Abstract
Selection of targets for deep brain stimulation (DBS) has been based on clinical experience, but inconsistent and unpredictable outcomes have limited its use in patients with heterogeneous or rare disorders. In this large case series, a novel staged procedure for neurophysiological assessment from 8 to 12 temporary depth electrodes is used to select targets for neuromodulation that are tailored to each patient's functional needs. Thirty children and young adults underwent deep brain stimulation target evaluation with the new procedure: Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation (SABERS). Testing is performed in an inpatient neuromodulation monitoring unit over 5-7 days, and results guide the decision to proceed and the choice of targets for permanent deep brain stimulation implantation. Results were evaluated 3-6 months postoperatively with the Burke-Fahn-Marsden Dystonia Rating Scale and the Barry-Albright Dystonia Scale. Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation testing allowed modulation to be tailored to specific neurologic deficits in a heterogeneous population, including subjects with primary dystonia, secondary dystonia, and Tourette syndrome. All but one subject were implanted with 4 permanent deep brain stimulation leads. Results showed significant improvement on both scales at postoperative follow-up. No significant adverse events occurred. Use of the Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation protocol with evaluation in the neuromodulation monitoring unit is feasible and results in significant patient benefit compared with previously published results in these populations. This new technique supports a significant expansion of functional neurosurgery to predict effective stimulation targets in a wide range of disorders of brain function, including those for which the optimal target is not yet known.
Deep brain stimulation is approved in the United States for patients with essential tremor (ET), advanced Parkinson's disease (PD), obsessive compulsive disorder, primary generalized dystonia, and medication-resistant focal epilepsy. 1 The vast majority of such surgeries are performed for patients with ET or PD. A few case studies document attempts at treating a variety of other diseases such as depression, secondary dystonia, addictive behavior, obesity, pain syndromes, and Tourette syndrome.2-4 Although these other diseases impart a huge burden on worldwide health care costs, clinical studies using single-target deep brain stimulation (DBS) have not yet shown consistent efficacy sufficient to warrant approval by regulatory agencies. We conjecture that lack of efficacy is in part due to failure to adjust targets to variations in location of injury and the individual pathophysiology of heterogeneous disorders. We thus developed the Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation (SABERS) technique for personalized identification of deep brain stimulation targets. Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation is a physiological assessment that guides functional neurosurgical interventions, just as magnetic resonance imaging (MRI) is an anatomical assessment that guides structural neurosurgical interventions. Here we report successful use of this technique for a heterogeneous group of children with pediatric movement disorders.
Pediatric movement disorders including dystonia, chorea, and tremor can be severely debilitating yet have few effective pharmacologic or surgical interventions. 5 Cerebral palsy is the most common cause of acquired movement disorders in children, with a prevalence of 2 per 1000 live births, 6 and is a common etiology of dystonia. 7 Other genetic and neurometabolic disorders cause movement disorder symptoms, either directly (eg, DYT1-associated dystonia) or indirectly through secondary brain injury (eg, type 1 glutaric aciduria). Deep brain stimulation was approved by the US Food and Drug Administration (FDA) for use in primary generalized dystonia beginning at 7 years under a Humanitarian Device Exemption. 8 There is no age restriction on its use in Europe, where it has been used in children as young as 3 years of age.9,10 Results from the use of deep brain stimulation in childhood movement disorders have been mixed, in part because of the heterogeneity of pathophysiology and symptomatology, even with similar underlying genetic or acquired causes. 11 Considering parents and caregivers often describe improvement in quality of life following deep brain stimulation, interest in this intervention has been maintained despite the mixed results. 12
Multiple potential targets for deep brain stimulation in pediatric and adult disorders have been proposed, 13 and target selection has been identified as a likely determinant of treatment success. 14 The heterogeneity of the population suggests that targets for deep brain stimulation are likely to vary between different children, but until recently no principled method for individualized selection of targets has existed.
We report our case series of 29 children with dystonia and one child with Tourette syndrome who underwent a novel personalized surgical targeting procedure developed by our group: Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation.15,16 This procedure uses 4 to 12 temporary depth electrodes to perform neurophysiological assessment of potential stimulation sites. Following implantation of temporary electrodes, patients are observed for at least 5 days in a neuromodulation monitoring unit using both stimulation and recording to identify the sites most likely to be beneficial for permanent lead implantation. The surgical technique is adapted from the relatively low-risk depth electrode placement and testing used in the surgical management of epilepsy, in which epileptogenic brain regions are identified individually based on recording and stimulation at multiple sites. 17 This study extends and includes the subjects from a smaller previously reported case series and report.15,18
Materials and Methods
Patient Selection
Patients identified as surgical candidates were diagnosed by a pediatric movement disorder specialist (T.D.S.) based on established criteria. 19 All patients were identified as having confirmed failure of symptomatic medical therapy at adequate dosing. 20 Standard deep brain stimulation approaches including both awake and asleep methods were discussed with all patients and families, although because of severity of dystonia they were often noted to be poor candidates for the more commonly used awake procedure. 21 It was discussed that utilization of the staged approach was not the conventional approach and represented a deviation from treatment at other centers. Based on the mixed or unknown response to deep brain stimulation in the patient's condition, the patient/parent elected to proceed with electrophysiological assessment of potential benefits of stimulation prior to proceeding with permanent lead implantation. Two subjects with inadequate response to prior placement of bilateral pallidal electrodes proceeded with test electrode implantation to assess for the possibility of additional benefit. Patients or parents of minor patients consented to surgical procedures according to standard hospital consent procedures. They also consented or assented as appropriate to HIPAA authorization for research use of protected health care information and institutional review board–approved consent for research use of electrophysiological data. Subject demographics and diagnoses are given in Table 1. To reduce reporting and outcome bias, we included all 31 patients treated consecutively from initiation of the procedure in 2016 through December 2020. One subject withdrew consent for research participation for reasons unrelated to the procedure. Thus, the data from 30 subjects are presented. Subjects 1 through 8, subject 10, and subject 23 were previously reported.15,18
Subject Demographics.
