Abstract
To expand current knowledge, we examined the safety and tolerability of subcutaneous interferon β-1a in patients with pediatric-onset multiple sclerosis. Records from 307 patients who had received at least 1 injection of subcutaneous interferon β-1a for demyelinating events when aged younger than 18 years were reviewed. Overall, 168 (54.7%) patients had at least 1 prespecified medical event related to or under close monitoring with subcutaneous interferon β-1a or specific to pediatric patients, 184 (59.9%) had nonserious medical events related to treatment or of unknown causality, and 12 (3.9%) had serious medical events irrespective of causality. The most common laboratory abnormalities were increased alanine (74/195; 37.9%) and aspartate aminotransferase levels (59/194; 30.4%). Annualized relapse rates were 1.79 before treatment and 0.47 during treatment. In conclusion, adult doses of subcutaneous interferon β-1a (44 and 22 μg, 3 times weekly) were well tolerated in pediatric patients and were associated with reduced relapse rates.
The onset of multiple sclerosis most frequently occurs at 20 to 40 years of age. 1 However, it is estimated that 3% to 5% of patients experience their first symptoms before the age of 16 years, 2 –5 and less than 1% before the age of 10 years. 4,6 In 98% of pediatric patients with multiple sclerosis, the disease initially follows a relapsing–remitting course. 7 Studies suggest that the onset of secondary progressive disease occurs later after the first attack in pediatric patients than in patients with adult-onset disease. 4,7,8 However, patients with childhood-onset multiple sclerosis reach the secondary progressive phase and have irreversible disability at younger ages than patients with adult-onset multiple sclerosis. 4,7,8
In adults with multiple sclerosis, disease-modifying drugs are recommended soon after diagnosis or at the first attack (provided clinical, laboratory, and magnetic resonance imaging features are consistent with multiple sclerosis), as clinical trials have demonstrated the benefits of early treatment. 9 –14 However, these trials did not enroll patients younger than 18 years. Given the potential severe long-term consequences of pediatric multiple sclerosis, the current recommendations are that children and adolescents with multiple sclerosis should be treated with disease-modifying drugs, and that treatment, as for adults, should be started as early as possible in the disease course, and not delayed until adulthood. 15 –18
Pediatric data on the use of interferon-β therapies, although available, are limited due to small sample sizes. Small cohort studies and isolated case reports indicate that interferon-β therapies are well tolerated and appear to be effective in the treatment of pediatric multiple sclerosis. 19 –28 To capture more comprehensive experience with subcutaneous interferon β-1a in a large, multinational pediatric multiple sclerosis population, this study reviewed the safety and tolerability of subcutaneous interferon β-1a in children and adolescents with demyelinating events, based on retrospective data from medical records. Although the study was not designed to evaluate efficacy, additional data were collected to explore the impact of treatment on clinical relapses.
Methods
Study Design
This was a retrospective, phase IV study (ClinicalTrials.gov identifier: NCT01207648) conducted at 18 centers, in the United States (6 sites); Italy (6 sites); and Russia, Argentina, Canada, France, Tunisia, and Venezuela (1 site each). Data were retrieved from medical health care records of patients evaluated between 1997 and 2009. The observation period for an individual patient began with the first medical record available on site and ended on December 31, 2009, or when the patient was lost to follow-up, whichever occurred first.
The study was conducted in accordance with the Declaration of Helsinki, Guidelines for Good Pharmacoepidemiology Practices, International Conference on Harmonisation Guidelines for Good Clinical Practice, and local regulations. An Institutional Review Board or Independent Ethics Committee approved the protocol as required by local laws. All patient data were recorded anonymously.
Study Population
Patients were eligible if they had received at least 1 injection of subcutaneous interferon β-1a for demyelinating events before the age of 18 years. Treatment with subcutaneous interferon β-1a must have been initiated prior to June 2009 to allow at least 6 months of possible observation for each patient. To minimize the potential bias inherent in retrospective patient selection, the aim was to assess all pediatric patients treated with subcutaneous interferon β-1a at each center. Therefore, for centers to participate in the study, it was essential that medical records were available for all patients treated with immunomodulatory drugs, including patients who died, became adults and moved to adult clinics, or were lost to follow-up.
Outcomes
The primary focus of the study was the safety and tolerability of subcutaneous interferon β-1a in a population with pediatric-onset multiple sclerosis. Data were collected on prespecified medical events of special interest, regardless of severity and causality. Prespecified medical events comprised medical events known to be related to subcutaneous interferon β-1a, such as injection-site reactions, influenza-like symptoms, blood cell disorders, hepatic disorders, allergic reactions, and thyroid disorders; medical events likely to occur in a pediatric population with or without multiple sclerosis, such as epilepsy and convulsive disorders, and bone/epiphyseal and cartilage disorders; and medical events under close monitoring with subcutaneous interferon β-1a, such as malignancies, serious infections, and autoimmune diseases.
Nonserious medical events considered by the investigator as related to treatment were recorded; nonserious medical events of unknown causality were collected by some sites and included in the analysis. Serious medical events, irrespective of causality, were also collected. Medical events that were listed as prespecified medical events could be counted again as nonserious and serious medical events. Only medical events that occurred after the initiation of subcutaneous interferon β-1a were eligible for analysis, including medical events that occurred after discontinuation of subcutaneous interferon β-1a therapy. Causality of medical events was assigned retrospectively only if well documented in the medical records. Laboratory parameters collected from the time of subcutaneous interferon β-1a initiation to the end of the observation period included total white blood cell count, differential hematology count, serum aspartate aminotransferase and alanine aminotransferase levels, and thyroid function (thyroid-stimulating hormone concentration).
