Abstract
Ultrasound (US) is an invaluable tool for evaluating the neonatal spine, offering a non-invasive, cost-effective, and radiation-free imaging alternative. This article reviews the indications, techniques, and sonographic features of congenital and acquired spinal pathologies in neonates. Common indications include the evaluation of atypical sacral dimples, spinal anomalies such as tethered cord and syringomyelia, congenital tumours like sacrococcygeal teratomas, and post-traumatic conditions such as spinal hematomas. Detailed sonographic spine anatomy, variants, and ultrasound guidance for interventions like lumbar puncture are also discussed. By enabling early diagnosis, US is crucial in guiding clinical management, especially in pediatric populations with spinal disorders.
Introduction
Advances in ultrasound (US) technology and imaging protocols have made it a preferred modality for evaluating the spine in neonates and young infants.1 -3 The unossified, cartilaginous posterior vertebral elements provide an excellent acoustic window for ultrasound imaging, typically up to 3-4 months of age.1,4,5 This non-invasive technique offers a rapid and cost-effective way to assess the pediatric spine without sedation or radiation exposure.1,2,6 Additionally, when performed by a skilled sonographer or radiologist, ultrasound is less prone to motion and technical artifacts making it a reliable imaging modality for neonatal spine evaluations.1,3,4 Ultrasound is comparable to magnetic resonance imaging (MRI) for detecting both congenital and acquired spinal abnormalities in very young children, allowing clinicians to determine the need for further imaging, such as MRI, and guide management decisions.1,4,7 An effective ultrasound examination requires a thorough understanding and familiarity with spinal anatomy, common variants, and primary pathologies.7 -9 This review aims to provide an overview of ultrasound techniques, key clinical indications, sonographic anatomy and variants, and both congenital and acquired spinal pathologies in the pediatric population.
Indications
The most common indication for spinal ultrasound in neonates is an atypical sacral dimple. According to the American Institute of Ultrasound in Medicine (AIUM) 2021 guidelines, spinal ultrasound is indicated for the following conditions2,3:
Lumbosacral stigmata: Midline or paramedian masses, skin discolourations, skin tags, hair tufts, haemangiomas, and atypical sacral dimples (above gluteal cleft, >2.5 cm from the anus, >5 mm in diameter, and cutaneous stigmata),4,6 are commonly associated with spinal dysraphism or tethered cord.
Caudal regression syndrome: Congenital anomalies such as sacral agenesis and anorectal malformations, including conditions like Currarino triad, VACTERL association, cloaca, and OEIS complex, which may involve spinal abnormalities.
Suspected spinal cord abnormalities: Evaluation of conditions such as cord tethering, diastematomyelia, hydromyelia, and syringomyelia.
Acquired spinal abnormalities: Patients with trauma, infection, or post-procedural complications, ultrasound can identify haematomas, CSF leaks, or misplaced devices, especially after lumbar punctures or spinal surgeries.
Congenital spine tumours: Ultrasound aids in detecting and evaluating congenital tumours such as sacrococcygeal teratomas and other neurogenic tumours involving the spine, including neuroblastoma.
Spinal haematoma: Detection of hematoma in neonates with intracranial haemorrhage.
Lumbar puncture guidance: Assisting with the safe and accurate needle placement.
Postoperative follow-up: Assessing for cord retethering or infection following spinal surgery.
However, preoperative ultrasound of an open spinal defect or a closed spinal defect with thin or compromised overlying skin is relatively contraindicated, while the area above and below it with intact skin can be evaluated.1,3
Technique
Ultrasound probe: Spinal sonography should be performed using a high-resolution, high-frequency (7-15 MHz) linear transducer and curved transducer (8-10 MHz).1,3,4,6,8
Patient positioning: Newborns are typically examined in the prone position, and a lateral decubitus position is also acceptable. An upright or reverse Trendelenburg position is desirable due to dependent distal thecal sac distension. A bolster under the lower abdomen or flexed knee can help improve positioning. For older children, paramedian angulation offers better visualization of the spinal canal due to ossifying spinous processes in midline.3,5,9,10
Imaging planes: Routine imaging includes sagittal and axial scans from the craniocervical junction to the tip of the coccyx, including conus medullaris, cauda equina, and coccyx. Sagittal imaging should be performed in the median and paramedian planes to ensure a comprehensive evaluation. Panoramic ultrasound helps in vertebral numbering and assessing the alignment, and it should include T12 to the coccyx in a single image.1,3,11,12 The focal zone is usually set between 1.5 and 2.5 cm, and the visualization depth extends from 3 to 5 cm.
Pulsatility: Cine imaging can assess the dynamic movement of the distal cord and conus, filum, and nerve roots, particularly in relation to CSF pulsations. M-mode ultrasound can be helpful. A normal, untethered cord may not exhibit significant pulsatility until few weeks of age, partially attributed to decreased CSF from physiological dehydration in neonates.1,3,9,11
Doppler ultrasound is not typically used for evaluating spine vascularity but can help assess haemangiomas and associated mass lesions.1,4,7
Gel pads: A standoff with gel pads is helpful for the visualization of superficial structures and assessment of sacral dimples and dorsal dermal sinus tract.3 -5
Tip: Due to the frequent association between spinal and renal abnormalities, both kidneys should be scanned in the longitudinal and transverse planes before concluding the examination. 5
Sonographic Anatomy
Cord and central echo complex: In the sagittal plane, the spinal cord appears as a hypoechoic tubular structure, with a central echo complex representing the central canal (Figure 1). The diameter of the spinal cord is variable, being widest at the cervical and lumbar levels due to the presence of the nerve roots for the cervical and the lumbar plexus and narrowest at the thoracic level. In the axial plane, the spinal cord appears as a round (thoracolumbar) or oval (cervical) hypoechoic structure, with the central echo complex approximately one-third to one-half of the distance between the anterior and posterior walls of the spinal canal. Paired dorsal and ventral nerve roots arise from the cord.4,6,11
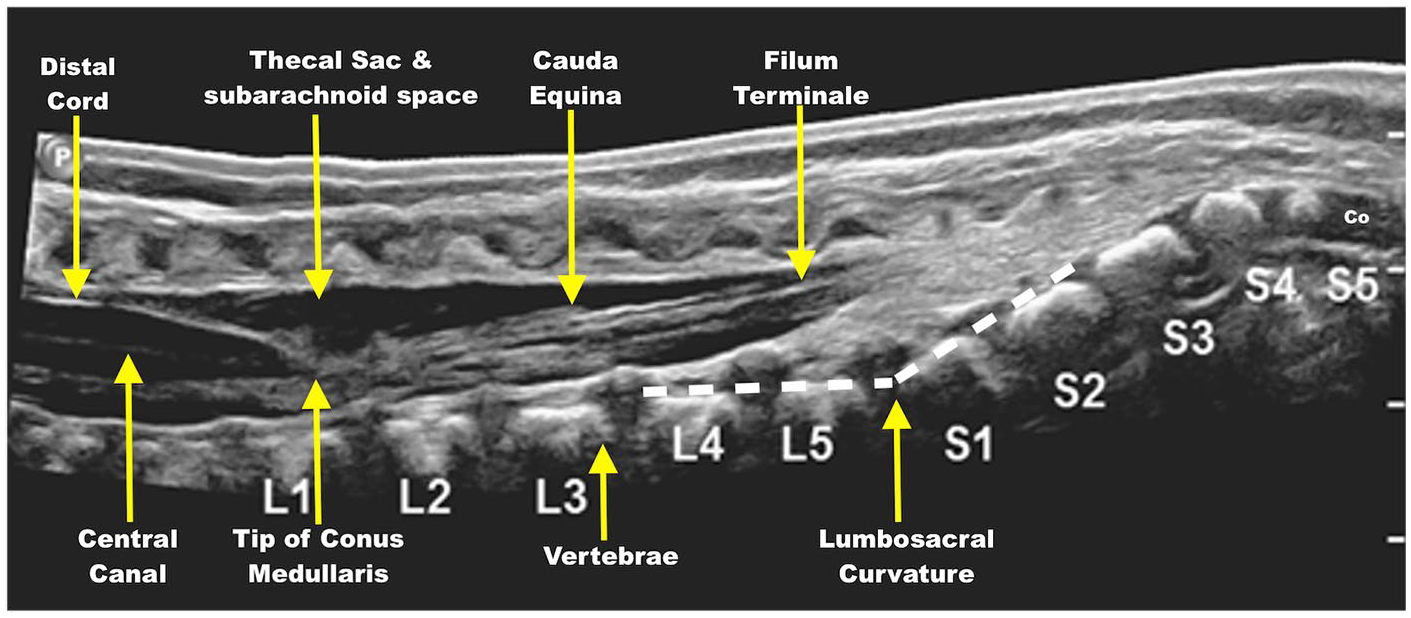
Sonographic anatomy of spine.
