Abstract
Rett syndrome is a neurodevelopmental disorder that primarily affects females and is often associated with sleep problems. To date, few studies have examined how sleep problems correspond to aspects of clinical severity in Rett syndrome. In this study, we examined how sleep architecture and sleep-disordered breathing correspond to psychometrically validated outcome measures that are being used in clinical trials. Thirteen participants (mean [M] = 5.96 years, standard deviation [SD] = 2.6), all with confirmed pathogenic variants in
Keywords
Rett syndrome (MIM 312750) is a severe neurodevelopmental disorder that primarily affects females and, in most cases, caused by loss of function variants in the X-linked gene Methyl CpG-binding Protein 2 (
Early studies of Rett syndrome described breathing abnormalities as being more prevalent during the daytime,9–11 including alternating hyperpnea followed by apnea, air swallowing, and/or Valsalva maneuvers. Recent studies using polysomnography, however, have shown that sleep-disordered breathing as well as changes to sleep architecture are also highly prevalent.12–15 Both obstructive sleep apnea and central sleep apnea (associated with hypoventilation) are common12,13 in Rett syndrome in non–rapid eye movement (non-REM) and rapid eye movement (REM) sleep, with the severity of apnea events being greater in REM sleep. 12 Hypoxemia throughout nocturnal sleep in Rett syndrome 12 is common, as well as increased limb movements during sleep. A recent study also showed that REM sleep is attenuated in Rett syndrome. 15 In terms of sleep macrostructure, individuals with Rett syndrome have decreased total sleep time, increased sleep latency, increased wake after sleep onset, and reduced sleep efficiency.6,12
There are very few studies to date that have examined how sleep problems correspond to aspects of clinical severity in Rett syndrome. One recent study examined the relationship between sleep architecture and discrete clinical features (hand function, epilepsy, sitting, walking, spoken language, scoliosis) and did not find any relationships except for increased stage 1 non-REM (N1) sleep corresponding to greater hand dysfunction. 14 This same study did not find any relationships between clinical criteria and those with or without sleep-disordered breathing. Another study used a subset of items from the Rett Syndrome Behavior Questionnaire 16 to examine how social profiles corresponded to sleep abnormalities in Rett syndrome and found that longer sleep onset latency corresponded to increased social impairment. 17 In contrast, a recent study that used parent-reported sleep questionnaires showed that sleep difficulties, autonomic dysfunction, and milder clinical severity are associated with higher levels of anxiety in Rett syndrome,18,19 suggesting adverse impacts of sleep problems on mental health as well as physical health. To summarize, sleep abnormalities are common in Rett syndrome across mutation subtypes and are prevalent throughout the life span but there are limited findings when examining how sleep problems relate to clinical severity. Some of this could be attributable, at least in part, to the outcome measures that were used when examining these relationships.
Clinical trials are underway in Rett syndrome, with many additional drugs in the development pipeline, and it is possible that these compounds could correct underlying sleep abnormalities.8,20,21 As part of these trials, there is a strong need to use psychometrically well-validated outcome measures that capture aspects of clinical severity in Rett syndrome, including how these measures relate to sleep problems. A commonly used clinical outcome assessment in Rett syndrome is the Motor Behavioral Assessment, which recently underwent a revision. 22 The Motor Behavioral Assessment is a clinician-reported outcome measure that has been expanded and recently revised to better define clinical features in Rett syndrome including gross and fine motor skills, respiratory features, social and communication skills, functional/adaptive skills (eg, feeding difficulties, toiler training), and seizures. 22 The Revised Motor Behavioral Assessment is conducted during a daytime, in-person clinical visit. The most recent version (Revised Motor Behavioral Assessment) was revised using data from the Rett syndrome natural history study of 1075 individuals. The Rett Syndrome Behavior Questionnaire is a very commonly used parent-reported outcome measure in current clinical trials. 8 The purpose of this present study was to extend existing work related to sleep abnormalities in Rett syndrome and examine how sleep architecture and sleep-disordered breathing correspond to these commonly used and psychometrically validated outcome measures. We specifically examine how aspects of sleep correspond to clinical severity using these outcomes, and present comparisons of those with and without sleep-disordered breathing. Given previous studies that have shown decreases in REM sleep, and the importance of REM sleep for learning and behavior, 23 we hypothesized that those with more limited REM sleep may show increased clinical severity on both the clinician-reported and the parent-reported outcomes.
