Abstract
Novel antiseizure medications are thought to be safer than their conventional counterparts, though no dedicated analysis of movement disorder risk among pediatric populations using novel antiseizure medications has been completed. We report a systematic review with meta-analysis describing the relationship between novel antiseizure medications and movement disorders in pediatrics.
MEDLINE, EMBASE, and the World Health Organization’s International Clinical Trials Registry Platform were searched up to October 2020 for randomized controlled trials investigating novel antiseizure medications in pediatric populations. Antiseizure medications included lacosamide, perampanel, eslicarbazepine, rufinamide, fenfluramine, cannabidiol, and brivaracetam. Outcomes were pooled using random effects models; risk difference (RD) and 95% confidence intervals (CIs) were calculated.
Twenty-three studies were selected from 1690 nonredundant manuscripts (n = 1912 total). There was a significantly increased risk of movement disorders associated with perampanel (RD 0.07, 95% CI 0.01-0.13; n = 133), though only 1 relevant trial was found. No increased risk of movement disorders was found with other antiseizure medications.
Our findings indicate most novel antiseizure medications are safe to use in pediatric populations with respect to movement disorders. However, findings were limited by quality of adverse event reporting.
The number of approved and marketed anti-seizure medications has drastically increased in recent years, 1 providing more options than ever for managing epilepsy. Concomitant with this diversity, further guidance is necessary to aid clinicians and patients in the rational choice of treatments. The pharmacokinetic implications of the patient’s age must also be considered when assessing the adverse event profile of a drug.
Many antiseizure medications exert their effect on GABAergic spiny projection neurons of the striatum as well as cholinergic and GABAergic interneurons.2,3 These, in addition to dopaminergic and cholinergic neurons, regulate the basal ganglia,3,4 and this connection may explain the mechanism of antiseizure medications treating and causing movement disorders.
The risk of movement disorders secondary to use of older antiseizure medications in children has been studied. Valproic acid has been reported to cause tremor in a dose-dependent manner in 15% of children. 5 Phenytoin and carbamazepine have also been shown to cause tremor in 14% and 1% of children, respectively. 5 Ataxia is recognized as a complication of various antiseizure medications including primidone, phenobarbital, phenytoin, carbamazepine, and lamotrigine.6,7 Various other movement disorders have been reported in association with older antiseizure medications, including choreoathetosis, 5 dystonia,5,6 and asterixis. 6 Novel antiseizure medications are thought to be better tolerated than their conventional counterparts but have also been shown to cause movement disorders.8–12
The risk of treatment-emergent movement disorders secondary to novel antiseizure medication use in adults has been well documented,13,14 but data specific to pediatric populations remains sparse. There are, to our knowledge, no dedicated assessments of the risk of movement disorders secondary to novel antiseizure medications in pediatric populations. To address this knowledge gap, we performed a systematic review with meta-analysis of all randomized clinical trials that quantitatively and exhaustively report adverse event incidence of novel antiseizure medications in pediatric populations. We sought to investigate whether pediatric populations receiving novel antiseizure medications have an increased risk of movement disorders relative to placebo.
Method
Protocol
A protocol was prepared according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for reporting harms 15 and with consultation from the Cochrane Handbook for Systematic Reviews and Interventions 16 in best practice for producing and disseminating a protocol. The study protocol was registered with the Open Science Framework. 17 Ethics committee approval was not sought for this study as per the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans. 18
Eligibility Criteria
Eligible studies were randomized controlled trials of pediatric populations in which a novel antiseizure medication was used and where adverse events were reported quantitatively. Studies were excluded if (1) the mean or median participant age was greater than 19 years unless a pediatric subgroup analysis was performed, (2) the study was not published in English, and (3) the study was not performed on humans. For the purpose of this review, novel antiseizure medications included lacosamide, perampanel, eslicarbazepine, rufinamide, fenfluramine, cannabidiol, cannabidiol with low-dose tetrahydrocannabinol (≥20:1), and brivaracetam. These antiseizure medications were selected as they have most recently entered clinical practice in our region; a review to date of adverse events was consequently deemed very timely.