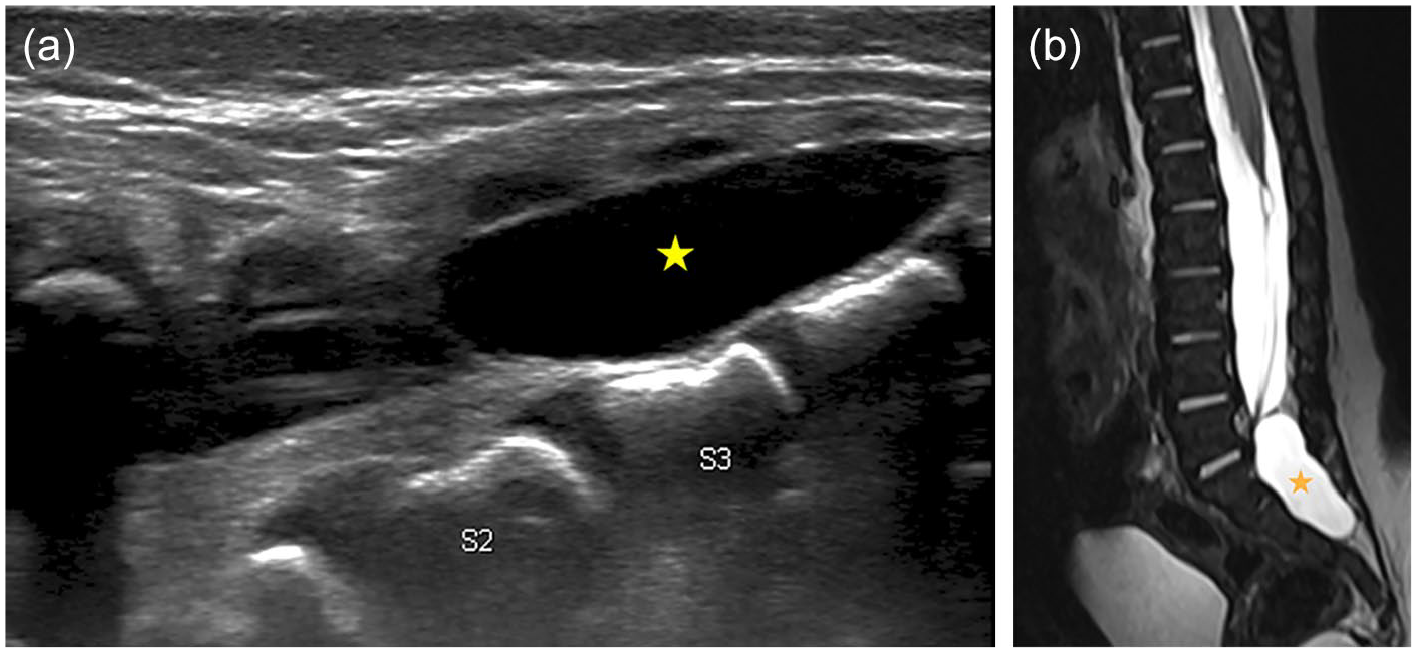
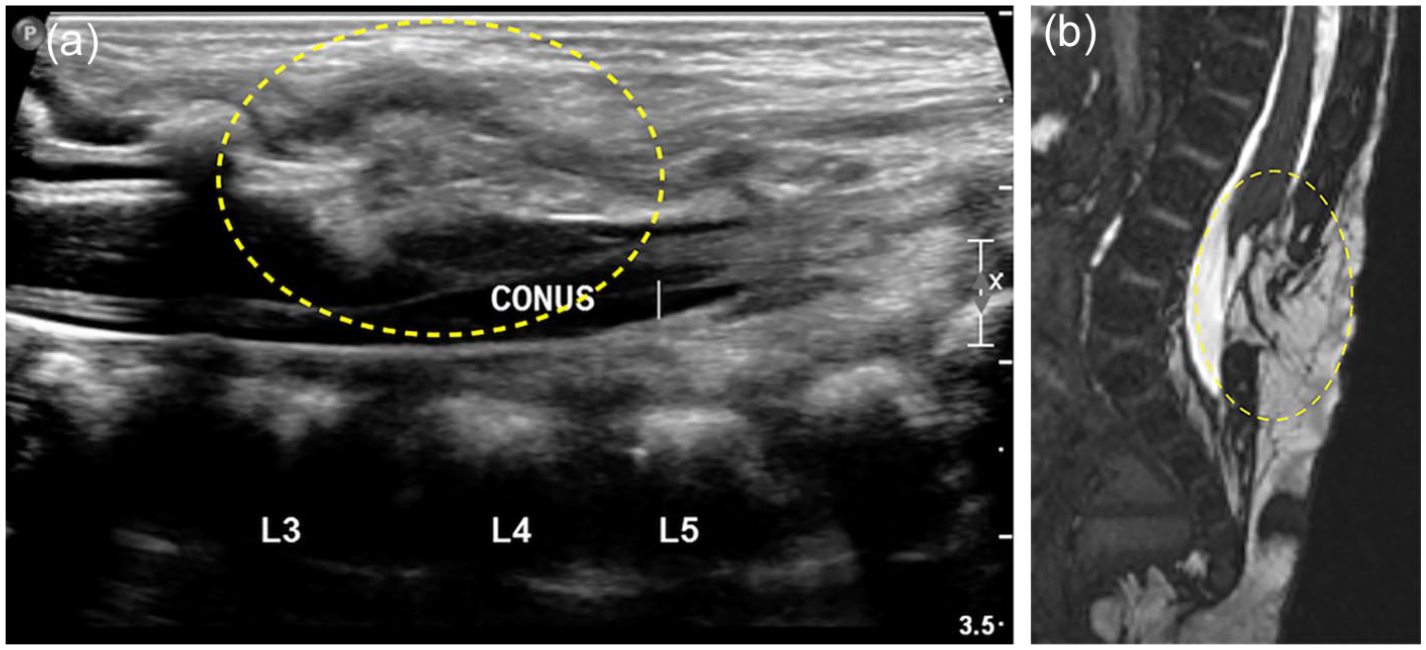
Conus Medullaris: The conus medullaris, at the caudal end of the cord continues into the filum terminale, surrounded by the cauda equina nerve roots (Figure 1). Localization of the conus medullaris is crucial for detecting low-lying conus or high termination. The conus medullaris usually terminates above the L2-L3 intervertebral disc in healthy newborns. The palpable landmarks for identification of the vertebral levels are the tip of the lowest rib corresponding to L2 and the iliac crest corresponding to L5. Vertebral labelling from the last rib at T12 may not be reliable in patients with 11 or 13 ribs. The lumbosacral junction (Figure 1) can be identified by distinct lumbosacral angulation, though this may be unreliable in the presence of transitional vertebrae. Alternatively, the coccyx, which is rounded and frequently unossified (Figure 1) compared to the sacral vertebrae, can be used as a reference point. The thecal sac typically ends at the S2 level and also aids in vertebral level determination. Conus medullaris at the mid-L3 level can be seen in normal preterm infants, but a follow-up is recommended after the corrected age (40 weeks to 6 months).3,4,6,13
Filum terminale: The filum terminale is a fibrous band extending from the conus medullaris to the caudal end of the spinal canal and is surrounded by cauda equina nerves (Figure 1). It typically measures less than 2 mm at the L5-S1 level, appearing relatively hypoechoic centrally with a hyperechoic periphery due to the CSF-filum interface on ultrasound.1,3,5,6,12
Cauda equina: The cauda equina nerves are suspended in the distal thecal sac (Figure 1). Cine imaging helps evaluate their dynamic movement and real-time imaging, particularly in relation to CSF pulsations.1,3,6,9
Thecal sac and subarachnoid space: Thecal sac extends up to S2 vertebrae (Figure 1), containing cord, filum, and cauda equina surrounded by anechoic CSF-filled subarachnoid space, interrupted by hyperechoic linear nerve roots and dentate ligaments.3,6,12
Vertebrae and paravertebral muscles: The vertebral bodies and discs are seen ventral to the thecal sac with posterior acoustic shadowing (Figure 1). Paravertebral muscles are best visualized below the L2 vertebral level.6,9
Variant Anatomy
Persistent ventriculus terminalis (Fifth ventricle): The central canal remnant at the conus medullaris appears as a focal widening of the central echo complex (Figure 2). The condition is asymptomatic and typically resolves within a few weeks after birth. On ultrasound, it appears as a well-defined round-to-oval anechoic area in the distal conus, close to the junction of conus and filum and usually measures less than 2 cm.4,6,9,14,15
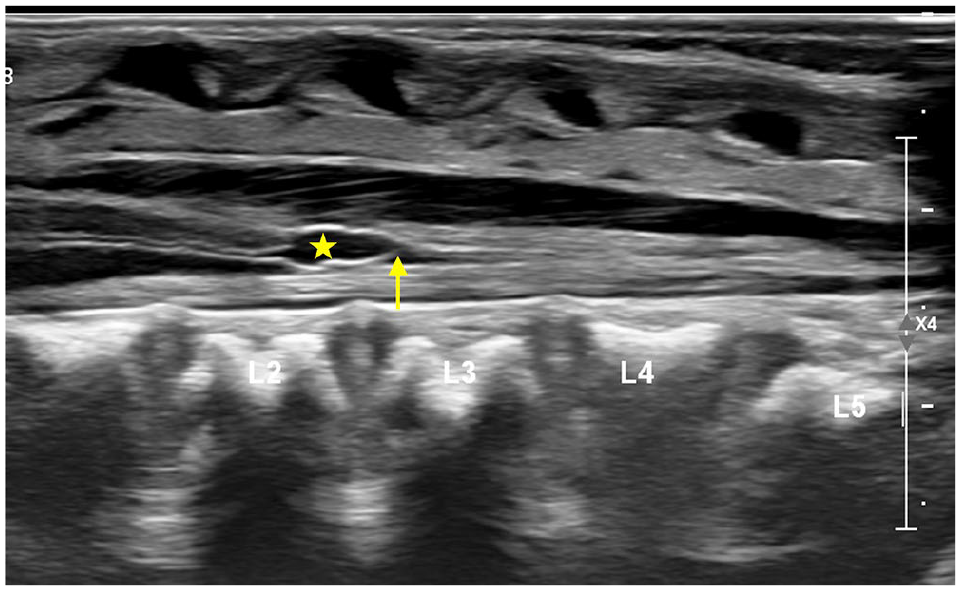
Sagittal US in 1-week-old shows a borderline conus at L2-L3 (arrow) with mild cystic dilatation of distal central canal at conus medullaris (star) in keeping with ventriculus terminalis.
