Abstract
This clinical guideline provides recommendations for the evaluation and management of individuals with
Background
Methods
Aims
This guideline results from review of the literature and the consensus of clinicians with experience treating individuals with PLAN, with input from caregivers of individuals with PLAN. The guideline includes all phenotypic presentations of PLAN. We looked broadly at interventions, therapies, or other management tools for treating different aspects of PLAN, including the primary disease process and the secondary manifestations (motor disorders, vision loss, developmental delay/regression, autonomic dysfunction, seizures, communication disorders, feeding and gastrointestinal disorders, psychosocial features and family support, end-of-life care). The guideline is intended for physicians, practitioners, therapists, and other professionals caring for individuals with PLAN.
Process Overview
As there are limited data on management of PLAN, expert opinion was sought in an approach similar to that used in the development of the pantothenate kinase–associated neurodegeneration 1 and β-propeller protein–associated neurodegeneration 2 guidelines and following the AGREE-II framework. 3 Specifically, a Guideline Development Group was formed including clinical experts in PLAN, a methodologic expert, and family members/caregivers of individuals with PLAN. The Guideline Development Group included 8 clinician members (MK, AS, NN, GZ, SH, PH, AG, JW), 1 methodology expert (AH), and 11 family members (MR, AH, DD, MT, SB, NS, PW, HM, AD, AS, LP). A modified Delphi method was used to develop the guidelines in the following steps:
Clinical Question Development
Members of the Guideline Development Group (clinician and family experts) independently proposed clinical questions important to the management of PLAN. PLAN clinician expert members of the Guideline Development Group then individually prioritized the questions as (1) less important, (2) moderately important, and (3) very important. The scores were averaged for each question. The members of the Guideline Development Group (clinician experts) met virtually to discuss question selection. The Guideline Development Group noted that many questions that had lower scores were still deemed valuable. For example, questions relating to parkinsonism and deep brain stimulation for adult PLAN were not in the top 15, yet the Guideline Development Group felt that these were very important topics for the adult PLAN subtype. Ultimately, the group decided to include all the questions except for 3 that were either too vague or outside of the scope of the guideline. Each clinical question is posed in bold starting in part 2 below, then answered accordingly.
Systematic Literature Search
A literature review was performed in June 2022 in consultation with a research librarian. Medline was searched using the terms
Grading the Evidence and Recommendation Development
The level of evidence was rated using the GRADE methodology. Individual studies were largely case reports or case series, which were assessed using the NIH study quality tools (https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools). The body of evidence for each outcome was assessed and rated on level of certainty (very low, low, moderate, high). If no studies addressed a particular outcome, the recommendation was developed by consensus opinion of the Guideline Development Group. As there was no research evidence to address the majority of the clinical questions, the recommendations were consensus opinion unless otherwise stated. The recommendations were drafted by the physician members of the Guideline Development Group. The items were circulated to all members of the Guideline Development Group. If consensus was not obtained on an item, the item was revised and recirculated to the Guideline Development Group until consensus was reached.
Stakeholder Input
We then sought further input from those who would be affected by this guideline. An open call to the PLAN community was made via the NBIAcure.org website for final review and comments.
Guideline Utilization and Updating
The published guideline will be shared through established NBIA, PLAN, and child neurology websites, and social media accounts and forums to reach a wide audience of practitioners. PLAN experts are encouraged to share these guidelines with their colleagues and the general practitioners caring for individuals with PLAN. Advocacy groups will share the document among their community through their websites and social media accounts. As new data emerge, the guidelines will be reevaluated and updated.
Limitations
Given the limited data available, this guideline largely reflects the clinical experience of the authors, some of whom are from the same institution and may have similar viewpoints. As data continue to emerge about PLAN, these recommendations are likely to need modification with time. Furthermore, as health care systems and resources vary, it may not be feasible or appropriate to apply some recommendations. The practitioner should understand these cautions when considering the care of an individual patient.
Part 1: Clinical Presentation and Natural History
Nomenclature
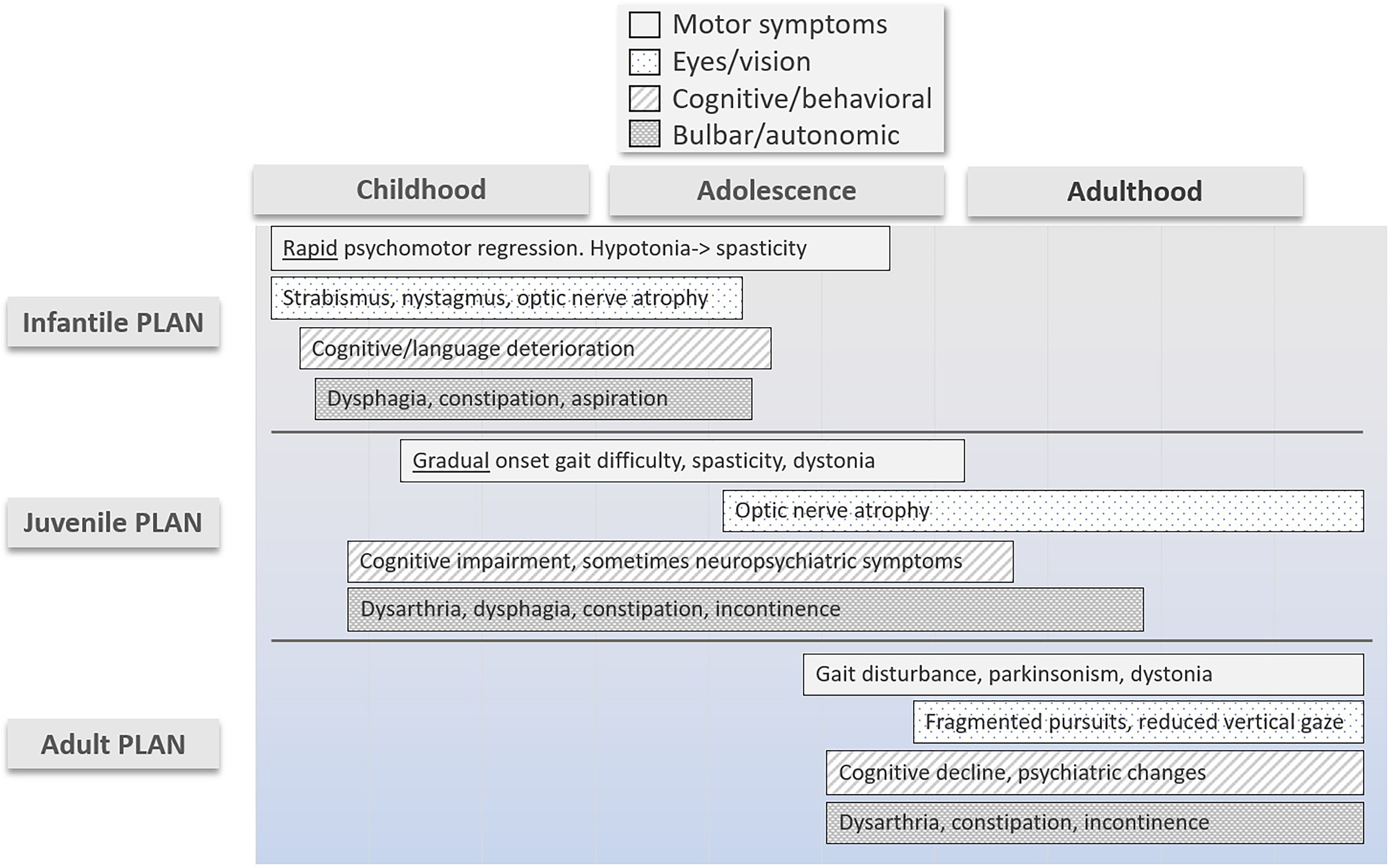
As with all the NBIA disorders, PLAN too has undergone name changes after the gene defect was identified and the phenotypic spectrum delineated. Also in common with the other NBIA disorders, PLAN includes a continuum of phenotypes. Despite this, there is benefit in categorizing the phenotypes into general groups based on age at onset. We favor use of the term “PLAN” to include all phenotypes, with each group designated further as infantile PLAN, juvenile PLAN, and adult PLAN. Infantile PLAN has long been called infantile neuroaxonal dystrophy, a name that could be used interchangeably with infantile PLAN. What was previously referred to as atypical NAD is now more appropriately termed juvenile PLAN. Finally, adult PLAN is one of the dystonia-parkinsonism disorders and is best designated without specific reference to symptoms that may or may not be present in affected individuals. Consistent use of the term PLAN with reference to its timing of onset to distinguish among the general phenotypes will enable the field to standardize the nomenclature for this disorder.
