Abstract
Bariatric surgery is an effective approach to management of morbid obesity. Given its increasing popularity worldwide, post-operative imaging of patients following bariatric surgery is frequently encountered in clinical practice. In this article we review the physiological principles, normal post-operative anatomy, and imaging appearance of common bariatric procedures (Roux-en-Y gastric bypass, vertical sleeve gastrectomy, and laparoscopic adjustable gastric banding) as well as less common bariatric procedures (including intragastric balloon and biliopancreatic diversion with duodenal switch). Technical considerations and imaging pearls for identifying perioperative and late complications following bariatric procedures will be illustrated through fluoroscopic upper GI studies and computed tomography case examples. Familiarity with expected anatomy and imaging appearances following bariatric procedures allows the radiologist to play a key role in early identification and management of postoperative complications.
Introduction
The prevalence of obesity has doubled over the last few decades, affecting an estimated 36% of adults over the age of 20 in the United States.1,2 Obesity is strongly associated with increased cardiometabolic risk including type 2 diabetes and cardiovascular disease,3,4 and also increases the risk of sleep apnea, gastroesophageal reflux disease (GERD), cancer, osteoarthritis, and early death. 5 Bariatric surgery is an effective treatment approach for morbid obesity and for obese patients with poorly controlled type 2 diabetes,6,7 with an estimated 252 000 bariatric procedures performed in the United States in 2018. 8 Despite increasing popularity of glucagon-like peptide-1 receptor agonists (GLP-1 RA) for weight loss and cardiovascular benefits in patients with severe obesity and type 2 diabetes, studies suggest that bariatric surgery remains an important treatment option. Compared to GLP-1 RA treatment, bariatric surgery was associated with lower risk of major cardiovascular adverse events, 9 and among individuals with a diabetes duration of 10 years or less, was associated with lower all-cause mortality. 10 Radiologists must be familiar with the altered gastrointestinal anatomy following bariatric surgery as well as the unique post-procedural complications in this patient population.
Bariatric surgical procedures may be categorized based on the mechanism of weight loss. Restrictive procedures decrease gastric volume to induce early satiety, and these include vertical sleeve gastrectomy (SG), laparoscopic adjustable gastric banding (LAGB), non-adjustable gastric banding (NABG), and vertical-banded gastroplasty (VBG). Malabsorptive procedures bypass portions of the small bowel to decrease nutrient absorption. Biliopancreatic diversion without or with duodenal switch is classically categorized as malabsorptive, however this procedure often includes a partial gastrectomy (a restrictive component). Combined restrictive and malabsorptive procedures, the most common of which is the Roux-en-Y gastric bypass (RYGB), result in weight loss by both reducing gastric volume and bypassing portions of small bowel, thus decreasing absorptive surface area.
The most common bariatric surgery performed in the United States is SG, accounting for 61% of procedures, followed by RYGB which accounts for 17%. 8 On the other hand, vertical-banded gastroplasty, LAGB, and biliopancreatic diversion are less commonly performed due to relatively higher re-operation rates, lower long-term efficacy and poorly tolerated adverse effects. 8 Here we aim to discuss and illustrate expected imaging appearances as well as early and late complications following bariatric surgery.
Roux-en-Y Gastric Bypass (RYGB)
RYGB involves creation of a small 15 to 30 mL gastric pouch from the proximal stomach, excluding the remainder of the stomach and duodenum from the path of food (Figure 1). The gastric pouch may be completely transected and separated from the excluded stomach, or separated by a staple line. 11 The excluded stomach and duodenum form the biliopancreatic (afferent or biliary) limb, which channels biliary and pancreatic secretions. The Roux (efferent or alimentary) limb through which food passes is formed by bringing up a segment of jejunum in either a retrocolic retrogastric or antecolic antegastric fashion, which is anastomosed to the gastric pouch in a side-to-side manner (gastro-jejunal anastomosis) (Figure 2A and B). The retrocolic approach requires creation of an opening in the transverse mesocolon, while the antecolic approach results in placement of the Roux limb anterior to the transverse colon. The Roux and biliopancreatic limbs meet at a Y-shaped side-to-side or end-to-side jejuno-jejunal (J-J) anastomosis, typically located in the left mid-to-upper abdomen.
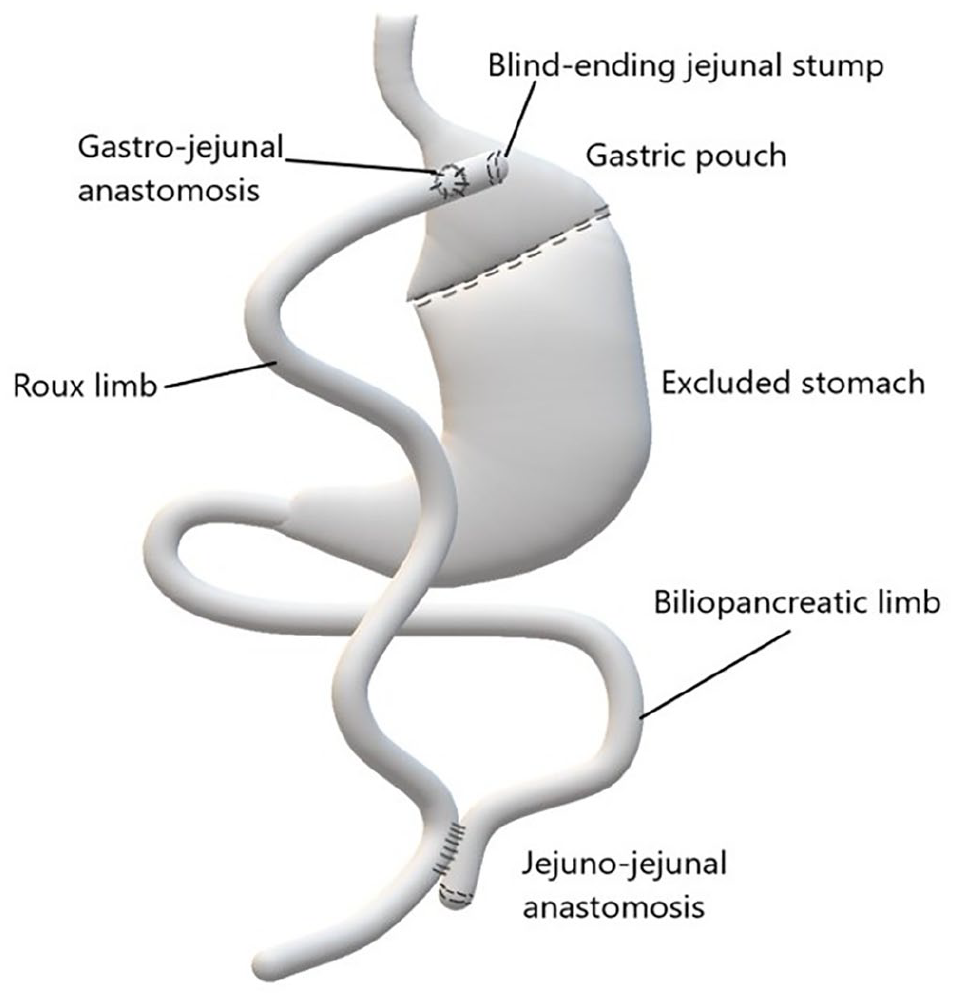
Schematic illustration depicting normal post-surgical anatomy following Roux-en-Y gastric bypass surgery.
