Abstract
This study investigates structural alterations of the corpus callosum in children diagnosed with infantile hydrocephalus. We aim to assess both macrostructural (volume) and microstructural (diffusion tensor imaging metrics) facets of the corpus callosum, providing insights into the nature and extent of alterations associated with this condition. Eighteen patients with infantile hydrocephalus (mean age = 9 years) and 18 age- and sex-matched typically developing healthy children participated in the study. Structural magnetic resonance imaging and diffusion tensor imaging were used to assess corpus callosum volume and microstructure, respectively. Our findings reveal significant alterations in corpus callosum volume, particularly in the posterior area, as well as distinct microstructural disparities, notably pronounced in these same segments. These results highlight the intricate interplay between macrostructural and microstructural aspects in understanding the impact of infantile hydrocephalus. Examining these structural alterations provides an understanding into the mechanisms underlying the effects of infantile hydrocephalus on corpus callosum integrity, given its pivotal role in interhemispheric communication. This knowledge offers a more nuanced perspective on neurologic disorders and underscores the significance of investigating the corpus callosum's health in such contexts.
Keywords
Infantile hydrocephalus is characterized by an atypical accumulation of cerebrospinal fluid in the brain, leading to progressive enlargement of the ventricular system and necessitating surgical intervention within the first 2 years of life. 1 One treatment for this condition is the surgical placement of a ventriculoperitoneal shunt system that redirects excess fluid to the peritoneum, effectively alleviating pressure on the brain. 2 This intervention has shown significant efficacy in reducing ventricular size and ameliorating associated symptoms. 3 Early hydrocephalus (with an onset before the age of 2), comes with a particular shape of ventricles, which is not seen in patients with a later onset, where the posterior horn is particularly affected and enlarged.4,5 Therefore, the posterior cortex, encompassing regions such as the parietal and occipital lobes, bears the brunt of damage.6,7 The direction of ventricular dilatation subjects the posterior cortex to sustained compression, inflammation, cortical thinning, and increased cerebrospinal fluid volumes.7,8 Research in both human and animal models indicate a correlation between hydrocephalus and corpus callosum thinning, primarily attributed to lateral ventricular dilatation exerting upward compression on periventricular and subcortical white matter.9–14 The corpus callosum, a principal interhemispheric commissure, connects neocortical areas and represents the largest white matter fiber bundle in the human brain. 15 Its role is to provide interhemispheric communication of motor, sensory, and higher-order problem-solving information. 16 Damages to the corpus callosum, including dorsal grooving from impingement on the falx cerebri, and myelin degradation, correlate with motor and cognitive deficits.17–20
The corpus callosum comprises 5 subdivisions, each with distinct functional connections. The posterior (splenium) part of the corpus callosum connects visual areas in the occipital lobe, whereas the midposterior (isthmus) part connects fibers of motor, somatosensory, and primary auditory areas.21–24 As the central (body) fibers traverse the cerebral cortex, they give rise to the corona radiata and contribute to the formation of various prominent white matter pathways. 25 The midanterior (genu) part of the corpus callosum connects the medial and lateral surfaces of the frontal lobes, 26 whereas the anterior portion (rostrum) serves as a vital bridge linking the orbital surfaces of the frontal lobes. 27
Understanding the intricacies of corpus callosum microstructure and volume is crucial for comprehending neurodevelopmental and pathologic processes. Microstructure, composed of intricate white matter fibers, holds essential information regarding the structural integrity and connectivity within this neural bridge. 28 Alterations in microstructural properties can provide insights into the underlying changes, shedding light on the adaptability and resilience of the corpus callosum in response to various conditions.29–33 Similarly, changes in volume offer a macroscopic view of the structural alterations.34–37 One study demonstrated a relationship between brain morphometry and functioning in children treated for hydrocephalus. Results revealed reductions in the size of the corpus callosum in the shunted hydrocephalus group of children aged 6-13 years and a relationship between the corpus callosum and deficits in nonverbal cognitive skills and motor abilities. 38 A study has yet to be reconciled in an infantile hydrocephalus population that explores the relationship between microstructural and macrostructural changes in the corpus callosum.
Diffusion tensor imaging is used to map and characterize the 3-dimensional diffusion of water as a function of spatial location.39,40 Diffusion anisotropy is mainly caused by cellular membranes in the brain's white matter, with some contribution from myelination and the packing of axons.28,41 Fractional anisotropy defines the degree to which water diffusion is constrained in a specific direction. 42 Fractional anisotropy is sensitive to the alignment of white matter fibers and their structural integrity with the degree of myelination. 43 Fractional anisotropy values range from 0 to 1, where 0 represents complete isotropy meaning diffusion occurs equally in all directions, and 1 represents maximum anisotropy meaning diffusion occurs in a single direction. 40 A study observed a correlation between the fractional anisotropy of the total corpus callosum and visuomotor interhemispheric processing speed, highlighting the functional implications of fractional anisotropy in neural processing. 44 Higher fractional anisotropy values are associated with more coherent and well-structured fiber bundles, whereas lower fractional anisotropy values could suggest possible damage or reduced fiber integrity.28,45,46 Mean diffusivity relates to the average diffusion of water molecules within a region of interest and can provide insight into tissue characteristics such as integrity and microstructural changes. 47
When the ventricles become enlarged in the hydrocephalus, inducing pressure on the surrounding brain tissue, this may lead to changes to the white matter integrity and diffusion properties.48–50 Research suggests that patients with hydrocephalus reveal reduced fractional anisotropy values, especially in the corpus callosum48,51–53 and splenium region 54 suggesting disrupted white matter integrity or axonal structure. Elevated mean diffusivity values have also been found in the corpus callosum of hydrocephalus patients,51,53 indicating less restricted movement of water molecules perpendicular to the axial direction, effectively reducing the integrity of the tissue structure and organization. To be sure, more research is needed to contribute to these findings, especially concerning a developing child population, as only 1 study considered a few children with hydrocephalus. 48 Fractional anisotropy and mean diffusivity are measures that help us understand brain and cognitive development, 55 while also considering how this may be impacted in neurodevelopmental disorders allowing for early identification of abnormal growth of white matter integrity.
