Abstract
Hypoxic-ischemic encephalopathy, caused by an intrapartum event leading to perinatal asphyxia, is the most frequent and well-studied cause of neonatal encephalopathy. 1 It is associated with later neurologic morbidities, including cerebral palsy, seizures, cognitive impairment, and behavioral challenges. Therapeutic hypothermia, initiated within 6 hours of birth and continued for 72 hours, has been established as a safe and effective therapy for neonates born at greater than 35 weeks of gestational age, who present with clinical evidence of moderate or severe neonatal encephalopathy.2‐6 It has been shown to decrease mortality or moderate to severe neurodevelopmental disability at 18-24 months of age,2,3 with a number needed to treat of 6 to 7. 3 Therapeutic hypothermia has also been shown to improve neurocognitive outcomes at school age. 7 At our institution, therapeutic hypothermia was implemented in 2008 with a specific referral and treatment algorithm shared with regional birthing centers.
In the acute neonatal period, outcomes of infants with hypoxic-ischemic encephalopathy are often difficult to predict. 8 Clinicians need to know what available information can be used to better counsel parents on long-term prognosis. The literature on hypoxic-ischemic encephalopathy, therapeutic hypothermia, and cerebral palsy is extensive. However, there are some gaps in the literature in terms of examining the profile of children with cerebral palsy who received therapeutic hypothermia after intrapartum asphyxia (IAP), both in terms of neonatal factors and long-term cerebral palsy outcomes. 8
In addition, parents of infants with hypoxic-ischemic encephalopathy will often ask concrete functional questions about long-term prognosis including: Will my child be able to walk? Will my child be able to speak and communicate with me? Will my child be feeding independently? These functional questions have a direct bearing on caregiver burden and eventual autonomy.
Our primary objective was to explore the profile of children with cerebral palsy secondary to intrapartum asphyxia who were treated with therapeutic hypothermia at a single regional tertiary center. Specifically, we set out to look at their clinical characteristics in the neonatal period and their cerebral palsy profiles as children. In addition, we compared this profile with the profile of children with cerebral palsy prior to therapeutic hypothermia to see if there had been any shift in the pattern of later cerebral palsy with the introduction of therapeutic hypothermia. Finally, we compared the neonatal characteristics (including birth characteristics, cord pH, Apgar scores, and magnetic resonance imaging [MRI] findings) between children with an eventual mild cerebral palsy phenotype and those with a more severe phenotype ultimately.
Methods
Our retrospective study was conducted using data extracted from patient charts in a single-center tertiary level neonatal intensive care unit. Children included in our study were term neonates with hypoxic-ischemic encephalopathy who were treated with therapeutic hypothermia from 2008 until 2018. We chose 2018 as our cut-off so that the children would be of sufficient age (at least 3 years old) for a firm diagnosis of cerebral palsy and associated characteristics at retrospective review.
Using a local research ethics board–approved registry of neonates with intrapartum asphyxia treated with therapeutic hypothermia, we systematically identified those neonates with a later diagnosis of cerebral palsy by either a neurologist or a developmental pediatrician. We then did a systematic chart review on those with cerebral palsy to collect neonatal information. Information collected, when available, included gestational age, cesarean section vs spontaneous vaginal delivery birth, birth weight, Apgar scores at 5 and 10 minutes, arterial cord pH, first blood gas pH, Sarnat score on admission, neonatal seizures if clinically present or seen on electroencephalograph (EEG), use of 2 or more antiepileptic medications to control neonatal seizures, the observation of burst suppression on EEG, and findings on MRI. For analysis of MRI findings, a neurologist categorized MRIs into one of 7 groups based on a previously used method, 9 in addition to an “other” category (see Table 4). We further categorized these into 2 groups for ease of analysis (see Table 4). In addition, we obtained information on outcome measures, including type of cerebral palsy, verbal capability, ambulation status, feeding independence, epilepsy occurrence, presence of substantial sensory impairments related to vision or hearing. CP functional scores was described using standardized measures, that is, Gross Motor Function Classification System (GMFCS), 10 Manual Ability Classification System (MACS), 11 and Communication Function Classification System (CFCS). 12
Furthermore, we divided our cohort into mild and severe phenotypes of cerebral palsy. The mild phenotype group included children who were verbal, ambulatory, and feeding independently. The severe phenotype involved children who were either nonverbal, nonambulatory, or feeding dependent or had more than 1 of these characteristics. We were then able to compare clinical characteristics in the neonatal period between these 2 groups to look for helpful indicators for clinical outcome.