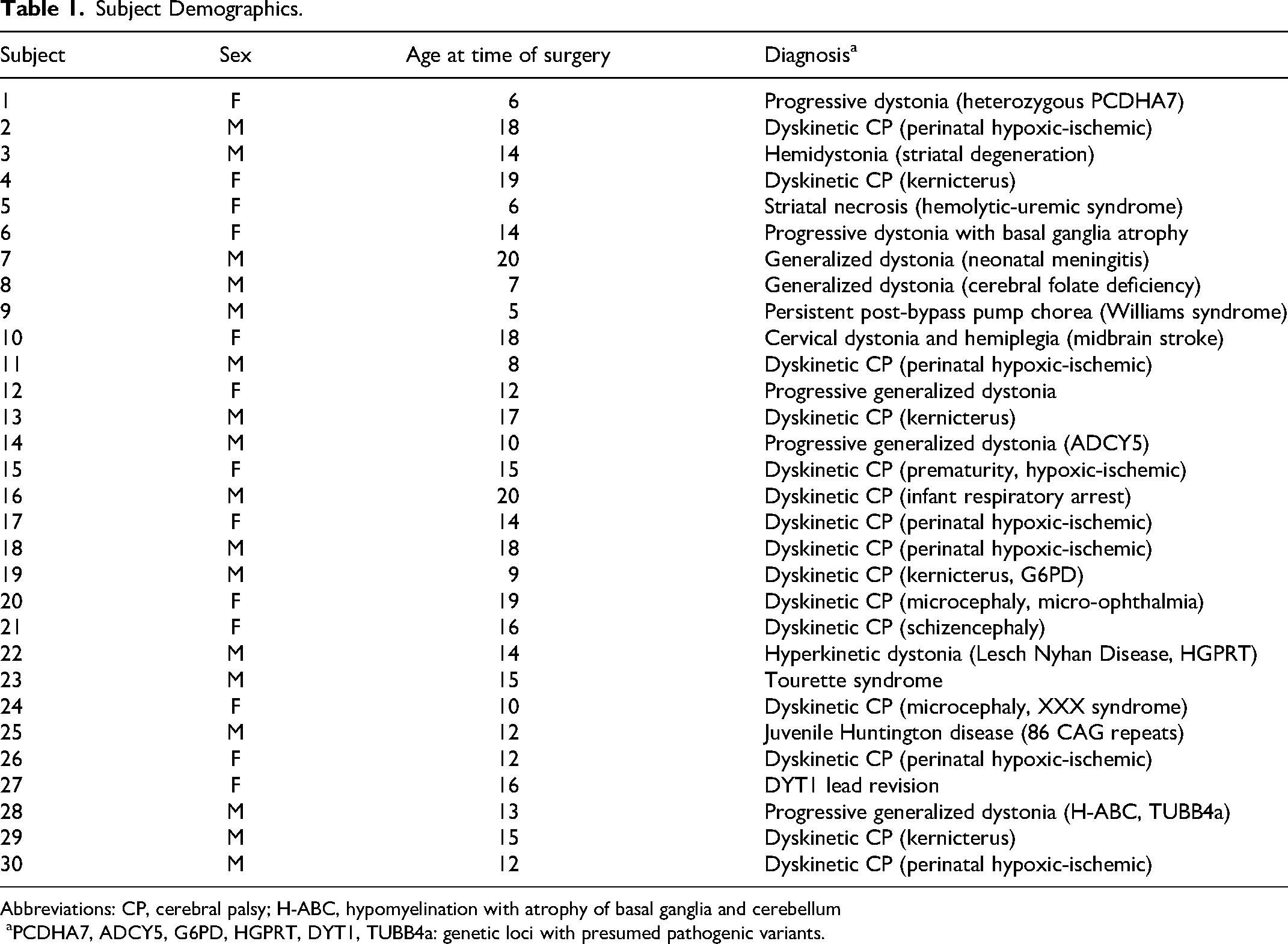
Abbreviations: CP, cerebral palsy; H-ABC, hypomyelination with atrophy of basal ganglia and cerebellum
aPCDHA7, ADCY5, G6PD, HGPRT, DYT1, TUBB4a: genetic loci with presumed pathogenic variants.
The technique has been previously described and will be summarized here.15,16 Potential targets for test stimulation and recording are identified based on reported case studies and the presumed functional connectivity pathways affected by the disorder. Targets are named based on Hassler's terminology 22 : ventralis intermediate nucleus thalamus (Vim), ventralis oralis complex of thalamus (Vo), ventral anterior nucleus of thalamus (VA), subthalamic nucleus (STN), and globus pallidus internus (GPi) anterior and posterior. The majority of patients had multiple leads implanted in globus pallidus internus and different thalamic nuclei, predominantly identified based on pattern of injury seen on imaging. The number of electrodes was limited at times because of the patient's anatomy, or need to limit time under general anesthesia because of additional health concerns. Nucleus accumbens (NA) and centromedian parafascicular nucleus of thalamus (CMPf) were used when tics or compulsive behaviors were present. Although variations in subjects’ anatomy was expected due to differences in underlying pathology, in all cases targeting performed using the Schaltenbrand-Wahren atlas aligned and scaled to the patient's AC-PC line appeared to identify appropriate targets, confirmed by 2 neurosurgeons. We have previously confirmed that this atlas adequately identifies the Vim/Vo boundary in thalamus 23
Phase 1 Surgery (Temporary Stereotactic Electrode Placement)
A preoperative stereotactic MRI scan with high-resolution T1 with and without intravenous gadolinium contrast and T2 sequences were obtained for each subject. Stereotaxy was performed using either a Radionics CRW head frame (Integra Life Sciences Corp, Burlington, MA, USA) with guidance from STEALTH stereotaxy software (Medtronic Inc, Minneapolis, MN, US), or a ROSA surgical robot with guidance from ONE software (Zimmer Biomet, Montpellier, France). Targeting was performed using standard surgical anatomical Schaltenbrand-Wahren atlas locations relative to the AC-PC line. After induction of general anesthesia, bone fiducial markers (for ROSA) or the CRW frame were secured to the skull, and a stereotactic computed tomography (CT) scan was obtained. The CT scan was aligned with the preoperative MRI to identify coordinates of stereotactic targets and to plan trajectories to avoid vessels, regions of high vasculature, or entry into the lateral ventricles. The head was stabilized using a Mayfield fixation device (Integra, Princeton, NJ, USA). When the ROSA stereotactic robot was used, registration to the fiducial markers was performed to ensure an error of <0.5 mm.
Under ROSA or Stealth stereotactic guidance, 4-12 Adtech MM16C depth electrodes (Adtech Medical Instrument Corp, Oak Creek, WI, USA) were placed via 2.4-mm drill holes at identified targets. MM16C electrodes are FDA-approved for invasive electrophysiological monitoring in humans. Each electrode has a diameter of 1.2 mm and includes 6 low-impedance (1-2 kΩ) contacts that run circumferentially around the electrode in a 2-mm band for stimulation. There is a 5-mm distance between the centers of each low-impedance contact. Each electrode also contains 10 high-impedance (70-90 kΩ) recording contacts that are approximately 50 μm in diameter and arranged in groups of 2 or 3 circumferentially around the electrode both near the tip and between the low-impedance electrode bands. Electrodes were secured to the skull using 13-mm anchor bolts (Adtech LSBK1-BX-06).
For infection control, patients use head and body antibacterial wash preoperatively, the hair is shaved immediately prior to surgery, and intravenous cefazolin is given perioperatively and throughout the following week of testing. With this protocol, no associated infections occurred. Postoperative CT was performed to confirm accurate localization of the electrodes, and to rule out complications. No implantation related hemorrhages occurred. Temporary depth electrodes have been associated with a lower occurrence of complications in the pediatric population compared with the adult population24,25 and are well tolerated when used for epilepsy focus identification.
Neuromodulation Unit Testing
Following placement of depth electrodes and recovery from general anesthesia, patients were transferred to a neuromodulation monitoring unit (NMU) on the inpatient Neurology ward for monitoring. Assessment did not begin until 24 hours after surgery to allow for full recovery from the effects of general anesthesia. If postoperative somnolence was noted, stimulant medications (methylphenidate or modafinil) were used to promote maintenance of daytime wakefulness and allow assessment of symptoms during activity. Children were in a regular hospital bed and unrestrained, although observed for safety and to avoid traction on the intracranial electrodes. At least 1 parent or guardian remained at the bedside throughout testing.