Search Strategy
We searched for randomized controlled trials meeting our criteria in 2 bibliographic databases, MEDLINE and EMBASE, supplemented by a search for unpublished studies through the World Health Organization International Clinical Trials Registry Platform (WHO ICTRP). The search used in Medline is given in Table 1 and includes validated search terms 19 for identifying pediatric studies, special Ovid filters to identify adverse drug events based on guidelines by the European Medicines Agency, and validated filters to screen for randomized controlled trials. 16 An analogous set of terms were used for Embase. 20 The WHO ICTRP was exported and locally sorted by drug and participant age. Relevant studies without published data were contacted by the authors.
Medline Search Terms.

Study Selection
Article screening and data extraction was performed using Covidence (Victoria, Australia). Two authors (DP, JY) independently screened all titles and abstracts of studies identified by the initial search. The 2 reviewers independently evaluated the selected full-text articles for inclusion in the systematic review. Inclusion at either stage was determined by consensus.
Data Extraction and Risk of Bias
Data from each included study were independently extracted by 2 reviewers (DP, JY). Extracted data included study design; sample size; antiseizure medication dose and duration; number of concomitant antiseizure medications; participant age at baseline; study inclusion and exclusion criteria; epilepsy syndrome, seizure etiology, or seizure type; prevalence of treatment-emergent movement disorders; study funding source; and study primary objective. When adverse event incidence was not reported exhaustively in the trial’s published manuscript but were exhaustively reported in public clinical trials databases such as clinicaltrials.gov, outcomes from the latter were extracted.
Risk of bias was assessed using the revised Cochrane risk-of-bias tool for randomized trials (RoB 2). 21 No limitations were set in our protocol on how movement disorders were ascertained, and so we used the Smyth Adapted ADE scale 22 to evaluate the quality of adverse drug event reporting. This tool assesses study design, methods for identifying adverse drug reactions, methods for determining causality, methods for determining avoidability, and methods for determining severity.
Data Synthesis and Analysis
The meta-analyses were performed using Stata S/E, version 16. 23 The primary outcome, risk difference of treatment-emergent movement disorders compared to placebo, was first calculated as a proportion in both treatment and control groups, and then presented as the risk difference as a measure of effect size. In addition, the risk difference, as opposed to relative risk, allows for “0%” proportions to be included in the analysis, which was likely to occur with this type of outcome. The overall risk differences and 95% confidence intervals, pooled by antiseizure medication, were calculated using Stata’s metan command, which presents pooled estimates with inverse-variance weights obtained from a random effects model. A forest plot was generated to display data from all included studies, along with pooled estimates, to visually explore the agreement between study-specific proportions, how close they are to the overall estimates, as well as inspect studies with large weights. Heterogeneity between estimates was assessed using the I2 statistic, which indicates the percentage of variance that is attributable to study heterogeneity. An I2 value >75% will indicate high heterogeneity. 24 In addition, Eggers test was used to formally test for small study effects.
Protocol Deviations
We deviated from protocol to adjust for finding fewer relevant studies and fewer cases of treatment-emergent movement disorders than expected. As such, we pooled all movement disorders as a single outcome instead of assessing each type of movement disorder individually. Additionally, it was also not feasible to assess for publication bias, and we did not perform meta-regression to understand movement disorder risk as a function of number of concomitant antiseizure medications, epilepsy syndrome or seizure etiology, age, risk of bias, and quality of adverse drug event content. We performed a subanalysis for the risk of included antiseizure medications on movement disorders only among children with epilepsy. We also analyzed the primary outcome by risk difference instead of prevalence to better account for any increased risk of movement disorders compared to placebo.
Results
The searches were performed during October 2020 and returned 1690 citations after duplicates were removed (Figure 1). After full-text screening, 23 studies represented by 23 publications were included for meta-analysis; no unpublished trials met criteria. Agreement between authors for full-text screening yielded kappa = 0.65 (95% CI = 0.4, 0.86), indicating substantial agreement. 25 Main characteristics of studies included for meta-analysis are described in Supplementary Table S1. Risk of Bias as per the RoB2 tool is represented in Figure 2, and quality of adverse event reporting is summarized in Supplementary Table S2. No studies met criteria using brivaracetam.
