Abstract
Introduction
Little is known about the longitudinal trajectory of brain growth in children with opsoclonus-myoclonus ataxia syndrome. We performed a longitudinal evaluation of brain volumes in pediatric opsoclonus-myoclonus ataxia syndrome patients compared with age- and sex-matched healthy children.
Patients and Methods
This longitudinal case-control study included brain magnetic resonance imaging (MRI) scans from consecutive pediatric opsoclonus-myoclonus ataxia syndrome patients (2009-2020) and age- and sex-matched healthy control children. FreeSurfer analysis provided automatic volumetry of the brain. Paired t tests were performed on the curvature of growth trajectories, with Bonferroni correction.
Results
A total of 14 opsoclonus-myoclonus ataxia syndrome patients (12 female) and 474 healthy control children (406 female) were included. Curvature of the growth trajectories of the cerebral white and gray matter, cerebellar white and gray matter, and brainstem differed significantly between opsoclonus-myoclonus ataxia syndrome patients and healthy control children (cerebral white matter, P = .01; cerebral gray matter, P = .01; cerebellar white matter, P < .001; cerebellar gray matter, P = .049; brainstem, P < .01).
Discussion/Conclusion
We found abnormal brain maturation in the supratentorial brain, brainstem, and cerebellum in children with opsoclonus-myoclonus ataxia syndrome.
Keywords
Opsoclonus-myoclonus ataxia syndrome, known as “dancing eye syndrome,” is a rare acute or subacute inflammatory disorder of the central nervous system that affects children at an average age of 18 months. 1 The estimated annual incidence in the United Kingdom is 0.18 new cases per million total population per year. 2 Clinical characteristics include the classic triad of ataxia, opsoclonus (an abnormality in eye movements consisting of involuntary and chaotic eye movements that change the point of fixation in all directions, without a period of time between the fixation points), and myoclonus. Irritability, sleep disturbances, and other behavioral abnormalities occur frequently, often pre-dating other neurologic manifestations.1,3–5 More than half of the cases are associated with neuroblastoma, and the remainder are presumed to be postviral, although no specific viral agent has been closely associated with opsoclonus-myoclonus ataxia syndrome. Ongoing symptoms and deficits may be observed in up to 60% of patients; however, whether these represent ongoing inflammatory activity or deficits due to central nervous system injury remains unclear.6,7
Knowledge of the long-term structural abnormalities of the brain associated with opsoclonus-myoclonus ataxia syndrome is limited. Cross-sectional studies of brain magnetic resonance imaging (MRI) changes in children with opsoclonus-myoclonus ataxia syndrome report no structural or leptomeningeal abnormalities on MRI examinations of the brain in the acute phase of the disease despite the presence of marked neurologic symptoms, 3 with some studies reporting cerebellar atrophy at follow-up in up to one-third of patients.8,9 One cross-sectional quantitative study evaluated brain volumes in 9 children with opsoclonus-myoclonus ataxia syndrome (mean 12.5 years from clinical onset), compared with 10 healthy children, and documented lower cerebellar gray matter volume (vermis, bilateral flocculonodular lobes, and bilateral cerebellar hemispheres) and motor and visual cortex volumes in children with opsoclonus-myoclonus ataxia syndrome. 10 The authors further evaluated the associations between atrophy and the presence of symptoms and reported a 65% reduction in the total cerebellar gray matter volume in the symptomatic opsoclonus-myoclonus ataxia syndrome group compared with asymptomatic opsoclonus-myoclonus ataxia syndrome, suggesting a possible structure-function correlation.
Notably, despite ongoing concerns about residual disease activity beyond the acute phase in many children and the documentation of cognitive and motor sequelae in most patients, little information is available regarding the longitudinal trajectory of brain volumes in this population. We therefore aimed to longitudinally assess regional brain volume trajectories in consecutive children presenting with opsoclonus-myoclonus ataxia syndrome in comparison with a cohort of age- and sex-matched children with normal clinical MRI examinations of the brain. We hypothesized that brain volumes in opsoclonus-myoclonus ataxia syndrome including supratentorial gray matter and white matter as well as cerebellar white matter and cortex would show significant differences in growth trajectory from healthy children.
