Abstract
Chronic migraines substantially burden patients, affecting their quality of life and daily functioning. The International Classification of Headache Disorders defines chronic migraines as headaches occurring more than 15 days per month for over 3 months, with at least 8 days featuring migraine-like symptoms. 1 Migraines in children can have significant consequences, including decreased school performance, limited social interactions, and reduced self-esteem. 2 The prevalence of chronic migraines in children is estimated to be 0.8% in those aged 12-17 years, with rates as high as 1.8% when chronic migraines are associated with medication overuse headaches. 3 The annual cost of migraines in the United States is estimated to be around $36 billion, considering the impact on health care costs and productivity. 4
Although several medication options exist for acute and preventive management of chronic migraines in adults, the pediatric population faces limitations and a scarcity of available treatments. Currently, only 1 preventive medication, Topamax, is approved by the Food and Drug Administration (FDA) for use in children. 5 Additionally, eNeura's (eNeura Inc, Baltimore, MD) Single-Pulse Transmagnetic Stimulator (sTMS) has received FDA approval for children 12 years and older. 6 The current standard of care for pediatric migraines involves nonsteroidal anti-inflammatory drugs, acetaminophen, antiemetics, anticonvulsants, and triptans. However, the efficacy of these medications has been inconsistent, and some have demonstrated significant side effects. 7 Beta-blockers, commonly used in adults, have shown mixed results in the pediatric population. 7 Tricyclic antidepressants, particularly amitriptyline, have proven to be the most efficacious among this class of drugs in children. 8 Among triptans, rizatriptan has shown statistically significant pain relief along with a reduction in associated symptoms such as nausea and vomiting. However, there are significant potential side effects, such as rebound migraines and malignant hypertension. 9 In the search for effective preventive medications for pediatric chronic migraines, the CHAMP trial compared topiramate, amitriptyline, and placebo, revealing that these medications were not more effective than a placebo, with approximately 40% of patients being nonresponders. 10 This highlights the need for further studies focusing on the pediatric population, as the available evidence remains limited.
The approval of OnabotulinumtoxinA (OnabotA) by the FDA in 2010 for the treatment of chronic migraines in adults provided a new therapeutic option. 11 Botox, a purified neurotoxin produced by Clostridium botulinum, is known to block the release of acetylcholine at the neuromuscular junction and inhibit the release of migraine-related neurochemicals such as substance P, CGRP, and glutamate. 12 These mechanisms of action contribute to the prevention of migraine attacks.13,14 However, there are limited data on the use of Botox in the pediatric population. Overall, evidence remains mixed, with a few small retrospective studies and case series having reported favorable outcomes. However, it was not found to be effective in a randomized controlled trial by Winner et al. 15 It is, therefore, essential to conduct further research with larger sample sizes to confirm the efficacy and safety of Botox injections for pediatric chronic migraines.
This manuscript presents our experience with the off-label use of OnabotulinumtoxinA for chronic migraine headaches in children. The objective of this study is to evaluate the efficacy and safety of OnabotulinumtoxinA in the management of chronic migraines in the pediatric population.
Methods
This study involved a retrospective single-center chart review conducted at the University of Illinois College of Medicine. The study was approved by the institutional review board (IRB) (#1796778-3, 4/1/2022). Due to the retrospective nature of the study, the requirement for informed consent was waived. The study population consisted of all children (<18 years of age) who were managed in the outpatient pediatric neurology/headache clinic at the Illinois Neurological Institute between January 1, 2015, and December 31, 2021. Inclusion criteria for the study included children who met the diagnostic criteria for chronic daily headache and chronic migraine headache, were on prophylactic medications for chronic migraines, and had not responded to previous medications for chronic daily headaches.
Chronic daily headache is described as frequent headaches that occur for 4 or more months, with more than 15 headaches per month that last 4 hours or more. 16 Chronic migraine is characterized by headaches on more than 15 days per month, of which at least 8 headache days per month meet criteria for migraine without aura or respond to migraine-specific treatment. 11
Patients who were followed in the pediatric neurology clinic but were >18 years of age were excluded from the study. Patients were identified from the records of the pediatric neurology clinic who presented with chronic migraines.
