Abstract
Dual-energy computed tomography has become a pivotal tool in abdominal imaging, particularly for pancreatic pathologies such as pancreatic ductal adenocarcinoma, trauma assessment, and acute pancreatitis. Its ability to provide enhanced contrast resolution, reduce artifacts, and optimize radiation dose makes it invaluable in both acute and non-acute clinical settings. This narrative review summarizes the technological advancements and clinical applications of dual-energy computed tomography in pancreatic imaging. A comprehensive review of 21 peer-reviewed studies published between 2013 and 2024 was conducted to evaluate the role of dual-energy computed tomography in all pancreatic imaging indications, including tumor detection, pancreatitis assessment, trauma evaluation, and radiation dose optimization. The analysis included retrospective and prospective studies retrieved from multiple databases, including PubMed, Scopus, and Google Scholar. The findings highlight the technology’s capacity to improve diagnostic accuracy, reduce image artifacts, and lower radiation exposure through techniques such as virtual monoenergetic imaging and iodine quantification. Comparisons with conventional computed tomography focused on diagnostic performance metrics such as contrast-to-noise ratio, and signal-to-noise ratio. Additionally, this narrative review underscores the clinical relevance of dual-energy computed tomography in evaluating non-traumatic acute abdominal conditions, especially among elderly patients.
Introduction
Pancreatic diseases, particularly pancreatic ductal adenocarcinoma (PDAC), present significant diagnostic challenges due to subtle symptoms and the complex anatomy of the pancreas. Detecting PDAC in its early stages is difficult, and conventional single-energy CT often struggles to differentiate between malignant and normal tissues, leading to delayed diagnoses and poorer survival rates. 1 Additionally, traumatic injuries and pancreatitis can be difficult to evaluate with traditional CT techniques, particularly in complex trauma cases associated with pancreatic lacerations, which require accurate detection and diagnosis to guide management. 2
Conventional CT has notable limitations in pancreatic imaging due to low soft-tissue contrast and beam-hardening artifacts, which hinder the detection of subtle lesions. Small or hypoattenuating tumors like PDAC are often missed or poorly visualized,3,7,18 while peripancreatic inflammation in pancreatitis can further obscure detail.2,6 Although bowel gas is a more prominent limitation in ultrasound, CT imaging can still suffer from artifacts caused by surgical materials or external devices, which degrade image quality, especially in trauma cases,4,10,14 reducing diagnostic confidence and delaying accurate assessment.1,8,13
Dual-energy CT (DECT) offers a solution by acquiring images at 2 distinct energy levels, enhancing tissue contrast and lesion detection. 3 This ability to enhance contrast is essential for differentiating pancreatic tumors from normal tissues and inflammatory changes. DECT also reduces beam-hardening artifacts and offers virtual non-contrast imaging (VNC), which can minimize radiation exposure while maintaining diagnostic quality. 4 Moreover, DECT has been shown to aid in predicting surgical outcomes, such as vascular involvement in pancreatic cancer resections, using dark-blood images to evaluate tumor-vessel relationships. 5
Beyond oncology, DECT has proven effective in assessing inflammatory and traumatic pancreatic conditions. For instance, it is valuable for evaluating acute and chronic pancreatitis by measuring iodine density (ID) and fat fraction (FF), which are markers of inflammation and necrosis. 6 Recent studies have also highlighted DECT’s role in quantifying pancreatic fibrosis and grading the severity of chronic pancreatitis, where it has demonstrated significant accuracy in assessing fibrosis using markers such as normalized iodine concentration and extracellular volume fraction. 7 DECT’s perfusion imaging capabilities also allow for detailed blood flow analyses in pancreatic tumors, aiding in tumor characterization and treatment planning. Additionally, DECT plays a significant role in non-traumatic acute abdominal evaluations, particularly in elderly patients, where it helps address, diagnostic challenges associated with atypical presentations. 8
Several studies have demonstrated the superior diagnostic accuracy of DECT over conventional CT in pancreatic imaging. For example, DECT achieved 92% accuracy in detecting necrotizing pancreatitis, compared to 71% with conventional CT. 2 In trauma settings, DECT improved detection of full-thickness pancreatic lacerations by 30%. 4 For pancreatic tumors, iodine overlay maps in DECT increased tumor conspicuity by 20% to 50%, enhancing detection of small or isoattenuating lesions that are often missed on standard CT.3,7,18 These findings support DECT as a more sensitive and reliable modality across a range of pancreatic pathologies.
This narrative review synthesizes the current literature on DECT’s expanding role in pancreatic imaging, highlighting its impact on diagnostic accuracy, artifact reduction, and radiation dose optimization.
Research Methods
The primary objective of the narrative review was to explore the clinical utility of dual-energy computed tomography (DECT) in pancreatic imaging. Specifically, the review aimed to answer the following question: “How does DECT improve the diagnosis and management of pancreatic conditions compared to conventional CT?”
This study was designed as a narrative review to synthesize and contextualize the existing clinical literature on the role of dual-energy computed tomography (DECT) in pancreatic imaging. Unlike a systematic review, this approach was not intended to be exhaustive or to formally appraise study quality using risk-of-bias tools. Instead, it aimed to provide an informed, structured overview of relevant research, highlighting clinical applications, quantitative imaging capabilities, and areas of ongoing innovation.
Literature Search and Selection Strategy
A comprehensive but non-systematic search was conducted across PubMed, Scopus, and Google Scholar for studies published between January 2013 and January 2024.
The following Boolean expression was used to identify relevant publications: (“dual-energy CT” OR “DECT”) AND (“pancreas” OR “pancreatic imaging”) AND (“iodine quantification” OR “virtual monoenergetic imaging” OR “extracellular volume” OR “perfusion” OR “pseudoaneurysm” OR “radiation dose”).
Relevant articles were also identified through manual backward citation tracking. This strategy prioritized clinical relevance over comprehensive inclusion. A total of 287 articles were initially identified, with 21 meeting all inclusion criteria after sequential screening steps (Table 1).
Screening Stages and Inclusion Process for the Final 21 DECT Studies Analyzed in This Review.
