Abstract
Aim
To evaluate the efficacy and safety of onabotulinumtoxinA for treating upper and lower limb spasticity among pediatric patients in 2 open-label extension trials.
Methods
Patients aged <18 years received ≤5 doses of onabotulinumtoxinA (maximum: 8 U/kg [300 U], cycle 1; 10 U/kg [340 U], cycles 2-5) over 60 weeks. Week 6 efficacy endpoints included mean change from baseline in Modified Ashworth Scale–Bohannon and Modified Tardieu Scale scores, and mean Clinical Global Impression of Overall Change score. Adverse events and laboratory assessments of bone health were monitored.
Results
A total of 580 patients received onabotulinumtoxinA. Modified Ashworth Scale–Bohannon change from baseline ranged from −1.01 to −1.9. Modified Tardieu Scale change from baseline was 13.6 to 18.1 (ankle), 25.8 to 44.1 (elbow), and −5.0 to −26.3 (wrist). Clinical Global Impression of Overall Change scores were 1.5 to 2.2. The most common treatment-emergent adverse events were upper respiratory tract infection (16.9%) and nasopharyngitis (15.7%).
Interpretation
Repeat administration of onabotulinumtoxinA was safe and efficacious for treating upper and lower limb spasticity in children.
Spasticity is classically defined as a neurologically mediated resistance to passive stretch and is characterized by a “velocity-dependent increase in tonic stretch reflexes (‘muscle tone’) with exaggerated tendon jerks, resulting from hyperexcitability of the stretch reflex.” 1 Spasticity can lead to impaired motor function, pain, and sleep disturbances, thereby impacting activity, participation, and quality of life.2,3 The leading cause of pediatric spasticity is cerebral palsy, a group of disorders of movement and posture development that cause activity limitation and are attributed to nonprogressive disturbances that occurred in the fetal or infant brain. 4 Approximately 90% of children with cerebral palsy have some form of spasticity.5,6
Treatment of spasticity is central to the clinical management of children with cerebral palsy, 7 with the primary goal of helping affected children achieve maximal potential development and preventing secondary complications. This is most often achieved with a combination of treatment modalities, including systemic and local pharmacologic treatments as well as surgical and nonsurgical treatments. 8 Intramuscular injection of onabotulinumtoxinA (BOTOX; Allergan plc, Dublin, Ireland) is a standard treatment for spasticity and pain from spasticity/hypertonia in children with cerebral palsy.7,8 When injected intramuscularly at therapeutic doses, onabotulinumtoxinA produces localized chemical denervation of the muscle, resulting in reduced muscle hypertonia for approximately 3 months. Relaxation of hyperactive muscles by onabotulinumtoxinA may augment the effects of physical and occupational therapy, which are commonly implemented to improve strength, balance, and motor function in individuals with cerebral palsy.9,10
Pediatric spasticity has been successfully treated with onabotulinumtoxinA since the early 1990s; however, until recently, most of the data available were from small studies. 11 In 2020, onabotulinumtoxinA was approved in the United States for the treatment of upper limb and lower limb spasticity in children, based primarily on 2 registrational phase 3, multinational, randomized, double-blind, placebo-controlled trials that showed onabotulinumtoxinA to be well tolerated and efficacious for reducing spasticity and improving functional outcomes in children with spasticity due to cerebral palsy or stroke.12,13
We report the results of 2 open-label extension trials that evaluated the long-term side effect profile and efficacy of up to 5 repeated doses of onabotulinumtoxinA for the treatment of pediatric upper limb and lower limb spasticity.
Patients and Methods
Study Design
This was an analysis of 2 multicenter, open-label extension trials (NCT01603615, conducted from October 2012 to September 2018; NCT01603641, conducted from November 2012 to August 2018) that followed 2 registrational phase 3, randomized, double-blind, placebo-controlled trials to evaluate the efficacy and side-effect profile of onabotulinumtoxinA for treatment of upper limb (NCT01603602) and lower limb (NCT01603628) spasticity in children under 18 years of age with cerebral palsy or stroke.12,13 The study design is summarized in Supplemental Figure S1.
The open-label extension trials were designed to further investigate the long-term efficacy and side-effect profile of up to 5 onabotulinumtoxinA treatments, with at least 12 weeks between treatments, in pediatric patients with upper limb and/or lower limb spasticity. In both trials, patients could receive treatment of the upper limb, lower limb, or a combination of both if clinically indicated. Each patient could receive up to 5 doses of onabotulinumtoxinA over 60 weeks. A maximum dose of 8 U/kg (300 U) could be administered in cycle 1 and in cycles 2 to 5 for treatment sessions of only 1 upper limb or lower limb, whereas a maximum dose of up to 10 U/kg (340 U) could be administered in cycles 2 to 5 for treatment of both lower limbs or a combination of upper limb and lower limb. The dose for each muscle and the total dose were determined by the investigator but were within the limits of protocol-defined recommendations. Muscles were injected using muscle localization techniques including e-stimulation, sonography, or electromyography.
Participants could be retreated during the trials if they had a Modified Ashworth Scale–Bohannon score of at least 1 + in the target muscle group(s) intended for injection, at least 12 weeks had elapsed since prior treatment, and there was no indication of unacceptable safety risk.
Patients
Trial patients were enrolled via rollover from the 2 phase 3 trials. Patients included medically stable monoplegic, hemiplegic, or triplegic (upper limb trial only) children (aged 2 to <18 years) with spasticity of the ankle due to cerebral palsy (lower limb trial) or of the upper limb involving the elbow and/or wrist flexor muscles due to cerebral palsy or stroke (upper limb trial). De novo enrollment was also permitted. Patients who did not participate in the double-blind placebo-controlled phase 3 trials could enroll if they were children with cerebral palsy from 2 to less than 18 years of age with an elbow or wrist flexor (upper limb trial) or ankle plantar flexor (lower limb trial) Modified Ashworth Scale score of 2 or more. Complete inclusion and exclusion criteria are listed in Supplemental Table S1.
Outcomes
Efficacy Measurements and Variables
Spasticity outcomes were assessed by calculating the change from baseline (defined as the baseline from the preceding double-blind, placebo-controlled phase 3 trials for rollover patients, and as the last nonmissing observation before receiving the first treatment for de novo patients) in Modified Ashworth Scale–Bohannon and Modified Tardieu Scale scores at each follow-up visit. The Modified Ashworth Scale–Bohannon grades the resistance encountered in a specific muscle group when a limb is passively moved through its range of motion at a study-specified velocity (1 second). The resistance encountered to passive stretch is graded from 0 = no increase in muscle tone to 4 = affected part(s) rigid in flexion or extension. For this analysis, conventional Modified Ashworth Scale–Bohannon scores (0, 1, 1+, 2, 3, 4) were coded on a 0 to 5 scale (Supplemental Table S2). In the lower limb trial, Modified Ashworth Scale–Bohannon was performed on the ankle(s) with knee extended and knee flexed, knee flexors, and other targeted muscles. In the upper limb trial, Modified Ashworth Scale–Bohannon was performed on elbow and wrist flexors at screening. At all other visits, Modified Ashworth Scale–Bohannon was performed on all elbow, pronator, wrist, finger, and thumb flexors and any other muscles that were being treated. The Modified Tardieu Scale is used to assess range of motion and the dynamic component of spasticity. 14 R2 (the angle of passive range of motion) and R1 (the angle at which the stretch reflex of spasticity is activated during fast stretch as per instrument) were assessed at each posttreatment visit using a goniometer according to standard procedures, 14 and differences from baseline in R2, R1, and R2-R1 (an indicator of the dynamic component of spasticity) were calculated. Modified Tardieu Scale testing was performed on the ankle with knee extended in the lower limb trial, and on elbow and wrist muscle groups in the upper limb trial.
