Abstract
Background
Current criteria help differentiate cluster headache from migraine. However, children may have overlapping features making it difficult to distinguish the 2 conditions, which may delay diagnosis. Differentiating cluster headache from migraine is important regarding treatment as well as diagnostic workup of secondary headache etiologies.
Methods
Cases at a single pediatric children's hospital from 2015 to 2023 diagnosed with cluster headache before the age of 18 years were reviewed.
Results
Twenty-five cases were identified of which 22 cases met criteria for either chronic, episodic, or probable cluster headache. Three cases were diagnosed with cluster headache by their provider, but documentation was insufficient to meet criteria for cluster headache. There were 16 females and 9 males between ages 6 and 17 years. Five cases were identified as symptomatic, 2 cases as chronic, 7 cases as episodic, and 13 cases as probable cluster headache. Symptomatic etiologies include Graves disease, optic neuritis, prolactinoma, hypothalamic pilocytic astrocytoma with carotid stenosis, and congenital right eye blindness. Migrainous features were common, including 76% with nausea, 36% with vomiting, 68% with photophobia, and 56% with phonophobia. Patients with cluster headache also had an independent diagnosis of migraine in 64%.
Conclusion
Children with cluster headache have a high frequency of migrainous symptoms and co-occurrent diagnosis of migraine. A careful history may differentiate cluster headache from migraine and treated accordingly. Children with cluster headache features should undergo screening for secondary causes with appropriate imaging and other studies. Except for prolactinoma, the symptomatic associations noted in this case series have not been reported before.
Background
Cluster headache is uncommon in adults and rare in children. The prevalence of cluster headache for all ages is approximately 0.1% and 0.03% to 0.09% among those aged 18 years.1–4 Cluster headache diagnosis peaks around 20-40 years of age, with childhood onset in 5% to 27.5% of cases.3,5,6 Pediatric onset cluster headache often has a significant delay to diagnosis, ranging from 0 to 33 years, with reported means of 6.2 and 21.2 years in 2 studies.3,6 Cluster headache has a male predominance with a male-to-female ratio of 2:1 to 3:1 in adults and 1.1:1 to 2.5:1 in pediatrics.3,7–10 Cluster headache can be familial in 5% to 10% of cases.6,11,12
Cluster headache is the most common trigeminal autonomic cephalgia9,13 with criteria defined by the third edition of the International Classification of Headache Disorders (ICHD-3; Table 1). 14 Cluster headache is a severe unilateral periorbital headache that lasts from 15 to 180 minutes and may include autonomic features or restlessness. Associated autonomic features are conjunctival injection, lacrimation, nasal congestion, rhinorrhea, eyelid edema, facial sweating, miosis, or ptosis. The most common autonomic feature is lacrimation followed by conjunctival injection. 9 It can have a circadian pattern, commonly waking individuals at night, and a seasonal pattern favoring spring or fall.2,7,15 Risk factors for cluster headache include smoking, positive family history, alcohol consumption, head trauma, and childhood exposure to secondhand smoking.9,16–19
ICHD-3 Criteria for Cluster Headaches.

Abbreviation: ICHD-3, International Classification of Headache Disorders, Third Edition.
Migraine is a distinct primary headache disorder with criteria defined by ICHD-3 in Table 2. 14 Although not part of ICHD-3 criteria, cluster headache can also have frequent migrainous features.3,20,21 The presence of migrainous features makes it difficult to differentiate cluster headache from migraine, which can be important for diagnostic evaluation and appropriate treatment.8,22 This is particularly true in children, where the lower prevalence of cluster headache may cause confirmation bias favoring migraine.
ICHD-3 Criteria for Migraine Without Aura.

Abbreviation: ICHD-3, International Classification of Headache Disorders, Third Edition.
Cluster headache has been associated with symptomatic causes. Examples in the literature include aneurysms, arteriovenous malformations, cerebral venous sinus thrombosis, carotid dissections, pituitary abnormalities, masses, inflammatory lesions such as multiple sclerosis and sarcoidosis, sinusitis, Chiari malformation, idiopathic intracranial hypertension, and sleep apnea. Only 6 pediatric symptomatic cases were found in the literature: 2 children with prolactinomas, an aneurysm, a sinusitis, a cardiac stent placement, and a left nasal septum spur.9,13,23–26 The frequency of secondary causes necessitates diagnostic imaging in patients with cluster headache, which is often not true for migraineurs.
The objective of this case series is to better understand the features, diagnosis, treatment, associations, and outcomes of pediatric cluster headache and its relationship to migraine and potential symptomatic causes.
Methods
Encounters in the electronic medical record system with International Classification of Diseases, Tenth Revision (ICD-10), codes for cluster headache given before the age of 18 years, from January 2015 to April 2023, were reviewed from one institution. Each case was evaluated and categorized according to the ICHD-3 criteria for chronic, episodic, or probable cluster headache. Cases were deemed symptomatic when there was a hypothalamic, sellar, suprasellar, vascular, ophthalmic, or other associated diagnosis that had potential causation. The presence of an established migraine diagnosis, as well as migrainous symptoms of photophobia, phonophobia, nausea, and vomiting, were recorded.
Response to treatment was divided into success or failure of treatment. Successful treatment was determined by patient reporting effectiveness and continued use of the medication, although detailed information about medication response (eg, time to effect) was not consistently available. Failure was determined by patient reporting inefficacy, loss of efficacy due to acute exacerbation or self-report of not working anymore, or intolerable side effects.
The first author gathered data and made the initial determination if the headache fulfilled criteria and results were verified by the second author.
