Abstract
Purpose:
Accurate vessel measurement is essential in endovascular thrombectomy (EVT) for acute ischemic stroke. Discrepancies between computed tomography angiography (CTA) and digital subtraction angiography (DSA) may impact procedural planning and device selection. This study compares vessel diameter measurements from CTA and DSA in patients with middle cerebral artery (MCA) M1 occlusions.
Methods:
In this single-center retrospective study, 90 consecutive patients who underwent EVT for MCA M1 occlusions between February 2020 and March 2024 were included. Vessel diameters were independently measured by 3 neuroradiologists using CTA and DSA (pre- and post-intervention). Statistical analysis included Wilcoxon signed-rank tests, intraclass correlation coefficient (ICC), and Bland–Altman analysis.
Results:
CTA consistently overestimated vessel diameter compared to DSA. The mean M1 diameter was 2.29 ± 0.27 mm on CTA and 2.16 ± 0.30 mm on pre-EVT DSA (P < .001), with a median difference of 0.4 mm (IQR: 0.2-0.6 mm). In 70% of cases, CTA values exceeded DSA. Bland–Altman analysis confirmed a mean difference of +0.13 mm (limits of agreement: −0.25 to +0.51 mm). No significant change was observed between pre- and post-EVT DSA measurements (P = .103). Clot-side M1 segments were significantly smaller than contralateral measurements on CTA (P = .003). Inter-rater agreement was good (ICC = .785).
Conclusions:
CTA overestimates MCA M1 diameter relative to DSA. While the discrepancy is modest, it may influence device selection in borderline cases. Awareness of this variability is important, and further research is warranted to explore its clinical implications.
Introduction
Stroke is a major global health concern and one of the leading causes of morbidity and mortality worldwide. 1 Among its subtypes, acute ischemic stroke due to large vessel occlusion (LVO) is particularly severe and demands rapid diagnosis and intervention to optimize clinical outcomes.2,3 Endovascular thrombectomy (EVT) is the standard of care for treating LVOs, significantly improving functional recovery when performed promptly. 4 The success of EVT is heavily dependent on accurate pre-procedural imaging to evaluate vessel morphology and guide device selection. 5
However, discrepancies in vessel diameter measurements between imaging modalities, particularly between computed tomography angiography (CTA) and digital subtraction angiography (DSA), may influence procedural planning and outcomes.6-8 Previous studies have highlighted that CTA can overestimate vessel size compared to DSA, potentially impacting thrombectomy strategy. Moreover, vessel morphology and thrombus are critical factors influencing EVT success, further emphasizing the need for precise vessel assessment.9,10
This study aims to compare vessel diameter measurements obtained from CTA and DSA in patients with middle cerebral artery (MCA) M1 occlusions, evaluating the reliability of each modality. Additionally, we analyze vessel caliber changes before and after EVT and assess the influence of thrombus on MCA diameter.
Methods
This single-center retrospective study included 90 patients who underwent EVT for MCA M1 occlusions between February 2020 and March 2024 at our institution. Only patients who underwent pre-treatment imaging at our institution were included to ensure standardization of CTA acquisition protocols and measurement techniques. Patients with tandem occlusions (ICA + M1) were excluded. During this period, the total EVT volume was approximately 271 patients, with M1 occlusions representing a substantial subset. Approximately 1 in 3 EVT patients were not reimaged prior to intervention and were taken directly to the angiography suite, which may partially account for under capture in the imaging database. The study cohort consisted of 39 males and 51 females, with a median age of 75 years (range: 29-96 years). The requirement for informed consent was waived by the institutional research ethics board (file number 1030481).
Vessel diameters were independently measured on both CTA and DSA by 3 board-certified neuroradiologists. CTA was performed using 256 slice-CT scanners (Siemens SOMATOM Definition Flash and SOMATOM Drive—Siemens Healthineers, Erlangen, Germany). Luminal measurements were obtained using a standardized protocol with a maximum zoom Estimated Radiographic Magnification Factor (ERMF) of 4 mm on coronal reformatted images. Measurements were taken at the M1 segment immediately proximal to the occlusion and at the corresponding site on the contralateral side (Figures 1 and 2).