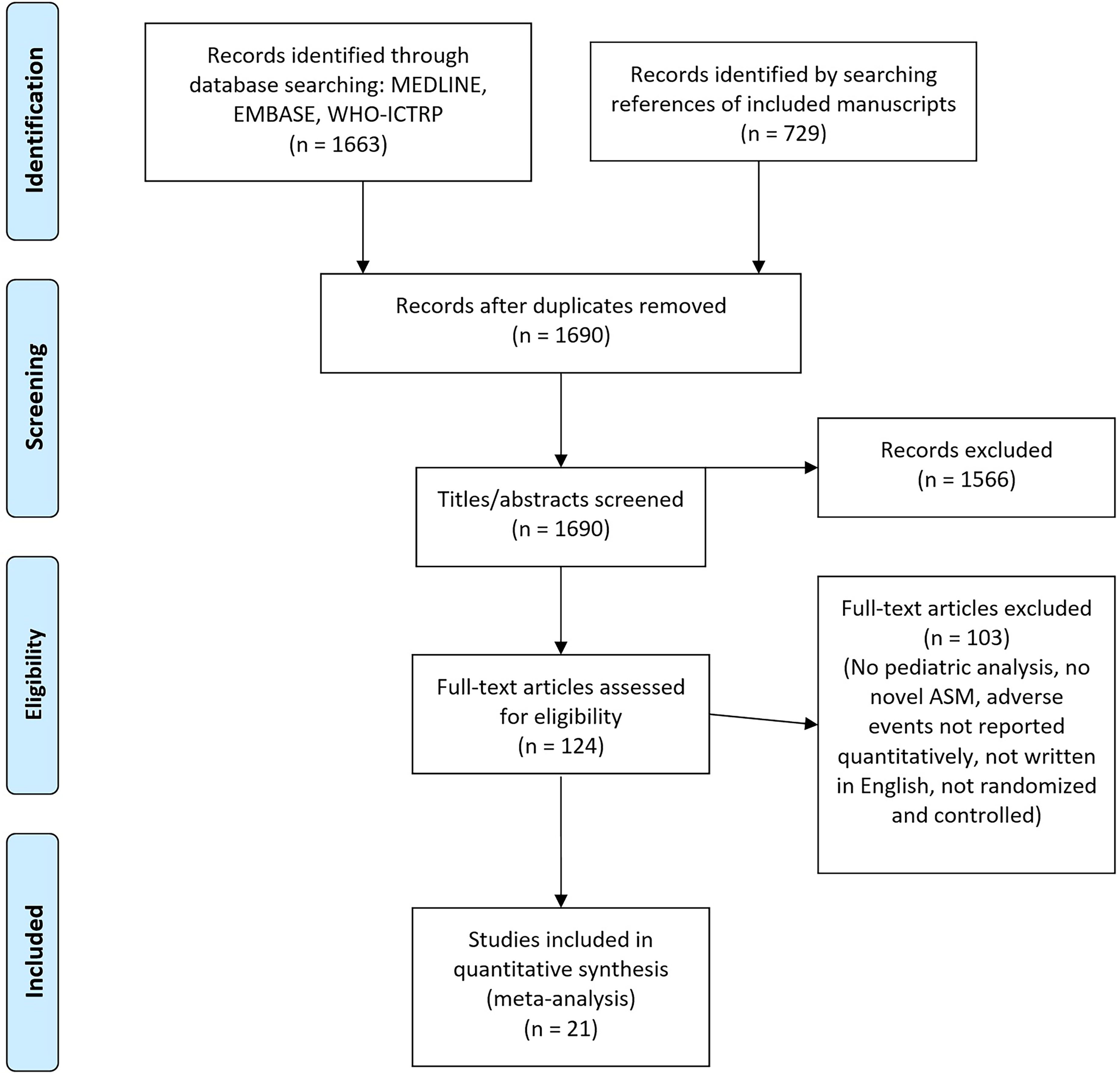
Manuscript selection, including number of manuscripts retrieved, screened, and selected for meta-analysis.