In addition to fractional anisotropy and mean diffusivity measures, analyzing radial diffusivity and axial diffusivity allows further characterization of various white matter pathologies. Radial diffusivity indicates the diffusion of water molecules perpendicular to the axonal fibers within white matter, reflecting diffusion across fiber bundles.56,57 Radial diffusivity values are often characterized by demyelination, myelin sheath damage, or axonal integrity loss.56,58 Axial diffusivity is involved in the diffusion of water molecules parallel to axonal fibers within white matter relating to the diffusion along the longitudinal direction of fiber bundles. 56 White matter damage influences axial diffusivity in that alterations include changes in axonal density or changes related to axonal loss.59,60
This study aims to characterize the extent of alterations to the corpus callosum in children who were affected by infantile hydrocephalus. We hypothesize that in children with infantile hydrocephalus, there will be discernible alterations in both volume and corpus callosum microstructure compared with typically developing children especially in its posterior portion. By establishing an understanding of the corpus callosum's health, we aim to provide a foundation for researchers to assess not only the individual subdivisions impacted by conditions such as infantile hydrocephalus but also the intricate network of connections facilitated by the corpus callosum, thus offering a more holistic perspective on neurologic disorders.
Methods
Participants
Eighteen infantile hydrocephalus patients were recruited from a pediatric neurosurgery clinic during follow-up (12 males, 6 females; mean age = 9 years and 4 months). They were matched by sex and age to 18 typically developing controls recruited from OurBrainsCAN Cognitive Neuroscience Research Registry at Western University. Inclusion criteria encompassed a diagnosis of hydrocephalus, treatment with a ventriculoperitoneal shunt within the first 2 years of life, asymptomatic status, and clinical stability. Exclusion criteria entailed neuropsychiatric diagnoses and major perinatal infections. None of the patients included in the study had colpocephaly, and no patient presented with fetal dilatation confirmed by an ultrasonography in utero. The control group consisted of healthy children devoid of any medical, neurologic, or developmental conditions.
The etiology of hydrocephalus varied among the patients, with cases stemming from intraventricular hemorrhage, Dandy-Walker malformation, and myelomeningocele. Six prematurely born infants with intraventricular hemorrhage–related hydrocephalus underwent shunt insertion between the ages of 1 and 8 months. Eleven full-term infants without intraventricular hemorrhage received a shunt insertion within a postnatal time frame of 2 days to 8 months. One full-term infant, diagnosed with intraventricular hemorrhage–related hydrocephalus, had a shunt inserted at 4 months of age. Shunt revisions were conducted in 8 instances.
Neuroimaging Acquisition
Images were acquired at the Robarts Research Institute's Centre for Functional and Metabolic Mapping, with a Siemens MAGNETOM Prisma whole-body 3-tesla magnetic resonance imaging (MRI) scanner (32-channel head coil). High-resolution anatomical brain images (sagittal interleaved slices with a voxel thickness of 2 mm × 2 mm × 2 mm, flip angle of 9°, repetition time of 2300.0 ms, echo time of 2.93 ms, and field of view of 256 mm) as well as 2 consecutive series of diffusion-weighted echo-planar images in opposing phase-encoding directions along the anterior-posterior axis were acquired. Diffusion volumes were acquired using the following parameters: gradient directions = 30; b-value = 1000s/mm2; isometric voxel size = 2 × 2 × 2 mm3; and matrix size = 192 × 192 mm.
Diffusion Tensor Imaging Preprocessing
The first preprocessing step taken for the diffusion-weighted imaging data included a correction distortion using the top-up tool from FSL (FMRIB Software Library). The data in the data set were collected with reverse phase-encode blips, resulting in pairs of images with distortions occurring in opposite directions (anterior to posterior and posterior to anterior). The susceptibility-induced off-resonance field was estimated using a method implemented in FSL,61,62 and the 2 images were combined into a single corrected one. After running FSL-Brain Extraction Tool on the data acquired from the previous step, we ran the eddy tool in FSL to perform several important tasks. Eddy corrects for susceptibility-induced distortions and compensates for motion artifacts that can occur because of participant movement during the scan. Such correction helps to align and correct the diffusion-weighted imaging data, ensuring more reliable measurements of diffusion properties. Next, DTIFIT in FSL was used to estimate and reconstruct diffusion tensors and used the input from the preprocessed data (eg, diffusion-weighted data, BET binary mask, gradient directions, and b-values). DTIFIT then applies mathematical algorithms to estimate the diffusion tensor at each voxel describing the magnitude measured by eigenvalues and direction of diffusion known as the principal eigenvector within that voxel. 63 Reconstructing diffusion tensors using DTIFIT allows for the calculations of fractional anisotropy, mean diffusivity, and tensor-derived metrics enabling us to assess tissue microstructure and white matter integrity of the corpus callosum. Track-based spatial statistics from FSL was used to facilitate further erosion of the mask to remove any artifacts in the subjects’ fractional anisotropy images. The diffusion tensor imaging data was then registered to the subject’s own anatomical brain-extracted T1 image.
Segmentations of the corpus callosum were executed manually through FreeSurfer v7. 4.0 on each diffusion-weighted imaging scan in native image space for both groups (each was reviewed by 2 authors). Segmenting the diffusion images in native space is beneficial for capturing information about the local tissue structure. In turn, this is important for accurate segmentation and preserving the microstructural information minimizing any errors or distortions especially relevant for a patient population with brain abnormalities. Quality assurance procedures were implemented during the segmentation process of the corpus callosum for microstructural diffusion data extraction. In addition to extracting individual subdivisions and their respective diffusion metrics, a corresponding standard deviation value was obtained for each subdivision. Notably, any deviation exceeding 1 standard deviation for a given metric prompted a resegmentation process. This stringent quality control measure ensured that the extracted data met accuracy and reliability standards, ultimately enhancing the robustness of our subsequent analyses. Volumes from specific corpus callosum subdivisions were obtained using fslmaths. For anatomical specifications, please refer to Figure 1.
