Abstract
Purpose:
To evaluate the clinical effectiveness, safety, and catheter durability of venting percutaneous transesophageal gastrostomy (PTEG) in patients with malignant bowel obstruction (MBO) who were not candidates for percutaneous gastrostomy (PG).
Materials and Methods:
This retrospective single-center study included 47 patients with MBO who underwent PTEG between 2018 and 2025. Outcomes included technical success, >72 hours clinical outcomes, paired pre–post analysis of nausea and vomiting severity, changes in antiemetic and opioid use, >72 hours catheter performance, and survival. Time-to-event analyses were performed using Kaplan–Meier estimates for overall survival and catheter event-free survival. Procedure-related adverse events were classified using the CIRSE system.
Results:
Technical success was achieved in all patients and clinical success within 72 hours occurred in 90.9%. Nausea and vomiting prevalence decreased from 87.2% to 8.5% and 4.3%, respectively (P < .001), with parallel reductions in antiemetic (87.2%-8.5%) and opioid use (53.2%-12.8%; P < .001). Catheter dysfunction occurred in 22.7% of patients. Median overall survival was 52.0 days (95% CI: 38.0-245.0), and median catheter event-free survival was 212 days (95% CI: 65-not reached). Mean overall survival was 136.2 days (95% CI: 75.5-196.8), while mean catheter event-free survival was 229.9 days (95% CI: 131.3-328.6). No major adverse events (CIRSE grade ≥3) were observed.
Conclusion:
Venting PTEG demonstrated favorable short- and long-term clinical outcomes in patients with MBO who were unsuitable for PG.
Introduction
Malignant bowel obstruction (MBO) is reported in 5% to 15% of patients with advanced malignancy, and is associated with severe symptoms including nausea, vomiting, abdominal distension, and inability to tolerate oral intake.1,2 Prognosis following MBO is poor and management is primarily palliative, with symptom control and quality of life as central goals.2-4 Consensus guidelines recommend nasogastric tube (NGT) or percutaneous gastrostomy (PG) for gastrointestinal decompression in patients with malignant bowel obstruction managed non-operatively.5-8 Although effective for acute relief, prolonged NGT use is poorly tolerated and associated with reduced quality of life, leading to a preference for PG for longer-term decompression.5,9,10
However, there are clinical scenarios in which PG may not be feasible or may carry a higher procedural risk. 11 Peritoneal carcinomatosis, malignant involvement along the gastrostomy tract, and large-volume or recurrent ascites can impair tract maturation and increase the risk of leakage, infection, tract seeding, and peritonitis; all of which can be highly morbid and reduce the quality of life for the palliative patient.11-15 In these scenarios, patients may be left with limited options beyond prolonged NGT decompression or incomplete symptom control. 2
Percutaneous transesophageal gastrostomy (PTEG) was developed in Japan as an alternative route to PG via the cervical esophagus under ultrasound and fluoroscopic guidance.16-18 Prior studies, including a randomized controlled trial demonstrating improved quality of life compared with NGT and several case series, have established the feasibility and short-term clinical efficacy of PTEG.19-22 More recent nationwide database analyses support an overall favorable safety profile, while noting tube-related complications and early replacement. 23 Despite this growing evidence, most studies emphasize placement outcomes, with limited data on longitudinal tube performance and durability. The present study reports real-world outcomes of venting PTEG in a Western palliative population, focusing on symptom control, catheter durability, and their relevance over the remaining disease course.
Material and Methods
Study Design
This study was approved and waived of informed consent by the Institutional Research Ethics Board (REB #2025_6928) of our institution and adheres to the STROBE Statement and the Declaration of Helsinki. A retrospective, single-center, observational study was designed to evaluate the safety and clinical outcomes of PTEG in patients with contraindications to PG. Consecutive patients who were assessed for PTEG placement between February 2018 and April 2025 were identified through a retrospective review of institutional procedural records and evaluated for inclusion. Inclusion criteria were patients ≥18 years who underwent PTEG at our institution for gastrointestinal decompression and who were deemed poor candidates for PG. Exclusion criteria were patients referred for PTEG in whom the procedure was not attempted, and patients in whom PTEG was indicated solely for feeding purposes.
Baseline Assessment
Baseline assessment included demographic variables, underlying disease, presence and grade of carcinomatosis and ascites and indication for PTEG placement. Symptom assessment prior to PTEG placement included the presence or absence of nausea, vomiting, abdominal pain, aspiration, and the use of antiemetic and opioids. Nausea and vomiting severity were recorded using an ordinal scale ranging from 0 to 3, as commonly applied in retrospective palliative MBO studies to enable standardized symptom grading from clinical documentation.19,24,25 Previous NGT placement and duration of use were recorded when applicable.
PTEG Procedure
All PTEG procedures were performed by board-certified interventional radiologists with 5 to 25 years of experience. Procedures were performed under local anesthesia and titrated conscious sedation. A 22-24 × 40 mm Atlas Gold balloon (BD®, Tempe, AZ, USA) was advanced into the cervical esophagus through the oral cavity under fluoroscopic guidance. Once inflated with iodinated contrast, percutaneous transesophageal access targeting the balloon was performed under ultrasound guidance using a 22-gauge Chiba needle (Cook Medical®, Bloomington, IN, USA). The optimal access site was the left cervical esophagus at the level of the thoracic inlet, targeting the window between the trachea and the left common carotid artery. A trans-thyroidal approach was used when thyroid enlargement precluded a direct transesophageal access. A 0.018-inch microwire (Cook Medical®) was advanced into the balloon lumen, and the balloon-wire unit was advanced into the mid-esophagus under fluoroscopic guidance. The access was then upsized, and a 0.035-inch Amplatz stiff wire (Cook Medical®) was advanced into the stomach. The tract was subsequently dilated, and a multipurpose pigtail catheter (Cook Medical®), ranging from 12 to 16 French and 40 to 60 cm in length, was advanced over the wire into the stomach. Technical success was defined as successful placement of the PTEG catheter in the intended position with immediate catheter functionality. After PTEG placement, patients were observed for 2 hours and subsequently transferred to the ward.
