Abstract
Anesthetic agents are hypothesized to have different degrees of influence on the intracranial pressure. Studies have shown differences in intracranial pressure with the same medication between adults and children. The factors that determine the response to an anesthetic are largely tied to cerebral blood flow, which in turn is influenced by multiple factors, including cerebral metabolic demand, mean arterial pressure, cerebral perfusion pressure, oxygen arterial partial pressure, carbon dioxide arterial partial pressure, and intrinsic cerebral vascular autoregulation. 1 The ability of an anesthetic to alter any of these parameters plays a vital role in its ability to influence intracranial pressure.
There are a multitude of anesthetic agents that can be used to assist in the completion of invasive medical procedures, including sevoflurane, which is one of the most commonly used inhaled anesthetics in pediatric anesthesia. Sevoflurane is a highly fluorinated methyl isopropyl ether anesthetic that exhibits low blood-gas and tissue blood solubilities. 2 These physical properties make sevoflurane popular, allowing a more rapid induction of anesthesia as well as a quicker emergence.2,3 Overall, sevoflurane possesses a strong safety record, although some studies have reported an increase in intracranial pressure and cerebral perfusion pressures in children at higher minimum alveolar concentration (MAC). 4
Accurate determination of the intracranial pressure is vital when dealing with conditions related to intracranial hypertension. There does remain debate about the influence of different anesthetic agents on this pressure in children. To our knowledge, the influence of sevofurane on cerebrospinal fluid (CSF) pressure measurements during diagnostic lumbar puncture procedures in pediatrics has not been studied. The goal of our study was to determine if sevoflurane exerts any influence on cerebrospinal fluid pressure in children presenting for a diagnostic lumbar puncture.
Methods
Approval was obtained from the institutional review board prior to initiating the study. All patients scheduled for a lumbar puncture to be performed by the corresponding author in the hospital outpatient surgery center were eligible for participation. Written consent for the study was obtained by the author on the day of the procedure. When indicated, preoperative anxiolysis was achieved with oral midazolam. Once in the operating room, an intravenous catheter was placed with the aid of nitrous oxide and oxygen. Nitrous oxide was discontinued once intravenous access was obtained. Induction was completed with intravenous propofol and maintained with inhaled sevoflurane and oxygen. Airway management consisted of a laryngeal mask airway. Narcotics and antiemetics were administered at the discretion of the treating anesthesiologist, after the pressure measurements were obtained.
Because of patient safety and comfort, it was not feasible or ethical to perform the lumbar puncture before anesthetizing the patient. There were also concerns regarding withdrawing the sevoflurane before completion of the clinical procedure, so the authors opted to study the pressure at the end of the procedure (ie, the closing pressure; T0). Following the patient over time from the start of sevoflurane withdrawal allowed the patient to serve as their own control.
The lumbar puncture was obtained with an initial opening pressure measurement using a 22-gauge 2.5- or 3.5-in. needle with the legs and neck extended. The procedure was completed with a closing pressure measurement immediately before the time sevoflurane was discontinued and represents time zero (T0). Temperature, pulse, respiratory rate, blood pressure, oxygen concentration, end tidal carbon dioxide and end tidal sevoflurane concentration were recorded at T0. Cerebrospinal fluid pressure, pulse, respiratory rate, oxygen concentration, end tidal CO2, and end tidal sevoflurane concentration measurements were then obtained at 2-minute intervals for a duration of 10 minutes (T1, T2, T3, T4, and T5). If the patient showed signs of arousal as evidenced by elevations in heart rate, respiratory rate, or movement, or if the end tidal sevoflurane concentration dropped below 0.65% end tidal sevoflurane, the study procedure was discontinued.
Because of obtaining multiple measurements for each subject, repeated measures analysis of variance models were used to analyze the collected data. A P value of <.05 was considered significant.
Results
A total of 21 patients were enrolled, and 3 patients had repeated lumbar punctures during the study period for a total of 24 encounters. Because a repeated measures analysis of variance model uses only different or independent subjects, duplicated subjects in the data set were excluded for this analysis. Of the 3 subjects who had multiple lumbar punctures, 2 had one of their observation periods shortened because of movement or tachycardia, and these encounters were excluded. For the third patient, only measurements from the first encounter were used. Three early patients did not have end tidal sevoflurane concentration measurements recorded. Indications for lumbar puncture included concerns associated with headache for 11 patients, optic nerve edema and headache in 3 patients, optic nerve edema only in 2 patients, primary (idiopathic) intracranial hypertension in 2 patients, and 1 patient each with diagnosis of optic neuritis, cranial nerve VI palsy, and monoparesis. The 3 patients with repeat encounters had individual diagnoses of primary (idiopathic) intracranial hypertension, headache, and cranial nerve palsy.