Efficacy outcomes included medically confirmed clinical relapses, defined as the emergence of new neurologic symptoms that occurred more than 30 days after a previous attack and persisted for at least 24 hours in the absence of known intercurrent illness. All records reporting clinical relapses were reviewed by members of the study steering committee to ensure relapses occurring in the pretreatment and treatment phases were evaluated reliably.
Statistical Analysis
Safety and tolerability data were analyzed descriptively for all patients in the study (total analysis set) and in 2 subgroups based on age at subcutaneous interferon β-1a initiation: children (aged <12 years) and adolescents (aged 12 to <18 years). Efficacy outcomes were evaluated only in patients with a final diagnosis of multiple sclerosis (multiple sclerosis analysis set). The annualized rate of medically confirmed clinical relapses was analyzed by Poisson regression with no covariates. For pretreatment attack rates, as every patient is considered to have 1 or more attacks prior to starting subcutaneous interferon β-1a treatment, the data were transformed by deducting 1 clinical attack for each patient, fitting a Poisson regression based on the transformed data, and retransforming the estimates of the Poisson regression by adding 1. For annualized relapse rates during subcutaneous interferon β-1a treatment, and from the end of subcutaneous interferon β-1a treatment until the end of observation, the log of the time on observation was used as the offset variable. A sensitivity analysis was performed by evaluating the annualized rate of medically confirmed clinical relapses in patients who were observed for at least 6 months before and after subcutaneous interferon β-1a initiation. The median times to the first medically confirmed clinical relapse, after the first demyelinating event and after starting subcutaneous interferon β-1a therapy, were evaluated using Kaplan–Meier cumulative probability estimates. All comparisons were purely exploratory and descriptive. Interpretation of results was based on point estimates and the corresponding 95% confidence intervals, not P values. All statistical analyses were performed using SAS version 9.1 (or higher) software (SAS Institute Inc, Cary, NC, USA).
A sample size calculation was not performed for this study as all patients who met the eligibility criteria at the participating centers were to be included. It was expected that data from approximately 250 patients would be available for analysis.
Results
Patients
A total of 307 patients were included in the total analysis set (United States, n = 139; Italy, n = 47; Russia, n = 38; Argentina, n = 33; France, n = 23; Canada, n = 21; Tunisia and Venezuela, n = 3 each). Data from 2 patients were collected but not included in the total analysis set as it could not be confirmed that these patients were aged younger than 18 years at the start of subcutaneous interferon β-1a treatment. Of the 307 patients, 298 had a final diagnosis of multiple sclerosis and were included in the multiple sclerosis analysis set. All participating centers were able to produce the required information from the first medical record available, as per the trial protocol.