Methods
Demographics
Participants were recruited from Rett syndrome clinics as well as through announcements from a patient advocacy group. Thirteen female participants who met the clinical diagnostic criteria for Rett syndrome
2
and had loss of function pathogenic genetic variants in
Clinical Evaluation
All participants received a detailed evaluation from a child neurologist and a clinical psychologist. Evaluations were typically conducted either the same day or within 1 week of polysomnography. A child neurologist (CF) assessed clinical severity using the Revised Motor Behavioral Assessment. The Revised Motor Behavioral Assessment was created using psychometric methods and is a 24-item clinician-reported outcome measure that has been used to assess children, adolescents, and adults with Rett syndrome 22 in clinical trials as well as clinical visits. Items are captured on a 5-point Likert scale with a total score, and 21 of the 24 items that load onto the following 5 scales that were derived using factor analysis: motor dysfunction (eg, bradykinesia, dystonia, scoliosis, hypertonia/rigidity), functional skills (eg, hand clumsiness, motor skills, communication skills, feeding difficulties, etc), social skills (poor eye gaze, lack of social interest, etc), aberrant behavior (self-injury, aggression, etc), and Rett-specific behaviors (eg, bruxism, breathing issues). The 3 remaining items are retained from the original Motor Behavioral Assessment for clinical relevance. Higher total scores indicate greater disease severity. Caregivers completed the Rett Syndrome Behavior Questionnaire, a caregiver-reported questionnaire made up of 45 items assessing a variety of behavioral and clinical features found in Rett syndrome, each scored on a 3-point Likert scale. Scoring the Rett Syndrome Behavior Questionnaire produces a total score as well as 8 subscales: general mood, breathing problems, hand behaviors, repetitive face movements, body rocking and expressionless face, nighttime behaviors, fear/anxiety, and walking/standing. Higher Rett Syndrome Behavior Questionnaire total scores and subscale scores indicate greater severity. Recently, the total Rett Syndrome Behavior Questionnaire has been shown to be sensitive to change within the context of a clinical trial. 8
Sleep Recording
Sleep was evaluated for each participant using overnight clinical polysomnography. A standard polysomnography protocol was used using the Nihon Kohden Polysmith Sleep System with monitoring of respiratory effort, capnometry sensor nasal pressure transducer and airflow sensor, blood oxygen saturation, heart rate, EEG, and video recording. Analysis and interpretation were performed by a board-certified sleep medicine specialist according to the American Academy of Sleep Medicine criteria. 24 Standard parameters for sleep duration included total sleep time, sleep onset latency, wake after sleep onset, and sleep efficiency. Sleep stages included: N1, N2, N3, non-REM, and REM sleep. The Pediatric Apnea Hypopnea Index is reported using the American Academy of Sleep Medicine definition for hypopnea 24 —that is, scored when the nasal pressure transducer or alternative signal (thermistor and/or SUM signal) excursions drop by 30% to 90% compared with baseline and for ≥90% of the respiratory event (defined as 2 missed breaths) and is associated with either a ≥3% desaturation from the preevent baseline or an EEG arousal. Obstructive sleep apnea in children is defined as an Apnea Hypopnea Index score of ≥1 event/hour with signs and symptoms of sleep-disordered breathing (snoring, witnessed apneas). In addition, a pattern of obstructive hypoventilation, with 25% or more of total sleep time with hypercapnia, a partial pressure of carbon dioxide in arterial blood >50 mm Hg associated with snoring, flattening of inspiratory nasal pressure waveform, or paradoxical thoracoabdominal motion is diagnostic of obstructive sleep apnea. Three degrees of obstructive sleep apnea severity are identified according to the Apnea Hypopnea Index: mild, 1-4; moderate, 5-9; severe, ≥ 10.
Statistical Analysis
Analyses were conducted using the Statistical Package for the Social Sciences (SPSS), version 29. Descriptive data (mean [M] ± standard deviations [SDs]) are presented for all sleep parameters. Spearman correlations were used to examine the relationships between all sleep parameters, the Revised Motor Behavioral Assessment, and the Rett Syndrome Behavior Questionnaire respectively. To examine differences between those with and without obstructive sleep apnea, data were log-transformed to minimize variability (given the small sample size), and data were tested for equality of variances.