The mean age in the study population was 11.05 years (range 2.42-16.75 years), 50% (12/24) were female, mean body mass index percentile was 66.44 (range 0.78-99.9) (see Table 1). An average of 8.8 mL of cerebrospinal fluid was removed before the start of the study period (SD = 2.8). The average change between opening and closing (ie, T0) pressures was 4.6 cm H2O (SD = 3.7). The mean patient temperature was 35.4 °C (range 32.8-36.4), and end tidal CO2 mean was 45.6 (range 36-59). End tidal CO2, respiratory rate, and oxygen concentration remained stable during the course of observation. The pulse remained stable in all but 2 encounters, which were both stopped because of a >20 beats/minute increase, suggesting patient awareness. Two observation periods were stopped early because of movement of an extremity by the participant and 3 because of the end tidal sevoflurane concentration of the participant dropping below the cutoff threshold.
Patient Demographics

Each age range included 1 repeat lumbar puncture.
End Tidal Sevoflurane Concentration and Cerebrospinal Fluid Pressure
End tidal sevoflurane concentration decreased from T0 to T5, whereas mean pressure measurements showed an initial increase between T0 and T2, before decreasing (see Figure 1). Seventeen patients (81.0%) had an increase in pressure from T0 to T1, and 15 (71.4%) had an increase at T2 compared with T0. The average pressure reached the maximal increase by T2. The Spearman correlation coefficient between sevoflurane level and cerebrospinal fluid pressure was nonsignificant (P = .133).
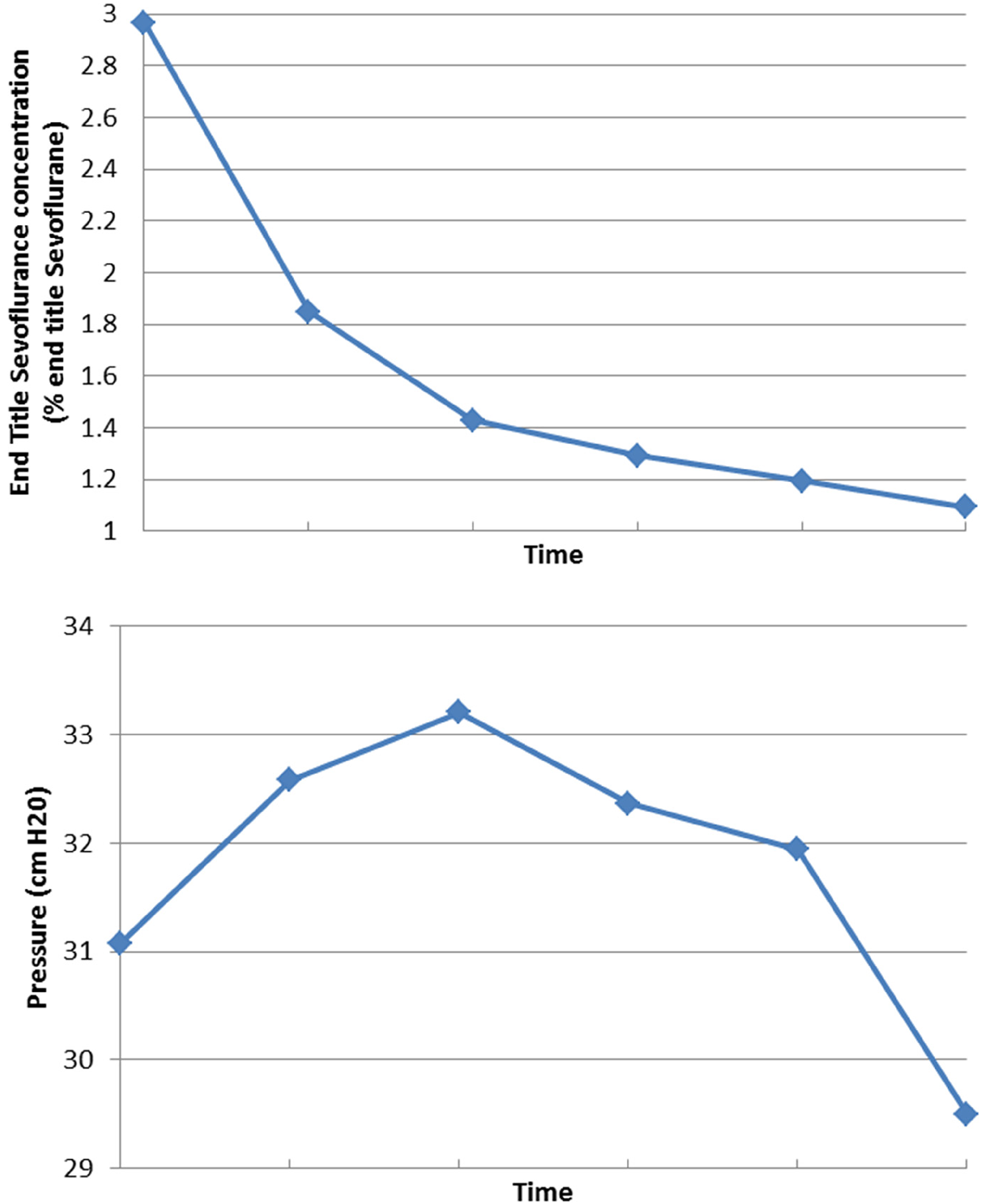
Sevoflurane concentration and cerebrospinal fluid pressure over time.
A repeated measures analysis of variance model confirmed that the mean pressure changed significantly over time (P = .001). To further determine the pattern of pressure change, pairwise comparisons were performed using Tukey's method. Mean cerebrospinal fluid pressure at T1 was significantly higher than that at T0 (P = .002). Mean cerebrospinal fluid pressure did not significantly change from T1 to T2 (P ≥ .999), or from T2 to T3 (P = .190). However, mean cerebrospinal fluid pressure significantly decreased from both T3 to T4 (P = .043), and T4 to T5 (P = .013). The pressure at T5 was not significantly different from that at T0 (P = .850). These results suggest a pattern of dynamics of the pressure where the increase after T0 and gradual decrease to T5 results in a negligible difference between times T0 and T5.
End Tidal CO2 and Cerebrospinal Fluid Pressure