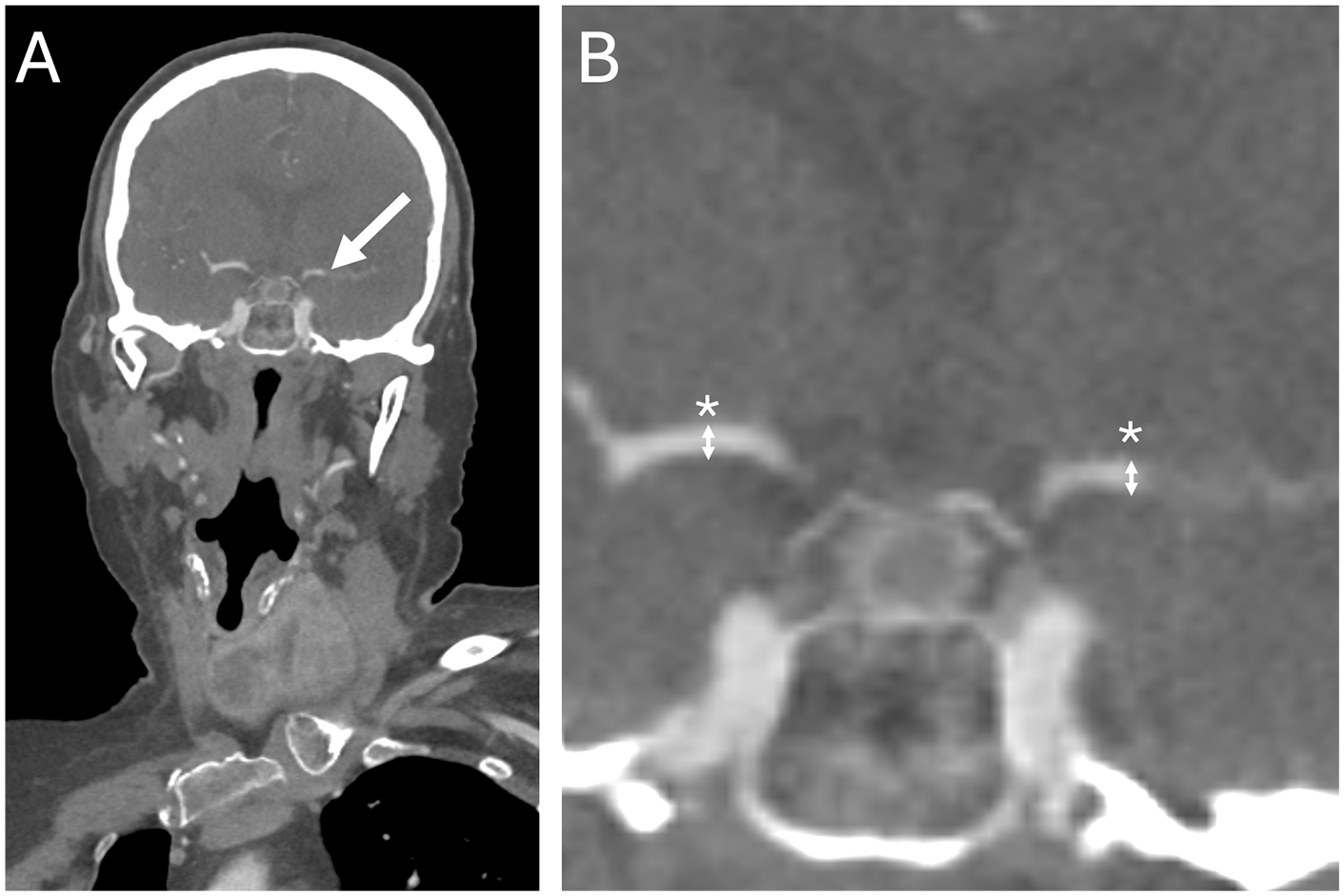
Coronal CTA images obtained before (A) and after (B) maximal zoom show a mid-segment occlusion of the left M1 (white arrow). Craniocaudal vessel diameter measurements were performed at the site of the occlusion and at the corresponding location on the contralateral M1 segment (*).