Results
Demographics and Diagnosis
Twenty-five cases were identified, and their characteristics, treatment, and outcomes are organized in Tables 3 and 4. There were 16 females and 9 males with a female-to-male ratio of 1.7:1. Age at diagnosis ranged from 6 to 17 years, with an average of 13.4 years. Twenty-two cases fulfilled criteria of chronic, episodic, or probable cluster headache. Two were chronic, 7 episodic, and 13 probable cluster headache. The remaining 3 cases with cluster headache features did not fulfill criteria for cluster headache or probable cluster headache based on review of the available data, but had been diagnosed as cluster headache by the treating physician. Five cases were considered symptomatic because of associated underlying pathology with potential for causation. Of the 5 symptomatic cluster headache cases, 4 fulfilled the probable cluster headache criteria and 1 the episodic cluster headache criteria. The episodic cluster headache symptomatic case was associated with a hypothalamic pilocytic astrocytoma. The probable cluster headache symptomatic cases were associated Graves disease, atypical optic neuritis, prolactinoma, and congenital right eye blindness ipsilateral to the attacks.
Cluster Headache Characteristics.
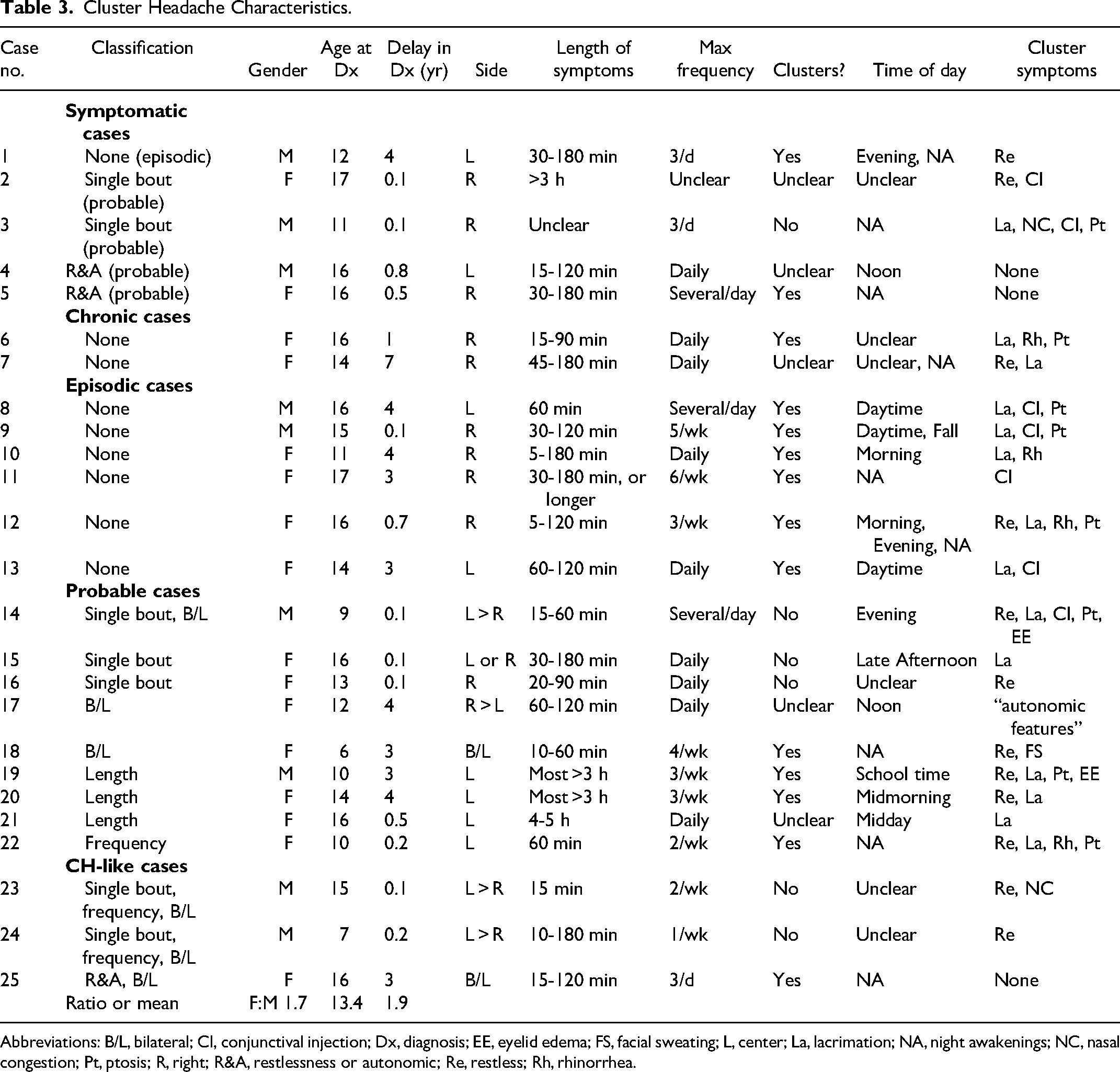
Abbreviations: B/L, bilateral; CI, conjunctival injection; Dx, diagnosis; EE, eyelid edema; FS, facial sweating; L, center; La, lacrimation; NA, night awakenings; NC, nasal congestion; Pt, ptosis; R, right; R&A, restlessness or autonomic; Re, restless; Rh, rhinorrhea.
Cluster Headache Treatment, Migrainous Features, Associations, and Outcomes.