Bipolar stimulation was performed through adjacent low-impedance contacts on the MM16C electrodes after lead integrity was confirmed by ensuring impedance between the stimulation contacts was between 1 kΩ and 5 kΩ. Stimulation and impedance testing were performed using a Medtronic external neurostimulator 37,022 with a Medtronic 8840 DBS programmer (Medtronic Inc, Minneapolis, MN, USA) at bilateral (homologous left and right) contacts, with settings of 90-μs pulse width, 60-Hz or 185-Hz frequency, and increasing voltage up to 5 V, for a total of 3-5 minutes per condition. Clinically beneficial contacts were probed initially bilaterally and subsequently unilaterally, with adjustments in frequency by 5-Hz steps and pulse width by 10-μs steps to find optimal settings. During this time, potential beneficial effects including decreased tone, decreased overflow or hyperkinetic movements, improved accuracy of movements, improved range of motion, and improved functional task performance could be observed by parent, patient, and examiner and were recorded by video and bedside notation.
Although it is widely believed that the effect of deep brain stimulation can take weeks or months to become apparent, in all cases we observed immediate effects. As in essential tremor and Parkinson disease, when stimulation in thalamus or subthalamic nucleus was effective the effects were seen within 3-5 seconds of stimulation onset. 26 Effects from pallidal stimulation took longer, but initial beneficial responses could be seen within 2-3 minutes of stimulation onset. Adverse sensory or motor effects were visible on similar time scales.
Testing was customized to the particular abilities and symptoms of each child, generally using standard clinical neurologic assessments (see Table 2). Objective improvements including improvements in passive tone, passive or active range of motion, voluntary strength, accuracy of movement during specific tasks, and speed of movement during specific tasks were recorded by video and documented by the examiner at each stimulation setting and compared with the same parameters off stimulation. Subjects were also monitored for deleterious effects including involuntary muscle contractions, worsening dystonia, paresthesia, discomfort, and changes in mood.
Standard Components of the Neurologic Examination used for Movement Assessment of Children in the Neuromodulation Monitoring Unit.

Tasks and clinical testing were also tailored to the specific function, abilities, and goals of each child. For example, children with significant cognitive deficits might be assessed for ease of care, toileting, opisthotonos, or painful spasms, whereas children with minimal cognitive deficits might be assessed for quality of handwriting, self-feeding, speech, or gait. Therefore, no standardized set of clinical scales could be used. At the beginning of the testing week, a set of goals is determined based on parent report and examination of the child. These goals are assessed clinically on the standard clinical neurologic examination, and documented by video. This documentation is designed to permit post hoc quantitative assessment, but decision making is based entirely on the clinical examination and parental assessment of improvement. Decisions include determining the locations and leads of the most beneficial stimulation contacts, and determining whether sufficient benefit was obtained that would merit implantation of permanent leads.
Potentially beneficial stimulation settings were maintained overnight in order to determine adverse or beneficial effects on sleep, and longer-term effects on motor function the following morning. If side effects were noted at or below routine therapeutic voltages (less than 3.5 V for pallidum, or less than 2.5 V for thalamus), those areas were not considered for permanent implantation. Once identified, testing at the effective parameters was repeated at least 24 hours apart, with results confirmed by multiple members of the clinical team and the parents to ensure consistent response. When possible, a clinical team member was blinded to stimulation parameters during assessment.
In our experience, because of the short duration of stimulation in the neuromodulation monitoring unit, any observed benefit underestimates the long-term benefit of permanent leads. However, in the absence of benefit in the neuromodulation monitoring unit, we would not have evidence that permanent leads would have long-term benefit. Therefore, the decision to implant permanent leads requires observation of at least a partial clinically relevant degree of benefit during the neuromodulation monitoring unit testing. Although the examination videos are sufficient for performance of quantitative ratings such as the Burke-Fahn-Marsden Dystonia Rating Scale 27 and the Barry-Albright Dystonia Scale, 28 these scales were not performed because such measures in the neuromodulation monitoring unit are not expected to be predictive of outcome. Furthermore, in our experience, these ratings do not have sufficient sensitivity 29 to be useful for decision making based on short-term improvement, and they do not reflect functional gains in performance, care, or comfort relevant to patients and parents.
Recording of spontaneous brain activity and evoked potentials in response to electrical stimulation was performed on the high-impedance contacts of the MM16C electrodes using a 256-channel 22-kHz electrically isolated recording system (Tucker-Davis Technologies, Inc). Evoked-potential data were not used for this study and will be reported elsewhere. Recording occurred continuously over the 5-12 days of neuromodulation monitoring unit testing, yielding 2-5 terabytes of data per child. In addition to continuous recording of intracranial data, scalp electrodes (limited 8-channel montage surrounding C3 and C4 scalp locations) and video were recorded. During arm movement and gait testing, surface electromyogram (EMG), accelerometer, and gyroscopic data are also recorded (Delsys Trigno system) and stored, although accelerometry and gyroscopic data were not used for this study and will be reported elsewhere. Data were stored for subsequent offline analysis. When recording is performed during therapeutic stimulation, removal of stimulation artifacts is done offline in order to isolate intrinsic brain activity and activity in response to stimulation (analytic methods and results reported elsewhere). Recording data are used to identify brain regions whose activity correlates with abnormal movement. This is particularly helpful to choose between 2 or more targets with similar clinical benefit. In such cases, the region whose activity was most highly correlated with abnormal movement was chosen as the target. Analysis includes bandpass filtering (300-3000 Hz) followed by principal components dimensionality reduction, and k-means clustering for spike identification (Tucker-Davis Technologies software). An example of the resulting recording data aligned to low-pass filtered surface EMG (5-450 Hz) is shown in Figure 1. In this figure, it can be seen that activity of the GPi1 (posterior globus pallidus internus lead) contacts was significantly greater than in GPi2 (anterior lead) contacts during dystonic spasms recorded on EMG, and thus the posterior globus pallidus internus lead was chosen for permanent implantation.