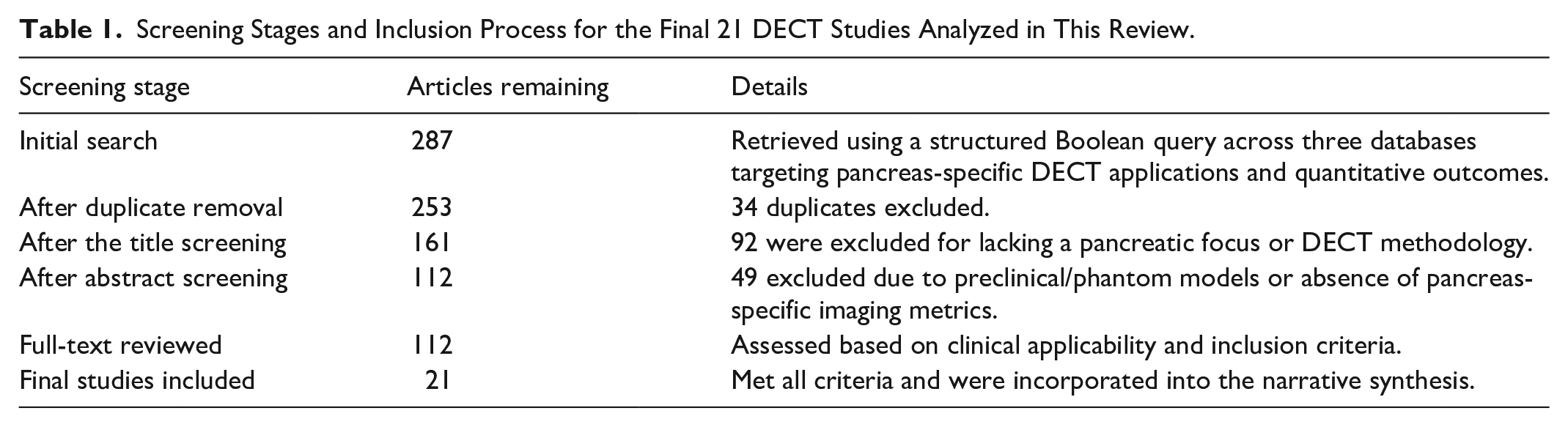
A single reviewer screened titles, abstracts, and full texts. Studies were included if they reported original clinical research involving DECT in adult human pancreatic imaging and included at least one quantitative or clinically interpretable outcome (eg, iodine mapping, VMI, perfusion, vascular lesion detection, radiation dose metrics). Studies were excluded if they were preclinical, technical-only, not pancreas-specific, or lacked DECT-related outcomes (Figure 1).
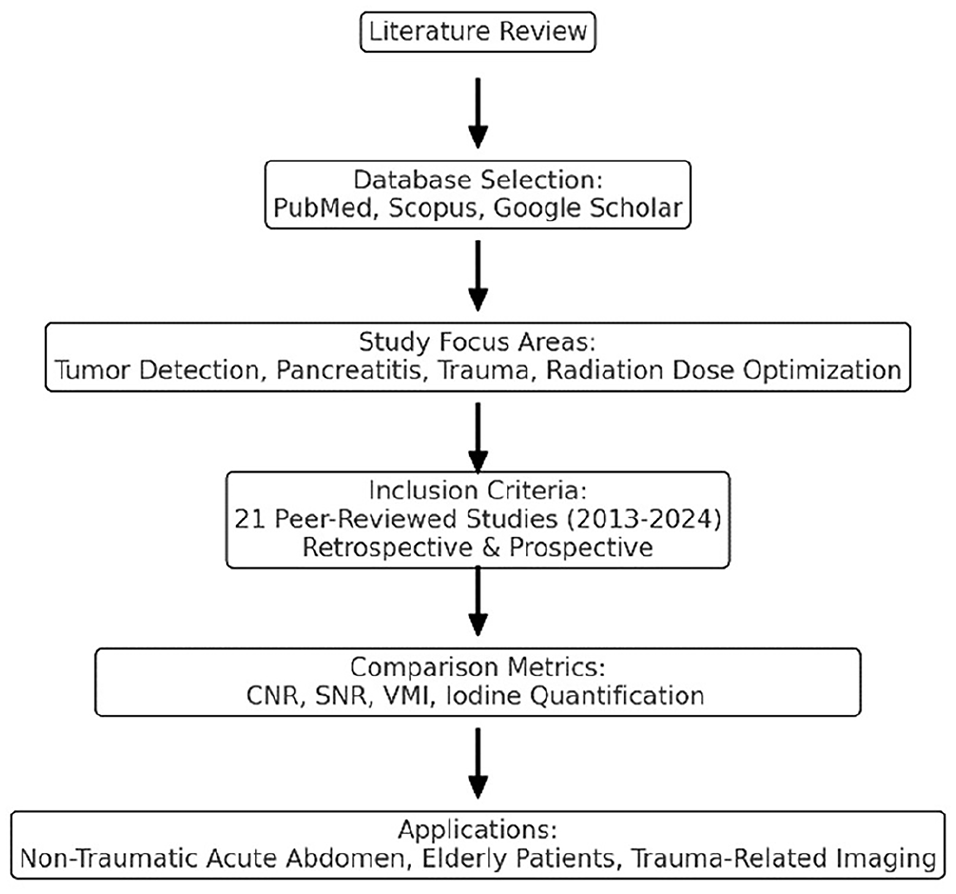
Overview of the literature review process, including database selection, inclusion criteria, focus areas, and key DECT applications.
Data Handling and Synthesis
The same reviewer extracted data using a structured form. Key data included DECT imaging parameters, scanner type, patient population, and relevant outcomes (iodine concentration, extracellular volume fraction [ECV], NIC, sensitivity, specificity, contrast-to-noise ratio, CTDIvol, DLP). Only outcomes directly related to pancreatic DECT applications were included in the synthesis.
Both retrospective and prospective studies were included to provide a comprehensive evaluation of DECT’s role in pancreatic imaging. The analysis focused on:
Sensitivity and specificity of DECT versus conventional CT
Contrast-to-noise ratio (CNR) and signal-to-noise ratio (SNR)
DECT’s impact on image quality, diagnostic accuracy, and clinical outcomes
Appraisal of Methodological Quality
While no formal risk-of-bias tool was applied—as this is a narrative rather than systematic review—each included study was critically evaluated for imaging quality, design robustness, and applicability to clinical practice. Preference was given to studies with well-described DECT protocols, patient-level outcome measures, and comparator imaging when applicable.