Demographics and disease characteristics for the total analysis set are shown in Table 1. The median (range) observation time for the total analysis set was 3.7 (0.4–16.0) years. In the multiple sclerosis analysis set, the median (range) observation time was 1.13 years (1 day to 13.8 years) prior to subcutaneous interferon β-1a initiation and 2.03 (0–12.5) years after starting subcutaneous interferon β-1a.
Demographics and Disease Characteristics (Total Analysis Set).
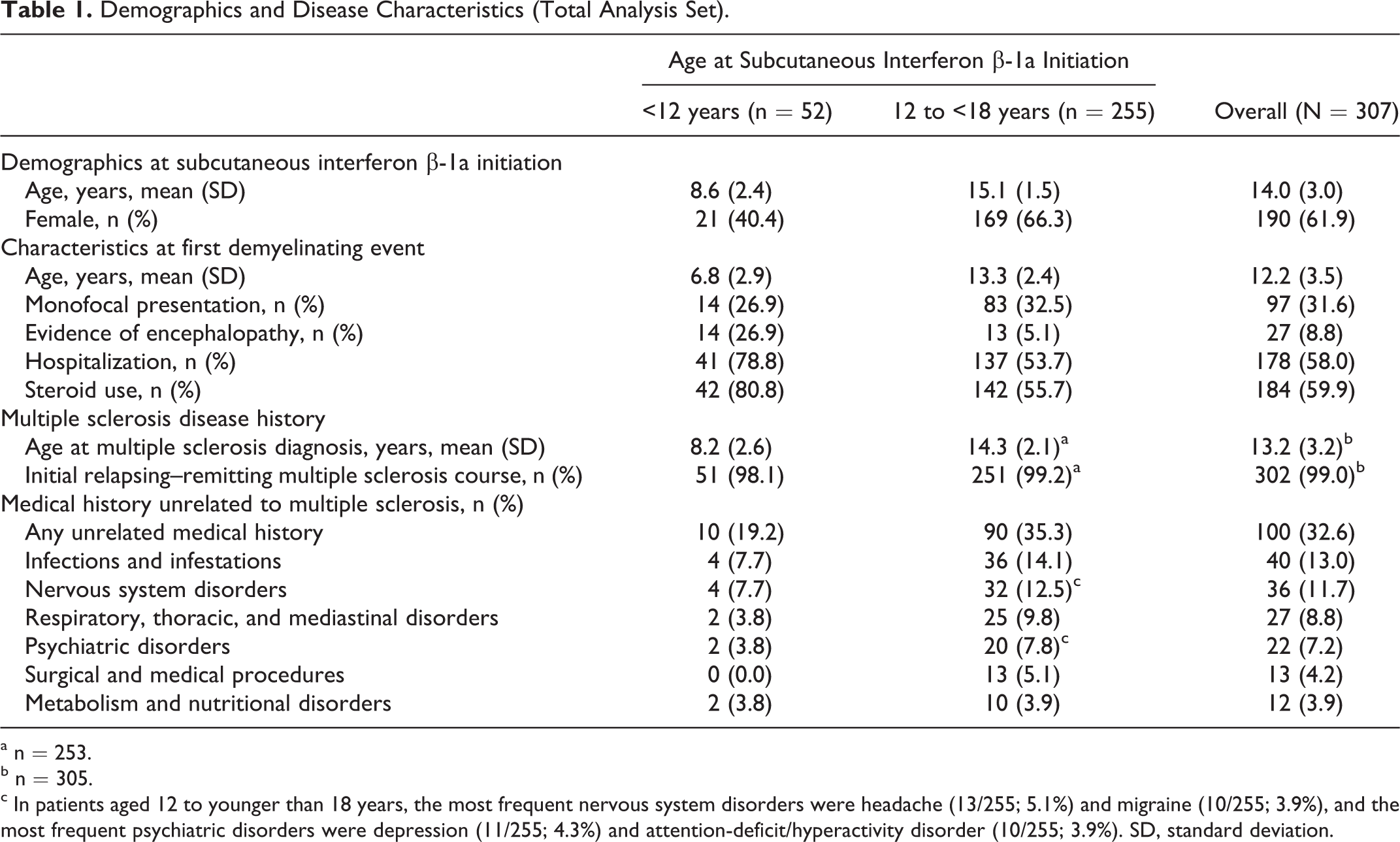
an = 253.
bn = 305.
cIn patients aged 12 to younger than 18 years, the most frequent nervous system disorders were headache (13/255; 5.1%) and migraine (10/255; 3.9%), and the most frequent psychiatric disorders were depression (11/255; 4.3%) and attention-deficit/hyperactivity disorder (10/255; 3.9%). SD, standard deviation.
Exposure to Subcutaneous Interferon β-1a
At treatment initiation, subcutaneous interferon β-1a doses were 44 μg 3 times weekly, 145 patients (47.2%) including 10 patients aged younger than 12 years (10/52; 19.5% of the children subgroup); 22 μg 3 times weekly, 117 patients (38.1%) including 32 patients aged younger than 12 years (32/52; 61.5%); other, 37 patients (12.1%) including 10 patients aged younger than 12 years (10/52, 19.2%); and unknown, 8 patients (2.6%; none aged <12 years). During the observation period, the mean (standard deviation) time on subcutaneous interferon β-1a therapy in the total analysis set was 2.12 (2.11) years; median 1.3 years (range, 1 day to 12.5 years). Overall, 82.7% (254/307) of patients were treated for at least 6 months and 59.3% (182/307) were treated for at least 12 months. A total of 99 patients permanently discontinued subcutaneous interferon β-1a therapy during the observation period. Reasons for discontinuation were clinical relapse (n = 31), other medical event (n = 26), patient decision (n = 15), magnetic resonance imaging activity without a clinical relapse (n = 9), and other reasons (n = 18).
Safety and Tolerability
Overall, 61.9% (190/307) of patients had at least 1 medical event. In total, 54.7% (168/307) of patients had at least 1 prespecified medical event (Table 2). The most frequently reported prespecified medical events were injection-site reactions (27.7%), influenza-like symptoms (24.4%), and hepatic disorders (14.3%). Individual prespecified medical events occurring in over 10% of patients were influenza-like illness (24.4%), injection-site erythema (15.6%), and injection-site pain (11.1%). The proportion and nature of prespecified medical events were similar in the 2 age groups, except that patients aged younger than 12 years, compared with those aged 12 to younger than 18 years, had a slightly higher incidence of injection-site erythema (19.2% vs 14.9%) and a lower incidence of injection-site pain (3.8% vs 12.5%).
Prespecified Medical Events of Special Interest (Total Analysis Set).