We followed the fixed 31 site injections16 and follow-the-pain protocol injections that was specific to the needs of our patients. The leftover medication was administered at patient-specific tender areas mostly in the temporalis muscle area. 16
Botox Treatment Protocol
The Botox protocol for the treatment of chronic migraines was adapted from phase 3 research evaluating migraine prophylaxis therapy (PREEMPT) protocol. 11 The treatment duration ranged from pretreatment to 6 months and 12 months, with an injection series occurring every 3 months. All patients were maintained on their chronic daily headache prophylactic medications and encouraged to keep a headache diary. The patients and their parents were provided with information regarding the procedure, associated side effects, and the off-label use of Botox in pediatric patients. Before each visit, patients were advised not to wear makeup, and a thorough assessment of their headache history was conducted at the headache clinic. Information was obtained regarding the frequency, duration, location, and quality of the headaches, as well as any associated symptoms and exacerbating or alleviating factors. Medication reconciliation was performed, and the impact of headaches on daily activities and other medical issues was addressed.
Botox was prepared by reconstituting the vials, which were kept refrigerated. Once reconstituted, the Botox was used within 1 hour or refrigerated if needed. Care was taken during reconstitution to avoid denaturing the Botox by shaking or violent movement. Each treatment session involved the administration of Botox injections via intramuscular (IM) injections at 31 fixed sites across 7 specific head and neck muscle groups, following the PREEMPT approach. 17 The Botox was reconstituted to a concentration of 100 units in 2 mL of normal saline, and 10 units per 0.1 mL were injected at each FDA-approved site, resulting in a total of 31 site injections. Side injections were performed following cleaning each area with alcohol and allowing it to dry. The administration of 155 units of Botox was carried out by a pediatric neurologist and a nurse practitioner, with the patient allowed to rest between the procedure. Trapezius muscle injections were typically the starting point because of their minimal discomfort. Fixed symmetrical injection sites were located, and medication was administered. After the completion of the 155 units of injections, the remaining medication was administered using the patient-specific follow-the-pain protocol. Following the injection procedure, patients were instructed not to rub the injection site and were asked to rest for approximately 1 hour.
Data Collection and Statistical Analysis
Data for the study were extracted from the electronic medical records, including demographic variables such as age and gender, and comorbidities. Other data extracted included information on other chronic migraine medications and brain magnetic resonance imaging (MRI) findings. Details regarding the patient's headache history, including Headache Impact Test (HIT 3) 18 scores, and Migraine Disability Assessment (MIDAS) scores 19 were also recorded. The pretreatment, 3-month, 6-month, and the last recorded headaches course were extracted from the electronic medical records. Patients reported quality of life measures including subjective improvement (yes/no), improvement in mood and concentration, as well as treatment-related side effects were also obtained. Standard descriptive analysis was performed, and values were presented as medians with interquartile ranges or frequencies with percentages as applicable. The headache score at the beginning of the treatment and at 6 months were compared using paired t test. Statistical analysis was conducted using JMP Pro version 17.0 (SAS Institute, Cary, NC).
Results
During the study period, a total of approximately 1121 unique patients between 5 and 17 years were seen in the neurology clinic for the principal complaints of headache (out of total 6895 patients). A total of 32 patients were offered Botox treatment, 8 patients declined, and our current manuscript describes our experience with the 24 patients. The median age of the study population was 15.4 years (interquartile range: 14.5, 16.4), with an age range of 12.0 to 17.5 years. A large proportion of the study population was female (21/24, 87.5%) and non-Hispanic White (20/24, 83.3%). The most common comorbidity observed was a history of depression, present in 41.6% (10/24) of the study population, followed by sleep disturbance, present in 33.2% (8/24) of the patients. Learning difficulties were present in 10.5% (2/19) of the patients on whom the data were available (Table 1).
Demographics.

Median (interquartile range).
Other comorbidities include chronic abdominal pain 2, anxiety, 4, attention-deficit hyperactivity disorder (ADHD) 1, asthma, 4.
Missing 5.