Statistics
Statistical analysis was performed using SPSS. For comparisons between the 2 groups (mild vs severe phenotype), the Student t test was used for parametric continuous variables and the Mann-Whitney U test was used for noncontinuous data. A χ2 test was used to compare distributions of MRI findings between mild and severe phenotypes. A P value of <.05 was selected a priori to indicate statistical significance.
Results
A total of 335 children were cooled between 2008 and 2018. Of these neonates, 52 neonates died in the neonatal period, 191 did not have an adverse outcome, 43 were lost to follow-up, and 49 had an adverse outcome (either cerebral palsy, global developmental delay / autism spectrum disorder, and/or seizure/epilepsy). Thirty children (8%) were identified later to have cerebral palsy (Table 1). The mean and median age in years of the last patient follow-up used for cerebral palsy phenotype was 6 and 7, respectively, with a range of 3 to 12.
Number of Children Undergoing TH in a Single-Center Tertiary Level Neonatal Intensive Care Unit between 2008-2018.

Abbreviations: CP, cerebral palsy; TH, therapeutic hypothermia.
Our current report focuses on these 30 children with eventual cerebral palsy. The perinatal characteristics of the children with cerebral palsy are outlined in Table 2. There were similar number of males and females, and similar numbers of cesarean section vs spontaneous vaginal delivery (Table 2). Twenty-nine (97%) of the children had neonatal seizures documented (22 of which had EEG or amplitude-integrated EEG confirmation of at least 1 seizure and 7 of which had only clinical suspicion but an abnormal interictal EEG), with 23 requiring 2 or more antiepileptic medications to control their neonatal seizures. Burst suppression on EEG was documented in 17 children (59%) (Table 2). The average birth weight was 3.17 kg (Table 2).
Neonatal Characteristics of Children With CP Post-TH (n = 30 Unless Otherwise Specified, if Patient Information Was Unavailable). a

Abbreviations: CP, cerebral palsy; TH, therapeutic hypothermia.
Some records were not available for all children: 2 children without documented 5- and 10-minute Apgar scores, 6 children without arterial cord pH documented (1 clotted, 5 records not available), and 2 children without a documented first blood gas pH, 16 children without arterial cord base excess documented, 6 children without first blood gas base excess not documented.
Sentinel events: placenta abruption, cord prolapse, uterine rupture, absent variability, bradycardia, repetitive late decelerations, shoulder dystocia.
We also explored long-term outcomes. We found that most children with cerebral palsy in our study had spastic quadraparetic cerebral palsy, with other types of cerebral palsy being less common (Table 3). Twenty children had a GMFCS Level of III-V, with 8 of these using a walker (GMFCS Level III). Eighteen (60%) had associated epilepsy. Sixteen children (53%) were nonverbal. Although visual impairments ranged from simple strabismus or amblyopia to cortical blindness, for the purpose of our study, we included only significant visual impairment (cortical visual impairment or legal blindness) in our count. Only 2 (7%) had legal blindness or cortical visual impairment. Likewise, hearing problems ranged from low-frequency hearing problems to complete deafness, but, for the purpose of our analysis, we included only complete deafness, or those requiring amplification or cochlear implants. There was variability in hearing impairment but only 3 (10%) were either deaf or required hearing aids or cochlear implants. Fourteen children (47%) were feeding dependent (gastric tube/gavage fed). Two children (7%) included in our cerebral palsy cohort died in the follow-up period (at 3 and 7 years): one of cardiorespiratory failure secondary to chronic aspirations and the other while overseas with family for whom a death certificate was not available to us.