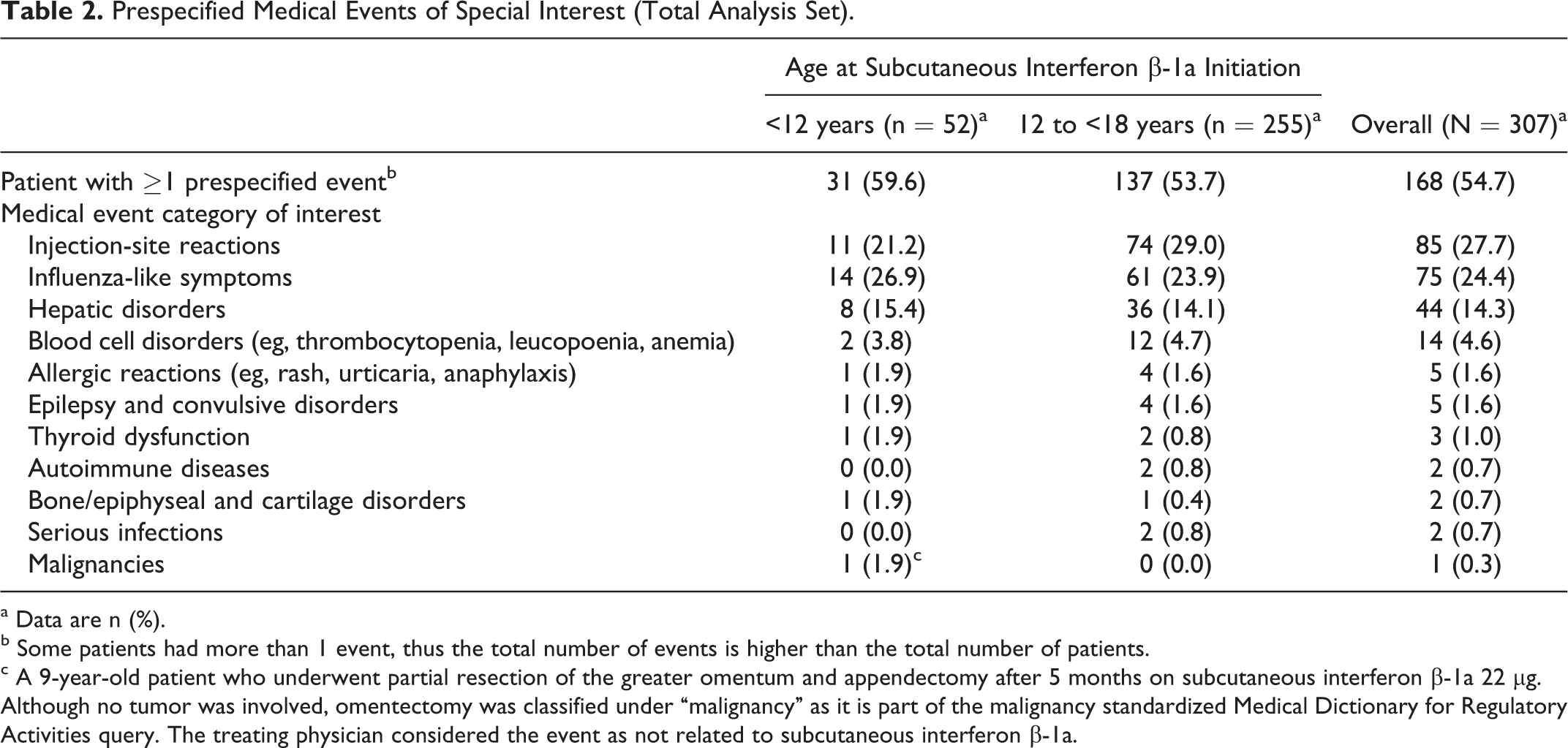
aData are n (%).
bSome patients had more than 1 event, thus the total number of events is higher than the total number of patients.
cA 9-year-old patient who underwent partial resection of the greater omentum and appendectomy after 5 months on subcutaneous interferon β-1a 22 μg. Although no tumor was involved, omentectomy was classified under “malignancy” as it is part of the malignancy standardized Medical Dictionary for Regulatory Activities query. The treating physician considered the event as not related to subcutaneous interferon β-1a.
Nonserious medical events considered by the investigator to be related to subcutaneous interferon β-1a treatment or of unknown causality occurred in 59.9% (184/307) of patients. Excluding prespecified medical events, the most frequent nonserious medical events reported in at least 1% of patients were headache (6.2%), pyrexia (2.9%), fatigue (1.6%), depression (1.6%), nausea (1.6%), myalgia (1.3%), chills (1.3%), and upper abdominal pain (1.3%). Analysis by age group revealed slight differences in patients aged younger than 12 years compared with those aged 12 to younger than 18 years: pyrexia (5.8% vs 2.4%) and upper abdominal pain (3.8% vs 0.8%) were reported more frequently, while headache (3.8% vs 6.7%) was reported less frequently. Depression was reported only in patients aged 12 to younger than 18 years (2.0%).
There were no deaths during the study period. A total of 18 serious medical events were reported in 12 (3.9%) patients. Serious medical events occurring in patients aged 12 to younger than 18 years were injection-site injury, injection-site necrosis, and injection-site cellulitis (1 patient); convulsion, auditory hallucination, and suicidal behavior (1 patient); and, in 1 patient each, irritability, autoimmune hepatitis, cholelithiasis, idiopathic thrombocytopenic purpura, abnormal liver function test, suicidal ideation, anaphylactic reaction, and cellulitis. Only 2 patients aged younger than 12 years had serious medical events: hypersensitivity, appendectomy, and omentectomy (1 patient), and epilepsy (1 patient).
Medical events leading to dose reduction were reported in 8 (2.6%) patients; the only medical event leading to dose reduction in more than 1 patient was increased hepatic enzyme levels (2 patients). A total of 36 (11.7%) patients permanently discontinued treatment due to medical events. The most frequent events leading to treatment discontinuation were injection-site pain (2.9%), influenza-like illness (2.0%), injection-site erythema (1.3%), and abnormal liver function test (1.3%).
The most common laboratory abnormalities were increased levels of alanine aminotransferase (74/195 patients; 37.9%) and aspartate aminotransferase (59/194; 30.4%). A similar proportion of patients aged younger than 12 years and 12 to younger than 18 years had elevated levels of alanine aminotransferase (15/38 [39.5%] and 59/157 [37.6%], respectively) and aspartate aminotransferase (11/38 [28.9%] and 48/156 [30.8%], respectively). No clinically relevant differences in laboratory values were observed between the 2 age groups.