Results
Participant Characteristics
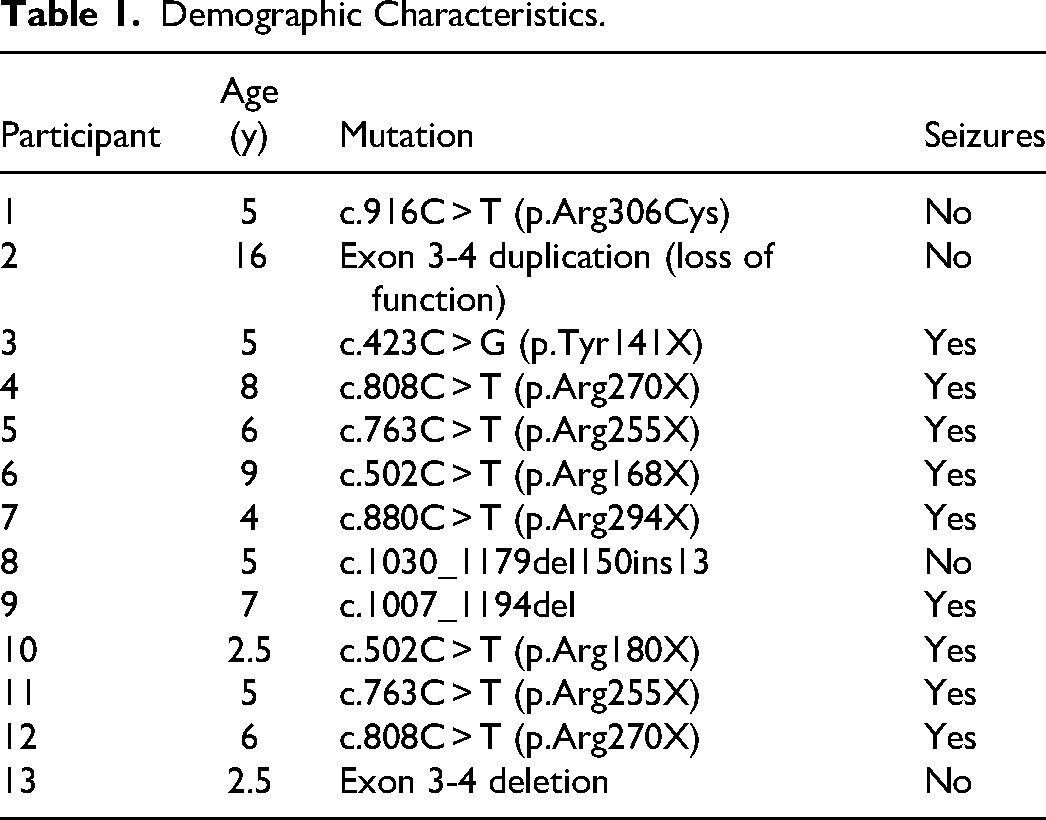
All participants in this study lived at home, and a primary caregiver accompanied them to the evaluation and completed questionnaires. Participants ranged in age from 2½ to 16 years (M = 5.96 years, SD = 2.6). Eleven of 13 participants were non-Hispanic White, 1 was Latino and White, and 1 was mixed race (African American and White). Eight participants had nonsense mutations, 1 had a missense mutation, 3 had small deletions, and 1 had an intragenic duplication of exons 3 and 4 causing loss of function (Table 1). Nine of the 13 participants had seizures (Table 1).
Demographic Characteristics.
Polysomnography
The mean total sleep time was 410.62 minutes (SD = 62.83), the mean sleep onset latency was 15.38 minutes (SD = 21.71), the mean wake after sleep onset was 59.54 minutes (SD = 51.65), and the mean sleep efficiency was 84.38 (SD = 11.01). In terms of sleep staging, mean N1 sleep was 8.92 minutes (SD = 13.48), mean N2 was 227.92 minutes (SD = 122), mean N3 was 135.54 minutes (SD = 130.66), and mean REM sleep was 37.77 minutes (SD = 37.32). Some participants had little to no REM sleep. No participants were observed having seizures during polysomnography. There were no aspects of sleep architecture that were related to age. Detailed results from polysomnography are shown in Supplementary Table 1 for each participant.
For sleep respiratory parameters, 6 of the 13 participants were diagnosed with obstructive sleep apnea. Those with obstructive sleep apnea were slightly younger (M = 5.08 years, SD = 1.74 years) compared with those without obstructive sleep apnea (M = 7.21 years, SD = 4.39 years) although the difference was not statistically significant (
Differences Between Those With and Without Obstructive Sleep Apnea for Sleep Parameters.