Functional outcomes were evaluated using the Clinical Global Impression of Overall Change, the Edinburgh Visual Gait score, and the Goal Attainment Scale. The Clinical Global Impression of Overall Change, a 9-point scale from −4 (very marked worsening) to +4 (very marked improvement), was determined by a physician at each follow-up visit. The Edinburgh Visual Gait score was calculated at each visit at a subset of sites among rollover patients from the lower limb trial with Gross Motor Function Classification System–Expanded and Revised level I to III. Patients’ gait patterns were evaluated using video recording as they walked down a 10-m pathway. The Goal Attainment Scale was used among rollover patients from the upper limb trial to evaluate the level of achievement of patient-specific functional goals on a 6-point scale from −3 (worse than start) to +2 (much more than expected) at each 6-week posttreatment visit and at trial exit. Functional goals, including 1 active and 1 passive goal, were selected at week −2 of the preceding study by the participant and caregiver with guidance from the investigator and/or treating therapist.
Safety Measurements and Variables
Safety assessments consisted of adverse events (including analysis of adverse events representing possible distant spread of toxin) and suicidality assessment using the Columbia-Suicide Severity Rating Scale (for patients ≥6 years of age at day 1). Safety data related to potential effects on long-term muscle and bone health were also collected, including laboratory assessments of bone metabolism (blood glucose, calcium, 25-hydroxyvitamin D, hemoglobin A1c [HbA1c], and bone fraction alkaline phosphatase). Laboratory measures were assessed on day 1 of each study, at 6 weeks after the third treatment, and at study exit. Patients underwent physical examinations and vital sign assessments throughout the study. Adverse events, treatment-emergent adverse events, and possible distant spread of toxin adverse events were described using preferred terms from the Medical Dictionary for Regulatory Activities (MedDRA, version 21.0).
Statistical Analysis
Outcomes were summarized descriptively, including N, mean, standard deviation (SD), median, minimum, and maximum for continuous variables; and N and/or frequency counts and percentages for categorical variables. Missing values were not imputed. For measurements that were not performed in the preceding double-blind, placebo-controlled studies, baseline was defined as the last nonmissing observation before the first treatment in the open-label trial.
Efficacy outcomes were analyzed among observed cases in the modified intent-to-treat population, which included all treated patients who had at least 1 Modified Ashworth Scale–Bohannon score recorded after day 1. Efficacy data were summarized by treatment cycle at each follow-up office visit. Safety outcomes were analyzed in the safety population, which included all patients who received at least 1 onabotulinumtoxinA injection. Safety data were summarized by dose group based on the mean dose received across all cycles within any onabotulinumtoxinA exposure period and for the overall safety population.
Results
Patients and Exposure
A total of 220 patients enrolled in the upper limb extension trial, including 218 from the preceding double-blind, placebo-controlled upper limb study and 2 de novo participants; of these, 213 received onabotulinumtoxinA. A total of 370 patients enrolled in the lower limb extension trial, including 366 from the preceding double-blind, placebo-controlled lower limb study and 4 de novo participants; of these, 367 received onabotulinumtoxinA. In the overall pooled safety population of 580 patients from the 2 trials, 63 patients received a mean dose of <4.5 U/kg of onabotulinumtoxinA across all treatment cycles, 313 patients received a mean dose of ≥4.5 to <7.5 U/kg, and 204 patients received a mean dose of ≥7.5 to ≤10 U/kg.
Patient disposition and baseline demographics are presented in Table 1. The pooled safety population had a mean age of 7.4 years, was 56.6% male, and was racially diverse (White, 61.0%; Asian, 29.3%; Hispanic, 5.3%; Black, 3.1%; other, 1.2%). In the upper limb and lower limb extension trials, 27 (12.7%) and 32 (8.7%) patients, respectively, discontinued the trial before completion. In both studies, the primary reasons for discontinuation were personal reasons and loss to follow-up. In the lower limb trial, 1 patient withdrew because of a serious adverse event (renal cancer, not related to study treatment) and 1 patient withdrew because of lack of efficacy. Most patients were classified as having level I or II functioning based on the Gross Motor Function Classification System–Expanded and Revised (∼80% and ∼95%, respectively, in the upper limb and lower limb extension trials).
Patient Baseline Demographics and Disposition.
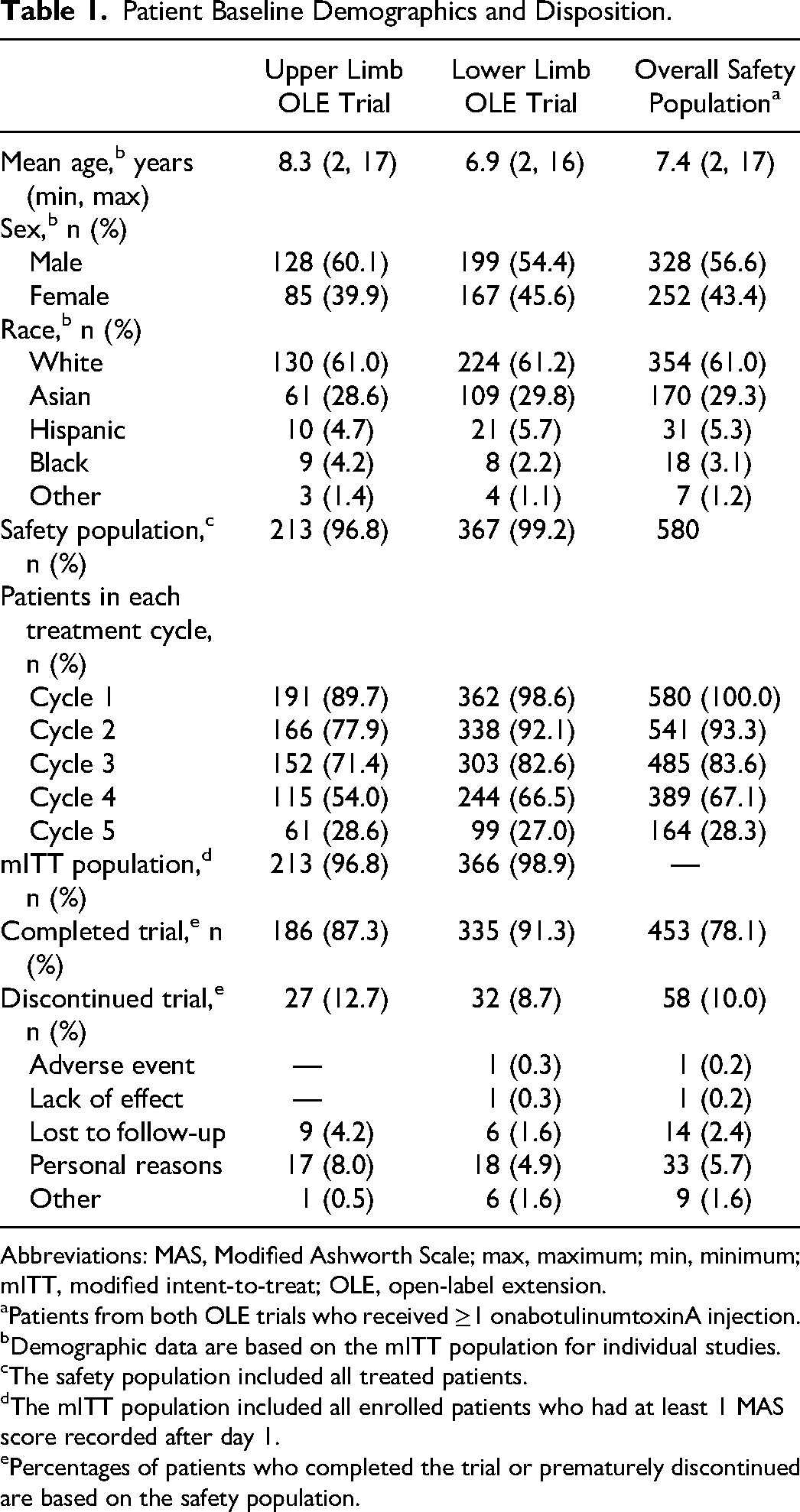
Abbreviations: MAS, Modified Ashworth Scale; max, maximum; min, minimum; mITT, modified intent-to-treat; OLE, open-label extension.