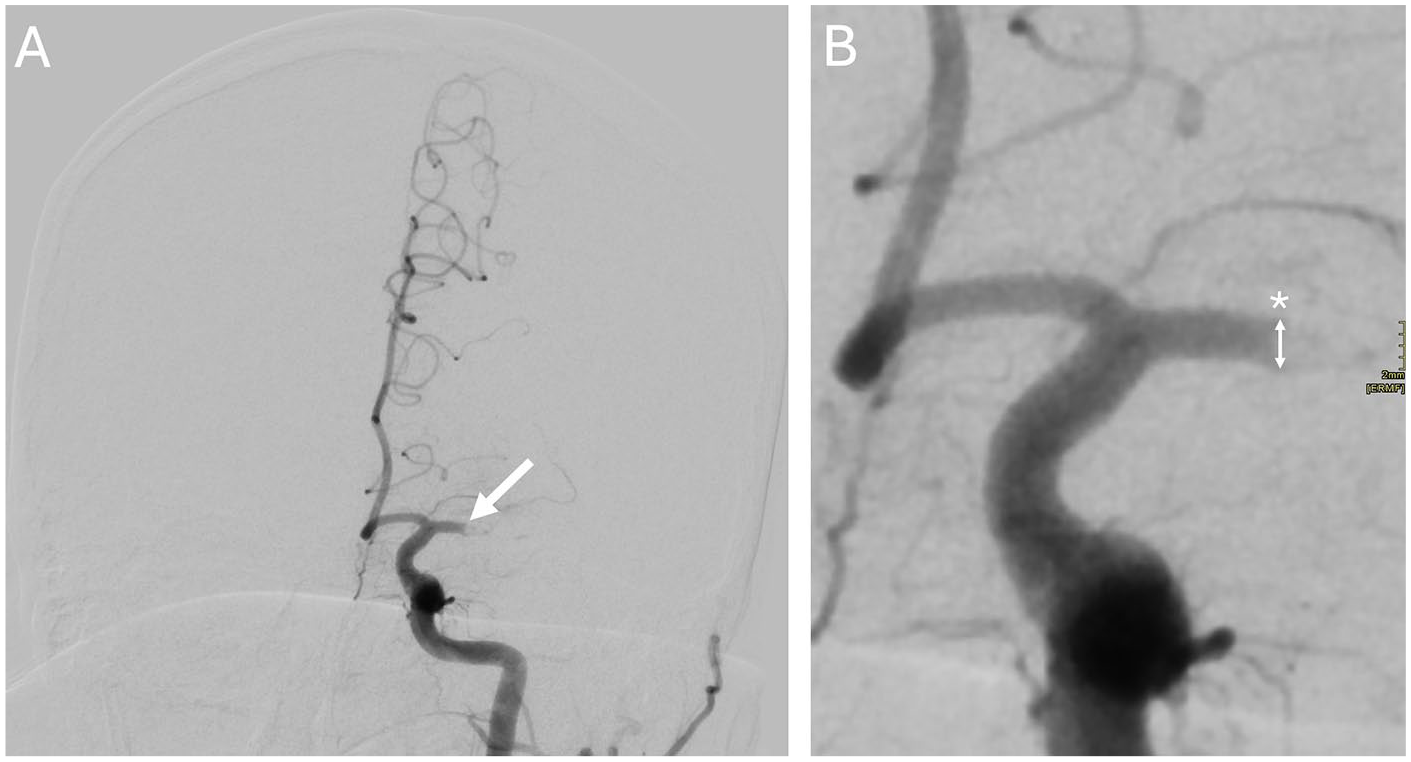
Anteroposterior left ICA angiograms obtained before (A) and after (B) maximal zoom confirm a mid-segment occlusion of the left M1 (white arrow). Craniocaudal vessel diameter was measured at the level of the occlusion (*).
DSA measurements were acquired using an anteroposterior projection at a maximum zoom ERMF of 2 mm for optimal accuracy. Craniocaudal diameter measurements were taken at the level of the occlusion, matching the CTA measurement site, both before (Figure 2) and after EVT (Figure 3). All imaging assessments were reviewed on high-resolution diagnostic monitors (Barco—Barco NV, Kortrijk, Belgium) to ensure consistency and precision in caliper placement.