PTEG Post-Procedural Follow-Up
Changes in nausea and vomiting were evaluated using the same ordinal scales applied at baseline up to 72 hours after insertion. Postprocedural abdominal pain, opioid and antiemetic use, length of hospital stay, discharge destination, and need for antibiotic therapy were also recorded. Beyond 72 hours, follow-up data were collected and included catheter dwell time, catheter checks and exchanges, catheter dysfunction, catheter-related readmissions, and survival status at last follow-up. Procedure-related adverse events (AE) occurring within 30 days were recorded using the CIRSE AE classification. 26
Outcomes
Primary outcomes included <72 hours clinical success and >72 hours clinical outcomes. Short-term clinical success was defined as a composite outcome reflecting effective gastrointestinal decompression within 72 hours, including resolution or meaningful improvement of nausea and/or vomiting, removal of nasogastric decompression when present, and/or ability to resume oral intake. Clinical outcomes beyond 72 hours were defined by symptom evolution, catheter-related outcomes, and survival during follow-up. The secondary outcome was procedural safety, assessed by the occurrence procedural-related AE.
Statistical Analysis
Continuous variables were summarized as medians with IQR, and categorical variables as counts and percentages. Paired pre- and post-procedural comparisons of binary variables were performed using the McNemar test. Paired ordinal variables were compared using the Wilcoxon signed-rank test. Time-to-event analyses were performed using Kaplan–Meier estimates for overall survival and catheter event-free survival, with censoring at the date of last follow-up. A two-sided P-value <.05 was considered statistically significant. All analyses were conducted using R, version 4.5.2 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline Characteristics
Between February 2018 and April 2025, 53 patients were evaluated for PTEG and 47 were included for analysis (Figure 1). Median age was 65.0 years (IQR: 51.0-73.0), and most patients were female (37, 78.7%). Ovarian cancer was the most frequent underlying malignancy (21, 51.2%). Intestinal obstruction was the predominant indication for PTEG (46, 97.9%), most commonly related to peritoneal carcinomatosis with or without ascites (46, 97.9%). Obstruction was predominantly mechanical (45, 95.7%). The majority of patients presented with nausea (41, 87.2%) and vomiting (41, 87.2%). All baseline characteristics are summarized in Table 1.
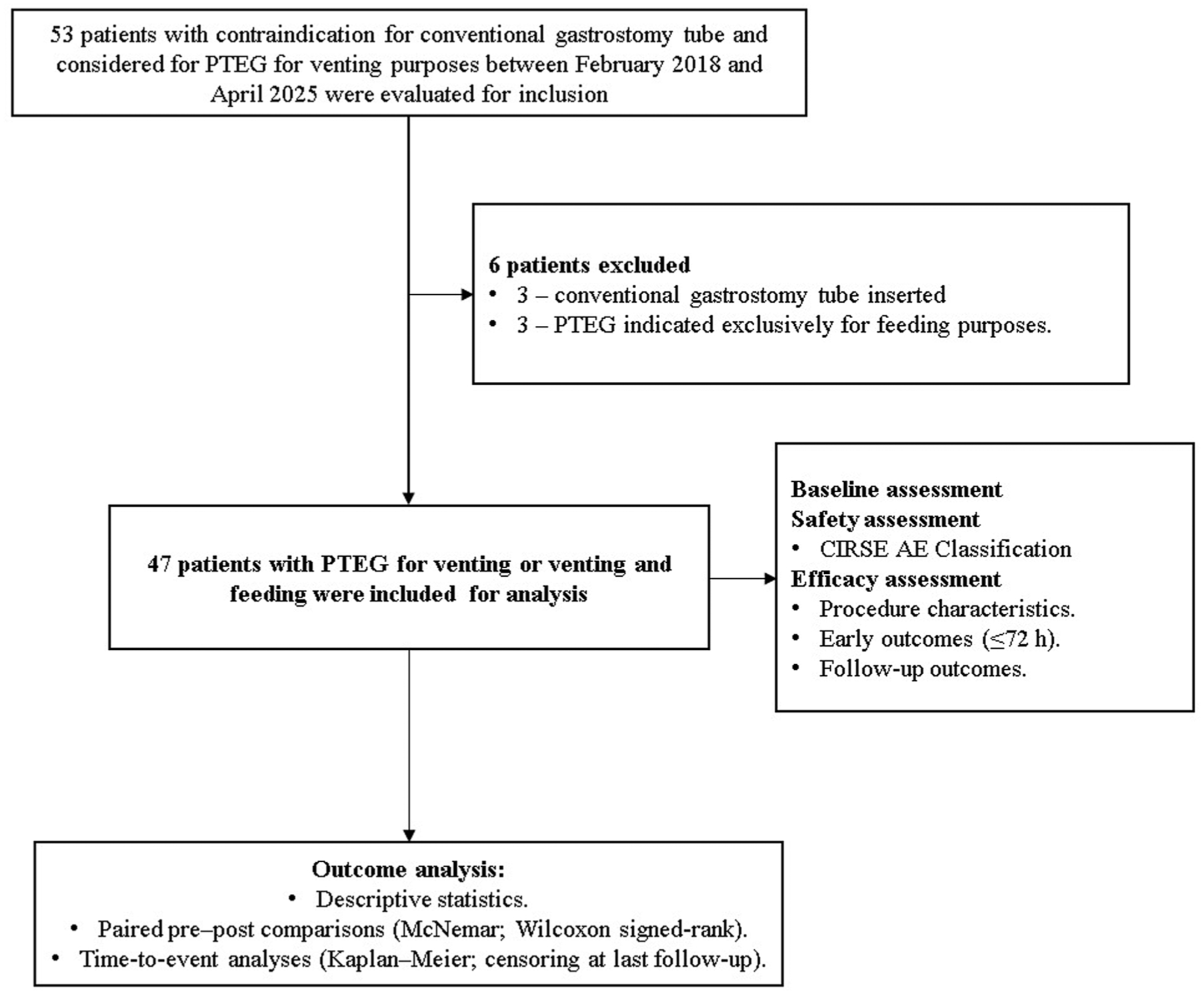
Study flowchart.
Baseline Characteristics.