Impact of Treatment on Clinical Relapses
For the multiple sclerosis analysis set population, the annualized rate of medically confirmed clinical relapses was 1.79 in the period prior to subcutaneous interferon β-1a initiation (Figure 1). During subcutaneous interferon β-1a treatment, the annualized relapse rate decreased to 0.47, and, in the subgroup of patients who discontinued subcutaneous interferon β-1a treatment, increased to 0.77 from the end of subcutaneous interferon β-1a treatment until the end of observation. For patients who stopped subcutaneous interferon β-1 and switched to another disease-modifying drug, the annualized relapse rate was 1.91 prior to subcutaneous interferon β-1a treatment, 0.98 during treatment, and 0.87 after stopping subcutaneous interferon β-1a. For patients who stopped all disease-modifying drug treatment, the corresponding rates were 1.85 prior to subcutaneous interferon β-1a, 0.39 during subcutaneous interferon β-1a treatment, and 0.37 after cessation of treatment. Similar results were observed when considering only the subgroup of patients with at least 6 months of observation before and after subcutaneous interferon β-1a initiation (Figure 1).
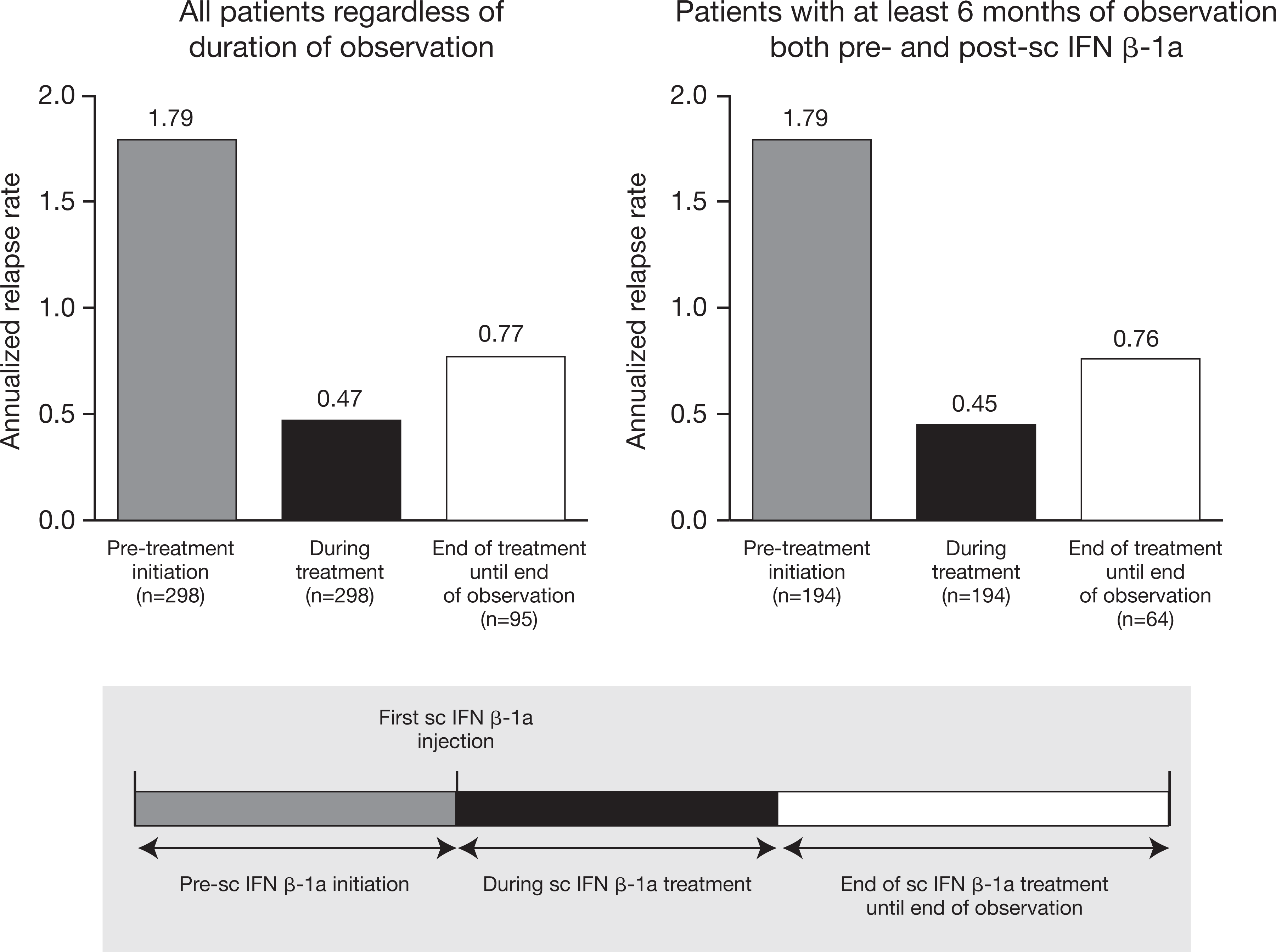
Medically confirmed clinical relapses (multiple sclerosis analysis set). As each patient had at least 1 clinical relapse prior to subcut-aneous (sc) interferon (IFN) β-1a initiation, annualized relapse rates in the pretreatment period were calculated by subtracting 1 relapse per patient, fitting the transformed data using the Poisson model, then adding back 1 relapse to the estimates.