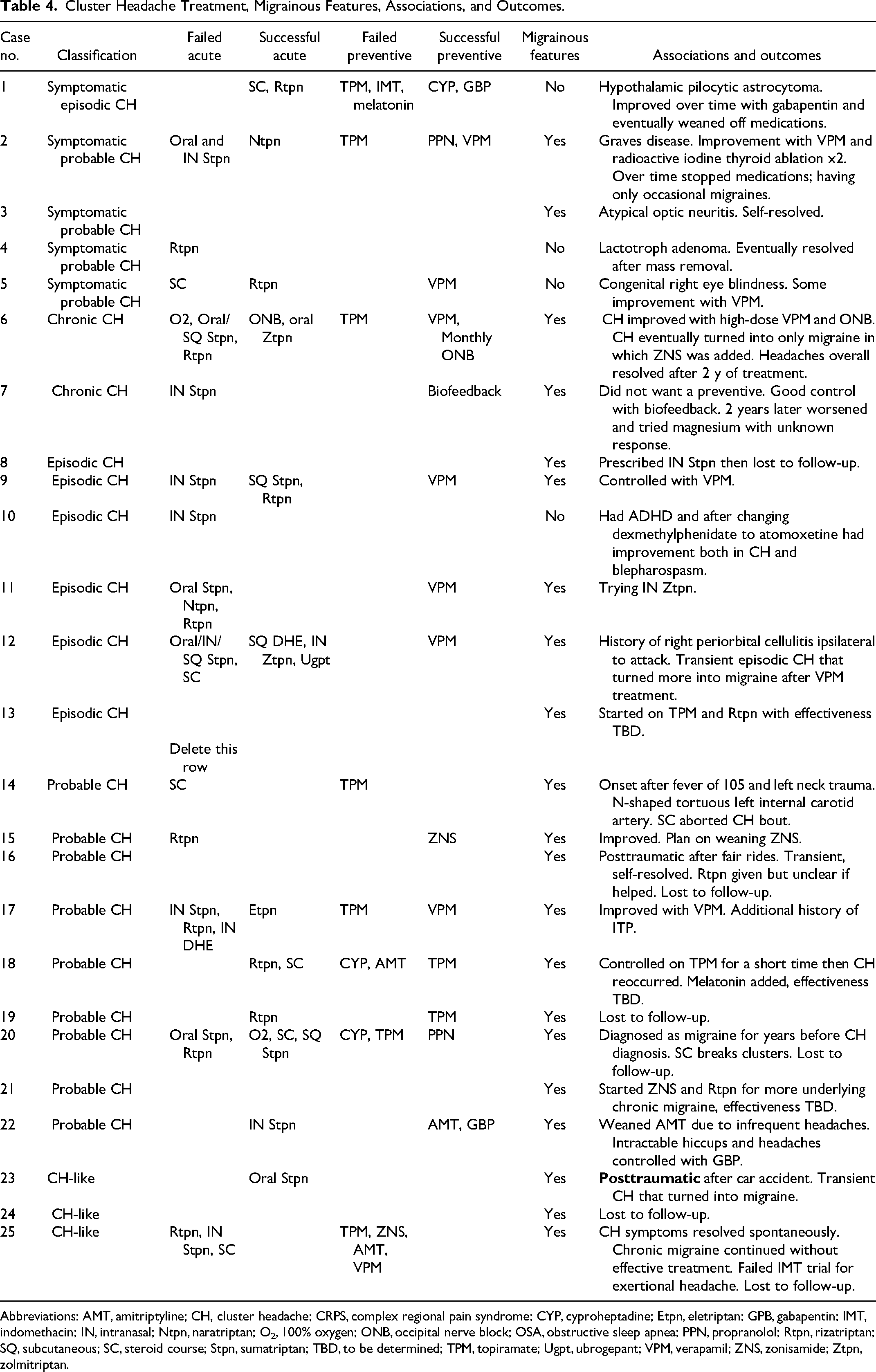
Abbreviations: AMT, amitriptyline; CH, cluster headache; CRPS, complex regional pain syndrome; CYP, cyproheptadine; Etpn, eletriptan; GPB, gabapentin; IMT, indomethacin; IN, intranasal; Ntpn, naratriptan; O2, 100% oxygen; ONB, occipital nerve block; OSA, obstructive sleep apnea; PPN, propranolol; Rtpn, rizatriptan; SQ, subcutaneous; SC, steroid course; Stpn, sumatriptan; TBD, to be determined; TPM, topiramate; Ugpt, ubrogepant; VPM, verapamil; ZNS, zonisamide; Ztpn, zolmitriptan.
Delay in diagnosis ranged from less than 1 month to 7 years with an average of 1.9 years. Prolonged delay in diagnosis >1 year was seen in 12 cases (48%), in which half saw a previous neurologist prior to cluster headache diagnosis by a headache specialist (cases 1, 6, 7, 12, 22, and 25).
Probable cluster headache was associated with a single bout in 4 cases and a single bout with bilateral headache in 1 case. The single cluster headache bout either self-resolved with no recorded reoccurrence or was diagnosed recently with the presence of recurrent bouts not yet determined by the time of this study. Other missing criteria included bilateral headache in 2 cases, no autonomic features or restlessness in 2 cases, headaches lasting greater than the 3-hour restriction in 3 cases, and not having at least every other day occurrence in 1 case. Of the 3 cases lacking criteria for cluster headache or probable cluster headache, 2 cases had a single bout with inadequate frequency and bilateral headache. One case did not fulfill criteria because of no autonomic features or restlessness and bilateral headache. Cluster headache features are delineated in Table 5.