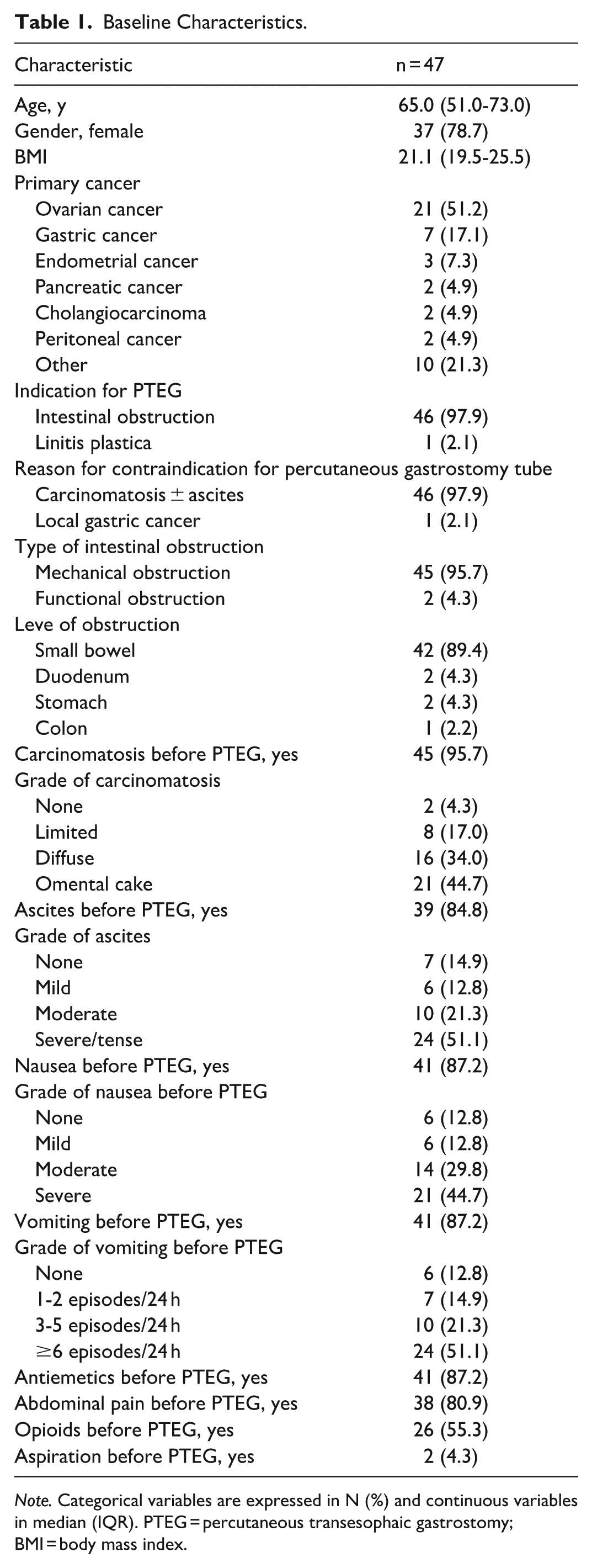
Note. Categorical variables are expressed in N (%) and continuous variables in median (IQR). PTEG = percutaneous transesophaic gastrostomy; BMI = body mass index.
PTEG Procedure
Technical success was 100%. Median procedure time was 23.0 minutes (IQR: 20.0-31.0). Thyroid enlargement was present in 14 patients (30.4%), with transthyroid placement performed in 13 (28.3%). The most frequently used PTEG tube size was 14 French (38, 82.6%), and the most common tube length was 60 cm (24, 53.3%; Figure Supplemental 1). Suction was required during admission in most patients (33, 70.2%). A NGT was in place before PTEG in the majority of cases (40, 85.1%) and was removed after PTEG in all patients (40, 100%; Table 2).
PTEG Procedure Characteristics.
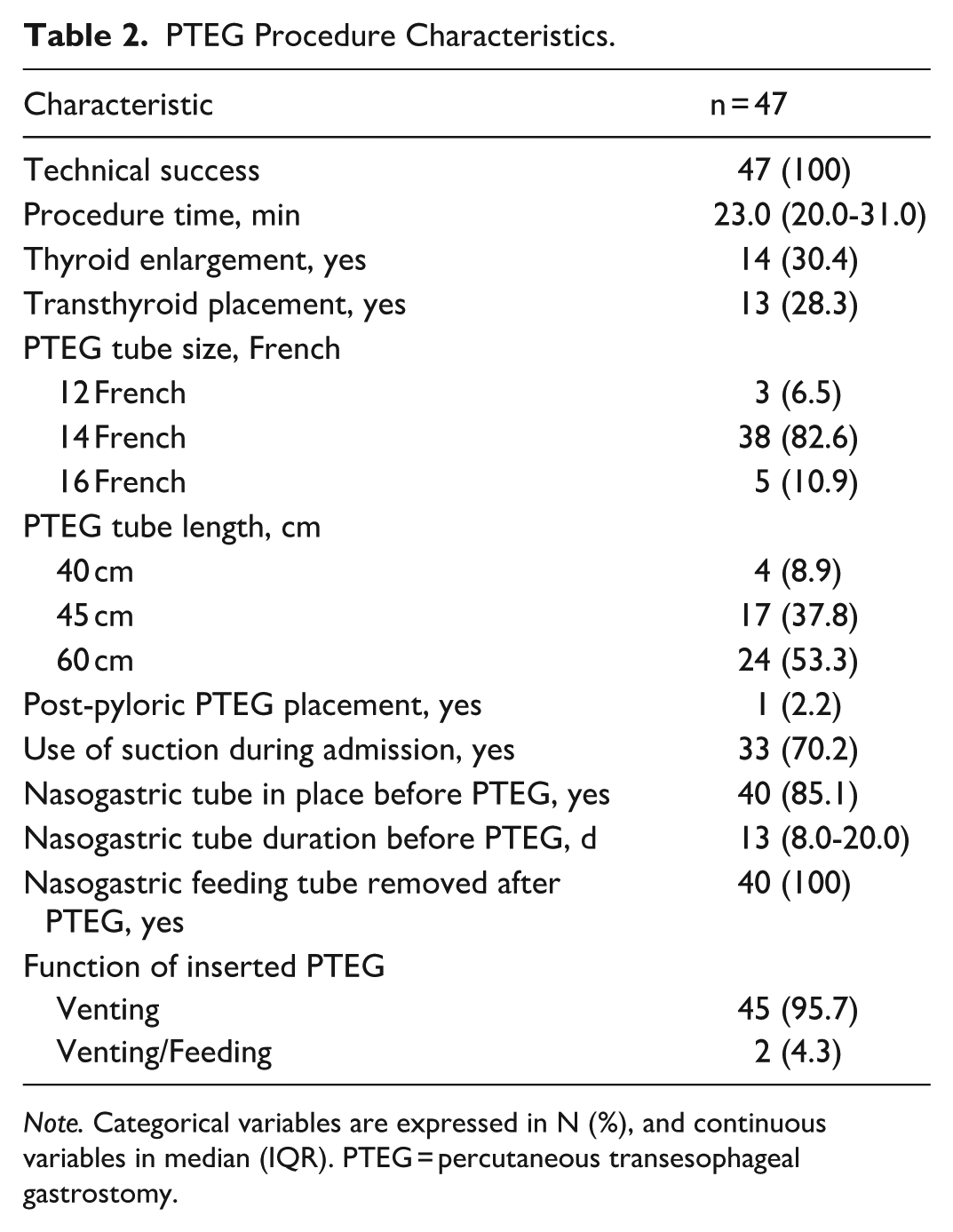
Note. Categorical variables are expressed in N (%), and continuous variables in median (IQR). PTEG = percutaneous transesophageal gastrostomy.