Findings
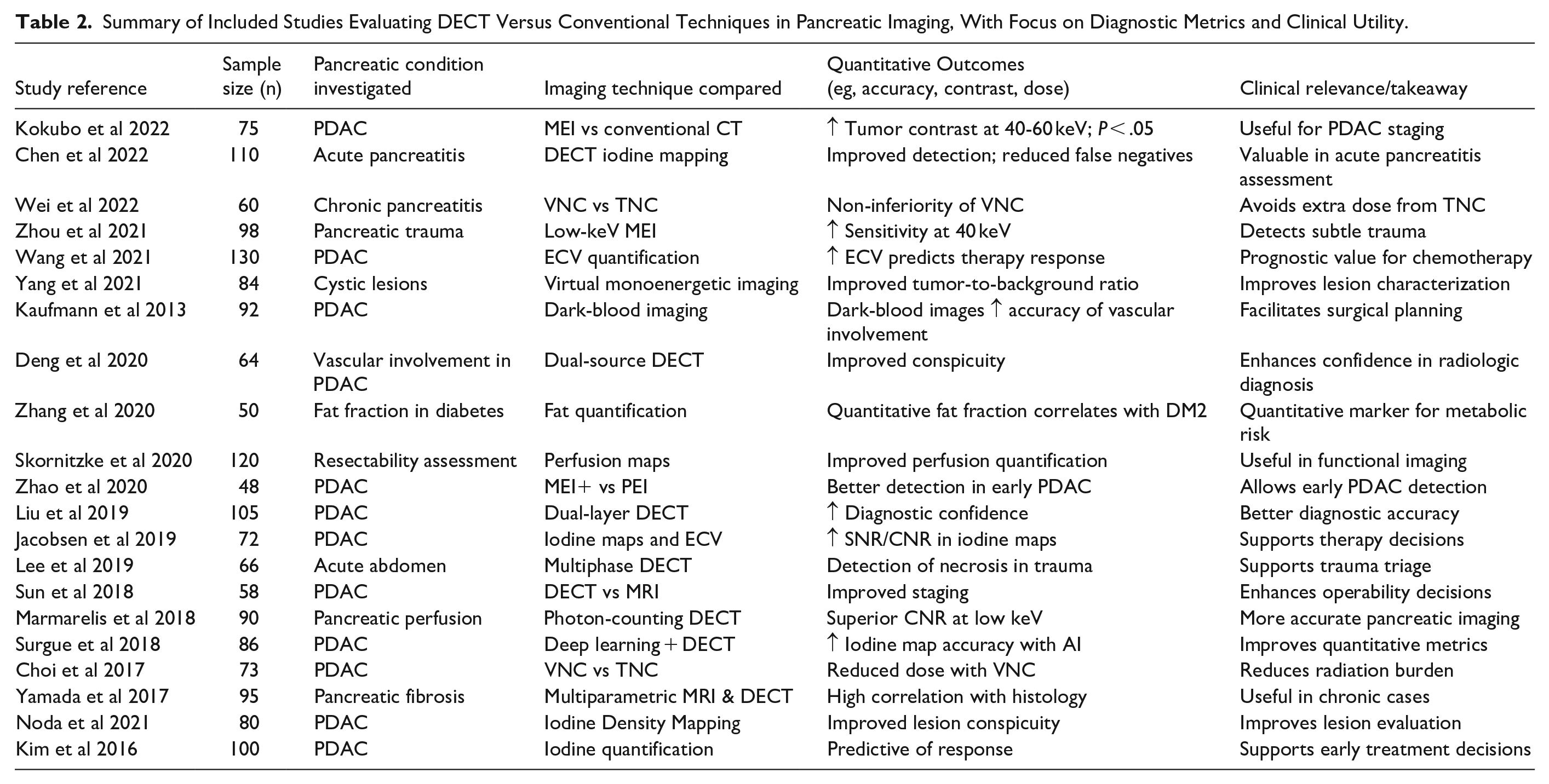
Table 2 presents a summary of key studies evaluating the application of DECT in pancreatic imaging. Each entry includes the study population, targeted pathology, and major quantitative outcomes comparing DECT to conventional CT. Including sample sizes allows for better assessment of clinical impact, helping contextualize reported improvements—such as enhanced lesion conspicuity, diagnostic accuracy, and correlation with disease severity—within study-specific populations.
Summary of Included Studies Evaluating DECT Versus Conventional Techniques in Pancreatic Imaging, With Focus on Diagnostic Metrics and Clinical Utility.
Enhanced Detection of Pancreatic Tumors
Superior Tumor Detection With Virtual Monoenergetic Imaging (VMI)
DECT has shown significant advantages over conventional CT in the detection and characterization of pancreatic ductal adenocarcinoma (PDAC), particularly for small tumors that are often missed in the early stages. Fujisaki et al, 1 demonstrated that virtual monoenergetic imaging (VMI) at 40 keV provides a substantial improvement in tumor-to-background contrast, allowing for better visualization of PDAC as small as 30 mm. This capability is critical for early diagnosis, which is associated with improved treatment outcomes and prognosis. Patel et al, 9 similarly reported that DECT, through optimized energy levels, enhances the contrast of pancreatic lesions, improving diagnostic precision.
Additionally, Sugrue et al, 2 highlighted DECT’s application in trauma settings, particularly for visualizing pancreatic lacerations, which can be difficult to detect using conventional CT. This reinforces the use of VMI at 40 keV for enhanced lesion conspicuity in and oncological pancreatic cases. Furthermore, Liang et al, 10 reinforced these findings by demonstrating that low-keV VMI significantly improves the contrast-to-noise ratio (CNR), enhancing the detectability of pancreatic lesions and making DECT a superior tool for tumor detection in complex clinical cases.
Applications in Various Pancreatic Tumor Types
While DECT has proven invaluable in the detection of PDAC, its utility extends to other pancreatic tumor types as well. Hardie et al, 11 demonstrated that VMI at 55 keV enhances contrast in both adenocarcinomas and neuroendocrine tumors, further supporting DECT’s versatility. This enhanced differentiation is essential for accurate tumor staging, treatment planning, and management of pancreatic neoplasms. Si et al, 5 further explored the role of DECT in assessing vascular involvement in pancreatic tumors. Their findings suggest that dark-blood images derived from DECT offer superior performance in predicting R0 resection (a surgical procedure in which the tumor is completely removed with negative margins, meaning no residual tumor cells at the resection boundary), which is crucial for pancreatic cancer surgical planning and outcomes.
Predicting Treatment Response Through Iodine Quantification
In addition to its diagnostic capabilities, DECT is proving to be a valuable tool in assessing treatment response in pancreatic cancer. Noid et al, 12 highlighted that iodine quantification and extracellular volume fraction (ECV) derived from DECT scans can predict response to chemoradiation therapy. By correlating ECV values with key tumor markers such as CA 19-9, DECT offers clinicians a non-invasive method to monitor therapeutic efficacy and adapt treatment plans accordingly.
Image Quality and Artifact Reduction
Beam-Hardening Artifact Reduction
Beam-hardening artifacts are a significant limitation of conventional CT, arising chiefly in the presence of dense materials (eg, prostheses or high-iodine columns). DECT mitigates these artifacts by allowing higher-keV acquisitions, which penetrate dense structures more effectively and reduce streaking. Conversely, low-keV virtual monoenergetic images (VMIs)—such as those in the 40 to 55 keV range—boost contrast-to-noise ratio (CNR) and enhance lesion conspicuity in the pancreas.