Methods
Participants
Children with a history of opsoclonus-myoclonus ataxia syndrome were consecutively recruited from the Pediatric Neuroinflammatory Disorders Clinic at the Hospital for Sick Children in Toronto, Ontario, Canada (2012-2023). This was a convenience sample of eligible participants with opsoclonus-myoclonus ataxia syndrome attending a specialized neuroinflammatory disorders clinic at a large tertiary care center; all patients were included if they had MRI scans available to reduce the possibility of bias. Inclusion criteria for the opsoclonus-myoclonus ataxia syndrome participants were (1) patients <18 years of age with a history of opsoclonus-myoclonus ataxia syndrome and (2) ≥1 MRI examination with a non–contrast-enhanced 3-D T1-weighted sequence at different time points of the disease course. Exclusion criteria were the presence of coexisting neurologic abnormalities or other medical comorbidities.
Children from a control cohort without significant abnormalities on clinical brain MRI and no underlying neurologic condition aged <18 years at the time of MRI were matched for age and sex to the opsoclonus-myoclonus ataxia syndrome group. Participants were included in this control cohort if they had undergone MRI scans requested for transient neurologic complaints such as headache and MRI examinations were reported as normal with subsequent discharge from neurologic care. Each control individual underwent a single scan at a single time point.
Standard Protocol Approvals, and Patient Consents
This study was approved by the institutional research ethics board at SickKids hospital. Informed consent was obtained for the opsoclonus-myoclonus ataxia syndrome group; however, our ethics board waived the requirement for consent from the control group because of the retrospective nature and large sample size.
MRI and Clinical Data
The collected clinical data included demographics, date of disease onset, date of diagnosis, and other relevant clinical information including treatment and diagnostic workup. The Mitchell and Pike OMS Rating Scale 3 was used to evaluate disease severity and functional recovery, and children were classified into those who had an underlying tumor and those who did not. We documented the use of acute therapies such as steroids, intravenous immunoglobulin and plasma exchange, and further identified children who received therapies such as rituximab within 6 months of symptom onset.
Deidentified 3D T1-weighted MRI sequences were made available from our local picture archiving and communication system (PACS) and were processed using FreeSurfer version 7 (Massachusetts General Hospital, Harvard Medical School; http://surfer.nmr.mgh.harvard.edu). 11 To manage image processing, we used in-house web-based data management software running on a local server (Lenovo Legion C730 with an i9 processor and 32 GB of RAM; Lenovo, Quarry Bay, Hong Kong). Technical details of the FreeSurfer pipeline have been described previously.11–13 Anatomical regions of interest included the cerebral white matter, cerebral gray matter, cerebellar white matter, cerebellar gray matter, and brainstem. Secondary to overlaying the neuroanatomical masks on the 3D T1-weighted MRI sequence, all regions of interest were visually inspected for the accuracy of alignment between the mask and the MRI.
Statistical Analysis
Volumes were charted as a function of age for both groups. We then fitted the charted data to a second-order polynomial, which allowed us to quantify how volumes of opsoclonus-myoclonus ataxia syndrome patients’ brain regions changed relative to those of controls. Paired t tests were performed on the curvature parameter of the second-order polynomial to determine whether there were significant differences between opsoclonus-myoclonus ataxia syndrome patients and controls. Statistical significance was set at P <.05. All P values were Bonferroni-corrected to avoid coincidental findings.
Data access
The principal author has full access to the data used in the analyses in this manuscript.
Data availability
All data are available by request from qualified investigators.
Results
Demographics
Fourteen pediatric patients with opsoclonus-myoclonus ataxia syndrome (12 girls, 2 boys; median age at clinical onset 1.6 years [IQR 1.4-1.9]) were included in this study. The baseline demographics and clinical characteristics are summarized in Table 1. The median number of MRI examinations per patient was 3 (IQR 1-4) for the opsoclonus-myoclonus ataxia syndrome group. The median disease duration was 1.02 years (IQR 0.4-3.4). Paraneoplastic opsoclonus-myoclonus ataxia syndrome was observed in 8 of 14 patients. All children received steroids and/or intravenous immunoglobulin within 6 months of symptom onset. All but one received therapy within 2 months of symptom onset. Twelve of 14 patients received rituximab. All patients receiving rituximab received it within 6 months of symptom onset. Two children who did not receive rituximab had neuroblastoma, and one had quick resolution of symptoms after steroid administration with follow-up surgery shortly after steroid administration. The other experienced delayed discovery of neuroblastoma 4 months after diagnosis in the context of relapse, with resection and symptom resolution thereafter. The control group comprised 474 individuals (406 girls and 68 boys). The median age at scan for the control cohort was 11.2 years (IQR 7.4-14.9).