The end tidal CO2 level consistently decreased from T0 to T5 despite the pressure increase over T0 to T2 followed by the decrease to T5 (see Figure 2). For the repeated measures analysis of variance model, pressure was used as a dependent variable and CO2 was used as the independent variable. Modeling showed that CO2 does not have significant effect on the dynamics of the cerebrospinal fluid pressure (P = .158). Spearman correlation coefficient between CO2 and pressure level was nonsignificant (P = .103).
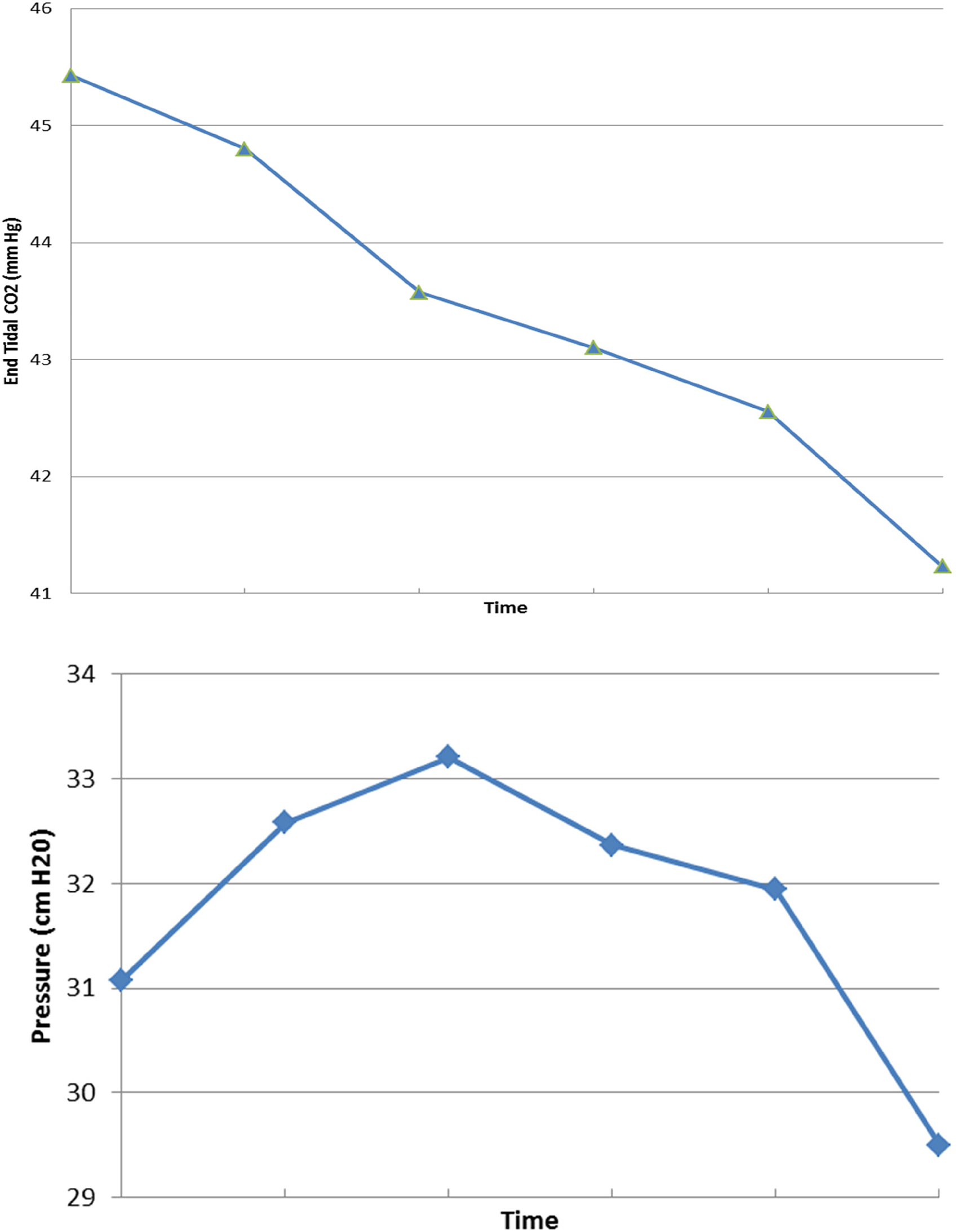
End tidal CO2 and cerebrospinal fluid pressure over time.
Spontaneous Respirations and Cerebrospinal Fluid Pressure
Seven patients required assisted ventilation at T0 due to apnea (see Figure 3). All but 1 patient had regained spontaneous respirations by T1 or T2, with the remaining patient regaining spontaneous respirations after completion of the observation period. The pressure of those requiring assisted ventilation (SR–) at T0 was consistently higher than that of those with spontaneous respirations (SR+) at T0. The repeated measures analysis of variance model suggested that ventilation status had a significant impact on pressure changes over time (P = .036).
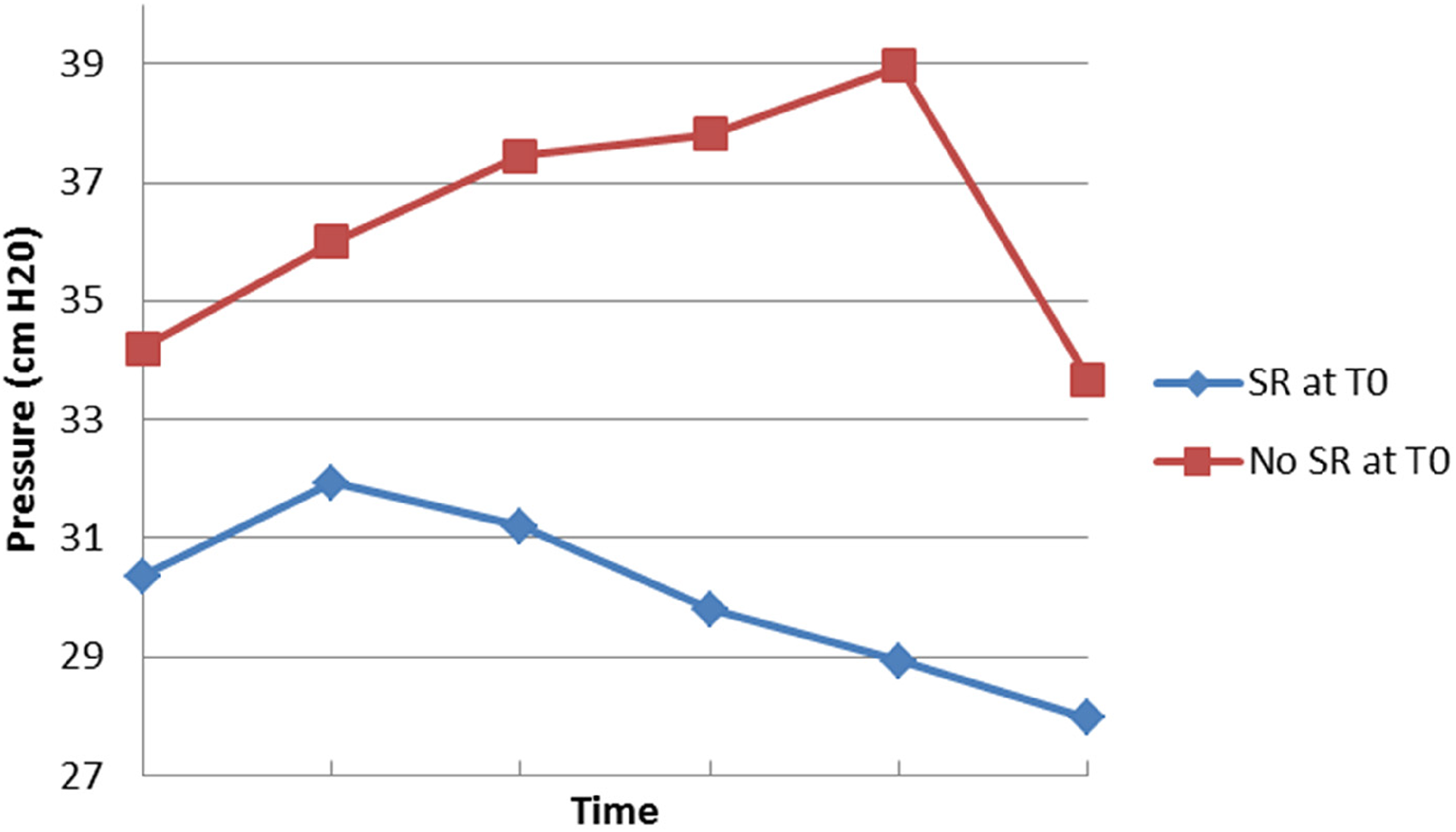
Spontaneous respirations vs pressure. Abbreviation: SR, spontaneous respirations.
Further comparisons of pressure between the 2 groups were done at each time point using P values adjusted by the Bonferroni-Holm method. The pressure in the SR– group was significantly higher than that in the SR+ group at both T4 (P = .032) and T5 (P = .039). Comparisons of pressure at time points T0 through T3 did not show a statistical difference between groups with P values of .068 for each time point.
Body Mass Index and Cerebrospinal Fluid Pressure
Nine patients had a body mass index percentile >85%ile and were classified as overweight or obese. The change in pressure over time for overweight vs normal-weight patients was not statistically significant (P > .05), suggesting that weight status did not have an effect on changes in cerebrospinal fluid pressure.
Optic Edema and Cerebrospinal Fluid Pressure
Five patients had presence of optic edema as an indication for their lumbar puncture. The change in pressure over time for patients with optic edema (ONE+) was compared to those without edema (ONE–). The mean cerebrospinal fluid pressure in the ONE+ group was higher than that of the ONE– group throughout the observation period with the exception of T5 (see Figure 4). The repeated measures analysis of variance model suggested that the difference in pressure between ONE+ and ONE− groups is consistent over time (P = .112). That is, the change in pressure was not significantly different for those with or without edema.