Within 72 Hours Clinical Outcomes
Clinical success within 72 hours after PTEG was achieved in most patients (40, 90.9%). Most patients presented a resolution of nausea (38, 90.5%), and vomiting (40, 95.2%). Use of antiemetics (4, 9.5%) and opioids (6, 14.3%) after PTEG was infrequent. Most patients were able to resume oral intake after PTEG (41, 91.1%). Median post-PTEG hospital stay was 6.0 days (IQR: 3.0-13.0). Most patients were discharged home or to palliative care (31, 66.9%; Table 3).
PTEG Early Clinical Outcomes (<72 Hours).
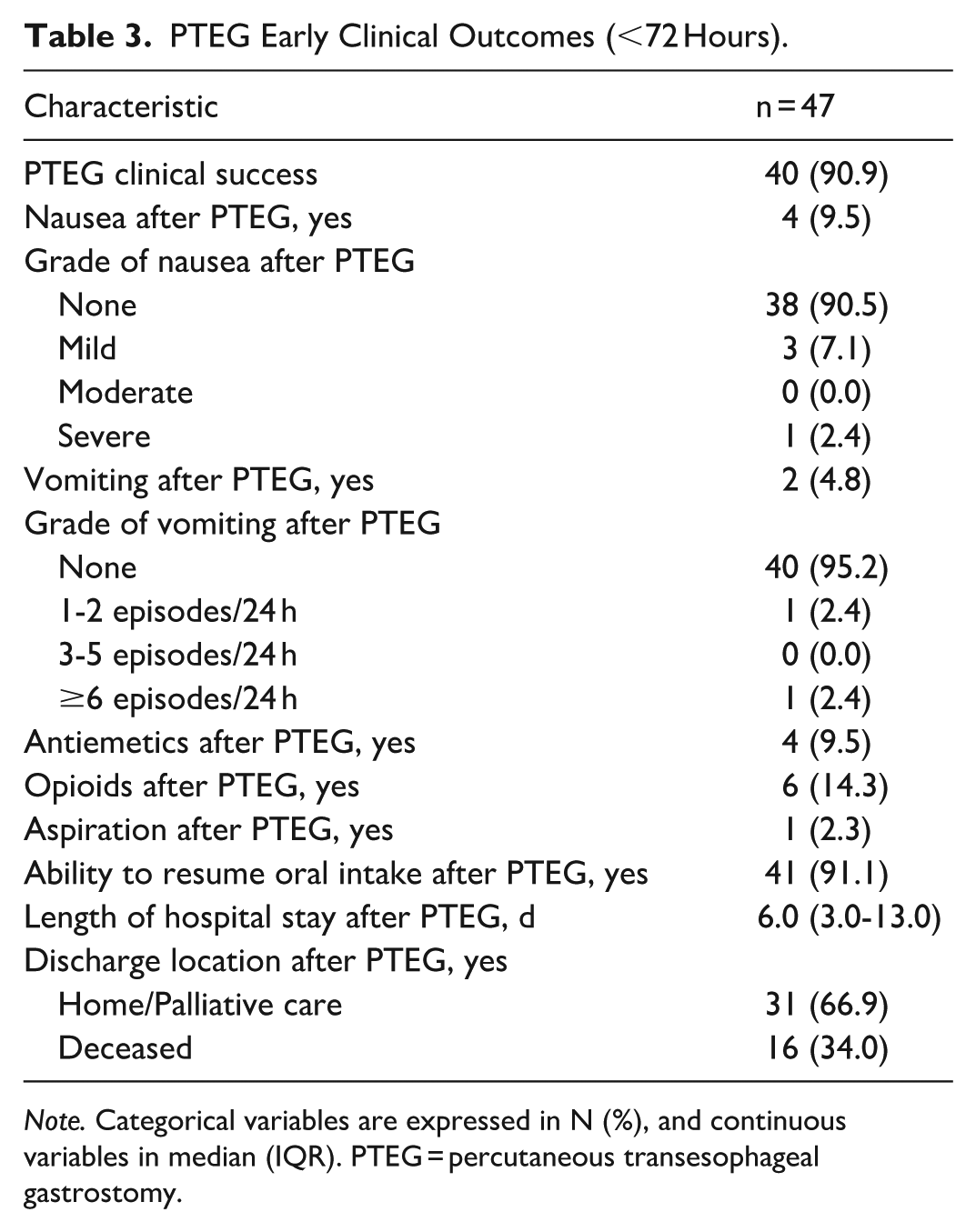
Note. Categorical variables are expressed in N (%), and continuous variables in median (IQR). PTEG = percutaneous transesophageal gastrostomy.
