Abstract
Participants were enrolled into a pilot randomized-controlled 4-week trial comparing the efficacy and feasibility of app-based cognitive behavioral therapy (CBT) to a stretching program. Headache-related disability and quality of life were assessed using the Pediatric Migraine Disability Scale (PedMIDAS), Kidscree27, and Pediatric Quality of Life Inventory. Multivariable regression analysis were performed to assess the group effects in the presence of adherence and other covariates. Twenty participants completed the study. Adherence was significantly higher in the stretching than in the CBT app group (100% vs 54%, P < .034). When controlling for adherence and baseline scores, the stretching group showed greater reduction in PedMIDAS score (average: 29.2, P < .05) as compared to the CBT app group. However, in terms of the Quality-of-Life Indicators, pre- and postintervention raw scores were not significantly different between groups (P > .05). App-based CBT was not superior to a stretching program in reducing headache-related disability in a select population of pediatric headache patients. Future studies should assess if implementing features to the CBT app, like tailoring to pediatric age groups, would improve outcomes.
Headaches are one of the most frequently reported health concerns among children,1,2 with 5% to 15% of youth suffering from migraine and 15% to 25% from tension-type headache.3–5 Although the prevalence of frequent or severe headaches has been largely constant over the years, headache-related disability, particularly school absenteeism,6,7 has increased. Children with headaches also suffer from poor sleep quality, 8 increased rates of anxiety and depression, 9 and overall lower quality of life.10,11
Pharmaceutical approaches for the prophylactic treatment of pediatric migraine and tension-type headache have traditionally been based on agents that are not headache-specific and have low efficacy in the pediatric age group, including amitriptyline, 12 propranolol, and topiramate. 13 More recent pharmacologic approaches such as CGRP inhibitors have not yet been approved in pediatric age groups. Nonpharmacologic therapies have shown similar effects to drug therapies for headache prevention; however, they are potentially more accessible and at times less expensive. 14 Success of migraine therapy is influenced by a number of factors including anxiety surrounding headache events, and psychological comorbidities.15,16 Factors contributing to the efficacy of nonpharmacologic interventions include patient preference, poor tolerance or negative side effects from medications, inability to cope with stress or pain, and comorbid psychological conditions. 17
Increased behavioral and emotional symptoms have been observed in headache patients, particularly related to somatic, anxiety-depressive, social, attention, and internalizing symptoms. 9 Cognitive behavioral therapy (CBT) in pain management, including headaches, has been successfully introduced in recent years. CBT targets 3 domains involved in triggering or worsening headaches: behaviors, emotions, and cognitions.18,19 CBT aims at altering patients’ thoughts, interpretations, and assumptions surrounding stressors, ultimately making them more aware of the relationships between stress, coping, and headaches.18,19 The ultimate goals of CBT for headache patients are to reduce headache frequency and severity, increase the control over headaches, reduce the disability associated with headaches, and limit reliance on medications. 20
CBT may thus provide an effective treatment alternative to pharmacotherapy for the reduction of pediatric headache related disability and has been reported to improve quality of life in children and adolescents.21,22 Health-related quality of life has become an important outcome measure for treatment studies in different diseases and has increasingly been used as a secondary outcome measure in headache trials. Quality of life in children with headaches has recently been shown to be decreased on all but 1 subscale (self-esteem) compared with a cohort of healthy children. 23 According to international headache treatment guidelines, improvement of health-related quality of life is to be among the main objectives of therapy and has been included in the guidelines pertaining to controlled trials of drugs in migraine. 24 We have included quality of life in our secondary outcome measures.
Traditional office-based CBT is cost- and resource-intensive. Recently, smartphone-based CBT apps have been developed as a more cost-effective way of distributing this treatment. Preliminary results in adults demonstrate a reduction in headache days following the use of CBT apps. 25 Similar studies using app-based platforms are not yet available for children. CBT apps are freely available and could become a practical and easily accessible treatment option for children with headaches. We therefore assessed the feasibility and effectiveness of an app-based CBT program in children with headaches.
Methods
Objectives
The objectives of this pilot study were to (1) assess the feasibility of an app-based CBT program for the management of pediatric headaches; (2) to compare headache-related disability between children randomized to app-based CBT and stretching, using the Pediatric Migraine Disability Assessment Scale (PedMIDAS); (3) to explore the influence of app-based CBT on quality of life in children with headaches; and (4) to measure the effect of app-based CBT on headache frequency, intensity, and duration.