Headache History
Nearly 45% of patients had made lifestyle modifications due to migraines, and 10 of 24 patients had used vitamin supplementation for migraines. Most patients had previously attempted multiple medications, with a median of 5 prior medications used (IQR 4, 7). Approximately one-fourth of patients (6/24, 25%) were using as-needed pain control medications more than 3 times a week and had experienced breakthrough emergency room visits for severe headaches (6/23, 26%). Six patients (26%) had abnormal MRI findings, and 3 patients had a concussion history (Table 2).
Headache History.

Abbreviations: ER, emergency department; IQR, interquartile range; MRI, magnetic resonance imaging; PRN, as-needed.
Missing 1.
Amitryptaline/sertaline.
Topiramate/valproate.
Stable postsurgical changes (tumor resection) 1, arachnoid cyst 2, arteriovenous malformation 1, low-lying tonsils 1, nonspecific diffusion restriction in splenium (resolved) 1, extracranial abnormalities 2.
Treatment Course
The pretreatment median Headache Impact Test and Migraine Disability Assessment scores were 71.5 (IQR 66.3, 78) and 67.5 (IQR 50, 95), respectively. Both headache scores significantly decreased at the 3- and 6-month visits. The last recorded median Headache Impact Test and Migraine Disability Assessment scores were 53 (IQR 39, 60) and 17.5 (IQR 10, 30), respectively. Paired t tests indicated a significant difference between the pretreatment and posttreatment headache scores for Headache Impact Test and Migraine Disability Assessment scores. The mean difference of the Headache Impact Test score (last score vs first) was 19.6 (95% CI −24.8, −14.3, P < .001), whereas the mean difference for the Migraine Disability Assessment scores (last score vs first) was −50.8 (95% CI −62.8, −39.0, P < .001).
Most patients (17/24, 71%) reported subjective improvement, with nearly 50% reporting improvement in quality-of-life measures such as mood and concentration. Treatment-related adverse effects were reported by 5 patients (20%) and included neurologic manifestations in 1 patient, gastrointestinal symptoms in 3 patients, and renal symptoms in 2 patients (Table 3). Injection site reaction and self-limited muscle weakness was reported by 5 patients (20%).