The median (95% confidence interval) time to first medically confirmed clinical relapse after the first demyelinating event and prior to subcutaneous interferon β-1a initiation in the multiple sclerosis analysis set was 8.1 (6.3–9.1) months. Considering subgroups of patients by treatment status at the end of observation, the median (95% confidence interval) time to first medically confirmed clinical relapse after the first demyelinating event was 7.8 (6.3–9.7) months in patients with ongoing subcutaneous interferon β-1a treatment, 8.1 (4.2–12.3) months in patients who switched to another disease-modifying drug, and 8.4 (5.6–12.2) months in patients who stopped all disease-modifying drug treatment. After starting subcutaneous interferon β-1a therapy, the median (95% confidence interval) time to the first medically-confirmed clinical relapse was 19.5 (14.5–27.2) months (Figure 2). For patients receiving ongoing subcutaneous interferon β-1a therapy at the end of the study (n =203), the median (95% confidence interval) time to the first medically confirmed clinical relapse after subcutaneous interferon β-1a initiation was 26.4 (18.4–37.0) months, compared with 10.0 (7.6–14.3) months for patients who stopped subcutaneous interferon β-1a and switched to another disease-modifying drug (n =60), and 27.2 (12.2 to not estimable) months for those who stopped subcutaneous interferon β-1a and did not switch to another disease-modifying drug (n =35).
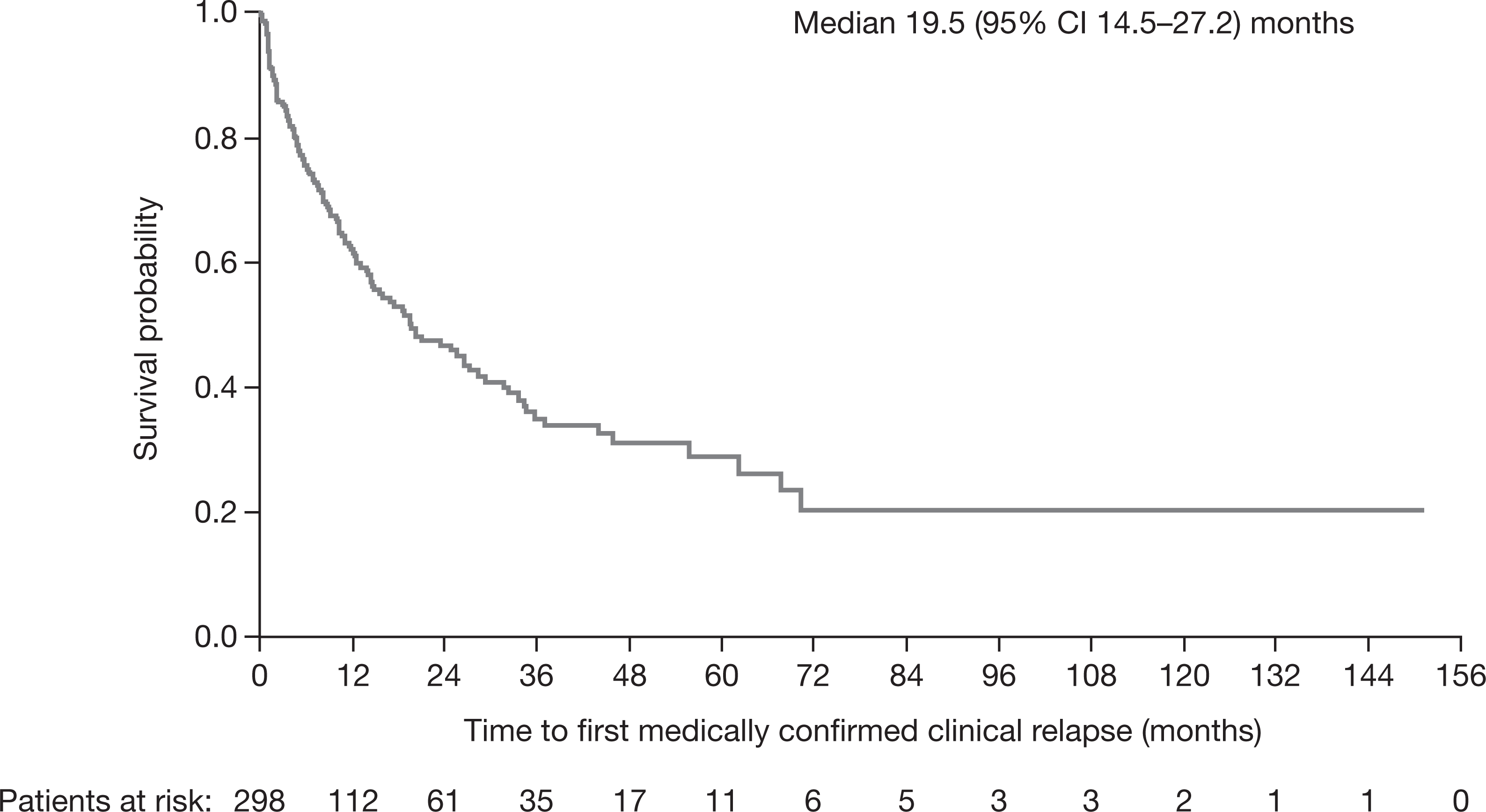
Time to first medically-confirmed clinical relapse from initiation of subcutaneous interferon β-1a (multiple sclerosis analysis set). CI, confidence interval.