Infantile PLAN (Infantile Neuroaxonal Dystrophy)
Children with infantile PLAN have normal early development or mild developmental delay until 6 months to 2 years of age when they experience a global developmental regression, hypotonia, and sometimes eye movement abnormalities like strabismus and/or nystagmus.4,5 Initial hypotonia evolves to spastic quadriplegia, although sometimes dystonia is present as well. 6 Many children are never able to walk independently. 7 With time, optic atrophy and visual loss develop. Some individuals may develop seizures. The motor disability affects bulbar function, putting children at risk for respiratory infections, and many children die before 10 years of age, although some live to the teenage years. 7
Juvenile PLAN (Atypical Neuroaxonal Dystrophy)
Individuals with juvenile PLAN have a later presentation with a slower deterioration. Early childhood may be characterized by an ataxic gait, autistic features, and psychiatric symptoms. In childhood or early adolescence, a neurodegenerative course ensues including visual deficits, optic atrophy, and nystagmus. The course of juvenile PLAN is less well described or consistent than infantile PLAN but tends to be slower. Individuals may have stable symptoms for several years, including gait difficulties due to ataxia, spasticity and dystonia, and language and cognitive deficits. Individuals with juvenile PLAN usually lack early hypotonia and are more likely than individuals with infantile PLAN to have dystonia. Individuals may also have psychiatric difficulties or autistic features. Other symptoms that may occur later in the disease include seizures, bladder dysfunction and parkinsonism. 6 The life expectancy varies, though a longer life span than infantile PLAN is expected.
Adult PLAN (PLA2G6-Related Parkinsonism)
Adult PLAN (PARK14, OMIM 612953) presents in young adulthood (mean age 26 years) 8 with neuropsychiatric symptoms, parkinsonism, and often dystonia. Motor phenotypes characterized by ataxia or spasticity have also been described.8,9 Cognitive decline, seizures, and autonomic dysfunction may also occur. Individuals with adult PLAN have a progressive course of motor deterioration that may include parkinsonism (usually exquisitely sensitive to levodopa with high risk of levodopa-related dyskinesia), dystonia, ataxia, and spasticity. In addition, they experience cognitive decline and, later, dysarthria and dysphagia (Figure 1).
Part 2: Diagnosis and Disease Monitoring
Neuroimaging Findings
Atrophy of cerebellar vermis and hemispheres is a nearly universal feature on brain magnetic resonance imaging (MRI) of the symptomatic child with PLAN, but may not be obvious early in the juvenile PLAN disease course.5,7,10–12 Hypointensity on T2 and iron-sensitive sequences in the globus pallidus and substantia nigra is another hallmark of the disorder. However, it may not be present in young children with PLAN and certainly is more obvious as the child becomes older and the disease progresses. Other findings often seen are hypertrophy of the clava (gracile tubercle), thin and vertically oriented splenium of the corpus callosum, T2 hyperintensity in the cerebellar cortex, dentate nuclei, or supratentorial white matter, and cerebral atrophy (Figure 2).6,7,13,14 In adult PLAN, most have abnormalities on imaging, typically cortical atrophy (47%) and cerebellar atrophy (40%). Some also have iron deposition in the globus pallidus and substantia nigra (25%). All have abnormalities on presynaptic dopaminergic terminal imaging.8,12,15
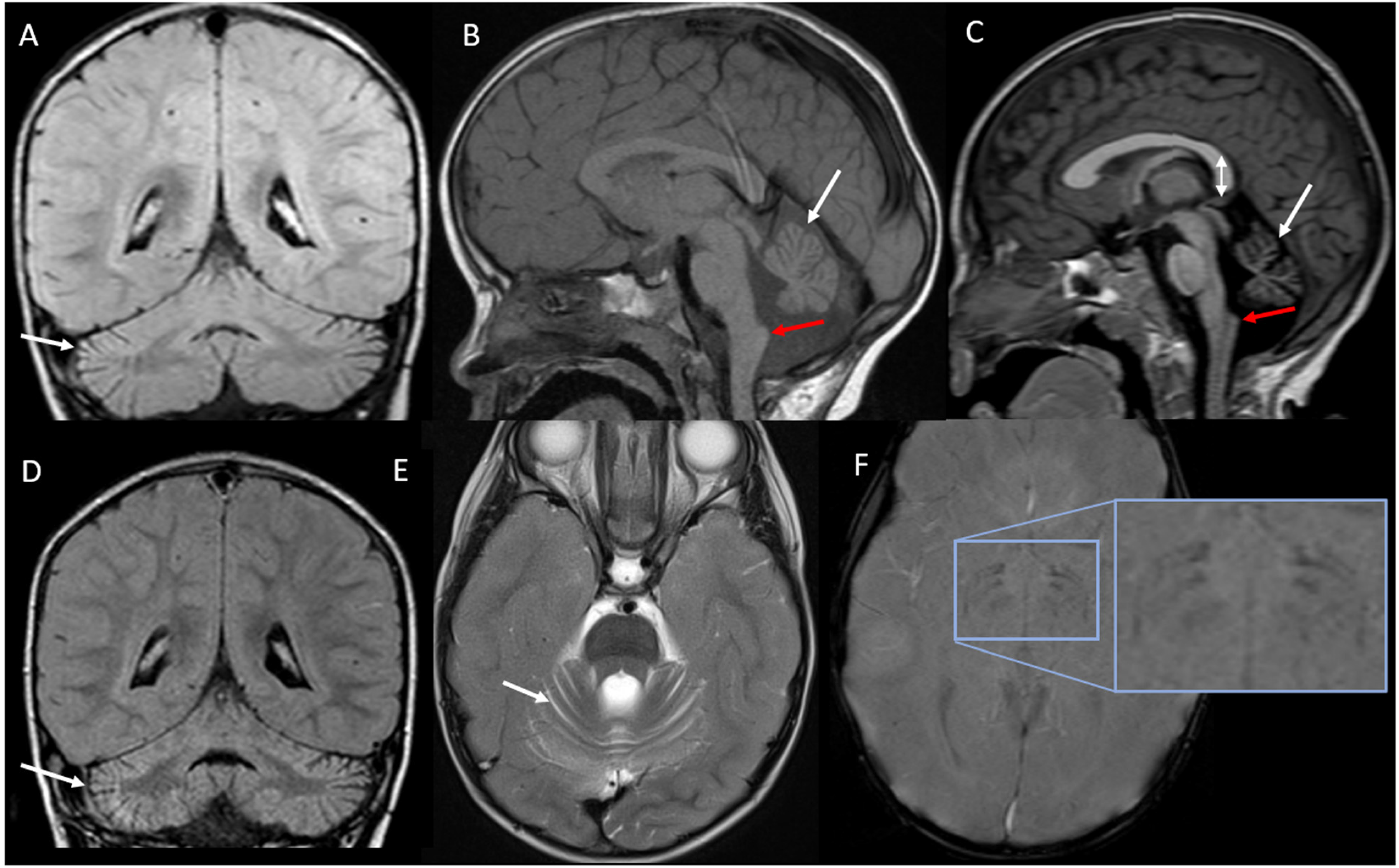
Brain magnetic resonance imaging findings in infantile PLAN. Images from children with infantile PLAN. (A and D) Cerebellar atrophy and gliosis (excess space between folia and hyperintensity of the cortex, white arrows) on coronal FLAIR MRI from the same patient with infantile PLAN at 2 years of age when the MRI was read as normal (A) and at 2.5 years of age (D). Cerebellar vermian atrophy is also demonstrated on sagittal T1 MRI (white arrows, (B) and (C)). (E) Cerebellar cortical atrophy on axial T2 MRI (white arrow). Claval hypertrophy (enlargement of the gracile tubercle) shown in (B) and (C), red arrow. Also in (C), vertically oriented splenium of the corpus callosum (double arrow). (F) shows hypointensity on susceptibility-weighted imaging in the globus pallidus and substantial nigra in a 3.5-year-old with infantile PLAN representing iron deposition (inset). FLAIR, fluid-attenuated inversion recovery; MRI, magnetic resonance imaging; PLAN,
A brain MRI is useful in the diagnostic evaluation of an individual presenting with symptoms of PLAN because the specific features (claval hypertrophy, cerebellar atrophy and gliosis and thin, vertically oriented splenium of the corpus callosum), can strongly suggest the diagnosis. If the clinical picture is in keeping with NBIA disorder, sequences that highlight iron accumulation (T2*, susceptibility-weighted imaging [SWI], quantitative susceptibility mapping [QSM], gradient echo [GRE]) should be performed where possible. In a child with genetic confirmation of PLAN, imaging is not recommended if the clinical symptoms are consistent with the diagnosis.