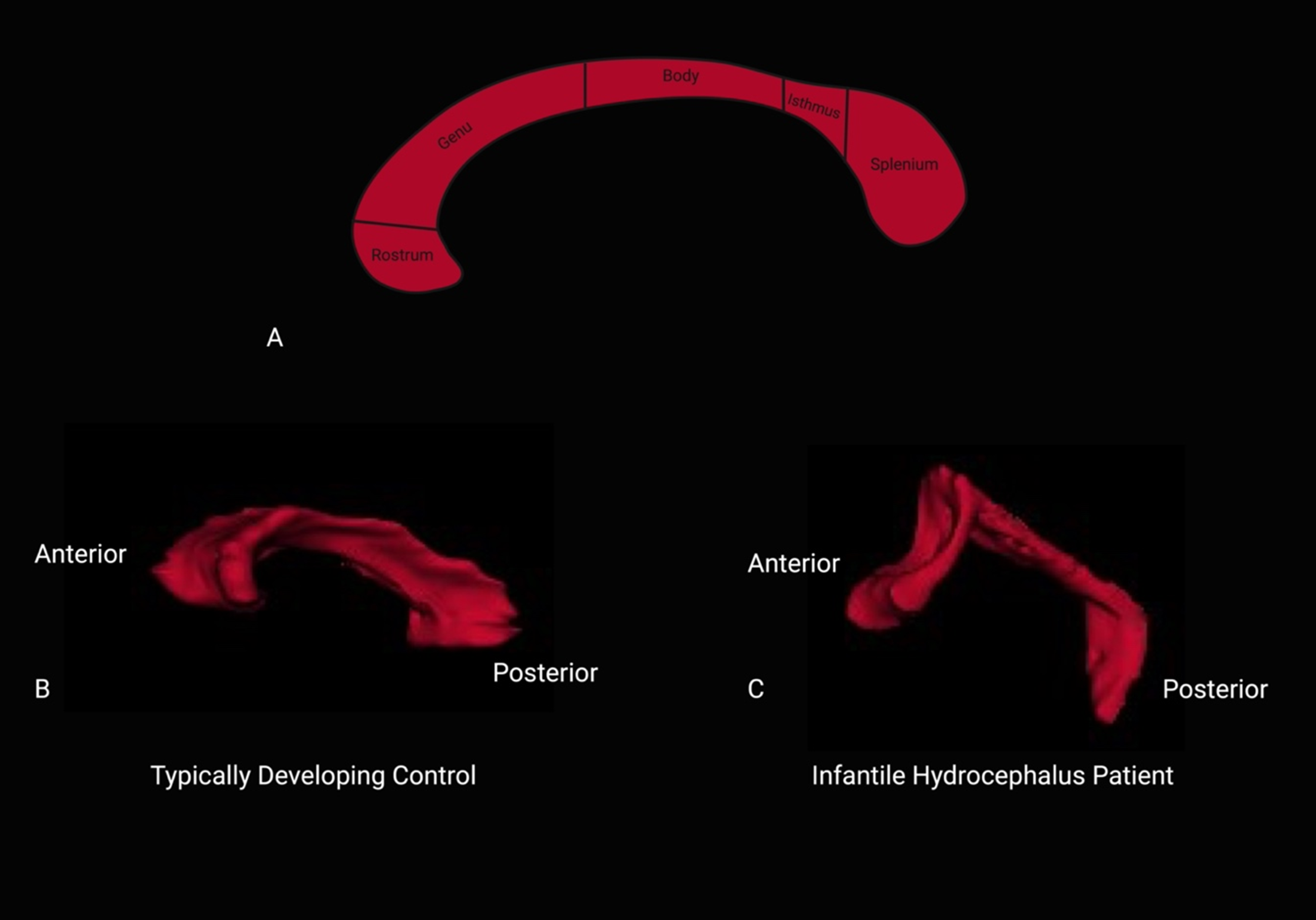
(A) Anatomical subdivisions of the corpus callosum, corresponding to the rostrum, genu, body, isthmus, and splenium. (B) Segmented corpus callosum of a typically developing male control subject aged 8 years 10 months and (C) a segmented corpus callosum of a male infantile hydrocephalus patient, aged 8 years 11 months.
Statistical Analysis
All statistical analyses were completed using Statistical Package for the Social Sciences (version 29). A repeated measures analysis of variance was conducted to assess whether diffusion tensor imaging microstructural metrics and volumes differed by corpus callosum subdivision, age, or group. This analysis was performed using a multivariate approach, evaluating the dependent variables of volume, fractional anisotropy, mean diffusivity, axial diffusivity, and radial diffusivity, with a predefined alpha level of 0.05 for determining statistical significance. The 5 corpus callosum areas served as the within-subjects factor, whereas age (used as a covariate) and group (comprising infantile hydrocephalus and a typically developing control group) were the between-subjects factors. Subsequent comparisons were conducted to assess discrepancies in diffusion tensor imaging metrics across contiguous corpus callosum subdivisions. Bonferroni corrections (P < .01) were applied to maintain control over reported univariate tests.
To examine specific comparisons between corpus callosum subdivisions and groups—a part of post hoc testing—paired and unpaired t tests were employed. These 2-tailed t tests were used to analyze differences in both volume and microstructural integrity, offering an examination of the nuanced alterations within the corpus callosum across different subdivisions and participant groups.
