Abstract
Mucormycosis is a rare, life-threatening opportunistic infection primarily affecting immunocompromised patients. The available literature on Rhizomucor pusillus (R pusillus) infections of the central nervous system (CNS) in children is very limited. We present the case of an immunocompromised adolescent with intracranial mucormycosis due to R pusillus manifesting with stroke-like symptoms. This case highlights this rare condition's diagnostic complexity and management challenges. We include a review of the available literature on pediatric R pusillus CNS infections to increase awareness among health care providers.
Mucormycosis is a life-threatening infection caused by fungi in the order Mucorales, which includes several genera of pathogenic fungi, such as Rhizopus, Mucor, and Rhizomucor.1,2 CNS mucormycosis is rare but with increasing incidence. These fungi are primarily opportunistic pathogens, typically affecting immunocompromised individuals, particularly those with uncontrolled diabetes mellitus, malignancies, hematopoietic or solid-organ transplantation, and, most recently, COVID-19.2-4 It can be particularly aggressive, with high morbidity and mortality rates ranging up to 60%, even with timely diagnosis and treatment. 3 Rhizomucor species account for up to 6% of mucormycosis, causing predominantly pulmonary and disseminated infections and less commonly cutaneous, rhino-orbito-cerebral, and gastrointestinal infections.1,2,4
Although its occurrence in the CNS is rare, when present, it can lead to infarction, necrosis, and hemorrhage because of its angio-invasive nature. Such cases are exceedingly uncommon in pediatric patients, and limited literature is available to guide clinical decision making.
This case report describes an unusual presentation of a likely isolated intracranial Rhizomucor pusillus infection in an adolescent with T-cell acute lymphoblastic leukemia, initially diagnosed as an arterial ischemic stroke. We also review the literature on pediatric rhizomucor infection and present an overview of the pathophysiology, diagnosis, and treatment options available.
Case Report
A 16-year-old right-handed female adolescent with T-cell acute lymphoblastic leukemia in induction therapy presented to the clinic with scleral icterus, conjugated hyperbilirubinemia, and elevated aspartate transaminase, alanine transaminase, and International Normalized Ratio (INR). During the visit, she experienced weakness and a minor fall, leading to hospitalization for further evaluation and management of acute liver failure with coagulopathy.
Her most recent chemotherapeutic plan included dexamethasone, daunorubicin, and vincristine administered 1 week before admission, with earlier courses including pegasparginase and methotrexate. She was also receiving prophylactic trimethoprim-sulfamethoxazole, micafungin, and norethindrone.
On admission, she reported a 1-week history of bilateral lower extremity weakness and numbness in her toes. Four days into hospitalization, she developed acute numbness in her left fingertips, which rapidly progressed to involve her entire left hand, causing difficulty holding objects. Intermittent left-sided abdominal, back, and hip pain were also reported. These new neurologic symptoms prompted the activation of an acute stroke alert.
Physical examination revealed an afebrile, cachectic adolescent with mild scleral icterus. Neurologic examination showed that she was alert and oriented, with intact speech and language. Mild left nasolabial fold flattening was noted, along with left-sided pronator drift and an inability to shrug her left shoulder. The rest of the cranial nerve examination was unremarkable. Sensorimotor examination revealed weakness on left distal upper extremity flexion and extension and bilateral hip flexion. Sensation to light touch was reduced distally in the left upper extremity and bilateral lower extremities. Vibration sense was decreased in the bilateral feet extending to the ankle on the right and the knee on the left. Coordination and deep tendon reflexes were intact. The National Institutes of Health Stroke Scale (NIHSS) was 9 for facial palsy, extremity weakness, and sensation deficit.
Initial brain magnetic resonance imaging (MRI; Figure 1a) showed focal diffusion restriction in the right peri-rolandic region and right centrum semiovale, involving the precentral more than the postcentral gyrus, suggesting acute ischemia. No evidence of stenosis or occlusion was seen on arterial and venous vessel imaging of the head and neck. Spine MRI was notable for lumbar disc degeneration but was otherwise unremarkable. Furthermore, additional blood workup showed no significant abnormalities (Table 1). COVID-19 test was negative.