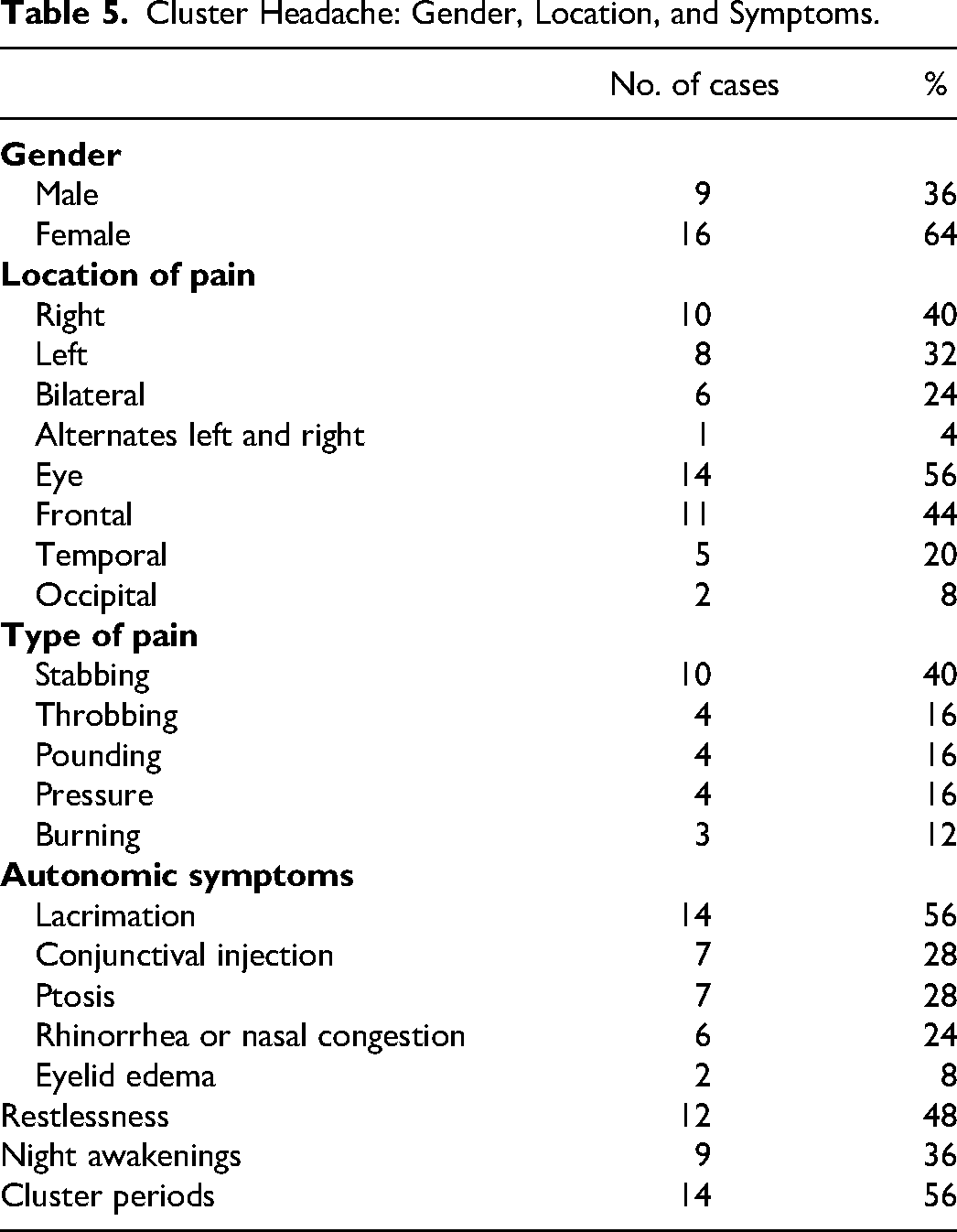
Cluster Headache: Gender, Location, and Symptoms.
Treatment
Seventeen patients (68%) received preventive treatment. The most-used and successful preventive treatment was verapamil, with 6 of 7 patients having improvement in cluster headache symptoms or frequency. Propranolol was successful in 2 patients. Gabapentin was successful in 2 patients. Biofeedback was successful in 1 patient. Seven patients received transitional treatment of oral corticosteroids, but only 4 had successful response. One patient received occipital nerve blocks that were successful. The first nerve block used a combination of bupivacaine and triamcinolone, and later she continued with monthly nerve blocks with anesthetic but without steroids as part of a preventive plan.
Acute oxygen therapy, intranasal zolmitriptan, subcutaneous dihydroergotamine, eletriptan, and ubrogepant were each only used once with successful treatment. Subcutaneous sumatriptan was used 4 times, with only 2 patients having success. Most other acute and preventive treatments had varied responses, with failure rates >50% due to either side effects or inefficacy (see Tables 4 and 6).
Cluster Headache treatments.
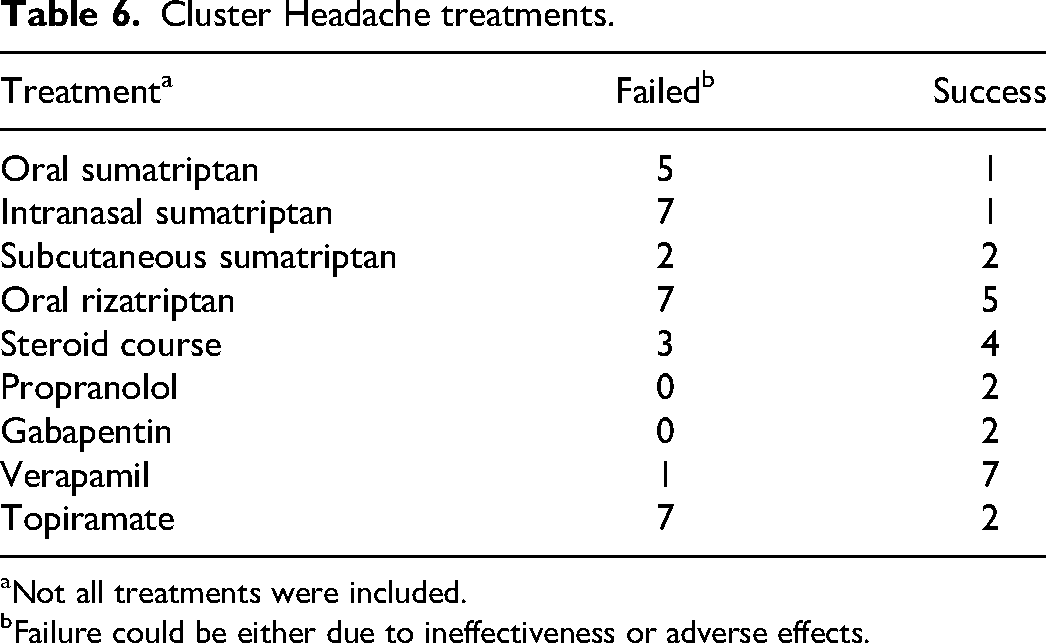
Not all treatments were included.
Failure could be either due to ineffectiveness or adverse effects.
Migrainous Features and Migraine Diagnosis
Associated migrainous features and co-occurrent diagnosis of migraine were frequent in these patients, as seen in Table 7. Migrainous features were present in 84% and included nausea (76%), photophobia (68%), phonophobia (56%), and vomiting (36%). An independent diagnosis of migraine was present in 16 cases (64%). A previous diagnosis of migraine prior to a cluster headache diagnosis was present in 9 cases (36%), and 8 of these 9 diagnoses came from a previous neurologist. Cluster headache resolution and subsequent transition into a new or recurrent migraine diagnosis appeared in 8 cases (32%).