Results
Segmentation of Ventricular and Corpus Callosum Volumes
The results of the segmentation for lateral ventricles in both groups revealed differences in the infantile hydrocephalus group compared with controls (Table 1). Patients treated lateral ventricles were larger as indicated by the total volume (mean = 37.12 cm3) compared to controls (mean = 10.7 cm3).
Treated Ventricular Volumes in Infantile Hydrocephalus Patients and Typically Developing Lateral Ventricular Volume Presented for Controls, Along With Corresponding Mean and SD.a

Abbreviation: SD, standard deviation.
Volumes are measured in cubic centimeters.
Group comparisons
Significant between-subject effects for group (patients vs controls) were evident across all diffusion tensor imaging metrics and volumes (Table 2). Specifically, group differences were evident in terms of volumes, F(1, 34) = 69.480, P < .001; fractional anisotropy, F(1, 34) = 279.578, P < .001; mean diffusivity, F(1, 34) = 19.622, P < .001; and radial diffusivity, F(1, 34) = 5.468, P = .026, values. However, axial diffusivity did not differ (P = .440) across different subdivisions when considering our entire pediatric population. No significant interactions between corpus callosum subdivision and age were evident, F(1, 34) = .610, P = .440. Age effects were not evident in the analyses. These findings collectively indicate that there are notable differences In subdivision volume and microstructure between hydrocephalus patients and typically developing controls, with the group distinction being a particularly influential factor in this analysis.
Repeated Measures Analysis of Variance Comparing Corpus Callosum Subdivision, Age, and Group for DTI Macrostructural and Microstructural Metrics.
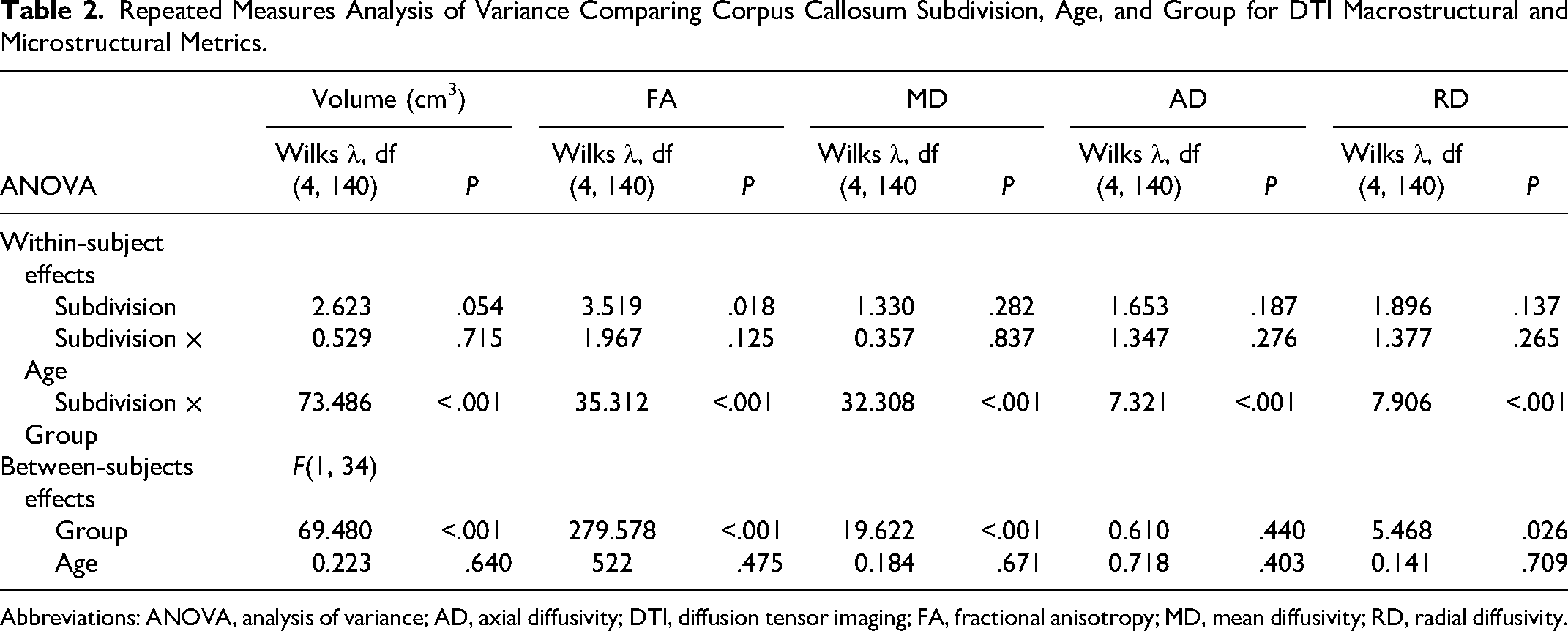
Abbreviations: ANOVA, analysis of variance; AD, axial diffusivity; DTI, diffusion tensor imaging; FA, fractional anisotropy; MD, mean diffusivity; RD, radial diffusivity.
Corpus callosum subdivision volumes
Patients had lower mean volumes across all 5 subdivisions especially in the posterior portions compared with healthy participants (Figure 2, Table 3). However, post hoc testing of the subdivisions of the corpus callosum indicated that only the splenium, isthmus, and rostrum were all significantly different between groups. Figure 1 provides a visual representation of the differences in the corpus callosum based on an anatomical scan for a single-participant comparison.