Study Design, Recruitment, and Participants
We conducted a 4-week pilot randomized controlled trial with approval from the research ethics board of the Children's Hospital of Eastern Ontario.
Children were recruited from the Children's Hospital of Eastern Ontario (CHEO) Neurology Clinic between October 2020 and November 2021. Children were eligible for participation if they were between 8 and 17 years old and diagnosed with migraine or tension-type headaches by a Pediatric Neurologist according to the International Headache Society Criteria. 26 Participants had to be fluent in English (verbal and written) with a reading ability at or above grade 3 level, have access to a mobile device, have no prior or current engagement in CBT, and an initial PedMIDAS score of greater than 10 points (indicating at least minimal disability). 27 Eligible children were approached by their neurologist during a regular scheduled appointment and, if interested in our study, consented by research staff. Written consent or assent was obtained from all participants, and written consent from parents of assenting participants. Participant allocation to one of the 2 study groups was determined by computer-generated permuted block randomization, with random block sizes of 2, 4, and 6.
Interventions
Both groups
At baseline, participants completed the PedMIDAS 27 (modified to 1 month), Kidscreen-27, and Pediatric Quality of Life Index (PedsQL). All participants attended a virtual meeting with the research coordinator to discuss the questionnaires, randomly assign the participant to either stretching or app-based CBT, and instruct them how to access and use the necessary apps and/or stretching booklet.
Stretching group
At the start of the intervention period, the stretching group was instructed to follow the stretching instructional booklet and perform the stretches 10 minutes daily for 4 weeks. The stretching booklet was repurposed—with permission—from the University of Buffalo Concussion Management Clinic, where it was used in a study on the effects of aerobic exercise on sport-related concussion in children. 28
CBT app group
At the start of the intervention period, the CBT group was instructed to use the app BoosterBuddy (Island Health) for 10 minutes daily for 4 weeks. Selection of the application was completed with the following considerations: Cost and accessibility (this app is free and widely available for both iOS and Android); age appropriateness (this app was geared toward a younger age group with adequate instructions and a broad range of activities available); and practicability for daily use (this app contained a broad range of activities that would be feasible within 10 minutes daily).
Data Collection
Demographic information was collected including patient age, sex, gender, school grade, and headache diagnosis.
Questionnaires
Questionnaire administration and data collection was performed with the use of RedCap, 29 an online survey tool. Participants and/or their parents/caregivers were emailed a series of links to access and complete the questionnaires through RedCap on their personal device. Three questionnaires were completed at baseline (0 weeks) and exit (4 weeks) of the study, PedMIDAS (modified for 1 month), KidScreen-27, and PedsQL. A feedback survey was administered after completion of all other study components.
Headache data
Participants in both groups used Migraine Buddy (Healint), an app-based headache diary, to document headache frequency, duration, intensity, additional symptoms, affected activities, potential triggers, and medications daily over the course of the intervention period (0-4 weeks). Participants were instructed to continue with their usual abortive and prophylactic headache medication regimens as instructed by their treating neurologist.
Outcomes
The primary outcome was the PedMIDAS score at exit compared to baseline. Secondary outcomes included Kidscreen-27 and PedsQL scores, headache frequency, severity (scale from 0 to 10), and duration.
Statistical Analyses
Quantitative variables were summarized as mean (SD) and median (minimum, maximum). A Wilcoxon (independent) test was used for univariate comparison between the CBT application and stretching groups for the continuous measurements. The adherence variable (proportion of days adhering to the study) was dichotomized into a binary categorical variable with 2 levels (eg, high adherence with attendance of ≥70% of the sessions; low adherence with attendance of <70% of the sessions). To verify that adherence was consistent between the CBT application and stretching groups, a Fisher exact test was used. The primary analysis tested for a difference in postintervention PedMIDAS scores between control (stretching) and CBT groups, when adjusting for baseline PedMIDAS and adherence (using an analysis of covariance [ANCOVA] and multivariate regression analysis). The R software 30 (version 4.2.2) was used to perform all of the statistical analysis.