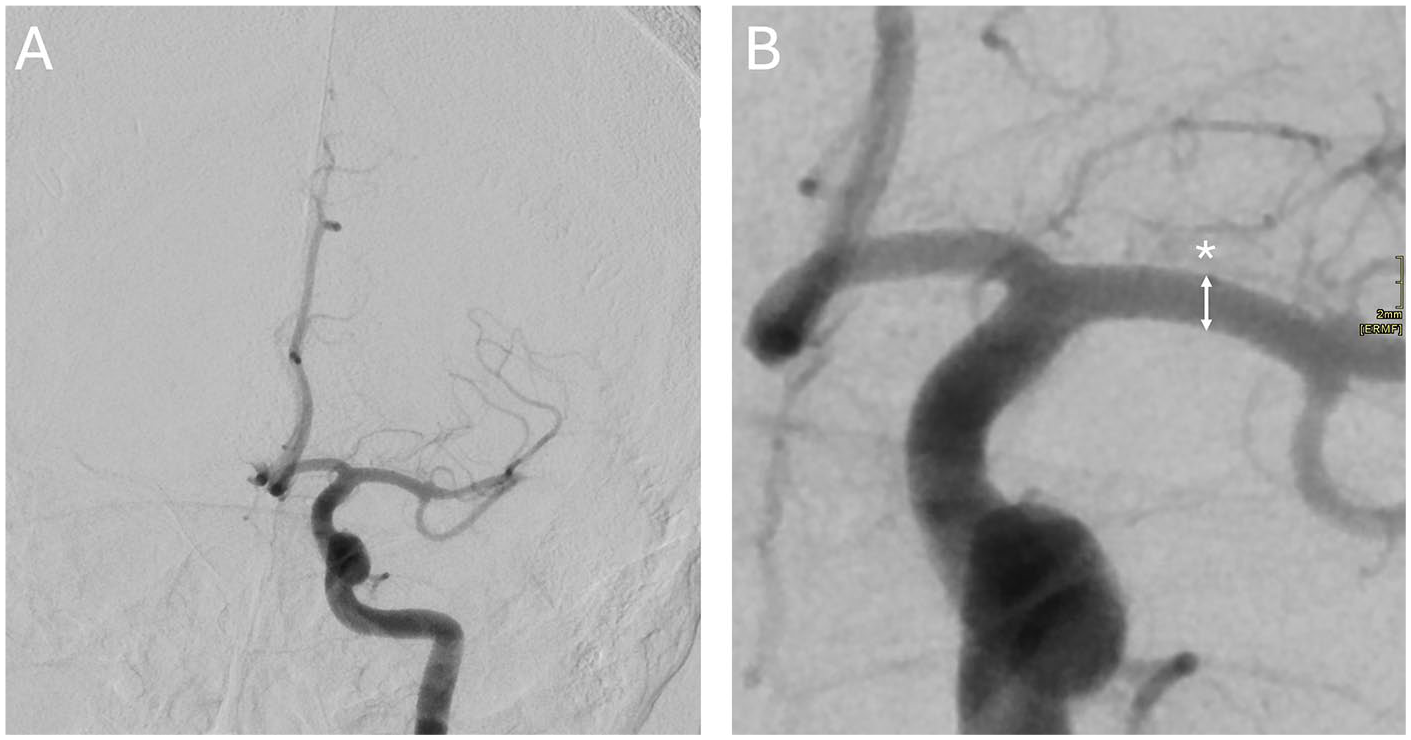
Anteroposterior left ICA angiograms obtained before (A) and after (B) maximal zoom following a single aspiration pass demonstrate successful recanalization of the previously occluded segment. Craniocaudal vessel diameter was remeasured at the site of the original occlusion (*).
For CTA, contrast was administered using 60 mL of Omnipaque 350 (GE Healthcare, Chicago, IL, USA), at 4 mL/s, followed by a 40 mL saline flush. EVT procedures were performed on an advanced biplane angiography system (Siemens ARTIS Q—Siemens Healthineers, Erlangen, Germany), with contrast injected manually using Omnipaque 200 (GE Healthcare, Chicago, IL, USA).
Statistical comparisons were conducted using Wilcoxon signed-rank tests to assess: (1) CTA pre-EVT (CTA-pre) versus DSA pre-EVT (DSA-pre), (2) DSA-pre versus DSA post-EVT (DSA-post), and (3) CTA clot-side M1 versus CTA contralateral M1 measurements. Inter-rater reliability was evaluated using the intraclass correlation coefficient (ICC) and further characterization of measurement agreement was performed using a Bland-Altman analysis.