Genetics
Heterogeneity and genotype-phenotype correlations
PLAN is an autosomal recessive condition caused by pathogenic variants in
When PLAN is suspected, genetic testing is recommended to confirm the diagnosis. Sequencing and deletion/duplication testing of the
Tissue Biopsy
Before discovery of the
Biopsy is rarely indicated in the diagnosis of PLAN. Biopsy may be considered in individuals with a clinical diagnosis of infantile PLAN or juvenile PLAN who have negative genetic testing and for whom other diagnostic studies described above are indeterminate. However, as axonal spheroids may not be present early in the disease course or in all tissues, a single negative biopsy does not exclude PLAN. 20 If biopsy is being considered, locations typically chosen include the sural nerve, skin overlying muscle, rectum, or conjunctiva, depending on patient, practitioner, and institutional experience.
Laboratory Evaluations
Elevated AST and LDH with normal alanine aminotransferase7,14,21 are frequent findings in infantile PLAN and may provide a clue toward the diagnosis, but do not need to be routinely evaluated.
Disease Monitoring
Monitoring the progression of disease includes a combination of clinical evaluations by a multidisciplinary team including physical therapy, occupational therapy, and speech therapy and various investigations.
Clinical assessment
Regular and detailed clinical evaluations by a neurologist or similar specialist are essential for monitoring the overall progression of disease. They may assess motor function, cognitive abilities, speech and language skills, and other neurologic symptoms such as movement disorders. Scales such as the infantile PLAN rating scale
22
may be used to quantify and monitor the severity of motor symptoms over time. As a general rule, only those studies that will change management are recommended.
The frequency and specific assessments may vary depending on the individual's symptoms, age, and clinical presentation. Close collaboration between the individual, caregivers, and their health care team are crucial to ensure optimal monitoring and management.
Part 3: Symptom-Based Treatment
Care of the person with PLAN requires anticipatory guidance for the person affected and his or her family, monitoring for complications and management of neurologic symptoms.
Body Function and Structure
Motor
Tone and movement
Spasticity can be problematic in children with infantile PLAN and individuals with juvenile PLAN. Spasticity may cause discomfort, interfere with bracing, make positioning and sitting difficult, and lead to contractures. Treatment of spasticity should be considered if the hypertonicity interferes with function or comfort. In some children with juvenile PLAN, increased tone may offset advancing weakness and enable continued motor activities. Therefore, treatment for spasticity should consider this balance.
Oral spasticity medications such as baclofen, tizanidine, and benzodiazepines may be indicated if spasticity is affecting quality of life. A “start low and go slow” approach can help with tolerance. Botulinum toxin may be considered for focal spasticity if there are worthy goals (eg, brace tolerance to allow stander use). However, the risk of systemic effect with the potential to cause worsening respiratory status is greater in children with more significant motor impairment.
23
The intrathecal baclofen pump has been used in individuals with juvenile PLAN,
24
although given the 30% complication risk reported in children,
25
this intervention should be reserved for severe spasticity not responsive to less invasive approaches.
Dystonia may be problematic in juvenile PLAN and adult PLAN. Although there are no data to support a specific treatment approach, baclofen, trihexyphenidyl (monitoring for constipation), gabapentin, and botulinum toxin are reasonable options. There has been one report of deep brain stimulation successfully treating status dystonicus in juvenile PLAN after failure of other treatments. 24 Deep brain stimulation has been more commonly used in the setting of adult PLAN.
Parkinsonism
Individuals with adult PLAN and parkinsonism often respond dramatically to dopaminergic agents, although the response may be short-lived, and dyskinesias and motor fluctuations typically emerge. The dyskinesias are expected to decline after discontinuation of dopaminergic agents, although individuals may prefer some dyskinesias to experiencing the “off state.” Thus, clinicians and the individuals they treat must consider at which point the dyskinesias are more problematic than the benefit derived from dopaminergic agents. Other treatments such as amantadine, levodopa intestinal gel, and deep brain stimulation (see below) may be considered as adjunctive therapies.
Individuals with infantile PLAN should not be treated with levodopa. Some individuals with juvenile PLAN may develop parkinsonism late in disease.
6
The juvenile PLAN phenotypic spectrum is broad and may overlap with adult PLAN.
19
For this reason, if there is clear parkinsonism, then a trial with a dopaminergic agent could be considered.
Although the reported outcomes of deep brain stimulation are favorable, there are few cases (10) reported, and long-term data are extremely limited. Deep brain stimulation has been reported to improve motor symptoms, including a reduction in freezing of gait, improved balance, and reduced dyskinesias. Nonmotor symptoms, such as pain, may also improve.15,24,26–28 The onset and severity of levodopa-induced dyskinesias may be the greatest influence in determining the best time for deep brain stimulation.
Orthopedic
Individuals with infantile PLAN and juvenile PLAN are at risk for contractures and other orthopedic problems.
7
They should be periodically evaluated by an orthopedic specialist. However, the benefits of surgery should be carefully weighed against the risks for children with respiratory compromise and shortened life expectancy.
Individuals with PLAN who are nonambulatory are at risk for fracture from low bone mineral density because of lack of weightbearing and potential nutritional deficiencies. For these reasons, we suggest individuals with PLAN adhere to the American Academy of Cerebral Palsy and Developmental Medicine (AACPDM) Care Pathway, which recommends adequate calcium and vitamin D intake and a weightbearing program that may include use of a standing frame for nonambulatory individuals, if feasible and not interfering with quality of life.