Results
Recruitment and Participant Demographics
A total of 113 patients from the Neurology Clinic at Children's Hospital of Eastern Ontario were approached for the study; 28 were recruited and 20 completed the study (Figure 1). Of those approached but not recruited to the study, 25 youth stated that they were no longer interested, 24 pursued other research projects being conducted at our center, and 33 were unable to be reached in follow-up. Of those recruited, 4 were no longer eligible and 3 left the study early stating they were no longer interested or unable to meet the time commitment any longer. Demographic characteristics are summarized in Table 1. Participants were on average 13.7 ± 1.6 years (78% female) in the stretching group and 13.6 ± 2.7 years (82% female) in the CBT app group (Table 1). Overall, there were more participants with a diagnosis of migraine (n = 14) than tension-type (n = 6) headaches.
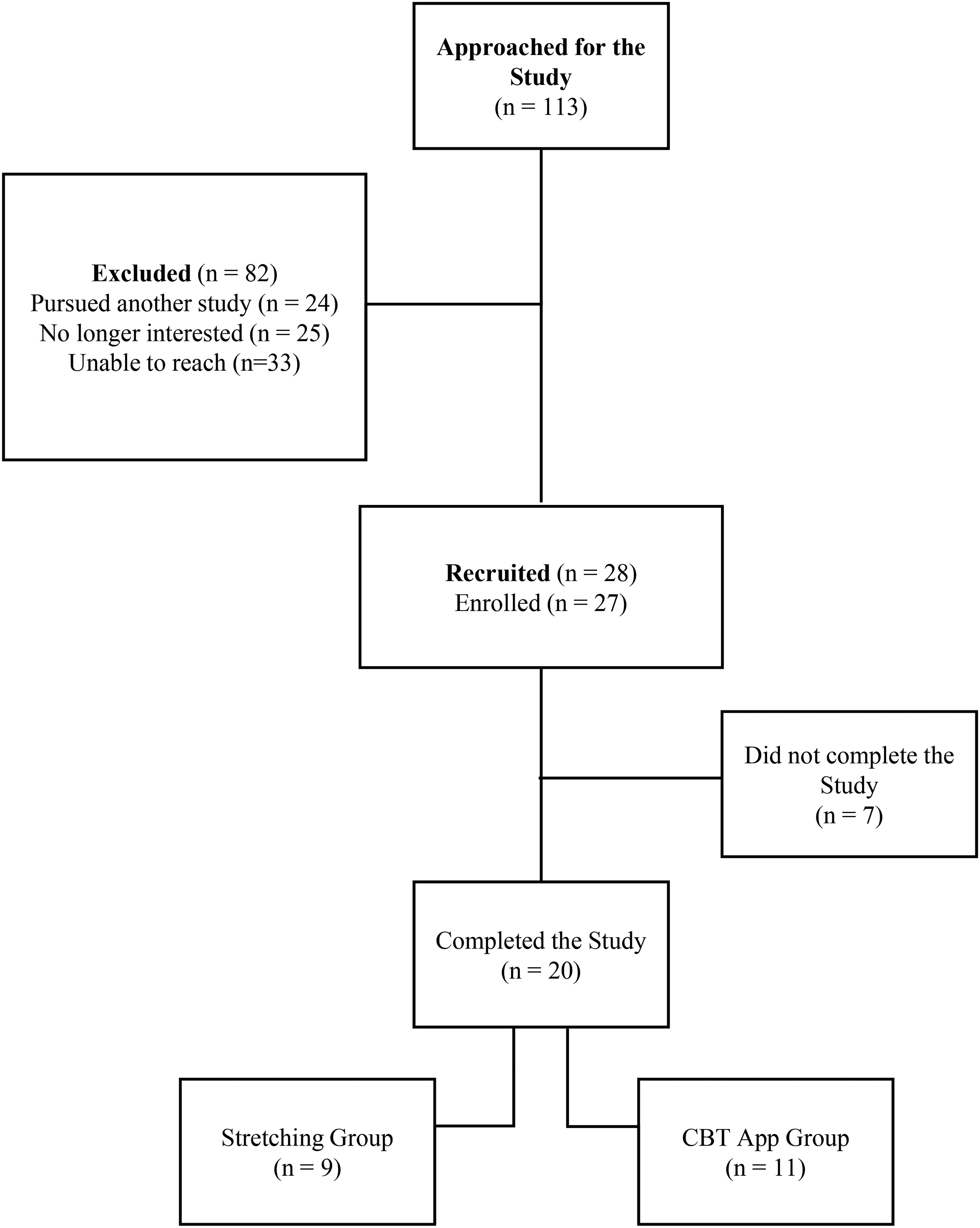
A schematic to outline recruitment for this study, with all participants recruited through the neurology clinic at the Children's Hospital of Eastern Ontario.