Cluster Headache–Associated Migraine Features and Diagnoses.

Abbreviation: CH, cluster headache.
Symptomatic Cases
Case 1: Hypothalamic pilocytic astrocytoma with Carotid Stenosis
A 12-year-old boy presented with severe left frontal headaches after a recent mild concussion. Pain was described as stabbing and excruciating, often causing him to lie down and scream, or awaken at night with restlessness. They occurred up to 3 times daily with an average frequency of 5 times per week. At age 8, he was diagnosed with a 2-cm intrasellar and suprasellar hypothalamic pilocytic astrocytoma extending into the right cavernous sinus and encompassing the right internal carotid artery and abutting the left internal carotid artery. Imaging at the time of these headaches showed new severe left internal carotid artery stenosis at the cavernous and supraclinoid segments (Figure 1). He did not respond to topiramate, indomethacin, or melatonin. Cyproheptadine caused significant weight gain. Oral steroids gave only transient relief. Verapamil was not used because of concerns of intracranial hypoperfusion due to the stenosis. He ultimately responded to gabapentin, and 1 year later he weaned off medications with no recurrence of his headaches. Follow-up imaging after the headaches resolved showed mild bilateral internal carotid artery stenosis (Figure 1).
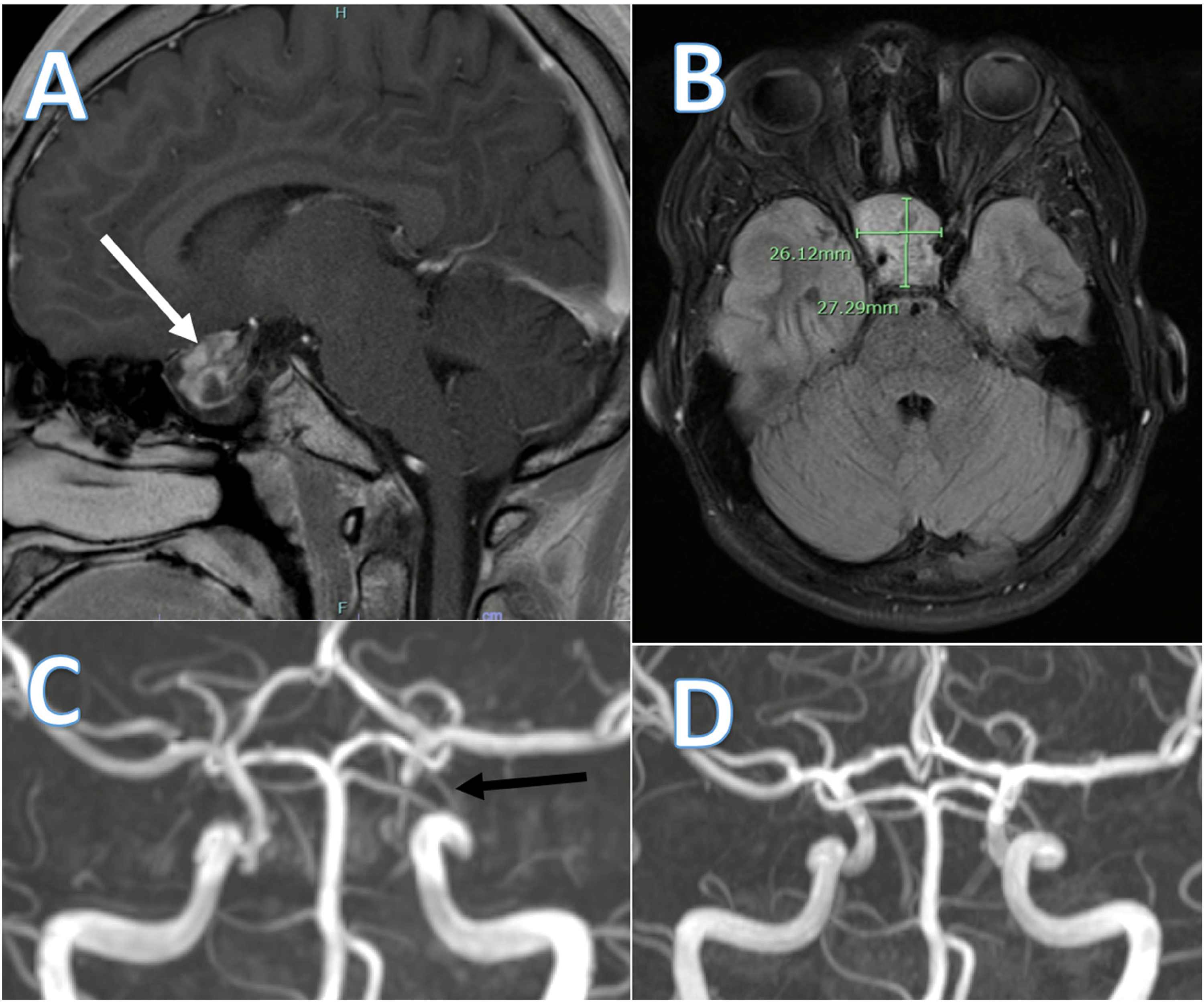
Case 1.
Case 2: Graves Disease
A 17-year-old female adolescent with history of migraine returned to the headache clinic with a new type of headache. The headaches were severe, right-sided, behind the eye, and associated with restlessness, conjunctival injection, nausea, vomiting, photophobia, and phonophobia with duration of hours to days. She started having migraine headaches at the age of 15 years for which topiramate was ineffective, but improvement was noted with propranolol 80 mg daily. After the appearance of new headache semiology consistent with cluster headache, she was started on low-dose verapamil and sumatriptan intranasal spray. Head imaging showed tonsillar and adenoid hypertrophy with correlating nasopharyngeal symptoms. She was treated for tonsilitis without symptomatic improvement. Further neck imaging showed thyromegaly and thyroiditis. Thyroid testing revealed Graves disease, and the patient was started on methimazole with propranolol 20 mg during the day. At follow-up, her headache frequency decreased with verapamil and propranolol, but intranasal sumatriptan caused vomiting. Six months later, the cluster headache resolved, but the patient continued to have migraine.