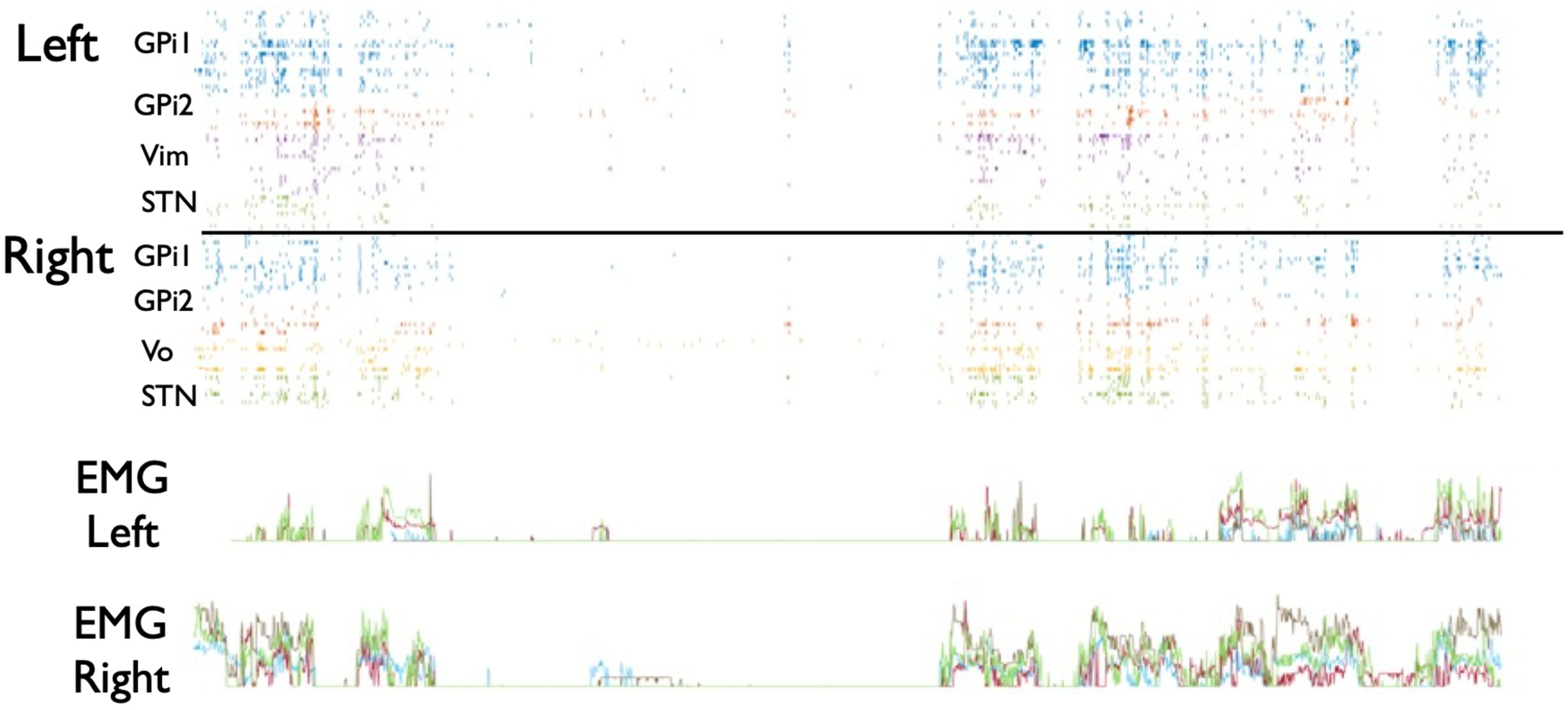
Sample spike activity recorded from multiple brain regions (color coded) in subject 1 during dystonic spasms. Horizontal axis is time (0-20 s). Each row shows spike timing for individual spikes in each of the recorded brain regions. High activity during spasms in GPi1 (posterior globus pallidus internus) leads justified selection of this target rather than GPi2 (anterior globus pallidus internus). EMG shows spontaneous dystonic spasms. Between spasms, all recorded areas are mostly silent. During spasms, most areas show activity.
Following the conclusion of inpatient testing, the clinical team and the family discussed whether or not to proceed with placement of permanent electrodes. In all cases with the exception of one subject in which neither parents nor clinicians appreciated benefits, the decision to proceed was made. A total of 4 permanent electrodes were placed in the phase 2 procedure, one on each side in each of the best 2 selected locations. Adtech depth electrodes were removed either at the bedside or in the operating room under general anesthesia (based on surgical considerations), and the patient was discharged (Figures 2 and 3).
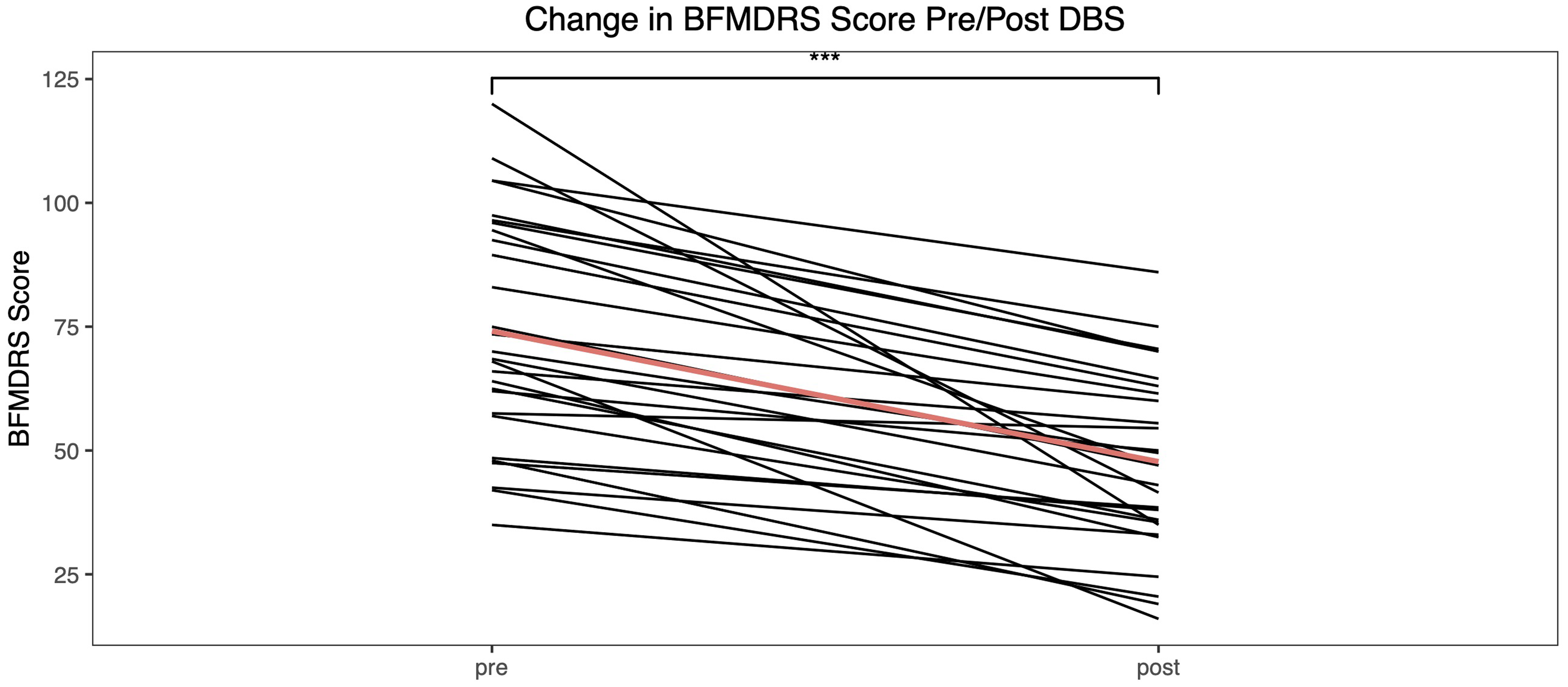
Change in Burke-Fahn-Marsden Dystonia Rating Scale for each of the 30 subjects included in the study. Red line shows mean change.