Demographic Summary of Cohort.

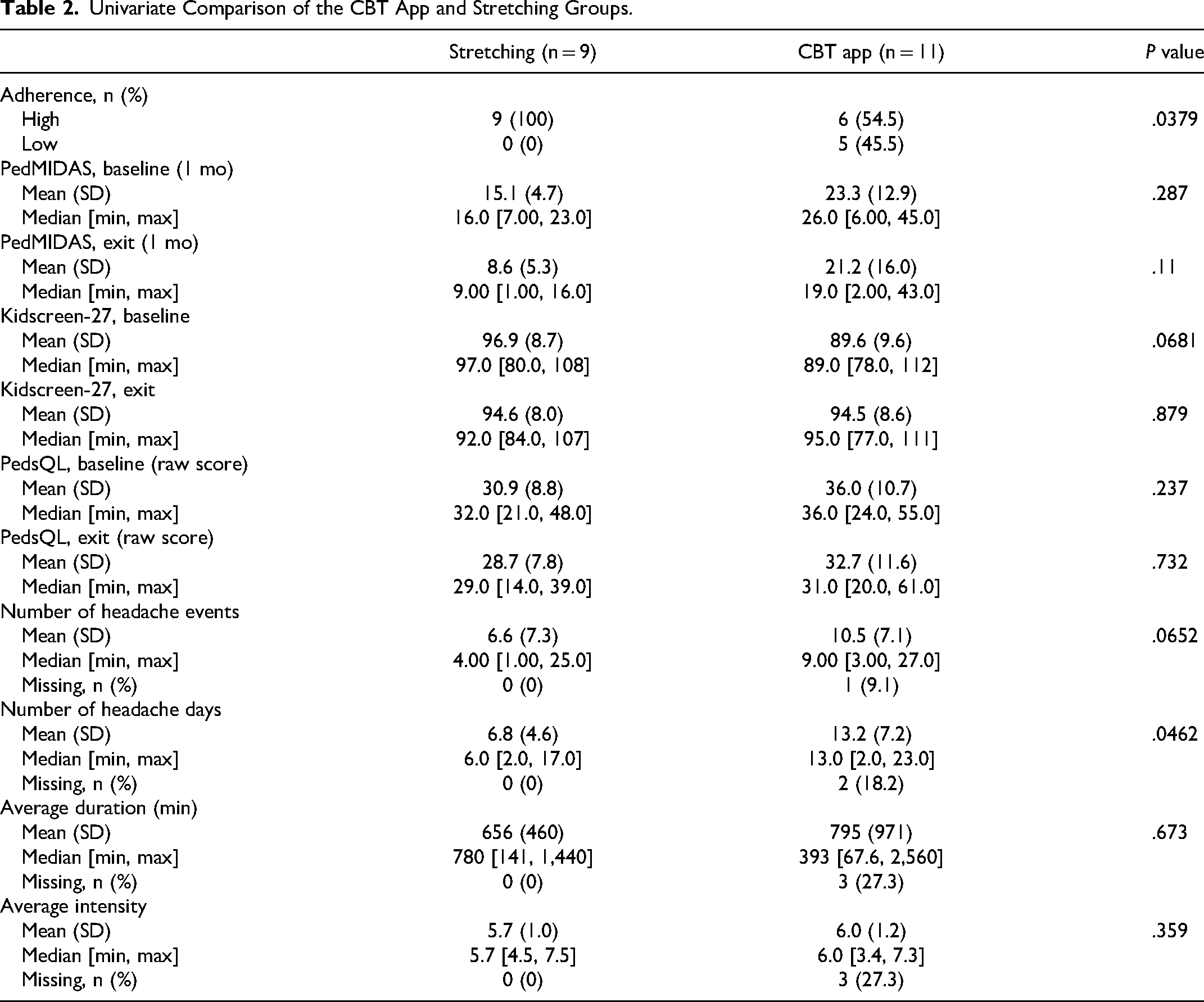
Univariate Comparison of the CBT App and Stretching Groups.