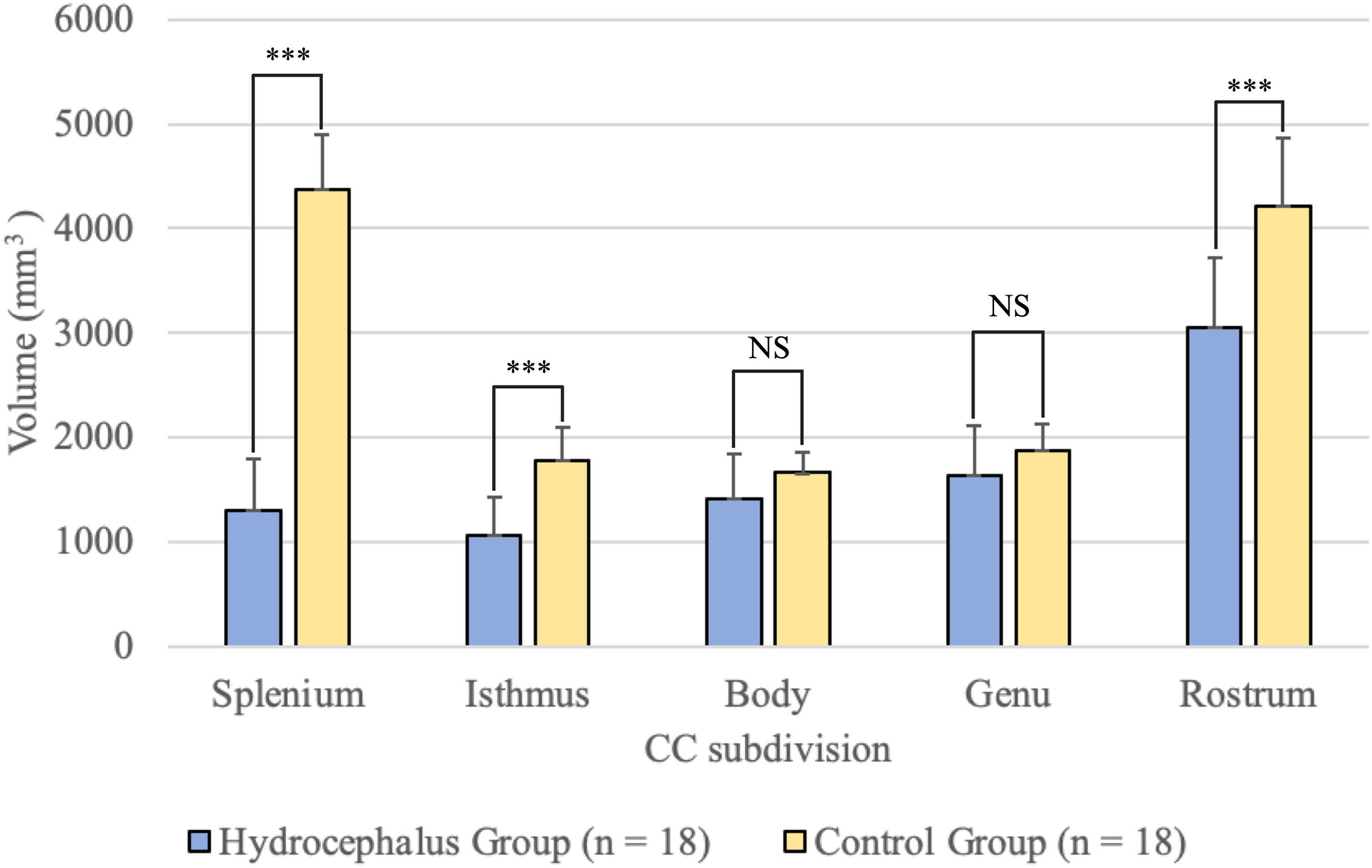
Bar graphs representing mean corpus callosum subdivisions compared by group, with standard deviation reported as error bars.
Corpus Callosum Subdivision Volumes Reported for Both Groups Along With Corresponding Mean and SD. a
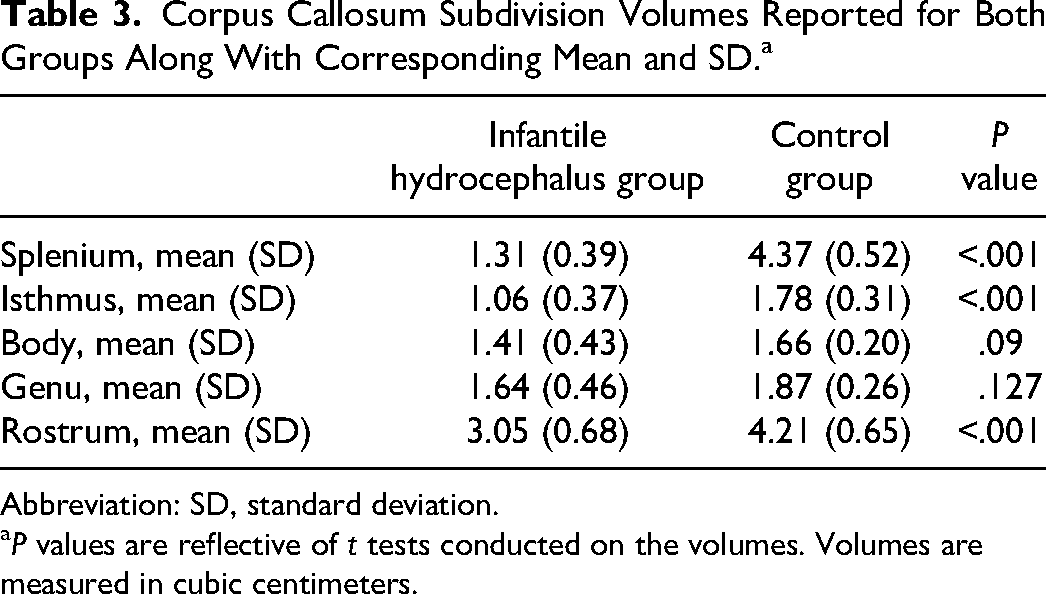
Abbreviation: SD, standard deviation.
P values are reflective of t tests conducted on the volumes. Volumes are measured in cubic centimeters.