Transient central canal dilatation: Mild, transient dilation of the central canal can be seen in the first few weeks of life (Figure 3) and may require a short follow-up to confirm resolution.2,6,16
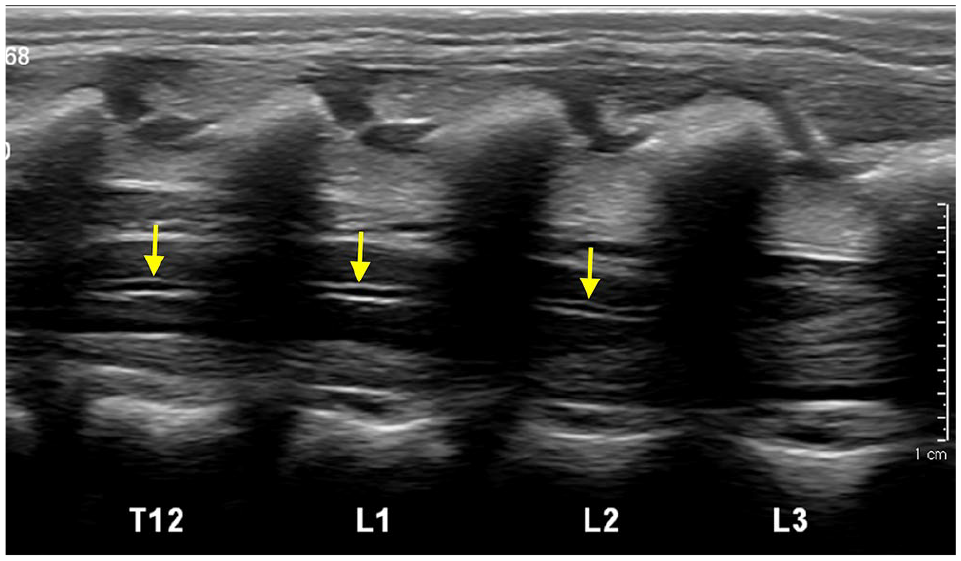
Sagittal US image of distal spine in a 2-days-old shows a borderline low conus with mild prominence of thoraco-lumbar central canal (arrows).
Filum terminale cyst: Small, midline, anechoic, and thin-walled cysts located in the filum terminale, just below the conus medullaris (Figure 4). Observed in less than 20% of infant spine ultrasounds. Their origin is uncertain but may represent a remnant of the ependymal lining or pseudocyst due to arachnoid reflection. They are typically not detectable on MRI or autopsy, and are unlikely to be of clinical significance.2,5 -7,17
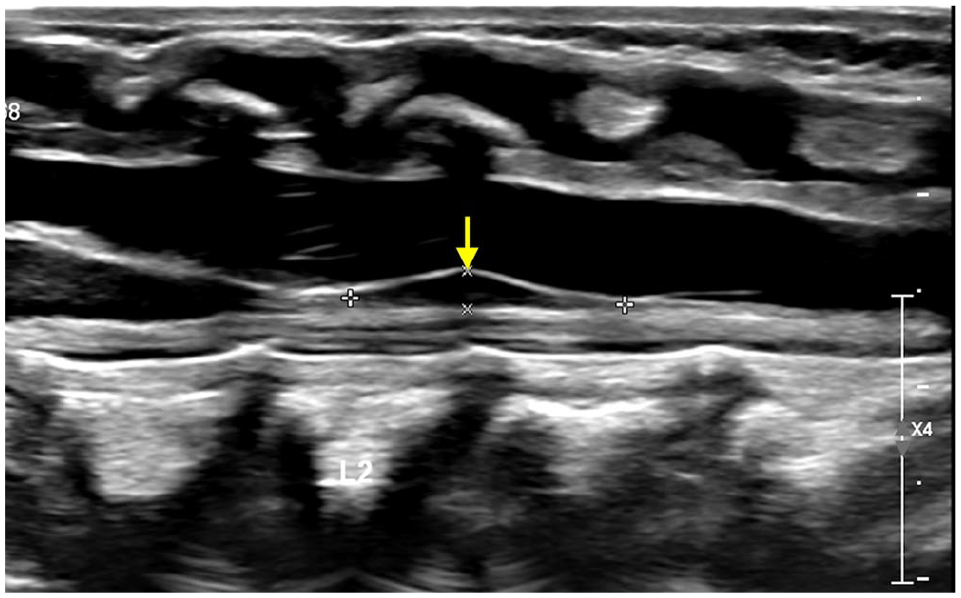
Sagittal US image of distal spine in a 1-week-old shows small filar cyst (arrow) in the proximal filum terminale just distal to conus medullaris.
Pseudomass of the cauda equina: This is a positional phenomenon where nerve roots appear clumped together during sonographic examination (Figure 5), commonly in decubitus position that resolves with a change in posture.3,7,18
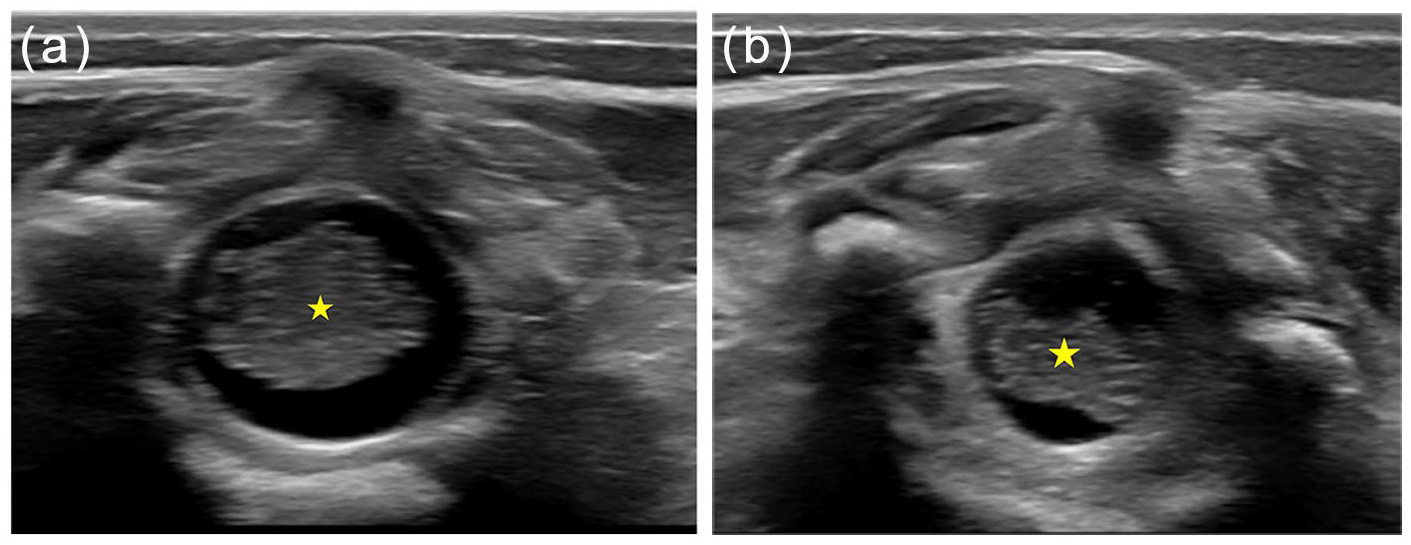
Axial US in 3-days-old shows pseudo-mass like appearance (star in a) due to physiological dehydration. Improved position of the nerve roots with change in positioning (star in b).