Comparative Analysis Between Groups
Univariate comparison of the Stretching and CBT app groups was completed for adherence, PedMIDAS baseline and exit scores, Kidscreen-27 baseline and exit scores, PedsQL baseline and exit scores, headache events, days, severity, and duration (Table 2). First, adherence was compared between groups and determined to be significantly higher in the stretching group (P < .05). Adherence was self-reported by participants and defined as the number of days they completed their assigned task out of the total number of days in the study (28 days). Next, the primary outcome, PedMIDAS scores, at baseline (0 weeks) and exit (4 weeks) were compared between groups. PedMIDAS scores reflect the number of days missing various activities due to headaches in a 1-month period. Mean PedMIDAS scores at baseline (P = .287) and exit (P = .11) were not significantly different between groups following univariate analysis. Secondary outcomes including Quality of Life measures and headache variables were compared between groups. Neither Kidscreen-27 baseline (P = .0681) and exit (P = .879) nor PedsQL baseline (P = .237) and exit (P = .732) scores were significantly different between groups. Further, all measured headache variables showed no significant differences between groups.
Primary Outcome: Headache-Related Disability
Before testing if headache-related disability was different between these groups, first we must establish if there are group-level differences that might confound this question. Adherence to an assigned activity was higher in the stretching group compared to the CBT app group (P < .05). All participants in the stretching group had high adherence (defined as greater than 70% adherence to their assigned activity: Figure 2). It was observed that within the CBT app group, 4 of 6 participants with higher adherence had higher PedMIDAS scores at the time of the exit interview (ie, worse outcomes) compared to baseline.
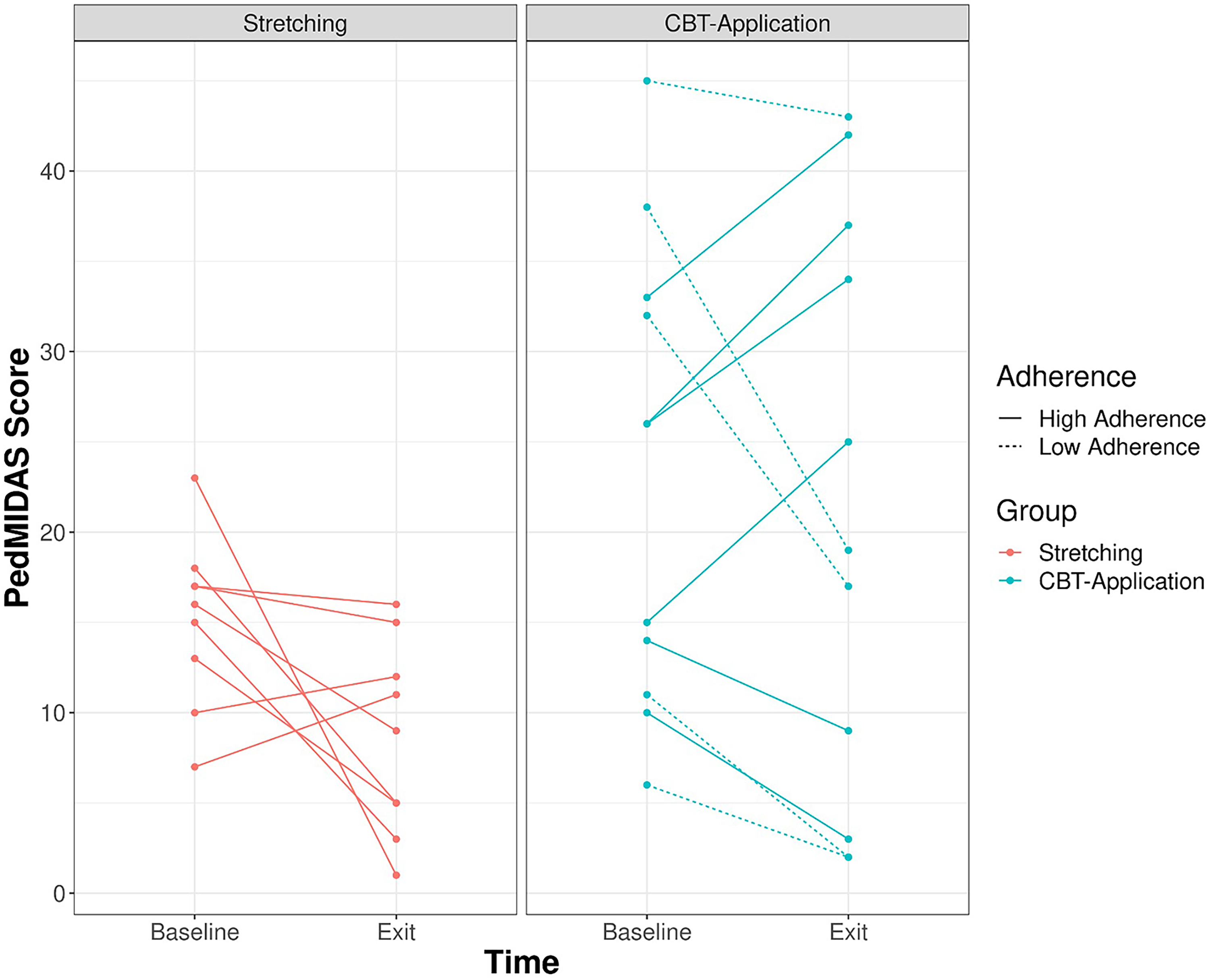
Comparison of the change between groups (Stretching = red; CBT app = green) on PedMIDAS (1 month), and how it varies by adherence. Lower PedMIDAS represent an improvement in quality of life. Participant high or low adherence is represented as either a solid or dashed line.
Multivariable Analysis
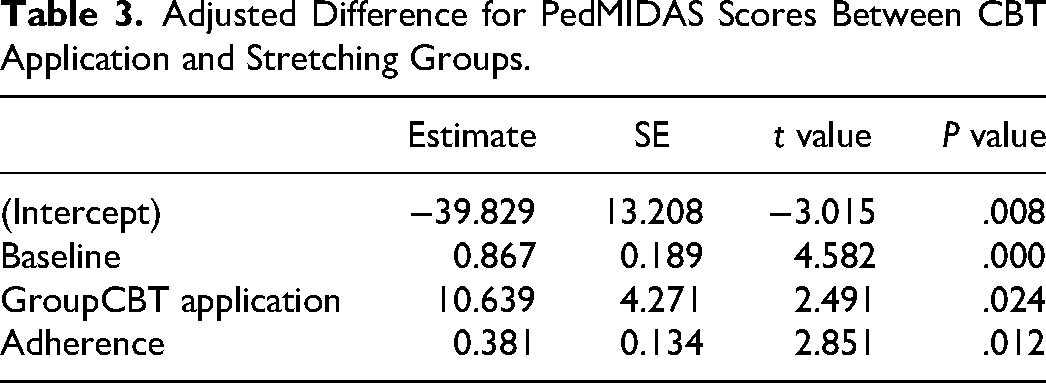
ANCOVA analysis was conducted adjusting for adherence and baseline PedMIDAS score. It was observed that the CBT app group had 11 units (P value = .024, Table 3) higher PedMIDAS scores. However, without adjusting for adherence, there is no significant difference in baseline to exit scores between the 2 groups.
Adjusted Difference for PedMIDAS Scores Between CBT Application and Stretching Groups.