White matter integrity of corpus callosum subdivisions
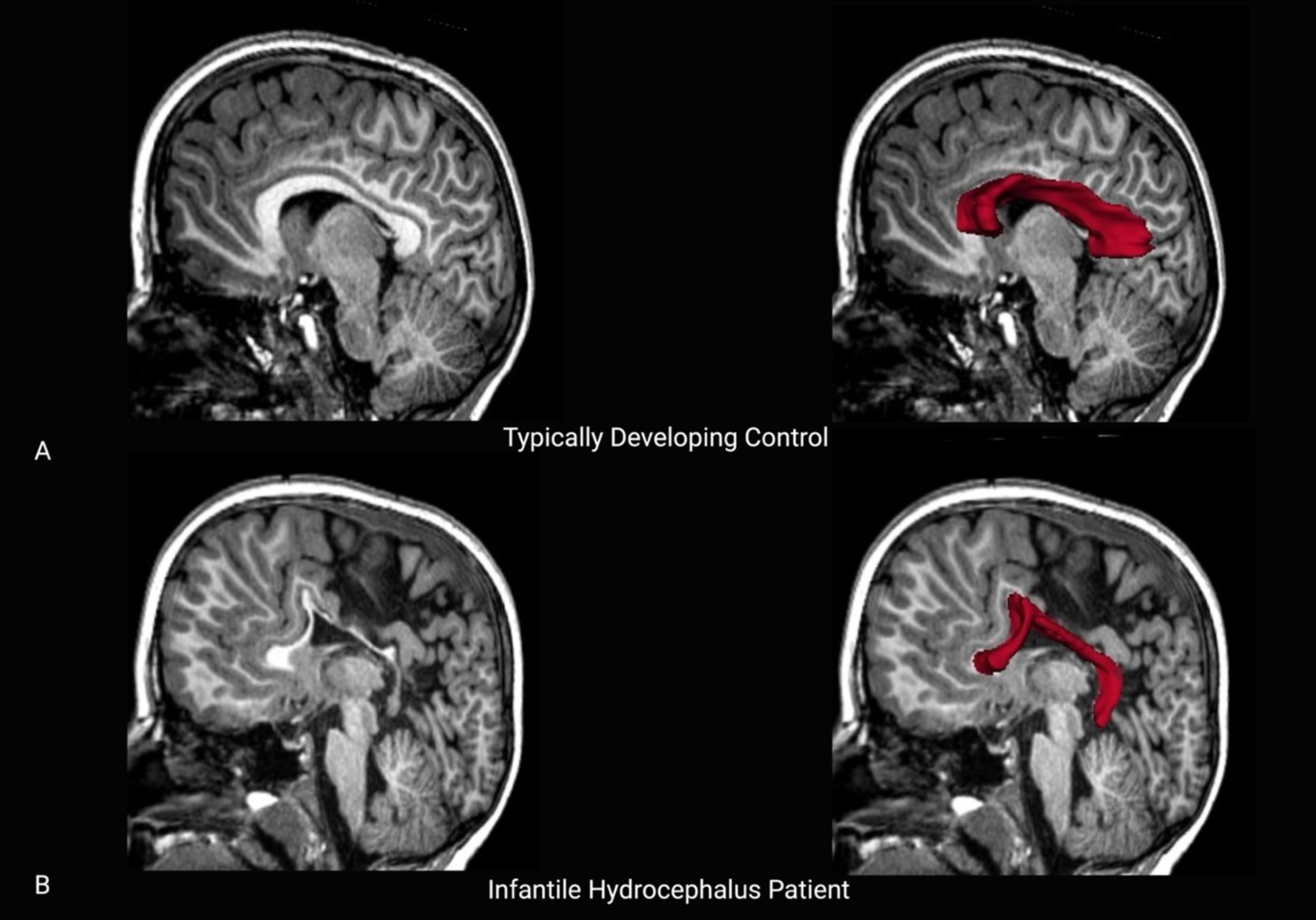
Healthy participants had significantly higher fractional anisotropy values in the subdivisions of the corpus callosum based on post hoc testing, suggesting increased white matter microstructural integrity (Table 4). Mean diffusivity was significantly lower in healthy participants relative to patients in the splenium and isthmus. Similar findings were evident for axial diffusivity and radial diffusivity values, whereby these values were lower in the splenium and isthmus but also the body of the corpus callosum in healthy participants compared with patients. Refer to Figure 3 for a midsagittal scan featuring a typically developing control and an infantile hydrocephalus patient.