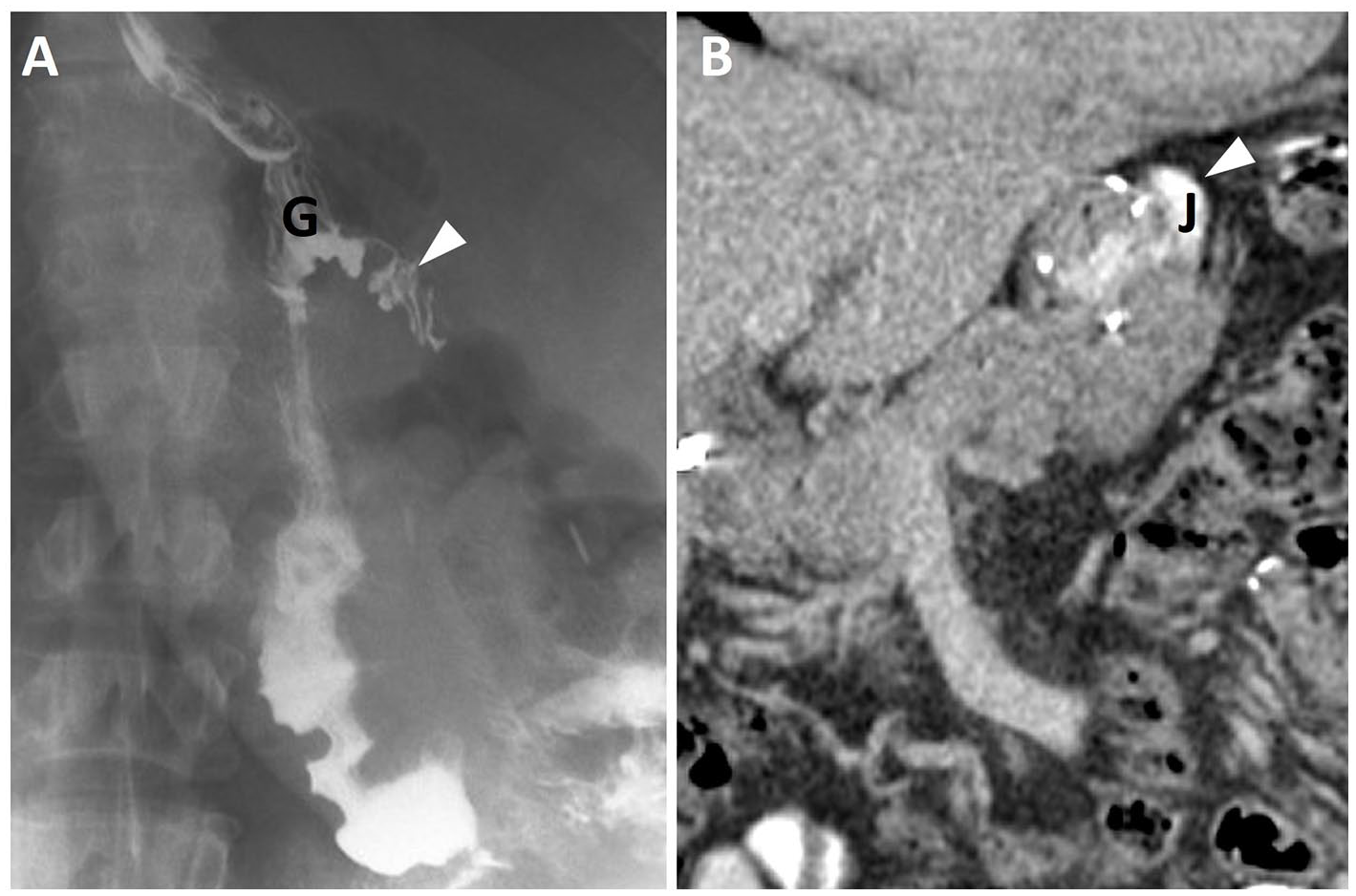
Roux-en-Y gastric bypass, normal appearance at fluoroscopy and CT. (A) Fluoroscopic upper gastrointestinal imaging (UGI) spot image reveals a small gastric pouch (denoted as G) with contrast filling of a short blind ending tubular structure demonstrating small bowel fold pattern, in keeping with the jejunal blind end related to side-to-side gastro-jejunal anastomosis (arrowhead), which may be mistaken for contrast leak. (B) Coronal contrast enhanced CT (CECT) acquired with oral contrast show the contrast opacified Roux limb (J), surgical staple line, and jejunal blind end depicted on the coronal image (arrowhead).
Studies suggest higher rates of early adverse events and hospitalization following RYGB as compared to LAGB and SG.12-16 Patients post-RYGB can present with complications in the early (<30 days) or late post-operative period (>30 days). Complications which can be evaluated at fluoroscopic upper GI examination (UGI) and computed tomography (CT) are summarized in Table 1.
Complications of Roux-en-Y Gastric Bypass Surgery Detected at Fluoroscopy and CT.

In the immediate post-operative period, complications may include infection, hemorrhage, or leak due to anastomotic/staple line disruption (Figure 3A). Early acute ischemia of the Roux limb can occur and may be self-limited or lead to bowel infarction. Bowel obstruction can develop anytime post-RYGB with an overall incidence of 4.4%.11,17 Causes of small bowel obstruction post-RYGB include adhesions and abdominal wall hernias, which are more common following open RYGB surgery. Other causes of obstruction include internal hernia, intussusception, and stricture at the gastro-jejunal (G-J) or J-J anastomosis. Patients with G-J anastomotic stenosis can present with progressive dysphagia, nausea/vomiting and malnutrition weeks to years post-RYGB. 11 Three patterns of small bowel obstruction have been described18,19: (A) obstruction of the Roux limb with decompressed biliopancreatic limb; (B) obstruction of the biliopancreatic limb with distension of the excluded stomach, presenting a high risk of closed-loop obstruction and perforation; and (C) obstruction of both the Roux and biliopancreatic limbs distal to J-J anastomosis. Intussusception most commonly occurs at the J-J anastomosis due to altered small bowel motility and function of the staple line acting as a lead point and is best detected at CT (Figure 3B). It is often transient, and clinical presentation can be non-specific. However, persistent long segment, retrograde, and symptomatic intussusception is worrisome, and can lead to small bowel obstruction or ischemia/necrosis, requiring prompt surgical consultation.
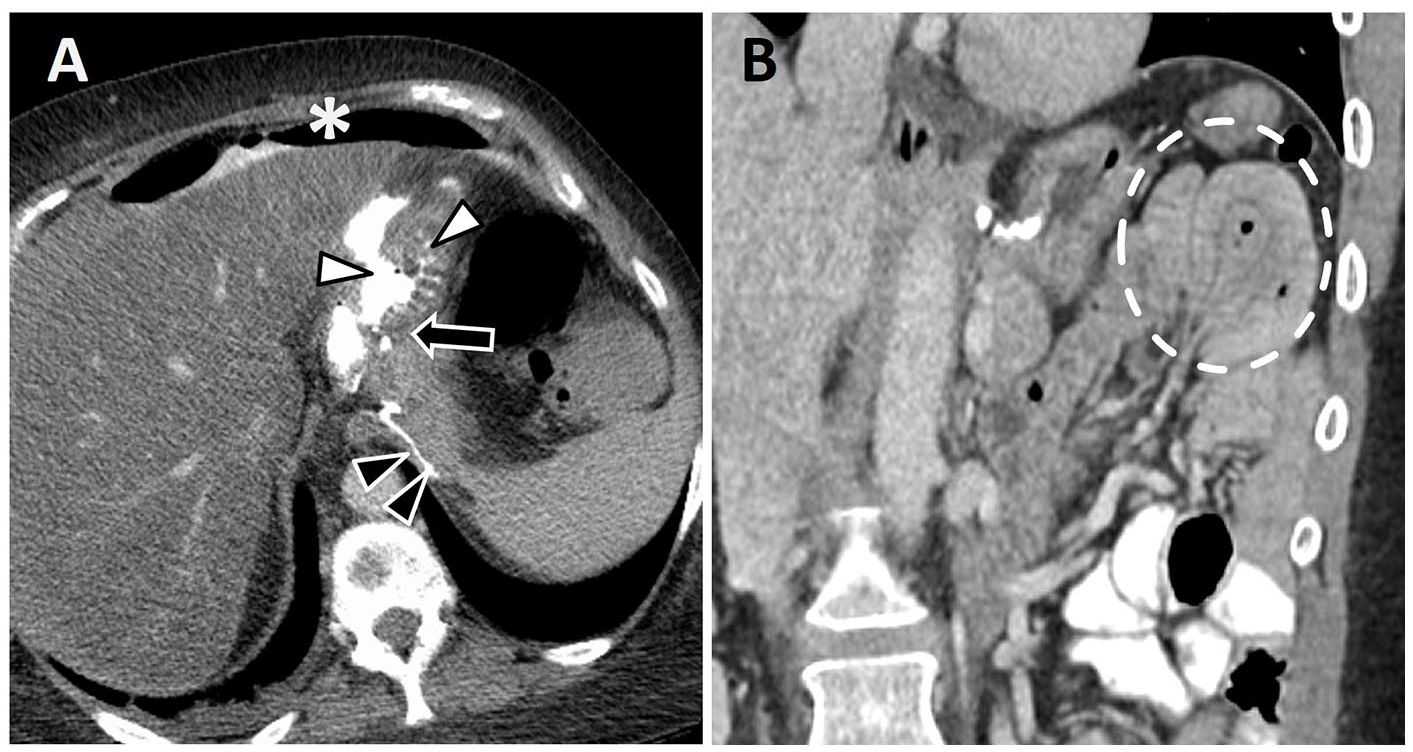
Complications following Roux-en-Y gastric bypass. (A) Anastomotic leak. Axial CECT image with oral contrast shows a defect (white arrowheads) at the gastrojejunal (GJ) anastomosis (black arrow). There is associated pneumoperitoneum and extravasated high density contrast (asterisk). Excluded stomach staple line (black arrowheads). (B) Intussusception in a patient with 2 days of worsening abdominal pain. Coronal CECT image shows a left upper quadrant enteroenteric intussusception (dotted line) associated with the jejunojejunal anastomosis. This was depicted at multiple imaging studies with recurrent intermittent abdominal pain post Roux-en-Y, and the patient was eventually taken back to surgery for anastomotic revision.
Creation of a mesenteric potential space/defect predisposes to development of an internal hernia which can occur as a late complication, with an incidence as high as 16%.20,21 CT is the modality of choice for detection of an internal hernia, which should ideally be performed while the patient is symptomatic as they can be intermittent. CT imaging features of internal hernia are further discussed below. If left untreated, an internal hernia can lead to bowel obstruction and necrosis requiring resection. 11 Three potential mesenteric defects are made during laparoscopic RYGB which can predispose to post-operative internal hernia: at the jejuno-jejunostomy (most common), between the transverse mesocolon and Roux-limb mesentery (Peterson’s defect), and at the transverse mesocolon in cases of retrocolic Roux limb.11,22 The retrocolic retrogastric approach is associated with higher rates of internal hernia and bowel obstruction compared with the antecolic antegastric approach.23-25 Factors which can contribute to development of an internal hernia include incomplete closure of defects, use of absorbable rather than non-absorbable suture material for closure, and weight loss leading to enlargement of the defects. 22
Another late complication after RYGB surgery is marginal ulceration, typically on the jejunal aspect of the G-J anastomosis, occurring in up to 16% of patients. 11 Exposure of jejunal mucosa to the acidic gastric secretions, poor local vascular perfusion and ischemia, and use of certain medications such as non-steroidal anti-inflammatory drugs (NSAIDs) are believed to contribute to ulcer development. 11 Marginal ulceration can lead to stenosis or perforation and is typically diagnosed with upper endoscopy. 11 Jejunal ischemia may occur which can cause giant ulcers in the Roux limb, a rare complication. Finally, recurrent weight gain months or years following surgery may occur because of dilation of the G-J anastomosis, gastrogastric fistula, or staple line dehiscence (in cases where the excluded stomach was not completely transected from the pouch).