29
Caregivers of individuals with very limited mobility should consult with their physical therapist, physiatrist, or orthopedist on handling methods and equipment to minimize risk of fracture.
Surveillance
Although specific orthopedic surveillance guidelines do not exist for PLAN, given the similarities in motor complications to individuals with cerebral palsy, it is reasonable to follow the POSNA 30 or AACPDM guidelines 31 that recommended beginning surveillance for hip subluxation or dislocation at 2 years of age with intervals dependent on functional motor status and prior screening results. An orthopedist or other subspecialist with expertise in this area (eg, physical medicine and rehabilitation) should also monitor for contractures and scoliosis at least annually.
Management
Management of orthopedic complications should be individualized based on symptoms and goals of care.
Hip dislocation
If a child with infantile PLAN develops a nonpainful hip dislocation, nonoperative management may be a reasonable alternative to surgery which is typically done to prevent pain in the teenage years. Surgical management may be appropriate if the dislocation is painful, making care and hygiene difficult, and the child is healthy enough to safely undergo surgery.
Contractures
Loss of mobility and spasticity may lead to contracture development. Bracing and splinting can be an effective prevention strategy. Some contractures are not bothersome or can be accommodated (eg, using a wedged ankle-foot orthotic to allow standing) and do not require treatment. A bothersome contracture (eg, impacting hygiene/skin integrity, comfort, ability to participate) can be treated with casting or surgery.
Scoliosis
Scoliosis may develop in some individuals because of increased truncal muscle tone or muscle weakness. Although the former has no proven preventive strategy, the latter may respond to bracing. Some individuals who are nonambulatory may develop more severe scoliosis that interferes with comfort, respiratory function, and digestion, and makes seating difficult. A spinal fusion may be considered after carefully weighing the risks for the particular individual and expected benefits, and with multidisciplinary operative and postoperative planning. 32
Rehabilitation
All children with PLAN are likely to benefit from physiotherapy. It is not known whether any specific PT or OT program is advantageous over the others as they have not been evaluated systematically. PT and OT should focus on tailored programs aimed at maximizing function, maintaining range of movement, and improving core strength. For example, a child who has some limited walking ability may benefit from a gait trainer to assist with walking, a stander to build bone-density, ankle-foot orthotics to maintain ankle flexibility, and task-specific OT. A child with advanced infantile PLAN may benefit from an equipment and home environment evaluation, regular stretching and bracing of joints at risk for contracture, and other approaches such as massage. Identifying a therapist with expertise treating individuals with similar needs is helpful in guiding this process.
Pragmatically, it is important for a person's multidisciplinary team to be aware of the progressive nature of the condition and ensure regular reviews and adjustments to their tailored therapy. It is also important to note that although this is a progressive, neurodegenerative condition, some children and adults may have stable periods (particularly juvenile PLAN children) and may continue to make forward progress with their development over the course of the disease. The caregivers should work closely with the therapist to identify realistic goals that optimize quality of life and determine an approach that will work best for the individual in their environment.
Pain
Pain may occur due to dystonia, gastrointestinal issues (reflux, constipation), and orthopedic complications. In the later stages when individuals have more difficulty with communication, it may be more difficult to know if they are experiencing pain. Although not specific to PLAN, studies have identified common pain behaviors in children with severe neurologic impairment including crying, grimacing, grinding or clenching teeth, irritability, agitation, sleep disruption, increased movements, pulling away when touched, stiffening, sweating, and breath-holding.
33
As children with infantile PLAN become less mobile, facial expressions, vital sign changes (increased heart rate or blood pressure), and disruptions of sleep may be the best indicators of pain. If pain is identified, the individual's physician should do a search for the source of pain which may include a careful skin exam, evaluation for corneal abrasion, hair tourniquet, or occult fracture, constipation evaluation and dental exam.
Treatment should be directed at the source of pain, if identified. In a child with reflux or constipation, ensure these are adequately treated. If no source is identified, empiric treatment for neurologic pain should be considered. Nonpharmacologic approaches may help with mild discomfort (eg, massage, music, repositioning, removing orthotics, use of a fan, vibratory therapy). Medications directed at neurologic pain such as gabapentin, tricyclic antidepressants (TCAs), or clonidine may be considered (Figure 3). 33 Some individuals also benefit from an as-needed medication, such as nonsteroidal anti-inflammatory drugs (NSAIDs) or acetaminophen (see “CBD” section for comment on cannabinoids). A pain or palliative care specialist may be a helpful care team member.
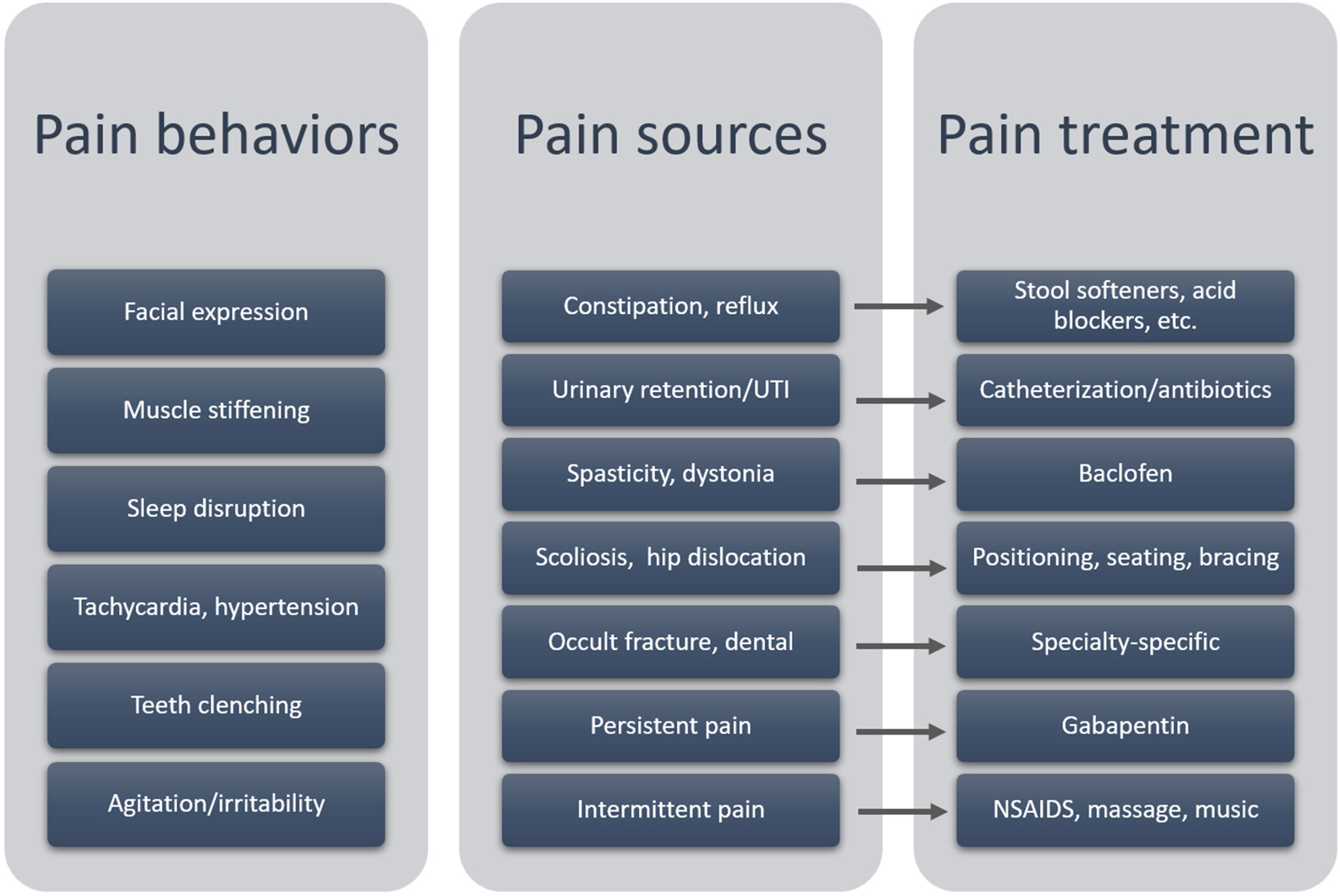
Pain sources, behaviors, and treatment in PLAN. Examples of behaviors that may indicate pain in an individual with PLAN, potential sources of pain and types of pain, and how those may be managed. NSAIDS, nonsteroidal anti-inflammatory drugs; PLAN,
Seizures
Seizures occur in a significant minority of individuals with PLAN, and seizure types may include generalized tonic seizures, focal seizures, and myoclonic seizures. Epileptic spasms have also been reported.7,10,34 Complex epilepsy phenotypes have not been reported. Seizures tend to occur later in the disease course and are not generally refractory to treatment.7,14,35,36 An EEG may show fast rhythms,5,10,11,14 although this does not need to be routinely obtained if there is no clinical suspicion for seizures.