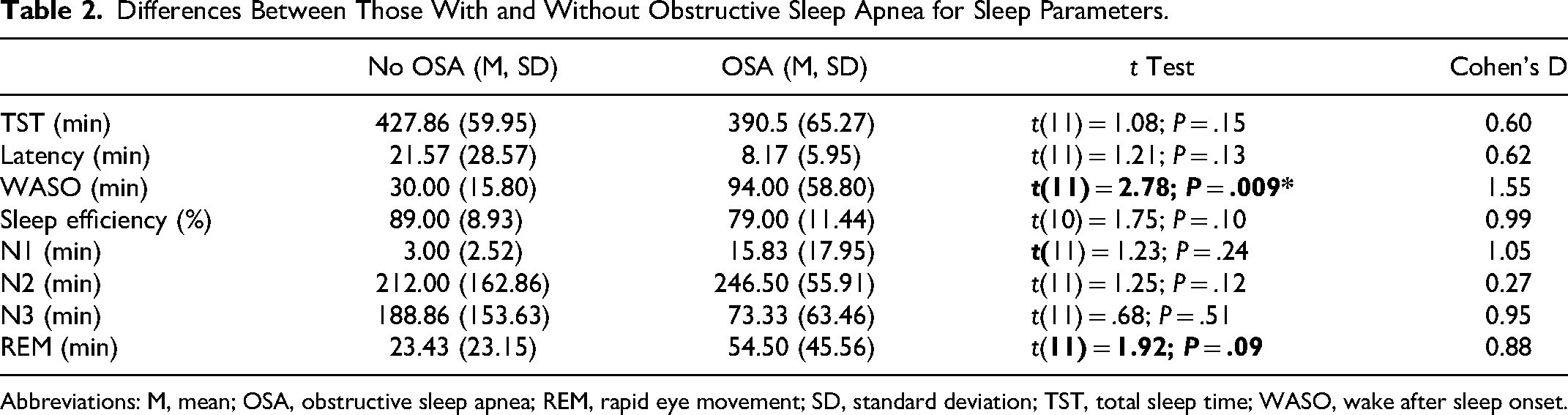
Abbreviations: M, mean; OSA, obstructive sleep apnea; REM, rapid eye movement; SD, standard deviation; TST, total sleep time; WASO, wake after sleep onset.
Correlations with Clinical Parameters
Correlations between sleep architecture parameters and the total and subscale scores of the Revised Motor Behavioral Assessment were examined. Revised Motor Behavioral Assessment total and subscale scores for each participant ranged from 7 to 48 (M = 21.20; SD = 13.11), with higher scores corresponding to increased severity (Supplementary Table 3 shows individual Revised Motor Behavioral Assessment scores for each participant). Decreased REM sleep time significantly correlated with increased overall clinical severity as assessed by the total Revised Motor Behavioral Assessment score, as well as increased severity on the Revised Motor Behavioral Assessment subscales Functional Skills and Social Skills (Table 3). Figure 1 shows a scatterplot of Total Revised Motor Behavioral Assessment scores by REM Sleep. Additionally, decreased REM sleep time showed a trend towards correlation with worsening Revised Motor Behavioral Assessment Respiratory