Dysmorphic coccyx: An abnormal curvature of the coccyx, often with a dorsal bend of the tip or bifid appearance, is common in neonates (Figure 6). It can be palpable and may mimic a mass, but it typically resolves with growth and does not require intervention.2,6,7,18
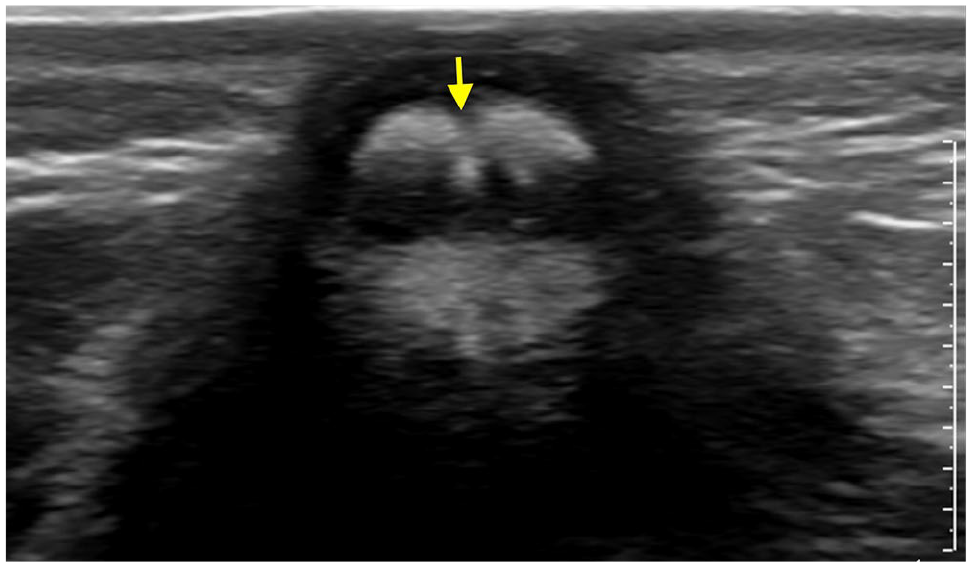
Axial US image in a 2-weeks-old for sacral dimple shows bifid coccyx (arrows).
Pseudosinus tract: An echogenic blind ending fibrous tract extending from a cutaneous dimple to the coccyx (Figure 7), no associated mass or CSF leak, and requires no further evaluation.2,5,7,18,19
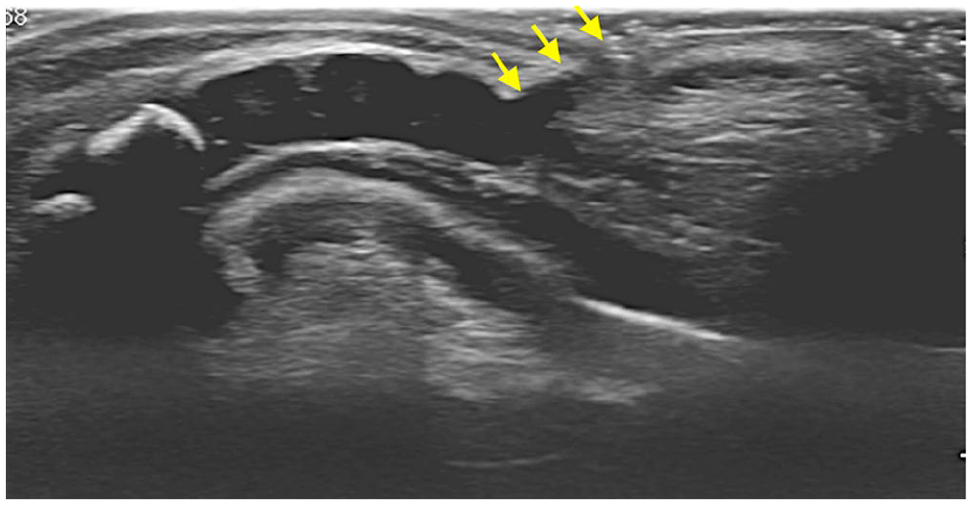
Sagittal US image of lower spine in a 5-days-old shows a subcutaneous hypoechoic tract ending blindly superficial to coccyx (arrows), in keeping with pseudo-sinus tract.
Mild epidural fat thickening: Mild thickening of the epidural fat without significant thecal sac indentation is usually incidental and considered within the spectrum of normal neonatal development.4,6
Prominent Filum Terminale: An echogenic filum terminale, measuring 1 to 2 mm, is a normal variant encountered in some neonates (Figure 8). It is present in approximately 6% of cases at autopsy (fatty filum terminale) and has no clinical significance when isolated, with a normal conus position and a filar thickness under 2 mm.2,3,6,7,9,20
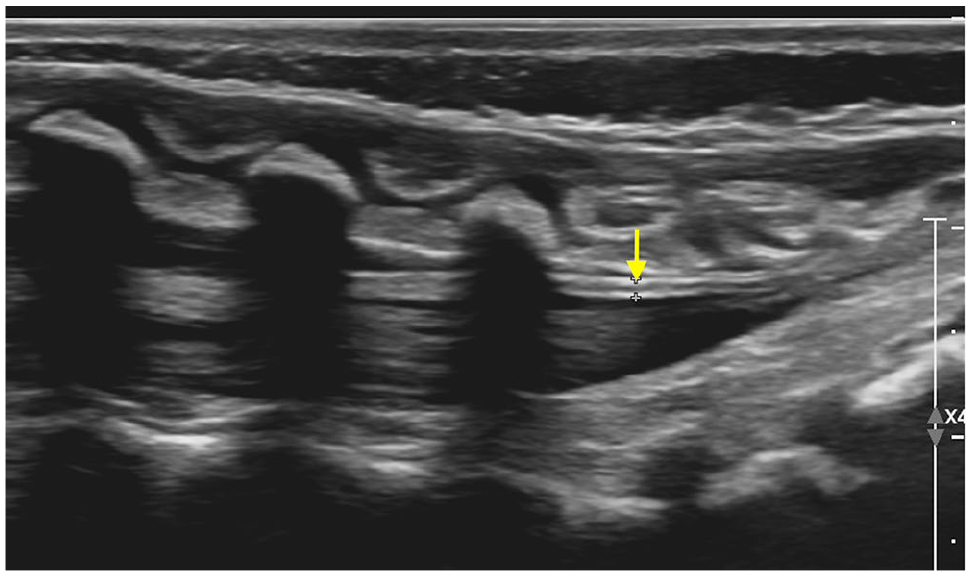
Sagittal US image of spine in a 10-days-old shows slightly increased echogenicity of the filum (arrow) measuring <2 mm and with a normal position of conus, suggesting prominent filum terminale.
Spinal Pathologies
Congenital
1. Haemangiomas: Lumbosacral haemangiomas are usually in the midline, large (>2.5 cm) and closely associated with spinal abnormalities. Ultrasound typically reveals a hyperechoic and vascular soft tissue lesion (Figure 9). Congenital haemangiomas are present at birth with their maximum size, and gradually regress over time. In contrast, infantile haemangiomas typically become noticeable within a few days to weeks after birth, initially grow for a period, and then undergo involution. Radiographic evaluation of LUMBAR (Lower body haemangioma, Urogenital abnormalities, Myelopathy, Bone deformities, Anorectal malformations, and Renal anomalies) syndrome should always include assessment of the spine for possible dysraphism.1,2,6,21,22
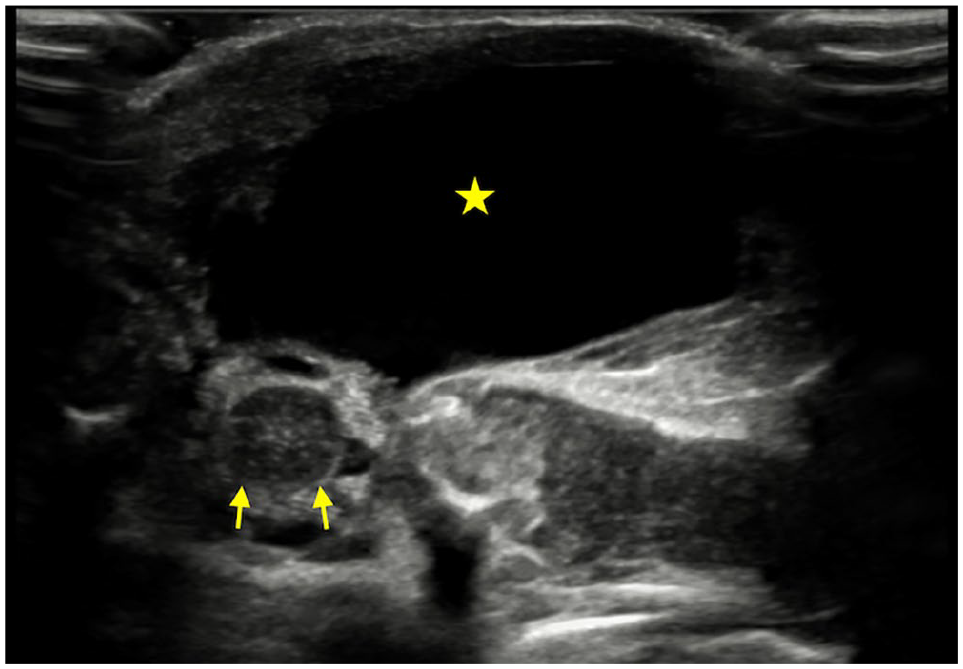
2. Arachnoid Cyst: These are CSF-filled, round to fusiform cysts within the spinal canal, typically located dorsal to the cord. When located ventrally, it is challenging to differentiate them from neuroenteric cyst. On ultrasound, they appear as anechoic, well-circumscribed, extramedullary lesions (Figure 10) with or without spinal dysraphism.4,9,10,12,18
3. Syringohydromelia: A syrinx appears as an anechoic, intramedullary, central cystic cavity of the cord. It can be isolated to the central canal (hydromyelia) or involve the parenchyma (syringomyelia). Syrinx is commonly associated with Chiari malformation, tethered cord, trauma, or neoplasms. Ultrasound features include an elongated, anechoic region within the cord (Figure 11), with cord parenchymal thinning in severe cases.4,5,9
4. Closed Spinal Dysraphism (CSD)
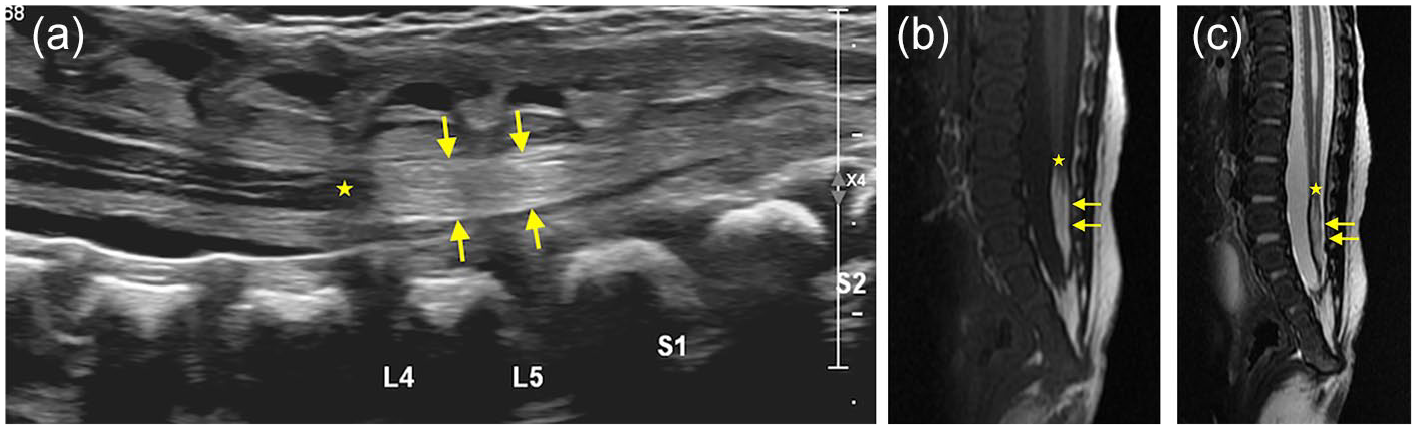
a. CSD Without Subcutaneous Mass Fibrolipoma of the Filum Terminale: Fibro lipoma of the filum terminale accounts for 12% of spinal lipomas. Ultrasound typically shows a thickened, echogenic filum terminale (>2 mm) often associated with a low-lying conus (Figure 12). This condition is frequently linked to other malformations, including syringomyelia, type II caudal agenesis syndrome, and tethered cord syndrome, though it generally lacks cutaneous stigmata.6,9,10,18,23,24 Intradural Lipoma: Intradural lipomas, comprising 4% to 24% of spinal lipomas, are located within the dural sac and can increase in size with age. Typically centred in the subpial region and closely associated with the neural placode, these lipomas are rarely intramedullary. Larger lipomas may displace the spinal cord, causing it to appear off-midline. On ultrasound, they present as well-defined, echogenic masses causing cord displacement with intact or dysraphic posterior elements.6,9,11,24 Dermal Sinus: A midline epithelial-lined fistula that connects the skin to the spinal canal, most commonly in the lumbosacral region. It occurs in approximately 1 in 2500 live births, with no sex predilection. On ultrasound, it appears as a thin, mildly hypoechoic track extending through the subcutaneous tissue to the spinal canal, where it may become a linear echogenic structure in the CSF-filled subarachnoid space (Figure 13) and can lead to complications such as meningitis. The associated cutaneous anomalies comprise capillary angiomas, hyperpigmented patches, hairy nevi, and inclusion cysts.4
-6,9,12,18,24 Limited dorsal myeloschisis: The combination of the dorsal dermal sinus with a distinct intraspinal tract and frequent tenting of the cord-tract junction just above the conus. Not frequently associated with inclusion cysts and meningitis.4,24,25 Diastematomyelia: Diastematomyelia is char-acterized by a complete or partial sagittal splitting of the spinal cord into 2 asymmetrical hemi-cords, each with its own pia mater and central canal. They are most common in the thoracolumbar spine, with a higher incidence in females. These hemi-cords may fuse distally or remain separate. Ultrasound typically reveals 2 hemi-cords with ipsilateral nerve roots in the axial plane. They can have 2 dural sacs with intervening osseocartilaginous spur in type I or a single dural sac in type II. Syringomyelia is commonly associated, seen in up to 50% of cases. The condition requires thorough scanning of the entire spinal canal to identify fusion and evaluate cutaneous stigmata.4