Individuals with PLAN, particularly infantile PLAN later in disease, may have nonepileptic stiffening or jerking episodes that can only be distinguished from seizure with an ictal EEG.
37
Without an EEG, there are other clues that can help with this determination. Seizures tend to be unprovoked, not interruptible, stereotyped, and involve loss of awareness and a postictal period. A video of the event may be helpful. If uncertainty remains after EEG, and the spells are frequent and bothersome, a trial of an antiseizure medication could be considered.
There is no evidence for a specific approach to seizure management in PLAN. As with other children, seizure medications are chosen based on seizure type and side effect profile.
Gastroenterology, Feeding, and Nutrition
Individuals with PLAN commonly experience dysphagia. Children with infantile PLAN gradually lose oral feeding abilities, which eventually result in the inability to maintain adequate nutrition and/or hydration, and risk of aspiration pneumonia. Discussion of this progression and early preparation for the eventual need for gastrostomy tube feeding will help make this transition smoother for families. A feeding specialist or team should continue to re-evaluate, make recommendations about food textures, safe feeding approaches, and nutritional supplementation and determine the timing of referral for gastrostomy tube placement.
Although early efforts are often directed toward increasing intake, in children with decreasing mobility (eg, those with infantile PLAN), caloric intake will likely later need to be decreased to avoid excessive weight gain.
A gastrostomy tube should be placed when the individual cannot eat safely or maintain nutritional or hydration status, or when oral feeding has become excessively difficult or time-consuming, if not sooner. The goal is to proceed while the nutritional status is still optimal, and the family is not in crisis. This is best achieved using shared decision making, informing and supporting caregivers while considering caregiver and individual values.
38
Providers should begin the conversation early, particularly for children with infantile PLAN, all of whom will require gastrostomy tube feedings. Connecting families to patient organizations could help provide practical information and support.
Constipation is often a significant challenge in PLAN that can impact comfort and quality of life. A principle of management is to develop a consistent regimen that prevents constipation rather than responding with as-needed treatments when constipation becomes problematic. Dietary management and physical activity and movement (eg, stander use) can be very helpful. Caution should be used around medications with constipation as a side effect (iron, trihexyphenidyl, glycopyrrolate). Stool softeners are typically used for constipation not responsive to dietary changes. Given that autonomic dysfunction in PLAN can affect gastric motility, the addition of a motility agent may be needed. Suppositories or enemas can be used as needed. In challenging cases, a gastroenterologist should be involved.
Reflux is common and can be problematic. There are no studies to suggest a specific approach with PLAN, and relevant clinical guidelines should be followed. The most benign interventions are dietary changes and positioning techniques. Medications such as proton pump inhibitors may be needed, although there are some concerns about long-term use of these medications. Motility agents such as erythromycin and baclofen are other options. Newer motility agents are being studied in children. For refractory symptoms, surgical approaches such as Nissen fundoplication may be considered.
Respiratory
Respiratory care strategies have not been evaluated systematically in PLAN. However, what is known is that because of bulbar dysfunction, children with infantile PLAN often have difficulty maintaining a safe swallow as the disease progresses, putting them at risk of aspiration. Muscle weakness and scoliosis may also contribute to respiratory insufficiency. Children with infantile PLAN should have their swallowing and mode of nutritional delivery routinely monitored. Those who have recurrent chest infections, frequent cough, rattly breathing, or snoring should be referred to a specialist respiratory team if available. Airway clearance techniques (medications, chest physiotherapy, nebulizers, suctioning) should be instituted proactively. General preventative strategies to maintain respiratory health for any child with neurologic disorder should be applied (eg, annual seasonal flu vaccines). Regular preventative antibiotics (such as azithromycin 3 times a week over the winter months) could also be considered.
Individuals with PLAN, particularly infantile PLAN, are at risk for obstructive and central sleep apnea. Children who have symptoms suggestive of sleep apnea (snoring or pauses in breathing during sleep) should also be considered for a polysomnogram to assess the severity of sleep apnea and determine if tonsillectomy and adenoidectomy or noninvasive ventilation (such as continuous positive airway pressure or bilevel positive airway pressure) may be indicated. The decision to commence noninvasive ventilation should be made with the child's multidisciplinary team and involve the family at every step of the process. The additional burden of care, benefits and risks of the intervention, and progressive nature of the condition should be discussed with the family in coming to a shared decision.
Treating obstructive sleep apnea may improve central sleep apnea. If central sleep apnea requires treatment, approaches include minimizing sedating medications and addition of supplemental nocturnal oxygen or positive pressure ventilation.
Cardiac
It is not known whether children with infantile PLAN should have cardiac surveillance as this has not been studied systematically. Cardiac disease is not commonly reported in PLAN. If a child has cyanotic episodes or dysautonomia, or other symptoms suggestive of a cardiac condition, they should be referred to a cardiology team for evaluation and/or further management.
Urologic
Providers and families should be aware of the risk of urinary retention and urinary tract infection in children with infantile PLAN. It is not known whether children with PLAN should have routine screening for neurogenic bladder. However, given this has been reported in case reports and series and may be underrecognized, it would be prudent at routine follow-up visits to inquire about long intervals between voiding or frequent urinary tract infections. These symptoms should prompt referral to pediatric urology. Constipation, a common symptom in childhood PLAN that can exacerbate urinary retention, should be actively managed. Individuals with adult PLAN frequently have incontinence, urgency, and other urinary symptoms that may also warrant urologic evaluation. 39
Vision
Individuals with infantile PLAN commonly experience nystagmus, strabismus, upgaze palsy, saccadic pursuits, saccadic intrusions, and optic atrophy. There may be some loss of vision later in disease.
40
Individuals with PLAN should establish care with ophthalmology on diagnosis. Some children may benefit from glasses. However, there are no treatments to improve the optic atrophy. Individuals should continue to see ophthalmology at least annually even if no abnormality is detected on initial examination. Individuals with visual impairment should seek early evaluation with a low vision specialist.