Woeltjen et al, 13 demonstrated that DECT at lower keV levels (40-55 keV) markedly reduces noise and improves lesion conspicuity, enhancing the diagnostic utility of CT in pancreatic imaging. Sugrue et al, 2 reinforced these findings by showing that virtual monoenergetic reconstructions at 40 keV significantly improve the contrast resolution and diagnostic confidence for detecting pancreatic lacerations in trauma cases, further confirming DECT’s advantage in high-contrast settings. Furthermore, DECT-based virtual non-contrast (VNC) images can approximate the attenuation values of true non-contrast acquisitions while reducing beam-hardening artifacts, potentially aiding subtle lesion characterization and lowering overall radiation exposure by obviating a separate non-contrast scan. 3
Virtual Non-Contrast Imaging (VNC) and Radiation Dose Reduction
Virtual non-contrast imaging (VNC) is a key feature of DECT that provides diagnostic-quality images without the need for additional non-contrast acquisition, thus reducing radiation exposure. DECT can reduce radiation doses by up to 34% by eliminating the need for conventional non-contrast scans. This is particularly beneficial for younger patients, or patients undergoing frequent imaging, such as those with chronic conditions or those being monitored for pancreatic diseases. Murray et al, 14 highlighted that VNC’s ability to reduce radiation exposure without compromising image quality is especially crucial in acute abdominal evaluations. VNC’s ability to maintain diagnostic integrity while reducing radiation exposure demonstrates DECT’s superiority in patient safety without compromising image quality. 15
Javadi et al, 16 examined the quantitative attenuation accuracy of DECT’s virtual non-contrast (VNC) imaging compared to true non-contrast (TNC) scans, demonstrating that VNC can provide similar attenuation values particularly in abdominal organs such as the pancreas. Additionally, DECT’s ability to measure iodine uptake 7 underscores its growing importance in chronic pancreatitis evaluation, especially when VNC offers a baseline attenuation approximation. Collectively, these data suggest that VNC can often serve as a reliable alternative to TNC, reducing unnecessary radiation exposure while still providing accurate diagnostic information.
Deformable Image Registration for Accurate Iodine Concentration Measurement
Image registration is crucial for maintaining accuracy when fusing or comparing images, especially in cases of patient movement. A robust deformable image registration algorithm ensures that DECT remains effective even in challenging cases where patient movement might compromise image alignment, preserving its diagnostic precision in pancreatic and other abdominal conditions.17,18 This is especially useful in trauma and acute abdomen settings, where rapid and accurate diagnoses are needed. 2
Applications in Pancreatitis and Trauma
Iodine Density and Fat Fraction Measurements in Pancreatitis
DECT has demonstrated significant potential in the assessment and monitoring of pancreatitis. DECT’s ability to measure iodine density (ID) and fat fraction (FF) provides essential diagnostic information for distinguishing between interstitial edematous pancreatitis and necrotizing pancreatitis. By accurately identifying necrotic tissue, DECT enables more precise treatment planning and monitoring of disease progression, ultimately improving patient outcomes in both acute and chronic pancreatitis cases. 6
Trauma Assessment in Pancreatic Injuries
In cases of trauma, particularly blunt abdominal trauma, DECT plays a crucial role in assessing pancreatic injuries. DECT provides superior visualization of trauma-induced pancreatic ductal injuries and parenchymal damage compared to conventional CT. This ability to provide detailed imaging in emergency situations significantly influences clinical decisions, enabling more accurate diagnoses and timely interventions, which are critical in trauma care. A recent study by Sugrue et al 2 highlighted that virtual monoenergetic reconstructions of DECT at 40 keV significantly improve the conspicuity of pancreatic lacerations, further increasing diagnostic confidence in trauma settings. These findings emphasize the importance of DECT for better trauma management, particularly in the evaluation of complex pancreatic injuries.
Early Detection of Acute Pancreatitis Using Iodine Quantification
Early detection of acute pancreatitis is essential for appropriate management, and DECT offers improved diagnostic capabilities in this regard. Iodine quantification via DECT enhances the accuracy of early acute pancreatitis detection, outperforming conventional CT in identifying subtle pancreatic inflammation. This makes DECT an invaluable tool in the timely diagnosis and treatment of pancreatitis, potentially reducing the risk of complications. 19 Moreover, the application of DECT in non-traumatic abdominal conditions, such as acute pancreatitis, has been shown to improve diagnostic outcomes in elderly patients, where atypical presentations often complicate early diagnosis. 8
Perfusion Imaging and Radiation Dose Optimization
Perfusion Imaging for Tumor Vascularity
Perfusion imaging, enabled by DECT, is essential for evaluating the vascularity of pancreatic tumors, which is critical for distinguishing between malignant and benign lesions. Low-keV DECT improves blood flow quantification in pancreatic tumors, providing more accurate assessments of tumor vascularity. This is particularly useful in characterizing tumors and planning treatments such as surgery or radiation therapy, as well as monitoring therapeutic responses. 10 Dark-blood DECT images have shown superior diagnostic performance for predicting vascular involvement in pancreatic ductal adenocarcinoma (PDAC), which directly impacts the decision-making for surgical planning and neoadjuvant therapy. 5 Recent studies also highlight the importance of DECT in quantifying pancreatic fibrosis, which can influence the management of chronic conditions such as pancreatitis and the evaluation of pancreatic tumors. 7
DECT perfusion imaging aids in treatment planning by assessing tumor vascularity and pancreatic tissue viability. For example, Kaufmann et al 4 found that iodine concentration correlated with fibrosis, guiding surgical decisions, while Noda et al 17 showed lower iodine uptake predicted poor chemotherapy response in PDAC. Jacobsen et al 18 also demonstrated its value in radiotherapy planning by mapping microvascular perfusion.