Baseline Demographic and Clinical Characteristics (N = 488).
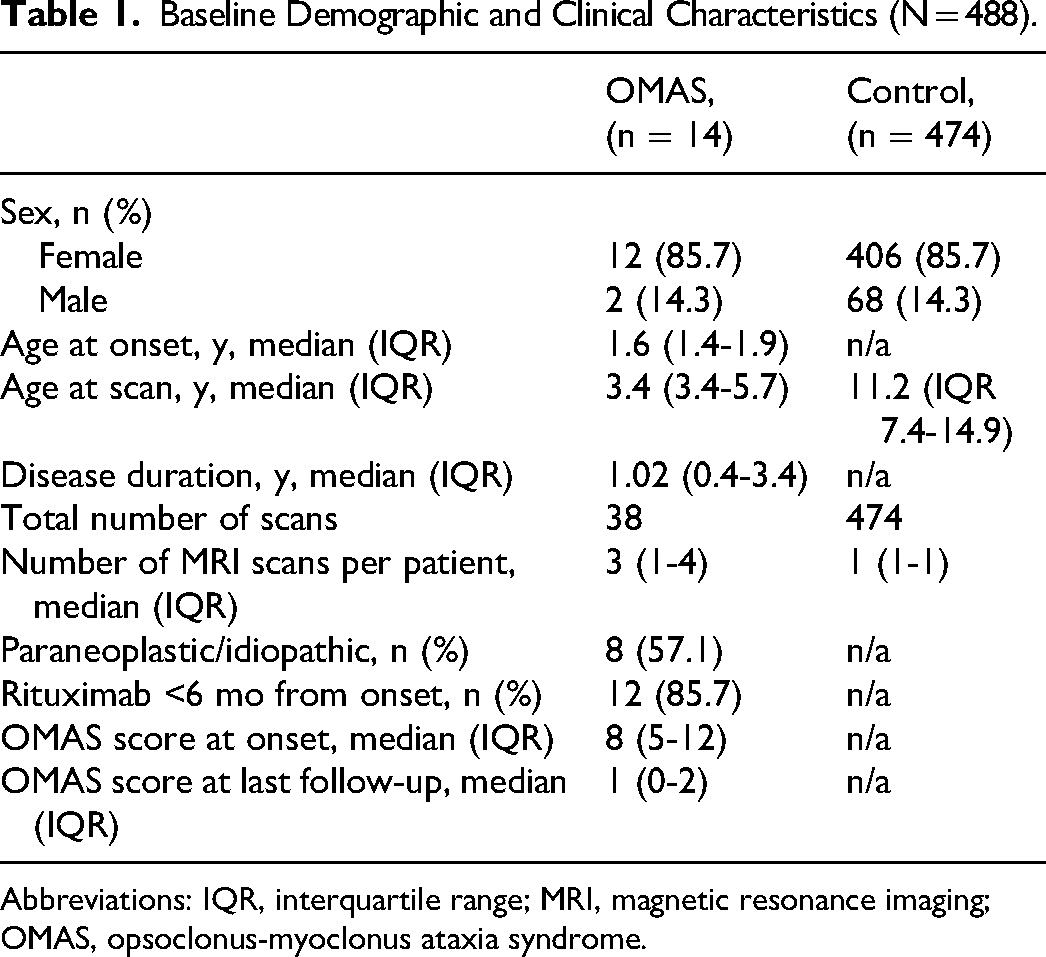
Abbreviations: IQR, interquartile range; MRI, magnetic resonance imaging; OMAS, opsoclonus-myoclonus ataxia syndrome.