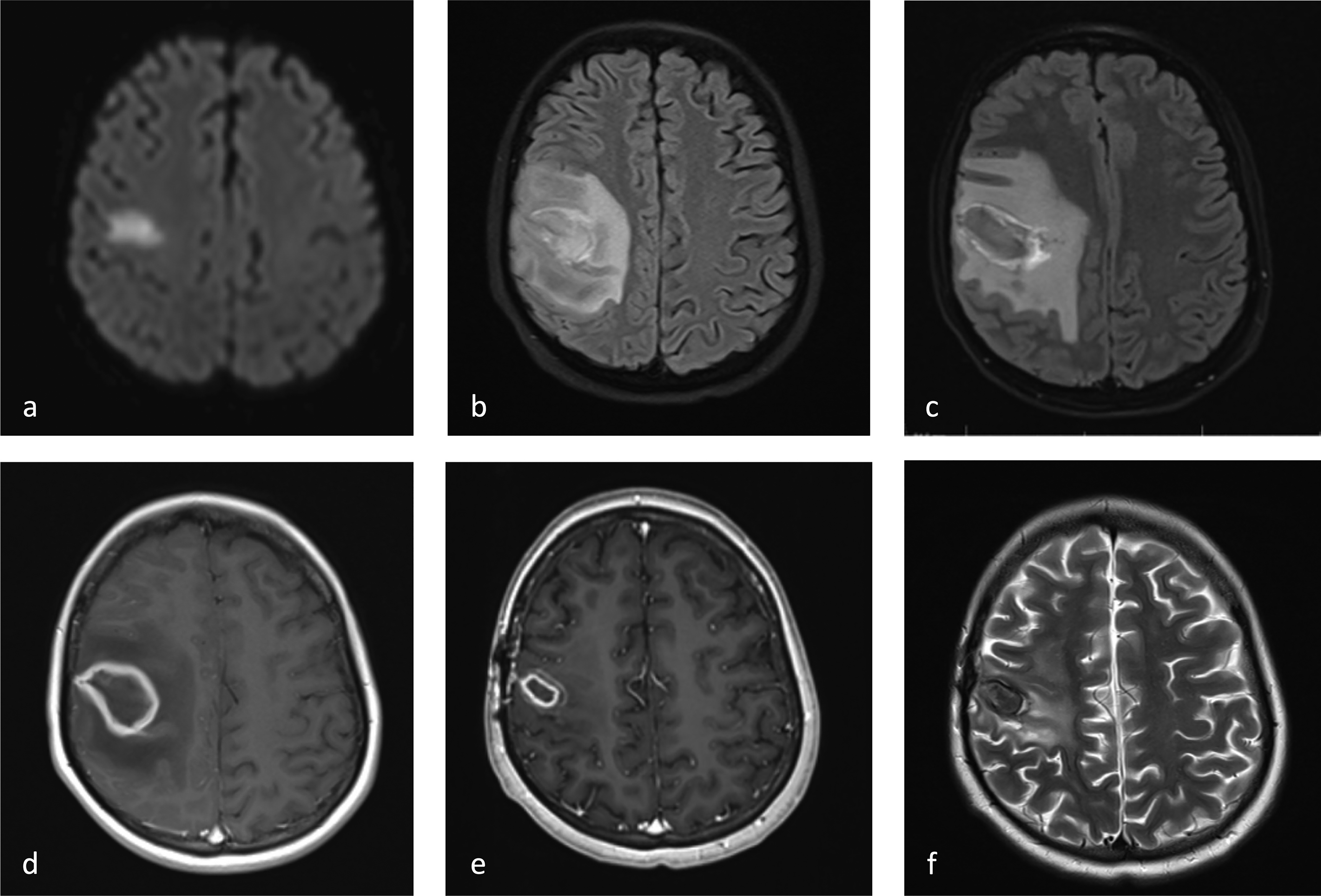
Sequential MRI changes over time. (a) Day 0: stroke MRI axial DWI showing restricted diffusion in the right perirolandic region and centrum semiovale, suggestive of acute ischemia at the time of presentation. (b) Day 13: axial FLAIR showing a small new collection at the prior suspected ischemia site with prominent surrounding vasogenic edema, concerning for hemorrhagic conversion. (c) Day 18: axial FLAIR demonstrating further increase in mass effect, vasogenic edema, and collection size, with a subtle developing rim of T2-weighted and FLAIR hypointensity. (d) Day 21: axial T1-weighted postcontrast showing enlarged lesion with thick rim enhancement, involving the cortex and subcortical white matter with central necrosis, vasogenic edema, mass effect with mild right-to-left midline shift. (e) Day 196: axial T1-weighted postcontrast and (f) axial T2-weighted showing postoperative contraction of the lesion with improvement in vasogenic edema and mass effect. DWI, diffusion-weighted imaging; FLAIR, fluid-attenuated inversion recovery; MRI, magnetic resonance imaging.