Behaviors. There were no other correlations between aspects of sleep architecture and the Revised Motor Behavioral Assessment or Revised Motor Behavioral Assessment subscales. Furthermore, no correlations were observed between sleep respiratory parameters (Apnea Hypopnea Index, the Oxygen Desaturation Index (ODI), and percentage of continuous oxygen saturation) and the Revised Motor Behavioral Assessment (Table 3). A trend was noted for having increased respiratory behaviors as measured on the Revised Motor Behavioral Assessment and a lower level of continuous oxygen saturation.
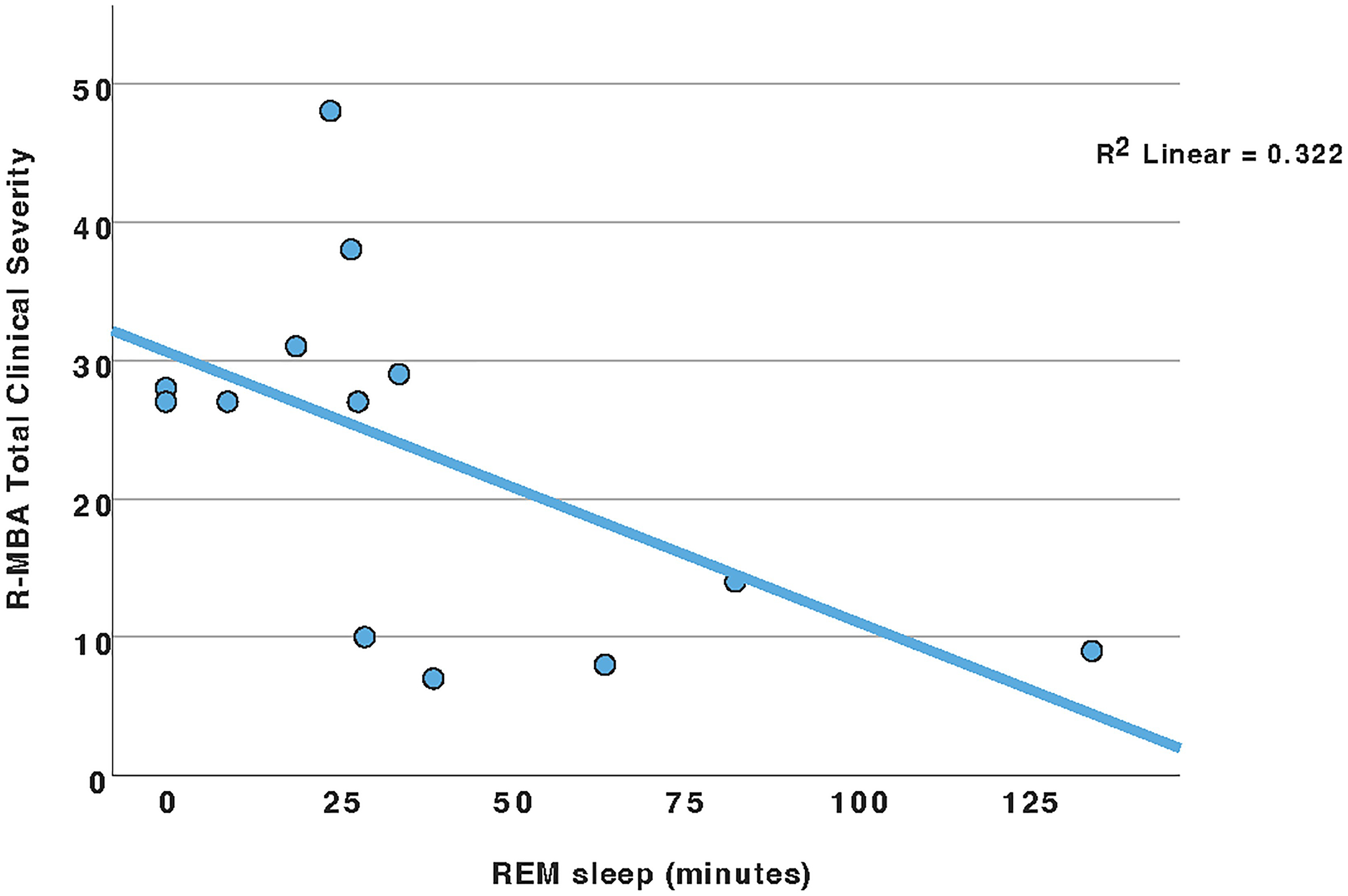
Revised motor behavioral inventory total score by REM sleep (minutes).
Correlations Between the Revised Motor Behavioral Inventory and Sleep Parameters.
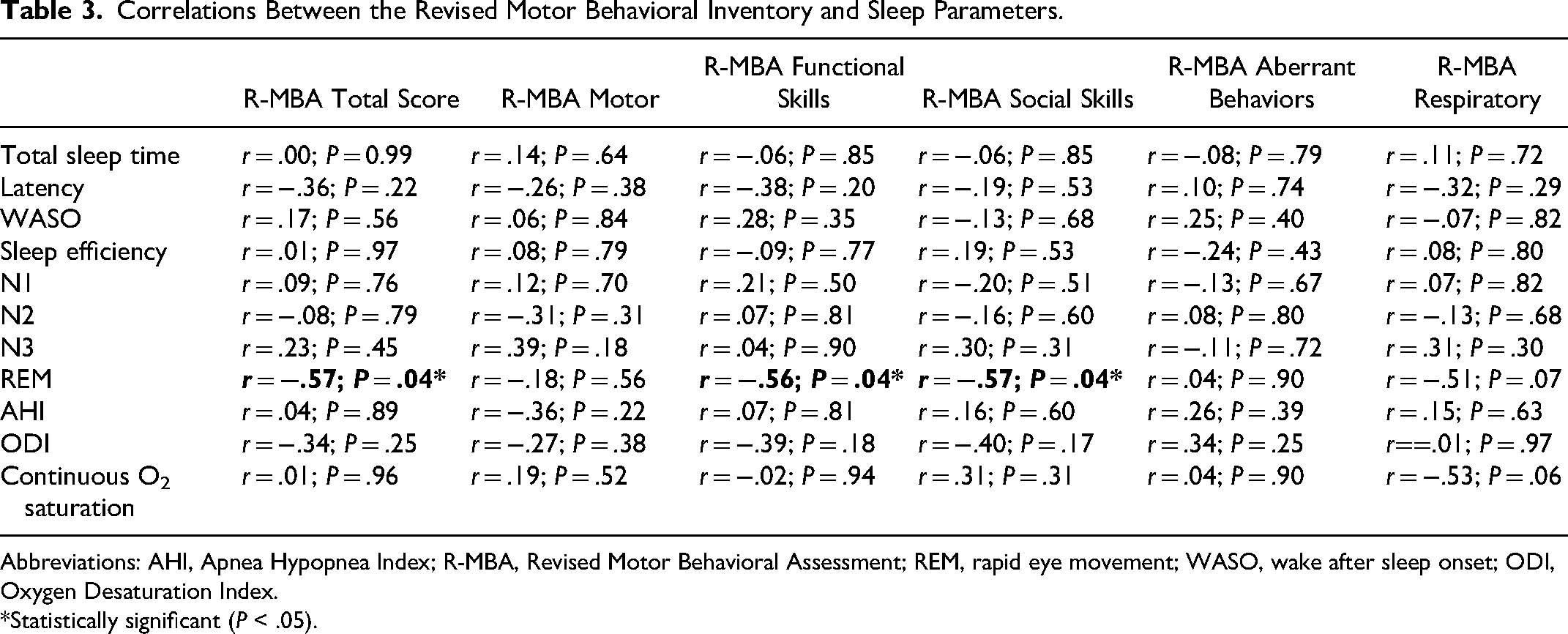
Abbreviations: AHI, Apnea Hypopnea Index; R-MBA, Revised Motor Behavioral Assessment; REM, rapid eye movement; WASO, wake after sleep onset; ODI, Oxygen Desaturation Index.
Statistically significant (