Supratentorial Volumes
The curvature of the growth trajectories of both cerebral white matter and gray matter volumes differed significantly from those of the control group (cerebral white matter, P = .01; cerebral gray matter, P = .01) (Figure 1). The curvature of the gray matter trajectory in opsoclonus-myoclonus ataxia syndrome followed an expected increase until preadolescence, with a marked downturn thereafter. Visual inspection of the curves suggests a relatively greater increase in the preadolescence years compared to controls and a steeper dropoff after adolescence. Similarly, curvature of the cerebral white matter volume differed in the opsoclonus-myoclonus ataxia syndrome group compared with healthy controls (P = .01).
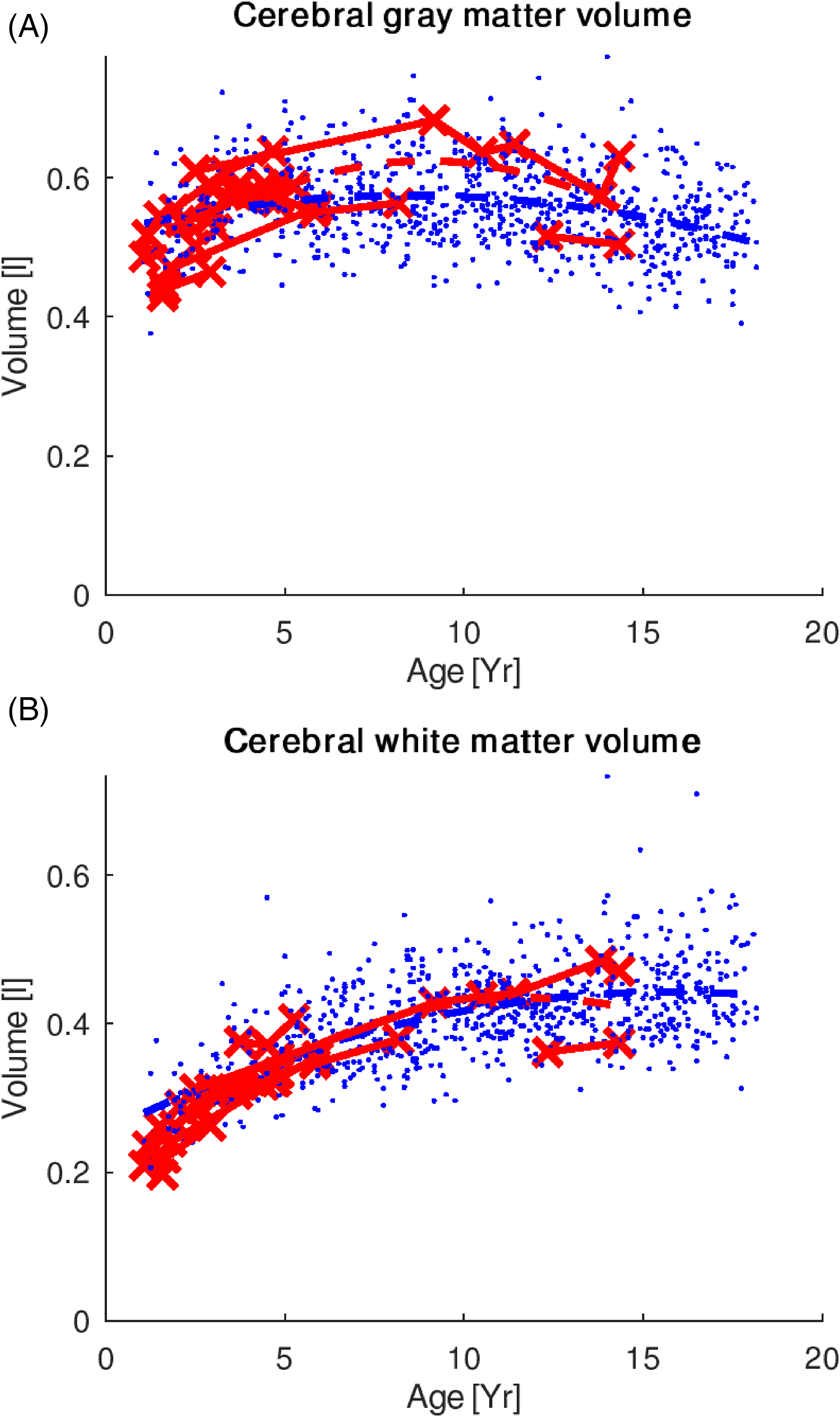
(A) Cerebral gray and (B) white matter volume trajectories in the OMAS (red) and control (blue) groups. The curvature of the growth trajectories of both cerebral white matter and gray matter volumes differed significantly from those of the control group (cerebral white matter, P = .01; cerebral gray matter, P = .01). OMAS, opsoclonus-myoclonus ataxia syndrome.