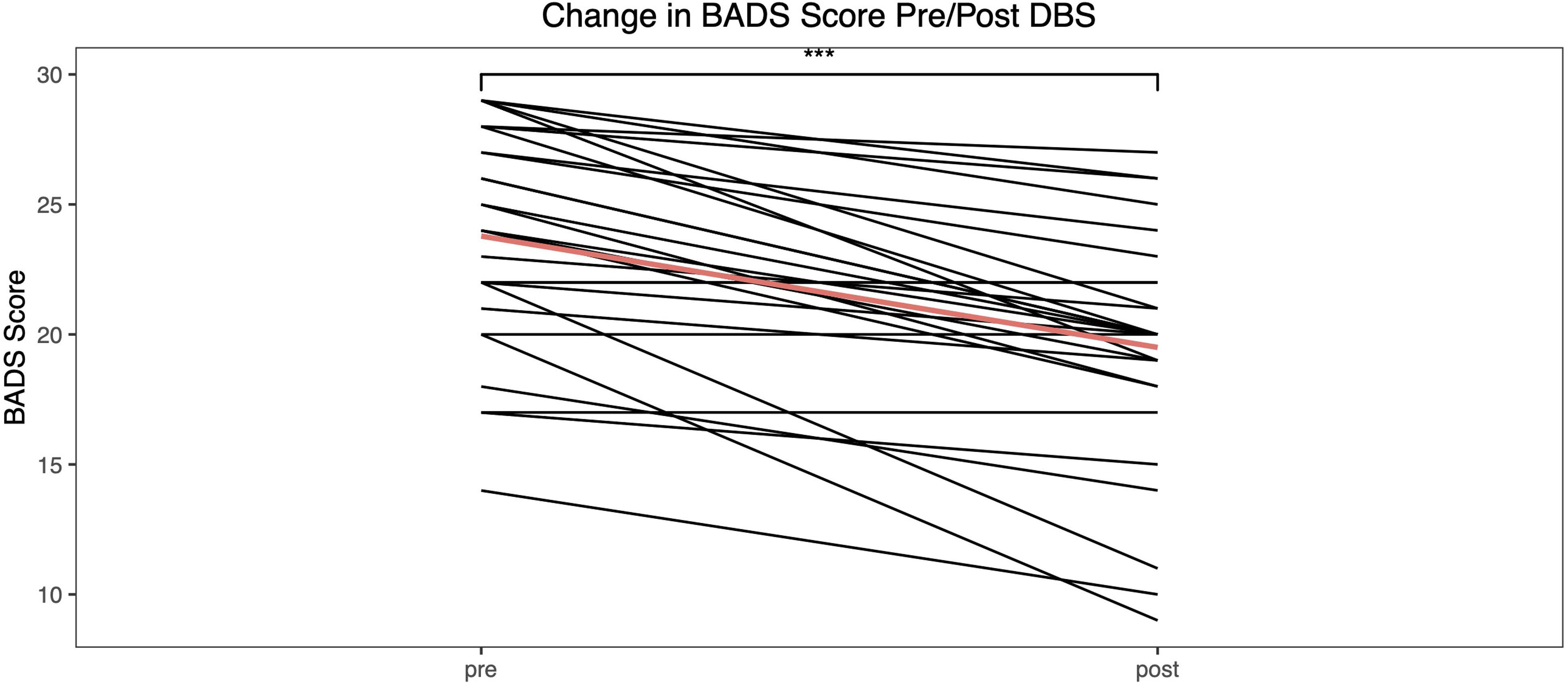
Change in Barry-Albright Dystonia Scale for each of the 30 subjects included in the study. Red line shows mean change.
Phase 2 Surgery (Permanent Deep Brain Stimulation Electrode Placement)
In order to ensure lack of infection, phase 2 surgery is scheduled only after a minimum interval of two weeks, during which the patient must be afebrile without antibiotics. Permanent electrodes (Model 3387 or 3389; Medtronic, Inc) were then implanted with the patient under general anesthesia using procedures similar to those used for the first-phase surgery. Targeting was determined by fusing the preoperative MRI and postoperative first-phase CT (showing the locations of the temporary depth electrodes) to the second-phase CT performed with the CRW frame or bone fiducials in place (for use of ROSA). This allows targeting permanent electrodes based on the location of the most effective depth electrodes. The entry points on each side are adjusted when possible so that both electrodes on the same side can be placed through the same burr-hole and fixed in place using the same cranial fixation device (Stimloc cap, Medtronic Inc). In most cases, the Stimloc cap needed to be altered during the surgical procedure by using a cutting burr to cut a second exit channel in the plastic so as to accommodate two electrodes. Extracranial leads were labeled, with the proximal ends placed in protective sleeves (“boots”) according to manufacturer recommendations and buried beneath the skin prior to wound closure. Intravenous (IV) vancomycin and ceftazidime were administered perioperatively and for 72 hours postoperatively, and the patient was discharged on 10 days of enteral dicloxacillin or equivalent. Antibiotics were adjusted for patients with medication allergies. To ensure sterility of the implanted components, the patient was required to have remained afebrile off antibiotics for at least 4 days before the next phase of surgery, thus requiring again a minimum of 2 weeks (10 days on antibiotics, 4 days off antibiotics) between phase 2 and phase 3 surgeries.
Phase 3 Surgery (Pulse Generator Placement)
Extension leads (Model 7483, Medtronic Inc) are connected to the intracranial electrodes and tunneled subcutaneously to implanted pulse generators (Activa RC, Medtronic Inc) placed one on each side of the chest. To facilitate programming with similar frequencies in homologous targets, homologous leads were routed to the same stimulator (when possible, for consistency we route thalamic leads to the stimulator in the right chest, and pallidal leads to the stimulator in the left chest). Intravenous cefazolin was given perioperatively, with the patient typically discharged on the day of surgery with 10 days of enteral dicloxacillin postdischarge.
Postoperative Programming
The initial programming visit is scheduled at least 2 weeks following implantation of permanent leads and pulse generators. At this visit, all homologous contact pairs are tested in monopolar montage, voltage mode (case positive) to determine side effects and initial beneficial effects. Pallidal contacts are tested at a frequency of 185 Hz and pulse width of 90 μs up to 5 V, and thalamic contacts at 90 Hz, 90 μs up to 4 V.
For subsequent programming, when pallidal contacts are used they are programmed with gradual increases to 3.5 V. To facilitate this, the patient home programmer is configured to allow gradual increases in voltages as tolerated in 0.1-V steps. Frequency may be adjusted to optimize benefit, with typical pallidal frequencies being in the range 120-250 Hz, and typical pulse widths 120-300 μs. Following stabilization of the pallidal settings, thalamic and subthalamic contact settings are adjusted using a similar procedure, with typical frequencies 40-90 Hz and typical pulse widths 60-200 μs.
Programming visits occur at 2- to 4-week intervals, and the patient home programmer can be configured to explore different settings using “groups” and adjustable voltages or pulse widths between visits. Programming and optimization of settings is based on motor components of the neurologic examination (Table 2), as well as parent report of functional benefit (communication, ambulation, school performance, daily care, safety, comfort, feeding, etc). Final programming settings vary considerably between children, and often need to be changed several times during the first year. In some cases, settings that are maximally effective during the initial months of programming subsequently become less effective than settings that were not initially found to be beneficial. This can occur due to reduction or change in side effects, improvement in motor function, or possibly due to neural plasticity in response to long-term stimulation. Understanding this phenomenon will require a larger cohort and a future study.