Patients from both OLE trials who received ≥1 onabotulinumtoxinA injection.
Demographic data are based on the mITT population for individual studies.
The safety population included all treated patients.
The mITT population included all enrolled patients who had at least 1 MAS score recorded after day 1.
Percentages of patients who completed the trial or prematurely discontinued are based on the safety population.
Treatment duration and dosage for the pooled safety population are presented in Table 2. Most patients (389/580, 67%) received at least 4 onabotulinumtoxinA treatments. A total of 167 patients received ≥1 treatment of 10 U/kg (or 340 U), and 35 received 4 consecutive treatments of 10 U/kg at 12-week intervals. The mean (SD) total dose injected was 4.1 (1.7) U/kg in the upper limb trial and 6.1 (1.5) U/kg in the lower limb trial. In the pooled safety population, the mean (SD) body weight–adjusted onabotulinumtoxinA dose (U/kg) administered among all patients (N = 580) across treatment cycles 1 through 5 ranged from 6.7 (1.6) U/kg to 7.5 (2.3) U/kg; the mean (SD) onabotulinumtoxinA dose (U) ranged from 171.2 (71.7) U at treatment cycle 1 to 213.7 (83.7) U at treatment cycle 5 (Table 2). Most patients received retreatment at week 12 for all treatment cycles. The median overall reinjection time for patients was every 12-13 weeks for upper limb or lower limb spasticity regardless of treatment cycle; however, in some cycles, some patients had a longer duration of response (ie, up to 24 weeks).
Onabotulinumtoxina Treatment Use by Treatment Cycle, Pooled Safety Population a .
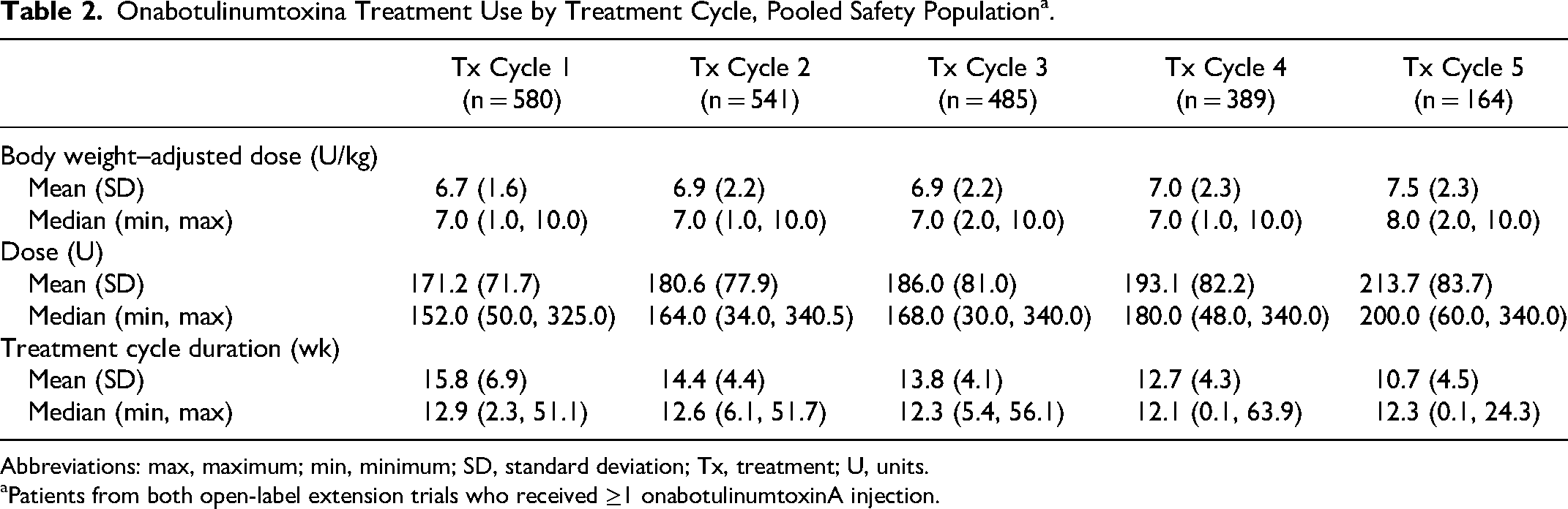
Abbreviations: max, maximum; min, minimum; SD, standard deviation; Tx, treatment; U, units.
Patients from both open-label extension trials who received ≥1 onabotulinumtoxinA injection.
Outcomes
Spasticity measures
At baseline, the mean coded Modified Ashworth Scale–Bohannon score of principal upper limb muscle group was 3.1 and the ankle score of lower limb(s) with knee extended was 3.5, indicating a marked increase in muscle tone and difficulty performing passive movement. With open-label onabotulinumtoxinA treatment, upper limb and lower limb coded Modified Ashworth Scale–Bohannon scores improved from baseline and showed sustained improvement across repeat cycles (Figure 1). In the upper limb trial, the mean reduction from baseline at week 6 ranged from −1.3 to −1.9 across all 5 treatment cycles, resulting in a coded Modified Ashworth Scale–Bohannon score of 1.5 to 1.9. In the lower limb trial, the point reduction from baseline in week 6 ranged from −1.01 to −1.45 at any time point across all 5 treatment cycles, resulting in a coded Modified Ashworth Scale–Bohannon score of 2.2 to 2.5 and indicating a descriptive improvement in muscle tone and range of motion. The majority of patients in both trials reported at least a 1-grade reduction in Modified Ashworth Scale–Bohannon score from baseline at week 6 across all cycles.