Fluoroscopic Upper GI Examination
UGI can be helpful in detecting strictures as well as suture line breakdown. Identification of the location of a leak is important for guiding management, including endoscopic stent or clip placement. 11 The patient drinks either water-soluble iodinated contrast or barium. Although higher density barium improves visualization and may increase sensitivity for detection of leaks, 26 it should be avoided as the initial oral contrast in the early post-operative phase when the clinical suspicion of leak is high. Spillage of barium into the peritoneum can lead to peritonitis or abscess formation and can produce artifact on subsequent CT scans. 27
In the early post-operative phase, contrast should reach the J-J anastomosis, while in the late post-operative phase contrast should ideally reach the terminal ileum to evaluate for small bowel obstruction or internal hernia. 28 The contrast column is observed as it traverses the esophagus and gastric pouch, passes through the G-J anastomosis and Roux limb into the jejunum. Attention should be paid to any contrast passage into the excluded stomach in the early phase of the fluoroscopic evaluation as later reflux of contrast into the biliopancreatic limb may opacify the excluded stomach, making this diagnosis challenging. 28 Reflux of contrast into the excluded stomach can simulate a gastrogastric fistula and can lead to a false positive result on UGI and CT studies but can be more reliably diagnosed on UGI with careful real time observation. 11
Extraluminal contrast extravasation may be seen secondary to suture line breakdown and leaks, while absence or poor opacification through the G-J or J-J anastomoses may suggest proximal stricture. Marginal ulcers, a late complication, can lead to G-J anastomotic strictures. These ulcers can appear as small focal outpouchings containing oral contrast along the G-J anastomosis, which can be difficult to detect on UGI studies when small, requiring endoscopy for definitive diagnosis. 28 Contrast opacification of the blind-ending outpouching of the efferent limb due to side-to-side G-J anastomosis should not be mistaken for a leak (Figure 2A).29,30 On UGI, this small pouch will opacify with contrast and subsequently empty. In contrast, a true leak will demonstrate persistent pooling of extraluminal contrast as the patient continues to drink. 30
Computerized Tomography With Oral and IV Contrast
In the peri-operative setting, CT can also be used for evaluation of intra-abdominal abscess formation, hemorrhage, or anastomotic leaks (Figure 3A). In the late post-operative setting, CT is commonly utilized to assess for internal hernia, small bowel obstruction, as well as marginal ulceration at the gastroenteric anastomosis. Positive oral contrast is administered 30 to 60 minutes prior to scanning and then again immediately prior to scanning to ensure opacification of the G-J junction and distally beyond the J-J anastomosis. 28 Marginal ulceration may present with subtle findings including perienteric fat stranding, wall thickening, and contour abnormalities. 31 Though less common, marginal ulcers can lead to perforation with imaging findings of extraluminal air, fluid, or oral contrast.
Presence of oral contrast is essential for evaluation of a gastrogastric or gastroenteric fistula and helps distinguish the gastric pouch/Roux limb (opacified), from the excluded stomach/biliopancreatic limb. Presence of oral contrast within the excluded stomach may be due to reflux via the biliopancreatic limb. However, the presence of oral contrast within the excluded stomach but not within the more distal portions of the biliopancreatic limb may increase the degree of suspicion for fistula. Additional complications following RYGB such as intussusception may be detected, which usually occurs retrograde at the jejunojejunal anastomosis, appearing as a classic target sign with invagination of a small bowel segment and its mesentery into the lumen of another bowel segment (Figure 3B). Intussusceptions can be transient in nature as discussed above, however if fixed can lead to bowel obstruction and/or necrosis. 32
CT is the preferred imaging modality for diagnosis of an internal hernia associated with RYGB (Figure 4A and B). It is important to maintain a high index of suspicion for presence of an internal hernia as the clinical presentation can be non-specific and the patient may present with intermittent symptoms. Delayed diagnosis can have serious consequences due to the risk of a small bowel obstruction, infarction, and perforation. 19 Internal hernias may present in a variety of ways at CT including a cluster of bowel loops in an atypical location, swirling of mesenteric vessels, SMV “beaking” (Figure 4A), “criss-cross” appearance, reversal of SMV and SMA anatomic relationship, bowel surrounding tubular distal mesenteric fat (“hurricane eye” sign), small bowel obstruction (Figure 4B), mushroom-shaped herniated mesenteric root, bowel loops other than duodenum posterior to the SMA, and displacement of the J-J anastomosis.19,23,33 Of these CT features, presence of a small bowel obstruction with SMV “beaking” or swirling of mesenteric vessels show highest sensitivity and specificity for diagnosis of internal hernia.23,33 Internal hernias may present without an associated small bowel obstruction which can make this diagnosis more challenging.
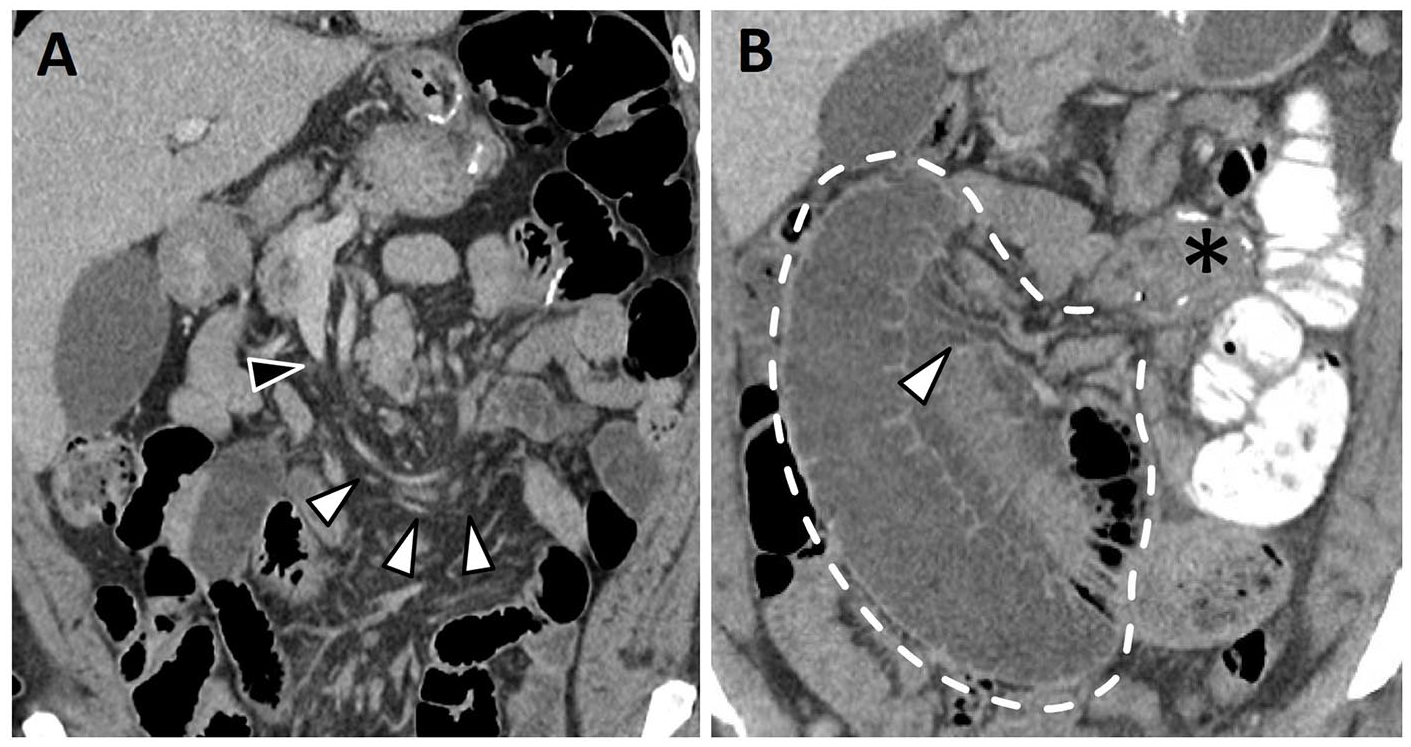
Internal hernia post Roux-en-Y gastric bypass. (A) Coronal CECT image from a patient with worsening abdominal pain shows swirling of the mesenteric vessels (white arrowheads) and “SMV beaking” (black arrowhead). (B) Coronal CECT image from a different patient presenting with abdominal pain shows an isolated dilated, fluid-filled small bowel loop (dotted line) consistent with closed-loop mechanical small bowel obstruction. There is associated mesenteric edema (arrowhead). Small bowel obstruction secondary to internal hernia was confirmed intraoperatively. Jejuno-jejunal anastomosis (asterisk).