-6,9,12,18,24,26,27 Segmental spinal dysgenesis: A localized malformation of the spine and spinal cord, typically seen in the thoracolumbar region, characterized by a sharp kyphotic deformity. Imaging shows absent or underdeveloped vertebrae at the affected segment, and relatively normal vertebrae above and below. The spinal cord at the site of deformity is either thinned or absent, with a low-lying conus and potential cord tethering. Associated findings may include cutaneous markers and other visceral abnormalities.18,24,28,29
b. CSD With Subcutaneous Mass ° Lipomas with Dural Defect (LDD): LDD including lipomyelomeningocele, lipomyelocele, and lipomyeloschisis, can present as skin-covered lumbosacral masses. Ultrasound shows a skin-covered lipoma attached to the neural placode, with the lipomatous fat often appearing iso- to hyperechoic compared to subcutaneous fat and dysraphic posterior elements. The subtypes are determined by the location of lipoma-placode interface, whether inside (lipomyeloschisis), at (lipomyelocele; Figure 14), or outside (lipomyelomeningocele; Figure 15) the defect. LDD accounts for over 80% of spinal lipomas and up to 20% of closed spinal dysraphism. These lesions may be associated with cutaneous haemangiomas or hairy nevi.11,18,20,23,24,30 Myelocystocele: Myelocystocele is a herniation of the dilated central canal through a posterior spinal defect. On ultrasound, a “nonterminal myelocystocele” shows herniation of the dilated central canal only, while the “terminal myelocystocele” also has protruding meninges covering the herniated dilated central canal. Terminal myelocystoceles are often associated with complex malformations, such as cloacal exstrophy or as a part of OEIS complex (omphalocele, cloacal exstrophy, imperforate anus, and spinal anomalies).9,11,12,18,20,24,31,32 Meningocele: It is a CSF-filled outpouching of the meninges through a vertebral defect that accounts for 2.4% of all CSD. The posterior meningocele, typically located in the lumbar region, is more common than the anterior meningocele (which often found in the sacral region and is associated with Currarino triad), or lateral meningocele (usually in the thoracic region and associated with NF1, Marfan, or Ehler-Danlos syndromes). Meningocele appears as an anechoic cystic structure devoid of neural tissue on ultrasound (Figure 16). Anterior sacral meningocele may be associated with the Currarino triad, which includes a presacral mass (dermoid or meningocele), a sacrococcygeal osseous defect (crescent-shaped sacrum), and anorectal anomalies such as anal stenosis or atresia.9,11,12,18,24,30
c. Caudal Regression Syndrome (CRS): CRS involves agenesis or dysgenesis of the lower spine (Figure 17) and is associated with genitourinary, anorectal, and limb anomalies, ranging from incomplete sacrococcygeal agenesis to sirenomelia (complete lumbosacral agenesis and fused lower extremities). Risk factors include maternal diabetes, vascular anomalies, drugs (eg, minoxidil, trimethoprim-sulfamethoxazole, tocilizumab), and genetic mutations such as HLXB9. Known associations are Currarino triad, VACTERL (Figure 18), cloaca, and OEIS complex. Ultrasound findings can identify CRS types based on the appearance and position of the conus: Group 1 shows a blunt, deformed conus above L1, while Group 2 features an elongated, tethered conus below L1.9,11,12,18,23,24,33 -35
5. Open Spinal Dysraphism Myelomeningocele/Myelocele: Myelomeningocele and myelocele account for over 98% of open spinal dysraphisms, with a slight female preponderance. These defects are most commonly located in the lumbosacral or thoracolumbar regions, frequently associated with Chiari II malformation (99%), syrinx (40%-80%), and diastematomyelia (20%-40%). Fetal ultrasound can demonstrate the neural placode in direct contact with the skin (myelocele) or protruding outside (myelomeningocele). Preoperative ultrasound is rarely performed in the postnatal period due to their intrauterine diagnosis and repair. Scanning should be avoided over exposed surfaces and is primarily used to assess associated abnormalities by scanning the area abobe and below the defect. Postoperative ultrasound is crucial for detecting complications like syrinx and cord retethering.5,9
-11,18,24,30 Hemi myelomeningocele/Hemi myelocele: Extremely rare and characterized by myelomeningocele or myelocele affecting one or both hemicords in diastematomyelia.6,24,36,37
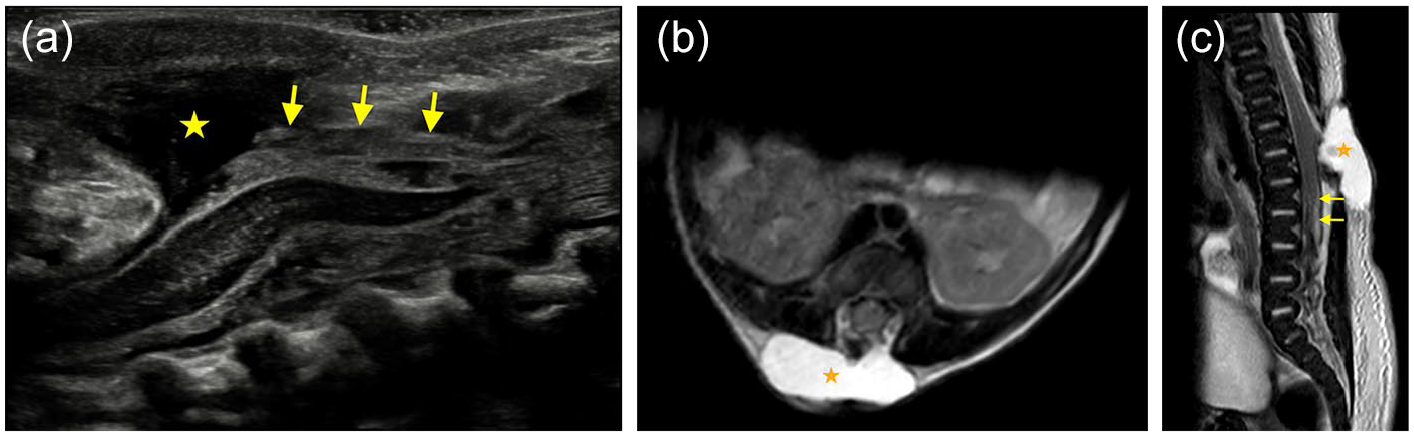
6. Congenital Spinal Tumours Sacrococcygeal Teratomas: Sacrococcygeal teratomas are the most common congenital tumours in neonates, with a female predominance, often presenting in the first days of life. Ultrasound findings are variable, ranging from solid to mixed cystic masses containing fat and calcifications (Figure 19). These tumours are classified into 4 types based on their intrapelvic extension: Type I (predominantly extrapelvic), Type II (significant intrapelvic component), Type III (predominantly intrapelvic), and Type IV (entirely intrapelvic or intra-abdominal).10,12,38 Neuroblastoma or other paraspinal tumours: Though rare in neonates, these tumours can invade the spinal canal. It is often challenging to assess the entire extent of the tumour on ultrasound.