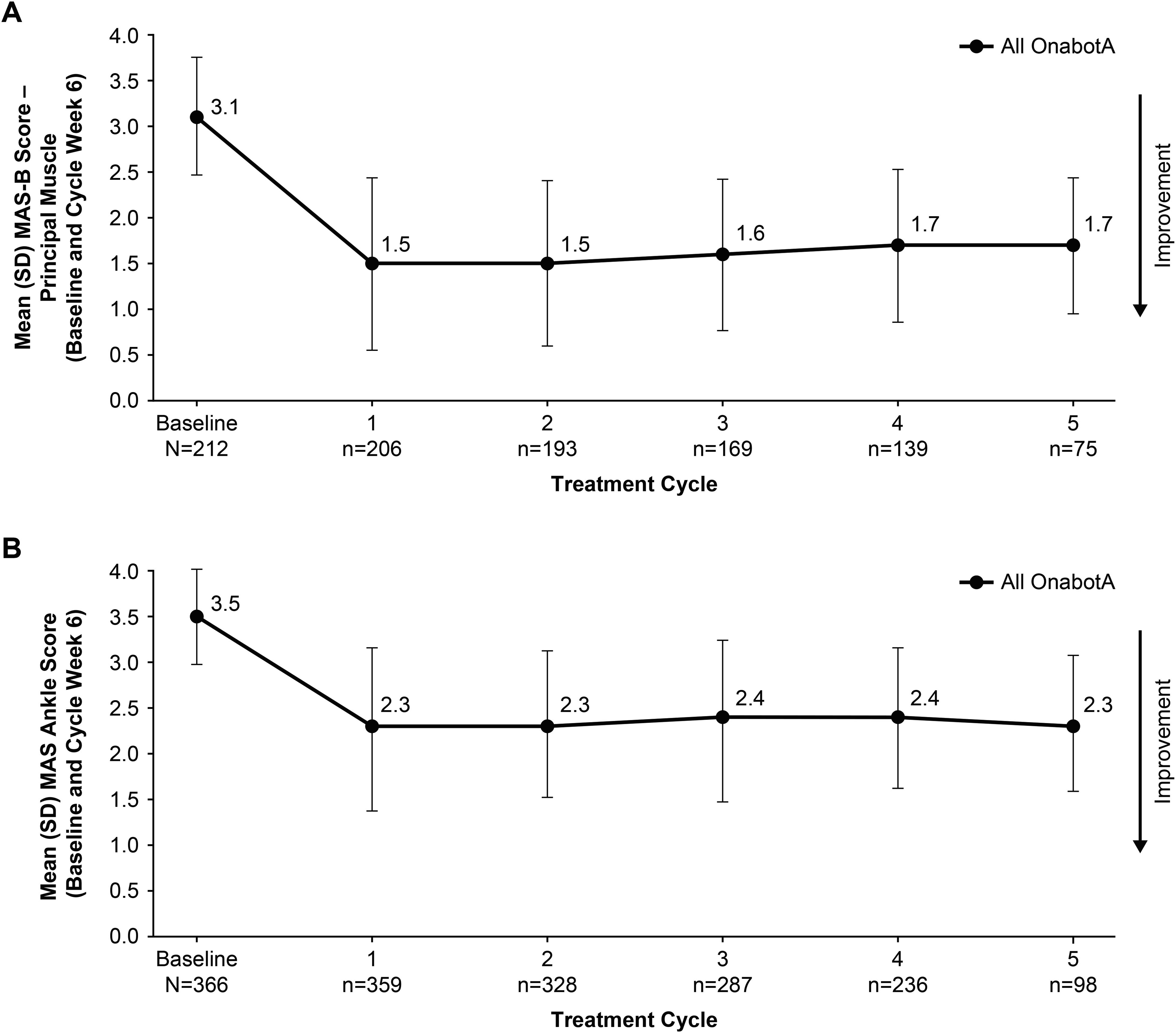
Reduction in muscle tone with repeated treatment cycles of onabotulinumtoxinA, as measured by MAS-B for (A) upper limb and (B) lower limb spasticity. Data shown were collected at week 6 for the mITT population following treatment for each cycle. MAS-B, Modified Ashworth Scale–Bohannon; mITT, modified intent-to-treat; onabotA, onabotulinumtoxinA; SD, standard deviation.
Modified Tardieu Scale scores in both trials showed numerical improvement compared with baseline for R1, R2, and R2-R1 at most time points. These improvements were consistent across repeat treatment cycles (Figure 2). In the upper limb trial, week 6 change from baseline in R2-R1 across all 5 treatment cycles was 25.8 to 44.1 degrees for the elbow and −5.0 to −26.3 degrees for the wrist (starting position 0 degrees), indicating consistent improvement in the dynamic component of spasticity. In addition, change from baseline in R2 (passive range of motion) across all 5 treatment cycles in the upper limb trial ranged from −0.8 to −8.3 degrees for the elbow (starting position 90 degrees), and −3.6 to 31.5 degrees for the wrist, indicating improved passive range of motion. In the lower limb trial, week 6 change from baseline in ankle R2-R1 ranged from 13.6 to 18.1 degrees at any time point across all 5 treatment cycles, indicating consistent improvement in range of motion. In addition, week 6 change from baseline in ankle R2 in the lower limb trial ranged from 0.95 to 4.61 degrees (starting position 90 degrees) at any time point across all 5 treatment cycles.
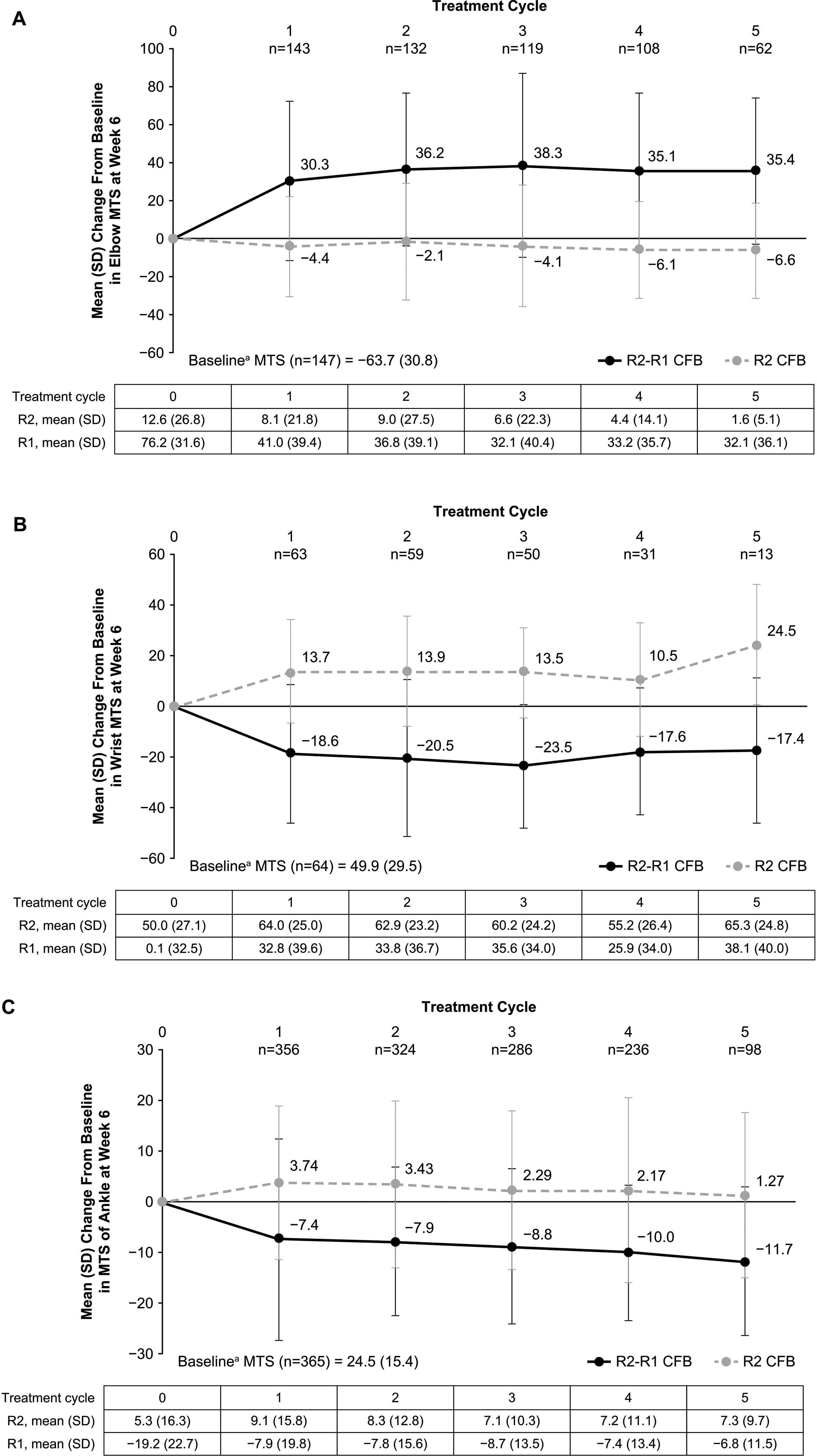
Consistent improvement in range of motion and dynamic component of spasticity measured by MTS week 6 change from baseline in R2 and R2-R1 for A) elbow (starting position 90°, full extension 0°), (B) wrist (starting position 0°, full extension 90°), and (C) ankle (starting position 90°, full flexion ∼45°) in the mITT populations. Error bars represent standard deviations. MTS ankle score was assessed with knee extended. Because of differences in starting angle/position and direction of movement during testing, improvement of spasticity from baseline is indicated by increasing values of R2-R1 for elbow and by decreasing values of R2-R1 for wrist and ankle. mITT, modified intent-to-treat; MTS, Modified Tardieu Scale; onabotA, onabotulinumtoxinA; SD, standard deviation.