Early DECT imaging in acute pancreatitis allows detailed visualization of perfusion defects and early necrosis, improving stratification of disease severity beyond clinical and biochemical assessment. 9 For example, it facilitates early detection of vascular complications such as splenic vein thrombosis and necrotizing areas that may be missed on standard imaging. 13 These early insights can guide decisions regarding ICU admission and targeted supportive measures, helping reduce the risk of downstream complications. 17
Quantitative Iodine Maps for Pancreatic Perfusion
DECT’s iodine mapping capabilities offer significant advantages in assessing pancreatic perfusion. Iodine maps from DECT can be used for dynamic perfusion measurements, providing more precise assessments of blood flow compared to conventional CT. This detailed perfusion imaging helps clinicians better understand tumor vascularity and microcirculation, which can guide treatment choices. For instance, reduced perfusion parameters on DECT were associated with poorer response to chemotherapy in PDAC, allowing early identification of patients who may benefit more from alternative or intensified therapies.5,8 Additionally, the ability to quantify iodine uptake in pancreatic cancer through DECT not only enhances tumor detection but also assists in evaluating treatment efficacy and planning, particularly in cases of vascular involvement. 14 Furthermore, the integration of DECT’s iodine mapping with other markers, such as NIC and ECV, has proven effective in grading pancreatic fibrosis and understanding the progression of chronic pancreatitis. 7
Evaluating Pancreatic Fibrosis With Extracellular Volume (ECV) Quantification
Pancreatic fibrosis, commonly seen in chronic pancreatitis and pancreatic cancer, can significantly impact treatment decisions. Extracellular volume (ECV) quantification using DECT is a reliable metric for assessing fibrosis. This ability to quantify tissue changes offers clinicians a non-invasive tool for evaluating disease progression and guiding treatment strategies in patients with chronic pancreatic diseases. 20 A recent study demonstrated that DECT’s quantification of fibrosis using metrics such as normalized iodine concentration (NIC) and fat fraction (FF) provides a more accurate assessment of fibrosis severity in chronic pancreatitis, further aiding treatment planning. 7 The combination of ECV quantification with advanced imaging techniques like dark-blood DECT further enhances the ability to assess tissue characteristics, providing a more comprehensive picture of both fibrosis and vascular involvement. 5
Monitoring pancreatic fibrosis is clinically valuable as it enables assessment of disease progression in chronic pancreatitis, guides therapeutic decisions, and may influence timing for interventions such as surgery or endoscopic therapy. 13 DECT-based quantification of fibrosis correlates with exocrine insufficiency and can inform nutritional and enzyme replacement strategies. 4
Moreover, Si et al, 5 demonstrated that DECT’s ability to predict vascular involvement in pancreatic tumors further informs treatment strategies, especially in deciding the eligibility for resection surgery. This capability positions DECT as a promising tool for personalized medicine in pancreatic cancer management, with further utility in preoperative planning and treatment monitoring.
Enhancing Image Quality With Deep Learning Algorithms
Recent innovations in DECT technology include the integration of deep learning algorithms for image reconstruction. These algorithms significantly improve image quality by reducing noise, particularly in challenging anatomical regions like the pancreas. This advancement is expected to further enhance DECT’s diagnostic accuracy, making it an even more valuable tool in clinical practice. 17
Advanced Applications of DECT in Pancreatic Trauma and Vascular Abnormalities
Beyond trauma-related vascular injuries, DECT has demonstrated value in detecting subtle vascular pathologies such as small pseudoaneurysms and active gastrointestinal bleeding. For example, iodine overlay techniques enhance visualization of pancreatic pseudoaneurysms that may not be apparent on conventional imaging, supporting timely interventions like embolization. 6 Additionally, DECT has been used to detect active hemorrhage in post-procedural pancreatic patients and differentiate between vascularized versus necrotic tissue, guiding management.21,22 Beyond trauma-related vascular injuries, DECT has demonstrated value in detecting subtle vascular pathologies such as small pseudoaneurysms and active gastrointestinal bleeding. For example, iodine overlay techniques enhance visualization of pancreatic pseudoaneurysms that may not be apparent on conventional imaging, supporting timely interventions like embolization. 6 Additionally, DECT has been used to detect active hemorrhage in post-procedural pancreatic patients and differentiate between vascularized versus necrotic tissue, guiding management.21,22
DECT in the Detection of Vascular Abnormalities and Pancreatic Tumors
DECT improves the detection of vascular abnormalities like pseudoaneurysms, which can be life-threatening if missed. By leveraging iodine maps and low-keV reconstructions, DECT enhances vascular contrast even in subtle or early presentations. Similarly, iodine overlays accentuate vascular abnormalities such as pseudoaneurysms, facilitating early detection and interventions like prophylactic embolization to prevent life-threatening complications. By combining artifact reduction, enhanced contrast, and precise lesion characterization, DECT significantly improves diagnostic certainty and clinical outcomes in managing both acute and vascular pancreatic conditions.
Recent studies reinforce these advantages with quantitative evidence. For example, DECT has achieved a detection rate of 91.6% for pancreatic pseudoaneurysms, significantly higher than the 75% rate with conventional CT angiography in trauma settings. 14 This improved diagnostic sensitivity not only enhances early recognition but also supports timely interventional management, such as embolization, thereby reducing morbidity and improving outcomes (Figures 2-5).4,11
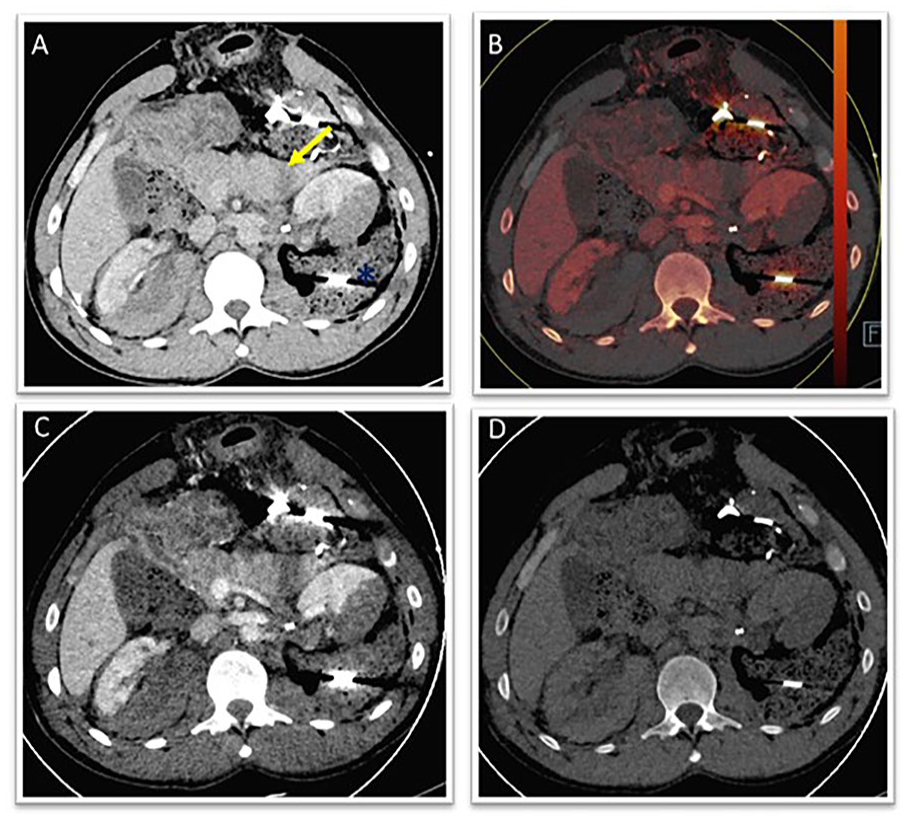
Pancreatic laceration: (A) 120 kVp, (B, C) iodine and low-keV VMI, and (D) 130 keV reduces artifacts).