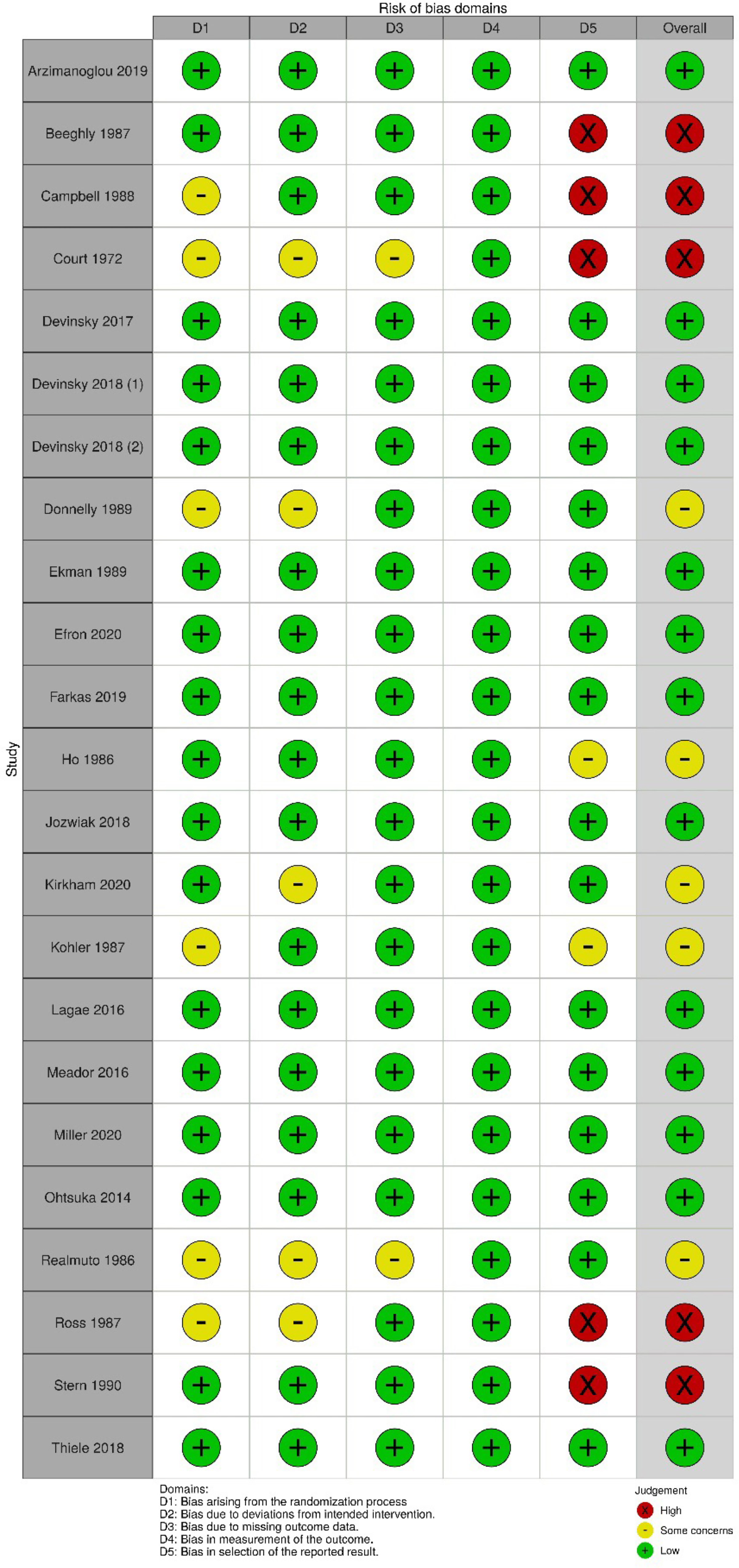
Quality of adverse event reporting among studies included for meta-analysis.