Cerebellar Volumes
The cerebellum, as the primary area in the brain that has been linked to opsoclonus-myoclonus ataxia syndrome, also showed differences in the curvature of the growth trajectory (cerebellar white matter volume, P < .001; cerebellar gray matter volume, P = .049) (Figure 2). Visual inspection suggests that divergence of the trajectories appears at about 5 years of age.
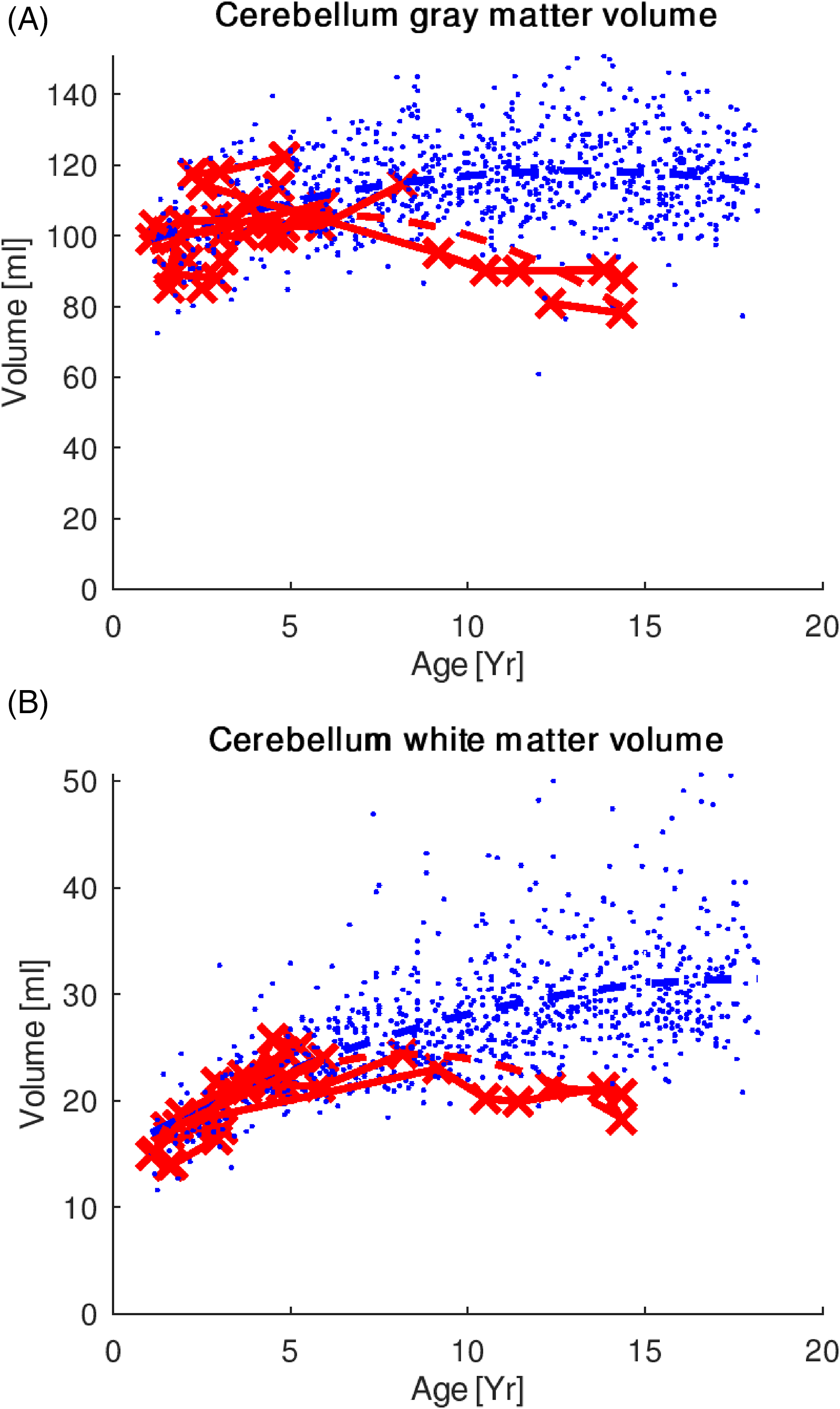
(A) Cerebellar gray and (B) white matter volume trajectories in the OMAS (red) and control (blue) groups. The cerebellum, as the primary area in the brain that has been linked to OMAS, also showed differences in the curvature of the growth trajectory (cerebellar white matter volume, P < .001; cerebellar gray matter volume, P = .049). OMAS, opsoclonus-myoclonus ataxia syndrome
Brainstem Volumes
Brainstem analysis included the whole brainstem, midbrain, superior cerebellar peduncle, pons, and medulla. Significant differences were found in the curvature of the growth trajectory in the whole brainstem of opsoclonus-myoclonus ataxia syndrome vs healthy controls (P < .01) (Figure 3).