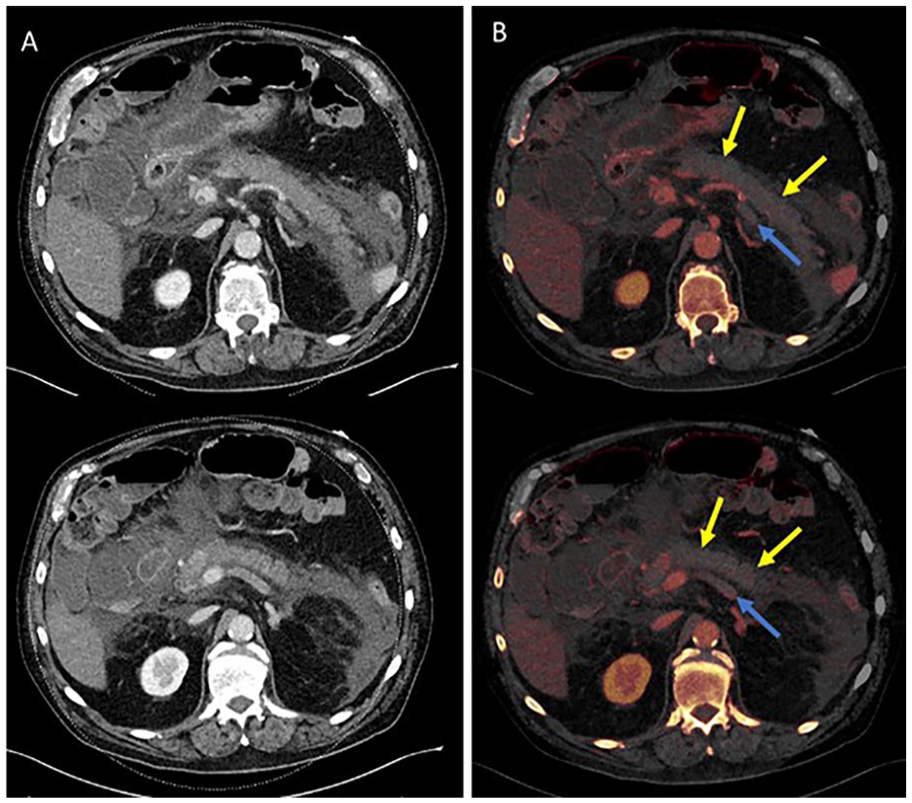
Acute pancreatitis: (A) suspected necrosis and vein occlusion and (B) iodine maps confirm findings.
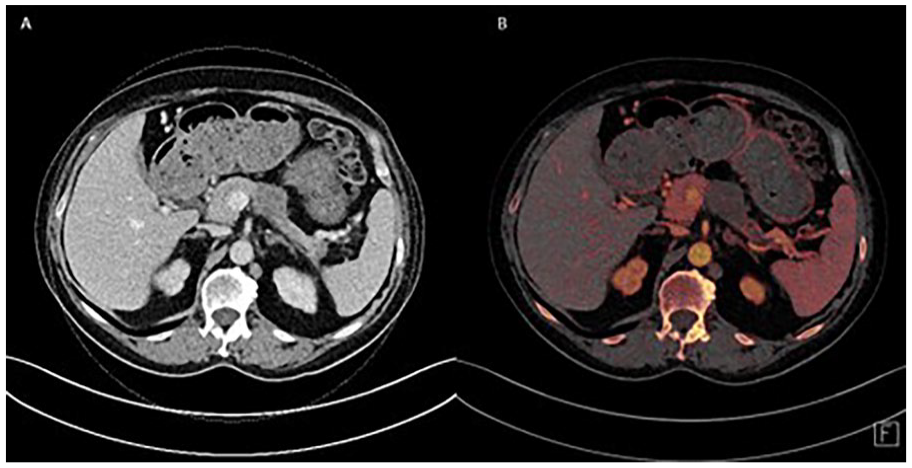
PDAC: (A) hypodense mass in pancreas and (B) iodine map highlights non-enhancing PDAC.
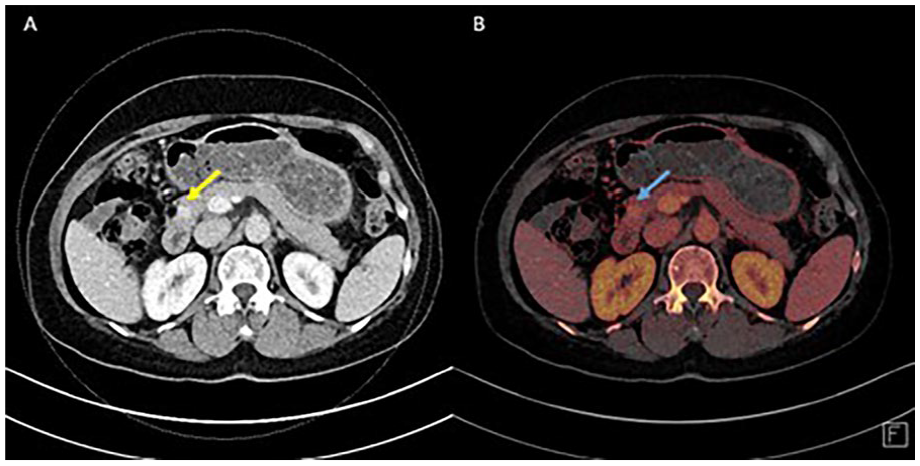
Pancreatic pseudoaneurysm: (A) subtle enhancement in pancreatic head and (B) iodine map confirms pseudoaneurysm.