Hearing
Children with infantile PLAN have a high likelihood of sensorineural hearing loss or auditory neuropathy spectrum disorder. 41 The time course of the hearing loss is not clear. Children should begin formal audiology testing at PLAN diagnosis and at least annually depending on the findings of initial examination. Children should receive an audiologic test battery that evaluates all components of the audiologic system, as appropriate for developmental level. A sedated auditory brainstem response (ABR) may be required. Children with hearing loss should receive directed speech therapy. Amplification may be indicated but should be considered on a case-by-case basis.
Sleep
It is important to inquire about sleep as it often has a major impact on quality of life of the individual with PLAN and those who care for and live with that person. However, if the sleep issue is not causing problems (eg, the child wakes up overnight but does not wake family members and is rested during the day), then no intervention may be required. The approach is to address behavioral components of insomnia, evaluate the sleep environment, investigate medical factors impacting sleep, and consider medications (see Table 1).
Sleep Difficulties, Testing, and Interventions in PLAN.
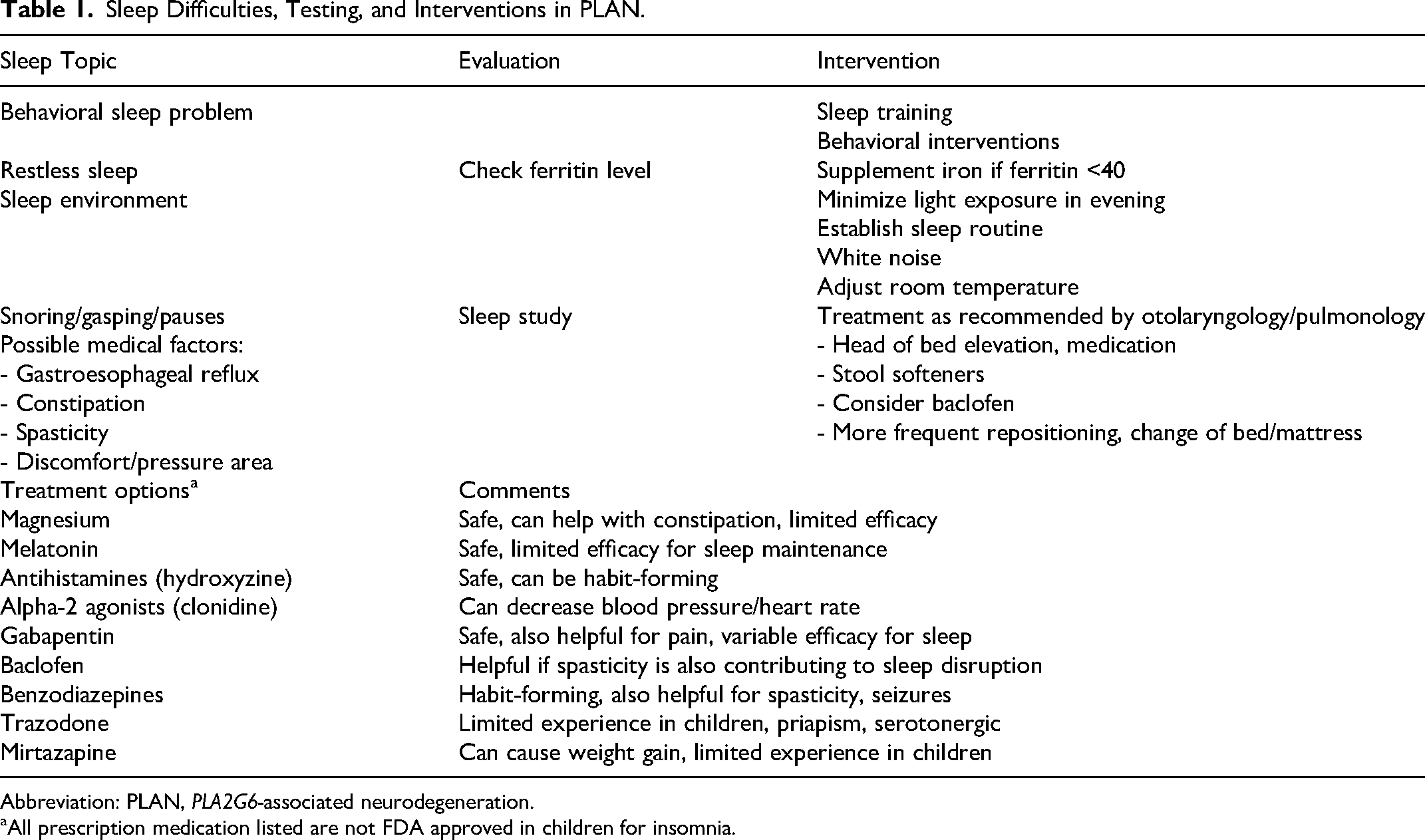
Abbreviation: PLAN,
All prescription medication listed are not FDA approved in children for insomnia.
Autonomic
Autonomic dysfunction may take the form of decreased circulation, constipation, urinary retention, decreased gastric motility, and temperature dysregulation. Treatment is supportive. Reassurance can be given that the discoloration in the extremities is not dangerous. In the later stages of infantile PLAN, caregivers may need to be cautious in hot or cold environments, which may lead to vital sign changes. Individuals with adult PLAN may also have orthostatic hypotension like adults with classic Parkinson disease, and it is reasonable to take a similar approach.
Psychiatric and Behavioral
Anxiety, depression, aggression, impulsivity, and psychosis can be early symptoms of adult PLAN. It may be helpful to involve psychiatry, psychology, and a behavioralist. In individuals with limited communication, challenging behaviors could be the manifestation of, or exacerbated by a physical problem (eg, constipation, dental pain, sleep deprivation), be the result of a psychiatric problem (eg, anxiety), and may serve a function such as avoidance of unwanted activities. Medical triggers should be sought. A functional analysis may help identify the factors that influence the behavior and guide behavioral interventions (eg, applied behavioral analysis). If there is an underlying psychiatric cause, medication directed at that cause may be appropriate. If other methods fail, pharmacologic interventions may be needed. In some individuals with PLAN, treatment with antipsychotics seems to precipitate dystonia or parkinsonism, although causality is not always clear as the motor symptoms generally persist after discontinuation of the medication.
Treatment of psychotic symptoms (hallucinations, paranoia, delusions) must be approached cautiously because of the potential for extrapyramidal side effects with antipsychotic medications. It is reasonable to take the same approach in PLAN as in Parkinson disease, where clozapine and quetiapine are preferred antipsychotic medications because their lower D2 receptor affinity decreases the risks of worsening parkinsonism. Aripiprazole has also been used to treat mood and psychotic symptoms in PLAN. 42
Environment
Equipment
Proper equipment is key to maximizing function, comfort, participation, well-being, and health of individuals with disabilities (and their caregivers). This may include the following:
mobility and positioning devices such as wheelchairs, standers, lifts, adaptive tricycles, and gait trainers; bracing such as ankle-foot orthotics, upper extremity splints, and thoracic-lumbar-sacral orthoses; communication devices; equipment to support activities of daily living such as adaptive silverware and drawing/writing devices, and bathing equipment; toys and games; and clothing and shoes.
In PLAN, the equipment needs will depend on the individual and will likely change over time. A physical therapist and occupational therapist with expertise in equipment often serves as a guide to appropriate equipment. Equipment can be obtained through an equipment vendor with a prescription from a physician. Insurance may fund some equipment, although the process can be time-consuming. Sometimes there are additional funds through grants or access to equipment for lending through hospitals and other programs.
Participation and activities
School and work
Children with PLAN have significant developmental, motor, and medical challenges that impact education. Children with PLAN should have an Individualized Education Plan to support their particular needs that should be reevaluated at least annually. An educational lawyer or advocacy center may be beneficial in supporting families working with educators.