Outcome Measures
The severity of each patient's dystonia was assessed using the Burke-Fahn-Marsden Dystonia Rating Scale motor component and the Barry-Albright Dystonia Scale 28 to evaluate video recordings preoperatively and at programming visits 3-6 months postoperatively, with timing based on the patient's scheduled follow-up visits in clinic. Scoring was performed by a staff member (J.A.M.) and confirmed independently by a pediatric movement disorders specialist (T.D.S.). Agreement from both raters was required for scoring. Video evaluators could not be blinded to pre- or postsurgery condition as those who had undergone surgery in some cases demonstrated visible aging due to growth. Assessment of the Tourette syndrome patient was performed using the Yale Global Tourette Syndrome Scale. 18 Concurrent with video recording for Burke-Fahn-Marsden Dystonia Rating Scale and Barry-Albright Dystonia Scale scoring, the parents and patients (when able) were queried on a 3-point Likert scale comparing current overall quality of life with preoperative quality of life as improved (+), no change (0), or worsened (–).
Results
Thirty-one patients underwent the Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation implantation procedure at 2 institutions, Children's Hospital Los Angeles and Children's Hospital Orange County. In order to maintain consistency, the same surgeon (M.A.L.) and neurologist (T.D.S.) were present and involved in procedures at both institutions. One patient withdrew consent for further participation in the study because of reasons unrelated to the procedure. Of the remaining 30 patients who underwent the initial testing procedure, 29 (96.7%) saw a significant improvement and elected to proceed with permanent deep brain stimulation implantation. Twenty-six of these patients had secondary dystonia, 2 had primary dystonia, and 1 had Tourette syndrome. No significant adverse events attributable to the staged surgical procedure or observation in the neuromodulation monitoring unit occurred. Outcomes for each subject are shown in Tables 3 and 4.
Pre and Postsurgical Scores.
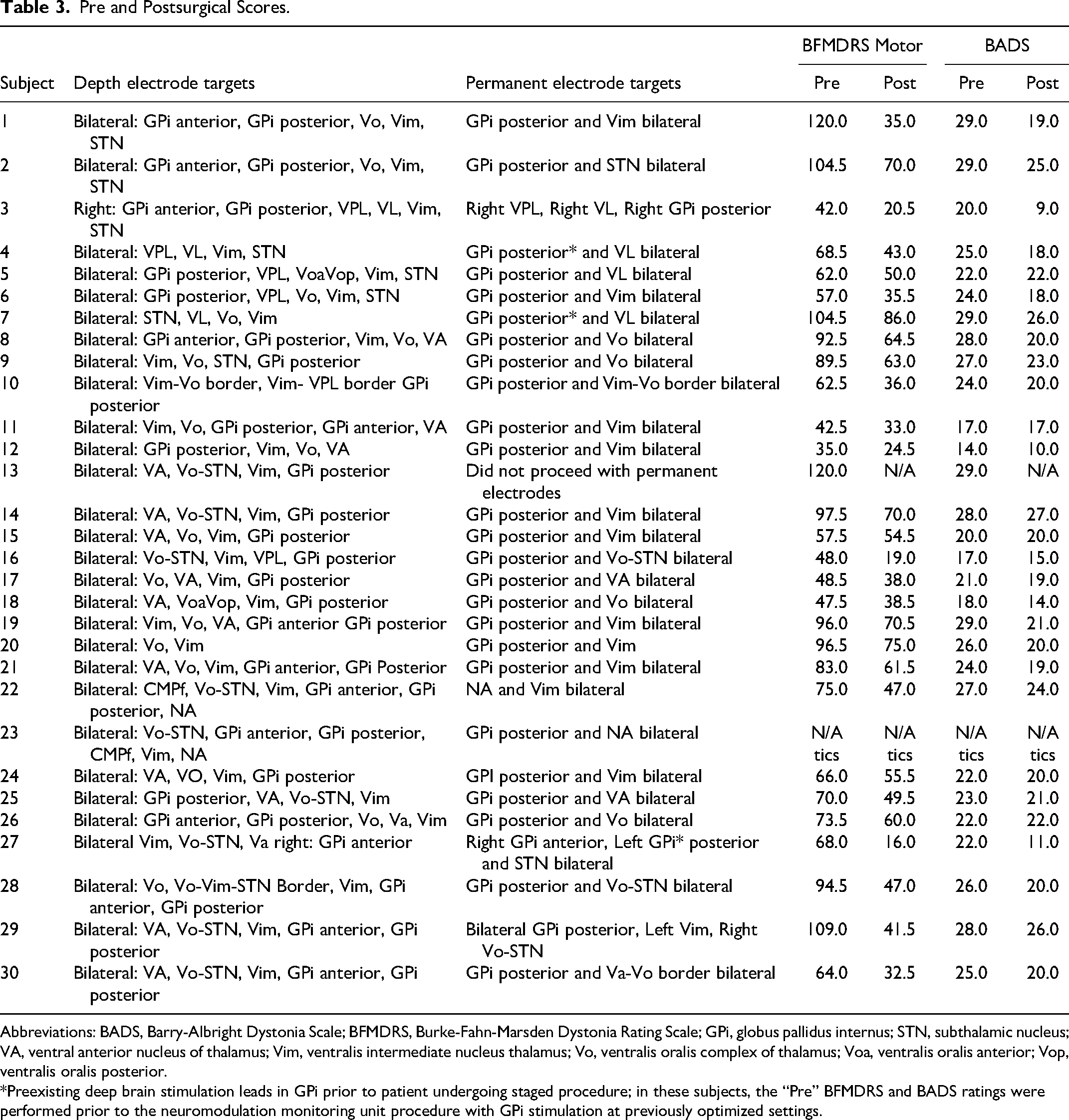
Abbreviations: BADS, Barry-Albright Dystonia Scale; BFMDRS, Burke-Fahn-Marsden Dystonia Rating Scale; GPi, globus pallidus internus; STN, subthalamic nucleus; VA, ventral anterior nucleus of thalamus; Vim, ventralis intermediate nucleus thalamus; Vo, ventralis oralis complex of thalamus; Voa, ventralis oralis anterior; Vop, ventralis oralis posterior.
*Preexisting deep brain stimulation leads in GPi prior to patient undergoing staged procedure; in these subjects, the “Pre” BFMDRS and BADS ratings were performed prior to the neuromodulation monitoring unit procedure with GPi stimulation at previously optimized settings.
