Abstract

Introduction
Coronavirus disease 2019 (COVID-19) is associated with adverse cardiovascular outcomes and long-term symptoms including post-acute sequelae of SARS-CoV-2 infection (PASC).1-3 Multiple inflammatory and endothelial cell activation biomarkers are associated with mortality in acute COVID-19. 4 However, there is limited understanding of longer-term changes to vascular function after COVID-19 and the relationship to cardiac symptoms. The purpose of this study was to evaluate non-invasive imaging and blood biomarkers of vascular inflammation and endothelial cell activation after recovery from COVID-19.
Methods
This prospective cohort study was approved by the research ethics board and written informed consent was obtained. Adult patients (≥18 years of age) who had a positive polymerase chain reaction SARS-CoV-2 test of nasopharyngeal swabs were invited for cardiovascular evaluation 1 to 3 months post-test (early post-COVID) or 3 to 6 months post-test (late post-COVID) and compared with a control group with negative SARS-CoV-2 test and no history of COVID-19. Cardiovascular symptoms assessed included chest pain and shortness of breath.
Participants in the early post-COVID and control groups underwent combined 18F-FDG-PET/MRI after diet preparation to suppress physiologic myocardial glucose uptake (Biograph mMR, Siemens Healthineers, Germany). Participants in the late post-COVID group underwent the MRI portion of the protocol only on the same MRI unit. 2 The MRI protocol included 4D-flow time resolved angiography acquired in coronal orientation centred over the thoracic aorta (2.5 × 2.5 × 2.5 mm3 spatial resolution; 36-40 ms temporal resolution; velocity encoding 250 cm/s) starting 5 minutes after injection of 0.15 mmol/kg Gadobutrol (Bayer Healthcare, Germany). After automatic 4D-flow segmentation of the thoracic aorta, peak systolic wall shear stress (WSS), pressure gradient, and speed were quantified in the ascending aorta at a level with the highest peak flow velocity (Arterys Cardio AIMR, San Francisco, USA). 5 Multiple planes were evaluated to ensure peak velocity and WSS were captured. Thoracic aortic inflammation was categorized as positive if aortic FDG-uptake was greater than or equal to FDG-uptake in the liver. 6
Peripheral blood samples were collected on the same day as the PET/MRI or MRI and processed within 3 hours after collection. Circulating levels of angiopoietin-2 (Ang-2), E-Selectin, intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion protein-1 (VCAM-1), endothelin-1, interleukin-6, interleukin-8, and soluble triggering receptor expressed on myeloid cells-1 (TREM-1) were quantified in platelet free plasma samples according to manufacturer’s protocol (Simple Plex Ella, ProteinSimple, San Jose, USA) with values reported as the average of triplicate readings. 7