When examining correlations between sleep architecture parameters and the Rett Syndrome Behavior Questionnaire (Table 4), increased wake after sleep onset was associated with greater severity in Rett Syndrome Behavior Questionnaire nighttime behaviors. Increased sleep onset latency was associated with more hand behaviors. For sleep respiratory parameters, a higher Oxygen Desaturation Index was associated with significantly higher anxiety on the Rett Syndrome Behavior Questionnaire. Also, a higher Apnea Hypopnea Index was associated with more repetitive face movements on the Rett Syndrome Behavior Questionnaire.
Correlations Between Sleep Parameters and Rett Syndrome Behavior Questionnaire Scores.

Abbreviations: AHI, Apnea Hypopnea Index; ODI, Oxygen Desaturation Index; REM, rapid eye movement; RSBQ, Rett Syndrome Behavior Questionnaire; WASO, wake after sleep onset.
Statistically significant (
Given the smaller number of participants,
Comparisons of Those With and Without OSA on the R-MBA.
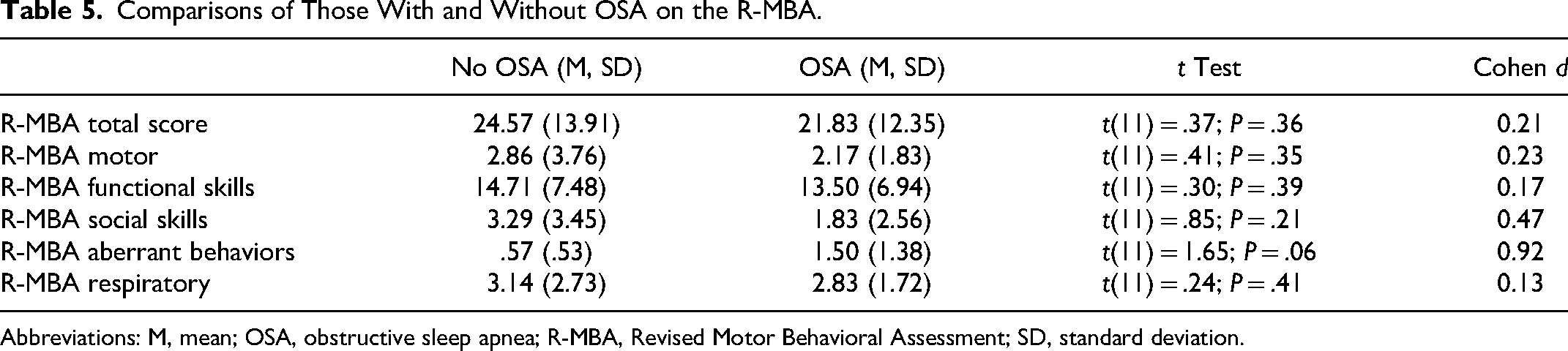
Abbreviations: M, mean; OSA, obstructive sleep apnea; R-MBA, Revised Motor Behavioral Assessment; SD, standard deviation.
Comparisons of Those With and Without OSA on the RSBQ.

Abbreviations: M, mean; OSA, obstructive sleep apnea; RSBQ, Rett Syndrome Behavior Questionnaire; SD, standard deviation.