Vertical Sleeve Gastrectomy (SG)
SG involves a partial gastrectomy, with removal of approximately 75% of the greater curvature, creating an elongated banana-shaped gastric “sleeve.” 19 This results in increased gastric emptying and metabolic changes from removing the fundal ghrelin-producing cells which are involved in appetite regulation. 34 It is an irreversible and purely restrictive procedure. Unlike RYGB, the stomach and small bowel connection is maintained. In morbidly obese patients, staged laparoscopic sleeve gastrectomy followed by RYGB may be a performed as a risk reduction strategy. 35 Imaging of early post-procedural complications may include assessing for intra-abdominal bleeding, staple line leak and abscess formation, while sleeve stricturing may be a late complication detected at imaging. 8
Fluoroscopic Upper GI Examination
Fluoroscopic imaging following oral administration of water-soluble iodinated contrast or barium, typically reveals a long tubular residual stomach. Loss of the narrow tubular-shaped gastric pouch in a patient with weight regain or insufficient weight loss suggests gastric dilation and may require surgical revision. 36 Staple line leak is the most common complication occurring in up to 2.9% of patients,34,37 typically along the superior aspect of the staple line, below the gastroesophageal junction. 8 Extravasation of water-soluble contrast and pooling of contrast adjacent to the greater curvature of the residual stomach may be seen on UGI (Figure 5). If a leak is suspected but not identified with water soluble contrast, barium may be administered to exclude a subtle leak. 19
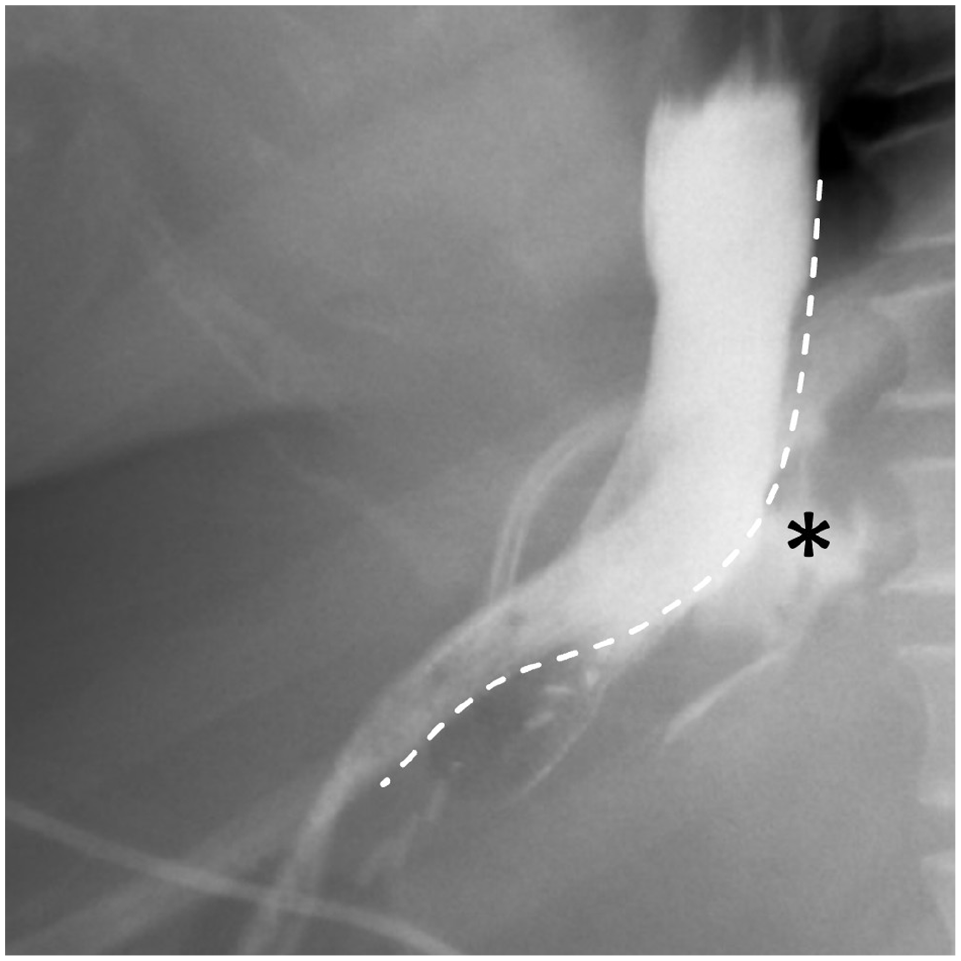
A patient on postoperative day 11 from vertical sleeve gastrectomy with an acute abdomen. UGI spot radiographic image demonstrates oral contrast extravasation (asterisk) along the staple line/greater curvature (dotted line) in keeping with an anastomotic leak.
Stenosis of the mid-portion of the sleeve may occur due to true mucosal or luminal stricturing or from twisting of the stomach at the incisura. 34 Endoscopic dilatation of focal strictures may be effective for treatment of the obstruction, however occasionally surgical revision is required.19,34 SG also increases the risk of developing GERD symptoms, erosive esophagitis, and Barrett’s esophagus, 11 which may prompt UGI evaluation.
Computerized Tomography With Oral and IV Contrast
CT following SG will reveal a staple line along the greater curvature of the stomach with reduced luminal diameter of the residual tubular-shaped stomach. CT can be helpful for identification of leaks, extraluminal collections, abscesses, and other complications following SG. 34 Postoperative bleeding, which occur in <2% of cases after laparoscopic SG can also be evaluated at CT. 34
Laparoscopic Adjustable Gastric Banding (LAGB)
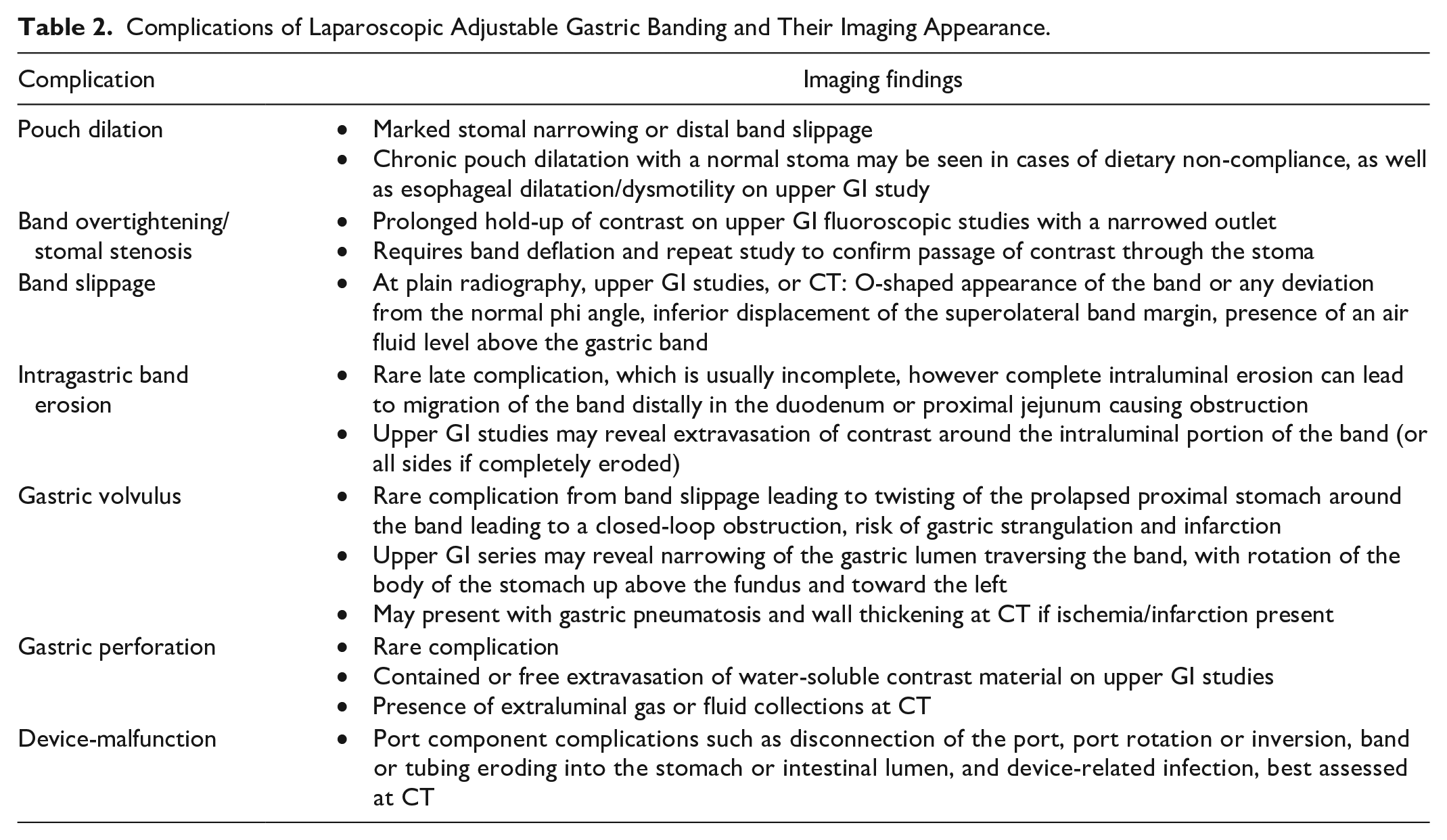
LAGB is one of the least invasive bariatric procedures and is reversible. While LAGB is an effective procedure for weight loss, studies suggest that RYGB and LSG are superior in terms of long-term weight loss.14,15,38,39 An adjustable silicone band is placed around the cardia of the stomach, which can be inflated or deflated by injecting/aspirating saline from a subcutaneous port attached via a connecting tube. This creates a gastric pouch with an approximate volume of 15 mL, leading to weight loss through reduced capacity for food intake. 11 Appropriate position of the band is confirmed by assessing the “phi angle”—the angle between the spinal column/vertical line, and the long axis of the gastric band, with a normal phi angle ranging from 4° to 58° (Figure 6). On abdominal radiograph, the band should be positioned approximately 5 cm below the left hemidiaphragm and appear rectangular on AP projection. LAGB has a low peri-operative complication rate, with band slippage (5%-10%) and erosion/intragastric migration (3%-7.5%) being the 2 most common late complications seen at imaging.22,40,41 The patient may present with reflux, epigastric discomfort, and poor oral intake, which could suggest an achalasia-like syndrome with esophageal dilation secondary to overinflation of the band. 11 Complications associated with LAGB are summarized in Table 2.