More Than 72 Hours Clinical Outcomes
Median follow-up duration after PTEG was 32.0 days (IQR: 9.0-75.0). At last follow-up, 18 patients were alive (38.3%). Recurrence of nausea and vomiting occurred in 3 patients each (6.4%). Hospital readmission after PTEG occurred in 9 patients (19.1%), most commonly unrelated to the procedure (7, 14.9%). PTEG dysfunction was observed in 10 patients (22.7%), with 9 (19.2%) requiring a tube check and 6 (12.8%) requiring tube exchange. Dysfunction was predominantly mechanical, with displacement or migration in 4 patients (8.2%) and blockage in 3 (6.1%). Median time from PTEG insertion to dysfunction was 16 days (IQR: 14-63), with 6 of 10 events (60%) occurring within the first 30 days after insertion. Median total PTEG duration was 32.9 days (IQR: 9.0-75.0; Table 4).
PTEG Follow-Up Outcomes and Durability.
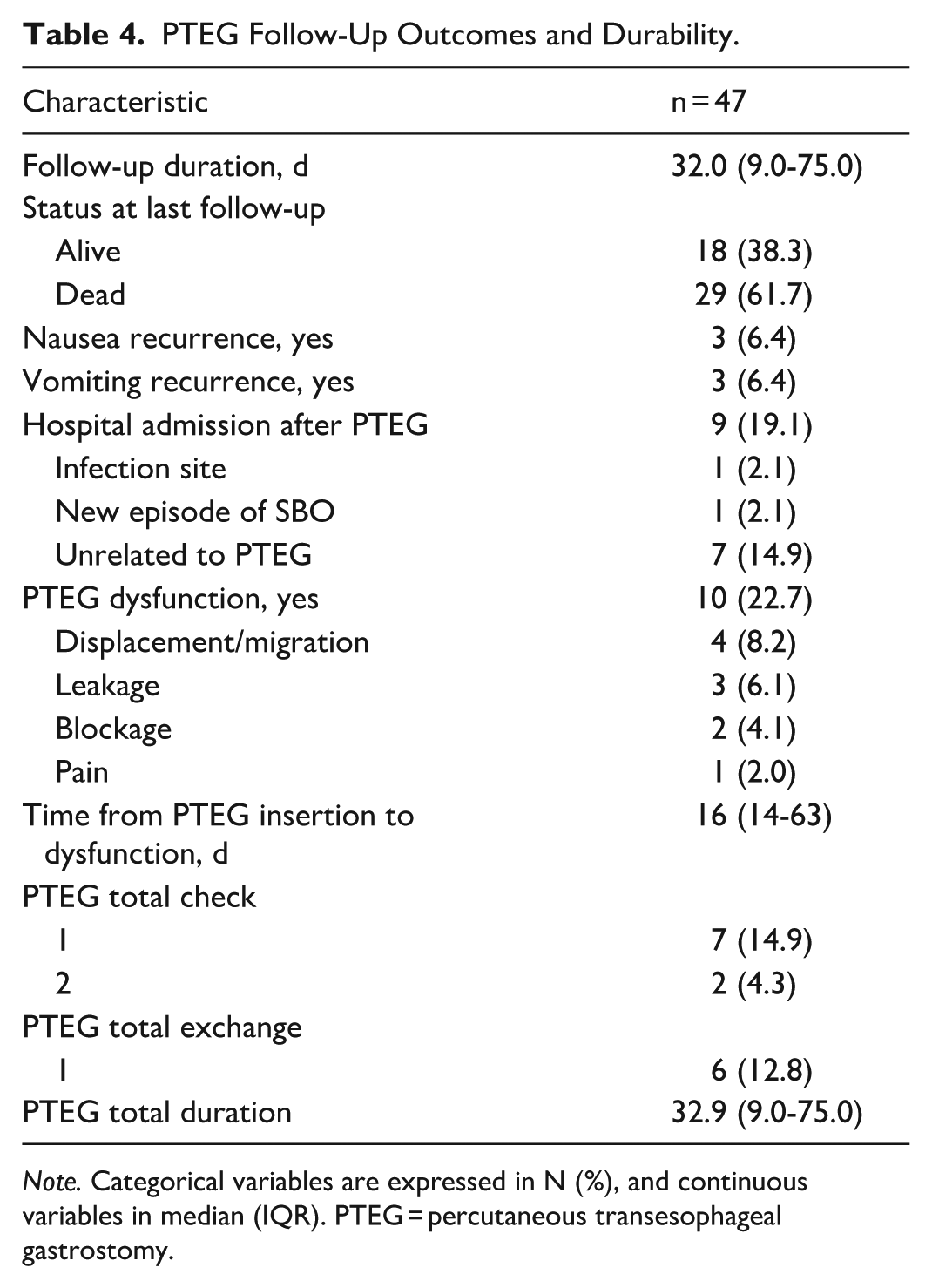
Note. Categorical variables are expressed in N (%), and continuous variables in median (IQR). PTEG = percutaneous transesophageal gastrostomy.
AEs Related to PTEG
CIRSE grade 1 AE occurred in 4 patients (8.5%), all related to pain, while a single CIRSE grade 2 AE due to pain was observed (1, 2.1%). No CIRSE grade 3 to 6 AEs were recorded (Table Supplemental 1).
Outcome Analysis
Proportion of patients with nausea decreased from 41/47 (87.2%) before PTEG to 4/47 (8.5%) after the procedure (P < .001), and median nausea severity decreased from 2.0 (IQR: 1.5-3.0) to 0.0 (IQR: 0.0; P < .001). Vomiting prevalence decreased from 41/47 (87.2%) to 2/47 (4.3%) (P < .001), with vomiting severity decreasing from a median of 3.0 (IQR: 1.0-3.0) to 0.0 (IQR: 0.0; P < .001). Use of antiemetics was reduced from 41/47 patients (87.2%) to 4/47 (8.5%) (P < .001), while opioid use decreased from 25/47 (53.2%) to 6/47 (12.8%; P < .001; Table Supplemental 2, Figure 2). Kaplan–Meier analysis showed a median overall survival of 52.0 days (95% CI: 38.0-245.0) and median catheter event-free survival of 212 days (95% CI: 65-not reached). Mean overall survival was 136.2 days (95% CI: 75.5-196.8) and mean catheter event-free survival was 229.9 days (95% CI: 131.3-328.6; Figure 3).