Case 3: Atypical Optic Neuritis
An 11-year-old boy with a history of left temporal lobe epilepsy controlled on oxcarbazepine presented with 1 week of new headache and 2 days of vision changes. He had a severe right frontal headache with tearing, conjunctival injection, congestion, ptosis, night awakenings, photophobia, phonophobia, and nausea. Length of headache was not documented. Frequency was 2 to 3 times per day, and they quickly resolved with ibuprofen. Five days after headache onset, he had blurry vision in his right eye with a yellow tint to his vision. His optometrist suspected optic disc edema and patient was subsequently evaluated by ophthalmology, who favored a diagnosis of retinal detachment and central serous retinopathy. Magnetic resonance imaging (MRI) of the orbits and brain showed a small right retinal detachment. Bartonella, tuberculosis, sarcoidosis, and syphilis testing were negative. He was subsequently evaluated by neuro-ophthalmology and diagnosed with optic neuritis. Headaches resolved in parallel to his optic neuritis and to date have not recurred.
Case 4: Prolactinoma
A 16-year-old boy presented with severe left frontal headaches of duration 15-120 minutes. Pain was described as stabbing or burning and associated with photophobia and phonophobia. There was no associated restlessness, autonomic symptoms, nausea, or vomiting. Frequency ranged from several per week to daily, most often at midday. After 8 months of headaches, brain MRI revealed a 2-cm sellar and suprasellar cystic mass with internal hemorrhage and compression of bilateral optic nerves and chiasm. The tumor was subsequently resected and identified as a prolactinoma. In the months following the resection, headache frequency and severity progressively decreased.
Case 5: Right Eye Congenital Blindness
A 16-year-old female adolescent with a history of congenital blindness of the right eye presented with 6 months of pain behind the right eye. Pain lasted from 30 minutes to a few hours. She would have cluster periods of multiple attacks per day for several days consecutively. In between clusters, she had no headaches. Pain was described as severe stabbing, burning, or pressure. She denied autonomic symptoms, restlessness, photophobia, phonophobia, nausea, and vomiting. Ibuprofen did not help. Oral rizatriptan showed benefit initially but eventually lost efficacy. She did not tolerate prednisone because of nausea and shakiness. She showed response to low-dose verapamil.
Discussion
This case series illustrates the many difficulties in accurately diagnosing cluster headache in children, which is important given risk of secondary headache and treatment implications. These headaches may overlap significantly with migraine and may even be co-occurring.
Symptomatic Cluster Headaches
Except for the prolactinoma, these symptomatic cases have never been reported in the literature. There are reported cases of glioblastoma multiforme and hypothalamic sarcoidosis causing cluster headache, but not hypothalamic pilocytic astrocytoma. There is a case of posterior scleritis, but not other associated eye-related diseases.25,27
The prolactinoma, hypothalamic pilocytic astrocytoma, and Graves disease may have been symptomatic due to the hypothalamic-pituitary role in cluster headache pathophysiology. Hypothalamic dysregulation is the leading theory behind cluster headache pathophysiology. Functional neuroimaging studies show ipsilateral posterior hypothalamic activation during a cluster headache. Patients with cluster headache have lower levels of testosterone, less response to thyrotropin-releasing hormone, and have decreased melatonin peak at night. 8 Graves disease ophthalmopathy of the eye muscles is another possible explanation of cluster headache–like symptoms, but signs of this were not seen on imaging.
Optic neuritis and congenital blindness may be symptomatic by causing hypothalamic and circadian dysregulation. 28 However, evidence for circadian disturbances would only be expected in absence of light perception, so unilateral visual pathway pathology may be an association rather than causation. 29 Although it is unclear if cluster headache was related to congenital blindness, the fixed location on the affected side raises the possibility.
Secondary causes have also been attributed to internal carotid artery pathology likely due to sympathetic pathway dysfunction or activation of trigeminal pain fibers in the internal carotid artery.30,31 Pathology and mass effect in the cavernous sinus where the ophthalmic branch of the trigeminal nerve passes is another potential mechanism for this case. 32 These could be potential explanations of cluster headache in the symptomatic hypothalamic pilocytic astrocytoma with carotid stenosis at cavernous segment.
Although not classified as symptomatic, there were other notable associated features with several cases. Case 12 had cluster headache ipsilateral to a prior periorbital cellulitis. Case 14 had left greater than right-sided cluster headache features after left-sided neck trauma and an N-shaped tortuous left internal carotid (Figure 2). Cases 16 and 23 were posttraumatic.