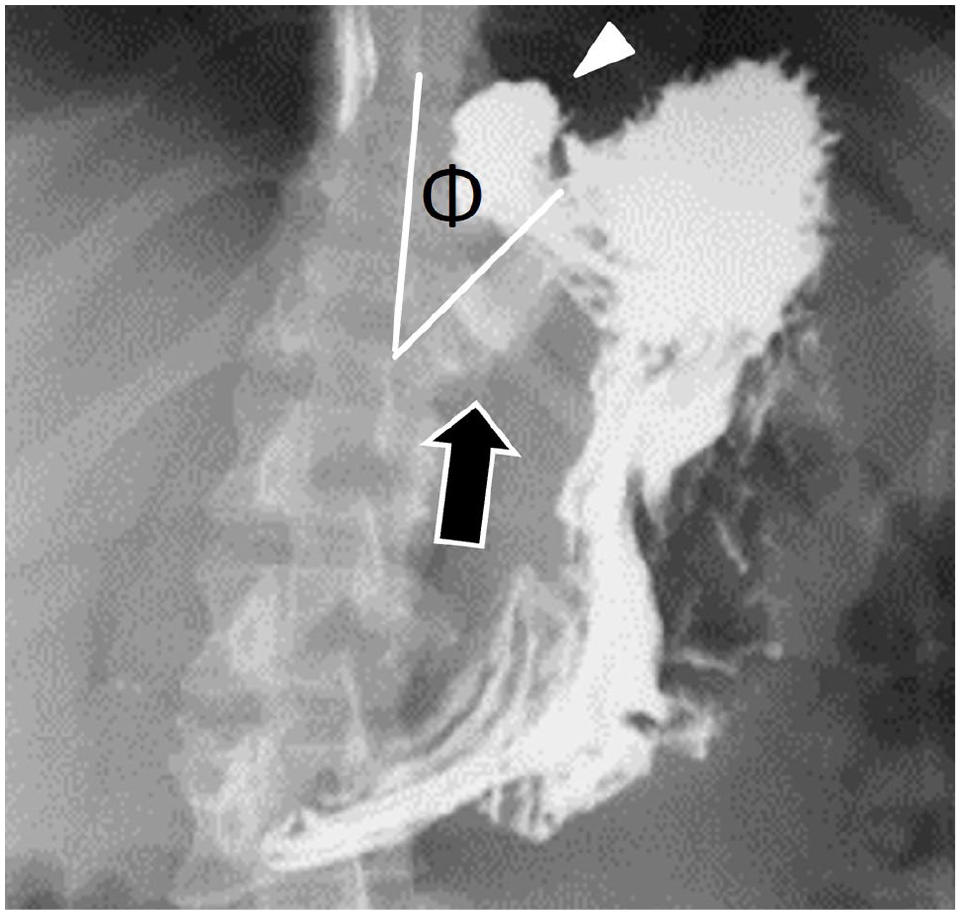
LAGB normal appearance at UGI series. Spot radiographic image demonstrates barium outlining a small gastric pouch (arrowhead) above the radiopaque gastric band (arrow). There is a normal ϕ angle (normal = 4°-58°), which is measured between the long axis of the vertebral column and gastric band (shown by the white lines).
Complications of Laparoscopic Adjustable Gastric Banding and Their Imaging Appearance.
Fluoroscopic Upper GI Examination
A scout image should be obtained to visualize the location of the band and confirm a normal phi angle (4°-58°). Orally administered contrast will normally reveal a small gastric pouch (normal diameter = 4 cm, ~15-20 mL), with luminal narrowing as it passes the band stoma. 19 Contrast should then pass into the larger stomach compartment below the band.
Stomal stenosis and band slippage are common complications of LAGB (Figure 7). 29 Stomal stenosis in the early post-operative period can be due to gastric edema, which improves spontaneously. Iatrogenic band overinflation, as well as gastric inflammation and thickening can cause stenosis in the later post-operative stage. 29 Band slippage more commonly occurs distally with posterior herniation of the distal stomach superiorly. This can be detected on plain abdominal radiograph or scout image by assessing an abnormal phi angle >58° (Figure 7A), a very sensitive (91%-95%) radiographic sign for band slippage. 42 Herniation of the stomach superiorly through a slipped gastric band can result in tilting of the band resulting in an o-shaped or “en face” configuration known as the “O sign” (Figure 7B) which has a specificity of 97% for distal gastric band slippage. 42 Anterior herniation of the distal stomach superiorly through the band results in a phi angle <4°. 29 UGI may reveal band malposition along with proximal pouch dilatation and comparison with prior post-operative studies is useful to assess for interval pouch dilatation. Proximal pouch dilatation alone in the absence of stomal stenosis may be seen in patients who overeat. The presence of an air-fluid level above the gastric band is highly sensitive (95%) and specific (100%) for band slippage with obstruction. 42 If band slippage is detected, immediate deflation is required under fluoroscopic or ultrasound guidance by withdrawing fluid through the injection port, and a follow-up fluoroscopic study to document patency of the stoma.19,22 If band deflation does not provide symptomatic relief, gastric necrosis and erosion should be considered, which can be sequelae of ischemia caused by pressure on the gastric wall. Band erosion is often diagnosed with endoscopy; however partial or full thickness erosion may manifest as flow of barium/contrast material around the eroded component of the band, which is pathognomonic on UGI. 40 A rare but known complication of LAGB that can be diagnosed on UGI is megaesophagus which occurs when there is proximal migration of the band mimicking achalasia and usually requires band removal. 22
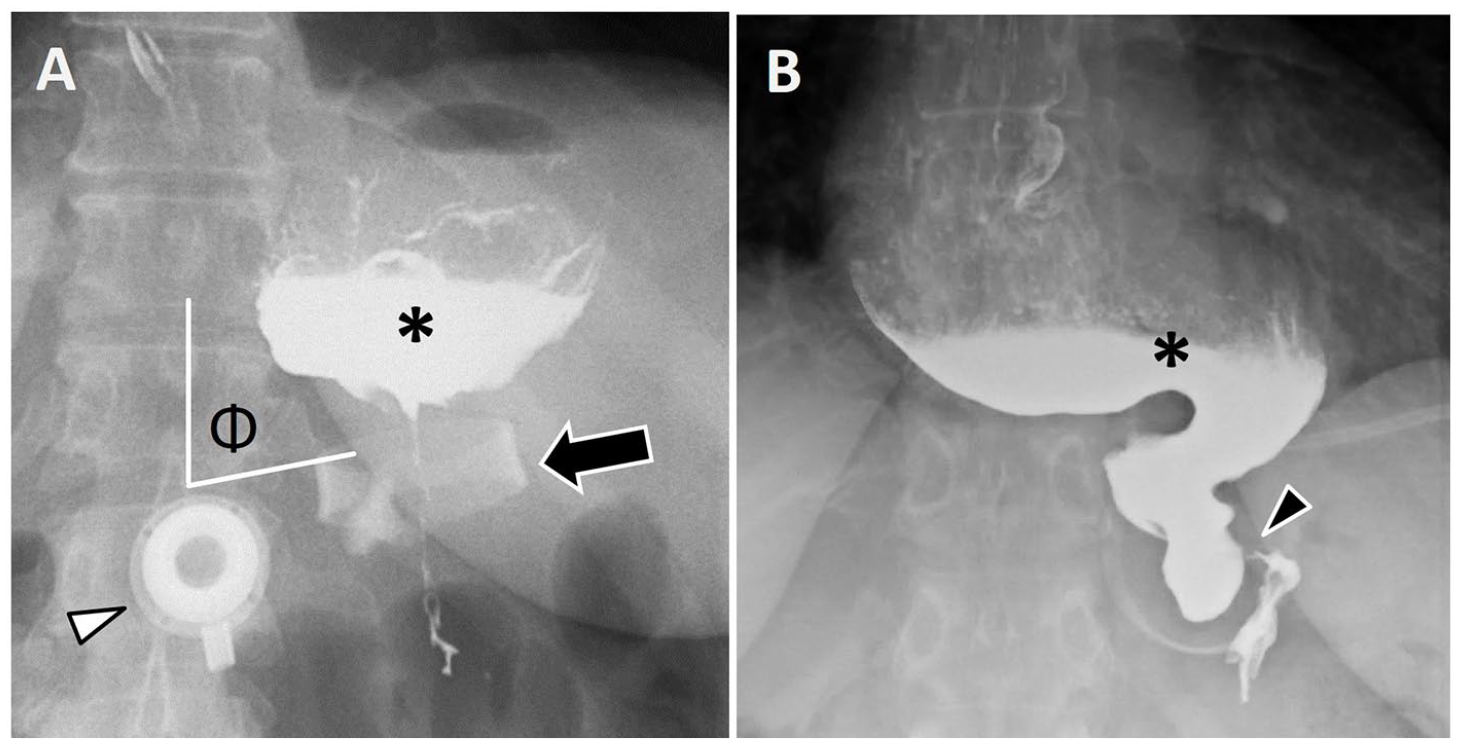
LAGB complication: distal band slippage. (A) A patient presenting 10 years post-gastric banding with progressive difficulty tolerating oral intake. Spot radiographic image from UGI series shows near horizontal position of the gastric band (arrow), resulting in increased ϕ angle measuring nearly 90° (normal 4°-58°) with proximal gastric pouch obstruction (asterisk). Access port (arrowhead). (B) Another patient 8 years post gastric band procedure with clinical concern for pouch narrowing. UGI series spot radiograph taken prior to administering oral contrast (not shown) demonstrated en face appearance of the laparoscopic band on the frontal projection (“O sign”), suggestive of band slippage. Following oral contrast administration, the stomach appears markedly narrowed as it traverses the laparoscopic band (arrowhead), with en face appearance of the band seen here. There is resultant marked distention of the esophagus (asterisk), with tapering toward the level of the laparoscopic band.