18
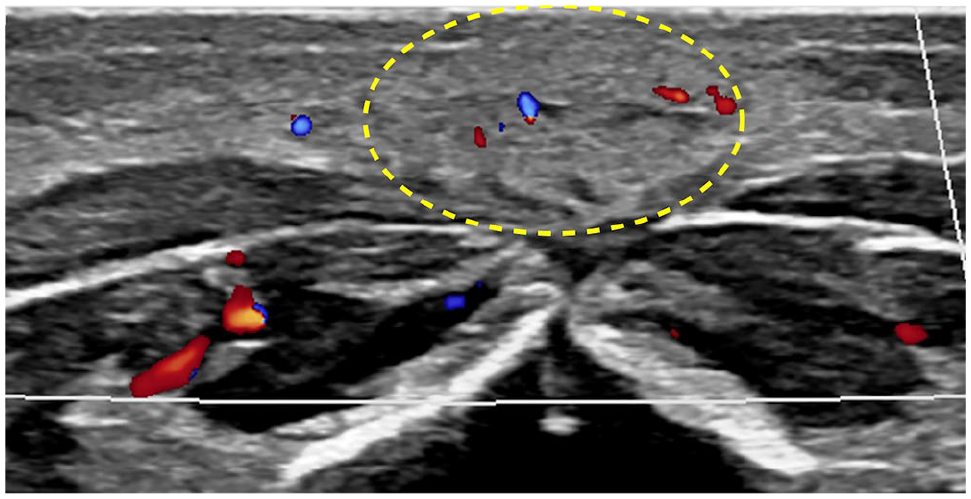
One-month-old with focal skin discolouration of the lower back, decreasing in size since birth. Axial US image shows focal echogenic lesion in the subcutaneous plane with mild internal vascularity (oval), in keeping with involuting congenital haemangioma.
Sagittal spine images of US (a) and T2W MRI (b) in a 2-week-old boy show an anechoic cystic structure at the level of S2-S4 (stars), indenting the thecal sac and displacing the filum anteriorly. Findings are most in keeping with an extradural arachnoid cyst.
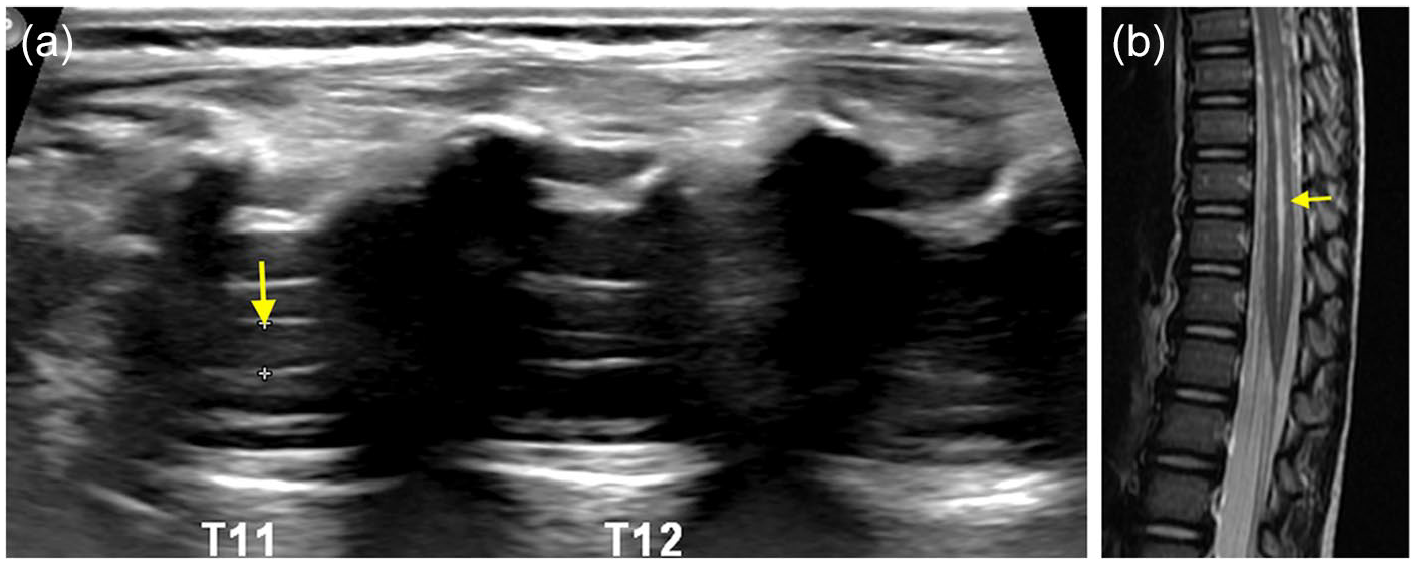
Sagittal ultrasound (a) and T2W MR (b) images of spine in a 3-months-old show dilated central canal (>2 mm) in the lower thoracic cord extending up to the conus (arrows) in keeping syringohydromelia.
Sagittal ultrasound (a), T1W (b), T2W (c) images in a 3-days-old shows low-lying conus (stars) at L4 and a thick, echogenic filum terminale (arrows), most consistent with low lying cord and fibro-lipoma of filum terminale.