School programs may incorporate vocational training and/or an extended school program so that individuals can continue in a school setting as young adults to aid in the transition to work. Families should familiarize themselves with available resources for disability supports (ie, vocational rehabilitation programs), which vary by location. Additional resources include social workers, transition specialists, or a transition clinic.
Activities of Daily Living
The support systems and approaches for individuals with PLAN in their daily activities will be specific to the individual and will change over time. Of primary importance is establishing a local team with expertise in developmental disabilities that can adapt their approach as needed.
Mobility
Maintaining mobility and weightbearing builds bone density, prevents contracture development, helps with digestion/constipation, may allow for more social interactions, can be enjoyable, and gives the individual more independence. Wheeled mobility may provide easier transport and participation in activities outside of the home. For some, it leads to an increase in independence. Children with infantile PLAN should be followed regularly by a PT with expertise in equipment as the needs of these children will change with time: they may shift from a walker or gait trainer to a stander. Individuals with juvenile PLAN and adult PLAN may need a wheelchair as they get older, although using a stander may still be important. A rehabilitation program developed by families working with rehabilitation specialists should have realistic goals in the context of the individual's prognosis.
Communication
A hearing evaluation is necessary for all children as hearing loss is an important contributor to communication deficits. Individuals with PLAN should be evaluated by a speech therapist and have ongoing communication support. The approach to communication should be individualized. Children with infantile PLAN experience loss of language, which can occur after the loss of motor function. However, they may retain receptive language and cognitive functions for years after symptom onset, and benefit from involvement of speech therapists with expertise in augmentative and alternative communication (AAC). Children with juvenile PLAN may have a longer period of static developmental deficits, benefiting from consistent speech therapy. Individuals with adult PLAN may experience dysarthria and cognitive decline requiring communication supports. As some individuals may have more difficulty with communication over time, augmentative communication strategies should be re-offered periodically.
Routine Care and Systems of Care
Individuals with PLAN often have needs from multiple specialists and subspecialists that focus on anticipatory and preventive measures as well as active interventions to address secondary complications from the disorder. Regular coordination of team care leads to the best management.
Neurology or neurodevelopmental specialist: Children benefit from a full evaluation by either pediatric neurology or a developmental specialist. Adults should see a neurologist or movement disorder specialist. Feeding specialists: Feeding specialists or teams should manage dysphagia and nutritional deficiencies particularly with disease progression. Ophthalmology or neuro-ophthalmology Audiology Medical genetics and genetic counseling Psychiatry: Evaluation and management of psychiatric complications of PLAN is an important component of care for most adults and some children. Rehabilitation therapy: Individuals with PLAN benefit from early involvement of PT, OT, and speech therapy. Orthopedics or physical medicine and rehabilitation: These specialists can oversee structural issues such as scoliosis and direct rehabilitation and equipment needs.
Individuals with PLAN should undergo a standard approach to immunization.
Although iron accumulation may be present on neuroimaging, we do not recommend a low iron diet as it leads to systemic iron deficiency without affecting brain iron levels or disease course. Dietary iron with supplementation as needed for age and nutritional state is recommended.
There are no specific dietary modifications shown to be beneficial for individuals with PLAN.
Individuals with PLAN may undergo anesthesia if indicated, such as for gastrostomy tube placement. It may be prudent to forgo sedated investigations that will not impact clinical management. Optimizing respiratory status and avoiding anesthesia around illness decrease anesthesia risk. Succinylcholine should be avoided as it can rarely induce hyperkalemia in individuals with denervation or limited mobility.
43
Oral hygiene recommendations are generally similar to those of other children; however, children with developmental disabilities including PLAN may be at higher risk for dental caries. Guidelines recommend establishing a dental home by age 1 year, daily brushing with fluoride-containing toothpaste beginning at first tooth eruption, and fluoride supplementation.
44
The American Academy of Pediatrics offers guidelines on menstrual management in adolescents with disabilities, which is a great resource for individuals with PLAN and their families. 45 Premenarchal suppression is generally not recommended. Postmenarchal menstrual management may be appropriate if menstruation is affecting quality of life. The choice is individual and should be discussed with a knowledgeable primary care physician or subspecialist.
Emergency Care Plan
Individuals with PLAN should have an emergency care plan, which is a concise overview of their condition and treatment recommendations. The care plan may include medical diagnoses, current medications, common medical issues and suggested management, medical providers and contacts, and specific factors about the individual such as how they communicate. The American Academy of Pediatrics has developed an emergency information form for children with special needs that would be suitable for children and adults with PLAN (https://www.acep.org/by-medical-focus/pediatrics/medical-forms/emergency-information-form-for-children-with-special-health-care-needs/).
End of Life
Although this has not been systematically reported, it is likely that respiratory failure often following illness is a common cause of death in PLAN. 7
Palliative Care
Individuals with infantile PLAN have a shortened life expectancy, typically living until late childhood. Clinicians should be transparent with families about the prognosis. It is important for the child's consistent provider(s) to discuss end of life care with families ideally before a time of crisis or hospitalization. Providers must be sensitive to the family's culture and experiences, using a shared decision-making approach to formulate a plan that aligns with the child's/family's values. Providers should give guidance. For example, it is appropriate to recommend no chest compressions nor intubation for a child nearing the end of their life. Although difficult, establishing an end-of-life care plan (preferred location of death, interventions and procedures not indicated, and code status) will help ensure that the child’s and family’s wishes are respected, and that the child is not subjected to harmful interventions. Involvement of a pediatric palliative care team or expert early after diagnosis is recommended to help facilitate these conversations support families. These teams often involve a physician, nurse, case manager, social worker and spiritual care advisor. Hospice care can provide valuable support as the child nears the end of life.
Even with careful planning, sometimes difficult decisions must be made during a hospitalization. The care team should be fully informed about PLAN. A useful question, as suggested by the “AAP Guidance for End of Life Care,” is to ask, “Are we doing this A parent does not want to be in the ICU trying to come to terms, unprepared, with how to proceed when a child is so ill, and you have specialists of every sort involved in just keeping the patient's vital signs going. When is it time to let go? Here is where social workers, hospice/palliative care team and chaplains are essential. There is a point at which continued medical intervention seems pointless and even cruel. Parents need to plan in advance and should be strongly encouraged by the medical team to do so.
Life expectancy in juvenile PLAN and adult PLAN is less clear from the literature, and palliative care may be appropriate for some individuals. Children and adults should be included in the discussions about their prognosis at a level appropriate to the individual. The American Academy of Pediatrics provides in-depth and practical guidelines applicable for children with PLAN. 46
Psychosocial Support
What are the most effective ways to provide psychosocial support to individuals with PLAN and their families?
Families affected by PLAN experience grief with the diagnosis and understanding of the progression of disease and ultimately the shortened life span associated with PLAN. They benefit from support systems during this time. Families affected by PLAN may feel isolated, given the rarity of the disorder, and overwhelmed by the complex future medical needs of their child. It can be helpful for these families to seek out connections and support through online resources such as PLAN social media groups. Participation in these groups can be a rich source of support and information but may be overwhelming for some newly diagnosed families, so clinicians should discuss these resources with appropriate balance and cautions. In some cases, it may be preferable to connect them to a few other families as a starting point, using the advocacy groups as resources.