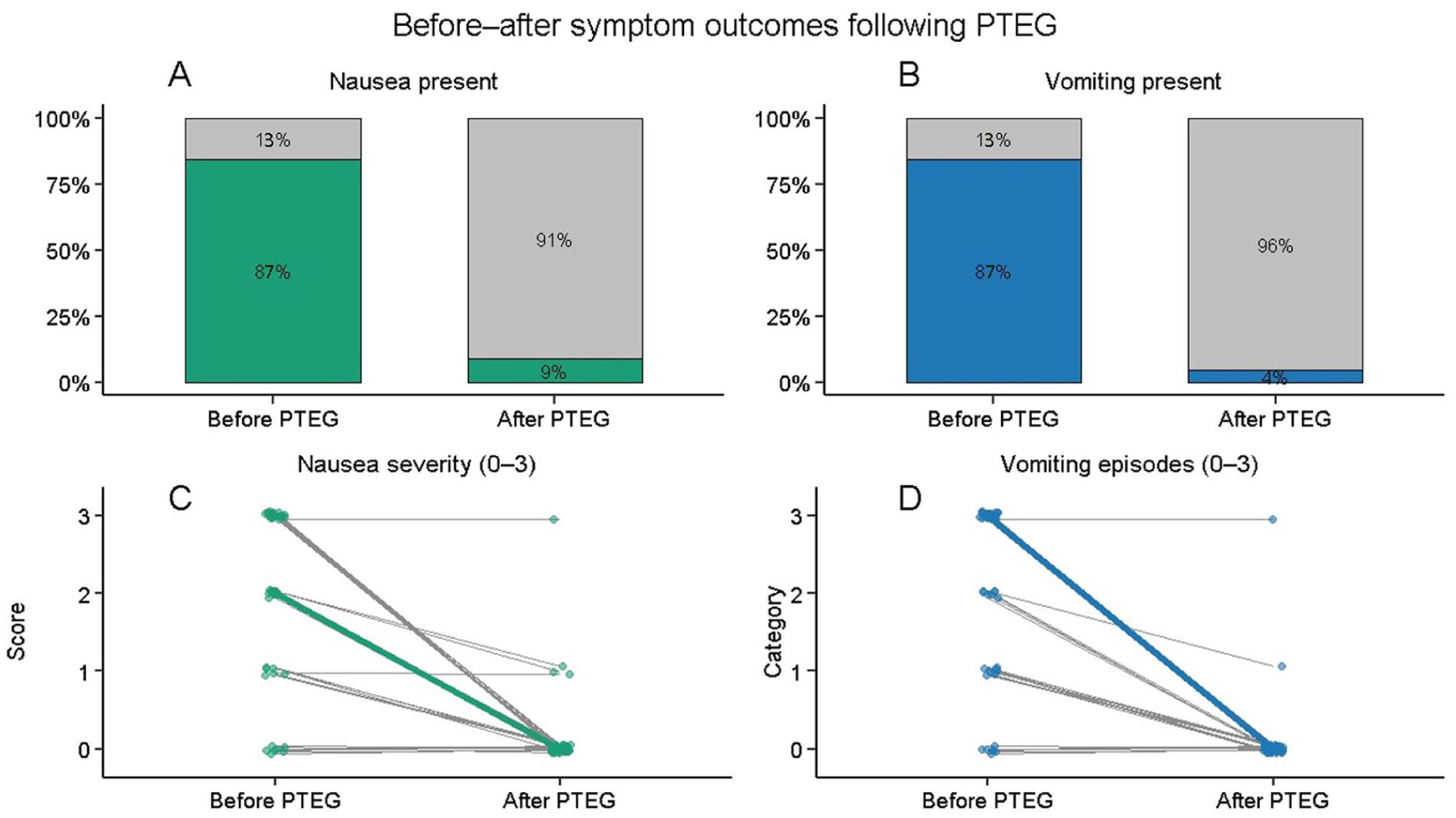
Before–after symptom outcomes following venting PTEG. (A) Proportion of patients with nausea before and after PTEG placement. (B) Proportion of patients with vomiting before and after PTEG placement. (C) Paired individual changes in nausea severity score (ordinal scale 0-3) before and after PTEG. (D) Paired individual changes in vomiting episode category (ordinal scale 0-3) before and after PTEG.