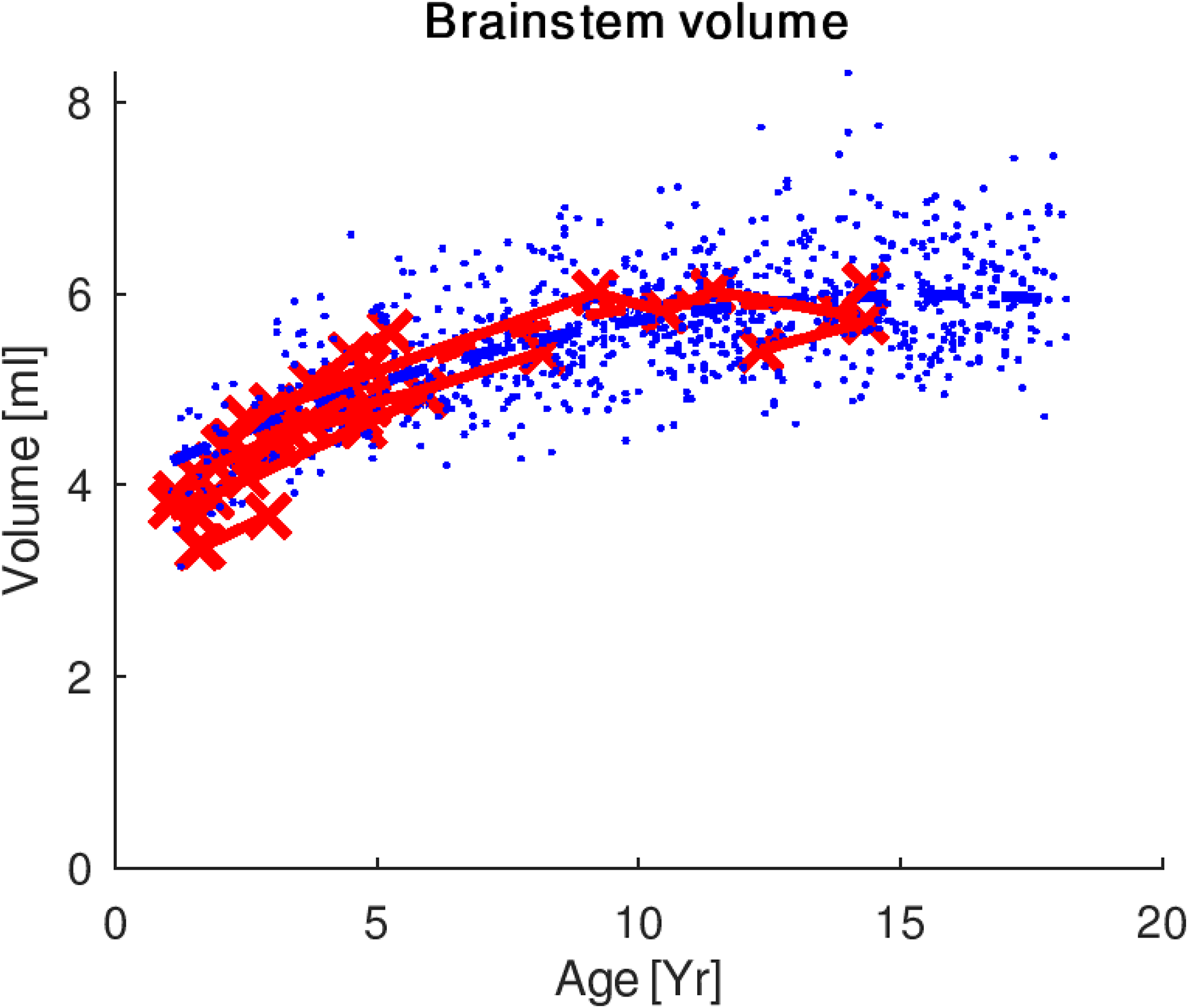
Brainstem volumes in OMAS (red) and control (blue) groups. Significant differences were found in the curvature of the growth trajectory in the whole brainstem of OMAS vs healthy controls (P < .01). OMAS, opsoclonus-myoclonus ataxia syndrome
Discussion
In this longitudinal analysis of cerebellar and extracerebellar brain volumes in children with opsoclonus-myoclonus ataxia syndrome compared with a large control cohort of children with normal clinical MRI examinations, we demonstrated differences in the growth trajectories of the brain in the cerebellar cortex. Surprisingly, we found evidence of a global process affecting all areas of the brain, including the cerebral gray matter, cerebral white matter, and brainstem, together with the cerebellar white matter. The divergence of growth trajectory curves was observed at variable times, starting as young as 5 years of age or approximately 3-3.5 years after the typical onset of opsoclonus-myoclonus ataxia syndrome.
Few studies have evaluated structural brain changes in opsoclonus-myoclonus ataxia syndrome patients during both the early and later stages of the disease. Cerebellar atrophy, particularly involving the vermis and flocculonodular lobes, has been reported in up to one-third of patients later in the disease course. 8 Furthermore, reductions in cortical thickness (motor and visual cortices) and subcortical gray matter have been documented at chronic stages of the disease.10,14 Our findings, in which the cerebellar gray matter volume in children with opsoclonus-myoclonus ataxia syndrome diverges from healthy children several years after disease onset, suggest that despite the use of aggressive therapies, a degree of injury may be sustained, which ultimately affects volume changes through time. Our data demonstrate differences between the expected inverted U–shaped trajectory of cerebellar gray matter growth in healthy children and children with opsoclonus-myoclonus ataxia syndrome, suggesting deviations in both the normal growth and pruning processes of