Statistical analysis included one-way analysis of variance, Kruskal-Wallis test, and Fisher exact test with post hoc tests for significance using Bonferroni correction; Spearman correlation; and linear regression (STATA software v14., StataCorp, Texas). All tests were 2-tailed, and P values less than .05 were considered statistically significant.
Results
Seventy-six participants were included, 22 early post-COVID (43 ± 12 years, 55% male), 40 late post-COVID (43 ± 14 years, 38% male), and 14 controls (41 ± 14 years, 36% male), Table 1. Persistent cardiovascular symptoms were reported by 36% and 50% of early and late post-COVID groups, respectively.
Clinical, Imaging, and Blood Biomarkers.
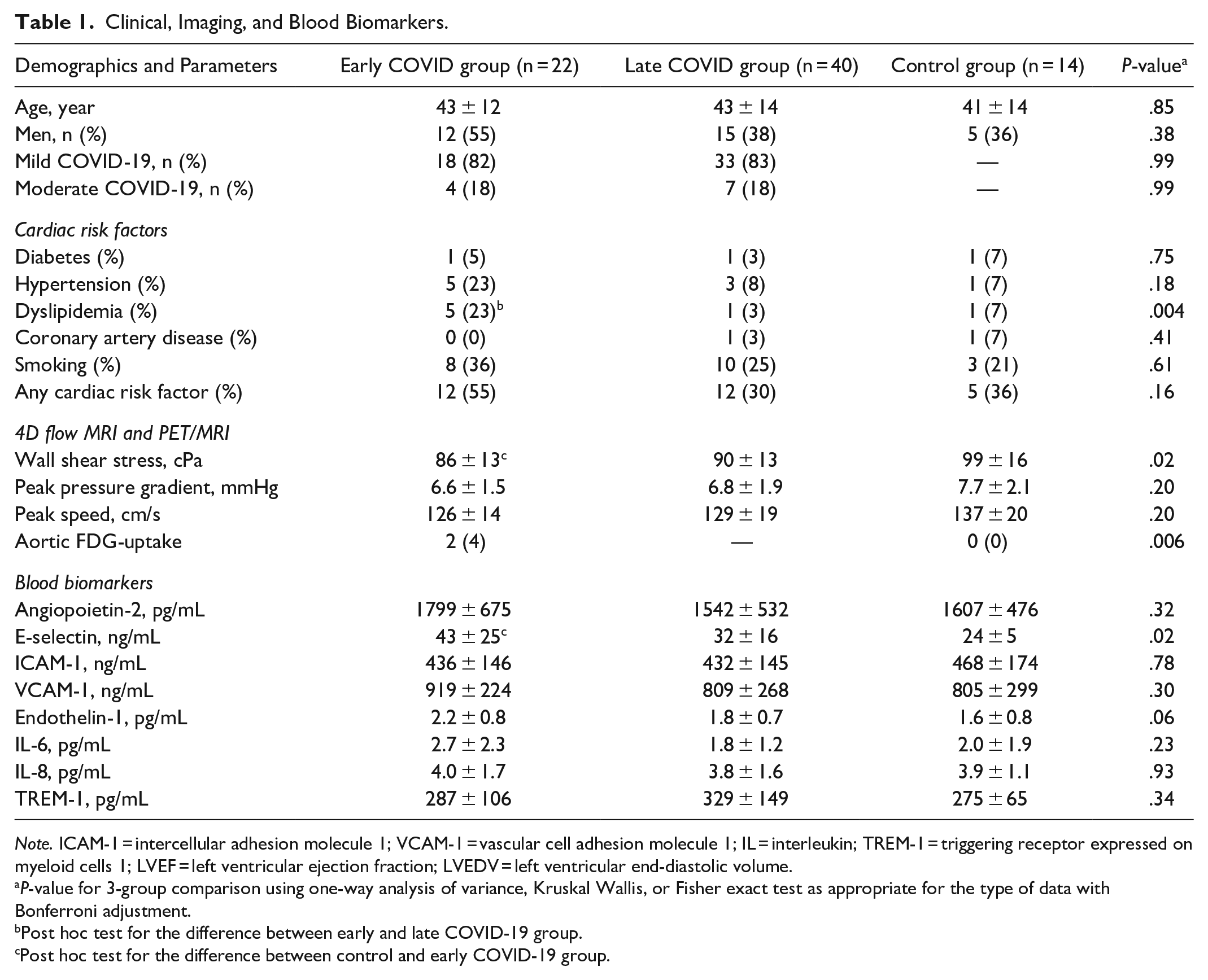
Note. ICAM-1 = intercellular adhesion molecule 1; VCAM-1 = vascular cell adhesion molecule 1; IL = interleukin; TREM-1 = triggering receptor expressed on myeloid cells 1; LVEF = left ventricular ejection fraction; LVEDV = left ventricular end-diastolic volume.
P-value for 3-group comparison using one-way analysis of variance, Kruskal Wallis, or Fisher exact test as appropriate for the type of data with Bonferroni adjustment.
Post hoc test for the difference between early and late COVID-19 group.
Post hoc test for the difference between control and early COVID-19 group.
Peak WSS was lower and E-selectin was higher in the early post-COVID group compared to controls (86 ± 13 cPa vs 99 ± 16 cPa, P = .022 and 43 ± 25 ng/mL vs 24 ± 5 ng/mL, P = .018, respectively), Figure 1. These differences remained significant in multivariable models adjusted for age, sex, and cardiovascular risk factors (B-coefficient −11.8 cPa, 95% CI −21.2, −2.4, P = .015 and 15.9 ng/mL, 95% CI 2.5, 29.3, P = .021, respectively). In unadjusted analysis, neither WSS nor E-selectin differed significantly between controls and the late COVID-19 group (P = .16 and P = .64, respectively). However, in a multivariable model, WSS was lower in the late post-COVID group compared to controls (B-coefficient −9.0 cPa, 95% CI −17.5, −0.5, P = .038). Mild thoracic aortic FDG-uptake was present in 2 early post-COVID participants (4%) but was not present in any of the controls (0%, P = .006). None of the other MRI parameters or blood biomarkers differed significantly between groups. WSS correlated significantly with E-selectin (r = −.31, P = .01) but not with the other blood biomarkers.