Computerized Tomography With Oral and IV Contrast
Normal appearance of an adjustable gastric band can be identified at CT showing the adjustable silicone device appearing as a radiopaque band positioned around the proximal stomach near the gastroesophageal junction, resulting in a small gastric pouch above the band. 19 The band is connected via tubing to an injection port in the anterior subcutaneous tissues. 19 The entire course of the banding device should be evaluated including the connecting tube and the subcutaneous port.
CT findings of extraluminal air or inflammation around the band itself should raise concern for intragastric band erosion. 40 Erosion of the band into the gastric fundal lumen may be visualized at CT. Device-related infection can occur as an early or late complication of LAGB. This may be secondary to manipulation of the access port and can present with gas/inflammatory changes tracking along the connector tubing toward the band, and abscess formation. 43 Hence, the distribution of inflammatory changes may be helpful in diagnosis, with inflammation centred at the band suspicious for band erosion and inflammation centred at the port suspicious for infection. Port malfunction due to disconnection of port components can also be detected at CT.
Band slippage can be identified at CT (Figure 8), with distal slippage characterized by more horizontal orientation of the band (phi angle >58°) or marked stomal narrowing leading to acute pouch dilation and proximal migration potentially resulting in esophageal obstruction. Acute pouch dilatation due to band slippage may require surgical correction to avoid adverse outcomes such as gastric volvulus, infarction, or perforation. 29 In contrast, chronic pouch dilatation typically demonstrates a normal stomal diameter on UGI with a normal phi angle and may occur in patients who cannot comply with the necessary dietary modifications. 19
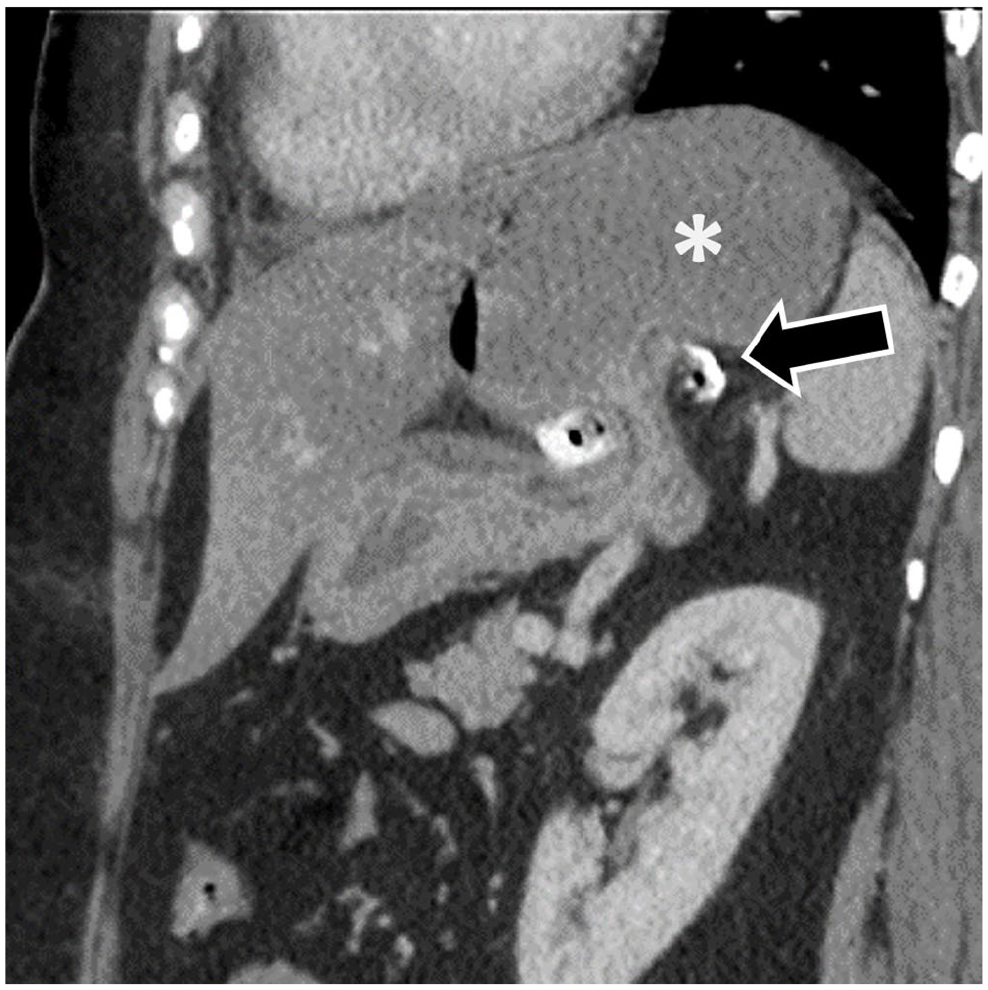
Distal slippage of LAGB at CT. A patient 4 days post-LAGB, presenting with vomiting and upper abdominal pain despite band decompression. Sagittal CECT image shows abnormal distal migration of the gastric band (black arrow), now around the mid gastric body, resulting in obstruction of the proximal stomach (asterisk).
Uncommon Bariatric Procedures
Biliopancreatic Diversion With Duodenal Switch (BPD-DS)
BPD-DS accounts for <1% of bariatric procedures. 44 The duodenum is divided above the level of the ampulla, separating biliopancreatic contents (biliopancreatic loop) from food (alimentary/digestive loop) while sparing the pylorus. The digestive loop bypasses approximately two thirds of the small bowel length (normal small bowel length ranging from 630 to 1510 cm45), before rejoining the biliary limb at a distal Y anastomosis to create a common channel (Figure 9A). The digestive loop (250 cm) as well as the common channel (50-125 cm), where digestion and absorption occur, reflect a marked reduction in surface area over which nutrient absorption may occur. 46 The procedure also typically includes a SG, or BPD-DS may be performed as a revisional procedure following SG. 44 Studies indicate that in comparison to RYGB, BPD-DS is more effective for sustained weight loss in patients with very high BMI, 47 however BPD-DS is associated with higher rates of re-operation. 38 BPD-DS is less frequently performed due to its surgical complexity and overall higher incidence of complications, including nutritional deficiencies.46,48 Appropriate selection of patients who can adhere to compliance with follow-up and lifelong vitamin supplementation are critical for improved long-term clinical outcomes.
