence interval, 0.34-0.75) on Avonex and 0.59 (95% confidence interval, 0.12-3.00) for Plegridy. Among patients treated with Avonex, those who did not experience a relapse during the initial 90-day acclimation period remained relapse-free for a median of 3.9 years after acclimation, compared to a median relapse-free duration of only 1.4 years after acclimation in those who experienced a relapse during that period.
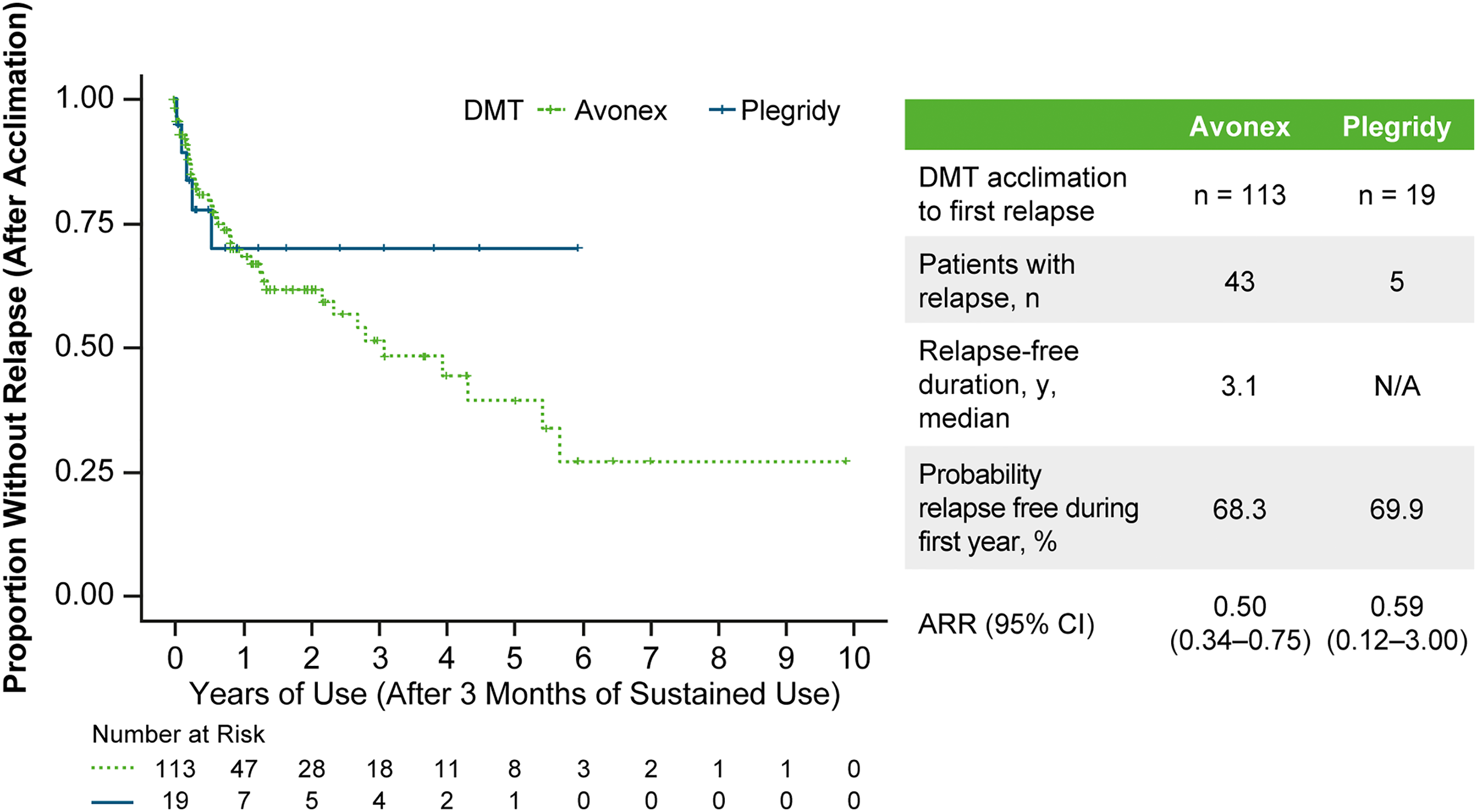
Kaplan-Meier Plot of Time to First Relapse in Patients on Avonex or Plegridy. Abbreviations: ARR, annualized relapse rate; DMT, disease-modifying therapy.