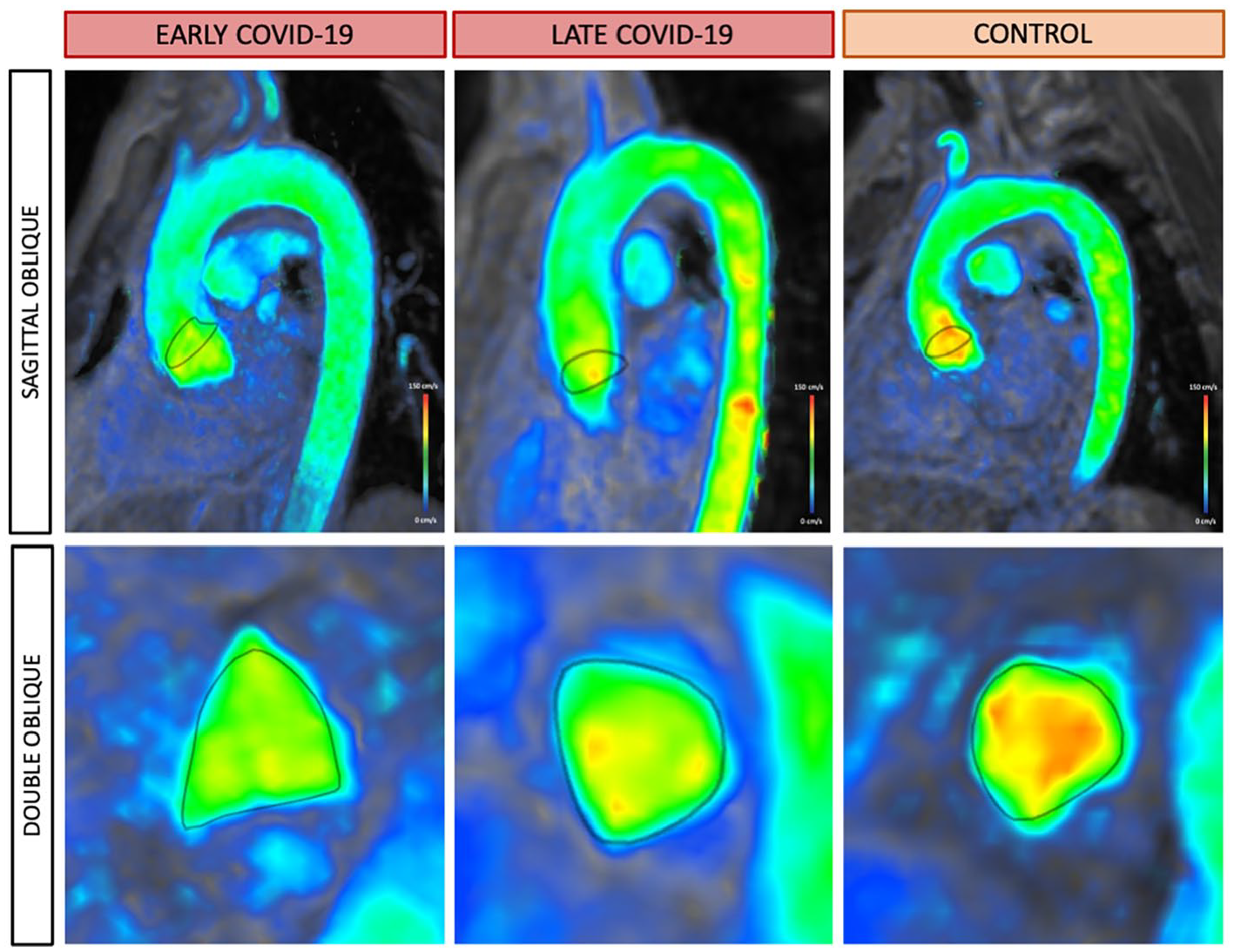
4D-flow velocity images in a patient recently recovered from COVID-19 (early post-COVID group), a patient 3 to 6 months post-COVID-19 (late post COVID group), and control. Peak wall shear stress (WSS) was assessed at the ascending aorta with region of interest (ROI) in dark grey shown in sagittal oblique (top) and double oblique (bottom) planes. Green indicates lower WSS values (peak 76 cPa in the early post-COVID patient) and orange indicates higher WSS values (peak 98 cPa in the control patient).
In both early and late post-COVID groups, there were no differences between participants with and without cardiac symptoms with respect to WSS (85 ± 18 cPa vs 86 ± 10 cPa, P = .80 and 93 ± 13 cPa vs 88 ± 15 cPa, P = .30, respectively) or E-selectin (39 ± 20 ng/mL vs 45 ± 28 ng/mL, P = .59 and 28 ± 8 ng/mL vs 35 ± 19 ng/mL, P = .31, respectively).
Discussion
In patients recovered from mild to moderate COVID-19, vascular inflammation is identified in a minority of patients at 1 to 3 months and endothelial activation detected by elevated E-selectin is identified at 1 to 3 months with recovery by 3 to 6 months, while abnormal low WSS may persist for up to 6 months. Neither WSS nor E-selectin were associated with persistent symptoms.
Low WSS and elevated E-selectin are associated with initiation and progression of atherosclerotic disease and endothelial dysfunction in other patient populations.8,9 Thus, vascular inflammation and endothelial dysfunction early after COVID-19 could potentially accelerate atherosclerotic disease as a mechanism underlying longer term risk of cardiovascular disease. In the absence of additional risk factors, endothelial resistance may help restore cardiovascular function post-COVID.
Limitations include a modest sample size and potential for selection bias given that participants volunteered for the study. Further study is needed to evaluate the association of WSS and E-selectin with larger cohorts of patients with PASC and long-term major adverse cardiovascular events after COVID-19.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from the Joint Department of Medical Imaging Academic Incentive Fund, Peter Munk Cardiac Center Innovation Committee, and Ted Rogers Center for Heart Research.