Functional Measures
Mean Clinical Global Impression of Overall Change scores for onabotulinumtoxinA-treated patients in both trials (from 1.8 to 2.2 in the upper limb trial and from 1.5 to 2.1 in the lower limb trial at any time point across all 5 treatment cycles) indicated slight to moderate improvement that was sustained across repeat treatment cycles (Figure 3). Most patients (from 84.7% to 100% in the upper limb trial and from 72.7% to 94.8% in the lower limb trial, depending on treatment cycle and dosage) had a Clinical Global Impression of Overall Change score of 1 or higher across repeat cycles, indicating that improvement was carried forward and maintained from the respective double-blind, placebo-controlled trials.
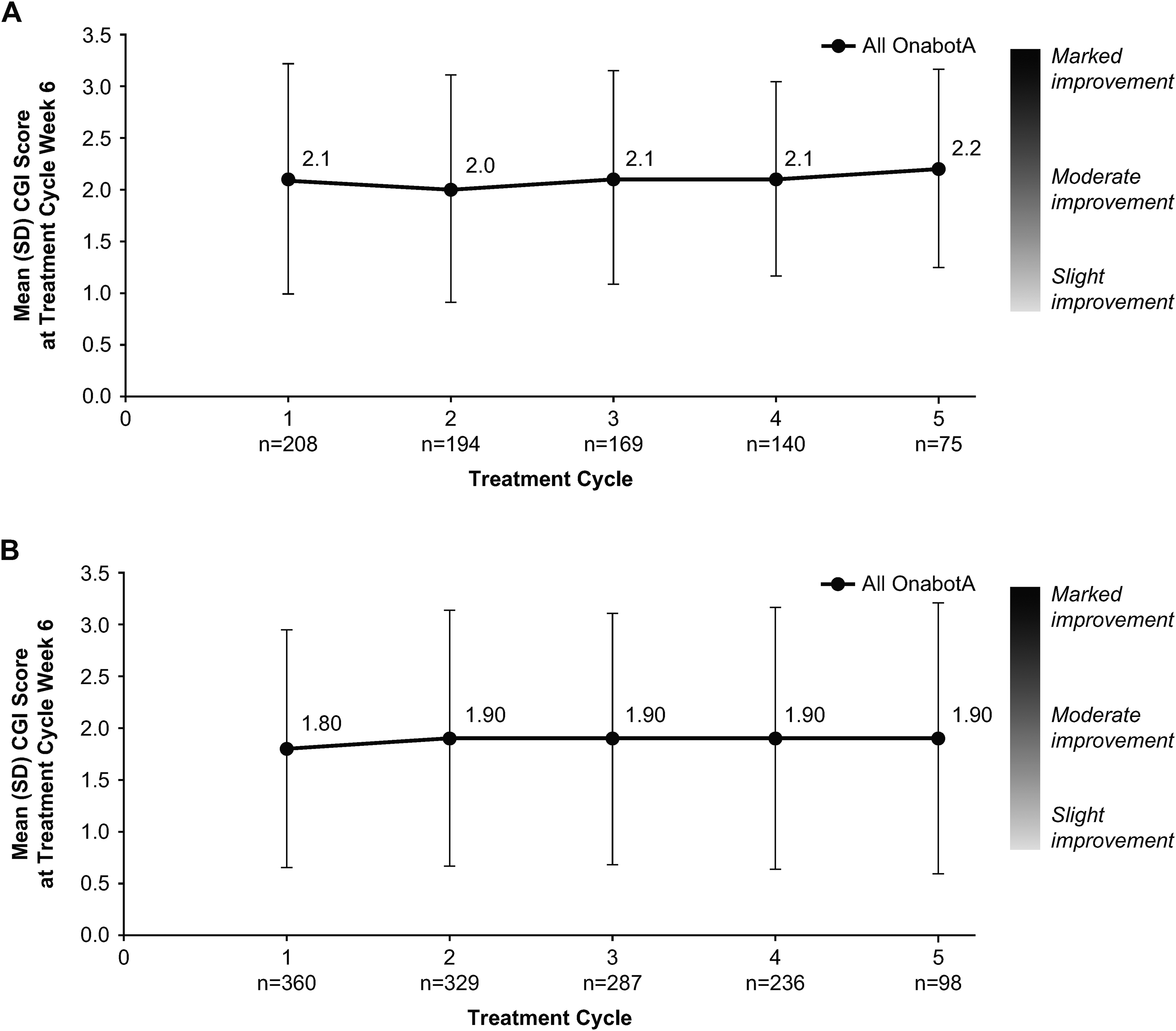
Mean week 6 CGI scores with onabotulinumtoxinA retreatment for the (A) upper limb and (B) lower limb open-label extension studies, mITT populations. The CGI, a 9-point scale with scores ranging from −4 (very marked worsening) to +4 (very marked improvement), was completed by a physician at each follow-up visit. CGI, Clinical Global Impression of Overall Change; mITT, modified intent-to-treat; onabotA, onabotulinumtoxinA; SD, standard deviation.
Edinburgh Visual Gait scores showed improvement compared with baseline at most time points for both treatment groups in the lower limb trial; findings were consistent and sustained across repeat cycles (Supplemental Table S3). Mean (SD) change from baseline in total Edinburgh Visual Gait scores at week 6 was −2.3 (2.9) for cycle 1 (n = 46), −2.4 (3.5) for cycle 2 (n = 35), −2.6 (3.3) for cycle 3 (n = 35), −2.8 (3.3) for cycle 4 (n = 24), and −6.8 (2.5) for cycle 5 (n = 4).
Goal Attainment Scale scores for active and passive goals in the upper limb trial indicated that goals were achieved, with most patients reporting a score of 0 (ie, attained the defined therapeutic goal) or better. Most patients showed sustained and consistent improvements in Goal Attainment Scale scores across repeat cycles (Supplemental Table S4). Across treatment cycles, more than 80% of patients were Goal Attainment Scale responders (Goal Attainment Scale score ≥0) for active and passive goals (Figure 4).