Statistically significant (
Discussion
This study contributes to the existing literature regarding sleep in Rett syndrome by not only showing that disruptions to sleep architecture and sleep-disordered breathing are common but that there are clinically meaningful correlations with the most used clinician- and caregiver-reported outcomes being used in clinical trials. In terms of sleep architecture, our findings show that consistent with other studies,6,14 there is reduced REM sleep in Rett syndrome. Reductions in REM sleep are not uncommon in children with developmental disabilities, and REM sleep is important for learning, language-functioning, cognition, and processing of emotional information.23,25,26 Our study is the first to show that in Rett syndrome, reduced REM sleep is related to more impairment in functional skills, social skills, and higher overall clinical severity as captured by the Revised Motor Behavioral Assessment. As such, our findings suggest that as found in studies with typical populations, 23 participants with Rett syndrome who have more REM sleep have higher functional skills as measured during direct assessment, and better social interaction. Although a prior study in Rett syndrome did not find a relationship between discrete clinical features and REM sleep, 14 this may be attributable, at least in part, to a difference in the outcome measure being used and is worthy of further investigation in larger samples. There were no other significant relationships between aspects of sleep architecture and the Revised Motor Behavioral Assessment. Regarding sleep-disordered breathing parameters (Apnea Hypopnea Index, Oxygen Desaturation Index, continuous oxygen saturation), there were no significant relationships with the subscales of the Revised Motor Behavioral Assessment, but there was a trend toward significance for nighttime level of continuous oxygen saturation and the Revised Motor Behavioral Assessment respiratory behaviors subscale (hyperventilation, breath holding). In future studies, it will be important to determine whether these daytime respiratory behaviors correspond to sleep-disordered breathing. One strategy would be to use wearable sensors to capture both day- and nighttime sleep-disordered breathing events, which would allow for continuous monitoring over a longer period as compared to a one-time clinical evaluation. 27
When examining correlations with the Rett Syndrome Behavior Questionnaire and aspects of sleep architecture, we found that increased wake after sleep onset was related to higher scores on Rett Syndrome Behavior Questionnaire subscale of Rett nighttime behaviors (eg, laughing, crying, and/or screaming at night), a finding that is consistent with other studies that have reported nighttime behavioral disturbances in Rett syndrome18,28 that result in periods of wakefulness. We also found a relationship between latency and the Rett Syndrome Behavior Questionnaire subscale of Rett hand behaviors (not using hands purposefully, repetitive hand movements, etc) such that those who have fewer impairments (i.e., better hand skills) took longer to fall asleep. Other studies have shown that individuals with Rett syndrome who have fewer impairments are more likely to exhibit challenging behaviors, 19 which also can encompass increased difficulties with sleep as noted in one recent study that found, using some similar Rett Syndrome Behavior Questionnaire items that longer sleep onset latency correlated with increased social impairment. 17 Regarding sleep-disordered breathing, we found that a higher Oxygen Desaturation Index corresponded to higher Anxiety on the Rett Syndrome Behavior Questionnaire, mirroring findings that have been noted in children who are typically developing. 29
Almost half of the participants in our sample had obstructive sleep apnea, and obstructive sleep apnea was not related to age. These findings support other studies12,13 that have shown that breathing difficulties in Rett syndrome are not solely attributable to central apneas or hypopneas, or daytime breath holding. We found that those with obstructive sleep apnea had more difficulties during REM sleep with noted disruptions to sleep architecture including increased wake after sleep onset compared with those without obstructive sleep apnea. Although prior studies have been inconclusive and/or have shown mixed results regarding the impact of obstructive sleep apnea on sleep architecture, our results are consistent with findings from other recent studies that show that children with obstructive sleep apnea exhibit greater sleep fragmentation and spend more time in lighter stages of sleep as compared to those without obstructive sleep apnea.30,31 With the Rett Syndrome Behavior Questionnaire, we found that those with obstructive sleep apnea had more problems with mood, anxiety, nighttime behaviors, and repetitive face movements. These findings related to obstructive sleep apnea mirror those in studies of typically developing children, as well as those with Down syndrome and autism that show that obstructive sleep apnea is related to increased behavioral difficulties.32,33 When examining comparisons between those with and without obstructive sleep apnea on clinical outcome measures, we did not find any significant differences on the Revised Motor Behavioral Assessment. There was a trend toward significance for those with obstructive sleep apnea having a higher incidence of aberrant behaviors on the Revised Motor Behavioral Assessment. Although we hypothesized that those with obstructive sleep apnea would have lower functional skills, this was not the case. This could be attributable to the very small sample size within each group, but also could reflect that those with obstructive sleep apnea were slightly younger, and younger participants tend to have lower scores (less severity) on the Revised Motor Behavioral Assessment. 22 In addition, the Revised Motor Behavioral Assessment may not be sensitive enough to detect underlying processing differences between those with and without obstructive sleep apnea. It will be important to examine differences between those with and without obstructive sleep apnea using other standardized assessment tools and incorporate assessments of auditory evoked potentials, a method that has been used in recent Rett syndrome studies, especially given that some studies in typical children suggest that obstructive sleep apnea adversely impacts auditory processing.34,35 Taken together, our findings suggest the need for more broadly screening for obstructive sleep apnea in Rett, especially in those who may be waking frequently at night. In addition, those individuals with Rett syndrome who have obstructive sleep apnea may benefit from closer monitoring of their behaviors, and appropriately targeted interventions to address any anxiety and/or mood disturbances.