Relevant Clinical Test Results and Their Clinical Significance for the Patient's Evaluation.

Abbreviations: CSF, cerebrospinal fluid; PCR, polymerase chain reaction; RBC, red blood cells; WBC, white blood cell.
At this stage, the acute ischemia noted on imaging was attributed to the hypercoagulable state associated with asparaginase and coagulopathy in the setting of liver failure and malignancy. Her lower extremity sensorimotor symptoms were attributed to vincristine-induced peripheral neuropathy.
Over the next 2 days, her weakness progressed to involve the face and more prominent weakness of the left upper extremity. A repeat brain MRI showed an enlarged region of diffusion restriction with new associated petechial hemorrhages. Given her persistent thrombocytopenia and risk of hemorrhagic conversion, anticoagulation was deferred, and the patient was started on aspirin. Despite physiotherapy, the patient's neurologic status continued to decline, with worsening hip flexor weakness. This deterioration was attributed to her critical illness and steroid use.
On day 13 of hospitalization, she developed stuttering episodes and complete paresis of her entire left upper extremity. Brain MRI (Figure 1b) suggested hemorrhagic conversion within the previous area of diffusion restriction and extensive vasogenic edema, despite improved thrombocytopenia and coagulopathy. Interval CTA was stable. Electroencephalogram (EEG) was without epileptiform activity and accompanying lab work was unremarkable.
By day 18, the patient developed worsening right-sided headaches, but remained afebrile and hemodynamically stable. A repeat brain MRI (Figure 1c) demonstrated increased vasogenic edema surrounding the hemorrhage, with associated midline shift, prompting the initiation of dexamethasone.
On day 21, contrast-enhanced MRI (Figure 1d) revealed a large ring-enhancing lesion with progressive surrounding edema at the location of initial restricted diffusion. Notably, laboratory results, including CSF studies, did not indicate an active infectious process.
A diagnostic brain biopsy was performed, and necrosis was observed on tissue examination. Routine tissue cultures were negative for microorganisms. Universal polymerase chain reaction (PCR) for bacteria, fungi, and mycobacteria was positive for Moraxella porci. Plasma metagenomic next-generation sequencing for pathogen identification identified Acinetobacter haemolyticus at low levels. Antimicrobials were not initiated because these organisms were considered highly unlikely to be the cause of her brain lesion. Although her left upper extremity weakness continued, she could walk again with assistance, and this was seen as an improvement off antibiotics.
On day 35, the patient had a 2-minute generalized tonic-clonic seizure that resolved spontaneously. Imaging revealed an enlarged lesion, prompting empirical initiation of metronidazole and ceftriaxone. After 2 weeks of antibiotic therapy, there was no reduction in lesion size on imaging.