Discussion
To date, this is the largest study assessing the safety, tolerability, and potential efficacy of subcutaneous interferon β-1a treatment in children and adolescents with multiple sclerosis. Adult doses of subcutaneous interferon β-1a (44 and 22 μg, 3 times weekly) were well tolerated in both children and adolescents with multiple sclerosis, with neither new nor unexpected adverse drug reactions. Generally, medical events were nonserious and manageable, and did not require dose reduction or treatment discontinuation. The frequency and type of medical events were consistent with those expected in adult patients with multiple sclerosis treated with subcutaneous interferon β-1a. 29,30 Limited data are available on the use of disease-modifying drugs to treat patients aged younger than 12 years: our study showed that there were no major differences in medical events observed in children (<12 years) compared with adolescents (12 to <18 years).
A limitation of this study is that the collection of laboratory data could not be standardized due to the retrospective nature of the study design. Regularly recorded laboratory values were available for some but not all patients, making the data difficult to interpret, particularly for the group of patients aged younger than 12 years, for which data were available for fewer patients. However, from the available data, the identified laboratory abnormalities were similar across the 2 age groups. Elevations in alanine aminotransferase and aspartate aminotransferase levels, which were the most common abnormal laboratory findings in this study, are known to be associated with interferon therapy and have been reported in up to approximately 60% of adults during the first 12 months of subcutaneous interferon β-1a therapy. 31 In fact, it is recommended that, in the absence of clinical symptoms, serum alanine aminotransferase levels are monitored prior to the start of therapy; at months 1, 3, and 6 during therapy; and periodically thereafter. 31
The impact of subcutaneous interferon β-1a treatment on the frequency of clinical relapses in this study population should be interpreted with caution as there was no control group and the natural history of multiple sclerosis tends toward a reduction in the frequency of relapses over time. 8,32 The annualized relapse rate after initiation of subcutaneous interferon β-1a treatment was lower than in the period prior to subcutaneous interferon β-1a initiation, which is in accordance with observations from smaller cohort studies of subcutaneous interferon β-1a in pediatric patients with multiple sclerosis. 23,24 The median time to first medically confirmed clinical relapse after starting subcutaneous interferon β-1a therapy was longer than that after the first demyelinating event. Patients who were receiving ongoing subcutaneous interferon β-1a therapy at study end, or had stopped subcutaneous interferon β-1a treatment and not switched to another disease-modifying drug, had a longer median time to the first medically confirmed clinical relapse after subcutaneous interferon β-1a initiation, compared with those who had stopped subcutaneous interferon β-1a treatment and switched to another disease-modifying drug. This suggests that patients who are doing poorly on subcutaneous interferon β-1a treatment tend to switch to another disease-modifying drug, whereas those who are doing well tend to either remain on subcutaneous interferon β-1a or cease disease-modifying drug treatment entirely.
Given the retrospective study design, there was variability among patients with respect to concurrent medications, comorbidities, age at the time of subcutaneous interferon β-1a initiation, and dosage and duration of subcutaneous interferon β-1a treatment. Furthermore, as this was a multinational observational study, regional variation in treatment practices might have introduced bias regarding data collection, completeness of data, and patient selection. Therefore, the findings of the present study should be considered in light of the inherent limitations of retrospective analyses and require confirmation in prospective, randomized, controlled studies.
Despite the challenges inherent in a retrospective study, data from this relatively large sample of patients with pediatric-onset multiple sclerosis indicate that both children and adolescents tolerate adult doses of subcutaneous interferon β-1a without raising new safety concerns. Furthermore, treatment in this subpopulation of patients with multiple sclerosis appears to be associated with a reduction in relapse rate.
Footnotes
Acknowledgments
The authors thank Ulrich Freudensprung for his valuable input into study design; and Eric Henninger, Delphine Issard, Shalini Jayasekar, Cedric Marchand, Jocelin McDonough, Claire McIlroy, and Daniel Mikol for their help in study implementation. The authors also thank Sharon Gladwin, PhD, of Caudex Medical (Oxford, UK) for assistance in the preparation of this article.
Author Contributions
SNT and DP drafted and revised the manuscript for content, developed study design and concept, performed analysis and interpretation and acquisition of data, supervised and coordinated the study. BB drafted and revised the manuscript for content, and performed analysis and interpretation and acquisition of data. LBK drafted and revised the manuscript for content and performed acquisition of data. AB drafted and revised the manuscript for content, performed acquisition of data, and supervised and coordinated the study. MM drafted and revised the manuscript for content, performed analysis and interpretation of data, and performed statistical analysis. LL drafted and revised the manuscript for content and performed analysis and interpretation of data. SR drafted and revised the manuscript for content, developed study design and concept, and performed analysis and interpretation of data. EVC drafted and revised the manuscript for content, performed analysis and interpretation of data, and supervised and coordinated the study. MSM drafted and revised the manuscript for content and performed analysis and interpretation