Comparison of Diffusion Metrics: Fractional Anisotropy (FA), Mean Diffusivity (MD), Axial Diffusivity (AD), and Radial Diffusivity (RD), in Infantile Hydrocephalus Patients and Controls Within Corpus Callosum Subdivisions. a
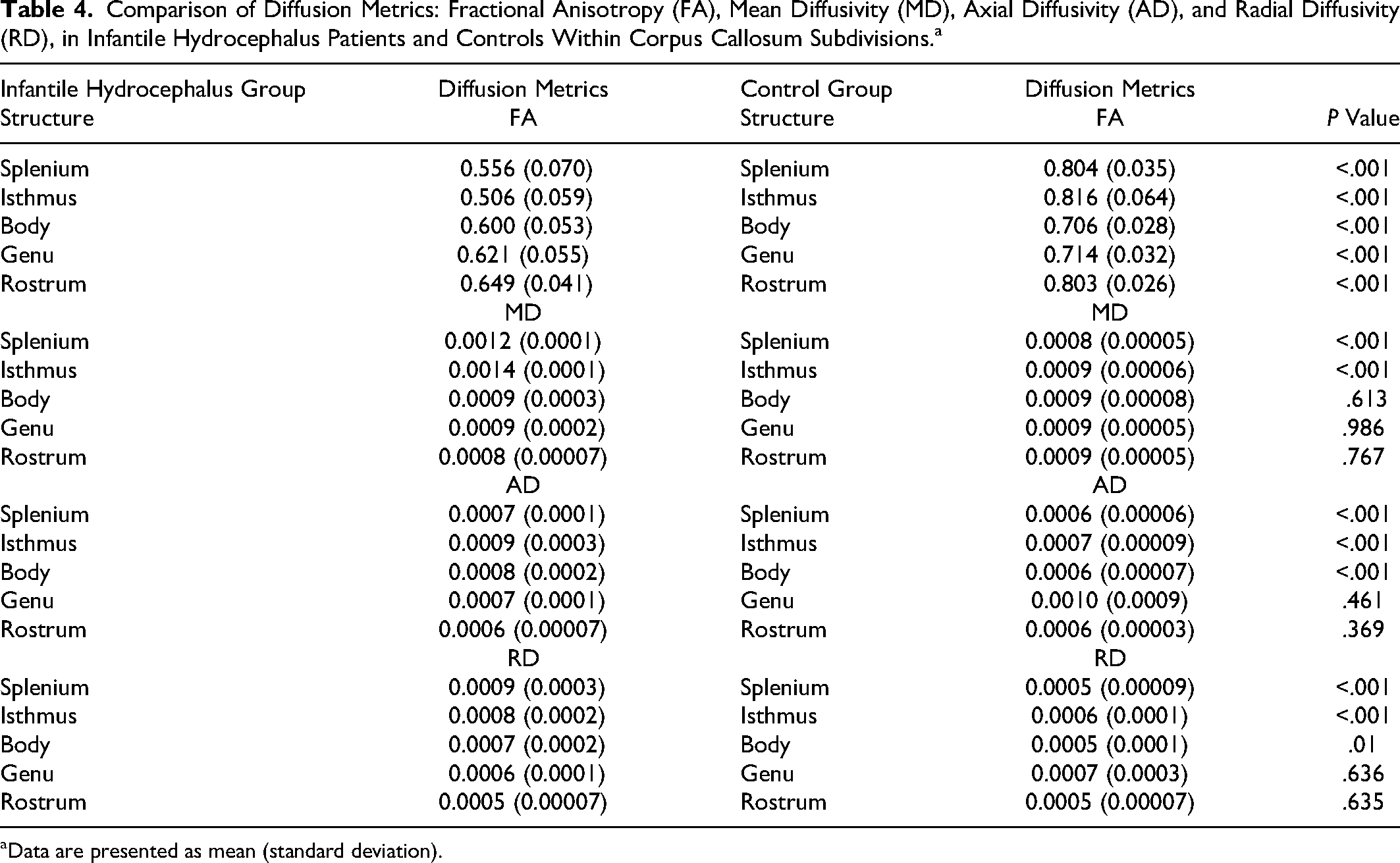
Data are presented as mean (standard deviation).
Discussion
The primary objective of this study was to investigate the long-term impact of infantile hydrocephalus on the corpus callosum at school age by examining both its macrostructure, represented by volume measurements, and microstructural components assessed through diffusion tensor imaging metrics. When examining the corpus callosum's macrostructure, represented by volume measurements, our results indicated a trend toward significance across different subdivisions. Although this P value falls slightly above our selected threshold of 0.05, it draws attention. This suggests subtle variations in corpus callosum volume within specific subdivisions, although these differences may not be as pronounced as in other subdivisions as will be discussed in this section. In terms of microstructural components, our analysis of fractional anisotropy demonstrated statistically significant differences across subdivisions. This finding underscores the importance of considering microstructural intricacies, as fractional anisotropy values exhibited variations depending on the specific subdivision being assessed. Extending our analysis to the interaction of subdivision and age, we found no significant effects, implying that age did not significantly contribute to the observed macrostructural and microstructural variations within the corpus callosum. Notably, the interaction of subdivision and group yielded significant findings. This suggests that the impact of subdivision on corpus callosum integrity differs significantly between the infantile hydrocephalus group and the typically developing control group. Specifically, the posterior segments of the corpus callosum, including the splenium and isthmus, and the rostrum exhibited distinct sensitivity to hydrocephalus-induced alterations in macrostructural integrity. Furthermore, examining the between-subjects effects, we observed significant main effects for group macrostructural measures, fractional anisotropy, mean diffusivity, and radial diffusivity.
In our study, we found differences in lateral ventricular volumes between infantile hydrocephalus patients and typically developing control subjects. Specifically, patients who had undergone surgical treatment for infantile hydrocephalus still had larger posttreatment lateral ventricular volumes compared with controls. Previous research has primarily focused on patients with moderate to severe ventricular dilatation, attributing changes in fractional anisotropy to white matter tract compression or stretching.65–67 Our findings, however, demonstrate a decrease in fractional anisotropy across all subdivisions of the corpus callosum, regardless of ventricle size and postshunt surgery, aligning with previous research. 68 In particular, we found significant differences in fractional anisotropy values among all subdivisions of the corpus callosum wherein patients with infantile hydrocephalus exhibited lower values compared with the control group. This finding is in line with previous studies that have reported alterations in corpus callosum white matter integrity in individuals with hydrocephalus.48,51–54,65 In the context of hydrocephalus, this widespread reduction in fractional anisotropy may be attributed to several underlying factors. Elevated intracranial pressure, a hallmark of hydrocephalus, can lead to mechanical stress on the white matter tracts, potentially resulting in altered axonal orientation or myelin breakdown.11,69 Additionally, altered cerebrospinal fluid dynamics and associated hydrodynamic forces may contribute to disruptions in white matter microstructure. 70 Furthermore, it is important to consider the developmental implications of hydrocephalus, particularly in pediatric populations as findings from cross-sectional data suggest that interhemispheric transfer of motor information mediated by the callosum increases from ages 4 to 11 years. 71 The growing brain is particularly susceptible to structural alterations, and hydrocephalus-related changes may have a profound impact on ongoing neurodevelopmental processes.9,11 It is also important to note that although the anterior commissure is not the primary focus of our study, we note its intact appearance in the MRI scans of these patients. As it relates to development, the anterior commissure forms 3 weeks earlier than the corpus callosum and tends to be less vulnerable to the effects of hydrocephalus, given its reduced exposure to ventriculomegaly. In cases of callosal agenesis, the anterior commissure may undergo enlargement, serving as an alternative site for midline crossing of aberrant callosal axons. 72
The smaller corpus callosum posterior volume in infantile hydrocephalus patients was not just a reflection of structural changes but we also found alterations in its microstructure, as indicated by fractional anisotropy measures and significantly elevated mean diffusivity, radial diffusivity, and axial diffusivity. The splenium, located at the posterior end of the corpus callosum, plays a crucial role in interhemispheric communication, particularly in the transmission of information related to visual perception.21–24 Visual information processing depends on the efficient transfer of data between the 2 brain hemispheres. Damage or absence of the posterior portion of the corpus callosum can severely impair the transfer of information related to the size, color, and shape of visual objects,73,74 as well as reading and interpreting digits. 75 Also, studies have documented correlations between motor abilities (eg, bimanual coordination) with the body fractional anisotropy 76 and visually guided motor function performance correlated with fractional anisotropy from the posterior body and isthmus in healthy persons. 77 The isthmus facilitates the transmission of neural projections from the primary motor cortex to the contralateral side of the brain, enabling the integration and coordination of motor commands between the 2 hemispheres.