Postacclimation Outcomes for Patients Treated With Avonex and/or Plegridy.

Abbreviations: ARR, annualized relapse rate; DMT, disease-modifying therapy; N/A, not applicable. All rows that present an n value indicate the number of patients who provided at least some data for the outcome's analysis.
The probability of having no N/NE T2 lesions in the year following the first postacclimation brain MRI was 62.0% for the Avonex-treated patients and 37.5% for the Plegridy-treated patients (Figure 3A). Median duration without postacclimation N/NE T2 lesions in patients on Avonex or Plegridy was 1.1 years and 0.8 years after the first postacclimation brain MRI, respectively. The probability of having no Gd+ lesions was 60.5% for the Avonex-treated patients and 61.7% for the Plegridy-treated patients at 1 year postacclimation (Figure 3B). The median postacclimation duration without Gd+ lesions was 2.2 years in the Avonex cohort.
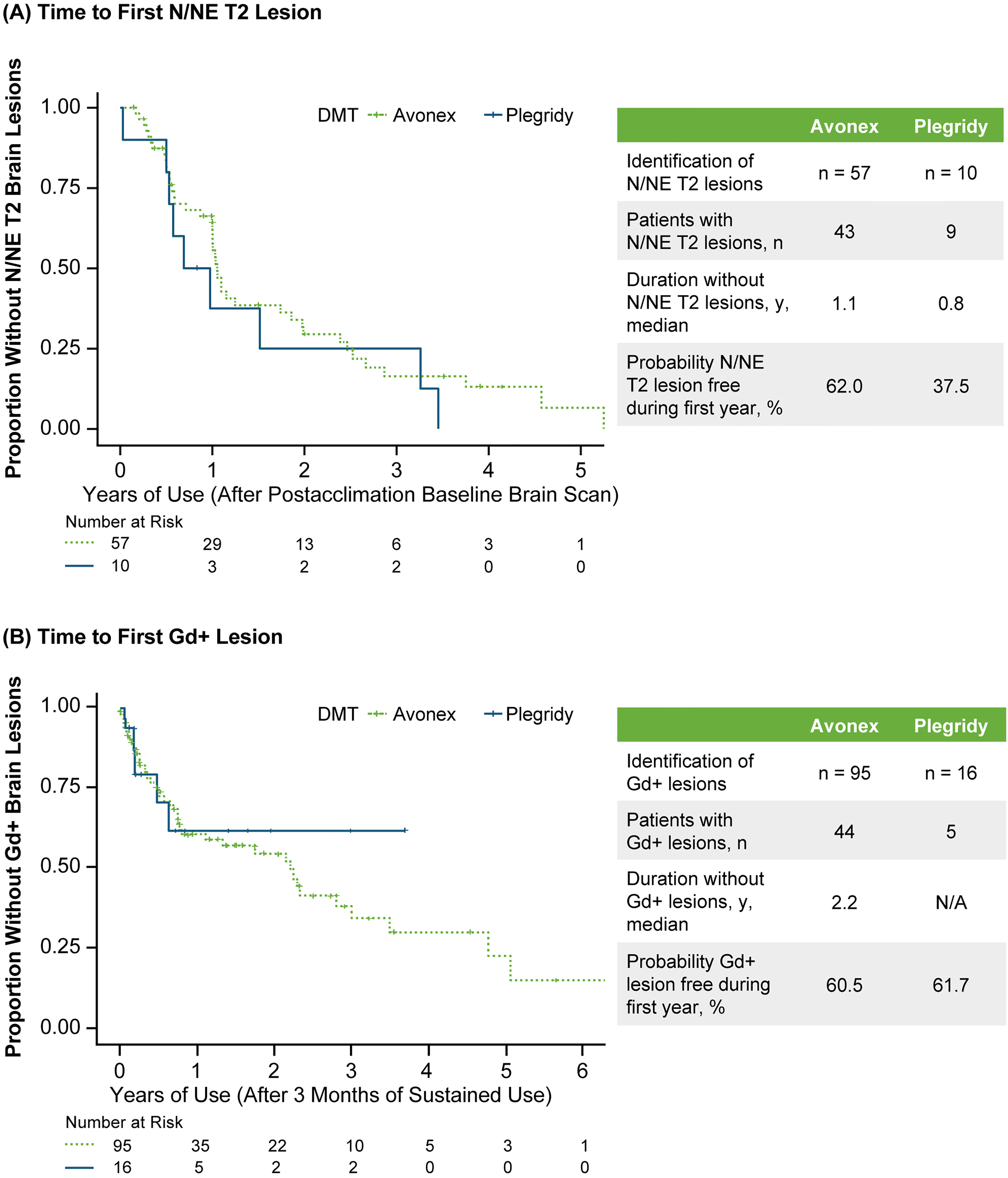
Kaplan-Meier Plots of Time to (A) First N/NE T2 Lesion and (B) First Gd+ Lesion in Patients on Avonex or Plegridy. (
Multiple postacclimation Expanded Disability Status Scale scores were available in 42 Avonex-treated patients and 12 Plegridy-treated patients. Eleven patients in the Avonex cohort and 6 patients in the Plegridy cohort had postacclimation Expanded Disability Status Scale progression. Of the patients with multiple postacclimation Expanded Disability Status Scale scores, the median number of years after the first postacclimation Expanded Disability Status Scale score to progression was 3.5 years for the Avonex group and 3.1 years for the Plegridy group (Figure 4).
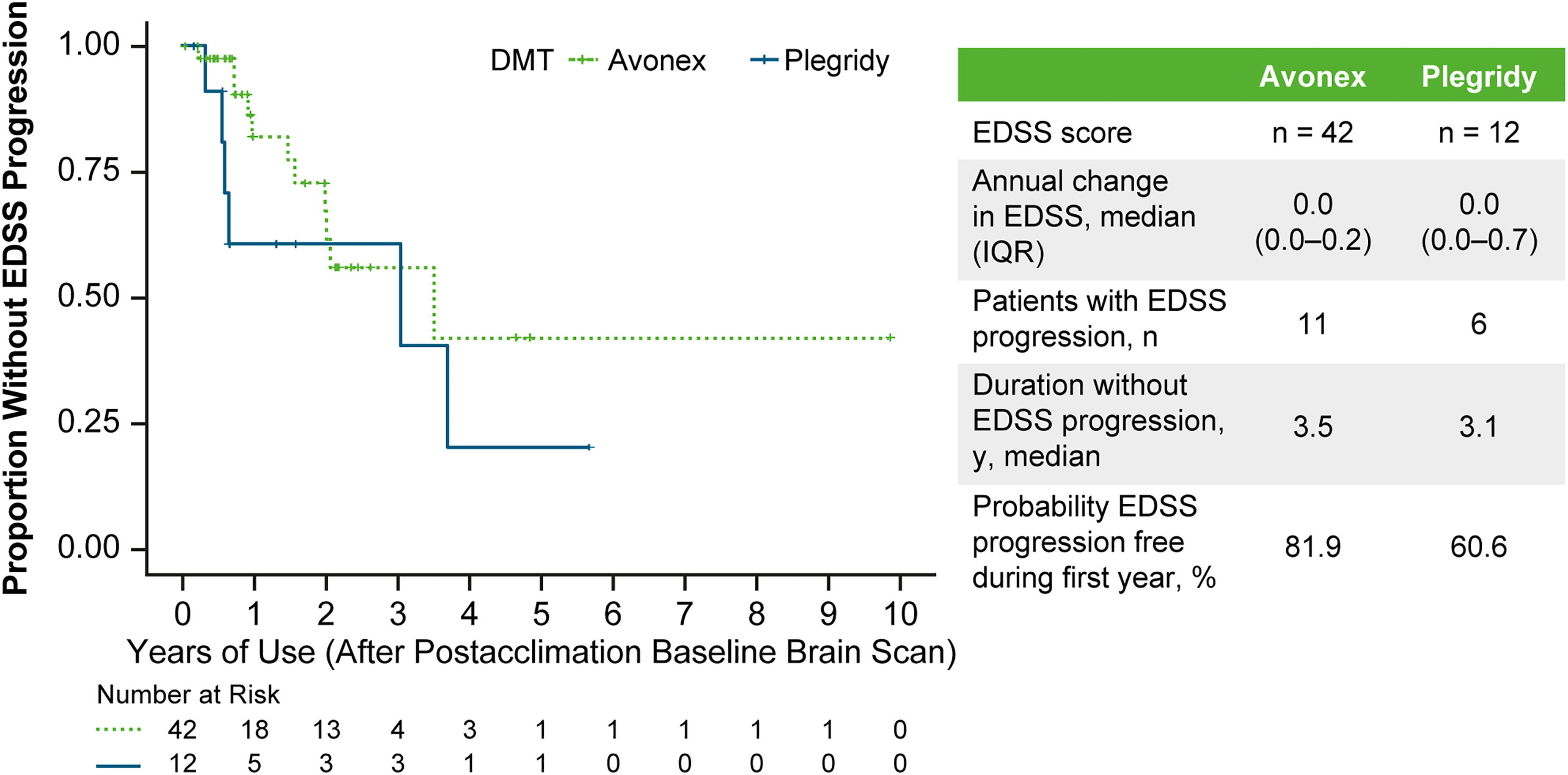
Kaplan-Meier Plot of Time to Disability Progression in Patients on Avonex or Plegridy. Abbreviations: DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; IQR, interquartile range.