the cerebellar gray matter. Notably, the cerebellum has been demonstrated to reach its peak volume later than the cerebrum, 15 with an age of peak volume at around 13 years in girls and 16 years in boys. Peak volume in our opsoclonus-myoclonus ataxia syndrome cohort was far earlier, at approximately 7 years of age, supporting a derangement in growth potential in this cohort.
As for cerebral cortex and white matter, peak cerebral cortex volume in healthy children occurs in females at around 8 years of age and males at around 10 years of age, with continued white matter growth through childhood. 16 In contrast to the cerebellar data, the peak volume appears similar in the cerebral gray matter of children with opsoclonus-myoclonus ataxia syndrome and healthy control children, but with a steeper growth trajectory and greater fall-off of the trajectory. The reasons for this aberration in growth pattern are unknown but suggest, again, that early injury may alter growth and pruning activity in the cortex. Importantly and of concern is the drop in the growth trajectory of the white matter that we observed: while ongoing and continued growth in the white matter is expected through childhood with no expected reduction in white matter volume in childhood, the opsoclonus-myoclonus ataxia syndrome cohort demonstrated a peak volume at around 8 years of age, with subsequent dropoff of volumes, again suggesting aberration in growth patterns with potential consequences to learning and higher-level cognitive functions. 17
Importantly, prior MRI studies have been limited by their cross-sectional nature and small patient cohorts. Our evaluation of longitudinal data compared to a large control cohort demonstrated differences in the growth trajectory in all areas of the brain. None of the previous studies demonstrated volume differences in the white matter, which we observed in our longitudinal analysis. These differences are potentially due to the small populations in cross-sectional studies and the inability of previous studies to evaluate changes in growth over time.