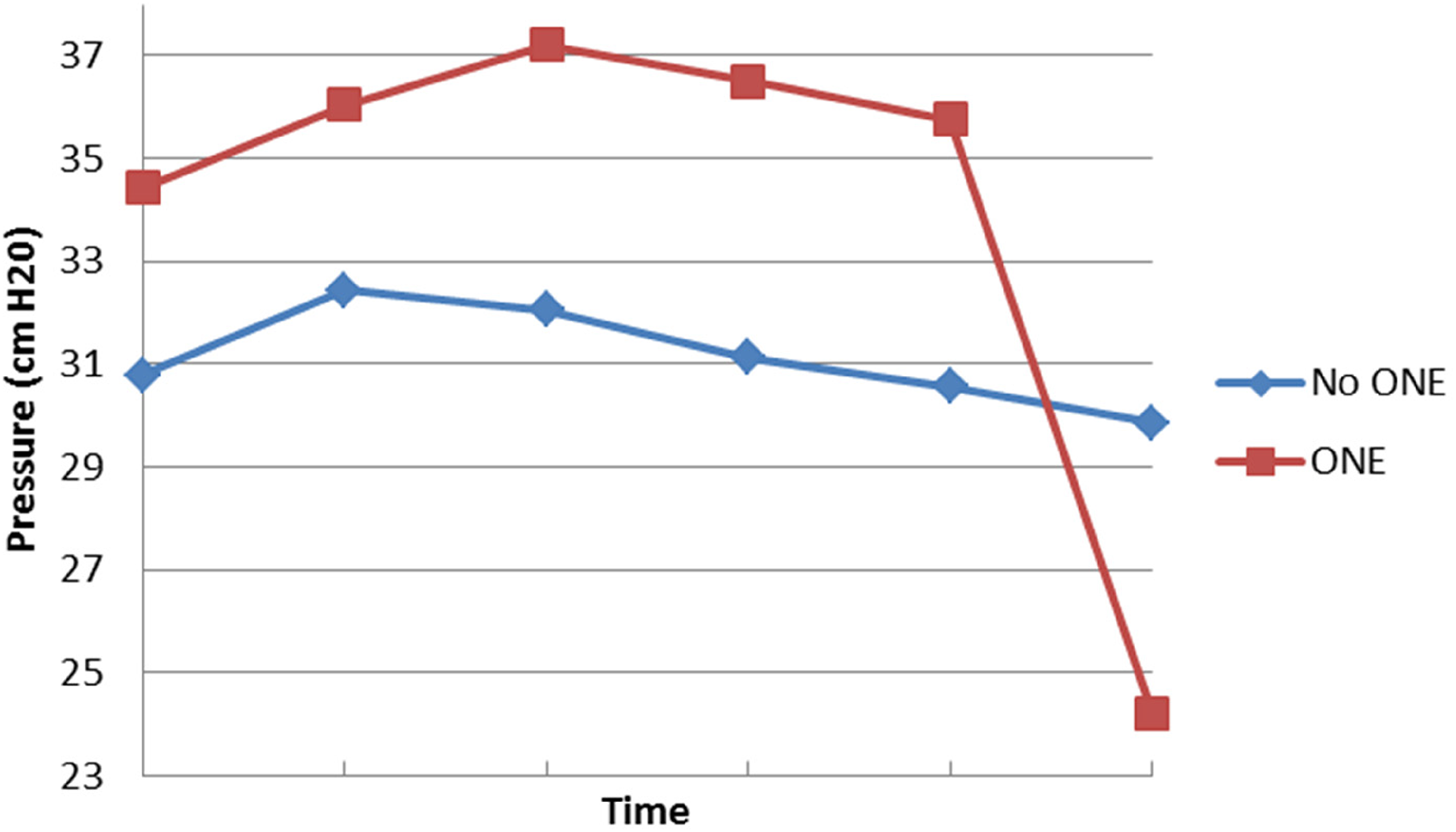
Optic nerve edema vs pressure. Abbreviation: ONE, optic nerve edema.
Age and Cerebrospinal Fluid Pressure
We divided the study group into 3 different age groups—group 1 (less than 8 years of age), group 2 (8-13 years), and group 3 (14-18 years)—to mimic prepubertal, pubertal, and postpubertal ages. The pattern of dynamics of the pressure appeared similar in each age group (see Figure 5). The repeated measures analysis of variance model suggested that, statistically, the difference in pressure between groups was consistent over time (P = .3099). In addition, the model suggests that the difference in pressure between age groups was not statistically significant (P = .131).