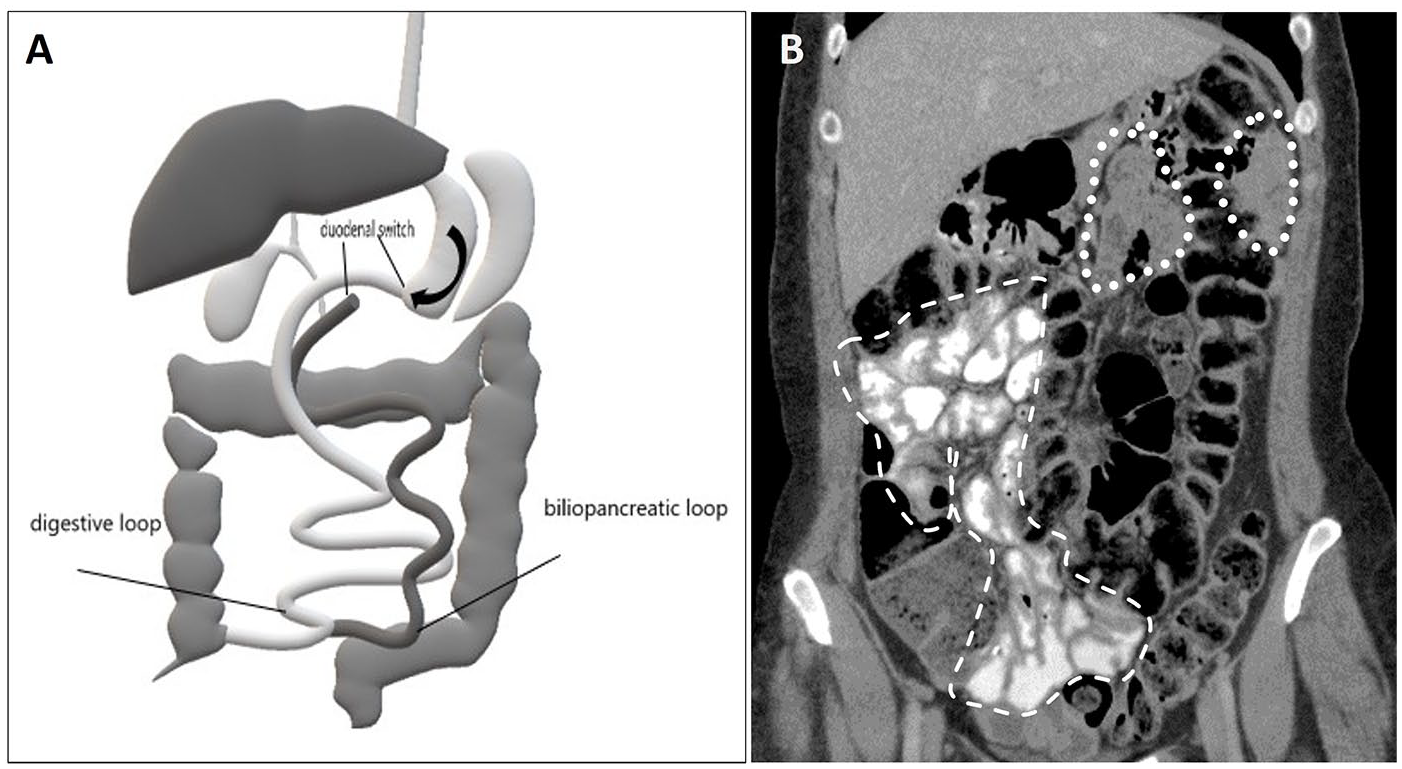
(A) Schematic illustration of a biliopancreatic diversion with duodenal switch (BPD-DS). A partial (sleeve) gastrectomy is performed. The residual gastric lumen is disconnected from the duodenum (depicted in dark grey) and reconnected to the distal small bowel (shown in light grey), bypassing a long segment of small bowel. The duodenum and proximal small bowel (biliopancreatic loop) rejoin the digestive (alimentary) loop at a very distal Y-anastomosis. (B) BPD-DS normal appearance at CT. Coronal CECT image demonstrate oral contrast opacifying the distal small bowel in the right hemiabdomen (dashed line) constituting the digestive loop. No oral contrast is noted within the bypassed small bowel (biliopancreatic loop) in the left hemi-abdomen (dotted line).
Peri-operative morbidity may include leaks at the gastrectomy staple line or at the duodenal or ileal anastomoses, bleeding, and abscess formation. Other potential postoperative complications include mechanical bowel obstruction and internal hernia.
Both contrast-enhanced CT with oral contrast agent and fluoroscopic small bowel follow-through studies can identify the presence of a BPD-DS. Fluoroscopic images will demonstrate opacification of the digestive loop and the common channel, with exclusion of the majority of the small bowel. 30 Key features on CT include presence of a sleeve, opacification of the digestive loop with oral contrast in the right hemiabdomen, and absence of oral contrast in the biliopancreatic loop in the left hemi-abdomen (Figure 9B).
Vertical-Banded Gastroplasty (VBG)
VBG involves creation of a small proximal gastric pouch along the lesser curvature, which is separated from the gastric fundus with a vertical staple line as illustrated in Figure 10A. Placement of a band/tight prosthetic mesh around the exit window from the pouch results in a narrow outlet (gastric stoma) that remains constant in diameter. 11 Figure 10B demonstrates the expected appearance following vertical-banded gastroplasty at CT. VBG is a purely restrictive procedure that is less commonly performed today, with nearly half of patients requiring revisional bariatric surgery such as conversion to a RYGB due to long-term complications or weight re-gain.49,50 Early post-operative complications include leak, staple line bleeding, and infection. Fistula formation between the gastric pouch and fundus due to staple line dehiscence may be seen in nearly half of patients, resulting in weight regain. 11 Band erosion, stomal stenosis, and pouch enlargement can occur which may warrant surgical revision. Stomal stenosis is an important long-term complication typically presenting as narrowing of the gastric stoma with delay of contrast outflow from the gastric pouch on UGI. UGI may reveal evidence of reflux, or less commonly, formation of gastric pouch diverticula. 29 In patients who cannot comply with strict dietary modifications, food may become impacted at the stoma, which is exacerbated by stomal stenosis and can lead to partial or complete obstruction. 29
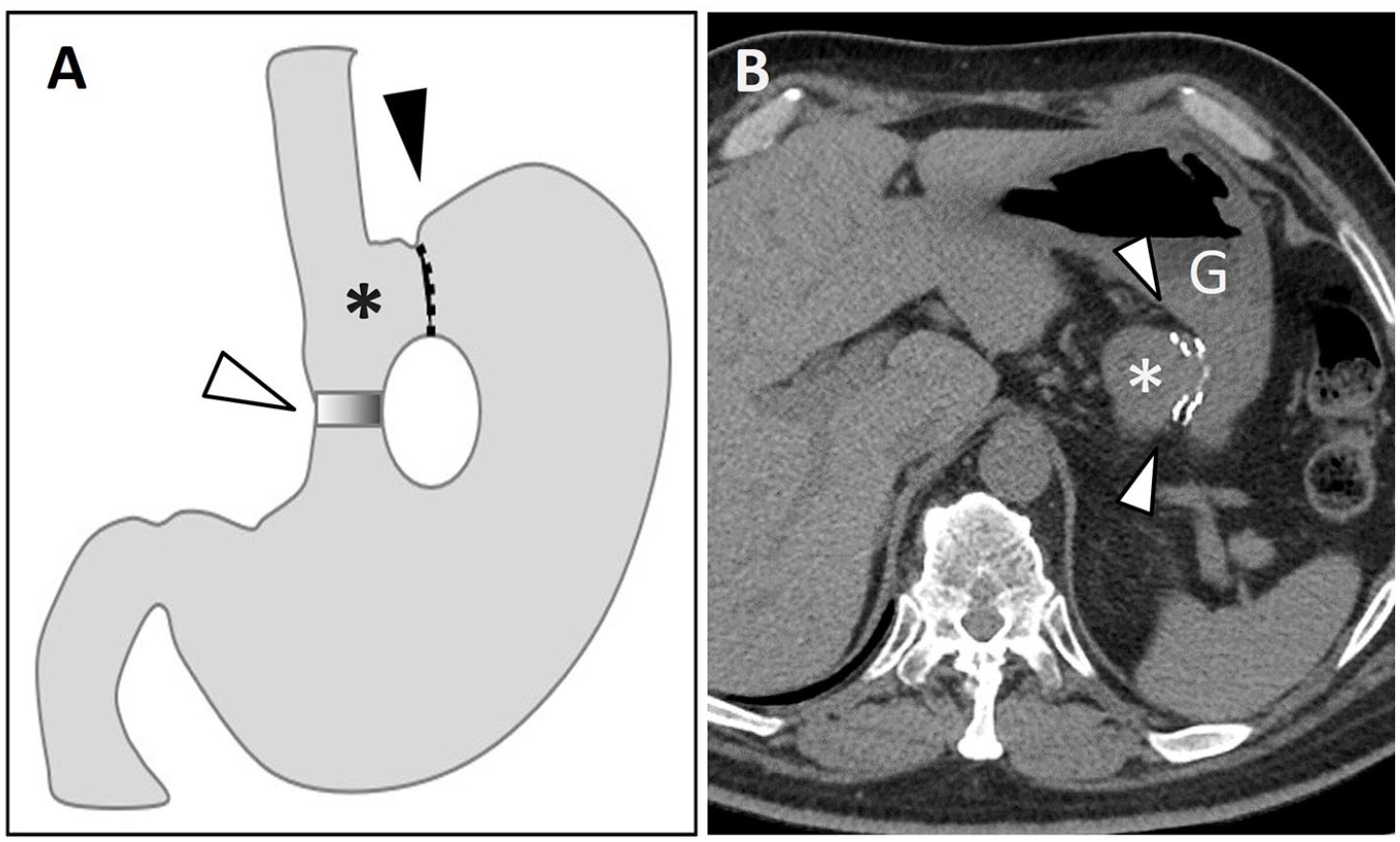
(A) Schematic illustration of vertical-banded gastroplasty. The stomach is divided, with the fundus separated from the gastric pouch (asterisk) by a vertical staple line (black arrowhead). A polypropylene mesh band (white arrowhead) creates a narrow opening from the gastric pouch into the remainder of the stomach. (B) Axial non-enhanced CT (NECT) image from another patient depicts the vertical staple line (arrowheads) separating the gastric pouch (asterisk) from the gastric fundus (denoted as G).
Intra-Gastric Balloon (IGB)
IGB is a space-occupying air or fluid-filled balloon device which is usually placed endoscopically (Figure 11), and may be used as a bridge between medical management and bariatric surgery. 51 IGB placement promotes weight loss by reducing intragastric volume, promoting satiety, modulating gastrointestinal hormones, and increasing gastric emptying time.51-53 IGB is a minimally invasive, non-permanent approach to weight loss and may be considered in non-surgical candidates, however there is limited data on long term efficacy. 51 Complications include balloon deflation, migration of the balloon causing intestinal obstruction, 54 gastric perforation, and pancreatitis due to compression of the pancreatic body.55,56 New developments include an ingestible gastric balloon designed to remain in the stomach for 4 months, self-deflate, and pass through the gastrointestinal tract, which alleviates the need for endoscopic placement and has shown some promising short term weight loss results in obese patients.57,58 IGB can be readily identified on CT as a large fluid or air attenuating balloon with a port, occupying the majority of the stomach lumen (Figure 11).