Treatment Course.
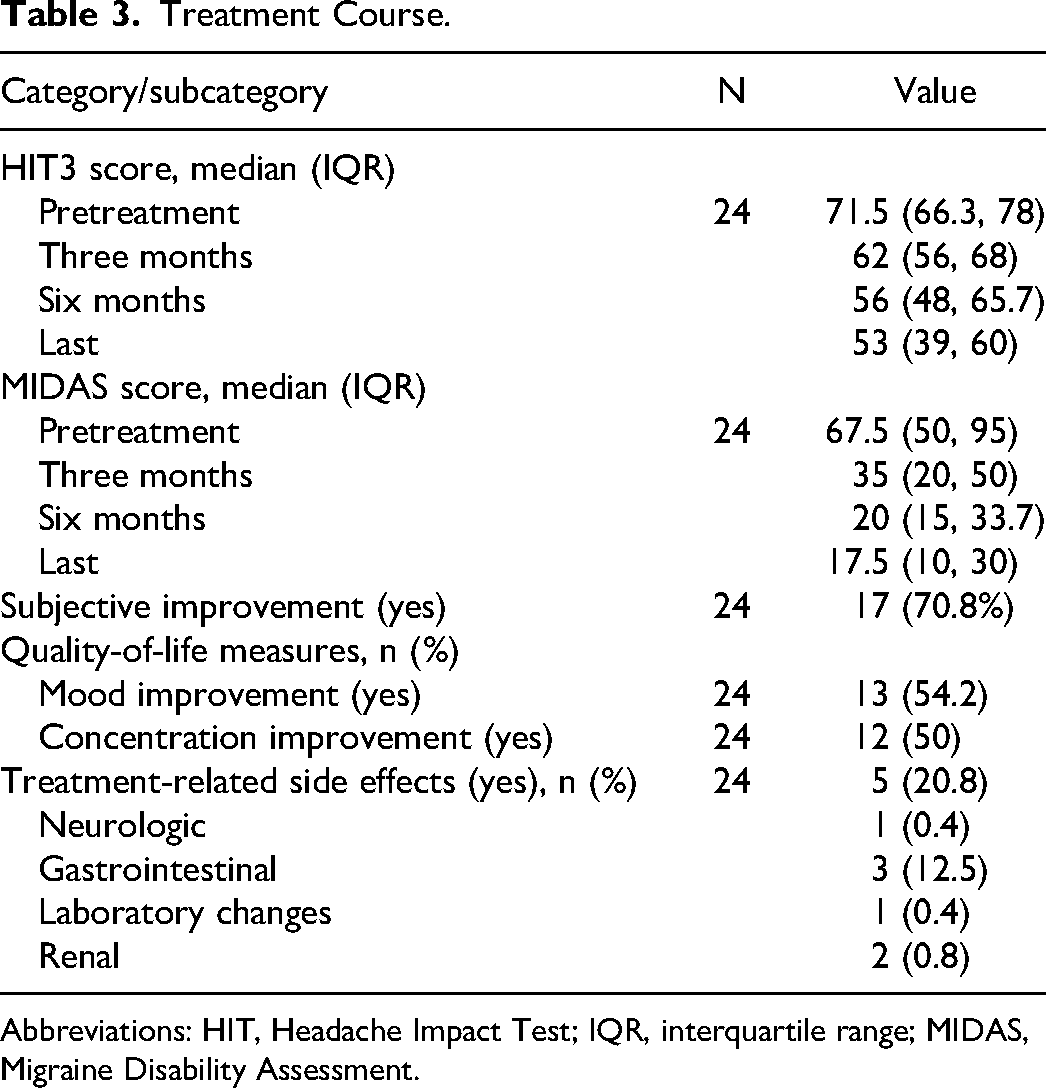
Abbreviations: HIT, Headache Impact Test; IQR, interquartile range; MIDAS, Migraine Disability Assessment.
Discussion
The results of this retrospective chart review demonstrated that Botox injections showed good clinical and subjective improvement in headache control with minimal adverse events in the studied population of 24 pediatric patients. Our findings add to the growing evidence on the safety and efficacy of Botox for pediatric migraines.
Botox injections, initially used for cosmetic purposes, have been adopted by several adult centers across North America for migraines and chronic tension-type headache prophylaxis. 20 Our findings of improvement in headache control and quality of life agree with the prior reports by Chan 21 (n = 6, 14-18 years, Botox 100 units), Shah 22 (n = 10, 8-17 years, 165-215 units), Bernhard 23 (n = 10, 13-17 years, 150 units), Schroeder 24 (n = 5, 10-16, 40-200 units), and more recently by Goenka 16 (n = 25, 13-21 years, 155 units). Our findings, however, conflict with the randomized controlled trial by Winner et al. 15 The dose utilized in the Winner trial (155 units in 45 participants and 74 IU in 74 participants) is lower than the one used in our study. Furthermore, we also used the “follow the pain” protocol, which is specific to the patient and may have yielded better results. Results from our study and other case series and chart reviews suggest that this procedure is well tolerated in the pediatric population.
In our study, we use the standard dose of 155 international units using a strict protocol and the rest of the 45 units as patient-specific follow-up pain protocol. The follow-the-pain protocol, as employed in this study, allows for a more individualized treatment approach based on the patient's specific pain profile and tenderness. 25 Future studies evaluating the efficacy of Botox injections using this protocol will be valuable in optimizing treatment outcomes. We have also consistently followed the patients at 3, 6, and 9 months, allowing us a longer time frame to assess improvements and side effects.