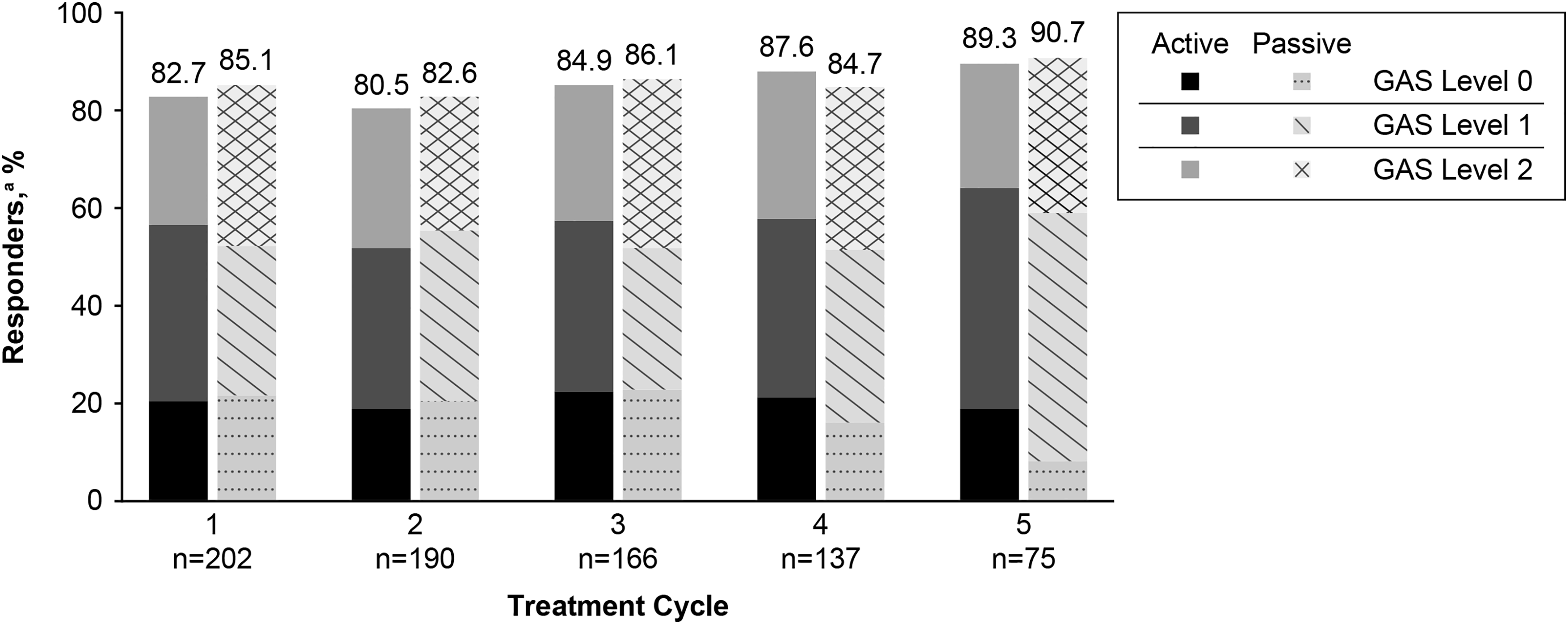
Percentage of GAS responders in the rollover patient mITT population; responder defined as active or passive GAS score ≥0. Level 0 = attained the defined therapeutic goal; level +1 = improvement slightly exceeded the defined therapeutic goal; level +2 = improvements clearly exceeded the defined therapeutic goal. Data shown were collected at week 6 following treatments for each cycle. GAS, Goal Attainment Scale; mITT, modified intent-to-treat.
Safety measures
The adverse event profile by total body onabotulinumtoxinA dose is summarized in Table 3. Overall, 374 of 580 (64.5%) patients reported a treatment-emergent adverse event during the trials, most commonly upper respiratory tract infection (16.9%) and nasopharyngitis (15.7%). Treatment-emergent adverse events decreased with each treatment cycle (Figure 5). The incidence of serious adverse events across the pooled analysis was 6.7% (n = 39), with no dose response (Supplemental Table S5). No serious adverse event was considered treatment related, no deaths occurred, and there were no reports of suicidal ideation.
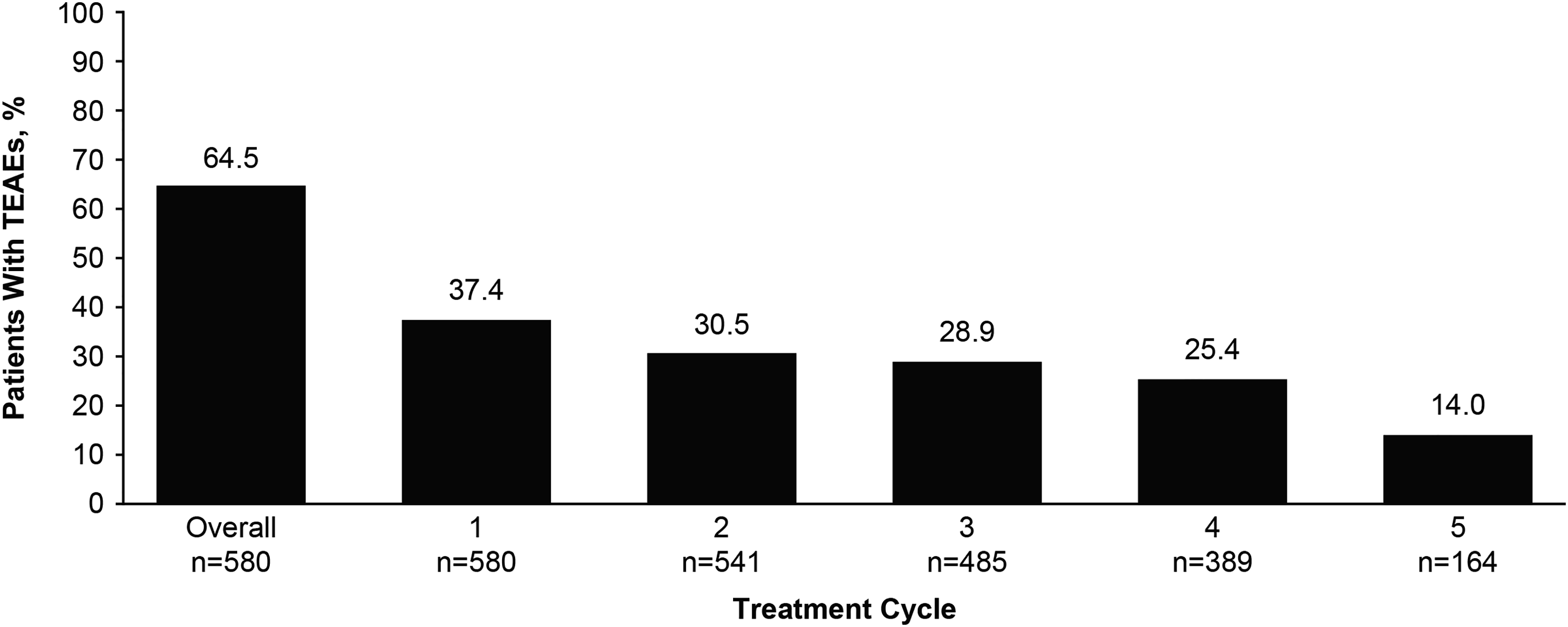
Decrease in TEAEs by retreatment cycle. TEAE, treatment-emergent adverse event.
Adverse Event Profile by Overall Total Body OnabotulinumtoxinA Dose,a Pooled Safety Population.
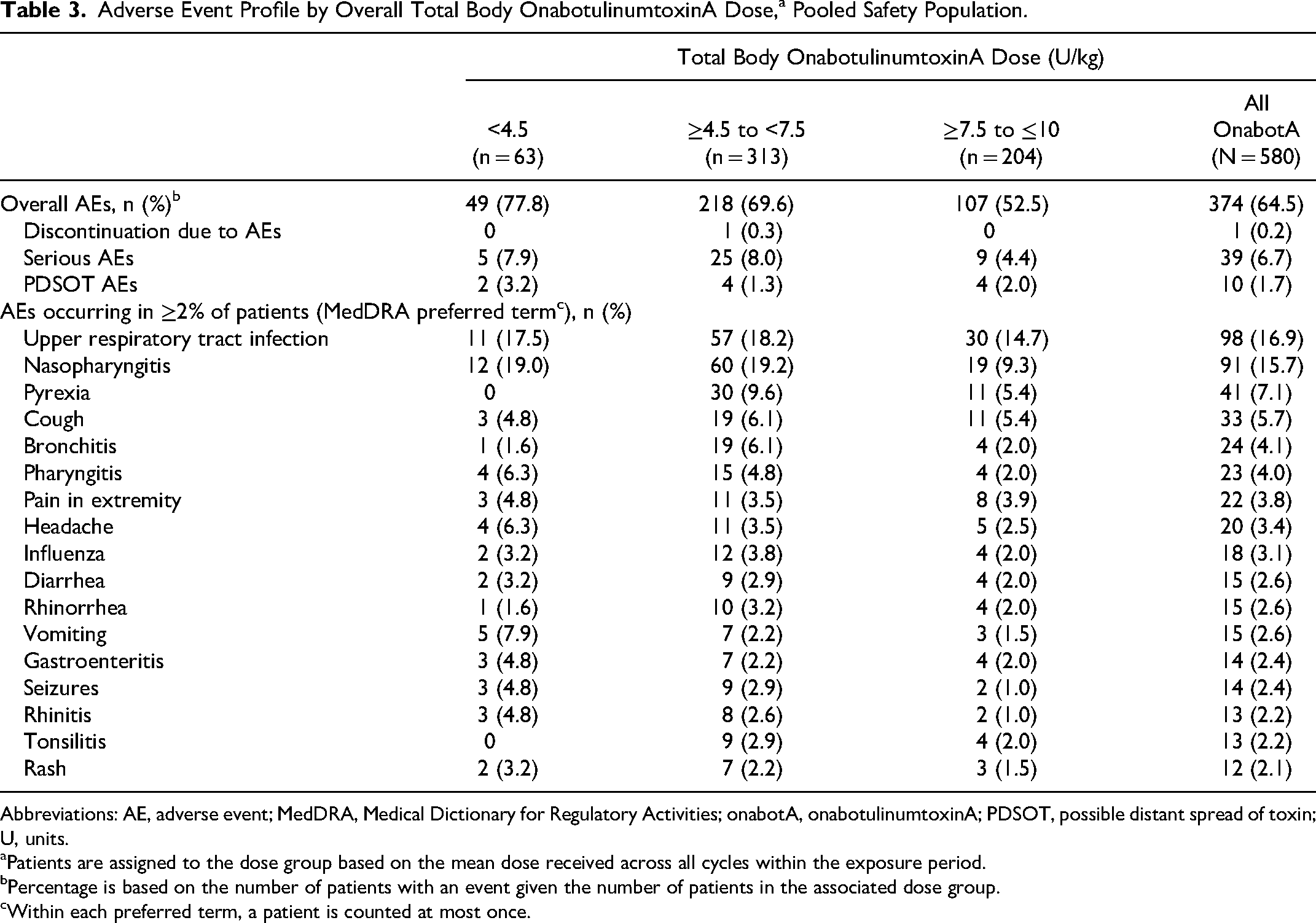
Abbreviations: AE, adverse event; MedDRA, Medical Dictionary for Regulatory Activities; onabotA, onabotulinumtoxinA; PDSOT, possible distant spread of toxin; U, units.