Quality of Life Change Following Deep Brain Stimulation.a
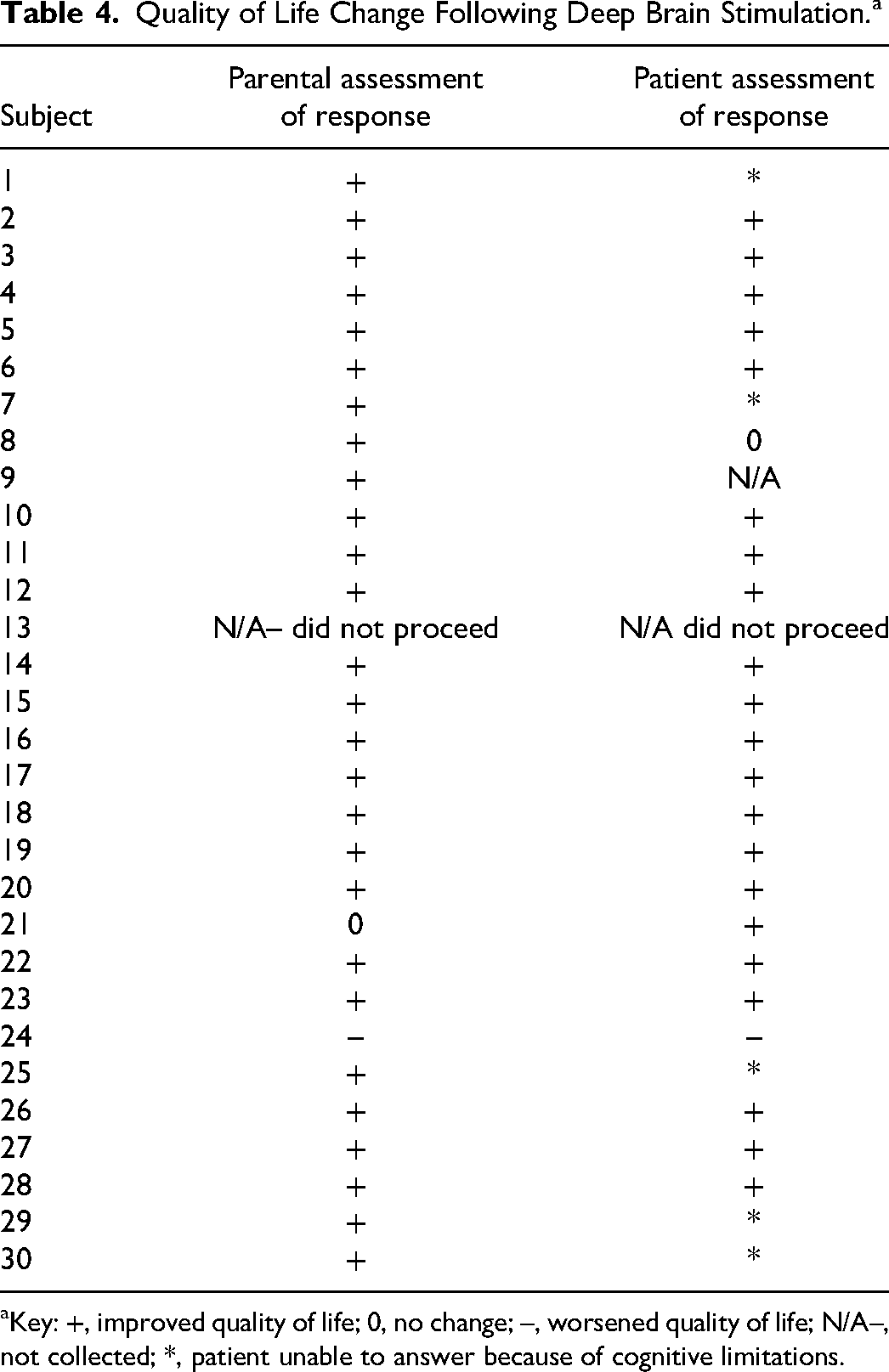
Key: +, improved quality of life; 0, no change; –, worsened quality of life; N/A–, not collected; *, patient unable to answer because of cognitive limitations.
Changes in Barry-Albright Dystonia Scale and Burke-Fahn-Marsden Dystonia Rating Scale motor scores for the 28 dystonic patients 3-6 months postimplantation were examined using mixed effects models, which were evaluated in the R environment (version 4.1.2) using the lme4 package. 30 Time (pre and post) was included as a fixed effect, whereas subject-level variability was modeled as a random effect. The parametric significance from these models is obtained in R using the Satterthwaite approximation implemented in the lmerTest package. 29 There was a statistically significant difference in both the Barry-Albright Dystonia Scale and Burke-Fahn-Marsden Dystonia Rating Scale scores. The mean reduction in Burke-Fahn-Marsden Dystonia Rating Scale was 26.4 points (95% CI: 19.5-33.2, P < .001). The mean reduction in Barry-Albright Dystonia Scale was 4.3 points (95% CI: 3.1-5.5, P < .001). For the Tourette syndrome patient, the reduction in Yale Global Tourette Syndrome Scale score was 57 points. Further details of the Tourette's case are reported elsewhere. 18
A Likert scale of positive change in quality of life, negative change in quality of life, or no change in quality of life was obtained concurrently with each subject's postsurgical motor assessment. Of the 29 subjects who proceeded with permanent lead implantation, 93% of parents saw an improvement in their child's quality of life. In addition, 91.3% of patients able to be assessed also saw an improvement in their quality of life following surgical intervention. One parent noted improved ability of their child to sit comfortably in wheelchair and use tablet, but decreased ability to stand with assistance, and hence no change in quality of life given this variable response. Both this patient and the single patient with a noted decrease in quality of life showed an objective improvement in the Burke-Fahn-Marsden Dystonia Rating Scale motor and Barry-Albright Dystonia Scale scores.
Clinically, it appears that stimulation in thalamic targets is most helpful for reduction of hyperkinetic components of dystonia, whereas stimulation in pallidal targets is most helpful for reduction of hypertonic components of dystonia. A research study to determine the statistical validity of this observation is currently ongoing.
Discussion
Successful use of deep brain stimulation neuromodulation in young patients and in rare disorders has been limited by the inability to perform individualized neurophysiological assessment for target selection in this heterogeneous population. 31 We present a population including 29 children and young adults with dystonia where use of the staged Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation process to identify optimal targets for neuromodulation resulted in consistent improvement postoperatively. The variability in targets identified during inpatient testing supports heterogeneity of the origin of symptoms in this population, and the possibility of future additional targets including anterior globus pallidus internus, substantia nigra reticulata, and the pedunculopontine nucleus. 32
The observed variability in response as measured by the Burke-Fahn-Marsden Dystonia Rating Scale and Barry-Albright Dystonia Scale scores likely reflects both symptomatic and etiologic heterogeneity, as well as limited sensitivity and reliability of these scales in adequately assessing changes in pediatric dystonia. 33 Parents consistently identify quality of life as a significant consideration for proceeding with deep brain stimulation; in this study, 96.7% of parents whose children were treated with the Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation protocol elected to proceed with placement based on the results of short-term testing. Patients demonstrated a mean improvement of 36.7% in the Burke-Fahn-Marsden Dystonia Rating Scale movement score at 3-6 months postoperatively with utilization of the staged procedure.
Some of the benefit reported here is likely due to the use of 4 wires instead of 2, as is more commonly performed. Additional benefit of 4 wires is readily apparent clinically, and none of the patients with 4 wires implanted has subsequently been reduced to stimulation of only 2 wires. Therefore, although a study of 4-electrode vs 2-electrode implantation is not currently available and may not be feasible, our results indicate that 4 electrodes provided additional benefit in all subjects compared with either pair alone, based on clinical assessment and selection of optimal stimulation patterns. This is similar to the justification for bilateral vs unilateral stimulation. In all our cases, bilateral stimulation was found to have greater effects than unilateral stimulation, which has been previously seen with utilization of deep brain stimulation in other conditions.34,35
It is also evident clinically that there are significant differences in the magnitude and effect of response based on location within pallidum and thalamus. This is particularly true for this heterogeneous population for whom prediction of targets prior to testing is limited. Further studies will be needed to delineate differences in clinical effects of the different target regions. Therefore, the benefits seen here are likely due to the full nature of the procedure, including both personalized targeting and subsequent implantation of 4 wires with two pulse generators.