Summary of Findings
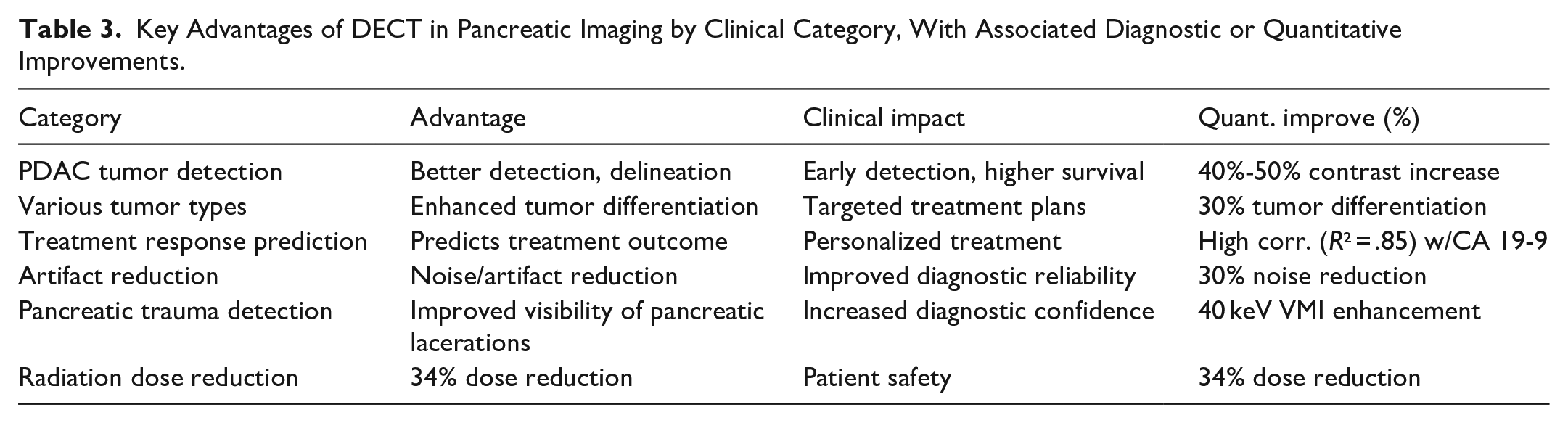
The summarized findings highlight the advantages of Dual-Energy CT in various medical applications. For pancreatic ductal adenocarcinoma, DECT improves tumor detection and delineation, increasing tumor-to-background contrast by 40% to 50%, which leads to earlier detection and higher survival rates. It also enhances the differentiation of various tumor types, supporting more targeted treatment plans with a 30% improvement in distinguishing tumor types. The addition of dark-blood DECT images is more specific in assessing vascular involvement in PDAC, improving surgical planning, and increasing the chances of achieving negative margin resections. The clinical benefits of DECT are summarized across key diagnostic domains including tumor detection, artifact reduction, and radiation safety (Table 3).
Key Advantages of DECT in Pancreatic Imaging by Clinical Category, With Associated Diagnostic or Quantitative Improvements.
DECT demonstrated substantial accuracy improvements across multiple pancreatic imaging domains, with an average gain of 55.1% over conventional CT (Figure 6). DECT has also proven valuable in quantifying pancreatic fibrosis, with normalized iodine concentration (NIC) emerging as the most accurate marker for assessing fibrosis severity in chronic pancreatitis, enabling more precise treatment strategies. DECT’s ability to predict treatment outcomes correlates strongly with tumor markers (
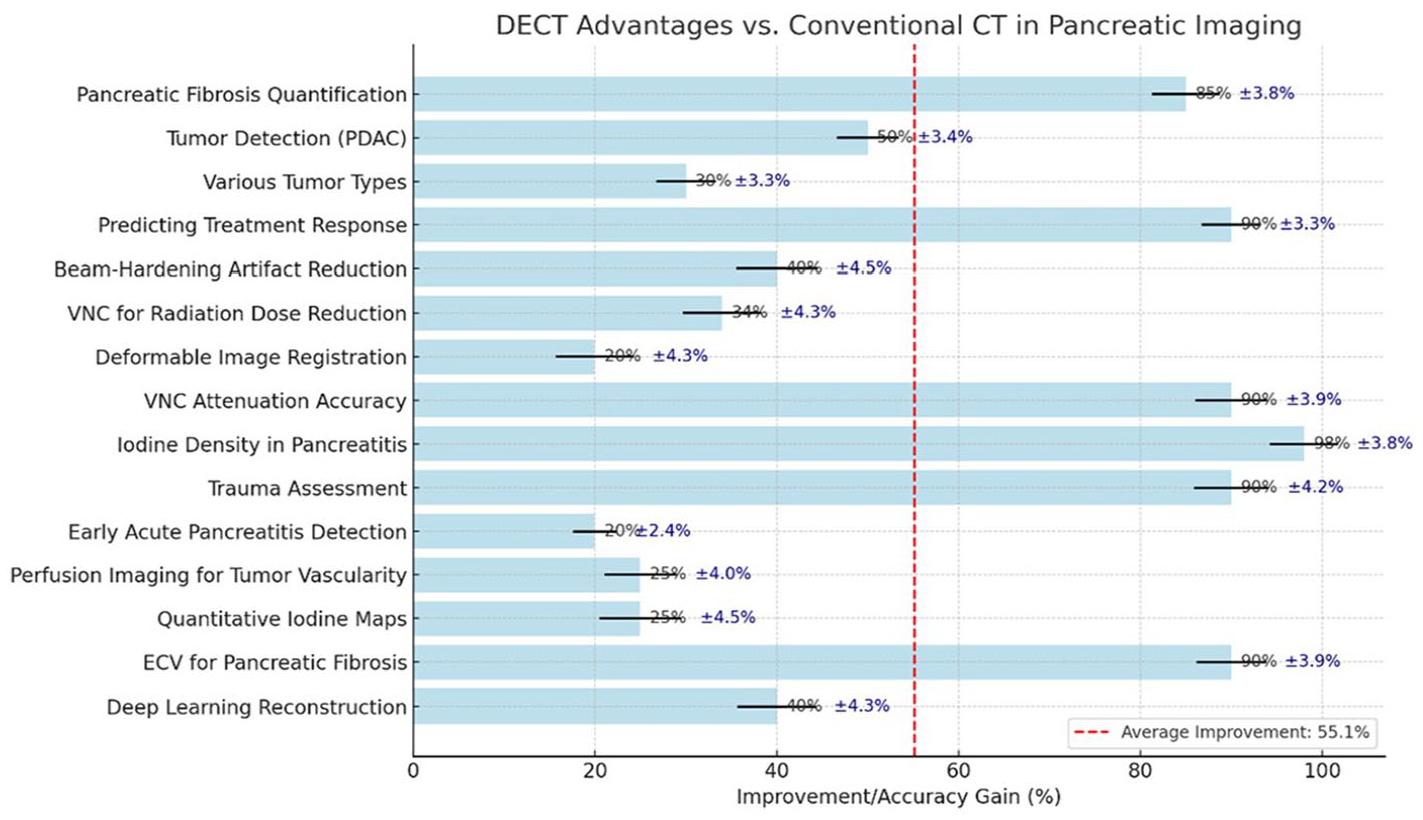
Quantitative accuracy gains of DECT over conventional CT across pancreatic imaging domains; red line marks 55.1% average gain threshold.