Patients are assigned to the dose group based on the mean dose received across all cycles within the exposure period.
Percentage is based on the number of patients with an event given the number of patients in the associated dose group.
Within each preferred term, a patient is counted at most once.
Ten of 580 patients (1.7%) reported a treatment-emergent adverse event that could indicate distant spread of toxin: muscular weakness (n = 7, 1.2%), constipation (n = 2, 0.3%), and facial paralysis (n = 1, 0.2%). The incidence of these events was not dose dependent. On review of individual cases, the events were found to be either local effect (ie, near the injection site) or confounded by other factors (eg, ongoing medical conditions, concomitant medications). None of the possible distant spread of toxin treatment-emergent adverse events were considered true distant spread of toxin effects, none were serious, all were mild or moderate in severity, and all resolved without sequelae.
No muscle atrophy events (eg, peroneal muscular atrophy, progressive muscular atrophy) were reported. Reported adverse events that may be indicative of effect on muscle function included muscular weakness (7 [1.2%]), fall (4 [0.7%]), and contusion (7 [1.2%]) (Table 4). The incidences of these events were low and not dose dependent.
Patients With TEAEs Associated With Muscle Function and Bone Health by Mean OnabotulinumtoxinA Dose, Pooled Safety Population.
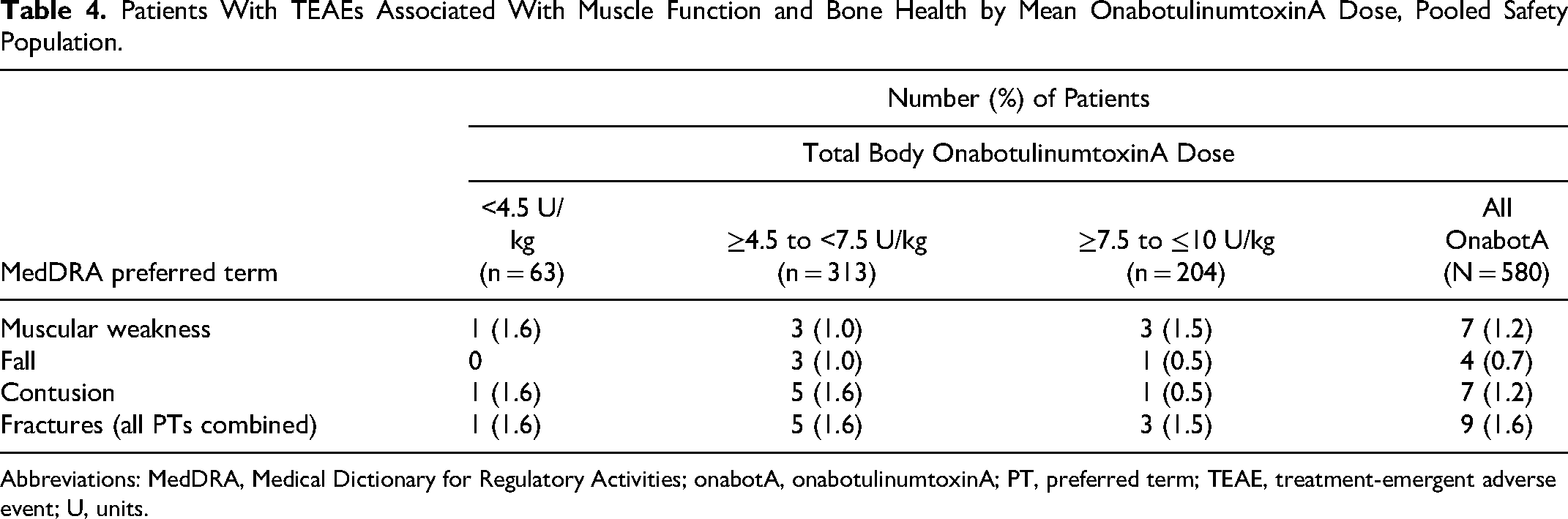
Abbreviations: MedDRA, Medical Dictionary for Regulatory Activities; onabotA, onabotulinumtoxinA; PT, preferred term; TEAE, treatment-emergent adverse event; U, units.
There were no clinically meaningful changes from baseline in laboratory indicators of bone metabolism (Supplemental Table S6), and the frequency of potentially clinically significant postbaseline values for these laboratory parameters was low (Table 5). The incidence of fracture events, an indicator of poor bone health, was low and not dose dependent (Table 4).
Frequency of Bone-Related Postbaseline Potentially Clinically Significant Laboratory Values for Patients With Normal Baseline Values by Mean OnabotulinumtoxinA Dose, Pooled Safety Population.

Abbreviations: H, high; onabotA, onabotulinumtoxinA; U, units.
Total patients included are those with a baseline laboratory value in the normal range and at least 1 postbaseline value in any treatment cycle.
Patients are assigned to the dose group based on the mean dose received across all cycles within the exposure period.
Discussion
Data from these 2 open-label extension trials show that, in children with upper limb or lower limb spasticity due to cerebral palsy or stroke, improvements in muscle tone and global treatment response with onabotulinumtoxinA observed in the double-blind placebo-controlled trials were maintained with up to 5 repeated treatments. Patients had sustained improvements in efficacy measures that remained generally consistent across repeat treatment cycles. Strengths of this study include its prospective, long-term design; large, diverse sample of patients; and adjustment of dosing and muscle treatment paradigms to optimize individual needs, which is more reflective of real-world clinical practice than the fixed-dose, fixed-muscle injection paradigm used in the double-blind, placebo-controlled trials.
Limited evidence is available for the effect of onabotulinumtoxinA on measured change in gross motor function and daily activities. The study focused on limited specific passive and active functional goals among patients with upper limb spasticity set during the preceding double-blind, placebo-controlled phase 3 study by the patient and family with input from the physician investigator and/or treating therapist. Focusing on these specific goals ensured that the goals being assessed were important to the family, were practiced at home, and were not “diluted” by being evaluated many times via a standardized functional assessment score. In this analysis, more than 80% of patients were Goal Attainment Scale responders for their predetermined active and passive goals.