Neurodevelopmental Outcome of Children With CP Post-TH (n = 30).

Abbreviations: CP, cerebral palsy; GDD, global developmental delay; ID, intellectual disability; TH, therapeutic hypothermia
Characteristics: nonambulatory, nonverbal, and feeding dependent.
When comparing mild vs severe cerebral palsy phenotypes, we found that there were more severe phenotypes (20/30) than mild phenotypes (10/30) (Table 4). Children with a severe cerebral palsy phenotype had significantly higher mean birth weight (P < .05) and lower 5- and 10-minute Apgar scores (P < .05) (Table 4), compared with those with mild cerebral palsy phenotypes. The 2 groups had similar cord pH and first blood gas pH, as well as similar modified Sarnat scores on admission (Table 4). In terms of imaging, we found that there was a difference in distribution of MRI findings when comparing mild vs severe phenotypes. The children with severe cerebral palsy phenotype had more often white matter injury with associated deep gray matter injury pattern or a near-total injury pattern (P < .05) (Table 4).
Comparison of Neonatal Characteristics Between Mild vs Severe CP Phenotypes. a
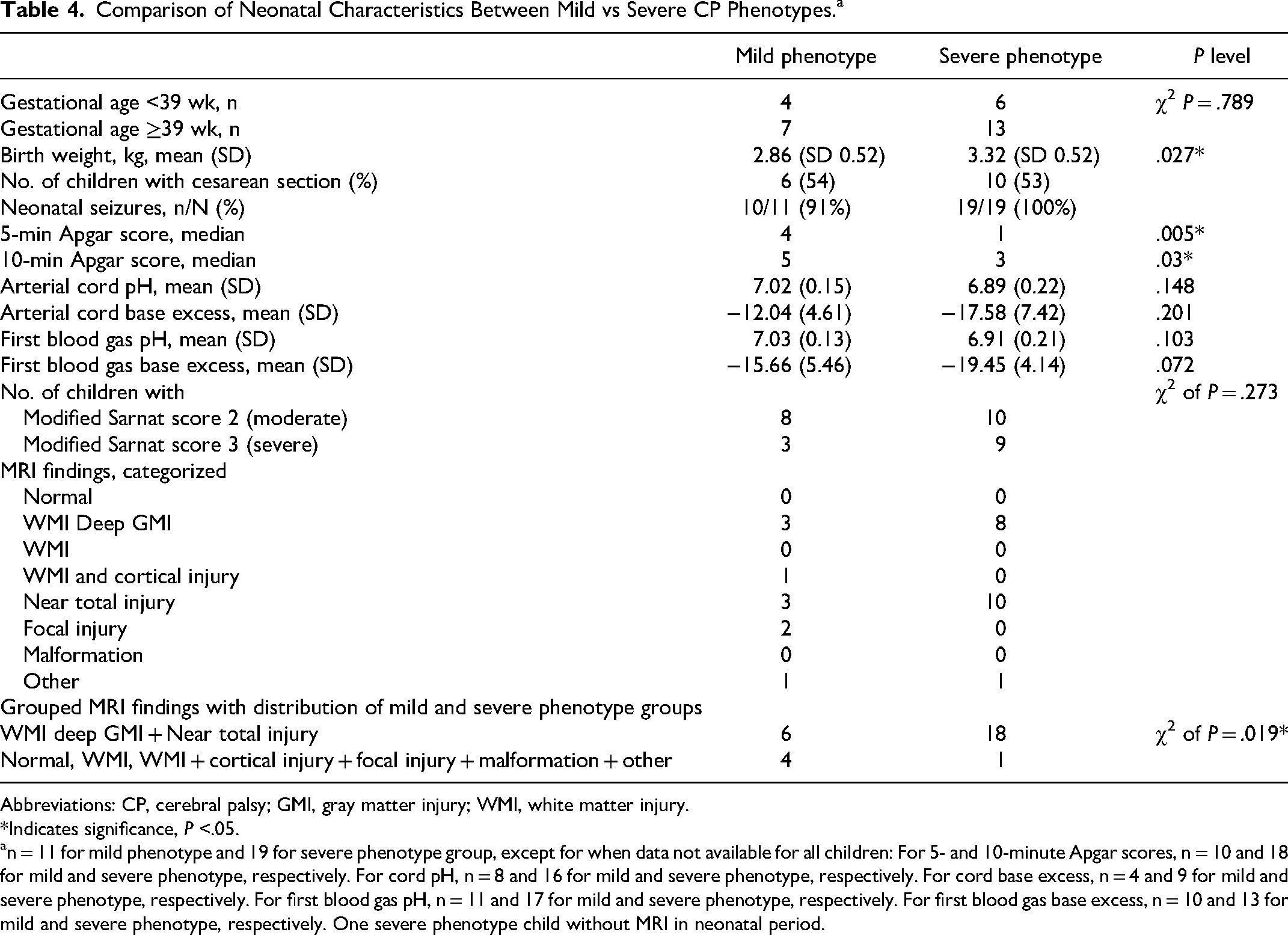
Abbreviations: CP, cerebral palsy; GMI, gray matter injury; WMI, white matter injury.
*Indicates significance, P <.05.
n = 11 for mild phenotype and 19 for severe phenotype group, except for when data not available for all children: For 5- and 10-minute Apgar scores, n = 10 and 18 for mild and severe phenotype, respectively. For cord pH, n = 8 and 16 for mild and severe phenotype, respectively. For cord base excess, n = 4 and 9 for mild and severe phenotype, respectively. For first blood gas pH, n = 11 and 17 for mild and severe phenotype, respectively. For first blood gas base excess, n = 10 and 13 for mild and severe phenotype, respectively. One severe phenotype child without MRI in neonatal period.
Discussion
To compare clinical characteristics of our cohort of children with cerebral palsy treated with therapeutic hypothermia after intrapartum asphyxia with characteristics of children with cerebral palsy in the pre–therapeutic hypothermia era, we reviewed the historical literature looking at clinical characteristics of children with cerebral palsy prior to therapeutic hypothermia.