Cannabidiol
Six studies of cannabidiol were included with doses ranging from 5 to 20 mg/kg/d, including a total of 460 treated participants26–31 (Supplementary Table S1). These included 5 add-on studies of children with epilepsy (Lennox-Gastaut syndrome or Dravet syndrome) and 1 monotherapy study of children with intellectual disability and severe behavioral problems. All citations were low risk of bias as per the RoB 2 (Figure 2). No significant increased risk difference of treatment-emergent movement disorders was found at 5, 10, or 20 mg/kg/d among any children receiving cannabidiol, and after subanalysis including only children with epilepsy (Figure 3). Heterogeneity of the primary outcome was moderate and significant at 20 mg/kg/d, nonsignificant at 10 mg/kg/d, and unable to be calculated at 5 mg/kg/d. 24
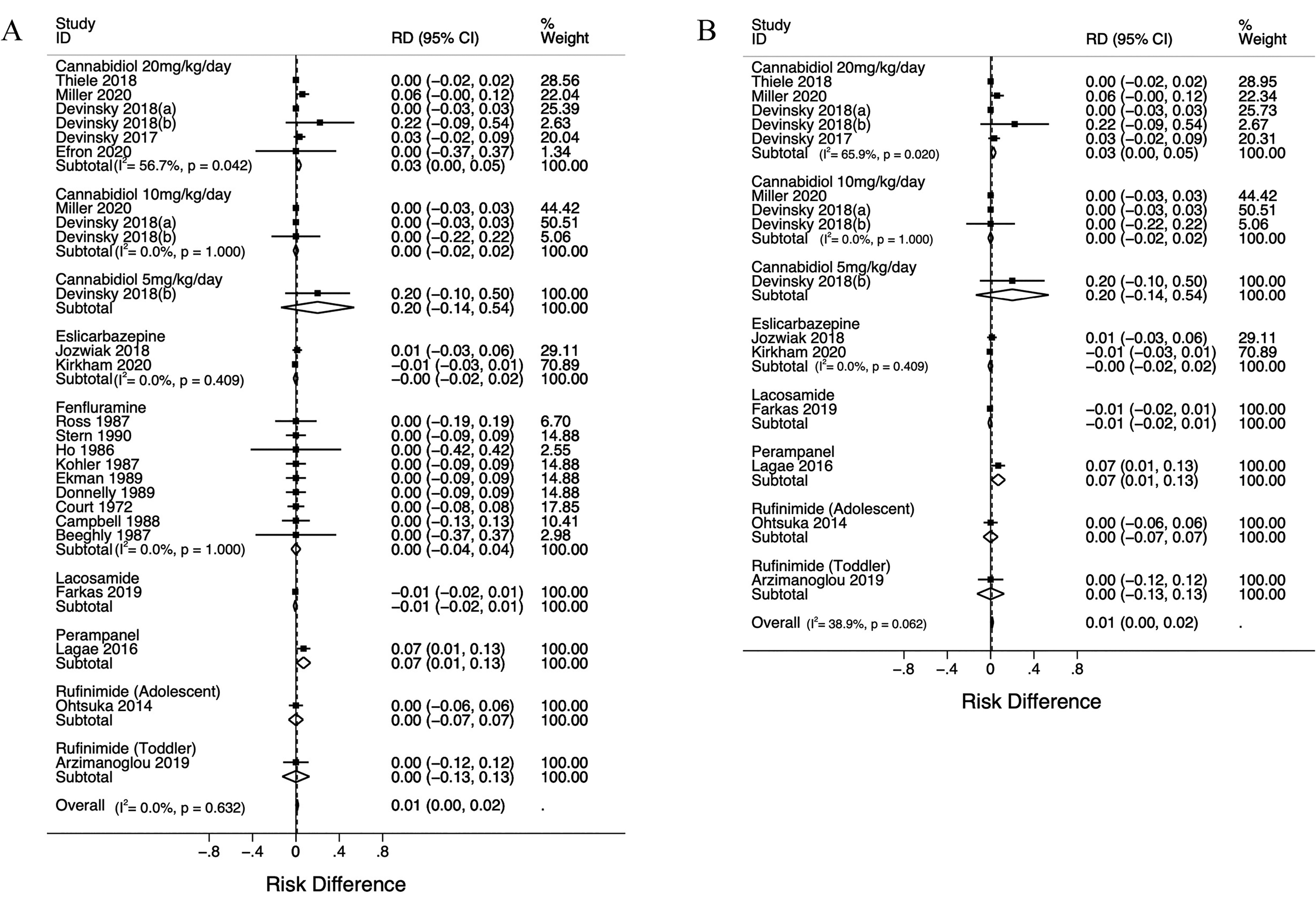
Risk difference (RD) of developing movement disorders among novel antiseizure medications compared to control among (A) all children or among (B) children with epilepsy.