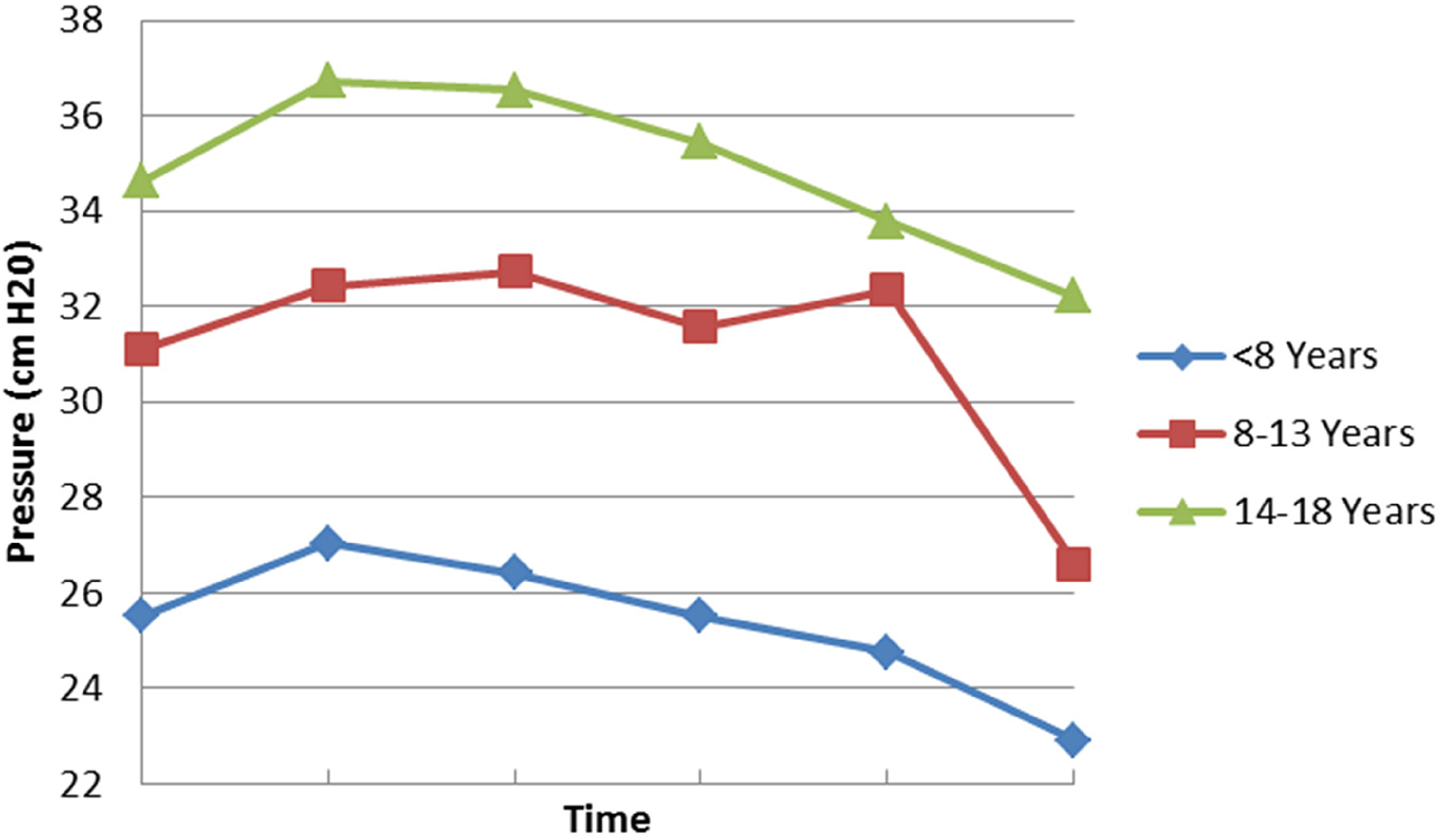
Age vs pressure.