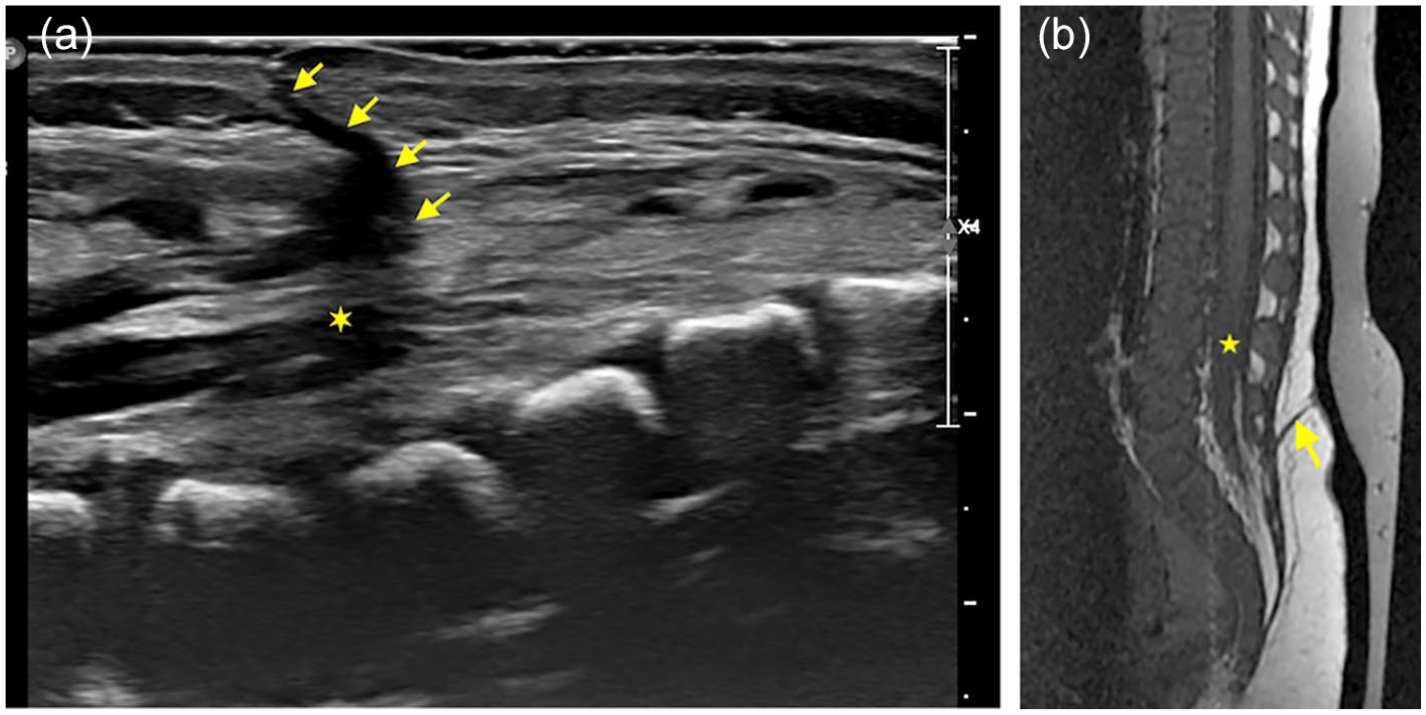
Sagittal ultrasound (a) and T1W MR (b) images in a 3-weeks-old show a hypoechoic dermal sinus tract (arrows) extending from skin to the dural sac with the low-lying conus (stars), in keeping with dorsal dermal sinus.
Sagittal ultrasound (a) and T2W MR (b) images of spine in a 20-days-old demonstrate low-lying fatty neural placode (ovals) that shows continuity with the dorsal subcutaneous fat through posterior spinal defect and no meningeal herniation, in keeping with lipomyelocele.
Two-weeks-old presented with subcutaneous mass. Sagittal US (a), axial T2W (b), sagittal T2W (c) images of distal spine reveal meningeal herniation (stars) and low-lying fatty neural placode (arrows) that shows continuity with the subcutaneous fat through the spinal defect in keeping with lipomyelomeningocele.
One-day-old for preoperative assessment of the lumbar meningocele. Axial ultrasound image of the spine shows a thin wall cystic structure in the posterior paraspinal soft tissue (star) that appears continuous with posterior spinal canal through defective T12-L2 posterior elements. Spinal cord (arrows) is seen deep to the sac within spinal canal.
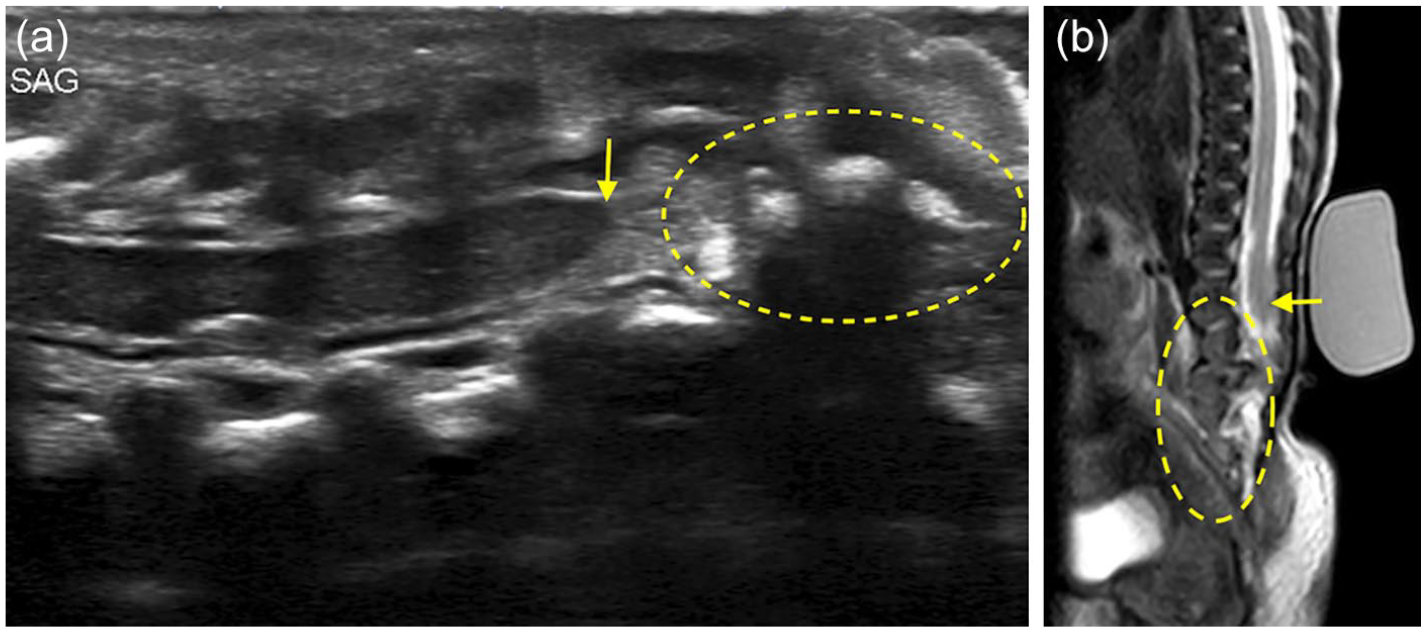
Sagittal US (a) and T2W MR (b) images in a 7-days-old demonstrate truncated appearance of the conus at L1 (arrows) and narrow and dysplastic appearance of distal spinal canal (ovals) due to dysgenesis of lower lumbar and sacral elements. Findings are in keeping with caudal regression syndrome.
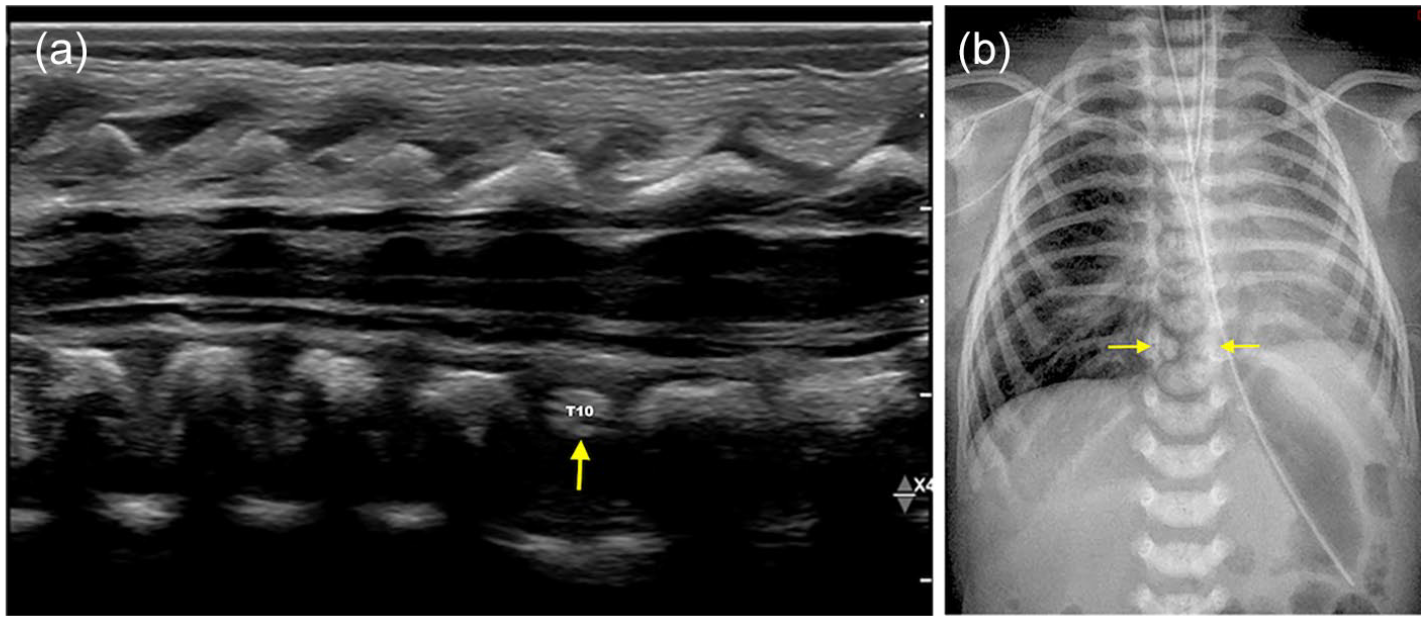
Sagittal US image of thoracic spine (a) and Xray chest (b) in a 3-days-old with suspected VACTERL syndrome shows small and dysplastic appearance of the T10 (arrows) vertebral body.