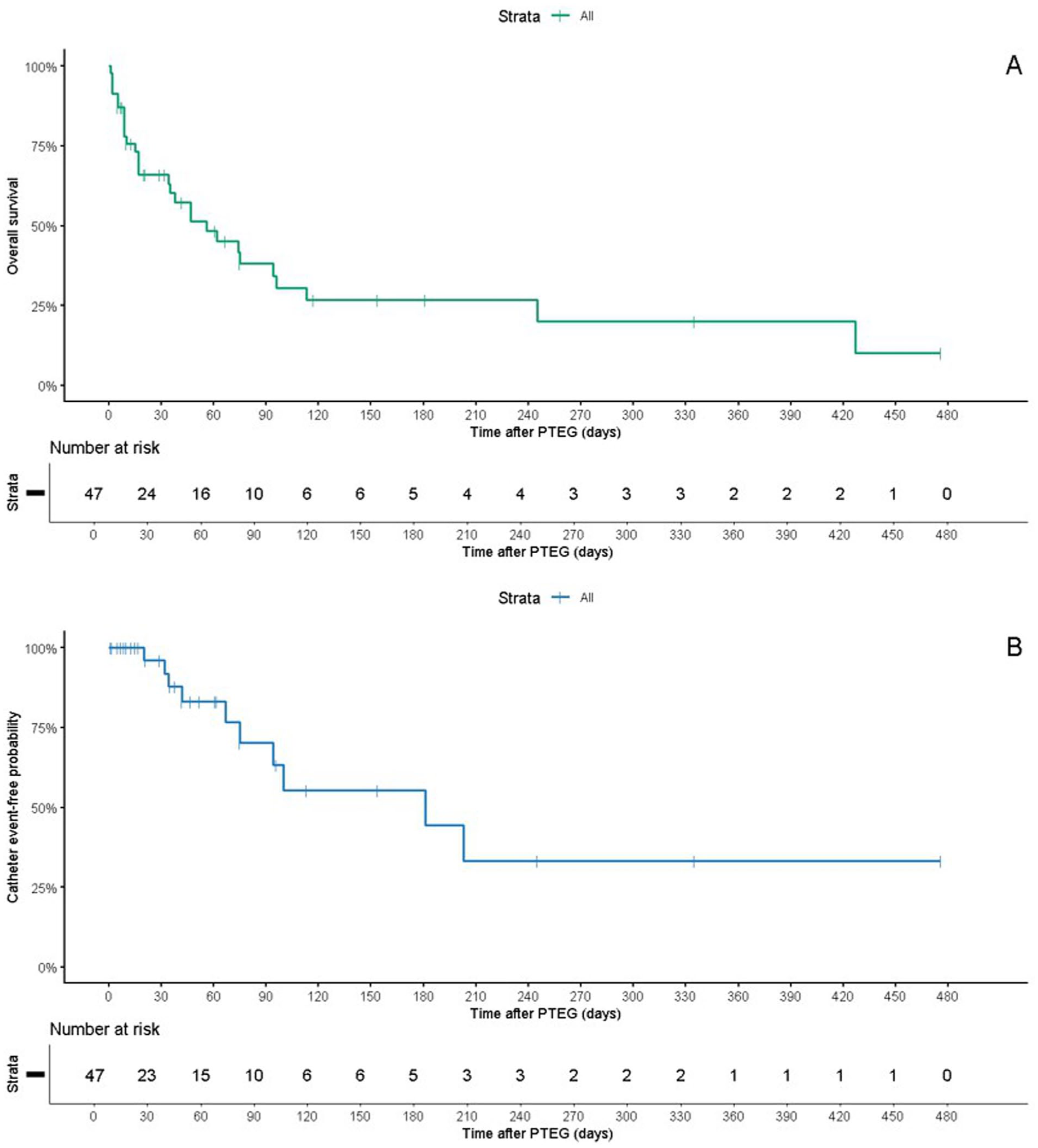
Kaplan–Meier analysis of survival and catheter durability after venting PTEG. (A) Overall survival following PTEG placement, with a median survival of 52.0 days (95% CI: 38.0-245.0) and mean survival of 136.2 days (95% CI: 75.5-196.8). (B) Catheter event-free survival, defined as catheter function without catheter-related complications requiring intervention, with a median of 212 days (95% CI: 65-not reached) and mean of 229.9 days (95% CI: 131.3-328.6).
Discussion
Patients with malignant bowel obstruction who are not candidates for transabdominal gastrostomy remain a clinically vulnerable population with limited durable decompressive options, often reliant on poorly tolerated nasogastric decompression. This study expands existing PTEG evidence by quantifying symptom control and catheter performance of venting PTEG in patients with MBO. Paired comparisons demonstrated a significant short-term reduction in symptoms, with nausea and vomiting decreasing from 87% before PTEG to 9% and 4% after placement, respectively, and median severity scores falling from moderate–severe ranges to zero. Median overall survival was 52.0 days, whereas median catheter event-free survival was 212 days (95% CI: 65-not reached), with mean estimates of 136.2 and 229.9 days respectively, with most dysfunction events clustering early after insertion, 60% within the first 30 days, and relative catheter stability thereafter. Catheter dysfunction occurred in 23% and was predominantly mechanical. Of the recorded dysfunction events, 70% were mechanical in nature, comprising displacement or migration and blockage, with leakage and pain accounting for the remainder. PTEG was also demonstrated to be safe, with only minor AE reported.
Our findings align with the established safety and technical feasibility of PTEG reported in previous series, while extending assessment into clinical and catheter-related outcomes that have been only partially addressed.21,22,27 Early Japanese experiences reported technical success rates approaching 100% with low rates of major complications, but were largely descriptive and focused on feasibility in small cohorts.21,28 Subsequent Western series confirmed high technical success but continued to emphasize short-term procedural outcomes and surrogate measures of effectiveness.22,29 More recently, Rotellini-Coltvet et al reported a 92% technical success rate in 38 patients with MBO, with a mean catheter dwell time of 56 days and tube exchanges required in 14%, but symptom assessment relied primarily on qualitative improvement rather than paired analysis. 27 In contrast, the present study combines paired pre–post symptom analysis with time-to-event evaluation, demonstrating symptom reduction exceeding 85% alongside a catheter dysfunction rate of 22.7% over extended follow-up. These results provide a more granular estimate of expected catheter behavior and sustained clinical benefit, positioning PTEG outcomes within a longitudinal palliative care framework rather than isolated procedural success.
Catheter durability has been variably reported, with early studies primarily describing short-term mechanical issues. Initial Japanese and early Western series described tube obstruction, dislodgement, or leakage as infrequent events but did not report denominator-based rates or follow-up beyond the immediate post-placement period.21,28 In these reports, catheter-related issues were generally described qualitatively, limiting comparison across cohorts. Subsequent Western experience began to quantify early tube-related events. Sanogo et al reported minor catheter complications in 43% of patients at 30 days, predominantly due to premature tube retraction or clogging (each 14.3%). 29 In contrast, catheter dysfunction in our cohort occurred in 22.7% of patients over extended follow-up, indicating a lower cumulative dysfunction rate despite substantially longer observation. More recent surgical series have provided additional but still limited durability data. Rotellini-Coltvet et al reported catheter exchanges in 27.8% of patients, with a median dwell time of 29 days, and most exchanges related to early dislodgement rather than loss of decompressive efficacy. 27 Our median catheter duration was comparable at 32.9 days, but with most patients requiring at most a single exchange (12.8%) or check (14.9%), suggesting similar early vulnerability but preserved function thereafter. At the population level, the nationwide administrative analysis by Nakama et al identified early tube replacement within 2 weeks in approximately 6% to 8% of cases, supporting the concept of an initial mechanical risk window. 23 This pattern parallels the early clustering of catheter events observed in our analysis, while the longer median catheter event-free survival relative to overall survival (212 vs 52.0 days) demonstrates that, once past the early period, catheter function is generally maintained for the remainder of palliative care. The apparent discrepancy between the short median catheter dwell time and the longer mean catheter event-free survival reflects survivorship bias and competing mortality, as many patients died with a functioning catheter before experiencing catheter dysfunction, resulting in censoring of catheter-related outcomes.