Our findings provide structural evidence for the global cognitive challenges reported in children with opsoclonus-myoclonus ataxia syndrome; up to 70% to 80% of patients in older series demonstrate neurologic or cognitive deficits, including motor (60%), speech (66%), learning (51%), and behavioral problems (51%). 6 Furthermore, although multiple previous studies have demonstrated symptom control with early and aggressive immunotherapies, 18 little is known about whether upfront therapy can change the long-term outcomes. Importantly, despite most children in our study receiving upfront and aggressive immunotherapy, a divergence of growth trajectories was found in these children compared with the control group. Because of the small number of children who received relatively delayed treatment, we were unable to evaluate the differences between those receiving upfront and aggressive immunotherapy and those who did not. Literature in other pediatric neuroinflammatory disorders, such as multiple sclerosis, suggests that earlier treatment with highly effective immunotherapy can change the trajectory of outcomes. 19 Whether the early use of highly effective therapies has improved the trajectory of brain growth in this population is unknown, but given the findings of this study, there is certainly more to be learned about prevention of long-term central nervous system abnormalities in opsoclonus-myoclonus ataxia syndrome. Knowledge of whether earlier therapeutic intervention results in improved outcomes has significant implications for future treatment decisions in opsoclonus-myoclonus ataxia syndrome. Further studies are required to address these issues. Furthermore, understanding the associations between specific cognitive deficits and brain volume changes is necessary to plan future targeted rehabilitation therapies for patients with opsoclonus-myoclonus ataxia syndrome.
Our study was limited by its small sample size, given the rarity of the condition. This may limit the generalizability of the results. Furthermore, our results should be evaluated with caution given the varying lengths of follow-up available and relative heterogeneity of etiologies in included opsoclonus-myoclonus ataxia syndrome patients. A small number of patients with opsoclonus-myoclonus ataxia syndrome had longer MRI observation times, potentially biasing the results. Given the small sample size, we were unable to include other regional brain volumes, which could have provided further insights into specific regional abnormalities in opsoclonus-myoclonus ataxia syndrome. Larger prospective multicenter studies are necessary to confirm and expand on our findings.
Conclusion
Our longitudinal study provides evidence for global brain involvement in the long-term follow-up of patients with opsoclonus-myoclonus ataxia syndrome, with the involvement of growth trajectories of gray and white matter in the cerebrum, cerebellum, and brainstem. Large-scale international multicenter longitudinal studies that include subsegmentation of different brain regions are warranted to confirm these results.
Footnotes
Acknowledgments
The authors wish to thank Edgar Martinez Chavez and Luke Chung for their assistance in the early stages of data analysis. We would also like to thank Tara Berenbaum for her assistance with study coordination and the patients and their families for their participation in this study.
Author Contributions
MA and EAY composed the first draft of the manuscript. EAY was responsible for study concept and design. MWW played a major role in data acquisition. GL, MA, and CY contributed to analysis and interpretation of data, and EAY, MWW, LV, and BE-W played a major role in the analysis and interpretation of data. All authors revised the article for content; EAY and CY also revised the medical writing for content. EAY was responsible for the funding of the study.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EAY has received research funding from National Multiple Sclerosis Society (NMSS), Consortium of Multiple Sclerosis Centers (CMSC), Canadian Institues of Health Research (CIHR), National Institutes of Health (NIH), Ontario Institute for Regenerative Medicine (OIRM), Stem Cell Network (SCN), The Garry Hurvitz Centre for Brain & Mental Health (GH-CBMH) Chase an Idea, SickKids Foundation, Rare Diseases Foundation, MS Scientific Foundation, McLaughlin Centre, Leong Center, and Peterson Foundation; investigator-initiated research funding from Biogen; and is on the scientific advisory board of Hoffman-LaRoche. She receives support as a member of the data safety monitoring board of a clinical trial from TG Therapeutics; speaker honoraria from Biogen, Johns Hopkins University (JHU), Saudi Epilepsy Society, New York University (NYU), MS-ATL (MS-At the Limits), Catamount Education, PRIME Education, and Canadian Neuropathology Society. The remaining authors report no disclosures relevant to the manuscript.
Ethical Approval
This study was approved by the institutional research ethics board at SickKids hospital. Informed consent was obtained for the opsoclonus-myoclonus ataxia syndrome group (SickKids REB#1000057228); however, our ethics board waived the requirement for consent from the control group because of the retrospective nature and large sample size.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Peterson Foundation (grant number n/a).