On day 53, she underwent an open brain biopsy. Multiple samples were taken, including fluid from the lesion and tissue from the leading edge of the lesion. Routine cultures and stains were again negative for microorganisms. PCR for bacteria and mycobacteria remained negative. Tissue examination showed nonspecific necrosis and granulation tissue. On day 61, universal fungal PCR identified R pusillus. Antifungal therapy was initiated with liposomal amphotericin B and posaconazole.
Initial MRI showed no evidence of sinus disease. However, at 3 weeks post-stroke, there was moderate mucosal thickening in the right frontal and right maxillary sinuses, with mild thickening in the bilateral ethmoid sinuses. These changes resolved spontaneously before antifungal treatment was initiated (please see supplementary material). The patient's clinical course was complicated by pancreatitis, which improved within days and was deemed unlikely infectious in etiology or related to the antifungal therapy. Chest computed tomography (CT) about 2 weeks into antifungal therapy showed left lingular consolidation, which raised concerns for fungal involvement and abnormal contrast enhancement around the central venous catheter, suggesting a possible thrombus. However, further investigations, including pathology, culture, and PCR of bronchoalveolar lavage samples, and echocardiogram did not suggest fungal infection or thrombus. This consolidation was not present on the chest radiograph 1 month prior to the stroke, and was almost completely resolved when CT chest was repeated 3 weeks later.
Liposomal amphotericin B was continued for 45 days, and posaconazole is being continued indefinitely at the time of this report. The patient continued to experience headaches, but her neurologic examination improved. Follow-up MRIs after day 80 (Figure 1e and f), demonstrated a collapsing abscess and reduced edema, along with new cystic changes in the right postcentral gyrus consistent with fungal disease.
Two years after the initial presentation, while in remission from acute lymphoblastic leukemia, the patient presented with a seizure and was found to have subtherapeutic levetiracetam and posaconazole levels. In the interim, she had developed steroid-induced diabetes mellitus. She subsequently underwent craniotomy for incision and drainage of 2 small adjacent abscesses, located slightly anterior to the original site. Pathology was consistent with an organizing abscess, and was negative for malignancy or fungal elements. Gram stain, cultures, and tissue PCR for bacteria, fungus, and mycobacteria remained negative. This was presumed to be a recurrence of her initial R pusillus infection related to her poorly controlled diabetes mellitus and subtherapeutic posaconazole levels. She received 6 additional weeks of amphotericin B and then continued long-term posaconazole therapy. Postsurgical MRI showed no residual abscess and decrease in surrounding edema.
Literature Review
Methods
A comprehensive literature search was conducted using the following electronic databases: PubMed, Embase, and CINAHL. The search covered all records from the database's inception to January 2024. The search strategy included keywords and Medical Subject Headings (MeSH) terms “rhizomucor pusillus” and “case report” or “case series.” No language or publication status restrictions were applied.
We included all studies in English or Spanish, cases of patients 18 years of age and younger who were diagnosed with intracranial R pusillus. We excluded conference abstracts and studies for which the full text was unavailable.
Two reviewers independently screened the titles and abstracts of all retrieved records. Full texts of potentially relevant articles were then assessed for eligibility based on the inclusion and exclusion criteria. Disagreements between reviewers were resolved through discussion.
The PRISMA flow diagram illustrates the study selection process (please see supplementary material for the selection process). Four studies met the inclusion criteria and were included in the systematic review.
Data were extracted using Excel, including information on study characteristics (author and year), participant demographics, diagnosis, intervention details, and clinical outcomes.
Results
Four articles met our inclusion criteria for our literature review, with 4 cases of intracranial R pusillus involving systemic pathology in pediatric patients (Table 2).5-8 Cases had disseminated disease, and we did not find any cases of isolated intracranial R pusillus in the pediatric literature. The identified cases presented with varied symptoms, including altered mental status, fever, hemiparesis, and seizures. Three of the 4 cases involved immunocompromised states because of genetic immunodeficiency, hematologic malignancies, chemotherapy, or immunosuppressive therapy. The fourth likely developed functional immunosuppression resulting from trauma, surgery, and corticosteroid use. Despite aggressive treatments, including antifungals and broad-spectrum antibiotics, the patients experienced significant morbidity, with mortality in 2 of the 4 cases due to rapid disease progression and multiorgan involvement. Diagnostics used across the cases included CT and MRI scans and blood cultures. In one of the surviving cases, decompressive craniectomy was performed. In those who survived, liposomal amphotericin B was used.