Discussion
We present a large, retrospective study assessing the efficacy of Avonex and Plegridy for pediatric multiple sclerosis. Estimates of patients who would be relapse-free 1 year after acclimation to these disease-modifying therapies was nearly 70% for both cohorts. The probability of having no Gd+ lesions in the first postacclimation year of treatment was around 60%. This is similar to clinical outcomes reported in prior studies of interferon beta therapies approved for use in the adult population. Our observations also align with previously published data on SC interferon beta-1a in pediatric populations, while specifically examining Avonex (designed for IM use) and peginterferon beta-1a (Plegridy) in a US registry. 11
In patients with a relapse within the first 90 days of starting treatment, median duration from the end of the acclimation period to next relapse was >2 years shorter than patients without a relapse during the acclimation period, which suggests these patients with early relapse may have overall higher disease activity compared to those without relapse during acclimation. We found patients had a longer median duration until clinical relapse as compared to duration until radiographic changes, highlighting the need for radiographic monitoring to ensure silent lesions are not occurring in the absence of clinical relapse and guide treatment efficacy while on these medications. Although the majority of the cohort was no longer on interferon beta treatments at the time of the data snapshot, some patients had long durations of use. This study was not designed to investigate whether termination was due to side effects, patient preference, relapse, radiographic progression, or a combination of factors; however, we note that side effects appeared to be more commonly reported as a reason for stopping Plegridy than for stopping Avonex.
In pediatric patients, there is evolving evidence suggesting that the use of newer disease-modifying therapies results in fewer relapses compared to injectables. 16 However, some are not without risk of complications related to immunosuppression, particularly higher risk of infections and hypogammaglobulinemia that has been seen with B-cell depleting therapies as compared to interferons, and long-term safety data are needed in pediatric patients.34–37 Although these long-term data are needed, increasingly, higher efficacy therapies are favored as first-line therapy in pediatric multiple sclerosis. Families may initially show a preference toward options with lower risk of immunosuppressive complications, and it is essential that providers are able to have thorough discussions regarding comparative efficacy of various disease-modifying therapies when having these conversations with families. Patients may have underlying conditions such as hypogammaglobulinemia, immune dysregulation disorders, or cytopenias in which certain disease-modifying therapy risk/side effect profiles are preferred, or those who develop rare but challenging complications from higher-efficacy disease-modifying therapy, necessitating a switch to injectables. These treatment decisions remain complex, and issues regarding both tolerability and compliance need to be considered. In making decisions regarding therapy, it is essential to understand side effects, safety, and efficacy of off-label disease-modifying therapy use in children. Having these data available allows for informed and shared decision-making between clinicians, caregivers, and patients.