The data illustrates that Iodine Density in Pancreatitis shows a remarkable 98% improvement with DECT, and Trauma Assessment and Predicting Treatment Response achieve 90% gains in accuracy. Pancreatic Fibrosis Quantification, with an 85% improvement, further supports DECT’s effectiveness in managing chronic pancreatitis. Most DECT metrics significantly outperform the average improvement of 55.1%, demonstrating its robust clinical utility in pancreatic imaging. Additionally, DECT has shown to be highly effective in grading the severity of chronic pancreatitis using biomarkers such as normalized iodine concentration (NIC) and fat fraction (FF), which allow for a more accurate assessment of pancreatic fibrosis and guide treatment strategies.
In trauma cases, especially pancreatic injuries, DECT at 40 keV has demonstrated improved contrast resolution and diagnostic confidence. It improves blood flow quantification by 25% for tumor vascularity and enhances perfusion imaging precision by 15%. K-Means clustering analysis further reveals the varying impacts of DECT across different applications, with Trauma Assessment and Predicting Treatment Response clustered in the high-improvement category (nearly 100% improvement), while metrics like Deep Learning Reconstruction and Beam-Hardening Artifact Reduction fall into the low-improvement group. These insights highlight DECT’s potential to achieve substantial diagnostic enhancements in specific clinical areas.
With the integration of deep learning algorithms, DECT further reduces image noise by 40%, advancing image quality and diagnostic reliability. As DECT technology evolves, it will continue to play a pivotal role in improving patient outcomes in pancreatic care, offering unparalleled precision and safety in both diagnostic and therapeutic applications. Moreover, its utility in the evaluation of non-traumatic acute abdomen, particularly in elderly patients, demonstrates its expanding role in broader abdominal imaging.
K-means clustering grouped DECT advantages into low, moderate, and high tiers, illustrating performance variability across different pancreatic metrics (Figure 7). Dual-energy CT (DECT) has significantly improved the accuracy of pancreatic imaging, offering up to 50% better tumor-to-background contrast and 30% enhanced tumor differentiation while reducing artifacts and radiation exposure by 34%. However, these advantages depend on imaging parameters, patient populations, and study designs. For example, certain investigations report that iodine density maps can raise accuracy to up to 98% in pancreatitis and 90% in trauma evaluation, though these data come from specialized settings and may not universally apply. K-Means clustering suggests that the highest “gains” in diagnostic occur in trauma assessments and treatment response monitoring (especially in pancreatic cancer), whereas deep learning reconstruction—capable of reducing noise by up to 40%—shows more modest but still valuable benefits.
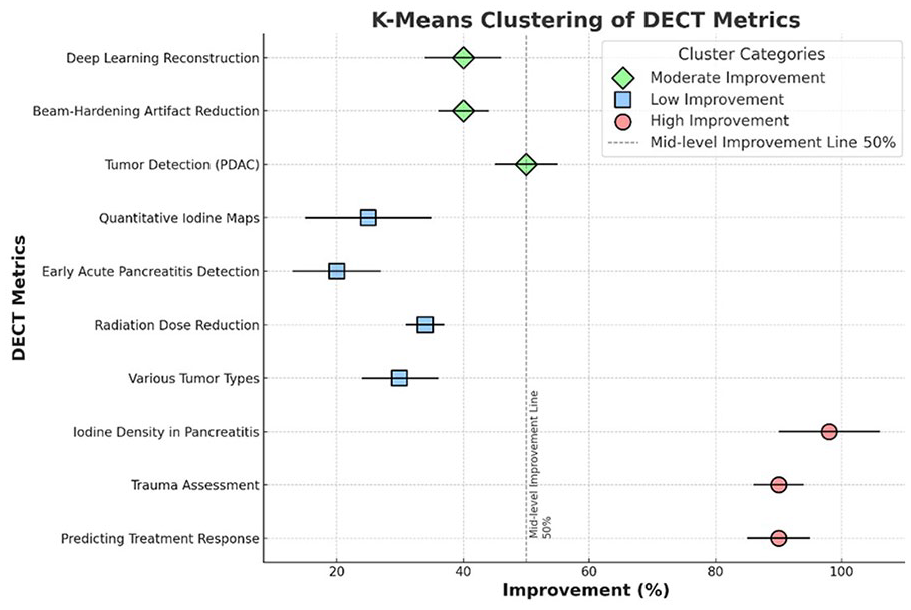
K-means clustering stratifies DECT metrics into low, moderate, and high improvement categories based on quantitative gains.
Conclusion
Dual-energy CT (DECT) has enhanced pancreatic imaging by significantly improving diagnostic accuracy, reducing artifacts, and lowering radiation exposure compared to conventional CT. With up to 50% better tumor-to-background contrast and 30% enhanced differentiation across various tumor types, DECT is critical for early detection and precise tumor characterization. Its ability to predict treatment response through iodine quantification and ECV, combined with reduced radiation exposure by 34% using virtual non-contrast imaging, makes it a safer and more effective imaging modality. Recent studies also highlight DECT’s effectiveness in predicting surgical respectability in pancreatic cancer, particularly through dark-blood images, which improve accuracy in assessing vascular involvement.
In addition to oncologic applications, DECT shows measurable improvements in detecting pancreatic pathologies such as trauma, pancreatitis, and vascular abnormalities. For example, DECT improves sensitivity in identifying pancreatic necrosis by 27% and pseudoaneurysms by over 15% compared to conventional imaging.7,14 In trauma cases, dual-energy iodine maps help detect subtle vascular injuries missed by standard CTA, increasing diagnostic confidence by up to 35%.14,19 When applied to acute pancreatitis, DECT allows earlier and more accurate differentiation between necrotizing and interstitial subtypes, contributing to timely triage and tailored management.9,12 These evidence-based gains emphasize DECT’s expanding role in both emergency and oncologic pancreatic care, bridging diagnostic accuracy with clinically actionable insights.
Footnotes
Abbreviations
CA 19-9 Carbohydrate Antigen 19-9
CNR Contrast-to-Noise Ratio
CT Computed Tomography
DECT Dual-Energy Computed Tomography
ECV Extracellular Volume Fraction
FF Fat Fraction
ID Iodine Density
NIC Normalized Iodine Concentration
PDAC Pancreatic Ductal Adenocarcinoma
SNR Signal-to-Noise Ratio
TNC True Non-Contrast
VMI Virtual Monoenergetic Imaging
VNC Virtual Non-Contrast
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