Results
A summary of patient demographic details and side of occlusion is presented in Table 1. CTA significantly overestimated vessel diameter compared to DSA prior to EVT, with a median diameter of 2.27 mm (IQR: 0.34) on CTA-pre versus 2.17 mm (IQR: 0.37) on DSA-pre (P < .001). In 70% of cases, CTA measurements exceeded the corresponding DSA-pre values, with a median difference of 0.4 mm (IQR: 0.2-0.6 mm).
Patient Demographics (n = 90).

No statistically significant difference was observed between DSA-pre and DSA-post measurements (P = .103), suggesting that vessel caliber remains stable during EVT. Conversely, CTA-derived measurements of the occluded (clot-side) M1 segment were significantly smaller than those of the contralateral M1 (P = .003), indicating potential thrombus-related narrowing.
Inter-rater reliability for vessel measurements was good, with an intraclass correlation coefficient (ICC) of .785 (95% CI: 0.528-0.887), indicating moderate to strong agreement among observers.
To further evaluate measurement agreement, a Bland–Altman analysis was performed, showing a mean difference of +0.13 mm between CTA and DSA-pre values, with 95% limits of agreement ranging from −0.25 to +0.51 mm (Figure 4), confirming a systematic overestimation by CTA.
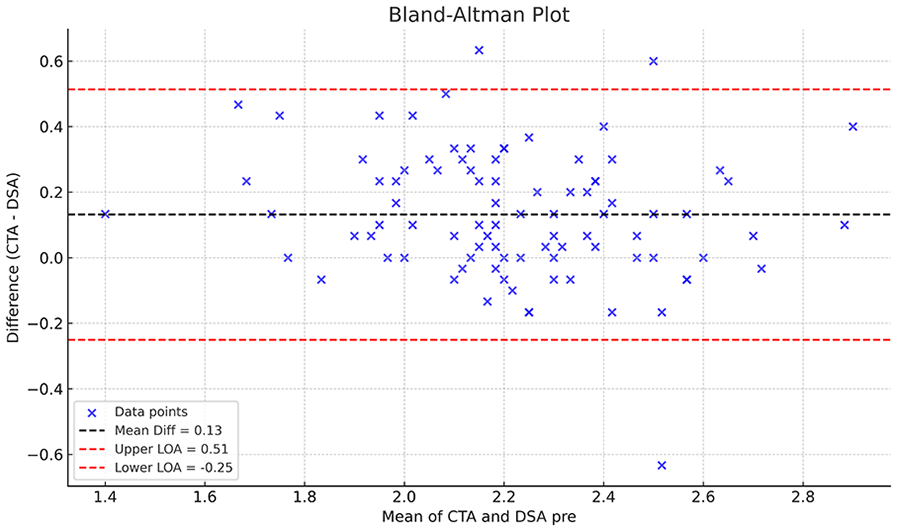
Bland–Altman plot comparing M1 vessel diameter measurements on clot-side CTA and pre-intervention DSA.
Discussion
This study demonstrates that CTA systematically overestimates vessel diameter compared to DSA in patients undergoing EVT for M1 occlusions. We observed a median difference of 0.4 mm (IQR: 0.2-0.6 mm) between CTA and DSA-pre measurements, with CTA values exceeding DSA in 70% of cases. A Bland–Altman analysis confirmed a systematic overestimation, with a mean difference of +0.13 mm and 95% limits of agreement ranging from −0.25 to +0.51 mm. Notably, DSA-based measurements remained stable before and after the procedure, and clot-side M1 diameters were significantly smaller than those on the contralateral side, likely reflecting thrombus-related narrowing.
The growing use of large-bore aspiration catheters in EVT has renewed focus on vessel sizing and device compatibility. Although larger catheters have been associated with improved first-pass efficacy and better aspiration mechanics, concerns remain regarding vessel trauma and oversizing, especially when device selection is based on CTA rather than intra-procedural DSA.11,12 Some studies suggest that the push toward larger catheters has overshadowed the importance of precise vessel measurement and tailored device selection, factors that are critical to optimizing both safety and efficacy. 13
Oversized catheters can result in excessive vessel wall contact, increasing the risk of endothelial injury, vasospasm, and procedural complications. 14 Nevertheless, current EVT practice often favors the largest available aspiration catheter, selected prior to the acquisition of intra-procedural digital subtraction angiography (DSA), in an effort to minimize time to reperfusion. 15 As such, thrombectomy device selection is frequently based on pre-procedural imaging, primarily computed tomography angiography (CTA).