Literature Search Results: 4 Cases of Intracranial Rhizomucor pusillus in Pediatric Patients.
Abbreviations: CRP, C-reactive protein; CSF, cerebrospinal fluid; CT, computed tomography; HSCT, hematopoietic stem cell transplantation; PCR, polymerase chain reaction; Pre-B ALL, pre-B acute lymphoblastic leukemia; WBC, white blood cell.
Discussion
Summary
We describe a case of likely isolated intracranial R pusillus infection. Our patient presented with focal neurologic deficits, with features more suggestive of arterial ischemic stroke and absent signs and symptoms of intracranial infection. The disease progression was slow. The hemorrhagic component of the lesion was unusual for an ischemic stroke because it was noted 2 weeks after her initial symptoms. Moreover, the degree of vasogenic edema appeared out of proportion compared to the size of the hemorrhage. Her initial workup revealed low levels of A haemolyticus on plasma metagenomic next-generation sequencing and M porci on tissue bacterial PCR. The former is not known to cause intracranial abscesses, and the latter is not a known human pathogen, and neither organism grew from tissue cultures. Additionally, the patient was not neutropenic for at least 3 weeks before the initial presentation. Although needle biopsy was unrevealing, universal PCR tissue from an open biopsy detected R pusillus. The fungus did not grow on culture, clinical examinations showed no evidence of sinus involvement, and there was no evidence of its spreading from adjacent structures on repeated imaging, supporting that the infection did not arise from contiguous spread. Incidentally identified pulmonary consolidation presented a possible source of noncontiguous spread, but this is considered unlikely as chest imaging before her stroke did not show evidence of infection nor did she have respiratory symptoms or fever in this period to suggest infection, and bronchoalveolar lavage showed no evidence of pulmonary fungal infection. Although the overall clinical course was atypical for such an infection, treatment was started based on a risk-benefit evaluation and resulted in improvement of neurologic symptoms.
Mucormycosis-associated intracranial infection has been reported in adults and children. Notably, we did not find a case of isolated intracranial R pusillus mucormycosis in pediatric literature in our systematic review. Cases have been reported in immunocompromised adults.9-11 Farid et al 11 reported a case of isolated R pusillus, an immunocompromised adult with chronic lymphocytic leukemia and a history of stem cell transplant, presenting with a generalized tonic-clonic seizure and expressive aphasia. The patient was afebrile, and otherwise, his examination was noncontributory. The lesion was resected. R pusillus was identified on histopathology and RNA sequencing, for which the patient was treated with liposomal amphotericin B, followed by posaconazole with a good response.