OnabotulinumtoxinA treatment was generally well tolerated during these long-term extension trials, with low rates of discontinuation. Adverse events occurred at a similar rate in all treatment groups for each cycle and generally decreased with subsequent treatment cycles. The most common adverse events were upper respiratory tract infection and nasopharyngitis, consistent with common transmittable illnesses observed in preschool- and school-aged children or with the known safety profile of onabotulinumtoxinA in patients with spasticity. Only 1 patient discontinued due to a serious adverse event (renal cancer). Serious adverse events were infrequent, none were considered related to onabotulinumtoxinA treatment by the treating physicians, and there were no reports of suicidal ideation. Safety findings were consistent with the results of the double-blind, placebo-controlled trials; no new safety concerns were identified.
The tested doses of onabotulinumtoxinA were well tolerated. A small number of patients (1.7%) reported a treatment-emergent adverse event that could indicate distant spread of toxin; however, on medical evaluation of each possible distant spread of toxin case by the sponsor safety team, none of these events were deemed to represent true distant-spread-of-toxin effects because the events were localized to the site of study drug injection, confounded by concomitant medications or underlying medical conditions, or not consistent with the mechanism of action of onabotulinumtoxinA. Similarly, no serious effects on bone health were observed, as evidenced by low incidence of adverse events as well as lack of dose-dependent effects and minimal change from baseline in muscle- and bone-related laboratory analytes. In addition, the percentage of patients with fracture events in the current study (2%) was lower than that reported in the published literature for children with cerebral palsy (12%-16%),15,16 and physical examination results were unremarkable over approximately 60 weeks of continuous onabotulinumtoxinA treatment.
This study had several limitations to consider. By nature, open-label trials lack a placebo group for comparison. As these studies were extensions of the registrational studies, efficacy outcomes were selected to fulfill regulatory requirements to support labeling and may not necessarily reflect the approach taken in clinical practice. Results are generalizable to children with cerebral palsy with Gross Motor Function Classification System–Expanded and Revised levels of I to III, but it is unknown how these results extrapolate to the more severely affected population, as very few study patients had Gross Motor Function Classification System–Expanded and Revised level IV. Although the improvement in passive range of motion (R2) over time in treated muscles was encouraging, we do not have detailed information on adjunct treatments that can impact R2 measurements, such as serial casting/splinting. 17 In addition, we did not collect information on R2 changes involving joints of non-injected muscle groups. Although experimental animal studies have demonstrated a fibrotic response with repeated injections of botulinum toxin A, 18 this study also did not include specific assessments for muscle atrophy or fibrosis. However, prior studies have shown that muscle atrophy does not occur to a significant extent in adults and children treated with onabotulinumtoxinA and may be offset by compensatory hypertrophy of neighboring muscles.19–21 Additional studies with longer follow-up times could provide insight regarding the long-term impact on potential muscle atrophy or fibrosis.
Conclusion
These findings demonstrated that onabotulinumtoxinA was an effective and well-tolerated long-term treatment for upper limb and lower limb spasticity in children. Dosing in this study was adjusted according to individual patient needs to mirror real-world usage and provided noticeable improvements in passive and dynamic range of motion, tolerance to stretching protocols, and attainment of desired functional goals among patients with Gross Motor Function Classification System–Expanded and Revised levels of I to III. Safety findings were consistent with the phase 3 registration trials, no new safety signals were identified, and no indications of long-term negative effects on muscle and bone were observed. These results highlight the favorable benefit-risk profile of onabotulinumtoxinA treatment for children with upper limb and lower limb spasticity.
Supplemental Material
sj-doc-1-jcn-10.1177_08830738241288431 - Supplemental material for Efficacy and Safety of OnabotulinumtoxinA for the Treatment of Pediatric Upper and Lower Limb Spasticity: Results From 2 Open-Label, Long-term Extension Trials
Supplemental material, sj-doc-1-jcn-10.1177_08830738241288431 for Efficacy and Safety of OnabotulinumtoxinA for the Treatment of Pediatric Upper and Lower Limb Spasticity: Results From 2 Open-Label, Long-term Extension Trials by Mark Gormley, Darcy Fehlings, Heakyung Kim, Marcin Bonikowski, Marta Banach, Fatma Gul, Jill Meilahn, Brad Racette, Nuoyu Huang, Xiaomeng Niu, Lynn M. James and Rozalina Dimitrova in Journal of Child Neurology
Supplemental Material
sj-docx-2-jcn-10.1177_08830738241288431 - Supplemental material for Efficacy and Safety of OnabotulinumtoxinA for the Treatment of Pediatric Upper and Lower Limb Spasticity: Results From 2 Open-Label, Long-term Extension Trials
Supplemental material, sj-docx-2-jcn-10.1177_08830738241288431 for Efficacy and Safety of OnabotulinumtoxinA for the Treatment of Pediatric Upper and Lower Limb Spasticity: Results From 2 Open-Label, Long-term Extension Trials by Mark Gormley, Darcy Fehlings, Heakyung Kim, Marcin Bonikowski, Marta Banach, Fatma Gul, Jill Meilahn, Brad Racette, Nuoyu Huang, Xiaomeng Niu, Lynn M. James and Rozalina Dimitrova in Journal of Child Neurology
Footnotes
Acknowledgments
The authors sincerely thank the patients who participated in the study and acknowledge the investigators and staff who contributed to the study conduct. The authors acknowledge medical writing support provided by Yvette M. Edmonds, PhD, of Peloton Advantage, LLC, an OPEN Health company, which was funded by AbbVie. Neither honoraria nor any other form of compensation was provided for authorship.
Author Roles
RD, MG, JM, and LMJ designed the study. MB, MG, FG, JM, and BR were the study investigators. MB, MG, FG, JM, and BR enrolled patients. MB, MG, and JM collected and assembled the data. XN performed the data analysis. RD and LMJ were responsible for data interpretation and manuscript preparation.
All authors took part in manuscript review and revisions and approved the final version of the manuscript.
Data Availability statement
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual, and trial-level data (analysis data sets), as well as other information (eg, protocols, clinical study reports, or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications. These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP), and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the US and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: https://vivli.org/ourmember/abbvie/ then select “Home.”
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Mark Gormley is an advisory board member and serves on the speaker's bureau for Allergan, an AbbVie Company. Heakyung Kim has received an honorarium from AAPM&R and is an advisory board member and consultant for Allergan, an AbbVie company, Ipsen, Merz, and Pacira Biosciences, Inc. Marcin Bonikowski serves as advisory board member, consultant, and speaker for Allergan, an AbbVie company, Ipsen, and Merz. Marta Banach serves as a speaker for Allergan, an AbbVie company, Ipsen, Kedrion, Merz, and Shire and is an advisory board member and consultant for Merz. Fatma Gul receives research support from AbbVie, Ipsen, Merz, and Supernus, and is a consultant to Medisys Consultants. Jill Meilahn receives research support and dedicated research time funding through Marshfield Clinic Research Institute. Darcy Fehlings and Brad Racette have no interests that might be perceived as posing a conflict or bias. Nuoyu Huang, Xiaomeng Niu, Lynn M. James, and Rozalina Dimitrova are employees of AbbVie and may hold AbbVie stock.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Allergan (prior to its acquisition by AbbVie) funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship.
Ethical Approval
Trial protocols were approved by a properly constituted Institutional Review Board (IRB) or Independent Ethics Committee (IEC) before trial initiation. Studies were conducted in conformance with the International Council for Harmonisation E6 guideline for Good Clinical Practice and the principles of the Declaration of Helsinki. Written informed consent and minor assent (as applicable) were obtained at the first trial visit, before any trial-related procedures or change in treatment.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