Secondary Outcomes: Headache Variables and Quality of Life With Adjustment for Adherence
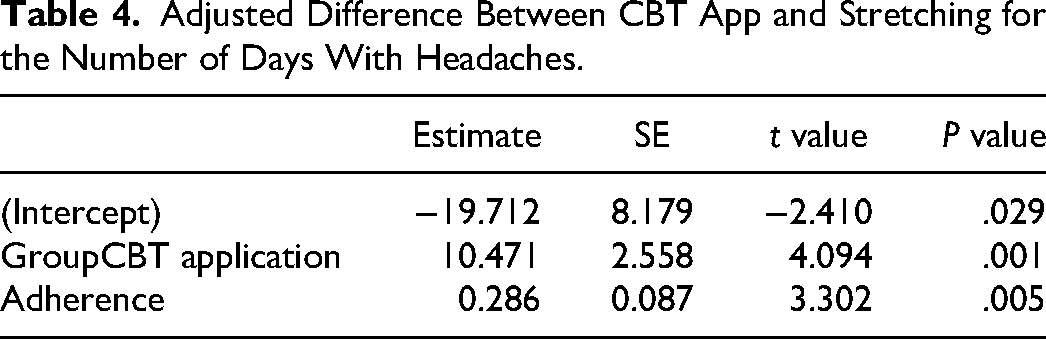
The number of headache days was significantly higher in the CBT app group (10.41) than the stretching group (P = .001, Table 4) when adjusting for adherence. There was not a significant difference between groups for average duration of headache, average intensity of headache, or the number of headaches when adjusting for adherence (P > .05). Suspected headache triggers recorded on Migraine Buddy by participants included stress (7 participants), sleep deprivation (6), decreased fluid intake (5), weather changes (4), skipped meals (3), having missed taking their preventative headache medication (2), premenstrual stage (2), and increased screen time (2). When adjusting for adherence, there remained no significant difference between the CBT app and stretching groups for Quality-of-Life Indicators, Kidscreen-27, and PedsQL pre- and postintervention raw scores.
Adjusted Difference Between CBT App and Stretching for the Number of Days With Headaches.
Feedback Questionnaire
A feedback questionnaire was developed to gather viewpoints of participants regarding their involvement in the study and intervention-specific feedback. A total of 12 (60%) participants completed the survey. Questions were answered on a 5-point Likert scale, 5 being the most positive rating. The questionnaire contained a mixture of general questions about their experience and intervention-specific questions. Users suggested improving ease of navigation through the CBT app and making the app more age appropriate. Average experience rating for the stretching group was 3.9/5.0 and 3.5/5.0 for the CBT app group.
Discussion
We explored the feasibility and efficacy of an app-based CBT program for children suffering from headaches. To the best of our knowledge, this is the first randomized control trial comparing headache related disability, frequency, severity, and duration, as well as quality of life, between a CBT and a stretching program. We did not obtain evidence supporting the efficacy of app-based CBT for decreasing headache related disability in youth suffering from headache,31–33 our CBT app intervention did not prove to be more effective than a stretching program. Additionally, our study did not demonstrate a difference between CBT and stretching on quality of life as measured by the Kidscreen-27 34 and PedsQL. 35 Finally, headache variables including number of headache events, days, duration, and severity of headaches were not significantly different between groups.
Interest for exploring this topic stemmed from a variety of studies incorporating CBT as an adjunct therapy for chronic migraine management, including a notable randomized clinical trial in youth that examined reduction in headache related disability following combined therapy of CBT and amitriptyline versus headache education and amitriptyline. A greater reduction in headache days and migraine-related disability was noted in the CBT plus amitriptyline group, supporting efficacy of CBT as an adjunct therapy for chronic migraine. 31 Another study in adult patients with headaches examined the long-term clinical effectiveness of combined medication therapy and individual or group-based CBT. The CBT sessions included aspects of psychoeducation, progressive muscle relaxation, coping strategies for pain and stress, and goal-setting skills. The combination of medical therapy with CBT led to clinically significant improvement in pain coping strategies, decreased headache intensity, and improved pain-related cognitions over a 12-month period. 14 Additionally, a more directly comparable pilot study, with a population of 21 youth, did observe reduction in headache days following the implementation of a hybrid CBT program. 36 Of note, the above-mentioned studies delivered CBT through in-person sessions, rather than self-directed technology-based platforms.