of data. AG drafted and revised the manuscript for content, performed analysis and interpretation and acquisition of data, and supervised and coordinated the study. Coinvestigators (REPLAY Study Group) include Marco Capobianco, MD (Centro di Riferimento Regionale Sclerosi Multipla, Azienda Ospedaliero Universitaria S. Luigi Gonzaga, Orbassano, Italy, principal investigator); Tanuja Chitnis, MD (Partners Pediatric MS Center at the Massachusetts General Hospital for Children, Boston, MA, USA, principal investigator); Kumaran Deiva, MD, PhD (Assistance Publique-Hôpitaux de Paris, Hôpitaux Universitaires Paris-Sud, Pediatric Neurology Department, and Université Paris Sud, National Referral Center for Neuro-Inflammatory Diseases in Children, Le Kremlin-Bicêtre, France, principal investigator); Neziha Gouider-Khouja, MD (Department of Pediatric Neurology, National Institute of Neurology, Tunis, Tunisia, principal investigator); Maria Giovanna Marrosu (Centro Sclerosi Multipla, Ospedale Binaghi, Università degli Studi di Cagliari, Cagliari, Italy, principal investigator); Vittorio Martinelli, MD (San Raffaele Scientific Institute, MS Centre, Milano, Italy, principal investigator); Jayne Ness, MD, PhD (Division of Pediatric Neurology, UAB Center for Pediatric Onset Demyelinating Disease, Children’s Hospital of Alabama, Birmingham, AL, USA, principal investigator); Marc C. Patterson, MD, FRACP (Mayo Clinic Children’s Center, Rochester, MN, USA, principal investigator); Francesco Patti, MD (Azienda Ospedaliero Universitaria, Policlinico-Vittorio Emanuele, Università di Catania, Cantania, Italy, principal investigator); Joaquin Pena, MD, PhD (Hopital Clinico de Maraicabo Neurología Pediátrica, Maracaibo, Venezuela, principal investigator); Carlo Pozzilli, MD, PhD (Multiple Sclerosis Centre, S. Andrea Hospital, University of Rome La Sapienza, Rome, Italy, principal investigator); Emmanuelle Waubant, MD, PhD (UCSF Regional Pediatric MS Center, San Francisco, CA, USA, principal investigator); Bianca Weinstock-Guttman (Pediatric MS Center of the Jacobs Neurological Institute, Buffalo, NY, USA, principal investigator); and Damiano Baroncini, MD (Centro Studi Sclerosi Multipla, Ospedale di Gallarate, Gallarate, Italy, site investigator)
Declaration of Conflicting Interests
SNT has received honoraria for speaking from Biogen Idec and Merck Serono; and for participation in an advisory group for Biogen Idec, Merck Serono, and Teva. BB has received honoraria from Biogen Idec, Merck Serono, Bayer, and Schering; and for participation in an advisory group for Biogen Idec and Merck Serono. DP has received honoraria or support for travel and accommodation and congress attendance from Bayer-Schering, Biogen Idec, Merck Serono, and Teva. LBK has received personal compensation for activities as a speaker, consultant, and/or participant on an advisory board from Teva Neuroscience, Biogen Idec, EMD Serono, the Multiple Sclerosis Association of America, Bayer Pharmaceuticals, Guidepoint Global, Pfizer, Axon Advisors, and Sanofi-Aventis; royalty or license fees or contractual rights payments from Abbott Laboratories, Genzyme Corporation, Health Professions Conferencing Corp., Bristol-Myers Squibb, Johnson and Johnson, MedImmune, Novartis, and Roche; grant support from the National Multiple Sclerosis Society; and research support from Serono and Biogen Idec. AB has been a member of advisory boards and participant in clinical trials sponsored by Bayer Schering, Merck Serono, Teva, Novartis, Biogen, Nycomed, Genzyme, and other companies. MM, LL, SR, EVC, and MSM were salaried employees of Merck Serono S.A., Geneva, Switzerland, at the time of the study. AG has received honoraria for speaking from Biogen, Merck Serono, Novartis, and Sanofi-Aventis; for consultancy from Merck Serono, Teva, and Novartis; and support for participation in national and international congresses from Bayer-Schering, Biogen-Dompè, Merck Serono, Novartis, and Sanofi-Aventis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The REPLAY study was funded by Merck Serono S.A., Switzerland, an affiliate of Merck KGaA, Darmstadt, Germany.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki, Guidelines for Good Pharmacoepidemiology Practice, International Conference on Harmonisation Guidelines for Good Clinical Practice, and local regulations. An Institutional Review Board or Independent Ethics Committee approved the protocol as required by local laws. The approving committees were Comité d’Ethique, Institut National de Neurologie (Tunisia); Ethics Committee of the State Budgetary Healthcare Institution “City Clinical Hospital #11” of the Moscow Healthcare Department (Russia); Mayo Clinic Institutional Review Board; University of Alabama at Birmingham Institutional Review Board for Human Use; University of California, San Francisco, Human Research Protection Program Committee on Human Research; Partners Human Research Committee; University of Buffalo, State University of New York; Children & Youth Institutional Review Board, Woman and Children’s Hospital of Buffalo; Office of Research Compliance, Division of Human Subject Protections, Stony Brook University (USA); Sick Kids, The Hospital for Sick Children Research Ethics Board/St Michael’s Research Ethics Board (Canada); Comite Independiente de Etica para Ensayos de Farmacologia Clinica, Fundacion de Estudios Farmacologicos y de Medicamentos (Argentina); Comitato Etico, Azienda Ospedaliera “S. Antonio Abate”; Comitato Etico Indipendente, Azienda Sanitaria Locale di Cagliari; Comitato Etico, Fondazione Centro San Raffaele del Monte Tabor; Comitato Etico AOU “Policlinico-Vittorio Emanuele” Catania, P.O. Policlinico “G. Rodolico”; Comitato Etico, Azienda Ospedaliera S. Andrea; Comitato Etico Interaziendale, Azienda Ospedaliero Universitaria S. Luigi Gonzaga (Italy); and Comité de Etica, Hospital de Especialidades Pediatricas/Fundacion Hospital de Especialdades Pediatricas (Venezuela). As this was a retrospective study involving data retrieved from medical records, informed patient consent was not obtained. Patient data were recorded anonymously.