These microstructural changes are indicative of disrupted white matter integrity, reduced neural connectivity, and potential alterations in axonal density and myelination within the splenium and isthmus. Distinguished by its dense axonal tracts and robust myelination, this subdivision is exquisitely sensitive to perturbations in white matter integrity. 78 Notably, the majority of callosal axons are established during prenatal development, with postnatal growth chiefly driven by myelination processes. 79 This developmental trajectory is marked through amplified area measurements in the isthmus and splenium during childhood and adolescence, indicative of heightened axonal diameter and augmented myelin deposition.80,81 It is noteworthy that oligodendrocytes, the precursors of myelin, may exhibit heightened vulnerability to insults during the rapid myelination phases of childhood. 82 Consequently, any disruptions in these myelination mechanisms bear significant implications for the microstructural architecture of the posterior corpus callosum. This observation suggests a dynamic interplay between hydrocephalic processes and the dynamics of myelination during the critical stages of development. These findings not only shed light on the nuanced vulnerability of the posterior corpus callosum but also offer insights into the underlying mechanisms steering white matter alterations in hydrocephalus. However, we did not observe significant differences in the genu and rostrum subdivisions microstructurally. This absence of significant findings in these possibly points to a unique resilience or uniformity in microstructural metrics in these particular segments.
Considering the health and integrity of the corpus callosum in research on infantile hydrocephalus is imperative because of its pivotal role in interhemispheric communication. The integrity and size of the corpus callosum are associated with the strength and efficiency of functional connectivity. 83 The corpus callosum serves as a critical conduit for transmitting information between the brain's hemispheres, facilitating various cognitive and sensory functions. Given its significance, any alterations or disruptions in the corpus callosum's microstructural integrity, as observed in our study, can have profound implications for overall brain function. In the context of hydrocephalus, where increased intracranial pressure and altered cerebrospinal fluid dynamics are prevalent, the corpus callosum is particularly vulnerable to structural changes. These changes can lead to altered axonal orientation, myelin breakdown, and potential disruptions in white matter microstructure. As our findings indicate, these alterations are not limited to specific regions but manifest across the entirety of the corpus callosum, underscoring the need to prioritize the assessment of corpus callosum health in hydrocephalic populations.
Future research should explore the longitudinal effects of infantile hydrocephalus and the potential for recovery or adaptation of white matter integrity. Further research and advanced imaging techniques may provide deeper insights into the specific mechanisms underlying these observed alterations. Despite the limitations in the current neuroimaging resolution, including investigations of the fetal brain reveal that infarcts may damage radial glial fibers that guide migratory neuroblasts and glioblasts. 84 In the context of hydrocephalus, where disruptions in the cerebral fluid dynamics play an important role, more research is needed to establish a correlation between the thickness of the corpus callosum and the degree of compression of the gray matter cortex. Although a correlation between the thickness of the corpus callosum and the extent of compression in the gray matter cortex in cases of fetal hydrocephalus may vary depending on the timing and severity of hydrocephalus along with other underlying conditions, more research is needed to explore this relationship. It is noteworthy that in cases of hydrocephalus, the stretching and elongation of radial glial fibers may further contribute to complexities in cellular migration, particularly affecting the cortical plate and layer 3, housing the small pyramidal cells pivotal in the origin of commissural axons.
Additionally, investigating the relationship between corpus callosum alterations and specific clinical outcomes, such as cognitive and motor impairments, can provide a more comprehensive understanding of the functional consequences of infantile hydrocephalus. However, this study has its limitations. The sample size was relatively small, and future research with larger samples and longitudinal designs is needed to further elucidate the impact of infantile hydrocephalus on the corpus callosum and its functional consequences. Additionally, although we focused on the corpus callosum, other brain structures and neural networks may also be affected in infantile hydrocephalus, warranting further investigation.
Footnotes
Acknowledgments
We extend our appreciation to the families who participated in this research, as their active involvement played a crucial role in the completion of this study. We would also like to thank the MRI technologists at the Centre for Functional and Metabolic Mapping for their support and assistance. Funding for the study was provided by the Academic Medical Organization of Southwestern Ontario and the Canada First Research Excellence Fund (CFREF) through OurBrainsCAN.
Author Contributions
All authors reviewed and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Health Sciences Research Ethics Board at Western University. Written informed consent was obtained from all participants prior to participation in the magnetic resonance imaging. The research was conducted according to the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Academic Medical Organization of Southwestern Ontario, Canada First Research Excellence Fund,