In terms of type of cerebral palsy, our study found 16 (76% of the spastic cerebral palsy group, and 53% of the total cerebral palsy group) had documented quadriplegia, which is increased compared to the 44% found by Koman et al13 in their study assessing 906 children with cerebral palsy in central and western North Carolina, USA, prior to therapeutic hypothermia. Of note, however, their report included all causes of cerebral palsy and not just children with hypoxic-ischemic encephalopathy. We found only 3% of our total cerebral palsy cohort (and 5% of the spastic group) to have documented diplegia, which is significantly less than that reported by Koman et al13 of 33%. Finally, 10% (14% of the spastic group) of our study group had documented hemiplegia, which is less than the 23% documented by Koman et al.13 The percentage of children who were nonambulatory, 33%, was a bit higher in our study compared to that reported prior to therapeutic hypothermia of 25%.13,14 The overall distribution of GMFCS scores was similar to previous reported distributions prior to therapeutic hypothermia. We found 33% of our cohort to have a GMFCS Level of I-II and 67% to have a GMFCS Level of III-V, which is close to the percentages of 39% and 61%, respectively, reported by the Canchild Centre for Childhood Disability in Ontario study. 15 Pappas and colleagues presented a comparison between a group treated with hypothermia and a group that were not treated with hypothermia, and reported a mild motor disability (GMFCS I) (19%), moderate disability (GMFCS II-III) (19%) and severe disability (GMFCS IV-V) (63%) in their group of patients who did not receive hypothermia. Although their groups were divided into 3 rather than 2, the overall pattern is similar to the distribution we found. 16 Our findings can also be compared to those of Jary and colleagues, 17 who looked at incidence, type, and severity of cerebral palsy in a regional cohort and compared them with historical outcomes in therapeutic hypothermia trials. Overall, their hypothermia group had lower mortality and comparable rates of cerebral palsy, although fewer children were severely affected. Of those who developed cerebral palsy, 67% had a GMFCS of I and 33% had a GMFCS of V, 17 in contrast to our 33% and 67% for GMFCS I-II and GMFCS III-V, respectively (Table 3).