Our findings highlight a potential pitfall of relying solely on CTA for sizing, particularly in workflows where catheters are selected before DSA is acquired. While the median difference of 0.4 mm may appear modest, it is comparable to the outer diameter (OD) difference between aspiration catheters with inner diameters of 0.072″ (2.16 mm OD) and 0.062″ (1.93 mm OD). Such variation may be relevant in borderline cases where vessel size influences device selection and performance. In borderline cases, particularly involving smaller M1 segments or tortuous anatomy, this discrepancy could influence the choice between catheter sizes, potentially affecting aspiration force, navigability, or safety. Although many institutions default to standardized device selections, especially for M1 occlusions, our findings highlight that even small variations in measured diameter may matter in select scenarios and deserve further investigation.
From a physiological standpoint, the relationship between catheter diameter and aspiration efficiency is governed by Poiseuille’s Law, which states that flow through a cylindrical tube is proportional to the fourth power of its radius. As a result, even modest increases in catheter inner diameter can lead to substantial gains in aspiration force and clot ingestion capacity. Multiple studies have demonstrated improved first-pass success with larger-bore catheters, particularly when aspiration is used as the primary technique. However, these benefits must be balanced against anatomical constraints, as oversized catheters may reduce navigability or increase the risk of vessel injury—especially in smaller or tortuous arteries. In this context, accurate vessel sizing becomes essential to maximize efficacy while minimizing procedural risks.
The STRATIS registry 13 demonstrated substantial variability in M1 diameters across stroke patients, reinforcing the concept that optimal device sizing should be individualized. While small-bore catheters underperform, registry data suggest diminishing returns when upsizing beyond medium-bore devices. 16 This suggests a threshold effect, beyond which increasing catheter size no longer improves efficacy, challenging the “bigger is better” assumption and reinforcing the importance of appropriate sizing. 17
The CANADAPT study 18 further supports this concept by emphasizing the physics of aspiration thrombectomy. It introduces the “push effect,” where arterial flow around the catheter tip enhances clot retrieval. An optimal catheter-to-vessel diameter ratio (~80%) facilitates this dynamic. In contrast, oversized catheters may impede flow, diminish aspiration efficiency, and elevate the risk of vessel trauma.19,20
Although our findings suggest that CTA overestimation could result in over-selection of device size, we agree that the clinical impact remains speculative. This study did not assess outcomes related to device performance or complication rates based on imaging modality, and therefore cannot assert that the CTA-DSA discrepancy should alter current EVT practice. Rather, our data serve as a cautionary insight into measurement variability and as a prompt for further investigation. We also acknowledge that undersized catheters may pose challenges, such as insufficient aspiration force or suboptimal clot engagement, highlighting the importance of avoiding both extremes of mismatch.
Future research should explore the relationship between vessel measurement accuracy and procedural outcomes, particularly in distal or borderline-caliber vessels where sizing discrepancies may be most impactful. Prospective studies incorporating AI-based vessel analysis, real-time intra-procedural sizing tools, or advanced imaging protocols may help refine device selection strategies and improve procedural efficiency and safety.
This study has several limitations. Its retrospective design introduces potential selection bias, as inclusion was based on the availability of imaging and procedural data. As a single-center study, generalizability may be limited. Despite standardized measurement protocols, inter-reader variability remains a factor, although we demonstrated good inter-rater agreement. Differences in contrast agents and injection techniques between CTA and DSA could have influenced vessel caliber measurements. Additionally, we did not control for vessel tortuosity, calcifications, or flow dynamics, all of which may alter apparent lumen diameter. Most importantly, we did not evaluate procedural or clinical outcomes in relation to device sizing. As such, the clinical implications of our findings remain uncertain and hypothesis-generating.
Conclusion
In this study, we found that CTA systematically overestimates MCA M1 vessel diameter compared to DSA, with a median difference of 0.4 mm. While this discrepancy may not affect device choice in most clinical workflows, it could be relevant in select cases—particularly when vessel size is near the threshold for different catheter calibers. These findings highlight the importance of understanding imaging-based variability in EVT planning. Further research is needed to determine whether improved vessel sizing strategies can enhance procedural outcomes.
Footnotes
Acknowledgements
The authors received no assistance or contributions to acknowledge.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.V. is a consultant for Penumbra Inc. This relationship did not influence the design, analysis, interpretation, or reporting of this study.