The staged procedure reduces potential complications that can arise from attempting to awaken children during surgery to confirm targeting intraoperatively. 36 This allows for consideration of deep brain stimulation in pediatric patients whose movement disorders may benefit from the intervention but are not sufficiently stable for intravenous anesthesia without endotracheal intubation. There were no perioperative complications associated with use of the staged procedure. In one patient with preexisting leads, temporary lead placement resulted in postoperative transient worsening of dystonia that improved after removal of the temporary leads, with overall beneficial outcome after placement of permanent leads. Approximately one-fourth of patients experienced increased somnolence during the observation and testing period in the neuromodulation monitoring unit, sometimes requiring the use of stimulant medications (caffeine, methylphenidate, or modafinil). All somnolence resolved within days following lead removal. Otherwise, there were no adverse events attributable to the staged surgical procedure or observation and testing in the neuromodulation monitoring unit. Long-term complications associated with permanent deep brain stimulation lead implantation are congruent with previously published results, with 6.8% of patients having wound or device infection in our cohort vs 10.3% of pediatric implants and revisions performed using other procedures. 37 All cases of infection in our series were due to mechanical trauma to the wound, usually due to wheelchair straps, involuntary head movement, or direct trauma due to involuntary movement. This supports the importance of exceptionally careful wound protection and parent education postoperatively.
Limitations of our study include the following: lack of availability of specific dystonia scales applicable to the secondary dystonia population, lack of availability of specific pediatric dystonia scales, and absence of blinding due to study investigators being involved in pre- and postoperative evaluations. It should be noted that assessment by multiple clinicians with blinding to stimulation was used for rating, but knowledge of pre- vs postoperative status could not be fully avoided when scoring videos because of visible physical changes in the patients.
Another limitation was the study population's heterogeneity, which, nonetheless, aligned with our desire to use this staged procedure to tailor neuromodulation to each patient's functional needs. This heterogeneity as well as the heterogeneity of previously published populations limits the comparison of the staged vs nonstaged procedure. However, it also reflects the reality of pediatric clinical practice and constitutes one of the important strengths of this procedure. The staged testing procedure allows assessment of effects and optimal targets with “n-of-1” testing. In a heterogeneous population, this type of individualized mechanistic and outcome evidence is superior to grouped statistical evidence. Population statistical evidence will be much less predictive in individual clinical situations when there is variability in etiology, location of injury, physical ability, age, and other demographic variables with limited control. Here, we elected to document the full heterogeneity of the patients seen for this procedure in order to document the applicability and effectiveness of the procedure across different disorders. With a larger number of subjects, it will eventually become possible to document effectiveness for specific subpopulations and varied disorders in future studies.
Deep brain stimulation has been performed for Parkinson disease and essential tremor for the past 25 years with FDA approval in the United States. Early promise and anticipation of subspecialty growth in new indications for deep brain stimulation has been tempered by the limited success of rigorous multicenter randomized clinical trials using a single-target approach. New indications for deep brain stimulation will be slow in coming because of the elevated expense of such trials, promulgated in part by regulatory requirements for target-specific approval. As many patients are impacted by movement and neuropsychiatric diseases, there is a large burden absorbed by administrators of health care and their payors, which may be ameliorated by using the Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation protocol to identify optimal targets in certain segments of these populations. Many movement and neuropsychiatric diseases have variable expressivity and nuanced and complex phenotypes, and are likely influenced by multiple deep brain foci that require multiple electrodes for optimal outcomes. Intraoperative awake testing while stimulating multiple deep brain targets is impractical in pediatric populations and also in adult populations where the process may be time consuming, risky, and disturbing to patients in the operating room setting. The application of the Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation protocol for movement and neuropsychiatric disorders that are presently underserved by the neurosurgical and neurologic communities will provide new opportunities for advanced symptomatic treatment. This type of evaluation is standard practice in epilepsy, a disorder recognized to have heterogeneous neuroanatomic and neurophysiological localization. We suggest that a similar approach can be used for movement and neuropsychiatric disorders.
Conclusions
We have shown efficacy in a group of 30 children and young adults implanted with permanent deep brain stimulation electrodes following Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation test stimulation with temporary electrodes in a neuromodulation monitoring unit setting. Improved outcomes with the new Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation procedure are likely to be due not only to improved targeting, but also to eventual implantation of 4 electrodes rather than 2, which provides greater flexibility for subsequent programming and greater ability to control multiple symptoms. The ability to find optimal targets and predict the potential efficacy of deep brain stimulation neuromodulation in pediatric movement disorders is essential for the decision to proceed. For many rare diseases, there are limited or no data on optimal targets. Thus, the neuromodulation monitoring unit staged procedure potentially allows deep brain stimulation to be applied to a wider range of children when symptoms suggest potential efficacy, but there is limited or no prior experience in selection of targets or prediction of outcomes because of rarity or heterogeneity of the disorder.
The Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation procedure presents a way to test multiple targets for efficacy in an individual patient prior to consideration of permanent lead implantation. Parents have identified uncertainty as a key hindrance to decision making for proceeding with pediatric deep brain stimulation. 11 The Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation staged procedure allows both parents and patients to assess the response to stimulation prior to committing to permanent invasive neuromodulation and thus to have informed participation in decision making. 38 Stereotactic Awake Basal ganglia Electrophysiological Recording and Stimulation is a physiological assessment that can guide functional physiological interventions. It enables broad expansion of deep brain stimulation and functional neurosurgery in the same way that advanced neuroimaging has enabled broad application of structural interventions.
Footnotes
Acknowledgments
We are appreciative of the significant efforts of the inpatient nursing staff and electroencephalography technicians at both CHOC and CHLA. Our thanks to Alexandra Himy, Kimberly Meckwood, and Andrew Perdue for their assistance in logistical arrangements for the procedure. We appreciate the surgical expertise of Dr Brian Lee and Dr Jason Chu; the clinical efforts of Aprille Tongol, Diana Ferman, Dr Quyen Luc, and Dr Allison Przekop; and the efforts of Dr Jessica Vidmark, Dr Estefania Hernandez-Martin, and Ruta Deshpande in operation of the bedside recording and stimulation equipment. We are immensely grateful to our patients and their families for their participation.
Author Contributions
T.D.S. M.A.L. were responsible for the conceptualization, methodology, formal analysis; T.D.S., J.A.M., and M.A.L. contributed to writing—original draft preparation, review, and editing; J.A.M. undertook outcome measurement and project administration; J.N. performed statistical analysis, review, and editing; M.K. and J.O. were responsible for the methodology, review, and editing. All authors read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Cerebral Palsy Research Alliance Foundation, The Crowley Carter Foundation.
Ethical Approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Boards of Children's Hospital of Orange County and Children's Hospital of Los Angeles (University of Southern California Human Subjects Institutional Review Board approval UP-13-00521, November 15, 2013, to June 30, 2020; Children's Hospital of Orange County Human Subjects Institutional Review Board approval 200330, July 13, 2020, to July 24, 2022). Written informed consent was obtained from all subjects involved in the study, and HIPAA authorization for use of protected health information was obtained.