From a clinical perspective, these findings help position venting PTEG within the spectrum of palliative options for MBO, bridging the gap between temporary decompression and durable symptom control. Prolonged NGT decompression is known to be poorly tolerated, with complication rates exceeding 30% to 40% and significant impact on comfort, mobility, and dignity, often leading to premature discontinuation despite ongoing obstruction.9,11 Conversely, PG, while effective in selected patients, has been associated with leakage, infection, and peritonitis rates of up to 20% to 30% in the presence of ascites or peritoneal carcinomatosis, frequently limiting its applicability in advanced disease.14,15 In patients managed conservatively without decompressive intervention, symptom burden remains high and spontaneous resolution is uncommon, with persistent nausea and vomiting reported in over 70% of cases and median survival measured in weeks rather than months.1,4 Against this background, our data demonstrate that venting PTEG provides sustained gastrointestinal decompression over the clinically relevant palliative timeframe. Symptom control was durable, with reductions in nausea and vomiting exceeding 85%, median severity scores reaching zero, and decreased reliance on antiemetics and opioids. Although catheter-related events occurred, they were predominantly early and mechanical, without loss of decompressive efficacy. This is supported by Kaplan–Meier analysis showing catheter event-free survival exceeding overall survival, indicating that catheter function was generally maintained for the remainder of patients’ palliative course. These findings support PTEG as a clinically meaningful decompressive strategy, with catheter maintenance representing an anticipated component of care rather than a limitation of the technique.
This study has several limitations. Its retrospective, single-center design limits generalizability and introduces selection bias in a palliative population with advanced disease. The lack of a comparator group, such as PG or prolonged NGT decompression, precludes direct comparison of efficacy, complications, or durability across decompressive strategies. Symptom response was derived from clinical documentation rather than standardized quality-of-life instruments, potentially underestimating variability over time. Finally, follow-up was heterogeneous and survival-dependent, which may have limited time-to-event assessment of catheter dysfunction and underestimated late device-related events.
Conclusion
Venting PTEG provides effective decompression in patients with MBO who are not candidates for PG. In this Western palliative cohort, it achieved rapid and sustained symptom relief with a favorable safety profile. Catheter dysfunction was infrequent, predominantly early, manageable, and did not compromise decompressive efficacy, with catheter function generally preserved throughout the palliative course. These data may inform procedural selection by supporting venting PTEG as a durable palliative decompression option in anatomically or clinically unsuitable PG candidates.
Supplemental Material
sj-docx-1-caj-10.1177_08465371261437627 – Supplemental material for Short- and Long-Term Outcomes and Catheter Durability After Venting Percutaneous Transesophageal Gastrostomy in Palliative Malignant Bowel Obstruction
Supplemental material, sj-docx-1-caj-10.1177_08465371261437627 for Short- and Long-Term Outcomes and Catheter Durability After Venting Percutaneous Transesophageal Gastrostomy in Palliative Malignant Bowel Obstruction by Alfredo Páez-Carpio, Romman Nourzaie, Matthew Squire, Elizabeth David, Chris Dey, Robyn Pugash and Gilbert Maroun in Canadian Association of Radiologists Journal
Supplemental Material
sj-docx-2-caj-10.1177_08465371261437627 – Supplemental material for Short- and Long-Term Outcomes and Catheter Durability After Venting Percutaneous Transesophageal Gastrostomy in Palliative Malignant Bowel Obstruction
Supplemental material, sj-docx-2-caj-10.1177_08465371261437627 for Short- and Long-Term Outcomes and Catheter Durability After Venting Percutaneous Transesophageal Gastrostomy in Palliative Malignant Bowel Obstruction by Alfredo Páez-Carpio, Romman Nourzaie, Matthew Squire, Elizabeth David, Chris Dey, Robyn Pugash and Gilbert Maroun in Canadian Association of Radiologists Journal
Supplemental Material
sj-tiff-3-caj-10.1177_08465371261437627 – Supplemental material for Short- and Long-Term Outcomes and Catheter Durability After Venting Percutaneous Transesophageal Gastrostomy in Palliative Malignant Bowel Obstruction
Supplemental material, sj-tiff-3-caj-10.1177_08465371261437627 for Short- and Long-Term Outcomes and Catheter Durability After Venting Percutaneous Transesophageal Gastrostomy in Palliative Malignant Bowel Obstruction by Alfredo Páez-Carpio, Romman Nourzaie, Matthew Squire, Elizabeth David, Chris Dey, Robyn Pugash and Gilbert Maroun in Canadian Association of Radiologists Journal
Footnotes
Abbreviations
AE – Adverse event
CIRSE – Cardiovascular and Interventional Radiological Society of Europe
KM – Kaplan–Meier
MBO – Malignant bowel obstruction
NGT – Nasogastric tube
PG – Percutaneous gastrostomy
PTEG – Percutaneous transesophageal gastrostomy
Ethical Considerations
This study was approved by the Research Ethics Board of Sunnybrook Health Sciences Centre, Toronto, Canada (REB #SBHSC2025_6928). All procedures performed were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments.
Consent to Participate
The requirement for informed consent was waived due to the retrospective nature of the study.
Author Contributions
GM the presented research study. APC collected the data presented. APC conducted the literature review, data analysis, and was the major contributor to the writing of the manuscript. APC selected the figures presented. GM, RN, MS, RB, ED, and CD supervised the writing of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The dataset used for this study is not publicly available as institutional consent for data sharing was not obtained from Sunnybrook Health Sciences Centre. However, data may be made available by the corresponding author upon reasonable request and subject to institutional approval.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