It is essential to consider potential side effects and adverse events associated with Botox injections. Although systemic side effects are rare, muscle weakness beyond the injection site can occur. Providers and headache neurologists should exercise caution when administering Botox to pediatric patients. We also reported very few treatment-related side effects. Prior retrospective reports by Chan, 21 Shah, 22 Schroeder, 24 and Goenka 16 also showed no serious adverse effects. In the case series by Bernard, 23 on 10 patients, the only side effects noted were redness and temporary pain. The randomized controlled trial by Winner et al, 15 although negative for pain control, also did not show any serious adverse events. Immunogenicity, particularly the development of toxin-neutralizing antibodies, can affect the clinical effectiveness of Botox treatment. However, the incidence of immunogenicity is uncommon, and the PREEMPT study 17 showed no positive toxin-neutralizing antibodies in the chronic migraine population. While we report data up to 9 months follow-up, even longer follow-up studies are needed to assess the immunogenicity of Botox in pediatric patients.
While our report affirms the beneficial effect of Botox with an excellent safety profile, comparative studies between Botox injections and other preventive medications, such as topiramate, are needed to provide a head-to-head comparison of their efficacy and safety profiles in the pediatric population. One pilot study comparing OnabotulinumtoxinA with topiramate demonstrated a significant reduction in headache frequency and improved quality of life with fewer side effects in the Botox group. 26 This suggests that Botox injections may be a more effective treatment option for pediatric chronic migraines. Other chronic headache conditions, such as occipital neuralgia and trigeminal neuralgia, may also benefit from Botox injections. Lidocaine injections, commonly used in adults, may have more side effects in the pediatric population, making Botox a potentially safer alternative. 27 New daily persistent headaches (NDPH) are another challenging subtype of chronic daily headaches encountered in pediatric patients, and further research is needed to investigate the effectiveness of Botox or lidocaine injections for managing new daily persistent headaches.
Deterrents to the use of Botox injection in the treatment of chronic migraine especially in the pediatric population among adults could also include phobia of needles and also the number of injections per session. Anecdotally we have observed that after seeing the response to Botox injections, some of the adolescents were able to overcome that fear of the injections once they realized that the injection discomfort was limited to the forehead area and not in other areas such as the neck, back of the head, and the shoulder muscles. Although we have not directly compared noncompliance, the fact that all of our patients completed the therapy suggests that the risk of noncompliance with Botox injection is significantly lower than taking daily preventative medication for the management of chronic daily headaches. 28
We monitored both Migraine Disability Assessment and Headache Impact Test–6 scores to assess response to treatment. Although both scores showed a significant decline, larger effect sizes were observed in the Migraine Disability Assessment scores. Both scores measure the headache-related disability; however, there are subtle differences with Headache Impact Test–6 being affected more by headache intensity and Migraine Disability Assessment more by headache frequency. 19 Migraine Disability Assessment score is also relatively simpler to do than Headache Impact Test–6 for the families and may have had a more reliable response than Headache Impact Test–6, 29 which may explain some of the observed differences.
Limitations of our study include its retrospective design and lack of a control population. Retrospective studies such as ours are inherently subject to investigators’ bias as well as placebo effect on the patient. Because of the relatively small sample size, we were also not able to perform any comparative analysis based on differences in risk factors and comorbidities. These limitations may be best addressed with a multicentric study. Although we used the validated headaches scores, they are still inherently subjective and are subject to measurement bias.
Conclusion
In conclusion, this study supports using Botox injections as a potentially effective therapeutic intervention for managing chronic migraines in the pediatric population. It demonstrates that Botox injections can lead to a decrease in pain scores and an improvement in the quality of life for pediatric patients with refractory migraines. Controlled trials with larger sample sizes and more extended follow-up periods are necessary to provide more robust evidence on the efficacy and safety of Botox injections for pediatric migraines.
Footnotes
Author Contributions
AA designed and conceptualized the study, assisted in data collection, analysis and interpretation, and manuscript drafting. JF assisted in the design of the study and data collection and provided critical input in drafting the manuscript. ST assisted in the study's design, statistical analysis and interpretation, and manuscript drafting. All authors have reviewed and approved the manuscript as submitted.
Author note
Asra Akbar is currently affiliated with Division of Pediatric Neurology, University of Saint San Antonio.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was conducted according to the guidelines of the Helsinki and approved by the institutional review board of Children's Hospital of Illinois (Universityof Illinois College of medicine in Peoria IL, human subject institution review board approval UP 1796778 12/14/2021) written informed consent was not required and HIPPA authorization for use of protected health information was obtained.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