Eslicarbazepine
Two studies of eslicarbazepine add-on therapy for focal-onset seizures were included at 20 to 30 mg/kg/d, including a total of 217 participants with focal-onset seizures32,33 (Supplementary Table S1). One citation was low risk of bias while there were some concerns with the other (Figure 2). No significant increased risk difference of treatment-emergent movement disorders was found (Figure 3).
Fenfluramine
Nine studies of fenfluramine were included in meta-analysis describing the use of 0.5 to 2.0 mg/kg/d among participants with autism spectrum disorder and/or attention-deficit hyperactivity disorder, including a total of 142 participants.34–42 No studies including children with epilepsy were found that met inclusion criteria. The most recent citation was published in 1990. Five citations had high risk of bias, there were some concerns in 3, and 1 was low risk as per the RoB2 (Figure 2); the latter were all high risk for bias in selection of the reported result No significant increased risk difference of treatment-emergent movement disorders was found (Figure 3). No included trial of fenfluramine treated more than 20 participants.
Lacosamide
One study was found matching our inclusion criteria. 43 This was a trial of lacosamide at 6 to 12 mg/kg/d for 12 weeks including 171 children with focal seizures. No significant increased risk difference of treatment-emergent movement disorders was found.
Perampanel
One study was found in which perampanel at 8 to 12 mg/d was given to 85 adolescents with partial onset seizures. 44 There was a significant increased risk difference of treatment-emergent movement disorders among participants treated with perampanel (0.07, 95% CI 0.01-0.13).
Rufinamide
We found 2 studies using rufinamide among participants with Lennox-Gastaut syndrome. The average treated ages were 16.0 and 2.4 years.45,46 Rufinamide given to 28 adolescents at 1000 to 3200 mg/d was compared to placebo, whereas rufinamide given to 25 children at 45 mg/kg/d was compared to any other antiseizure medication at the investigator’s discretion. No significant increased risk difference of treatment-emergent movement disorders was found.
Discussion
We present the first systematic review of novel antiseizure medications with focus on treatment-emergent movement disorder risk in a pediatric population. We also provide an up-to-date listing of all randomized controlled trials in which novel antiseizure medications were used in pediatrics and in which the incidence of any adverse events are exhaustively and quantitatively reported.
Of the drugs investigated in this review, only perampanel was associated with a significant increased risk of movement disorders compared to placebo (95% CI 1-13 percentile increase). Most abnormal movements reported were ataxia or tremor. Notably, only 1 randomized controlled trial exists investigating perampanel that met our inclusion criteria. The present meta-analysis assessed 9 different drugs, and so correcting for multiple hypothesis testing renders the perampanel finding not statistically significant. The clinical significance of increased movement disorder risk secondary to perampanel use in children will be better understood when more controlled data are made available. It is notable that an open-label trial of perampanel in children with focal seizures also found a 6.7% incidence of movement disorders, in agreement with the randomized controlled trial data. 47
Treatment emergent movement disorder risk reported in placebo-controlled trials is generally consistent with safety data reported in longer-term open label trials for cannabidiol,48,49 eslicarbazepine, 32 fenfluramine, 50 perampanel, 47 and rufinamide.51,52 An open-label trial of lacosamide reported a total movement disorder prevalence of 10.6%, which is discordant with the prevalence of 0% reported in the randomized controlled trial. 53 The open-label lacosamide trial was performed with the same strength and a comparable age group, but with fewer children who were observed for a shorter period. The reason for the discrepancy is unclear but could be related to different standards in reporting adverse events. A recent case series has also demonstrated a dose-dependent relationship between lacosamide administration and dyskinesias of the face and/or upper extremity in children with histories suggestive of opercular dysfunction. 54 Two modern randomized controlled trials of fenfluramine in Dravet syndrome have been reported, though neither reports the incidence of treatment-emergent adverse events occurring in less than 10% of children.55,56 Brivaracetam was approved for use in pediatric populations based on a randomized controlled trial including participants as young as 16 years and data showing similar pharmacologic properties in pediatrics as in adults. These data are supported by an open-label trial wherein 149 children with focal seizures or generalized tonic-clonic seizures were treated for 2 years and reported no movement disorders, though adverse events were only reported if they had an incidence of >10% or >2% if considered drug-related. 57 Postmarketing surveillance data also exist for the included drugs, but interpretation of these with respect to movement disorder risk is challenging as reporting of adverse events is typically voluntary and from a population of uncertain size.