Among the studies included in a Cochrane Review of 20 studies 32 and a meta-analysis of 14 randomized control trials 33 showing the efficacy of CBT in headache management, were computerized web-based CBT programs.37,38 One pilot study showed reduction in headache frequency in children and adolescents following internet-based self-help training, rooted in concepts of CBT, in comparison to a psychoeducation intervention group. 39 The use of a CBT-based app for adult headache sufferers showed a reduction in the number of headache days. 25 However, the CBT app used in our study did not result in a clinically meaningful decrease of headaches. Reasons for this may include that the CBT app we used was not developmentally or headache specific. The CBT app did address suspected headache triggers such as elevated stress while it did not address other commonly reported triggers such as decreased caloric and fluid intake, and sleep deprivation. This could be another reason why this intervention did not succeed. Additionally, it should be considered that CBT may be difficult for pediatric patients to practice in a self-guided manner. CBT without extensive professional or parental involvement may not be feasible in young patients, as they may not be able to fully understand the behavioral and cognitive strategies taught through CBT and how to incorporate these concepts into their daily life. A willingness to participate in adopting skills in everyday life and the ability to cognitively participate in CBT is dependent on cognitive development and can impact the effectiveness of treatment. 40
Our stretching group patients demonstrated better improvement of headaches-related disability compared to the CBT patients, after completing multivariate analyses with consideration of adherence rates. Physical-based therapy has been widely used by patients suffering with headache, particularly tension-type, as the techniques geared toward release of muscle tension target a proposed trigger of this headache type.41–43 This has been demonstrated by studies implementing therapeutic exercise for tension-type headaches, showing superior reduction in pain than no treatment or placebo 41 as well as a pilot trial implementing contract-relax technique and static stretching in migraineur women, showing improvement in headache and migraine-related disability following both interventions. 42 Nevertheless, there is a paucity of evidence-based data to support its efficacy in pediatric headache management.
Both app-based CBT and stretching were well tolerated by our participants, with no adverse events reported. However, our results demonstrate higher adherence among the stretching group compared to the CBT app group. This finding may be a result of participants in the stretching group feeling more actively involved and engaged. A subset of participants described completing their daily stretches with family members, which helped them adhere to it. We hypothesize that the CBT app was not engaging and interactive enough, as suggested by our participants’ feedback. Another pediatric headache study using CBT reported good adherence to CBT, with participants regularly attending treatment sessions and engaging in practicing skills at home. 44 Of note, this study used an in-person CBT program. A consideration for improving adherence to an inherently less active or physically engaging modality, such as the CBT app, is described by Ramsey et al. 45 They suggested the use of an app-based progressive reminder system for daily prevention treatment in young adults with migraine. 45 We attempted to implement a tracking system by requesting participants to mark their adherence on the Migraine Buddy app to encourage accountability; however, an app that sends reminders may be more successful.
Study Limitations
One of the main limitations of this study was the sample size, creating difficulty when exploring statistically significant and clinically meaningful conclusions. Recruitment slowed during periods when participants were undergoing pandemic virtual schooling and during summer months, as headache burdens seemingly decreased during these periods. Additionally, transitioning to and from virtual schooling and into summer months could have impacted scores on the PedMIDAS. Given the nature of the questions asked on the scale (ie, days missed from school or extracurricular activities), it could be expected that during summer months or during periods in which school or social activities were limited because of pandemic lockdowns, scores would reflect this and be lower than expected.
Conclusions
The data from our study questions the effectiveness of CBT via an app for reducing pediatric headache-related disability. Interestingly, a standardized stretching program was adhered to more and resulted in superior improvement in headache-related disability compared with CBT app treatment. Possible explanations for this are that the app used in this study was not age or headache specific, or that self-guided CBT is difficult and may not be feasible for all patients depending on their cognitive, social, and emotional maturity. Given the small size of this pilot study, future investigation is warranted to explore the role of CBT apps for headache management in a larger pediatric population, with the creation of an age-appropriate and headache-specific app with attractive features that would increase engagement as seen with more actional activity, like the stretching program.
Footnotes
Acknowledgements
We would like to thank all children and their families for participating in this study, and Drs. Doja, Muir, Sell, Thulasirajah, and Venkateswaran for their assistance with recruitment.
Author Contributions
CM, DP, NW: study conception and design; CM: data collection; RB, RW, CM, PD: analysis and interpretation of results; CM, DP, RB, RW: draft manuscript preparation. All authors reviewed the results and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the folllowing financial support for the research, authorship, and/or publication of this article: This study was supported by an internal grant from the Children's Hospital of Eastern Ontario Research Institute.
Ethical Approval
This study was approved by and conducted in accordance to the regulations of the Children's Hospital of Eastern Ontario Research Institute Ethics Board. Written informed consent was obtained by all participants or legal guardians of minor participants. Written assent was provided by all minor participants.