Pathophysiology
R pusillus is thermophilic, allowing growth in febrile patients.1,12 This fungus is ubiquitous in the environment and can be airborne both outdoors and inside hospitals.1,12 Rhizomucor is transmitted through environmental exposure to fungal spores by inhalation, direct contact, or rarely ingestion. 12
Cerebral mucormycosis is commonly rhinosinogenic in origin, spreading by direct tissue invasion from adjacent infected tissues, usually the paranasal sinuses. This is particularly prevalent in diabetic patients, especially in the setting of diabetic ketoacidosis. Less often, infection occurs by hematogenous spread, typically in patients with hematological malignancies or recipients of stem cell transplants. Mucormycosis usually originates in the maxillary sinuses then spreads directly into adjacent tissue, including the periorbital tissues and cavernous sinus, which can cause cavernous sinus thrombosis. With perineural invasion, the fungus can extend into the brain parenchyma. The fungus in the sphenoid sinuses can spread to the cavernous sinus or infiltrate the carotid artery into the frontal and parietal lobes. Less likely infection can extend to the sagittal sinus, epidural and subdural spaces, and meninges.13,14 Orbital infections are also seen. 15 Isolated intracranial infections represent a less frequent but distinct category and are seen with intravenous drug abuse.4,13
Rhizomucor virulence is associated with angioinvasion, leading to blood vessel occlusion and tissue ischemia, infarction, and necrosis. The hyphal growth in blood vessel lumens damages the endothelium and forms fungal emboli, causing occlusion and infarction. The fungus can also extravasate to the brain parenchyma, forming an abscess and predisposing to strokelike symptoms. 10 The vascular complications, tissue inflammation, necrosis, and continued hyphal invasion manifest as headaches, sensory and motor deficits, cognitive disturbance, and seizures. Infection may also lead to coma and death. This hypoxic necrotic environment may impair immune cells’ access to the infection. 13 Challenges to hemostasis, notably disease-related factors such as elevated serum sodium chloride levels, hyperglycemia, and hyperferremia, create a conducive environment that facilitates fungal invasion and further compromises the immune response.13,16
Diagnosis
The diagnosis of intracranial mucormycosis is challenging, particularly in cases with atypical presentations like our patient. Signs and symptoms can be nonspecific, including headache, focal neurologic deficits, seizures, and altered mental status with or without signs of infection, occurring in both immunocompromised and even immunocompetent individuals.5-11 Cases of strokes secondary to CNS fungal infections are reported with a negative CSF profile. These commonly involved anterior circulation strokes and hemorrhagic conversion have been reported. 17
In our patient, CSF studies did not indicate CNS infection. Although pleocytosis in CSF is a common finding in CNS fungal infections, isolated abscesses, including fungal balls, are poorly evaluated with CSF analysis, particularly in the absence of leptomeningeal involvement.
18
Mucorales do not release 1,3-beta-
MRI may show a lesion with a T1-hyperintense rim with peripheral contrast enhancement and a T1-hypointense core in the fungal abscess. The abscess wall and projections show restricted diffusion on DWI with core sparing. Often, more than 1 abscess is seen. 21 Certain spectroscopic profiles on MRS can help support diagnoses of fungal abscesses, such as elevated amino acids, lactate, and lipids, with decreased N-acetyl-aspartate, choline, and creatine. 22
Our patient's initial brain MRI demonstrated restricted diffusion in a nonvascular distribution without an associated T1/T2 abnormal signal or mass effect, which is suspicious for ischemic stroke. Vascular imaging was not suggestive of vasculopathy, occlusion, or stenoses. We observed persistent restricted diffusion, which is atypical for a stroke, as the ADC signal typically trends toward facilitated diffusion within a week. Second, hemorrhagic conversion occurred 2 weeks after the initial presentation, which is delayed compared with the expected time frame of within the first few days. Third, the mass effect and vasogenic edema significantly increased over time, contrary to the typical expectation of a gradual decrease in these parameters following a stroke.23,24 Follow-up MRI without contrast (Figure 1b) showed the development of a collection, later with increasing mass effect and vasogenic edema (Figure 1c), which was further characterized in a subsequent brain MRI with contrast on day 21, demonstrating a thick layer of peripheral enhancement, surrounding vasogenic edema and trace peripheral rim of susceptibility artifact (Figure 1d). There was no evidence of interval rebleeding or evolution of blood products. The smooth peripheral rim was more suggestive of a developing abscess capsule. The imaging findings over time could be summarized as an initial focus of microvascular injury and ischemia, eventually developing a cerebral abscess. Posttreatment imaging showed a further thickened, collapsed abscess capsule (Figure 1e and f) with postprocedure changes and no definite residual purulent material. Of note, the initial MRI did not demonstrate sinus involvement. However, at the time of hemorrhagic conversion, there was opacification of the paranasal sinuses, which varied in degree over time and resolved completely before the initiation of antifungal therapy.