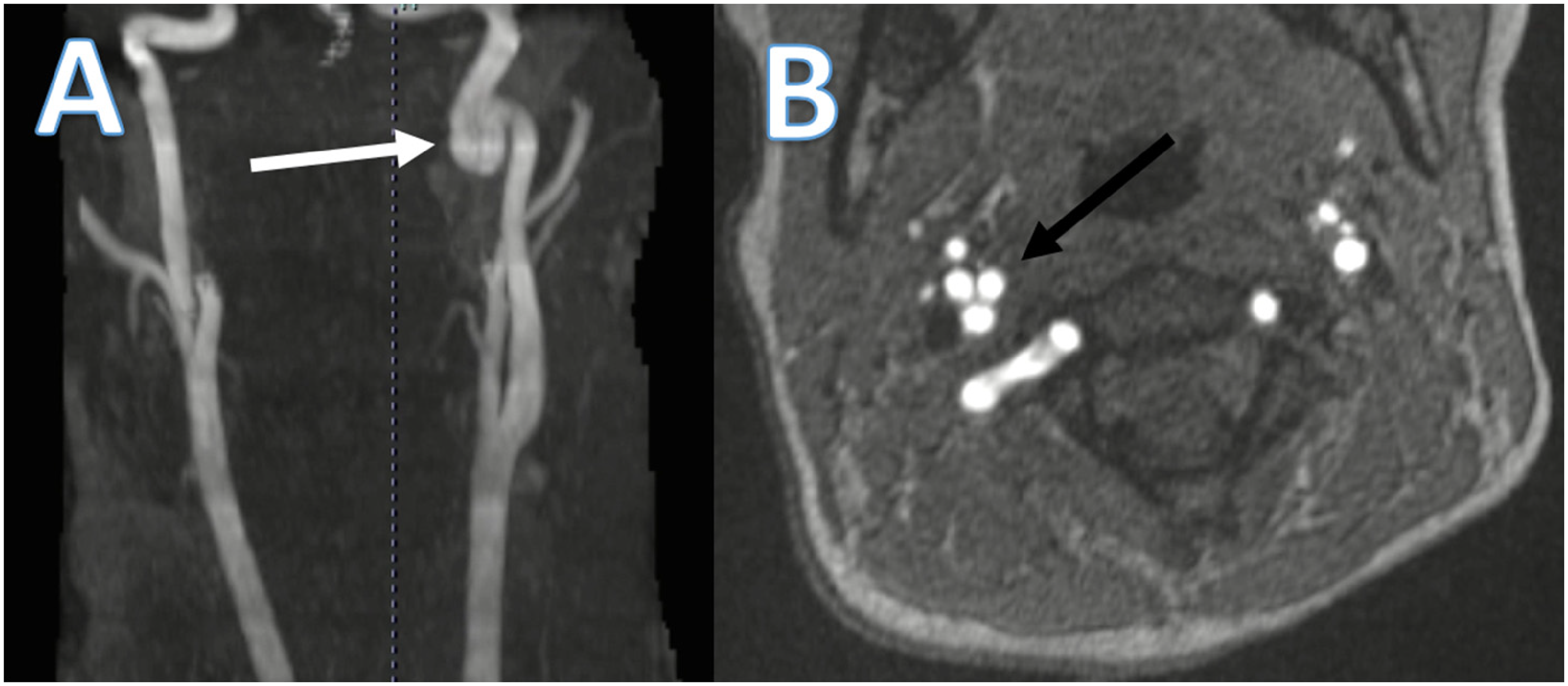
Case 14. (A) Magnetic resonance angiography showing tortuous course of the left internal carotid artery with an N-shape (white arrow). (B) Axial view of the N-shaped internal carotid artery capturing the vessel at 3 points in one plane.
Because of the possibility that cluster headache can be symptomatic, patients should have appropriate neuroimaging, with further consideration of additional testing, such as sleep study or serologic testing, when relevant history or clinical findings are present. Because pituitary tumors and carotid dissection seem to be the most common symptomatic causes in the literature, it is recommended to perform, at minimum, brain MRI and imaging of the cerebral vessels. 24
The Migraine Relationship
History has given names to the connection between migraine and cluster headache as “migraine-cluster,” “cyclical migraine,” “clustering episodes of migraine,” “cluster with aura,” “atypical cluster without autonomic symptoms or severe pain,” or “evolution of migraine to cluster.” 33 Patients can also move between migraine and cluster headache, commonly with migraine first and subsequent transition to cluster headache. 34
The crossover seen in migraine and cluster headache may be due to common pathophysiology. Central nervous system centers, such as the trigeminovascular system, trigeminocervical complex, superior salivatory nucleus, autonomic nervous system, and hypothalamus, have been implicated in the pathophysiology of both migraine and cluster headache. Both have similar pharmacologic triggers such as histamine and calcitonin gene-related peptide (CGRP). Many similar acute and preventative treatments show efficacy for both headache types. One of the main epidemiologic differences between cluster headache and migraine is gender predominance. This may indicate hormonal factors, such as low testosterone in males causing cluster headache.1,8,35 However, females are more prevalent in this retrospective case series, which, if accurate, contradicts the general trend of male predominance in this pediatric population. This could be in part due to selection bias in this retrospective study, higher rates of migrainous features seen in females with cluster headache, 21 or may be due to the more equal gender ratios in younger patients.
Cluster headache with migrainous features were more common in this study than in reported literature. Previous studies show that nausea in cluster headache can range from 13% to 52%, vomiting 19% to 26%, photophobia 34% to 63%, and phonophobia 13% to 41%. The 24% prevalence of bilateral headaches in this study is similar to a previous study showing 18%.3,6,9,15,17,36,37
Both cluster headache and migraine can have autonomic features. Bilateral autonomic features can be common in migraine. 38 Alternatively, it is uncommon for migraine to have unilateral autonomic features. Only 89 of 2862 (3%) patients with migraine in 1 study had unilateral autonomic features. 20 A stabbing headache was more likely to indicate cluster headache (38%) vs migraine (1.7%). 9
Cluster headache can have circadian patterns, with 1 case series in children noting night and morning awakenings in about 76%, 15 which is more than this case series at 36%. Migraine night and morning awakenings have also been reported, ranging from 39% to 62%.35,39 However, a recent meta-analysis clarifies and suggests that cluster headache tends to happen in the evening and night whereas migraine tends to happen more in the early morning and daytime. 40 Night awakenings in children with cluster headache can also be misdiagnosed. Case 18 in this series and 4 patients in another case series were misdiagnosed with night terrors. 41
It may be difficult for clinicians to differentiate migraine from cluster headache. A prolonged delay may be due to concordant migrainous features leading to a misdiagnosis of migraine, 22 or often the headaches not meeting full cluster headache criteria. This case series suggests that a shorter lag to cluster headache diagnosis may be achieved by being sensitive to cluster headache features even when migrainous features are predominant. Symptoms that are more suggestive of cluster headache are short duration of time, restlessness, side-locked unilateral headaches, unilateral autonomic features, evening or night headaches, or clustering.20,34 Figure 3 contains a proposed framework of thinking when differentiating and diagnosing cluster headache or migraine.