We were unable to find data on distribution of MACS or CFCS scores prior to the therapeutic hypothermia era, likely because the emergence of development and use of these standardized scores coincides in time roughly with the emergence of therapeutic hypothermia. In our study, 18 (60%) had epilepsy. Prior to therapeutic hypothermia, this number had been documented as lower: that is, 35% with seizures, although this percentage was again suggested for all causes of cerebral palsy and not just children with cerebral palsy due to hypoxic-ischemic encephalopathy, 13 or one third of children with cerebral palsy had epilepsy. 18 Pappas and colleagues 16 found that 22% of children in their normothermic group had epilepsy, compared with their hypothermia group, which was documented as 16%. One suggestion why there would be an increase in epilepsy in our patient cohort is our cohort includes a bias for a more severe phenotype given the children would need to meet eligibility criteria and have a moderate to severe hypoxic-ischemic encephalopathy to qualify for therapeutic hypothermia, compared with the previously documented literature that also included other causes of cerebral palsy without preceding moderate to severe neonatal encephalopathy, although this would not explain the discrepancy between our findings of epilepsy in the hypothermia group of Pappas et al. 16 One area of bias in our study is the loss of 43 children to follow-up (Table 1). It is possible that those children lost to follow-up may have had a milder phenotype and may have been less likely to have epilepsy.
In terms of visual impairments, prior to therapeutic hypothermia, reports of visual impairment were 20% to 40%, 13 and in our study, we found 7%. Pappas and colleagues found blindness in 11% of the normothermia group, and 2% of their hypothermia group. 16
Of course, there are limitations in comparing our cohort with literature prior to therapeutic hypothermia. When comparing characteristics of our cerebral palsy cohort and that of the pre–therapeutic hypothermia era, we must remember that some of the pre–therapeutic hypothermia era data, such as those of Koman et al,13 included all causes of cerebral palsy, including other causes of cerebral palsy such as prematurity and neonatal stroke, and these children would not have met criteria for therapeutic hypothermia. It has been previously speculated that therapeutic hypothermia may lead to a deferral in the decision to withdraw active care in some severely asphyxiated neonates after 72-hour treatment is completed, and by then any initial cardiorespiratory compromise may have improved sufficiently to enable survival. 19 In addition, there have been other advances in general neonatal care (ie, cardiorespiratory support, management of seizures, feeding strategies, etc) over the last decade that may also contribute to greater survival.
In terms of our objective to look for neonatal predictor markers, we found that birth weight, and 5- and 10-minute Apgar scores were significantly different (P < .05) when comparing severe vs mild cerebral palsy phenotype groups. The differences in 5- and 10-minute Apgar scores between the 2 groups are unsurprising to us as we often consider these scores, as well as individual components of these scores, as part of our global evaluation to reflect on the possible severity of an asphyxial event. This information may be useful in discussing prognosis regarding neonates receiving therapeutic hypothermia. The risk of cerebral palsy has been found to be higher in children with low 5- and 10-minute Apgar scores.20,21 In terms of birth weight, it is interesting that the milder phenotype had a statistically significantly lower mean birth weight. One possibility is difficulty with extracting larger neonates, although only 1 neonate (ultimately in the mild phenotype category) had documented shoulder dystocia during birth. Another possibility would be the larger neonates having a more advanced gestational age; however, there was no significant difference in distribution in terms of gestational age between the groups. Finally, this finding could simply be a statistical variation. Future work could look at the precise mechanism for moderating outcome in more detail. Cord pH and first blood gas pH were not found to be significantly different between the 2 groups, which comes as surprise given we also use this information in the clinical setting to evaluate for the severity of an asphyxial injury. In a clinical setting, we often consider these as potential indicators of eventual child outcome. Low cord pH has been shown to have a strong association with neonatal morbidity and long-term outcome, including cerebral palsy. 22 Again, the fact that cord pH and first blood gas pH were not different could be explained by all these children needing to meet eligibility criteria to qualify for therapeutic hypothermia and cord/first blood pH belongs to these criteria.