Treatment
Amphotericin B is the drug of choice for initial management of mucormycosis, usually as a liposomal formulation. Amphotericin concentrations in CSF tend to be low as it does not readily cross the blood-brain barrier, and fungal vascular occlusion may inhibit the drug from reaching the target tissue. However, some animal models suggest it may have good brain tissue penetration when tissue is inflamed. The antifungal azoles posaconazole and isavuconazole generally retain in vitro activity against the mucorales and are commonly used as salvage or step-down therapy, or in combination with amphotericin B, but this practice has not been adequately investigated. Treatment duration is case-dependent and usually lasts months, and depending on the patient's immune status, may be continued indefinitely to prevent relapse of infection, as seen in our patient. Medical treatment is usually combined with surgical debridement, which has been shown to improve survival.10,13,25
Conclusion
R pusillus mucormycosis of the CNS is a rare, often fatal opportunistic infection. Isolated intracranial mucormycosis is especially rare. To our knowledge, this is the first reported case of likely isolated intracranial R pusillus infection in pediatrics, offering valuable insights into the presentation and management.
The case underscores the potential for intracranial mucormycosis to mimic stroke, without clear signs of infection, in an immunocompromised adolescent, emphasizing the need for clinicians to maintain a high index of suspicion for fungal infections even when initial presentations suggest other pathologies. The diagnostic challenges highlighted the critical role of serial imaging and advanced molecular techniques like PCR in identifying rare pathogens, particularly when conventional methods prove inconclusive.
Despite the delayed diagnosis, our patient's improvement with prolonged antifungal therapy demonstrates the potential for favorable outcomes with aggressive and sustained treatment, even in cases of advanced infection. This finding is particularly significant given the typically poor prognosis associated with intracranial mucormycosis.
We review 4 pediatric cases of intracranial R pusillus, all presenting with severe systemic pathology and resulting in significant morbidity and mortality. The study has several limitations. The rarity of intracranial mucormycosis in pediatric patients limits the generalizability of the findings. Additionally, the heterogeneity of cases, including variability in patient demographics, underlying conditions, and treatment regimens, poses challenges in drawing standardized conclusions.
Future research efforts should focus on developing more sensitive and specific diagnostic tools for early detection of fungal CNS infections, optimizing antifungal treatment regimens, and investigating long-term outcomes in survivors. Additionally, exploring preventive strategies for high-risk patients could prove valuable in reducing the incidence of these life-threatening infections.
Supplemental Material
sj-docx-1-jcn-10.1177_08830738251334939 - Supplemental material for Intracranial Rhizomucor Pusillus Mucormycosis in an Adolescent Triggering a Stroke Alert: A Case Report and a Systematic Review of Pediatric Cases
Supplemental material, sj-docx-1-jcn-10.1177_08830738251334939 for Intracranial Rhizomucor Pusillus Mucormycosis in an Adolescent Triggering a Stroke Alert: A Case Report and a Systematic Review of Pediatric Cases by Rima El Atrache, Melissa Mizerik, Joaquin Francisco Ruiz Lopez, Eric Engstrom, Karla Patricia Salazar, Mikael C. Guzman-Karlsson, Daniel Davila-Williams, Sonali Sen, Kristen S. Fisher and Jennifer C. Erklauer in Journal of Child Neurology
Footnotes
Acknowledgments
We thank the intensive care, infectious disease, hematology and oncology, neurology, and neurosurgery teams, as well as the nurses and physical therapists at Texas Children's Hospital, who contributed to the patient's recovery.
Author Contributions
REA contributed to conception and design, EE and MGK contributed to design, REA, MM, JRL, EE, and KS contributed to acquisition, analysis, and interpretation, MGK contributed to interpretation, REA, JRL, EE, and MGK drafted manuscript, DDW, SS, KF, and JE contributed to analysis and interpretation. All authors critically revised manuscript, gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Consent to Participate and Publication
Written informed consent was obtained from the patient and legally authorized representative for anonymized patient information to be published in this article.
Data Availability
The case report and literature review do not contain any data sets or quantitative/qualitative data as it is a clinical case description.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
No ethical approval was required.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