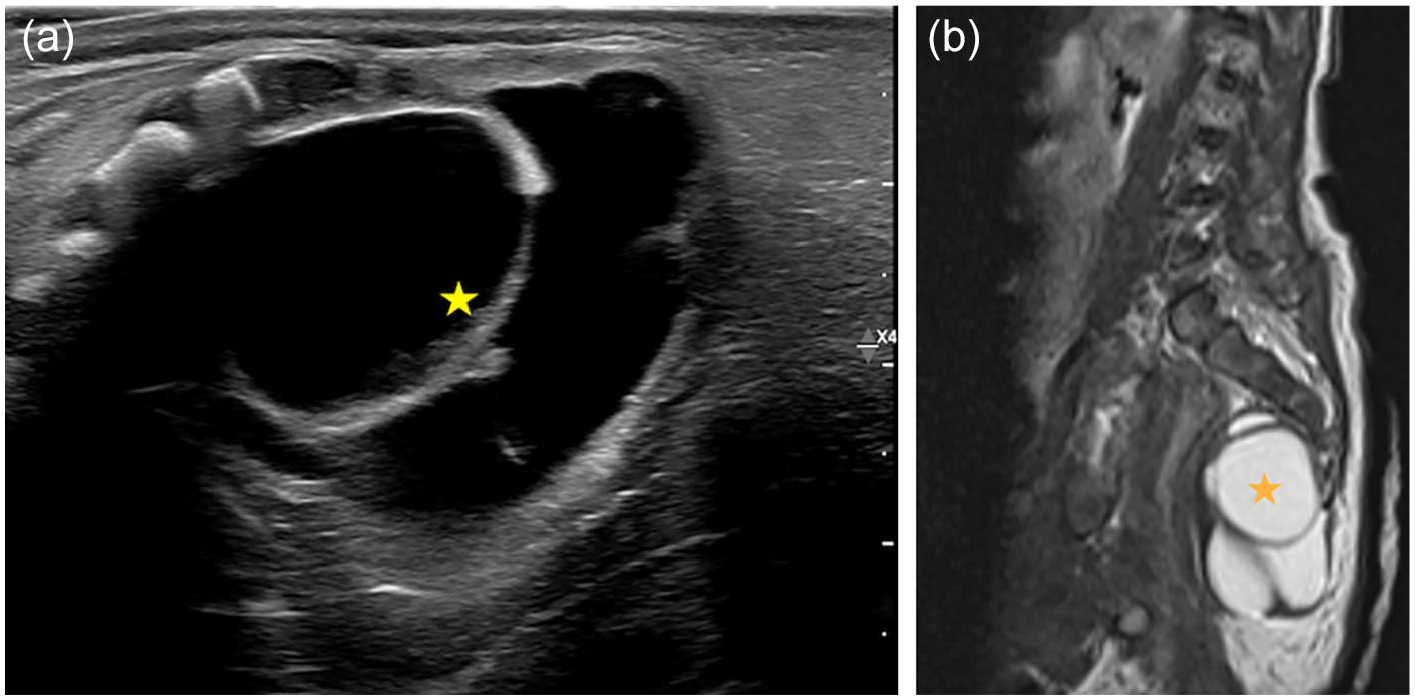
Two-days-old female admitted for erythematous lesion of lower back and possible anterior meningocele. Sagittal US (a) and T2W MR (b) images show a large multilocular cystic lesion (stars) in the presacral space with a small superficial component. Findings are in keeping with sacrococcygeal teratoma (Type III).
Acquired Pathologies
Spinal Cord Injury/Intraspinal Haematoma: Spinal cord injury or intraspinal haematoma post-delivery or intervention shows increased cord echogenicity on ultrasound due to oedema, haemorrhage, or venous congestion, with possible cord displacement by epidural or subdural haematoma (Figure 20). Small epidural haematomas post-lumbar puncture are common and should not raise major concerns. Follow-up US/MRI to monitor haematoma resolution and assess for myelomalacia could be consider.3,9,11,18
Meningitis/Arachnoiditis: Inflammatory processes affecting the pia-arachnoid and subarachnoid space, often appear as increased echogenicity and septations in the subarachnoid space on ultrasound.3,18
Ultrasound Guided Interventions: Spinal procedures such as caudal anaesthesia and lumbar puncture can be accurately performed using ultrasound guidance, which assists in proper needle placement and minimizes injury to delicate structures.3,9,11,12,18 In neonates with multiple failed attempts, ultrasound is used to assess for complications and level of patent thecal sac.
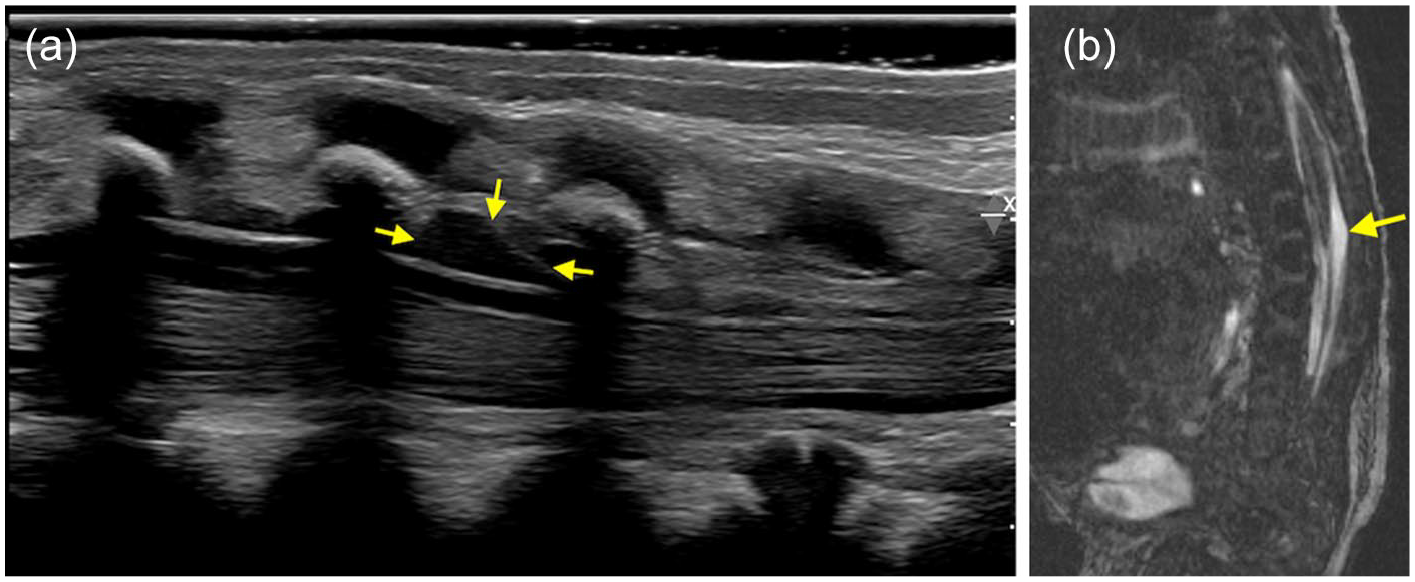
Sagittal ultrasound (a) and FIESTA (b) images of distal spine in a 1-week-old following lumbar puncture, show a small extra-axial collection (arrows) at the level of L3-L4 with no significant surrounding mass effect, suggestion of a collection post lumbar puncture.
Ultrasound Versus MRI
Ultrasound is a first-line imaging technique for neonatal spinal evaluation that enables real-time dynamic assessment of cord, cauda equina, and filar movement. Its ability to be portable and provide bedside screening make it particularly advantageous for screening neonatal spine, including the detection of tethered cord and other congenital anomalies. However, its limited penetration and reliance on operator expertise restrict its broader clinical applicability, particularly in less experienced settings and peripheral centres.
In contrast, MRI offers superior anatomical detail and excellent soft tissue contrast, making it indispensable for evaluating complex spinal pathologies across all age groups. It provides comprehensive imaging of the cord, nerves, and bony structures, allowing for the detection of subtle and complex multisystem abnormalities that may be challenging on ultrasound. Despite its advantages, MRI is associated with higher cost, longer acquisition time, and limited availability with need for sedation in infants and young children. The Table 1 highlights a detailed comparison between ultrasound and MRI in the evaluation of the pediatric spine.
Comparison of Ultrasound and MRI in the Assessment of Pediatric Spine.

Conclusion
Ultrasound is a valuable tool for diagnosing congenital and acquired pathologies of the neonatal spine, offering high sensitivity and specificity, especially for visualizing soft tissue structures, fluid collections, and spinal cord anomalies. Early identification of spinal pathologies can guide management decisions and improve outcomes in affected infants. Syndromic associations, such as VACTERL, OEIS, LUMBAR syndrome, and the Currarino triad, should also be considered during evaluation, as they help identify underlying conditions and guide further management.
Footnotes
Acknowledgements
To the CAR-ASM 2024 team for inviting our work for submission in the CARJ.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