In terms of the MRI pattern, we found that there was a difference in distribution of MRI findings, with the severe phenotype group having more white matter injury associated with deep gray matter injury or a near-total injury pattern. Imaging, as early as day 2 of life, have been shown to be associated with later abnormal neurodevelopmental outcome. 23 MRI has shown association with neurologic subtype and cerebral palsy severity. 24
Importantly, it has been previously discussed that parameters, including Apgar scores and severity of acidemia have low sensitivity and positive predictive value for outcome. 25 However, a combination of clinical values is more predictive of outcome than individual values in terms of prognostic utility. 25 Our study is limited by consideration of individual factors, and not in combination. Another important point is that 52 children died after therapeutic hypothermia in the neonatal period. 26 Many of those who died in the neonatal period would likely have had sufficient criteria to merit a later diagnosis of cerebral palsy if they had survived beyond 30 days. This may have also impacted our results, as they would have almost surely fit (if they had survived) into our severe phenotype group. As mentioned previously, 43 children were lost to follow-up, which may have introduced a bias for a more severe phenotype, as those lost to follow-up may have had a greater likelihood for having a mild phenotype.
Our cohort was all children who were cooled in only a single-center tertiary-level neonatal intensive care unit vs historical literature that regrouped children from different centers. One area for future direction is that we only looked at neurodevelopmental characteristics in early childhood to determine our “severe” and “mild” phenotype groups. A later look at outcomes might also be beneficial (ie, levels of education attained, employment, learning disorders, overall quality of life), as cerebral palsy has a lifelong impact.27‐29 Further studies could consider these later endpoints to further categorize the “milder” phenotype, as they would also be important in parental discussions on prognosis. In addition, a multicenter study may be beneficial in obtaining information on more universal outcomes and not just outcomes based on practices (follow-up plan, available therapeutic hypothermia resources) specific to the one center. A multicenter study would also enable the possibility of comparing local neonatal intensive care unit practices and their influence on modifying later neurodevelopmental outcome.
In conclusion, we found differences in cerebral palsy profiles between children prior to therapeutic hypothermia and children in the post–therapeutic hypothermia era. Our data demonstrated more infants with severe cerebral palsy rather than mild cerebral palsy in our cohort of children treated with therapeutic hypothermia for intrapartum asphyxia. Five- and 10-minute Apgar scores were significantly different between mild and severe cerebral palsy phenotypes; however, cord pH and first blood gas pH were not significantly different between these 2 groups. This information can guide clinicians how to better weigh these neonatal factors, when predicting and counseling parents on later infant outcomes.
Footnotes
Author Contributions
HP drafted manuscript. HP and MS contributed to conception and design. HP, MS, PW, FA, PM contributed to acquisition, analysis and interpretation of data, critically revised manuscript and gave final approval.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pia Wintermark receives research grant funding from the FRSQ Clinical Research Scholar Career Award Senior (grant 297362) and a CIHR Project Grant (grant 406737).
Ethical Approval
The ethics review board approval number for the database of neonates with hypoxic ischemic encephalopathy treated with therapeutic hypothermia is 11-602-PED.