The INADcure Foundation and Cure INAD UK have become hubs for PLAN advocacy, and support families through research efforts, fundraising, and engagement of investigators and partners. More formal activities are also carried out through the NBIA Alliance (https://www.nbiadisorders.org/family-support/nbia-alliance), an international federation of patient advocacy organizations for NBIA disorders. Alliance members work together and individually to raise awareness about NBIA disorders, support patients and families, and promote research. They offer additional opportunities for families and care providers to interact and learn from one another through group forums and family conferences. The largest international family conference, hosted regularly by the NBIA Disorders Association (https://nbiadisorders.org/), provides updates to families on the latest research and treatments and promotes networking and support for affected individuals, as well as their family members.
Part 4: Future Directions
Disease-Specific Treatments
There are currently no disease-specific treatments available. The basic biology of PLAN is still not well understood. The
Food and Drug Administration (FDA) Approved for Another Indication
Desipramine
Based on studies of fruit fly and human cell models of PLAN, there has been interest in targeting the accumulation of ceramides as a therapeutic end point with drugs such as myriocin or desipramine. 47 An uncontrolled clinical trial of desipramine in children with infantile PLAN closed, but no data have been published (https://clinicaltrials.gov/study/NCT03726996). Anecdotal reports following a short course of desipramine in a few children with PLAN suggest that the drug is poorly tolerated and may cause agitation (personal communication). Studies of the drug are needed in a good mammalian model of PLAN to determine if further human trials are warranted. Desipramine is not recommended for PLAN given the risks and lack of data.
Iron Chelating Agents
Iron chelation has been an area of active research in NBIA disorders. A randomized controlled trial of deferiprone in individuals with pantothenate kinase–associated neurodegeneration, a distinct NBIA disorder, found no significant difference between placebo and treatment groups in change in dystonia measurement after 18 months. 48 In pantothenate kinase–associated neurodegeneration and many other neurodegenerative disorders, insufficient bioavailable iron leading to iron overload is thought to be a common theme that may also be active in PLAN.49–53 MRI demonstrates iron accumulation in the basal ganglia in some individuals with PLAN, but it is not hypothesized to be the cause of the clinical symptoms of PLAN. Two individuals with PLAN treated with deferiprone after 2 and 7 years had stable levels of brain iron in one and a decrease in brain iron in the other, although both experienced worsening of clinical symptoms in this time period. 54 Iron chelation is not recommended for PLAN as there are risks of treatment without evidence of clinical benefit.
Semaglutide
Semaglutide, an GLP1 receptor agonist used in the treatment of type 2 diabetes mellitus, has been shown to decrease neuronal loss and improve symptoms when given in high doses to a mouse model of infantile PLAN. 55 One formulation of semaglutide has FDA approval for people 12 years and older for treatment of obesity, although there is potential for serious side effects. Further research is needed. Semaglutide is not recommended for PLAN given the risks and lack of data.
Other
Researchers have identified 4 medications (ambroxol, azoraminde, desipramine, and genistein) that lessen abnormalities seen in flies and patient-derived cells lacking
Not FDA Approved
Gene therapy
There are 2 in vivo, adeno-associated viral vector–delivered gene therapies being developed that are at preclinical stages of development. These preclinical studies have shown that infantile PLAN mice treated with gene therapy have prolongation of life span with improved body weight and locomotor function.56,57
Stem cells
Delivery of stem cells directly into patients has not been evaluated for safety or efficacy in PLAN. The authors advocate strongly that this should only be administered to children in the context of a clinical trial where there will be regulatory and ethics oversight and no cost to the families.
RT001
RT001 is a di-deuterated form of linoleic acid that has been under investigation for a number of neurodegenerative conditions including infantile PLAN. There is one report of 2 individuals with infantile PLAN who were safely treated with RT001. 58 A larger single-arm open-label study closed but has not published results. Clinicaltrials.gov indicates that of the 19 individuals enrolled, 1 patient died and 63% had serious adverse events during the 1-year study. 59 This medication is not available for individuals with PLAN.
Supplements
Docosahexanoic acid (DHA)
The loss of phospholipase activity in PLAN results in a decrease in DHA and lysophospholipids. There is nonhuman preliminary data supporting a possible benefit of DHA supplementation. 60 Given the low toxicity, DHA supplementation at dosages appropriate for age and weight may be tried in individuals with PLAN but clinical benefit has not been demonstrated.
Vitamin E
Preclinical studies show vitamin E has helped in cellular models of PLAN (https://doi.org/10.1016/j.nbd.2022.105649). However, there are risks associated with excess vitamin E, and further work is needed to determine if this is safe and effective in animal models and humans. What are the benefits and effectiveness of using cannabidiol (CBD) for PLAN? (symptom relief, pain, etc)
Cannabis products have not been specifically studied in PLAN. Cannabis products containing CBD without tetrahydrocannabinol (THC) have low potential for harm, although they are not well studied for symptomatic management of neurodevelopmental disabilities. Epidiolex is the only FDA-approved CBD product (indications are epilepsy due to Lennox-Gastaut syndrome, Dravet syndrome, and tuberous sclerosis). Cannabis products containing THC have data for use in adults with spasticity due to multiple sclerosis 61 and have been studied in a limited way in children with hypertonicity due to neurodevelopmental disabilities.62–66 However, there is a greater likelihood of adverse effects with THC, and products available are not FDA-regulated. Cannabinoids are considered third-line treatment for pain in children with severe neurologic disorders in a clinical report from the section on hospice and palliative medicine, council on children with disabilities. 33 Therefore, we suggest caution in use of these products; however, they may be considered for individuals with severe hypertonicity/pain not responsive to standard treatments.
Footnotes
Author Contributions
JLW made a significant contribution to the design, acquisition, analysis and interpretation of data and drafted and revised the article. AKSS made a significant contribution to the design, acquisition, analysis and interpretation of data and drafted and revised the article. AG made a significant contribution to the concept, design, acquisition, analysis and interpretation of data and drafted and critically revised the article. NN made a significant contribution to the analysis and interpretation of data and critically revised the article. GZ made a significant contribution to the analysis and interpretation of data and critically revised the article. MR made a significant contribution to the design, analysis and interpretation of data and critically revised the article. AH made a significant contribution to the design, analysis and interpretation of data and critically revised the article. DWD made a significant contribution to the design, analysis and interpretation of data and critically revised the article. MT made a significant contribution to the design, analysis and interpretation of data and critically revised the article. SB made significant contribution to the analysis and interpretation of data and critically revised the article. NAS made a significant contribution to the design, analysis and interpretation of data and critically revised the article. PW made a significant contribution to the concept, design, analysis and interpretation of data and critically revised the article. Heather Massey made a significant contribution to the design, analysis and interpretation of data and critically revised the article. AD made a significant contribution to the design, analysis and interpretation of data and critically revised the article. AS made a significant contribution to the design, analysis and interpretation of data and critically revised the article. LP made a significant contribution to the design, analysis and interpretation of data and critically revised the article. PH made a significant contribution to the concept, design, acquisition, analysis and interpretation of data and drafted and critically revised the article. MAK made a significant contribution to the concept, design, acquisition, analysis and interpretation of data and drafted and critically revised the article. SJH made a significant contribution to the concept, design, acquisition, analysis and interpretation of data and drafted and critically revised the article. All authors approved the final version of the article for publication and agreed to be accountable for all aspects of the work and resolved any issues related to its accuracy or integrity.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the INADcure Foundation, the NBIA Disorders Association, Hoffnungsbaum eV, and AISNAF. Manju Kurian is funded by the Sir Jules Thorn Award for Biomedical Research, GOSH Charity, MDBR, Rosetrees Trust, MRC and LifeArc.
Ethical Approval
This study was approved by the OHSU IRB (protocol e8165).