Discussion
Performing a lumbar puncture for opening pressure is a diagnostic consideration in the workup and management of pediatric headache. The lumbar puncture is often at the forefront in the presence of optic edema; however, up to 17.8% of patients lack optic edema in the setting of other symptoms consistent with intracranial hypertension.5‐7 Propofol is a known vasoconstrictor, yet it preserves cerebral autoregulation, and it has been found to lower the intracranial pressure compared to inhaled anesthetic agents in adult patients. 8 Another study in children found that propofol resulted in elevated pressures as the patient woke from sedation. 9 Nitrous oxide has been shown to increase cerebral blood flow volume, and halogenated inhaled anesthetic agents increase cerebral blood flow to varying degrees.10,11 Similar to propofol, sevoflurane has been shown to preserve cerebral autoregulation and thus cerebral blood flow in both adults and children.12‐14
With concerns around patient safety and comfort in addition to issues with withdrawing the sevoflurane prior to completion of the clinical procedure, we opted to study the changes as the inhaled agent left the patient. Although practitioners are typically more concerned about the opening pressure, the changes noted as sevoflurane was metabolized in patients can be extrapolated back to its influence on the opening pressure. As would be expected, the end tidal sevoflurane concentration decreased over time once discontinued. However, we did not expect there to be an initial increase in cerebrospinal fluid pressure. The changes in mean pressure over time were only found to be statistically significant between T0-T1 (increase), T3-T4 (decrease), and T4-T5 (decrease), though the change at the final endpoint (T5) compared to T0 was not statistically significant. There was no significant correlation between the end tidal sevoflurane concentration and the pressure, which is different than reported by Sponheim et al 4 and Talke et al. 15 Specifically, Sponheim et al4 studied a group of children undergoing intraparenchymal pressure device implantation for pressure monitoring. The authors directly measured intracranial pressure via the implanted transducer and noted increases in heart rate and intracranial pressure with decreases in mean arterial pressure and cerebral perfusion pressure with a change from 0.5 to 1.0 MAC of sevoflurane. 4 The mean increase in intracranial pressure was 5 mm Hg (6.8 cm H2O). Similarly, Talke et al. monitored cerebral perfusion pressure and lumbar cerebrospinal fluid pressure in children undergoing transsphenoidal hypophysectomy. 15 They noted a statistically significant (though clinically insignificant) 2 mm Hg (2.7 cm H2O) rise in lumbar cerebrospinal fluid pressure with concurrent decrease in cerebral perfusion pressure with increasing MAC of sevoflurane. Because our study was done as sevoflurane concentrations decreased in the patient, Sponheim's4 and Talke's 15 results would predict that the intracranial pressure would have decreased as the end tidal sevoflurane concentration decreased. However, we found a small initial increase in the pressure with the initial decrease in end tidal sevoflurane concentration but the final endpoint was not statistically different from the initial time point. There were no changes in any of the other measured variables such as heart rate, or respirations during this period. It is possible this change represented an initial washout effect resulting in transient changes in cerebral vascular tone as it did correlate with the period of largest change in the end tidal sevoflurane concentration. We noted poor correlation of cerebrospinal fluid pressure with decreasing end tidal CO2 measurements during the study period. Sevoflurane retains the cerebrovascular response to CO2 levels as well as autoregulation. 2 Thus, it is possible that the change in end tidal CO2 (4.1 mm Hg) was not enough to result in cerebral vasoconstriction to impact overall cerebrospinal fluid pressure. The influence of patient age and the presence of optic nerve edema when individually compared with the sevoflurane concentration did not produce a clinically significant difference in pressure. Patient body mass index also did not have a statistical impact on cerebrospinal fluid pressure.
In our cohort, the lack of spontaneous respirations did produce a clinically significant increase in the intracranial pressure. As shown in our other analyses, this is not a direct result of the sevoflurane on the pressure but rather the sevoflurane resulting in respiratory depression and subsequent hypercarbia. Hypercarbia has been proven to increase intracranial pressure in both human and animal models.16‐18 The cause is likely due to the loss of cerebral autoregulation. 16 This may explain the slightly higher opening pressure reported by Avery et al 19 in those under moderate to deep sedation, although they did not specify anesthetic agents used or presence of spontaneous respirations.
Conclusion
Our study shows that sevoflurane itself does not influence the intracranial pressure when used as the primary anesthetic agent. However it does support that when measuring the intracranial pressure, the most accurate measurement is with the patient maintaining spontaneous respirations.
Footnotes
Acknowledgements
Thank-you to the anesthesia department and staff for assistance in completing this study.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. S. Aylward receives salary support from Biomarin pertaining to Batten Disease research, Biohaven Pharmaceuticals Holding Company Limited for a study on Rimegepant, and NeuroNEXT/NINDS a fellowship grant award. He also receives an honorarium as associate editor of Pediatric Neurology.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