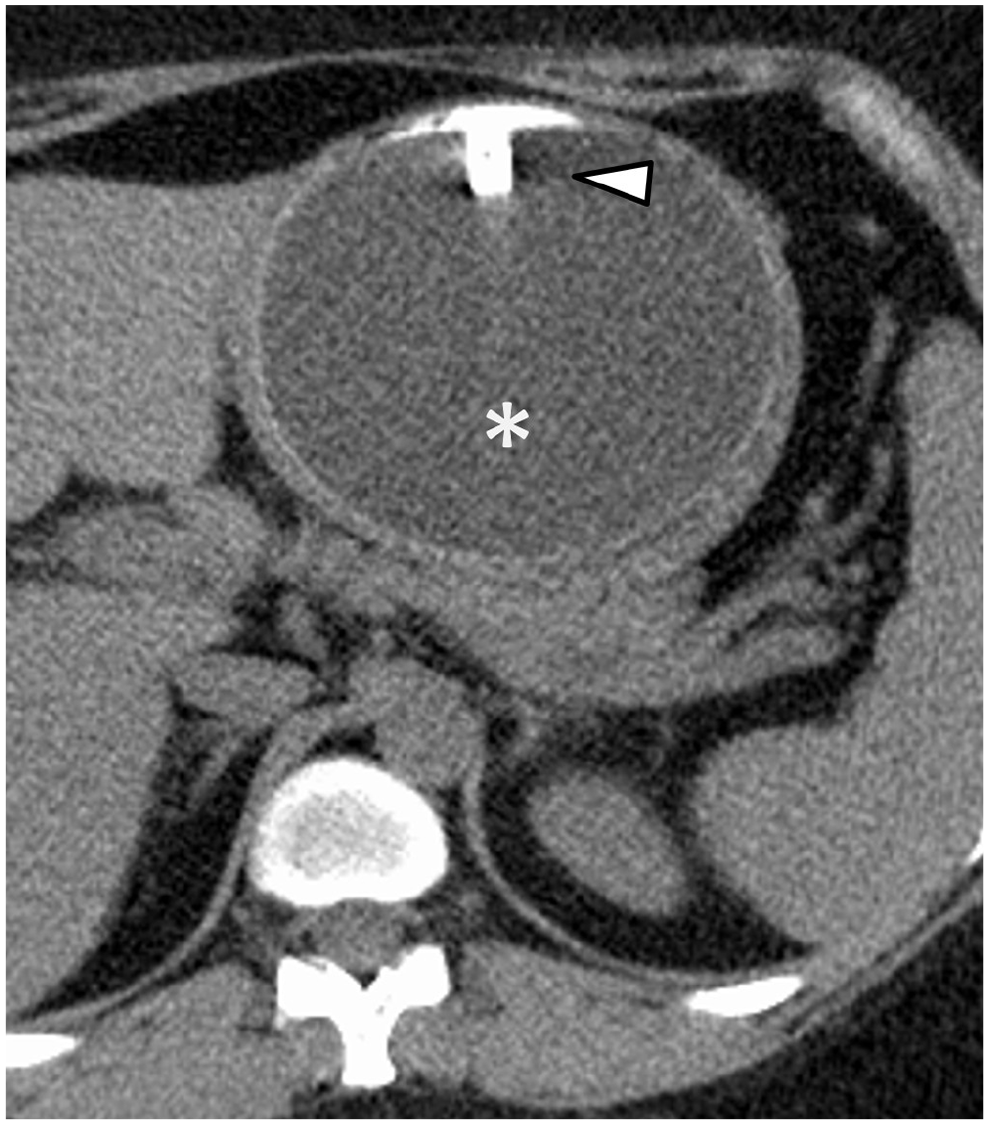
Axial NECT image depicting a large fluid attenuation balloon occupying the majority of the gastric lumen (asterisk) with port noted anteriorly (arrowhead).
Non-Adjustable Gastric Banding (NAGB)
NAGB procedure was pioneered in the late 1970s. 59 A prosthetic material is placed around the upper stomach creating gastric segmentation without transecting the stomach (Figure 12A). 60 NAGB is a purely restrictive procedure as delayed emptying through the narrowing created by the band results in restriction of food intake. Despite short-term weight loss, weight regain and persistent vomiting resulted in high rates of re-operation which eventually led to abandonment of this procedure.59-61 However, nonadjustable gastric bands may still be found incidentally on imaging studies (Figure 12B and C) or patients may present with complications decades after the procedure, often related to dysphagia and proximal gastric outlet obstruction.60,62 UGI can help with evaluation of the gastric anatomy and evaluate for reflux into the esophagus or gastric distention due to excessive narrowing of the stomach by the gastric band. Band erosion with extravasation of contrast around the site of erosion may be visible on UGI, prompting surgical removal.
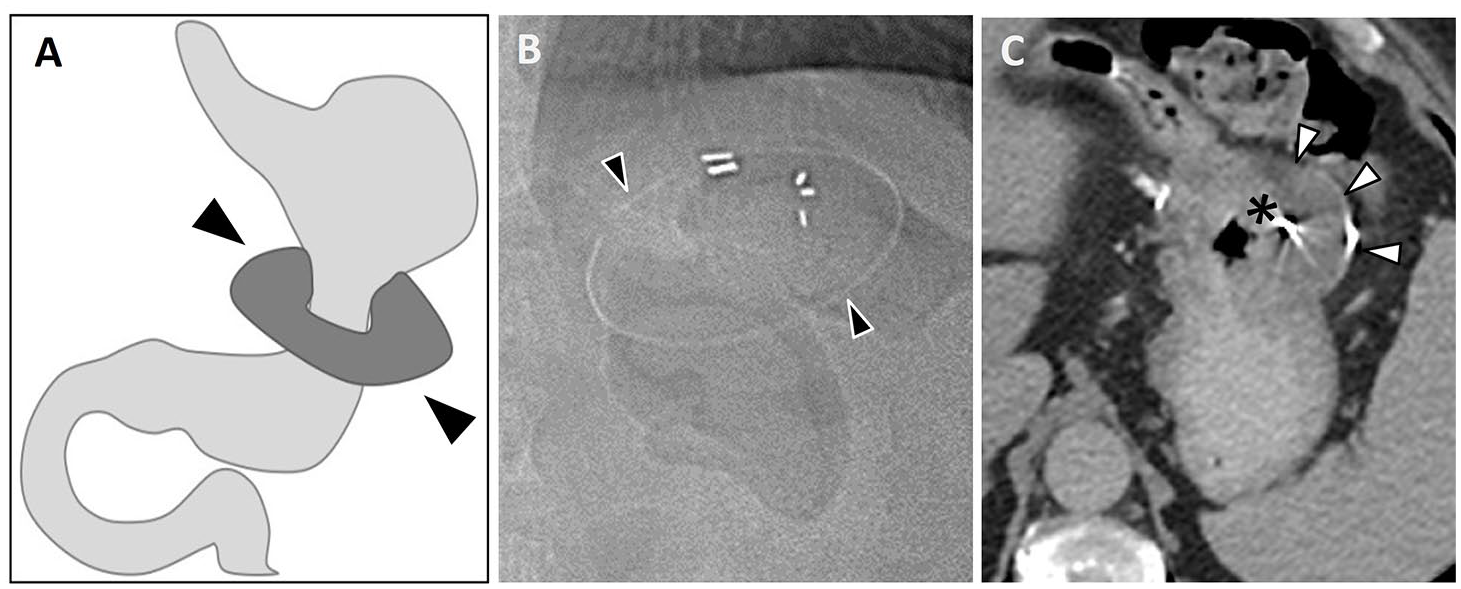
(A) Schematic illustration of a non-adjustable gastric band. A constrictive ring (arrowheads) is circumferentially placed around the proximal stomach/gastric body, restricting the lumen. It lacks the access port of more contemporary devices for adjustment of band tightness. (B) Off-label use of silicone Angelchik anti-reflux prosthesis as an early form of non-adjustable gastric band circa early 1980s. Coronal CT scout image demonstrates a radiopaque marker demarcating location of the gastric band in the left upper quadrant (arrowheads). Axial CECT image (C) demonstrates a hypodense silicone prosthesis with peripheral radiopaque marker (arrowheads) surrounding and restricting the gastric lumen (asterisk).
Conclusion
Bariatric surgery is an effective and increasingly utilized treatment approach for weight loss and management of obesity-related comorbidities in morbidly obese patients. Imaging interpretation in patients following bariatric procedures can pose a challenge, requiring familiarity of the anatomic changes and expected post-operative appearances. Fluoroscopic UGI and CT are invaluable for detection of early and late post-operative complications of bariatric procedures including identification of band slippage, internal hernia, anastomotic leak, bowel obstruction, and anastomotic strictures. While sleeve gastrectomy and Roux-en-Y gastric bypass are most frequently performed, familiarity with less common bariatric procedures which may on occasion be encountered allows the radiologist to confidently identify potential complications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