Although not a focus of this study, the underlying etiology of sleep problems in Rett syndrome warrants further investigation. Studies in animal models to date show that highly fragmentated sleep and circadian rhythm alterations are common and could be implicated in the proper functioning of specific brain areas and neurotransmitters involved in the sleep-wake cycle.36,37 The possible mechanistic underpinnings of the sleep-wake cycle in Rett syndrome might be attributable to MeCP2 binding and transcriptionally activating the circadian clock genes, Per1 and Per2, 38 and that MeCP2 protein is highly expressed in the suprachiasmatic nucleus. 39 There is a general disorganization of the circadian system that suggests a role for MeCP2 in the circadian timing system and provide a possible mechanistic explanation for the sleep/wake disturbances observed in Rett syndrome patients.
The current study has some limitations that should be addressed in future work. First, the sample size was small and, as such, there was more limited statistical power. Although effect sizes are reported, comparisons could not be made across different genetic mutations that could confer differing severity regarding sleep problems.14,18 Future studies should include larger samples to determine whether similar relationships will be found between clinical outcomes, sleep macrostructure, and sleep-disordered breathing. Almost all the participants in this study had seizures, and seizure medications can have an impact on sleep architecture. 40 Also, some studies have suggested that obtaining 1 night of polysomnography is not sufficient because of concerns about adaptation effects, which primarily affect sleep architecture but can be less significant for respiratory parameters and a diagnosis of obstructive sleep apnea in children.41–44 Nonetheless, given the challenges associated with polysomnography (cost, accessibility, and sensory sensitivities), it will be important in future studies to determine more typical sleep over time using other novel methods of remote sleep assessment (eg, at-home sleep apnea testing, wearable sensors). 45 Wearable sensors are being used more in sleep research studies because of their sensitivity and ease of use, which have made them suitable for longitudinal monitoring of patients within their home environment and are beginning to be pilot-tested for assessing sleep in Rett syndrome.27,46
To summarize, our study demonstrates that changes to sleep architecture and sleep-disordered breathing are common in Rett syndrome and are associated with increased severity as captured using clinician- and parent-reported outcomes. Reduced REM sleep is very common in Rett syndrome and is associated with more impairments in functional and social skills. Obstructive sleep apnea is also very common, and is associated with changes to sleep architecture, higher anxiety, and more mood disturbances. Taken together, these findings add to the existing literature and suggest that screening for sleep problems and implementing targeted interventions can impact the quality of life of individuals with Rett syndrome and their families.
Supplemental Material
sj-docx-1-jcn-10.1177_08830738251347561 - Supplemental material for Sleep Problems and Clinical Severity in Rett Syndrome
Supplemental material, sj-docx-1-jcn-10.1177_08830738251347561 for Sleep Problems and Clinical Severity in Rett Syndrome by Sarika U. Peters, Cary Fu, Althea Robinson Shelton, Jeffrey L. Neul and Beth Malow in Journal of Child Neurology
Footnotes
Acknowledgments
We graciously acknowledge the children and families who participated in this study.
Ethical Approval
This study was approved by the Institutional Review Board at Vanderbilt University Medical Center (IRB number 210217).
Consent to Participate
Informed consent was obtained from all participants.
Consent for Publication
Not Applicable
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Institutes of Health (NIH), National Center for Advancing Translational Sciences (grant number 1R21TR003942-01, to SUP).
Data Availability
Additional data will be made available on reasonable request
Supplemental Material
Supplemental material for this article is available online.
Author Contributions
This study was conceptualized by SUP, CF, & JLN. SUP, ARS, and BM were involved with methodology. SUP, CF, and ARS collected data. SUP, BM, and ARS analyzed the data. SUP drafted the manuscript. CF, JLN, ARS, and BM reviewed and edited the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