This review is the first of its kind to systematically assess the risk of movement disorders associated with novel antiseizure medications in pediatric populations. It is, however, limited by study type and fulsomeness of reporting in the primary literature. To maintain high standards of evidence in the present review, we excluded observational studies such as open-label extensions of included studies and postmarketing safety surveillance data. We also did not include manuscripts written in languages other than English; this may have resulted in bias toward including publications with positive outcomes. 58 Patient-level data were not available to us for larger trials, and so it is possible that multiple types of abnormal movements may have originated from a single patient. As such, we reported the number of abnormal movements rather than number of affected participants. Our analysis did include trials with very low sample sizes, for which imbalances in randomization could affect the risk of reported adverse events. In the case of fenfluramine, which would be most affected by this, outcomes were generally consistent. Additionally, large open-label studies of fenfluramine in children with Dravet syndrome were also consistent with the randomized controlled trial findings. 50 Additionally, we did not include in meta-analysis any studies that did not exhaustively report treatment-emergent adverse events; this led to the exclusion of both modern randomized controlled trials investigating fenfluramine in children. Pooled analyses of otherwise unpublished randomized controlled trials describing the use of novel antiseizure medications in children were also excluded as sufficient information from the study sponsors could not be obtained to perform risk-of-bias assessment.
Our analysis was unable to ascertain the effect of various factors that could contribute to risk of treatment-emergent adverse events such as participant age, number and type of concomitant antiseizure medications, epilepsy syndrome or seizure etiology, age, risk of bias, and quality of adverse drug event content because of the limited number of included studies. We performed a subanalysis by which all nonepilepsy indications for the included antiseizure medications were excluded; no meaningful difference was observed. We also separated quantitative analysis for different drugs, different drug strengths, and substantially different participant age groups. The result is a limited quantitative understanding of how the above factors influence movement disorder risk secondary to antiseizure medication use in children, but still provides an important first step in understanding their relationship. Future analyses may be able to ascertain further risk factors for movement disorder development in children using antiseizure medications when more data are available, and particularly if the labeled indications for these antiseizure medications expands to pathophysiologies with different underlying risks of developing treatment-induced movement disorders.
The amount of safety information from randomized double-blind controlled trials describing the safety of each novel antiseizure medication in children is sparse and often without rigorous assessment of drug safety. Methods of identifying adverse events were often detailed, but assessment of causality was almost exclusively relegated to clinical judgment without the use of standardized methods such as the Naranjo 59 or the WHO-UMC scales. 60 Assessment of adverse event preventability was not performed in any included citation. The use of reproducible methods for assessing causality and preventability of adverse events would allow us to generalize the safety of interventions more accurately.
Conclusion
Using a rigorous study design, we found that perampanel was the only novel antiseizure medications in our set associated with more movement disorders than placebo though only 1 trial of perampanel was found. No increased risk of movement disorders was found in association with cannabidiol, eslicarbazepine, fenfluramine, lacosamide, or rufinamide. No conclusion could be made regarding brivaracetam because of no randomized controlled trial meeting inclusion criteria. We maintained rigorous standards of evidence by not including observational data, though clinical reliance on these reports would be less if randomized controlled trials were powered for safety rather than efficacy. We have also identified multiple problems in existing literature including the assessment of adverse events and transparency of findings. From this, we advocate for improved monitoring of movement disorders in children treated with antiseizure medications by powering studies for safety, including standardized assessment of causality, and by including analyses of adverse event preventability.
Footnotes
Author Contributions
All authors contributed to the study design, interpretation of data, and gave final approval of the manuscript. DP and CB contributed to conception of the study. DP, JY, and MvD contributed to acquisition of data. DP, JY, and JS contributed to analysis of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethics committee approval was not sought for this 73 study as per the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