We acknowledge several limitations to this study. The sample size of patients who received Plegridy was quite small, and outcome data in this group should thus be interpreted cautiously. Overall median duration of therapy in Avonex and especially Plegridy groups was relatively short, limiting interpretation of these outcomes. Safety and tolerability of Avonex and Plegridy in the cohorts were not investigated, as this was beyond the scope of the study. Despite lack of systematic safety data, the most common side effects reported from the first duration of Avonex use were “flu-like symptoms,” with “other” and “headache” being the next most common; for Plegridy these were “flulike symptoms” and “other.” Recommended laboratory monitoring for patients receiving Avonex or Plegridy includes periodic complete blood counts with differential, platelet counts, liver function tests, and thyroid function tests, with frequency guided by clinical judgment and patient history. Furthermore, this study did not analyze whether patients switched to high-efficacy therapies after stopping Avonex or Plegridy, which may impact the future annualized relapse rate and the absence of other comparisons should be considered when interpreting our results. Because our outcome summaries were predicated on having postacclimation data, interpretations may not generalize well to the experiences of those whose medication use was abbreviated, perhaps because of safety, tolerability, or efficacy concerns. Finally, because new “baselines” could be established for some outcomes and because all outcomes excluded the acclimation period, our results do not describe possible acute effects during acclimation.
Conclusions
Although interferon beta therapies are not currently approved for use in patients with pediatric multiple sclerosis, this study demonstrates that patients with pediatric multiple sclerosis may experience outcomes similar to those seen in adult populations when treated with Avonex or Plegridy. While there is an increasing trend toward upfront higher efficacy disease-modifying therapies at diagnosis of pediatric multiple sclerosis for improved outcomes, injectables are at times still utilized in the pediatric population, and in unique situations these may be preferred by clinicians and/or families due to other comorbidities. Understanding efficacy of disease-modifying therapies in pediatric multiple sclerosis is essential to making informed treatment decisions and thoroughly discussing risks and benefits of specific disease-modifying therapies with families.
Footnotes
ORCID iDs
Consent to Participate
Informed consent was obtained as required by each center's review board. 33
Ethical Considerations
The Pediatric Multiple Sclerosis and Other Demyelinating Diseases database was reviewed and approved by each participating center's institutional review board. 33
Author Contributions
MAW, BJB, MW, MV, TCC contributed to study design. All authors contributed to manuscript preparation and/or critical review or revision of the manuscript. AA, EW, YW, MR, JMT, LK, MG, LB, TL, NS, SM, TC, TS, contributed to data collection. MAW, BB, SP, MW, TCC contributed to data analysis. All authors reviewed and approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Biogen (Cambridge, MA, USA). Editorial support for the preparation of this manuscript was provided by Excel Scientific Solutions (Fairfield, CT, USA): funding was provided by Biogen.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MAW: research funding to institution from Biogen. BJB: funding to institution (received or pending) from Biogen, HRSA, Intermountain HealthCare, Johns Hopkins, National MS Society, NIH, Owlet, and members of the Four Corners Youth Consortium. SP, MW, JR, MRensel, AA, EW, YW, MRodriguez, JMT, TL, NS, and TCC: no conflicts of interest to disclose. LK: consultant for Biogen, Bristol Myers Squibb, Celgene, F. Hoffmann-La Roche, and MCIC Vermont; and receives royalty payments for the Fatigue Severity Scale. MG: research funding to institution for clinical trial site participation from Roche Genentech; advisory board for Arialys Therapeutics. LB: research funding from NIH, Roche, ROHHAD Fight, and Rosamund Stone Zander Translational Neuroscience Center; speaker honorarium from Novartis; consultant to Massachusetts Department of Public Health and the National Vaccine Injury Compensation Program. SM: site PI for clinical trials with Roche and Biogen. TS: consultant for CDC; scientific advisory board or data safety monitoring board for Biogen; research support from Roche Genentech. MV: employee of and holds stock/stock options in Biogen. CC: research funding to his institution from Biogen, F. Hoffman-La Roche, and UCB.
Data Availability
Individual participant data collected during the trial may be shared after anonymization and on approval of the research proposal. Biogen commits to sharing patient-level data, study-level data, CSRs, and protocols with qualified scientific researchers who provide a methodologically sound proposal. Biogen reviews all data requests internally based on the review criteria and in accordance with our Clinical Trial Transparency and Data Sharing Policy. Deidentified data and documents will be shared under agreements that further protect against participant reidentification. To request access to data, please contact pedms@hsc.utah.edu.