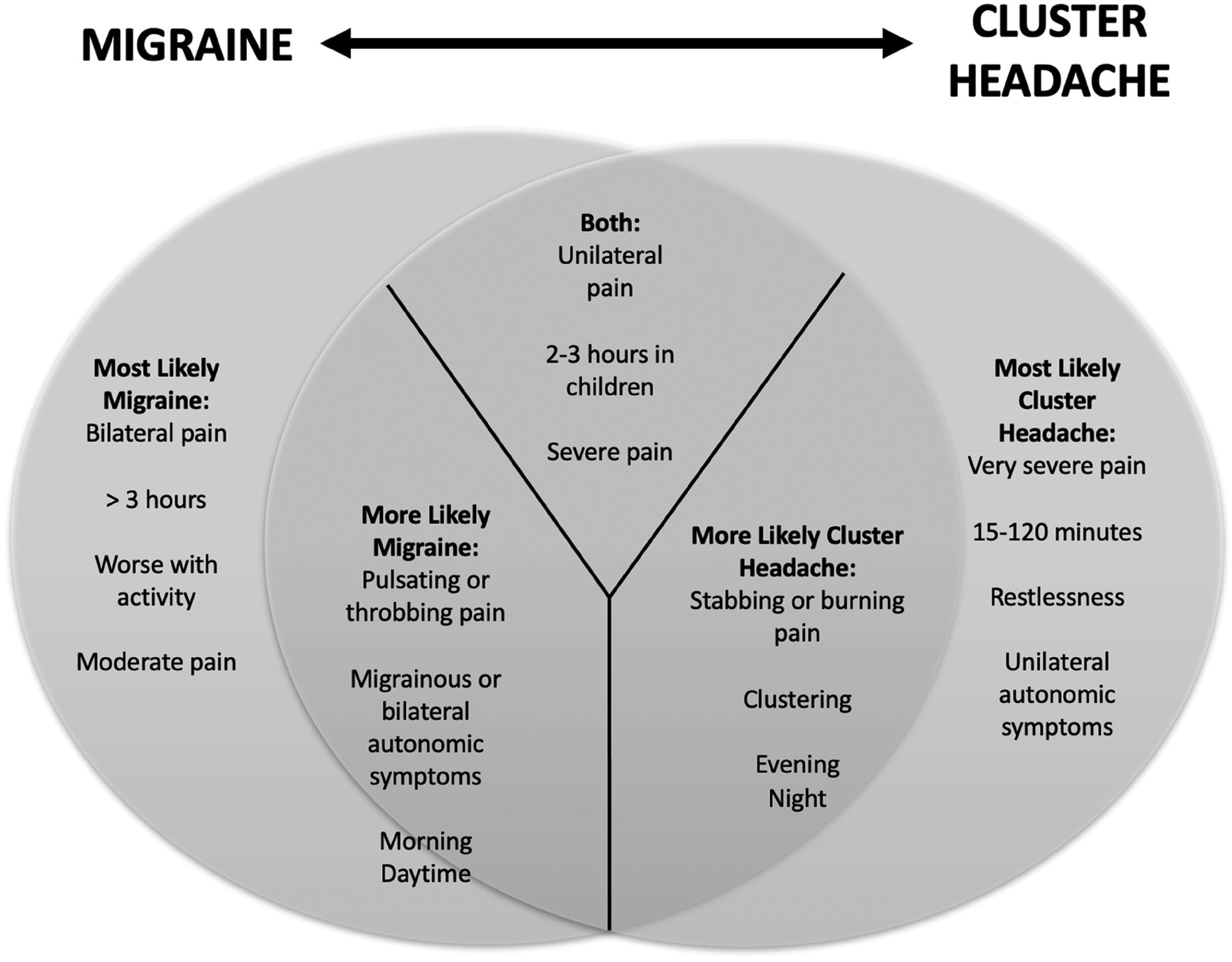
Venn diagram showing the relationship of migraine and cluster headache symptoms. Symptoms more on the left are more likely to lead to a migraine diagnosis. Symptoms on the right are more likely to lead to a cluster headache diagnosis.
Oxygen therapy has level A evidence efficacy but was limited often because of a lack of convenience and availability. Sometimes patients reported an effective response to oral triptans, which are often found to be unsatisfactory for cluster headache because of their long Tmax and slower onset of action. As this study was retrospective and uncontrolled, this may be related to placebo effect. It may also represent patient preference for oral agents instead of subcutaneous or intranasal, which children may not tolerate. This may be why intranasal zolmitriptan and subcutaneous sumatriptan were not used as much despite level A evidence. 42
In patients with features of cluster headache and migraine, a migraine preventive such as topiramate was used more often than verapamil despite the known, and reinforced here, efficacy of the latter in cluster headache. Perhaps verapamil should be considered earlier when migrainous headaches have features of cluster headache.
Limitations
Limitations of this study include missing cluster headache criteria or migrainous concordant features that may bias the diagnosis, insufficient data in the electric medical records, poor follow-up, inability to see outcomes after age 18 years, and limited headache specialists at the institution. Determining treatment effectiveness based on retrospective chart review is fraught with limitations of limited data, placebo effect, subjectivity, and small sample size.
Conclusion
This retrospective case series of pediatric cluster headache is the largest reported in the literature. Pediatric cluster headache has an evident relationship to migraine, with frequent migrainous symptoms and independent diagnosis of migraine. This may result in delay of diagnosis and ineffective treatment, particularly in children. Alternatively, it is necessary to distinguish cluster headache from migraine with autonomic features. Careful history should be obtained to differentiate cluster headache from migraine to identify the most appropriate treatment. When considering a diagnosis of a primary headache disorder, it is important to rule out secondary causes, and patients with cluster headache should have cerebral and neurovascular imaging. The novel symptomatic cases described in this report, including Graves disease, optic neuritis, hypothalamic pilocytic astrocytoma with carotid stenosis, and right eye congenital blindness, indicate the wide range of conditions that can cause or be associated with cluster headache.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
Spencer W. Eberhard: Drafted the manuscript and provided the majority of contribution to conception, design, data acquisition and anaylsis, interpretation, and revisement. Christopher T. Jackman: Advisor and edited manuscript with minor contribution to conception, design, data acquisition and anaylsis, interpretation, and revisement.
Ethical Approval
Indiana University Institutional Review Board (IRB) waived the need for ethics approval and the need to obtain consent for the collection, analysis and publication of the retrospectively obtained and anonymized data for this non-interventional study. Approval date Jan 27, 2023. Approval ID #18079.
